Smoking-Mediated Upregulation of the Androgen Pathway Leads to Increased SARS-CoV-2 Susceptibility
Abstract
1. Introduction
2. Results
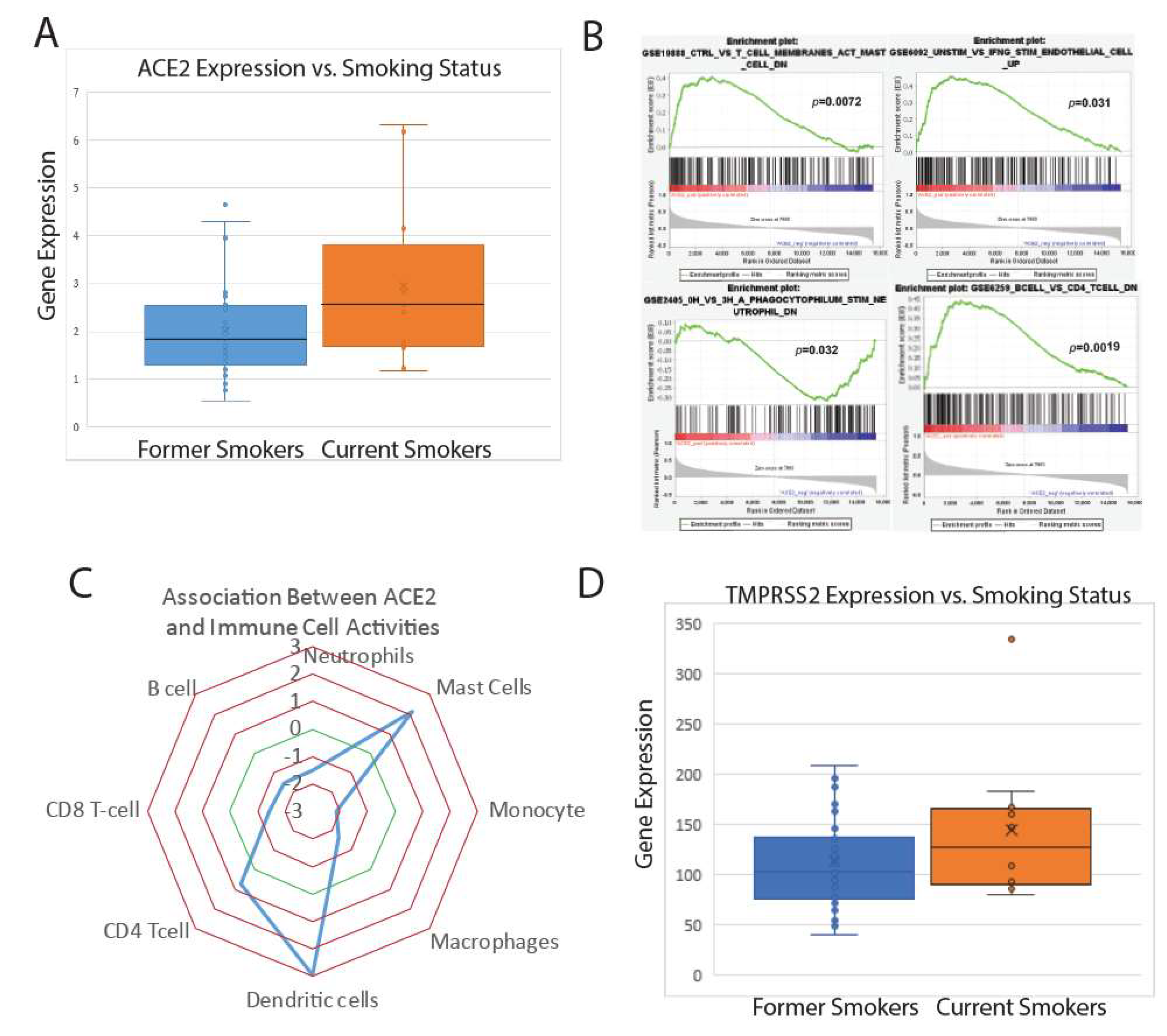
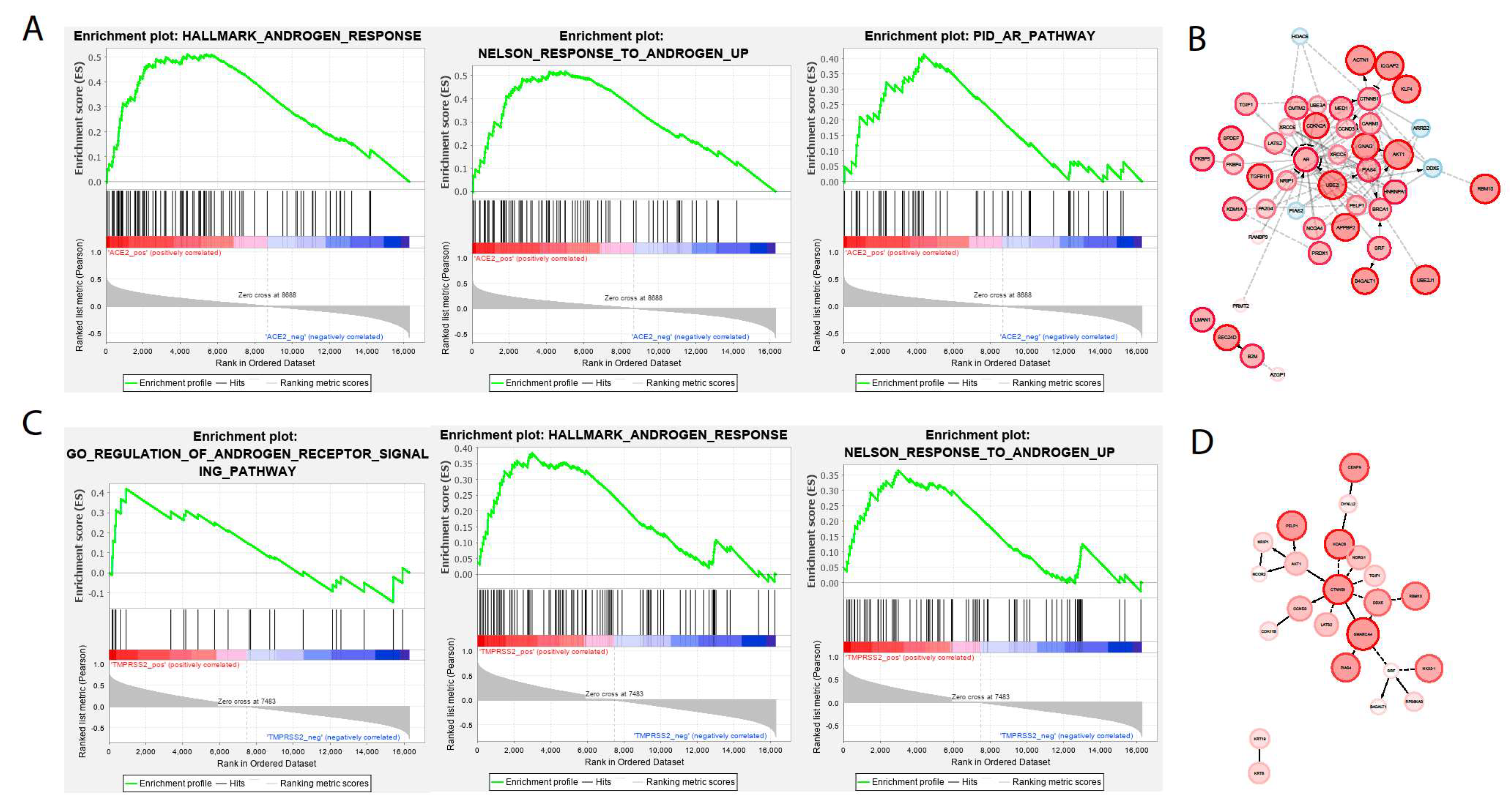
2.1. Analysis in Lung Epithelium
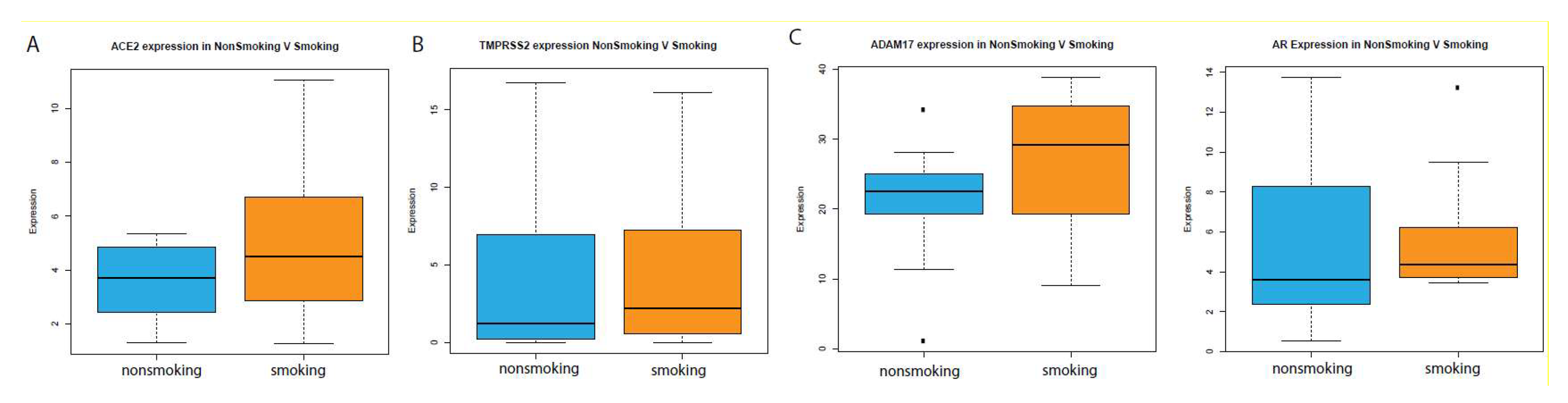
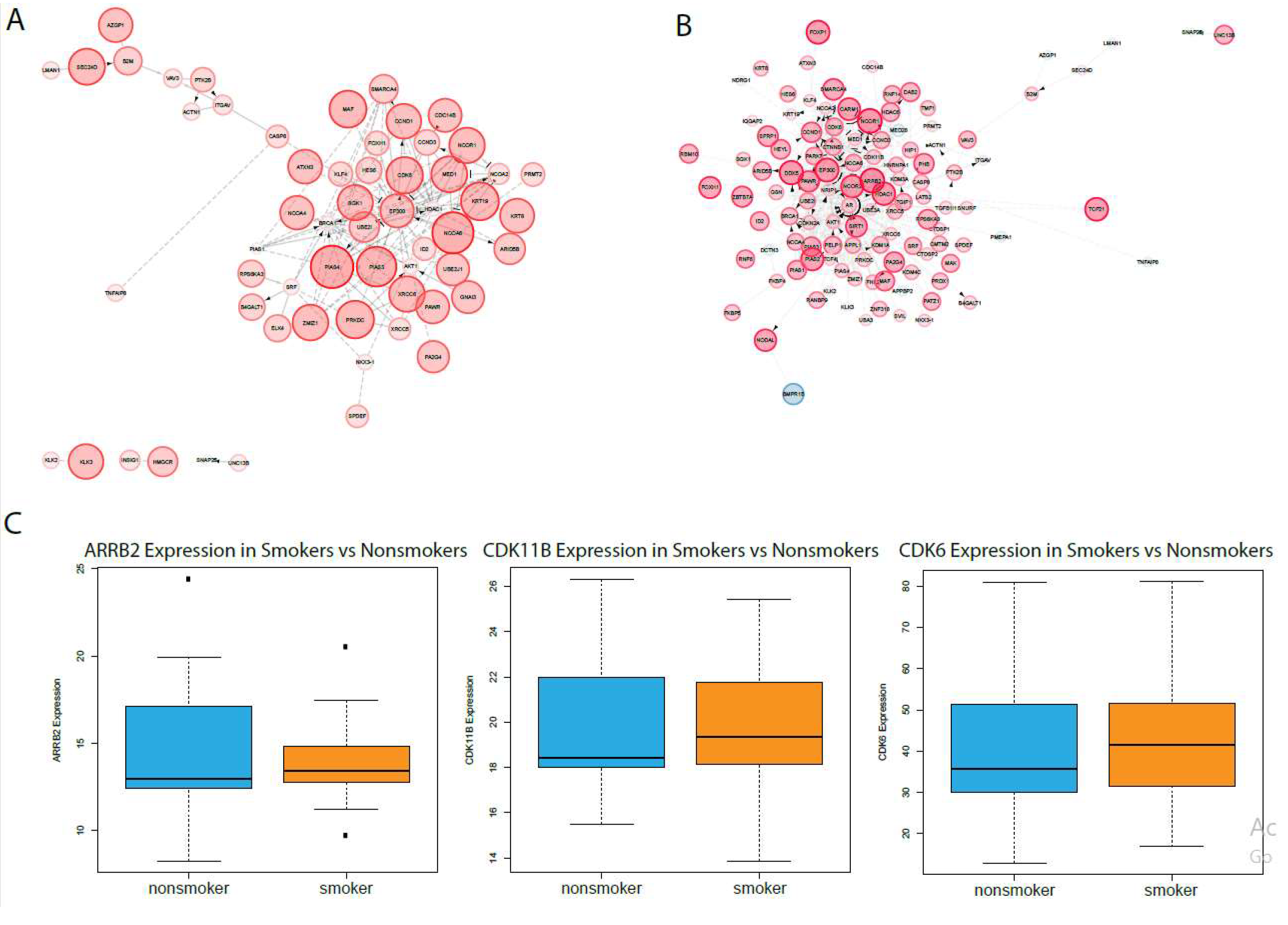
2.2. Analysis in Oral Epithelia
3. Discussion
4. Materials and Methods
4.1. RNA-Sequencing Data Acquisition
4.2. Differential Expression Analysis
4.3. Immune Pathway Association Using GSEA
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| TCGA | The Cancer Genome Atlas |
| GSEA | Gene Set Enrichment Analysis |
| MSigDB | Molecular Signature Database |
| SARS | Severe acute respiratory syndrome |
| MERS | Middle east respiratory syndrome |
| SARS CoV-19 | Severe acute respiratory syndrome coronavirus 2 |
| Mers-CoV | Middle east respiratory syndrome coronavirus |
| COVID-19 | Coronavirus disease 2019 |
| CPM | Counts per million |
| AR | Androgen receptor |
References
- Fauci, A.S.; Lane, H.C.; Redfield, R.R. Covid-19—Navigating the Uncharted. N. Engl. J. Med. 2020, 382, 1268–1269. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.-C.; Chen, C.-S.; Chan, Y.-J. The outbreak of COVID-19: An overview. J. Chin. Med. Assoc. 2020, 83, 217–220. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, M.; Kleine-Weber, H.; Schroeder, S.; Krüger, N.; Herrler, T.; Erichsen, S.; Schiergens, T.S.; Herrler, G.; Wu, N.-H.; Nitsche, A.; et al. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell 2020, 181, 271–280. [Google Scholar] [CrossRef]
- Glowacka, I.; Bertram, S.; Müller, M.A.; Allen, P.; Soilleux, E.; Pfefferle, S.; Steffen, I.; Tsegaye, T.S.; He, Y.; Gnirss, K.; et al. Evidence that TMPRSS2 activates the severe acute respiratory syndrome coronavirus spike protein for membrane fusion and reduces viral control by the humoral immune response. J. Virol. 2011, 85, 4122–4134. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.C.; Sheltzer, J.M. Cigarette smoke triggers the expansion of a subpopulation of respiratory epithelial cells that express the SARS-CoV-2 receptor ACE2. bioRxiv 2020. [Google Scholar] [CrossRef]
- Astuti, I. Ysrafil, Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2): An overview of viral structure and host response. Diabetes Metab. Syndr. Clin. Res. Rev. 2020, 14, 407–412. [Google Scholar] [CrossRef] [PubMed]
- Vardavas, C.I.; Nikitara, K. COVID-19 and smoking: A systematic review of the evidence. Tob. Induc. Dis. 2020, 18. [Google Scholar] [CrossRef]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- Guan, W.-J.; Ni, Z.-Y.; Hu, Y.; Liang, W.-H.; Ou, C.-Q.; He, J.-X.; Liu, L.; Shan, H.; Lei, C.-L.; Hui, D.S.C.; et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef]
- Tikellis, C.; Bernardi, S.; Burns, W.C. Angiotensin-converting enzyme 2 is a key modulator of the renin–angiotensin system in cardiovascular and renal disease. Curr. Opin. Nephrol. Hypertens. 2011, 20, 62–68. [Google Scholar] [CrossRef]
- Meng, Y.; Yu, C.-H.; Li, W.; Li, T.; Luo, W.; Huang, S.; Wu, P.-S.; Cai, S.-X.; Li, X. Angiotensin-Converting Enzyme 2/Angiotensin-(1-7)/Mas Axis Protects against Lung Fibrosis by Inhibiting the MAPK/NF-κB Pathway. Am. J. Respir. Cell Mol. Biol. 2013, 50, 723–736. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Li, Z.; Chen, Y.; Li, S.; Lv, Y.; Zhou, W.; Liao, M.; Zhu, F.; Zhou, Z.; Cheng, X.; et al. Autoantibodies targeting AT1 receptor from patients with acute coronary syndrome upregulate proinflammatory cytokines expression in endothelial cells involving NF-κB pathway. J. Immunol. Res. 2014, 2014, 342693. [Google Scholar] [CrossRef] [PubMed]
- Crowley, S.D.; Rudemiller, N.P. Immunologic Effects of the Renin-Angiotensin System. J. Am. Soc. Nephrol. 2017, 28, 1350–1361. [Google Scholar] [CrossRef] [PubMed]
- Glowacka, I.; Bertram, S.; Herzog, P.; Pfefferle, S.; Steffen, I.; Muench, M.O.; Simmons, G.; Hofmann, H.; Kuri, T.; Weber, F.; et al. Differential Downregulation of ACE2 by the Spike Proteins of Severe Acute Respiratory Syndrome Coronavirus and Human Coronavirus NL63. J. Virol. 2010, 84, 1198–1205. [Google Scholar] [CrossRef]
- Wang, W.; Yang, X.; Liang, J.; Liao, M.; Zhang, H.; Qin, X.; Mo, L.; Lv, W.; Mo, Z. Cigarette smoking has a positive and independent effect on testosterone levels. Hormones 2013, 12, 567–577. [Google Scholar] [CrossRef]
- Green, S.M.; Mostaghel, E.A.; Nelson, P.S. Androgen action and metabolism in prostate cancer. Mol. Cell Endocrinol. 2012, 360, 3–13. [Google Scholar] [CrossRef]
- Qing, E.; Hantak, M.P.; Galpalli, G.G.; Gallagher, T. Evaluating MERS-CoV Entry Pathways. Methods Mol. Biol. 2020, 2099, 9–20. [Google Scholar]
- Lukassen, S.; Chua, R.L.; Trefzer, T.; Kahn, N.C.; Schneider, M.A.; Muley, T.; Winter, H.; Meister, M.; Veith, C.; Boots, A.W.; et al. SARS-CoV-2 receptor ACE2 and TMPRSS2 are primarily expressed in bronchial transient secretory cells. EMBO J. 2020, 39, e105114. [Google Scholar] [CrossRef]
- Cai, G.; Bossé, Y.; Xiao, F.; Kheradmand, F.; Amos, C.I. Tobacco Smoking Increases the Lung Gene Expression of ACE2, the Receptor of SARS-CoV-2. Am. J. Respir. Crit. Care Med. 2020. [Google Scholar] [CrossRef]
- Stopsack, K.H.; Mucci, L.A.; Antonarakis, E.S.; Nelson, P.S.; Kantoff, P.W. TMPRSS2 and COVID-19: Serendipity or Opportunity for Intervention? Cancer Discov. 2020. [Google Scholar] [CrossRef]
- Pedersen, K.B.; Chodavarapu, H.; Porretta, C.; Robinson, L.K.; Lazartigues, E. Dynamics of ADAM17-Mediated Shedding of ACE2 Applied to Pancreatic Islets of Male db/db Mice. Endocrinology 2015, 156, 4411–4425. [Google Scholar] [CrossRef] [PubMed]
- Purayil, H.T.; Zhang, Y.; Dey, A.; Gersey, Z.; Espana-Serrano, L.; Daaka, Y. Arrestin2 modulates androgen receptor activation. Oncogene 2015, 34, 3144–3151. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.T.E.; Mansukhani, M.; Weinstein, I.B. Cyclin-dependent kinase 6 associates with the androgen receptor and enhances its transcriptional activity in prostate cancer cells. Proc. Natl. Acad. Sci. USA 2005, 102, 5156. [Google Scholar] [CrossRef] [PubMed]
- Gregory, C.W.; Presnell, S.C.; Mohler, J.L.; French, F.S. Androgen Receptor Regulation of G1 Cyclin and Cyclin-Dependent Kinase Function in the CWR22 Human Prostate Cancer Xenograft. J. Androl. 2001, 22, 537–548. [Google Scholar] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chakladar, J.; Shende, N.; Li, W.T.; Rajasekaran, M.; Chang, E.Y.; Ongkeko, W.M. Smoking-Mediated Upregulation of the Androgen Pathway Leads to Increased SARS-CoV-2 Susceptibility. Int. J. Mol. Sci. 2020, 21, 3627. https://doi.org/10.3390/ijms21103627
Chakladar J, Shende N, Li WT, Rajasekaran M, Chang EY, Ongkeko WM. Smoking-Mediated Upregulation of the Androgen Pathway Leads to Increased SARS-CoV-2 Susceptibility. International Journal of Molecular Sciences. 2020; 21(10):3627. https://doi.org/10.3390/ijms21103627
Chicago/Turabian StyleChakladar, Jaideep, Neil Shende, Wei Tse Li, Mahadevan Rajasekaran, Eric Y. Chang, and Weg M. Ongkeko. 2020. "Smoking-Mediated Upregulation of the Androgen Pathway Leads to Increased SARS-CoV-2 Susceptibility" International Journal of Molecular Sciences 21, no. 10: 3627. https://doi.org/10.3390/ijms21103627
APA StyleChakladar, J., Shende, N., Li, W. T., Rajasekaran, M., Chang, E. Y., & Ongkeko, W. M. (2020). Smoking-Mediated Upregulation of the Androgen Pathway Leads to Increased SARS-CoV-2 Susceptibility. International Journal of Molecular Sciences, 21(10), 3627. https://doi.org/10.3390/ijms21103627



