6-Shogaol Protects Human Melanocytes against Oxidative Stress through Activation of the Nrf2-Antioxidant Response Element Signaling Pathway
Abstract
1. Introduction
2. Results
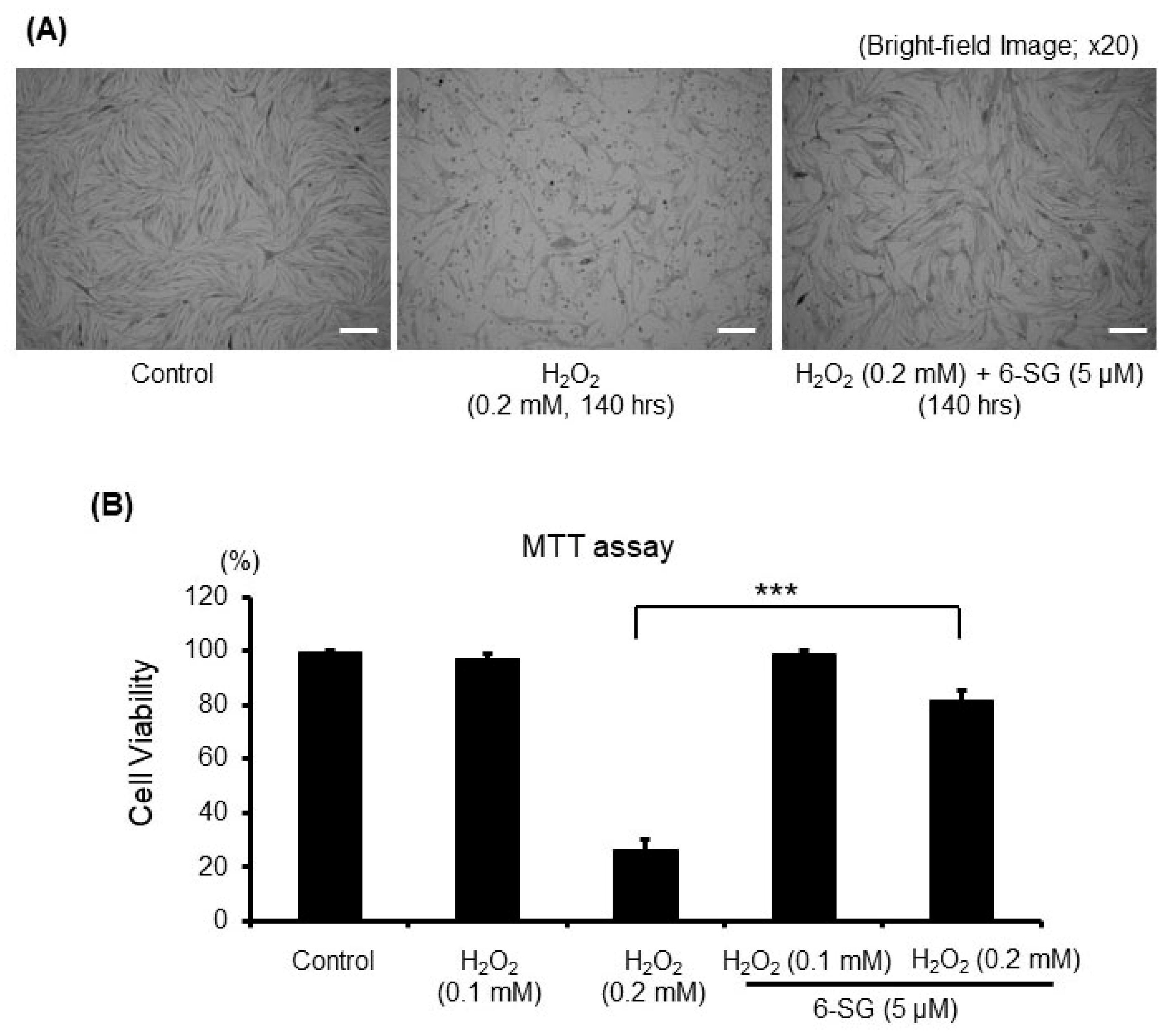
2.1. 6-SG Attenuates H2O2-Induced Cytotoxicity in Human Primary Epidermal Melanocytes
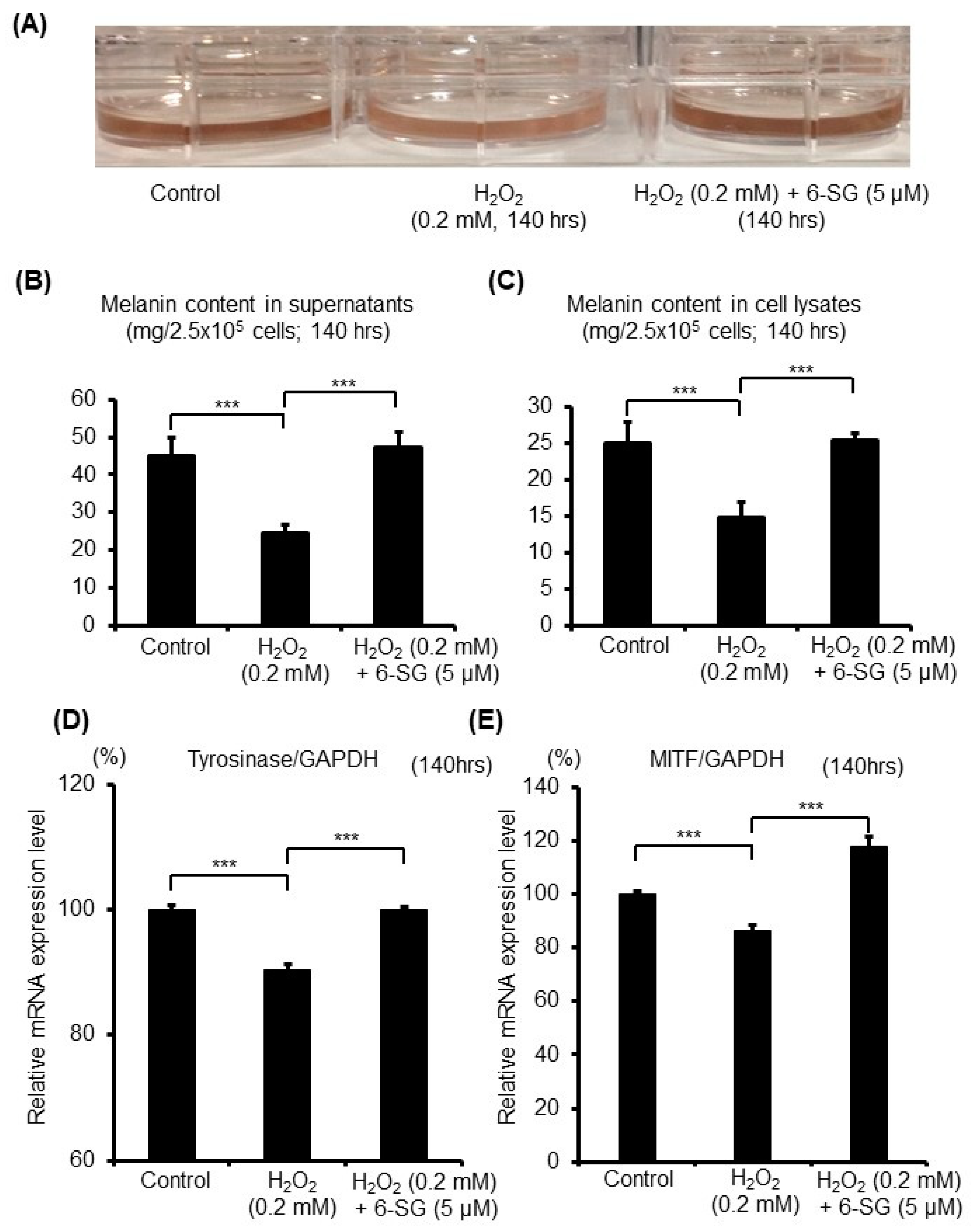
2.2. 6-SG Attenuates Reduced Melanogenesis in H2O2-Treated Human Primary Epidermal Melanocytes
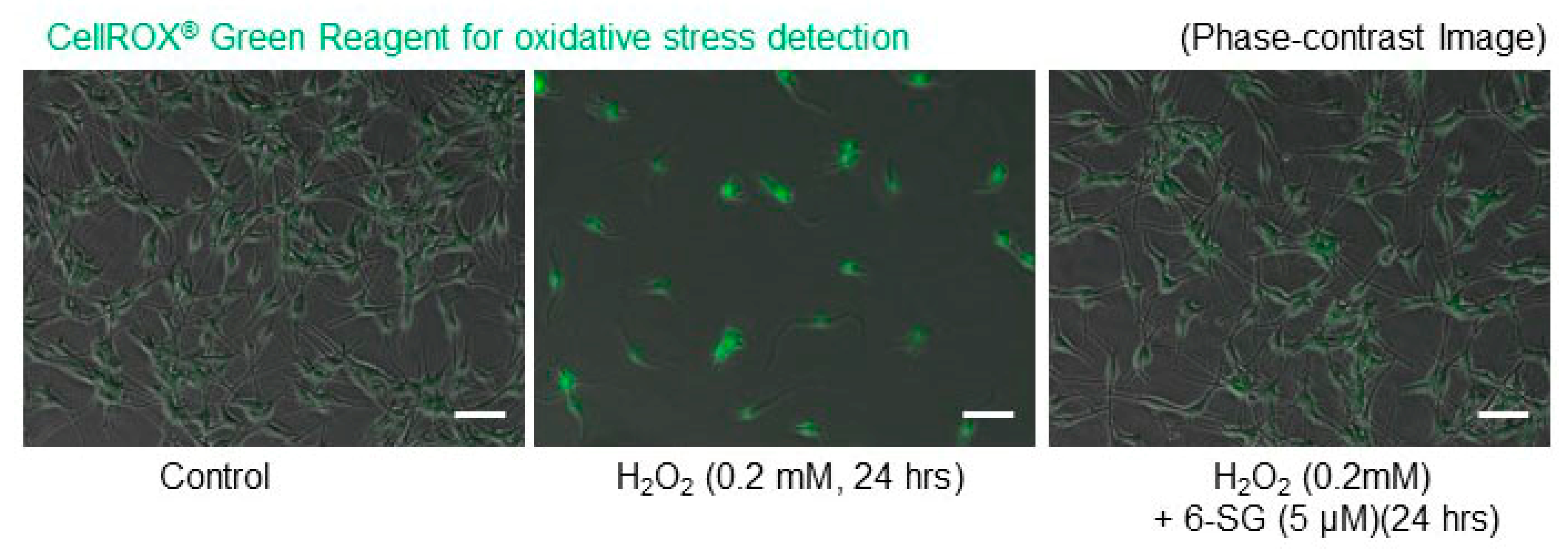
2.3. 6-SG Reduces H2O2-Induced Oxidative Stress in Human Epidermal Melanocytes
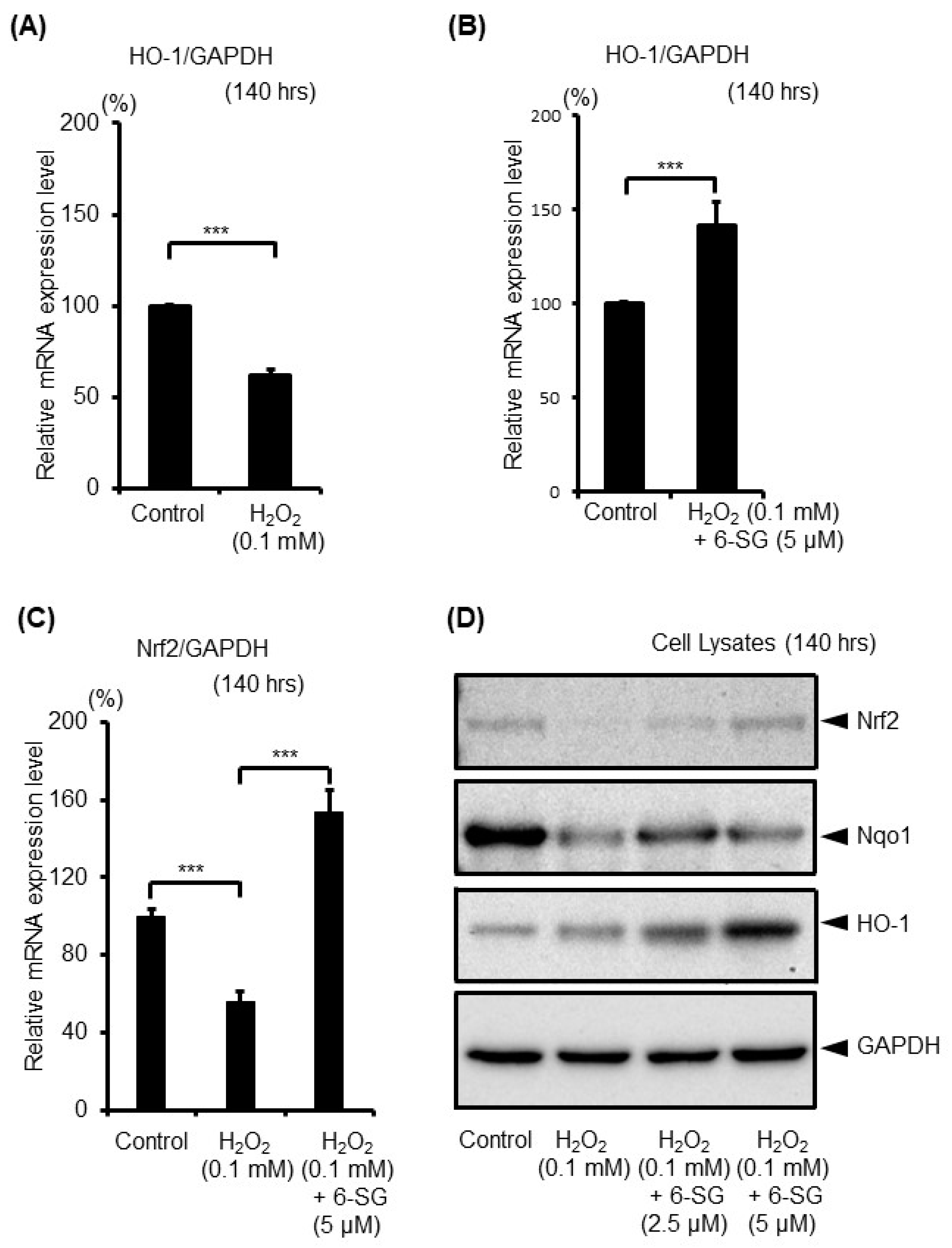
2.4. 6-SG Activates Intrinsic Antioxidant Defense Response in Human Epidermal Melanocytes
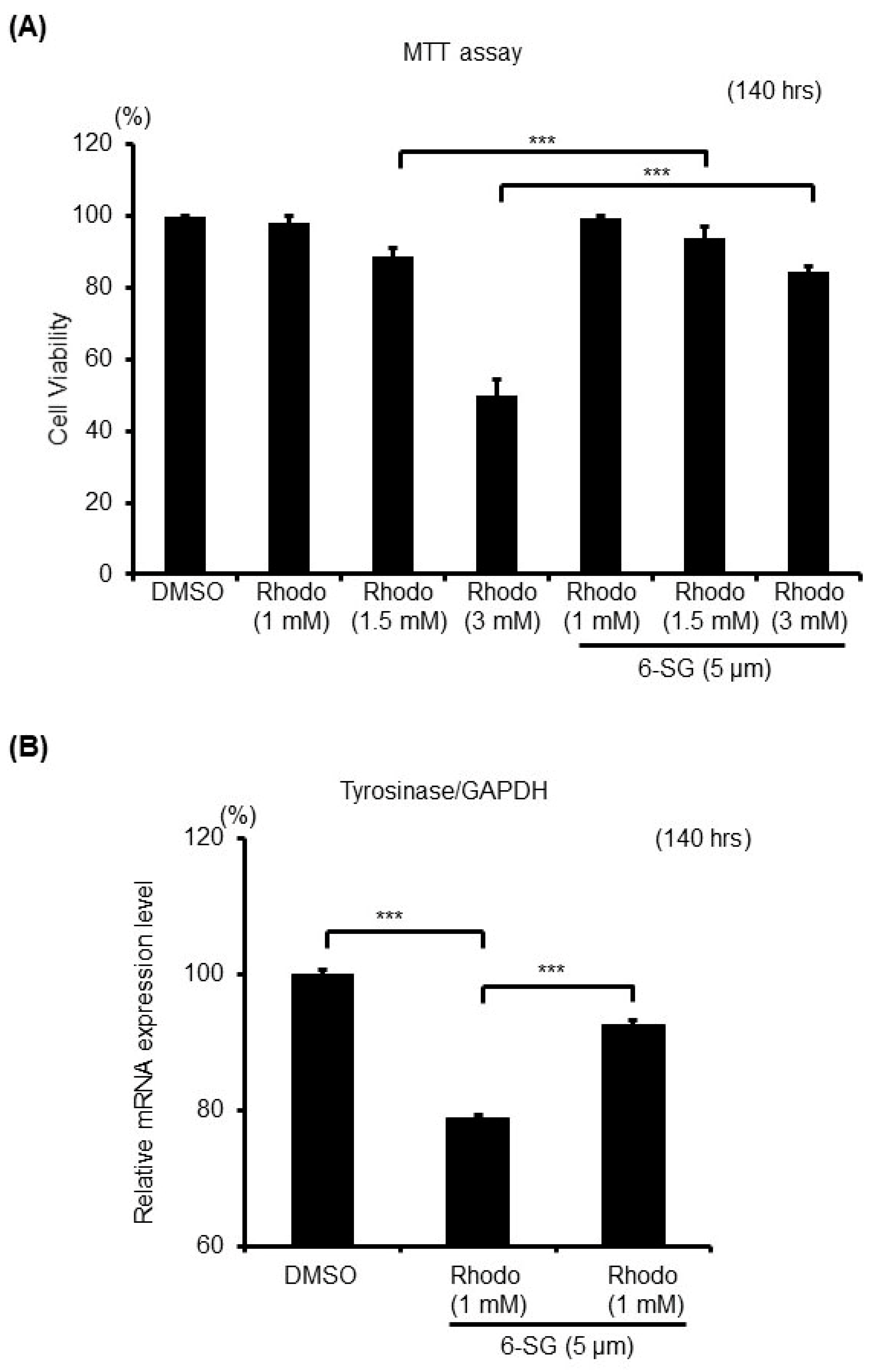
2.5. 6-SG Protects Melanocytes Against Rhododendrol-Induced Oxidative Stress
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Cell Culture
4.3. Cell Viability Assay
4.4. Melanin Content Assay
4.5. RNA Isolation and Real-Time RT-PCR Analysis
4.6. Western Blot Analysis
4.7. Oxidative Stress Assessment
4.8. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Denat, L.; Kadekaro, A.L.; Marrot, L.; Leachman, S.A.; Abdel-Malek, Z.A. Melanocytes as instigators and victims of oxidative stress. J. Investig. Dermatol. 2014, 134, 1512–1518. [Google Scholar] [CrossRef] [PubMed]
- Taieb, A.; Picardo, M. Clinical practice. Vitiligo. N. Engl. J. Med. 2009, 360, 160–169. [Google Scholar] [PubMed]
- Picardo, M.; Dell’Anna, M.L.; Ezzedine, K.; Hamzavi, I.; Harris, J.E.; Parsad, D.; Taieb, A. Vitiligo. Nature reviews. Dis. Prim. 2015, 1, 15011. [Google Scholar]
- Taieb, A. Intrinsic and extrinsic pathomechanisms in vitiligo. Pigment. Cell Res. 2000, 13, 41–47. [Google Scholar] [CrossRef]
- Maresca, V.; Roccella, M.; Roccella, F.; Camera, E.; Del Porto, G.; Passi, S.; Grammatico, P.; Picardo, M. Increased sensitivity to peroxidative agents as a possible pathogenic factor of melanocyte damage in vitiligo. J. Investig. Dermatol. 1997, 109, 310–313. [Google Scholar] [CrossRef]
- Schallreuter, K.U.; Moore, J.; Wood, J.M.; Beazley, W.D.; Gaze, D.C.; Tobin, D.J.; Marshall, H.S.; Panske, A.; Panzig, E.; Hibberts, N.A. In vivo and in vitro evidence for hydrogen peroxide (H2O2) accumulation in the epidermis of patients with vitiligo and its successful removal by a UVB-activated pseudocatalase. J. Investig. Dermatol. Symp. Proc. 1999, 4, 91–96. [Google Scholar] [CrossRef]
- Passi, S.; Grandinetti, M.; Maggio, F.; Stancato, A.; De Luca, C. Epidermal oxidative stress in vitiligo. Pigment Cell Res. 1998, 11, 81–85. [Google Scholar] [CrossRef]
- Yildirim, M.; Baysal, V.; Inaloz, H.S.; Can, M. The role of oxidants and antioxidants in generalized vitiligo at tissue level. J. Eur. Acad. Dermatol. Venereol. JEADV 2004, 18, 683–686. [Google Scholar] [CrossRef]
- Yildirim, M.; Baysal, V.; Inaloz, H.S.; Kesici, D.; Delibas, N. The role of oxidants and antioxidants in generalized vitiligo. J. Dermatol. 2003, 30, 104–108. [Google Scholar] [CrossRef]
- Akoglu, G.; Emre, S.; Metin, A.; Akbas, A.; Yorulmaz, A.; Isikoglu, S.; Sener, S.; Kilinc, F. Evaluation of total oxidant and antioxidant status in localized and generalized vitiligo. Clin. Exp. Dermatol. 2013, 38, 701–706. [Google Scholar] [CrossRef]
- Yanishlieva, N.V.; Marinova, E.; Pokorny, J. Natural antioxidants from herbs and spices. Eur. J. Lipid Sci. Technol. 2006, 108, 776–793. [Google Scholar] [CrossRef]
- Afzal, M.; Al-Hadidi, D.; Menon, M.; Pesek, J.; Dhami, M.S. Ginger: An ethnomedical, chemical and pharmacological review. Drug Metab. Drug Interact. 2001, 18, 159–190. [Google Scholar] [CrossRef] [PubMed]
- Zeng, G.F.; Zhang, Z.Y.; Lu, L.; Xiao, D.Q.; Zong, S.H.; He, J.M. Protective effects of ginger root extract on Alzheimer disease-induced behavioral dysfunction in rats. Rejuvenation Res. 2013, 16, 124–133. [Google Scholar] [CrossRef] [PubMed]
- Ha, S.K.; Moon, E.; Ju, M.S.; Kim, D.H.; Ryu, J.H.; Oh, M.S.; Kim, S.Y. 6-Shogaol, a ginger product, modulates neuroinflammation: A new approach to neuroprotection. Neuropharmacology 2012, 63, 211–223. [Google Scholar] [CrossRef]
- Lee, C.; Park, G.H.; Kim, C.Y.; Jang, J.H. [6]-Gingerol attenuates beta-amyloid-induced oxidative cell death via fortifying cellular antioxidant defense system. Food Chem. Toxicol. 2011, 49, 1261–1269. [Google Scholar] [CrossRef]
- Bellei, B.; Pitisci, A.; Ottaviani, M.; Ludovici, M.; Cota, C.; Luzi, F.; Dell’Anna, M.L.; Picardo, M. Vitiligo: A possible model of degenerative diseases. PLoS ONE 2013, 8, e59782. [Google Scholar] [CrossRef]
- Donata, S.; Kesavan, M.; Austin, S.; Mohan, K.S.; Rajagopalan, K.; Kuttan, R. Clinical trial of certain ayurvedic medicines indicated in vitiligo. Anc. Sci. Life 1990, 9, 202–206. [Google Scholar]
- She, J.; Bai, Y.; Liu, Z. Clinical Observation on The Curative Effect of Plum Blossom Needle, Ginger and Acupoint Injection of Autologous Venous Blood in The Treatment of Vitilligo. J. Pract. Tradit. Chin. Intern. Med. 2012, 2012, 40. [Google Scholar]
- Shukla, Y.; Singh, M. Cancer preventive properties of ginger: A brief review. Food Chem. Toxicol. 2007, 45, 683–690. [Google Scholar] [CrossRef]
- Suekawa, M.; Ishige, A.; Yuasa, K.; Sudo, K.; Aburada, M.; Hosoya, E. Pharmacological studies on ginger. I. Pharmacological actions of pungent constitutents, (6)-gingerol and (6)-shogaol. J. Pharm. 1984, 7, 836–848. [Google Scholar]
- Kim, E.C.; Min, J.K.; Kim, T.Y.; Lee, S.J.; Yang, H.O.; Han, S.; Kim, Y.M.; Kwon, Y.G. [6]-Gingerol, a pungent ingredient of ginger, inhibits angiogenesis in vitro and in vivo. Biochem. Biophys. Res. Commun. 2005, 335, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Koo, K.L.; Ammit, A.J.; Tran, V.H.; Duke, C.C.; Roufogalis, B.D. Gingerols and related analogues inhibit arachidonic acid-induced human platelet serotonin release and aggregation. Thromb. Res. 2001, 103, 387–397. [Google Scholar] [CrossRef]
- Dugasani, S.; Pichika, M.R.; Nadarajah, V.D.; Balijepalli, M.K.; Tandra, S.; Korlakunta, J.N. Comparative antioxidant and anti-inflammatory effects of [6]-gingerol, [8]-gingerol, [10]-gingerol and [6]-shogaol. J. Ethnopharmacol. 2010, 127, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Weng, C.J.; Wu, C.F.; Huang, H.W.; Ho, C.T.; Yen, G.C. Anti-invasion effects of 6-shogaol and 6-gingerol, two active components in ginger, on human hepatocarcinoma cells. Mol. Nutr. Food Res. 2010, 54, 1618–1627. [Google Scholar] [CrossRef] [PubMed]
- Peng, F.; Tao, Q.; Wu, X.; Dou, H.; Spencer, S.; Mang, C.; Xu, L.; Sun, L.; Zhao, Y.; Li, H.; et al. Cytotoxic, cytoprotective and antioxidant effects of isolated phenolic compounds from fresh ginger. Fitoterapia 2012, 83, 568–585. [Google Scholar] [CrossRef]
- Prota, G. Progress in the chemistry of melanins and related metabolites. Med. Res. Rev. 1988, 8, 525–556. [Google Scholar] [CrossRef]
- Yasumoto, K.; Yokoyama, K.; Shibata, K.; Tomita, Y.; Shibahara, S. Microphthalmia-associated transcription factor as a regulator for melanocyte-specific transcription of the human tyrosinase gene. Mol. Cell Biol. 1995, 15, 1833. [Google Scholar] [CrossRef]
- Motohashi, H.; Yamamoto, M. Nrf2-Keap1 defines a physiologically important stress response mechanism. Trends Mol. Med. 2004, 10, 549–557. [Google Scholar] [CrossRef]
- Kaspar, J.W.; Niture, S.K.; Jaiswal, A.K. Nrf2:INrf2 (Keap1) signaling in oxidative stress. Free Radic. Biol. Med. 2009, 47, 1304–1309. [Google Scholar] [CrossRef]
- Nguyen, T.; Nioi, P.; Pickett, C.B. The Nrf2-antioxidant response element signaling pathway and its activation by oxidative stress. J. Biol. Chem. 2009, 284, 13291–13295. [Google Scholar] [CrossRef]
- Tokura, Y.; Fujiyama, T.; Ikeya, S.; Tatsuno, K.; Aoshima, M.; Kasuya, A.; Ito, T. Biochemical, cytological, and immunological mechanisms of rhododendrol-induced leukoderma. J. Dermatol. Sci. 2015, 77, 146–149. [Google Scholar] [CrossRef] [PubMed]
- Ito, S.; Ojika, M.; Yamashita, T.; Wakamatsu, K. Tyrosinase-catalyzed oxidation of rhododendrol produces 2-methylchromane-6,7-dione, the putative ultimate toxic metabolite: Implications for melanocyte toxicity. Pigment Cell Melanoma Res. 2014, 27, 744–753. [Google Scholar] [CrossRef] [PubMed]
- Miyaji, A.; Gabe, Y.; Kohno, M.; Baba, T. Generation of hydroxyl radicals and singlet oxygen during oxidation of rhododendrol and rhododendrol-catechol. J. Clin. Biochem. Nutr. 2017, 60, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Gabe, Y.; Miyaji, A.; Kohno, M.; Hachiya, A.; Moriwaki, S.; Baba, T. Substantial evidence for the rhododendrol-induced generation of hydroxyl radicals that causes melanocyte cytotoxicity and induces chemical leukoderma. J Dermatol. Sci. 2018, 91, 311–316. [Google Scholar] [CrossRef] [PubMed]
- Jian, Z.; Li, K.; Liu, L.; Zhang, Y.; Zhou, Z.; Li, C.; Gao, T. Heme oxygenase-1 protects human melanocytes from H2O2-induced oxidative stress via the Nrf2-ARE pathway. J. Investig. Dermatol. 2011, 131, 1420–1427. [Google Scholar] [CrossRef]
- Hasse, S.; Gibbons, N.C.; Rokos, H.; Marles, L.K.; Schallreuter, K.U. Perturbed 6-tetrahydrobiopterin recycling via decreased dihydropteridine reductase in vitiligo: More evidence for H2O2 stress. J. Investig. Dermatol. 2004, 122, 307–313. [Google Scholar] [CrossRef]
- Spencer, J.D.; Gibbons, N.C.; Rokos, H.; Peters, E.M.; Wood, J.M.; Schallreuter, K.U. Oxidative stress via hydrogen peroxide affects proopiomelanocortin peptides directly in the epidermis of patients with vitiligo. J. Investig. Dermatol. 2007, 127, 411–420. [Google Scholar] [CrossRef]
- Spencer, J.D.; Gibbons, N.C.; Bohm, M.; Schallreuter, K.U. The Ca2+-binding capacity of epidermal furin is disrupted by H2O2-mediated oxidation in vitiligo. Endocrinology 2008, 149, 1638–1645. [Google Scholar] [CrossRef][Green Version]
- Guan, C.P.; Wei, X.D.; Chen, H.Y.; Zhang, L.; Zhou, M.N.; Xu, A.E. Abnormal nuclear translocation of nuclear factor-E2 related factor 2 in the lesion of vitiligo. Zhonghua Yi Xue Za Zhi 2008, 88, 2403–2406. [Google Scholar]
- Guan, C.P.; Zhou, M.N.; Xu, A.E.; Kang, K.F.; Liu, J.F.; Wei, X.D.; Li, Y.W.; Zhao, D.K.; Hong, W.S. The susceptibility to vitiligo is associated with NF-E2-related factor2 (Nrf2) gene polymorphisms: A study on Chinese Han population. Exp. Dermatol. 2008, 17, 1059–1062. [Google Scholar] [CrossRef]
- Qiu, L.; Song, Z.; Setaluri, V. Oxidative stress and vitiligo: The Nrf2-ARE signaling connection. J. Invest Dermatol. 2014, 134, 2074–2076. [Google Scholar] [CrossRef] [PubMed]
- Jian, Z.; Li, K.; Song, P.; Zhu, G.; Zhu, L.; Cui, T.; Liu, B.; Tang, L.; Wang, X.; Wang, G.; et al. Impaired activation of the Nrf2-ARE signaling pathway undermines H2O2-induced oxidative stress response: A possible mechanism for melanocyte degeneration in vitiligo. J. Investig. Dermatol. 2014, 134, 2221–2230. [Google Scholar] [CrossRef] [PubMed]
- Ademiluyi, A.O.; Oboh, G.; Ogunsuyi, O.B.; Akinyemi, A.J. Attenuation of gentamycin-induced nephrotoxicity in rats by dietary inclusion of ginger (Zingiber officinale) and turmeric (Curcuma longa) rhizomes. Nutr. Health 2012, 21, 209–218. [Google Scholar] [CrossRef]
- Bhandari, U.; Kanojia, R.; Pillai, K.K. Effect of ethanolic extract of Zingiber officinale on dyslipidaemia in diabetic rats. J. Ethnopharmacol. 2005, 97, 227–230. [Google Scholar] [CrossRef] [PubMed]
- Ajith, T.A.; Nivitha, V.; Usha, S. Zingiber officinale Roscoe alone and in combination with alpha-tocopherol protect the kidney against cisplatin-induced acute renal failure. Food Chem. Toxicol. 2007, 45, 921–927. [Google Scholar] [CrossRef]
- Shang, H.; Yang, D.; Zhang, W.; Li, T.; Ren, X.; Wang, X.; Zhao, W. Time course of Keap1-Nrf2 pathway expression after experimental intracerebral haemorrhage: Correlation with brain oedema and neurological deficit. Free Radic. Res. 2013, 47, 368–375. [Google Scholar] [CrossRef]
- Yang, F.; Yang, L.; Wataya-Kaneda, M.; Yoshimura, T.; Tanemura, A.; Katayama, I. Uncoupling of ER/Mitochondrial Oxidative Stress in mTORC1 Hyperactivation-Associated Skin Hypopigmentation. J. Investig. Dermatol. 2018, 138, 669–678. [Google Scholar] [CrossRef]
- Yang, L.L.; Murota, H.; Serada, S.; Fujimoto, M.; Kudo, A.; Naka, T.; Katayama, I. Histamine Contributes to Tissue Remodeling via Periostin Expression. J. Investig. Dermatol. 2014, 134, 2105–2113. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, L.; Yang, F.; Teng, L.; Katayama, I. 6-Shogaol Protects Human Melanocytes against Oxidative Stress through Activation of the Nrf2-Antioxidant Response Element Signaling Pathway. Int. J. Mol. Sci. 2020, 21, 3537. https://doi.org/10.3390/ijms21103537
Yang L, Yang F, Teng L, Katayama I. 6-Shogaol Protects Human Melanocytes against Oxidative Stress through Activation of the Nrf2-Antioxidant Response Element Signaling Pathway. International Journal of Molecular Sciences. 2020; 21(10):3537. https://doi.org/10.3390/ijms21103537
Chicago/Turabian StyleYang, Lingli, Fei Yang, Lanting Teng, and Ichiro Katayama. 2020. "6-Shogaol Protects Human Melanocytes against Oxidative Stress through Activation of the Nrf2-Antioxidant Response Element Signaling Pathway" International Journal of Molecular Sciences 21, no. 10: 3537. https://doi.org/10.3390/ijms21103537
APA StyleYang, L., Yang, F., Teng, L., & Katayama, I. (2020). 6-Shogaol Protects Human Melanocytes against Oxidative Stress through Activation of the Nrf2-Antioxidant Response Element Signaling Pathway. International Journal of Molecular Sciences, 21(10), 3537. https://doi.org/10.3390/ijms21103537




