Vitamin D and Cardiovascular Risk: Which Implications in Children?
Abstract
1. Introduction
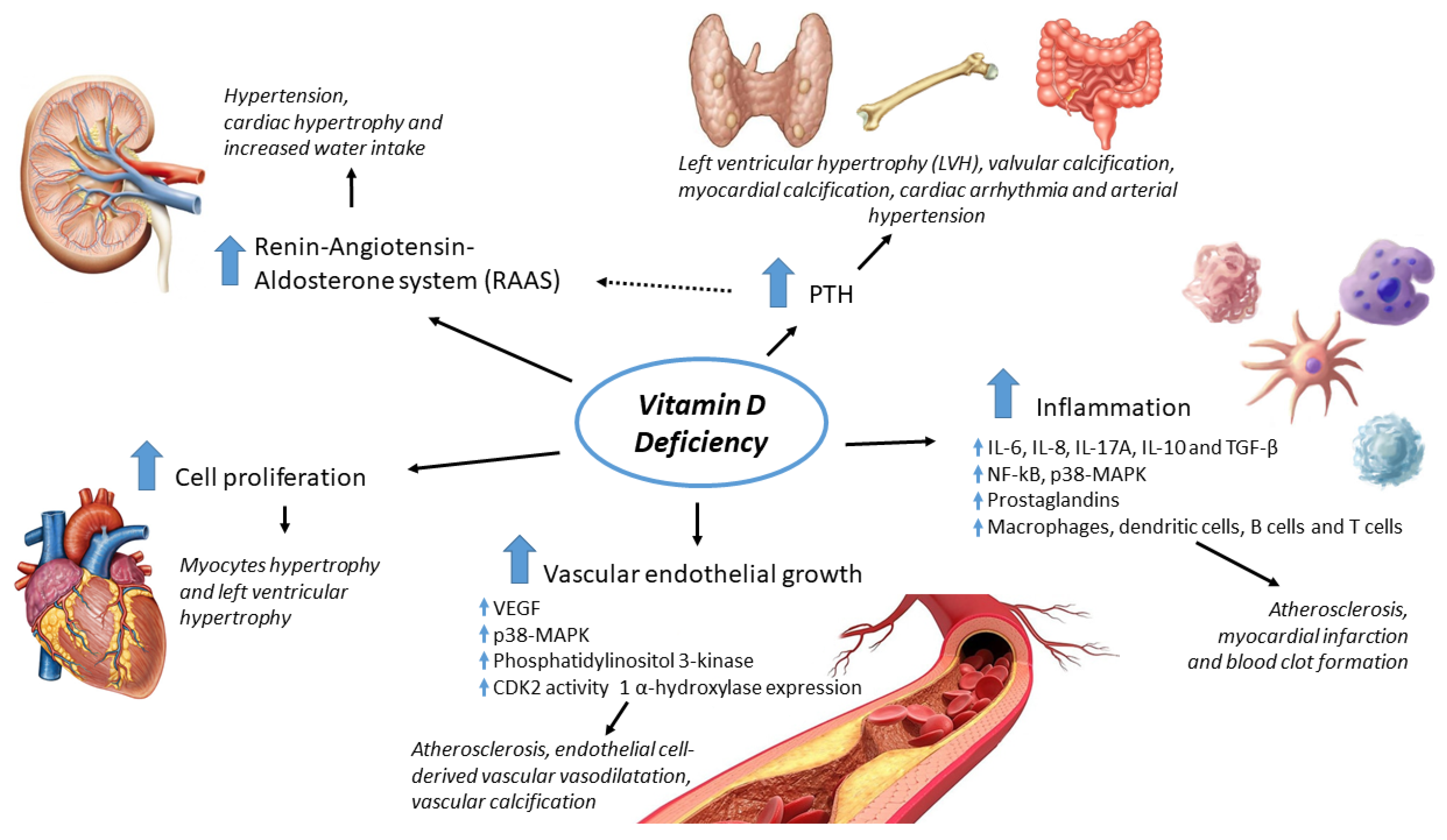
2. Vitamin D and the Cardiovascular Tissue: Pathophysiological Effects
3. Vitamin D Deficiency and Cardiovascular Risk Factors
4. The Impact of Vitamin D Supplementation
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| 1,25(OH)2D | 1,25-dihydroxyvitamin, calcitriol |
| 15-PGDH | 15-Hydroxyprostaglandin Dehydrogenase |
| 25OHD | Vitamin D, 25-hydroxyvitamin D |
| aIMT | Aortic intima media thickness |
| BMI | Body Mass Index |
| BMI-z | BMI-for-age z-score |
| CAD | Coronary artery disease |
| c-HDL | Cholesterol High Density Lipoprotein |
| cIMT | Carotid intima-media thickness |
| c-LDL | Cholesterol Low Density Lipoprotein |
| COX-2 | Cyclooxygenase-2 |
| CV | Cardiovascular |
| CVD | Cardiovascular disease |
| CYP27B1 | Cytochrome P450 27B1, 1-alpha hydroxylase |
| DBP | Diastolic blood pressure |
| HbA1c | Glycated hemoglobin |
| HDL | High Density Lipoprotein |
| HOMA-IR | Homeostasis Model Assessment of Insulin Resistance |
| hs-CRP | high-sensitivity C-Reactive Protein |
| IMT | Intima-media thickness |
| LDL | Low Density Lipoprotein |
| LVH | Left ventricular hypertrophy |
| PTH | Parathormone |
| RAAS | Renin-angiotensin-aldosterone system |
| RAR | Retinoic Acid Receptor |
| RCT | Randomized controlled trials |
| RXR | Retinoid X Receptors |
| SBP | Systolic blood pression |
| TC | Total cholesterol |
| TG | Triglycerides |
| UV | Ultraviolet |
| VDBP | Vitamin D binding protein |
| VDR | Vitamin D receptor |
| VDRE | Vitamin D response element |
| VEGF | Vascular endothelial growth factor |
| VSMC | Vascular smooth muscle cell |
| WBISI | Whole Body Insulin Sensitivity Index |
References
- Al-Ishaq, R.K.; Kubatka, P.; Brozmanova, M.; Gazdikova, K.; Caprnda, M.; Büsselberg, D. Health implication of vitamin D on the cardiovascular and the renal system. Arch. Physiol. Biochem. 2019, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Swier, V.J.; Boosani, C.S.; Radwan, M.M.; Agrawal, D.K. Vitamin D Deficiency Accelerates Coronary Artery Disease Progression in Swine. Arterioscler. Thromb. Vasc. Biol. 2016, 36, 1651–1659. [Google Scholar] [CrossRef] [PubMed]
- Sirajudeen, S.; Shah, I.; Al Menhali, A. A narrative role of vitamin d and its receptor: With current evidence on the gastric tissues. Int. J. Mol. Sci. 2019, 20, 3832. [Google Scholar] [CrossRef] [PubMed]
- Bikle, D. Vitamin D: Production, Metabolism, and Mechanisms of Action; MDText.com, Inc.: South Dartmouth, MA, USA, 2000. [Google Scholar]
- Tripkovic, L.; Lambert, H.; Hart, K.; Smith, C.P.; Bucca, G.; Penson, S.; Chope, G.; Hypponen, E.; Berry, J.; Vieth, R.; et al. Comparison of vitamin D2and vitamin D3 supplementation in raising serum 25-hydroxyvitamin D status: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2012, 95, 1357–1364. [Google Scholar] [CrossRef] [PubMed]
- Norman, A.W. From vitamin D to hormone D: Fundamentals of vitamin D endocrine system essential for good health. Am. J. Clin. Nutr. 2008, 88, 491S–499S. [Google Scholar] [CrossRef]
- Dalan, R.; Liew, H.; Tan, W.K.A.; Chew, D.E.K.; Leow, M.K.S. Vitamin D and the endothelium: Basic, translational and clinical research updates. IJC Metab. Endocr. 2014, 4, 4–17. [Google Scholar] [CrossRef]
- Maestro, M.A.; Molnár, F.; Mouriño, A.; Carlberg, C. Vitamin D receptor 2016: Novel ligands and structural insights. Expert Opin. Ther. Pat. 2016, 26, 1291–1306. [Google Scholar] [CrossRef]
- Hii, C.S.; Ferrante, A. The non-genomic actions of vitamin D. Nutrients 2016, 8, 135. [Google Scholar] [CrossRef]
- Norman, A.W. Minireview: Vitamin D receptor: New assignments for an already busy receptor. Endocrinology 2006, 147, 5542–5548. [Google Scholar] [CrossRef]
- Bellan, M.; Andreoli, L.; Mele, C.; Sainaghi, P.P.; Rigamonti, C.; Piantoni, S.; De Benedittis, C.; Aimaretti, G.; Pirisi, M.; Marzullo, P. Pathophysiological role and therapeutic implications of vitamin D in autoimmunity: Focus on chronic autoimmune diseases. Nutrients 2020, 12, 789. [Google Scholar] [CrossRef]
- Antonucci, R.; Locci, C.; Clemente, M.G.; Chicconi, E.; Antonucci, L. Vitamin D deficiency in childhood: Old lessons and current challenges. J. Pediatr. Endocrinol. Metab. 2018, 31, 247–260. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Kong, J.; Duan, Y.; Szeto, F.L.; Liao, A.; Madara, J.L.; Yan, C.L. Increased NF-κB activity in fibroblasts lacking the vitamin D receptor. Am. J. Physiol. Endocrinol. Metab. 2006, 291, E315–E322. [Google Scholar] [CrossRef] [PubMed]
- Janjetovic, Z.; Zmijewski, M.A.; Tuckey, R.C.; DeLeon, D.A.; Nguyen, M.N.; Pfeffer, L.M.; Slominski, A.T. 20-Hydroxycholecalciferol, product of vitamin D3 hydroxylation by P450scc, decreases NF-kappaB activity by increasing IkappaB alpha levels in human keratinocytes. PLoS ONE 2009, 4, e5988. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Kong, J.; Sun, T.; Li, G.; Szeto, F.L.; Liu, W.; Deb, D.K.; Wang, Y.; Zhao, Q.; Thadhani, R.; et al. 1,25-Dihydroxyvitamin D3 suppresses inflammation-induced expression of plasminogen activator inhibitor-1 by blocking nuclear factor-κB activation. Arch. Biochem. Biophys. 2011, 507, 241–247. [Google Scholar] [CrossRef]
- Valen, G.; Yan, Z.Q.; Hansson, G.K. Nuclear factor kappa-B and the heart. J. Am. Coll. Cardiol. 2001, 38, 307–314. [Google Scholar] [CrossRef]
- Low Wang, C.C.; Hess, C.N.; Hiatt, W.R.; Goldfine, A.B. Clinical update: Cardiovascular disease in diabetes mellitus. Circulation 2016, 133, 2459–2502. [Google Scholar] [CrossRef]
- Derakhshanian, H.; Djazayery, A.; Javanbakht, M.H.; Eshraghian, M.R.; Mirshafiey, A.; Jahanabadi, S.; Ghadbeigi, S.; Zarei, M.; Alvandi, E.; Djalali, M. Vitamin D downregulates key genes of diabetes complications in cardiomyocyte. J. Cell. Physiol. 2019, 234, 21352–21358. [Google Scholar] [CrossRef]
- Al-Rasheed, N.M.; Al-Rasheed, N.M.; Bassiouni, Y.A.; Hasan, I.H.; Al-Amin, M.A.; Al-Ajmi, H.N.; Mohamad, R.A. Vitamin D attenuates pro-inflammatory TNF-α cytokine expression by inhibiting NF-кB/p65 signaling in hypertrophied rat hearts. J. Physiol. Biochem. 2015, 71, 289–299. [Google Scholar] [CrossRef]
- Liu, W.; Zhang, L.; Xu, H.J.; Li, Y.; Hu, C.M.; Yang, J.Y.; Sun, M.Y. The anti-inflammatory effects of vitamin D in tumorigenesis. Int. J. Mol. Sci. 2018, 19, 2736. [Google Scholar] [CrossRef]
- El-Fakhri, N.; McDevitt, H.; Shaikh, M.G.; Halsey, C.; Ahmed, S.F. Vitamin D and its effects on glucose homeostasis, cardiovascular function and immune function. Horm. Res. Paediatr. 2014, 81, 363–378. [Google Scholar] [CrossRef]
- Weng, S.; Sprague, J.E.; Oh, J.; Riek, A.E.; Chin, K.; Garcia, M.; Bernal-Mizrachi, C. Vitamin D deficiency induces high blood pressure and accelerates atherosclerosis in mice. PLoS ONE 2013, 8, e54625. [Google Scholar] [CrossRef] [PubMed]
- Jia, G.; Aroor, A.R.; Hill, M.A.; Sowers, J.R. Role of renin-angiotensin-aldosterone system activation in promoting cardiovascular fibrosis and stiffness. Hypertension 2018, 72, 537–548. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.C.; Kong, J.; Wei, M.; Chen, Z.-F.; Liu, S.Q.; Cao, L.-P. 1,25-Dihydroxyvitamin D3 is a negative endocrine regulator of the renin-angiotensin system. J. Clin. Invest. 2002, 110, 229–238. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.C.; Qiao, G.; Uskokovic, M.; Xiang, W.; Zheng, W.; Kong, J. Vitamin D: A negative endocrine regulator of the renin-angiotensin system and blood pressure. J. Steroid Biochem. Mol. Biol. 2004, 89, 387–392. [Google Scholar] [CrossRef] [PubMed]
- Chandel, N.; Ayasolla, K.; Wen, H.; Lan, X.; Haque, S.; Saleem, M.A.; Malhotra, A.; Singhal, P.C. Vitamin D receptor deficit induces activation of renin angiotensin system via SIRT1 modulation in podocytes. Exp. Mol. Pathol. 2017, 102, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Zittermann, A.; Ernst, J.B.; Prokop, S.; Fuchs, U.; Dreier, J.; Kuhn, J.; Knabbe, C.; Börgermann, J.; Berthold, H.K.; Pilz, S.; et al. Effects of Vitamin D Supplementation on Renin and Aldosterone Concentrations in Patients with Advanced Heart Failure: The EVITA Trial. Int. J. Endocrinol. 2018, 2018, 5015417. [Google Scholar] [CrossRef]
- Andersson, P.; Rydberg, E.; Willenheimer, R. Primary hyperparathyroidism and heart disease—A review. Eur. Heart J. 2004, 25, 1776–1787. [Google Scholar] [CrossRef]
- Foley, R.N.; Parfrey, P.S.; Harnett, J.D.; Kent, G.M.; Martin, C.J.; Murray, D.C.; Barre, P.E. Clinical and echocardiographic disease in patients starting end-stage renal disease therapy. Kidney Int. 1995, 47, 186–192. [Google Scholar] [CrossRef]
- Anderson, J.L.; Vanwoerkom, R.C.; Horne, B.D.; Bair, T.L.; May, H.T.; Lappé, D.L.; Muhlestein, J.B. Parathyroid hormone, vitamin D, renal dysfunction, and cardiovascular disease: Dependent or independent risk factors? Am. Heart J. 2011, 162, 3313–3339. [Google Scholar] [CrossRef]
- Van Ballegooijen, A.J.; Reinders, I.; Visser, M.; Brouwer, I.A. Parathyroid hormone and cardiovascular disease events: A systematic review and meta-analysis of prospective studies. Am. Heart J. 2013, 165, 655–664. [Google Scholar] [CrossRef]
- Brown, S.J.; Ruppe, M.D.; Tabatabai, L.S. The Parathyroid Gland and Heart Disease. Methodist Debakey Cardiovasc. J. 2017, 13, 49–54. [Google Scholar] [CrossRef] [PubMed]
- O’Connell, T.D.; Berry, J.E.; Jarvis, A.K.; Somerman, M.J.; Simpson, R.U. 1, 25-Dihydroxyvitamin D3 regulation of cardiac myocyte proliferation and hypertrophy. Am. J. Physiol. 1997, 272, H1751–H1758. [Google Scholar] [CrossRef]
- Patange, A.R.; Valentini, R.P.; Gothe, M.P.; Du, W.; Pettersen, M.D. Vitamin D deficiency is associated with increased left ventricular mass and diastolic dysfunction in children with chronic kidney disease. Pediatr. Cardiol. 2013, 34, 536–542. [Google Scholar] [CrossRef] [PubMed]
- Şeker, T.; Gür, M.; Uçar, H.; Türkoğlu, C.; Baykan, A.O.; Özaltun, B.; Harbalıoğlu, H.; Kalkan, G.Y.; Kaypaklı, O.; Kuloğlu, O.; et al. Lower serum 25-hydroxyvitamin D level is associated with impaired myocardial performance and left ventricle hypertrophy in newly diagnosed hypertensive patientshipertrofisi ile ilişkilidir. Anatol. J. Cardiol. 2015, 15, 744–750. [Google Scholar] [CrossRef]
- Yamamoto, T.; Kozawa, O.; Tanabe, K.; Akamatsu, S.; Matsuno, H.; Dohi, S.; Hirose, H.; Uematsu, T. 1,25-dihydroxyvitamin d3 stimulates vascular endothelial growth factor release in aortic smooth muscle cells: Role of p38 mitogen-activated protein kinase. Arch. Biochem. Biophys. 2002, 398, 1–6. [Google Scholar] [CrossRef]
- Cardus, A.; Panizo, S.; Encinas, M.; Dolcet, X.; Gallego, C.; Aldea, M.; Fernandez, E.; Valdivielso, J.M. 1,25-Dihydroxyvitamin D3 regulates VEGF production through a vitamin D response element in the VEGF promoter. Atherosclerosis 2009, 204, 85–89. [Google Scholar] [CrossRef]
- Martinez-Moreno, J.M.; Herencia, C.; De Oca, A.M.; Muñoz-Castañeda, J.R.; Rodríguez-Ortiz, M.E.; Diáz-Tocados, J.M.; Peralbo-Santaella, E.; Camargo, A.; Canalejo, A.; Rodriguez, M.; et al. Vitamin D modulates tissue factor and protease-activated receptor 2 expression in vascular smooth muscle cells. FASEB J. 2016, 30, 1367–1376. [Google Scholar] [CrossRef]
- Rebsamen, M.C.; Sun, J.; Norman, A.W.; Liao, J.K. 1α,25-dihydroxyvitamin D3 induces vascular smooth muscle cell migration via activation of phosphatidylinositol 3-kinase. Circ. Res. 2002, 91, 17–24. [Google Scholar] [CrossRef]
- Zhou, W.; Wang, Q. Vitamin D suppresses lipopolysaccharide-induced inflammatory response in vascular smooth muscle cells via inhibition of the p38 MAPK signaling pathway. Pharmazie 2019, 74, 168–174. [Google Scholar] [CrossRef]
- Chen, S.; Law, C.S.; Grigsby, C.L.; Olsen, K.; Gardner, D.G. A role for the cell cycle phosphatase Cdc25a in vitamin D-dependent inhibition of adult rat vascular smooth muscle cell proliferation. J. Steroid Biochem. Mol. Biol. 2010, 122, 326–332. [Google Scholar] [CrossRef]
- Borges, A.C.R.; Feres, T.; Vianna, L.M.; Paiva, T.B. Cholecalciferol treatment restores the relaxant responses of spontaneously hypertensive rat arteries to bradykinin. Pathophysiology 2002, 8, 263–268. [Google Scholar] [CrossRef]
- Torremadé, N.; Bozic, M.; Panizo, S.; Barrio-Vazquez, S.; Fernandez-Martín, J.L.; Encinas, M.; Goltzman, D.; Arcidiacono, M.V.; Fernandez, E.; Valdivielso, J.M. Vascular Calcification Induced by Chronic Kidney Disease Is Mediated by an Increase of 1α-Hydroxylase Expression in Vascular Smooth Muscle Cells. J. Bone Miner. Res. 2016, 31, 1865–1876. [Google Scholar] [CrossRef]
- Rai, V.; Agrawal, D.K. Role of Vitamin D in Cardiovascular Diseases. Endocrinol. Metab. Clin. N. Am. 2017, 46, 1039–1059. [Google Scholar] [CrossRef] [PubMed]
- Krishna, S.M. Vitamin D as A Protector of Arterial Health: Potential Role in Peripheral Arterial Disease Formation. Int. J. Mol. Sci. 2019, 20, 4907. [Google Scholar] [CrossRef] [PubMed]
- De Jesus, J.M. Expert panel on integrated guidelines for cardiovascular health and risk reduction in children and adolescents: Summary report. Pediatrics 2011, 128, S213. [Google Scholar]
- Valerio, G.; Maffeis, C.; Saggese, G.; Ambruzzi, M.A.; Balsamo, A.; Bellone, S.; Bergamini, M.; Bernasconi, S.; Bona, G.; Calcaterra, V.; et al. Diagnosis, treatment and prevention of pediatric obesity: Consensus position statement of the Italian Society for Pediatric Endocrinology and Diabetology and the Italian Society of Pediatrics. Ital. J. Pediatr. 2018, 44, 88. [Google Scholar] [CrossRef]
- Skinner, A.C.; Ravanbakht, S.N.; Skelton, J.A.; Perrin, E.M.; Armstrong, S.C. Prevalence of obesity and severe obesity in US children, 1999–2016. Pediatrics 2018, 141, e20173459. [Google Scholar] [CrossRef]
- Colak, R.; Anil, M.; Yasar, F.; Rahmi Bakiler, A.; Pirgon, O.; Helvaci, M.; Dündar, B. Metabolic disturbances and cardiovascular risk factors in obese children with vitamin D deficiency. Arch. Pediatr. 2020. [Google Scholar] [CrossRef]
- Censani, M.; Hammad, H.T.; Christos, P.J.; Schumaker, T. Vitamin D Deficiency Associated With Markers of Cardiovascular Disease in Children with Obesity. Glob. Pediatr. Health 2018, 5. [Google Scholar] [CrossRef]
- Iqbal, A.; Dahl, A.; Lteif, A.; Kumar, S. Vitamin D Deficiency: A Potential Modifiable Risk Factor for Cardiovascular Disease in Children with Severe Obesity. Children 2017, 4, 80. [Google Scholar] [CrossRef]
- Lee, M.; Ebert, J.R.; Kadakia, M.P.; Zhang, J.; Czerwinski, S.A. Inverse associations between cardiometabolic risk factors and 25-hydroxyvitamin D in obese American children and adolescents. Am. J. Hum. Biol. 2016, 28, 736–742. [Google Scholar] [CrossRef] [PubMed]
- Kao, K.-T.; Abidi, N.; Ranasinha, S.; Brown, J.; Rodda, C.; McCallum, Z.; Zacharin, M.; Simm, P.J.; Magnussen, C.G.; Sabin, M.A. Low vitamin D is associated with hypertension in paediatric obesity. J. Paediatr. Child Health 2015, 51, 1207–1213. [Google Scholar] [CrossRef] [PubMed]
- Earthman, C.P.; Beckman, L.M.; Masodkar, K.; Sibley, S.D. The link between obesity and low circulating 25-hydroxyvitamin D concentrations: Considerations and implications. Int. J. Obes. 2012, 36, 387–396. [Google Scholar] [CrossRef] [PubMed]
- Petersen, R.A.; Dalskov, S.M.; Sorensen, L.B.; Hjorth, M.F.; Andersen, R.; Tetens, I.; Krarup, H.; Ritz, C.; Astrup, A.; Michaelsen, K.F.; et al. Vitamin D status is associated with cardiometabolic markers in 8–11-year-old children, independently of body fat and physical activity. Br. J. Nutr. 2015, 114, 1647–1655. [Google Scholar] [CrossRef]
- Kim, M.R.; Jeong, S.J. Relationship between vitamin D level and lipid profile in non-obese children. Metabolites 2019, 9, 125. [Google Scholar] [CrossRef]
- Liang, X.; Chen, M.; Qu, P.; Hao, G.; Huang, Y.; Chen, J.; Li, T. The Association of Vitamin A and Vitamin D with Hypertension in Children: A Case-Control Study. Int. J. Hypertens. 2018, 2018, 9295147. [Google Scholar] [CrossRef]
- Paul Baker, C.; Kulkarni, B.; Radhakrishna, K.V.; Charyulu, M.S.; Gregson, J.; Matsuzaki, M.; Taylor, A.E.; Prabhakaran, D.; Mamidi, R.S.; Wells, J.; et al. Is the association between Vitamin D and cardiovascular disease risk confounded by obesity? Evidence from the Andhra Pradesh Children and Parents Study (APCAPS). PLoS ONE 2015, 10, e0129468. [Google Scholar] [CrossRef]
- Arman, D.; Çetiner, Z. The relationship between serum vitamin D levels and intima-media thickness in term infants. Eur. J. Pediatr. 2019, 178, 1087–1093. [Google Scholar] [CrossRef]
- Sauder, K.A.; Stamatoiu, A.V.; Leshchinskaya, E.; Ringham, B.M.; Glueck, D.H.; Dabelea, D. Cord Blood Vitamin D Levels and Early Childhood Blood Pressure: The Healthy Start Study. J. Am. Heart Assoc. 2019, 8, e011485. [Google Scholar] [CrossRef]
- Salonen, J.T.; Salonen, R. Ultrasonographically assessed carotid morphology and the risk of coronary heart disease. Arterioscler. Thromb. 1991, 11, 1245–1249. [Google Scholar] [CrossRef]
- Theodoratou, E.; Tzoulaki, I.; Zgaga, L.; Ioannidis, J.P.A. Vitamin D and multiple health outcomes: Umbrella review of systematic reviews and meta-analyses of observational studies and randomised trials. BMJ 2014, 348, g2035. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Liu, X.; Bartell, T.R.; Pearson, C.; Cheng, T.L.; Wang, X. Vitamin D Trajectories from Birth to Early Childhood and Elevated Systolic Blood Pressure During Childhood and Adolescence. Hypertension 2019, 74, 421–430. [Google Scholar] [CrossRef] [PubMed]
- Miliku, K.; Felix, J.F.; Voortman, T.; Tiemeier, H.; Eyles, D.W.; Burne, T.H.; McGrath, J.J.; Jaddoe, V.W.V. Associations of maternal and fetal vitamin D status with childhood body composition and cardiovascular risk factors. Matern. Child Nutr. 2019, 15, e12672. [Google Scholar] [CrossRef]
- Stagi, S.; Rigante, D.; Lepri, G.; Matucci Cerinic, M.; Falcini, F. Severe vitamin D deficiency in patients with Kawasaki disease: A potential role in the risk to develop heart vascular abnormalities? Clin. Rheumatol. 2016, 35, 1865–1872. [Google Scholar] [CrossRef] [PubMed]
- Le, T.Y.L.; Ogawa, M.; Kizana, E.; Gunton, J.E.; Chong, J.J.H. Vitamin D Improves Cardiac Function After Myocardial Infarction Through Modulation of Resident Cardiac Progenitor Cells. Heart. Lung Circ. 2018, 27, 967–975. [Google Scholar] [CrossRef] [PubMed]
- Yin, K.; You, Y.; Swier, V.; Tang, L.; Radwan, M.M.; Pandya, A.N.; Agrawal, D.K. Vitamin D Protects Against Atherosclerosis via Regulation of Cholesterol Efflux and Macrophage Polarization in Hypercholesterolemic Swine. Arterioscler. Thromb. Vasc. Biol. 2015, 35, 2432–2442. [Google Scholar] [CrossRef] [PubMed]
- Xiang, W.; Hu, Z.-L.; He, X.-J.; Dang, X.-Q. Intravenous transfusion of endothelial progenitor cells that overexpress vitamin D receptor inhibits atherosclerosis in apoE-deficient mice. Biomed. Pharmacother. 2016, 84, 1233–1242. [Google Scholar] [CrossRef]
- Beveridge, L.A.; Struthers, A.D.; Khan, F.; Jorde, R.; Scragg, R.; Macdonald, H.M.; Alvarez, J.A.; Boxer, R.S.; Dalbeni, A.; Gepner, A.D.; et al. Effect of vitamin D supplementation on blood pressure a systematic review and meta-analysis incorporating individual patient data. JAMA Intern. Med. 2015, 175, 745–754. [Google Scholar] [CrossRef]
- Legarth, C.; Grimm, D.; Krüger, M.; Wehland, M.; Infanger, M. Potential beneficial effects of vitamin d in coronary artery disease. Nutrients 2020, 12, 99. [Google Scholar] [CrossRef]
- Legarth, C.; Grimm, D.; Wehland, M.; Bauer, J.; Krüger, M. The impact of vitamin d in the treatment of essential hypertension. Int. J. Mol. Sci. 2018, 19, 455. [Google Scholar] [CrossRef]
- Rejnmark, L.; Bislev, L.S.; Cashman, K.D.; Eiríksdottir, G.; Gaksch, M.; Grübler, M.; Grimnes, G.; Gudnason, V.; Lips, P.; Pilz, S.; et al. Non-skeletal health effects of Vitamin D supplementation: A systematic review on findings from meta-Analyses summarizing trial data. PLoS ONE 2017, 12, e0180512. [Google Scholar] [CrossRef] [PubMed]
- Beveridge, L.A.; Khan, F.; Struthers, A.D.; Armitage, J.; Barchetta, I.; Bressendorff, I.; Cavallo, M.G.; Clarke, R.; Dalan, R.; Dreyer, G.; et al. Effect of vitamin D supplementation on markers of vascular function: A systematic review and individual participant meta- analysis. J. Am. Heart Assoc. 2018, 7, e008273. [Google Scholar] [CrossRef] [PubMed]
- Hauger, H.; Laursen, R.P.; Ritz, C.; Mølgaard, C.; Lind, M.V.; Damsgaard, C.T. Effects of vitamin D supplementation on cardiometabolic outcomes in children and adolescents: A systematic review and meta-analysis of randomized controlled trials. Eur. J. Nutr. 2020, 59, 873–884. [Google Scholar] [CrossRef] [PubMed]
- Hauger, H.; Mølgaard, C.; Mortensen, C.; Ritz, C.; Frøkiær, H.; Smith, T.J.; Hart, K.; Lanham-New, S.A.; Damsgaard, C.T. Winter Cholecalciferol Supplementation at 55° N Has No Effect on Markers of Cardiometabolic Risk in Healthy Children Aged 4–8 Years. J. Nutr. 2018, 148, 1261–1268. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.J.; Tripkovic, L.; Hauger, H.; Damsgaard, C.T.; Mølgaard, C.; Lanham-New, S.A.; Hart, K.H. Winter Cholecalciferol Supplementation at 51° N Has No Effect on Markers of Cardiometabolic Risk in Healthy Adolescents Aged 14–18 Years. J. Nutr. 2018, 148, 1269–1275. [Google Scholar] [CrossRef] [PubMed]
- Ferira, A.J.; Laing, E.M.; Hausman, D.B.; Hall, D.B.; McCabe, G.P.; Martin, B.R.; Hill Gallant, K.M.; Warden, S.J.; Weaver, C.M.; Peacock, M.; et al. Vitamin D Supplementation Does Not Impact Insulin Resistance in Black and White Children. J. Clin. Endocrinol. Metab. 2016, 101, 1710–1718. [Google Scholar] [CrossRef]
- Tavakoli, F.; Namakin, K.; Zardast, M. Vitamin D supplementation and high-density lipoprotein cholesterol: A study in healthy school children. Iran. J. Pediatr. 2016, 26, e3311. [Google Scholar] [CrossRef]
- Brar, P.C.; Contreras, M.; Fan, X.; Visavachaipan, N. Effect of one time high dose “stoss therapy” of vitamin d on glucose homeostasis in high risk obese adolescents. Arch. Endocrinol. Metab. 2018, 62, 193–200. [Google Scholar] [CrossRef]
- Shah, S.; Wilson, D.M.; Bachrach, L.K. Large doses of vitamin D fail to increase 25-hydroxyvitamin D levels or to alter cardiovascular risk factors in obese adolescents: A pilot study. J. Adolesc. Heal. 2015, 57, 19–23. [Google Scholar] [CrossRef]
- Javed, A.; Vella, A.; Balagopal, P.B.; Fischer, P.R.; Weaver, A.L.; Piccinini, F.; Dalla Man, C.; Cobelli, C.; Giesler, P.D.; Laugen, J.M.; et al. Cholecalciferol Supplementation Does Not Influence β-Cell Function and Insulin Action in Obese Adolescents: A Prospective Double-Blind Randomized Trial. J. Nutr. 2015, 145, 284–290. [Google Scholar] [CrossRef]
- Magge, S.N.; Prasad, D.; Zemel, B.S.; Kelly, A. Vitamin D3 supplementation in obese, African-American, vitamin D deficient adolescents. J. Clin. Transl. Endocrinol. 2018, 12, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Sethuraman, U.; Zidan, M.A.; Hanks, L.; Bagheri, M.; Ashraf, A. Impact of vitamin D treatment on 25 hydroxy vitamin D levels and insulin homeostasis in obese African American adolescents in a randomized trial. J. Clin. Transl. Endocrinol. 2018, 12, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Varshney, S.; Khadgawat, R.; Gahlot, M.; Khandelwal, D.; Oberoi, A.; Yadav, R.; Sreenivas, V.; Gupta, N.; Tandon, N. Effect of high-dose Vitamin D supplementation on beta cell function in obese Asian-Indian children and adolescents: A randomized, double blind, active controlled study. Indian J. Endocrinol. Metab. 2019, 23, 545–551. [Google Scholar] [CrossRef] [PubMed]
| Study | Participants | Vitamin D Deficiency Cut-Off | 25OHD Mean Levels | Endpoint | Results |
|---|---|---|---|---|---|
| Colak R et al. 2020 [49] | 40 obese children (7–14 years) 30 controls (7–14 years) | Deficiency: ≤ 20 ng/mL 25OHD levels | Obese children 16.4 ng/mL Lean children 19.6 ng/mL | Vitamin D levels and lipid profile, fasting glucose and blood pressure Vitamin D levels and ultrasound imaging (cIMT and left ventricular wall thickness) | 25OHD concentrations were negatively correlated with 24-h ambulatory blood pressure and cIMT (p <0.05) No associations between 25OHD values and fasting plasma glucose, HOMA-IR or lipid profile were present |
| Censani M et al. 2018 [50] | 178 overweight and obese children (6–17 years) | Deficiency: ≤ 20 ng/mL 25OHD levels | 20.7 ± 9.2 ng/mL | Vitamin D levels and lipid profile | Patients with 25OHD < 20 ng/mL had significantly higher non-HDL cholesterol, TC, TG and LDL levels (p ≤ 0.03) and a significantly higher TG/HDL and TC/HDL ratios (p ≤ 0.03) |
| Iqbal AM et al. 2017 [51] | 376 obese children (2–18 years) | Deficiency: ≤ 20 ng/mL 25OHD levels Sufficiency: > 30 ng/mL 25OHD levels | 25.2 ± 10.10 ng/mL | Vitamin D levels and lipid profile | 25OHD values were negatively associated with BMI z-score (p = 0.004) and were positively correlated with c-HDL also after adjustment for age, sex, BMI metric and season of blood draw |
| Lee M et al. 2016 [52] | 209 overweight or obese children (6–18 years) | Deficiency: ≤ 20 ng/mL 25OHD levels | 20.3 ± 6.4 ng/mL | Vitamin D levels and lipid profile | A 10 mg/dl increase in TC and oxidated-LDL was associated respectively with a 1,3% and 0,8% decrease in 25OHD concentrations |
| Petersen R et al. 2015 [55] | 782 children (8–11 years) | Deficiency: ≤ 25 nmol/L 25OHD levels | 60.8 ± 18.7 nmol/L | Vitamin D levels and glucose concentration, lipid profile, insulin, blood pressure and heart rate weighted for fat mass index | Serum 25OHD was negatively associated with diastolic blood pressure, total and c-LDL, TG and lower metabolic syndrome score, also after adjustment for fat mass index |
| Kim MR et al. 2019 [56] | 243 non-obese healthy volunteers (9–18 years) | Deficiency: ≤ 20 ng/mL 25OHD levels | 17.27 ± 6.89 ng/mL | Vitamin D levels and lipid profile | Vitamin D levels significantly inversely associated with TG level and TG/c-HDL ratio (p < 0.001) |
| Liang X et al. 2018 [57] | 164 children (6–12 years) Hypertensive vs control subjects | Deficiency: <50 ng/mL 25OHD levels | 38.22 ± 12 nmol/L in hypertension group 43.28 ± 12.3 nmol/L in control group | Vitamin D levels and blood pressure, the transcription level of RARs and RXRs, 25OHD receptor | Serum 25OHD in children with hypertension was lower than that in the control group (p = 0.02). Serum 25OHD and 25OHD receptor were significantly associated with blood pressure level, and both breastfed and c-HDL were independent protective factors of blood pressure level |
| Arman D et al. 2019 [59] | 135 term healthy neonates | Deficiency: ≤ 20 ng/mL 25OHD levels Sufficiency: > 30 ng/mL 25OHD levels | 15.17 ± 9.66 ng/mL | Vitamin D levels and aIMT and cIMT | Neonates with vitamin D sufficiency had a lower aIMT than the others (p = 0.001) |
| Sauder KA et al. 2019 [60] | 1410 birth cohort of ethnically diverse pregnant woman and their offspring 715 children evaluated at 4 to 6 years old | Childhood: 55.8± 21.1 nmol/L | Vitamin D levels and CV risk factors (blood pressure, arterial stiffness, body size, and adiposity) | Higher vitamin D levels in cord blood are associated with lower systolic and diastolic blood pressure at 4 to 6 years of age, regardless of childhood 25OHD levels, race/ethnicity, and other covariates | |
| Wang G et al. 2019 [63] | Birth cohort study of 775 children, followed prospectively up to 18 years | Deficiency: < 11 ng/mL on cord blood and < 25 ng/mL in early childhood 25OHD levels | Birth: 13.5 ± 9.9 ng/mL Childhood: 32.6 ± 10.8 ng/mL | Vitamin D levels and blood pressure | Low vitamin D status at birth was associated with higher risk of elevated SBP at ages 3 to 18 years. Low vitamin D status in early childhood was associated with a 1,59-fold higher risk of elevated SBP at age 6 to 18 years |
| Miliku K et al. 2018 [64] | 4903 mother-children pairs re-evaluated at 6 years | Deficiency: <50 ng/mL 25OHD levels | Birth: 28.8 ± 9.9 nmol/L; Childhood: 64 ± 10.8 nmol/L | Vitamin D levels and blood pressure, lipid profile, BMI | 25OHD concentrations were not associated with cardiovascular risk factors |
| Study | Where and Season | Participants | Baseline Mean 25OHD Levels | Treatment | Control Group | Duration | Endpoints | Results |
|---|---|---|---|---|---|---|---|---|
| Hauger et al. (2018) [75] | Denmark Winter | 130 Normal weight children 4–8 years White | 10µg/d: 56.9 ± 12.7 nmol/L 20µg/d: 58.1 ± 13.5 nmol/L Control group: 55.2 ± 10.8 nmol/L | D3 10 or 20 µg/day | Placebo | 20 weeks | 25OHD levels BMI, SBP, DBP, lipid profile, glucose, insulin, HbA1c | 25OHD increased to 61.8 ± 10.6 nmol/L in the 10 µg/d group, to 75.8 ± 11.5 nmol/L in the 20 µg/d group No effect on any of the cardiometabolic risk markers Marginal dose-response effect on triglycerides, which increased by 0.03 nmol/L per 10 nmol/L increase in 25OHD (p = 0.07) |
| Smith et al. (2018) [76] | UK Winter | 110 Normal weight adolescents 14–18 years White | 10µg/d: 49.2 ± 12.0 nmol/L 20µg/d: 51.7 ± 13.4 nmol/L Control group: 46.8 ± 11.4 nmol/L | D3 10 or 20 µg/day | Placebo | 20 weeks | 25OHD levels BMI, waist circumference, SBP, DBP, glucose, lipid profile | Baseline serum 25OHD was inversely associated with BMIz (p < 0.001) and waist circumference (p = 0.002) 25OHD increased to 56.6 ± 12.4nmol/L in the 10µg/d group, to 63.9 ± 10.6nmol/L in the 20µg/d group No significant differences in cardiovascular risk factors within either group or between groups |
| Ferira et al. (2016) [77] | USA Winter | 323 Normal weight children and adolescents 9–13 years Mixed | Mean: 70.0 ± 1.0 nmol/L | D3 400,1000, 2000 or 4000 IU/day | Placebo | 12 weeks | Dose-response effects of vitamin D on fasting glucose, insulin and HOMA-IR | Baseline 25OHD was inversely associated with BMI (p = 0.003), insulin (p = 0.005) and HOMA-IR (p = 0.012) No significant difference in fasting glucose, insulin and HOMA-IR between groups over time after supplementation |
| Tavakoli et al. (2016) [78] | Iran Not known | 47 Normal weight children and adolescents 10–14 years Caucasian (Iranian) | Treatment group: 7.55 ± 4.96 ng/mL Control group: 9.71 ± 5.48 ng/mL | D 1000IU/day | Placebo | 4 weeks | 25OHD levels c-HDL | 25OHD increased in the treatment group (11.50 ± 5.84ng/mL, p < 0.001) c-HDL significantly increased in the treatment group (+ 4.10 ± 6.10mg/dL, p = 0.007) |
| Brar et al. (2018) [79] | Not known All year | 20 Obese adolescents 12–18 years Mixed, 75% Hispanic | Mean levels: 16.7 ± 2.9 ng/mL | D2 300000 IU once | Placebo (crossover at week 6) | 6 weeks | 25OHD levels Insulin metabolism | 25OHD treatment group: 19.5 ± 4.5 ng/mL (p = 0.0029), control group: 17.2 ± 4.7 ng/mL (p 0.5262) WBISI showed a trend towards improvement in the treated group (p = 0.0577) |
| Shah et al. (2015) [80] | USA All year | 40 obese adolescents 11–17 years Mixed | Treatment group: 19.6± 1.4 ng/mL Control group: 25.8 ± 2.6 ng/mL | D2 150000IU baseline and at 12 weeks | Placebo | 24 weeks | 25OHD levels BMI, lipid profile, HbA1c | Baseline 25OHD was inversely associated with BMI No significant difference in 25OHD levels and no significant changes in any of the markers analysed after vitamin D2 supplementation |
| Javed et al. (2015) [81] | USA All year | 51 Obese adolescents 12–18 years Caucasian | Treatment group: 23.5 ± 8.5 ng/mL Control group: 24.4 ± 7.7 ng/mL | D3 2000 IU/day | 400 IU/day | 12 weeks | 25OHD levels Insulin metabolism Lipid profile | A significant increase in 25OHD in the 2000IU/d group (p = 0.04) No change in parameters of insulin metabolism or lipid profile |
| Magge et al. (2018) [82] | USA All year | 26 Obese adolescents 12–17 years African American | Treatment group: 12.3 ± 3.5 ng/mL Control group: 11.7 ± 4.1 ng/mL | D3 5000 IU/day | 1000 IU/day | 12 weeks | 25OHD levels BMI-z, HOMA-IR, lipid profile, hs-CRP | 25OHD treatment group: 28.8 ± 11.4 ng/mL (p < 0.0001), control group: 18.8 ± 3.9 ng/mL (p = 0.0006) No significant difference in cardiometabolic markers within either group or between groups following Vitamin D3 supplementation |
| Sethuruman et al. (2018) [83] | USA All year | 29 Obese adolescents 13–17 years African American | Treatment group: 12.1 ± 3.8 ng/mL Control group: 12.4 ± 3.8 ng/mL | D2 50000IU once per week + 500 mg/day calcium carbonate | Placebo once per week + 500mg/day calcium carbonate | 12 weeks | 25OHD levels Insulin metabolism Lipid profile | 25OHD treatment group: 32 ng/mL (p < 0.0001), control group: 13 ng/mL (p = 0.126) 25OHD was positively correlated with HDL (r = 0.6, p < 0.05) and fasting insulin (r = 0.5, p < 0.05), but not HOMA-IR (r = 0.5, p = 0.08) |
| Varshney et al. (2019) [84] | India All year | 202 Obese children and adolescents 11–17 years Asian Indian | Treatment group: 8.36 ± 5.45 ng/mL Control group: 9.01 ± 5.59 ng/mL | D 120,000 IU once a month | 12,000 IU once a month | 12 months | 25OHD levels Insulin metabolism Lipid profile, pulse wave velocity and augmentation index | 25OHD treatment group: 26.89 ± 12.23 ng/mL; control group 13.14 ± 4.67 ng/mL (p < 0.001) No changes in insulin metabolism or in cardiovascular risk factors within either group or between groups after supplementation |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Savastio, S.; Pozzi, E.; Tagliaferri, F.; Degrandi, R.; Cinquatti, R.; Rabbone, I.; Bona, G. Vitamin D and Cardiovascular Risk: Which Implications in Children? Int. J. Mol. Sci. 2020, 21, 3536. https://doi.org/10.3390/ijms21103536
Savastio S, Pozzi E, Tagliaferri F, Degrandi R, Cinquatti R, Rabbone I, Bona G. Vitamin D and Cardiovascular Risk: Which Implications in Children? International Journal of Molecular Sciences. 2020; 21(10):3536. https://doi.org/10.3390/ijms21103536
Chicago/Turabian StyleSavastio, Silvia, Erica Pozzi, Francesco Tagliaferri, Roberta Degrandi, Roberta Cinquatti, Ivana Rabbone, and Gianni Bona. 2020. "Vitamin D and Cardiovascular Risk: Which Implications in Children?" International Journal of Molecular Sciences 21, no. 10: 3536. https://doi.org/10.3390/ijms21103536
APA StyleSavastio, S., Pozzi, E., Tagliaferri, F., Degrandi, R., Cinquatti, R., Rabbone, I., & Bona, G. (2020). Vitamin D and Cardiovascular Risk: Which Implications in Children? International Journal of Molecular Sciences, 21(10), 3536. https://doi.org/10.3390/ijms21103536





