Heterobasidion annosum Induces Apoptosis in DLD-1 Cells and Decreases Colon Cancer Growth in In Vivo Model
Abstract
1. Introduction
2. Results and Discussion
2.1. Taxonomic Identification of M-0969 Strain Classifies to HA
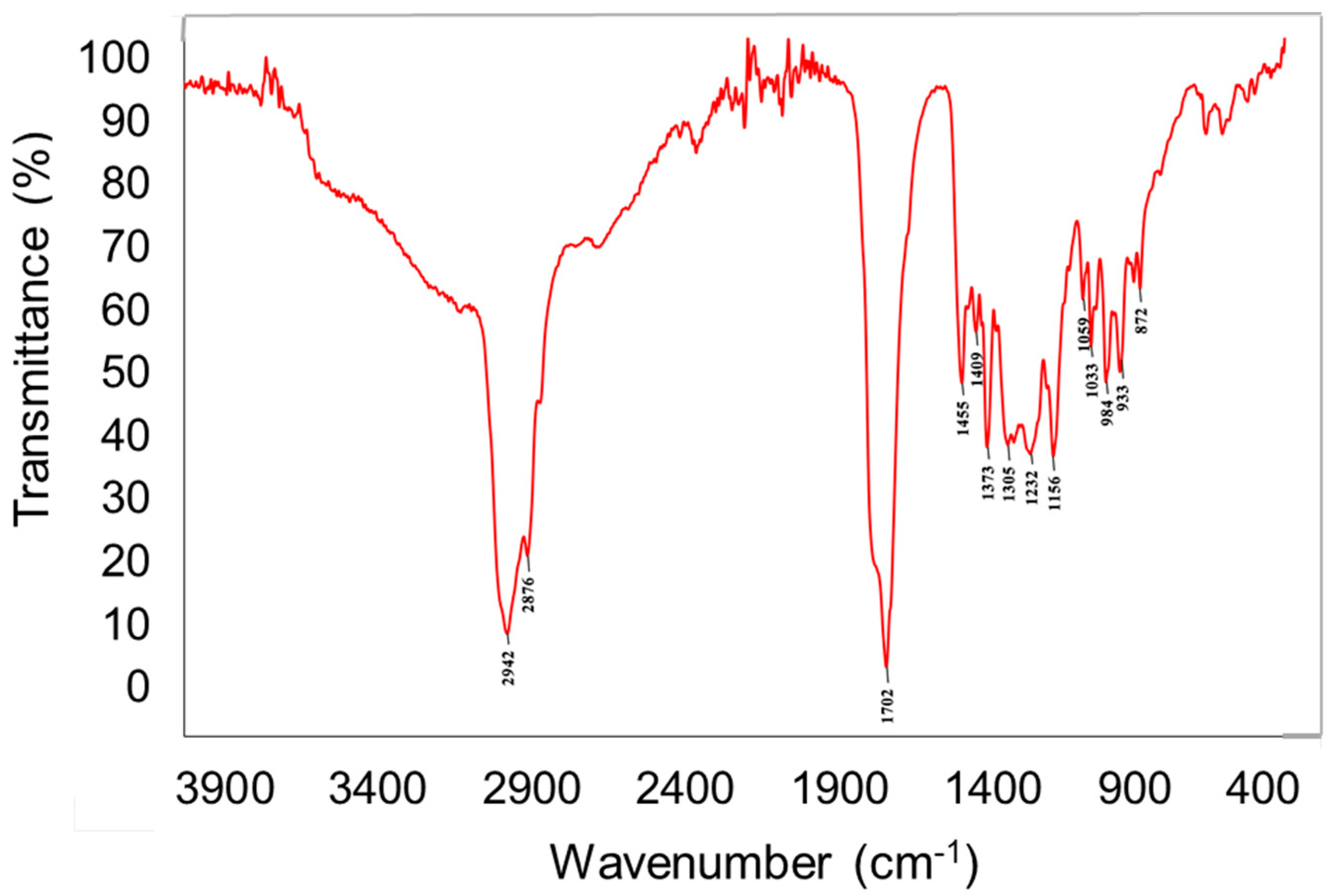
2.2. Chemical Characteristic of M-0969 Strain Determine Their Biological Properties
2.3. Hemocompatibility and Cytotoxic Activity of HA Extract at In Vitro and In Vivo Level
2.3.1. High Compatibility and Restriction of CRC Cell Viability after Exposure to HA Extract
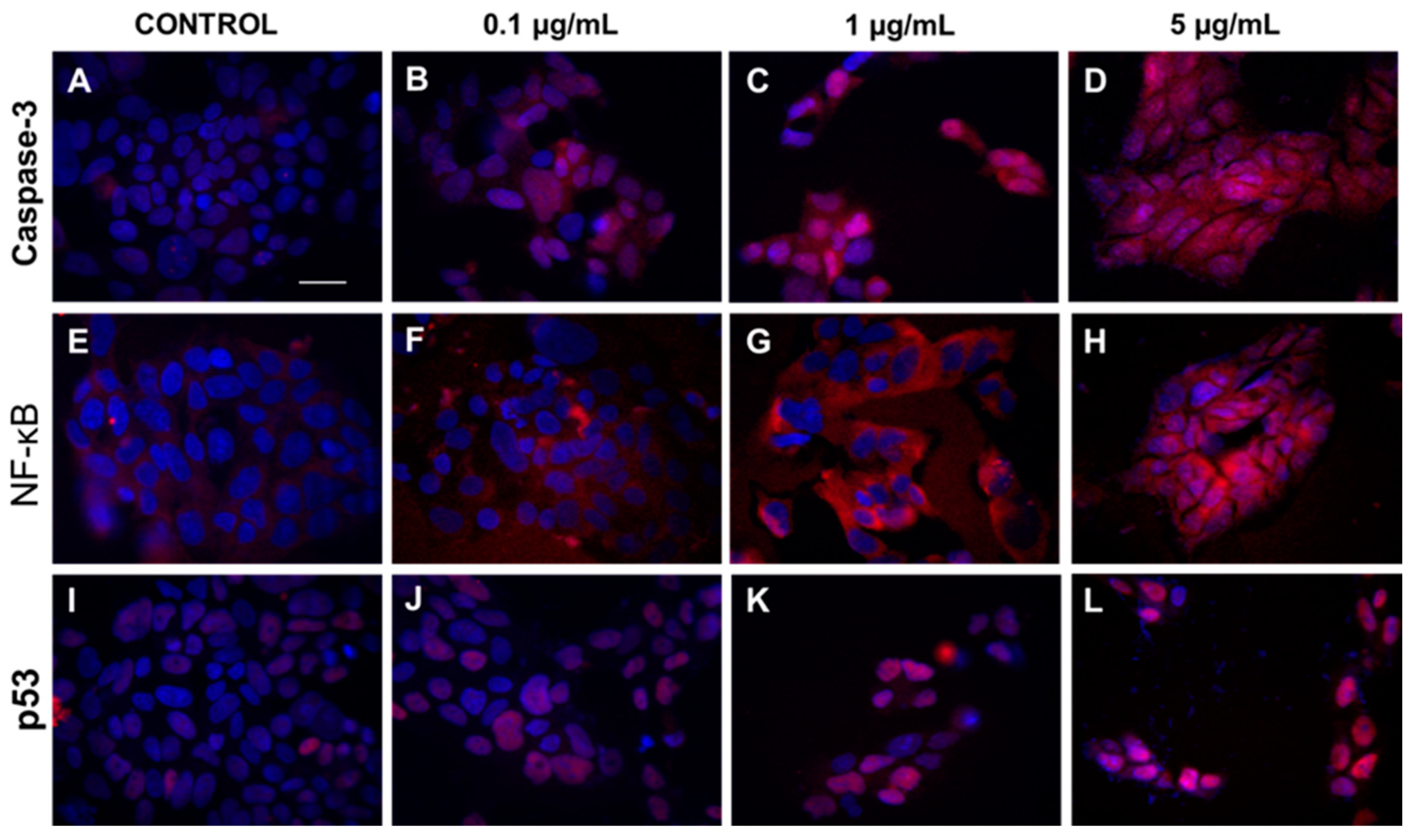
2.3.2. Exposition to HA Extract Induces Apoptosis via the Complex Mode of Action
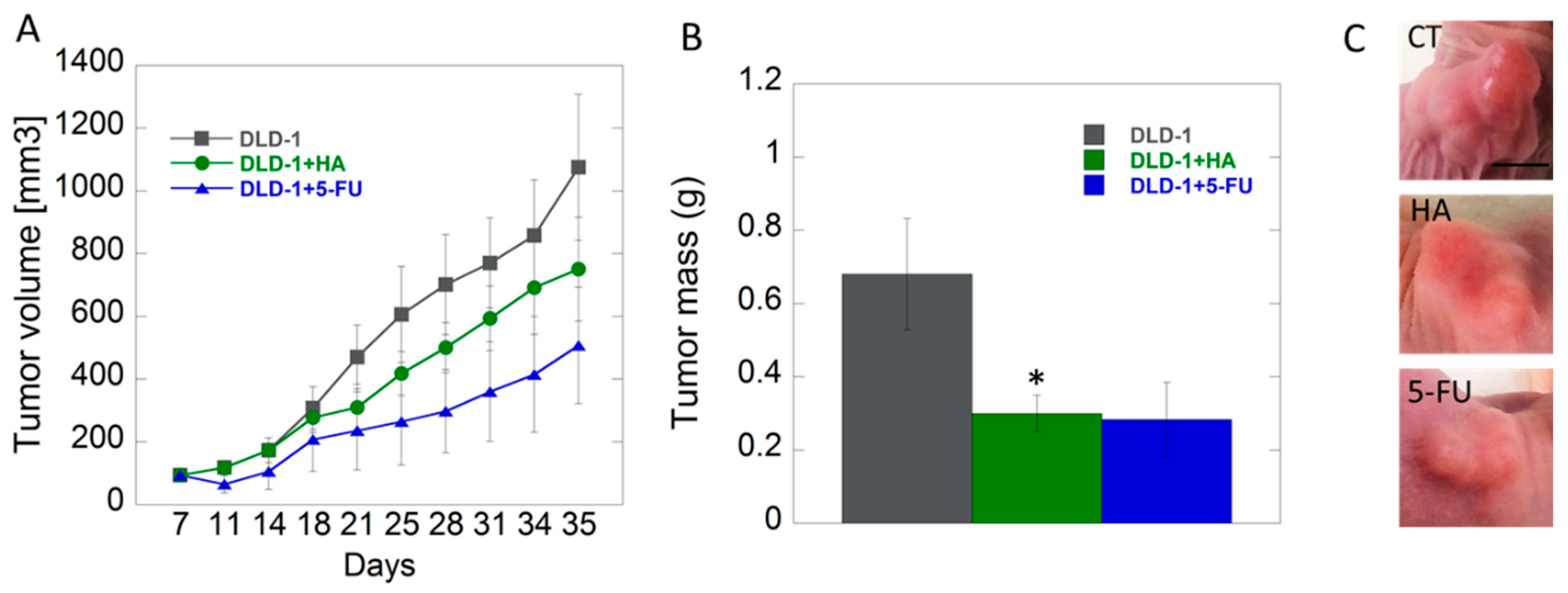
2.3.3. Effects of HA Extract in Xenograft CRC Model
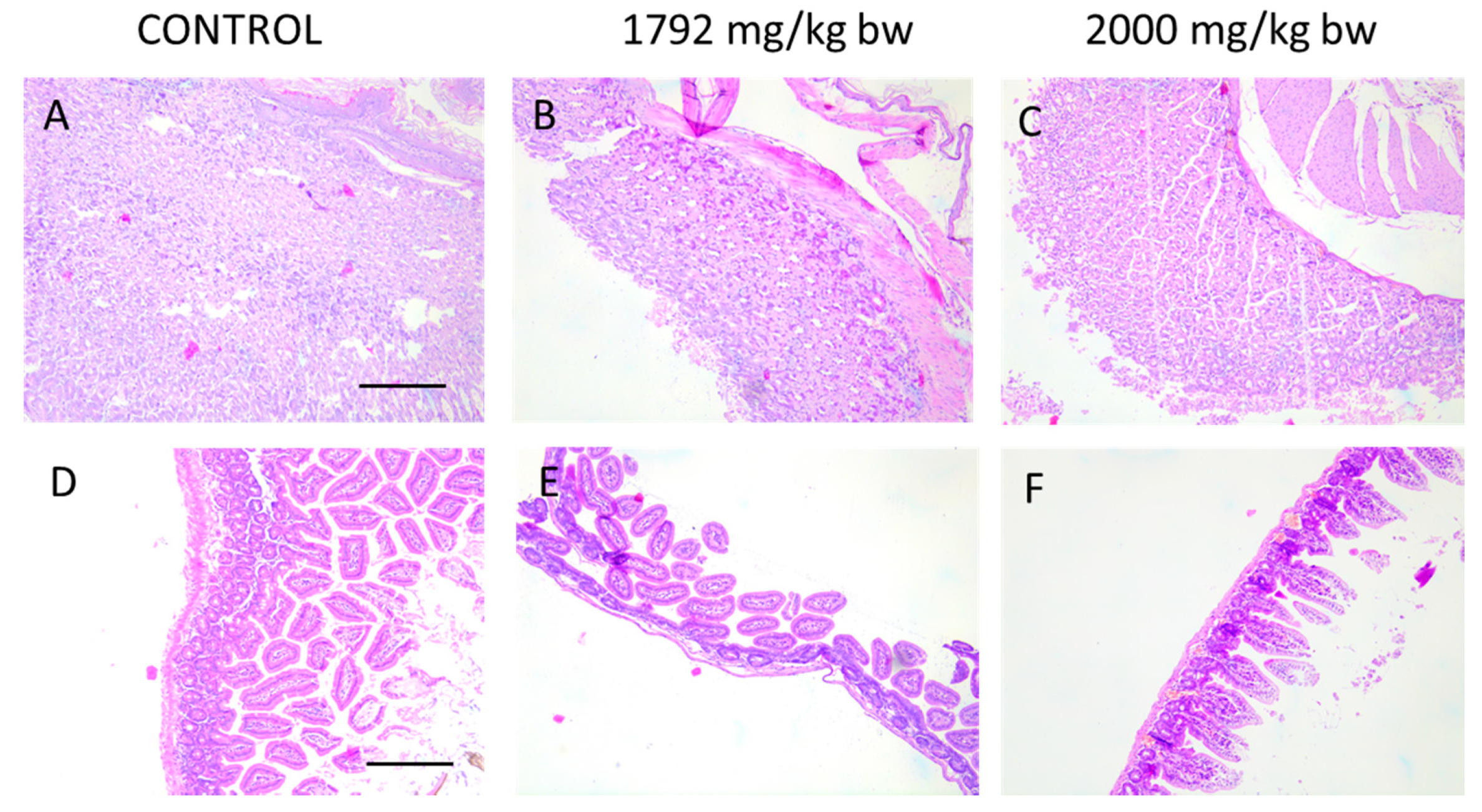
2.3.4. HA Extract Applied in Doses up to 2000-Fold Effective Dose Caused Mild or Moderate Acute Toxicity
3. Materials and Methods
3.1. Biological Material and Morphological Identification
3.2. PCR Amplification and Sequencing of the Fungal ITS Region
3.3. Preparation of HA Extract
3.4. Physicochemical Analysis of HA Extract
3.4.1. FTIR Spectroscopy Analysis.
3.4.2. Gas Chromatography–Mass Spectrometry Analysis
3.4.3. High-Performance Liquid Chromatography
(a) Analysis of Indole Compounds
(b) Analysis of Phenolic Compounds
(c) Extraction and Analysis of Sterol
3.4.4. F-AAS Analysis of Selected Bioelements
3.5. In Vitro Evaluation of HA Extract
3.5.1. Hemolytic Activity
3.5.2. Cell Culture
3.5.3. Cell Viability Assay
3.5.4. [3H]-Thymidine Incorporation (DNA Biosynthesis Assay)
3.5.5. Mitochondrial Potential Assay
3.5.6. Vitality Assay: Analysis of the Cellular Thiols (SH)
3.5.7. Cell Bioimaging
3.6. In Vivo Evaluation of HA Extract
3.6.1. Xenograft Studies in Nude Mice
3.6.2. Acute Oral Toxicity of HA Extract
3.6.3. Histopathological analysis
3.7. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CRC | Colorectal cancer |
| HA | Heterobasidion annosum |
| RBCs | human red blood cells |
| GSH | Glutathione |
| GC/MS | Gas chromatography–Mass spectrometry |
| HPLC | High-Performance Liquid Chromatography |
| F-AAS | Atomic Absorption Spectroscopy |
| FTIR | Fourier-Transform Infrared Spectroscopy |
| 5-FU | 5-fluorouracil |
References
- Korhonen, K.; Stenlid, J. Biology of Heterobasidion annosum. In Heterobasidion Annosum: Biology, Ecology, Impact and Control; CAB international: Wallinford, UK, 1998. [Google Scholar]
- Korhonen, K.; Capretti, P.; Karjalainen, R.; Stenlid, J. Distribution of Heterobasidion annosum intersterility groups in Europe. In Heterobasidion Annosum: Biology, Ecology, Impact and Control; CAB international: Wallinford, UK, 1998. [Google Scholar]
- Dai, Y.C.; Yuan, H.S.; Wei, Y.L.; Korhonen, K. New records of Heterobasidion parviporum in China. For. Pathol. 2006, 36, 287–293. [Google Scholar] [CrossRef]
- Dai, Y.C.; Vainio, E.J.; Hantula, J.; Niemela, T.; Korhonen, K. Investigations on Heterobasidion annosum s. lat. in central and eastern Asia with the aid of mating tests and DNA fingerprinting. For. Pathol. 2003, 33, 269–286. [Google Scholar] [CrossRef]
- Otrosina, W.J.; Garbelotto, M. Heterobasidion occidentale sp. nov. and Heterobasidion irregulare nom. nov.: A disposition of North American Heterobasidion biological species. Fungal Biol. 2010, 114, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Korhonen, K. Intersterility groups of Heterobasidion annosum. Commun. Inst. For. Fenn. 1978, 94, 23–30. [Google Scholar]
- Capretti, P.; Korhonen, K.; Mugnai, L.; Romagnoli, C. An Intersterility group of Heterobasidion annosum specialized to Abies alba. Eur. J. For. Pathol. 1990, 20, 231–240. [Google Scholar] [CrossRef]
- Niemelä, T.; Korhonen, K. Taxonomy of the genus Heterobasidion. In Heterobasidion Annosum: Biology, Ecology, Impact and Control; CAB internatinal: Wallinford, UK, 1998. [Google Scholar]
- Asiegbu, F.O.; Johansson, M.; Woodward, S.; Hüttermann, A. Biochemistry of the host–parasite interaction. In Heterobasidion annosum: Biology, Ecology, Impact Control; Woodward, S., Stenlid, J., Karjalainen, R., Hüttermann, A., Eds.; CAB International: Wallingford, UK, 1998; pp. 167–193. [Google Scholar]
- Asiegbu, F.O.; Abu, S.; Stenlid, J.; Johansson, M. Sequence polymorphism and molecular characterisation of laccase genes of theconifer pathogen Heterobasidion annosum. Mycol. Res. 2004, 108, 136–148. [Google Scholar] [CrossRef] [PubMed]
- Haars, A.; Chet, I.; Hütterman, A. Effect of phenolic compounds and tannin on growth and laccase activity of Fomes annosus. Eur. J. For. Pathol. 1981, 11, 67–76. [Google Scholar] [CrossRef]
- Daniel, G.; Asiegbu, F.; Johansson, M. The saprotrophic wood-degrading abilities of Heterobasidium annosum intersterility groups P and S. Mycol. Res. 1998, 102, 991–997. [Google Scholar] [CrossRef]
- Hansson, D.; Wubshet, S.; Olson, Å.; Karlsson, M.; Staerk, D.; Broberg, A. Secondary metabolite comparison of the species within the Heterobasidion annosum s.l. complex. Phytochemistry 2014, 108, 243–251. [Google Scholar] [CrossRef]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Moehler, M.; Göpfert, K.; Lenz, H.J. Outlook: Immunotherapy in Gastrointestinal Carcinoma—Innovative Strategies. Oncol Res. Treat. 2018, 41, 313–315. [Google Scholar] [CrossRef]
- Jo, W.S.; Hossain, M.A.; Park, S.C. Toxicological profiles of poisonous, edible, and medicinal mushrooms. Mycobiology 2014, 42, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Lindequist, U.; Niedermeyer, T.H.; Jülich, W.D. The pharmacological potential of mushrooms. Evid. Based Complement. Alternat. Med. 2005, 2, 285–299. [Google Scholar] [CrossRef] [PubMed]
- Grienke, U.; Zöll, M.; Peintner, U.; Rollinger, J.M. European medicinal polypores—A modern view on traditional uses. J. Ethnopharmacol. 2014, 154, 564–583. [Google Scholar] [CrossRef]
- Kladar, N.V.; Gavarić, N.S.; Božin, B.N. Ganoderma: Insights into anticancer effects. Eur. J. Cancer Prev. 2016, 25, 462–471. [Google Scholar] [CrossRef] [PubMed]
- Glamočlija, J.; Ćirić, A.; Nikolić, M.; Fernandes, Â.; Barros, L.; Calhelha, R.C.; Ferreira, I.C.; Soković, M.; van Griensven, L.J. Chemical characterization and biological activity of Chaga (Inonotus obliquus), a medicinal “mushroom”. J. Ethnopharmacol. 2015, 162, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.; Goyal, A. Recent developments in mushrooms as anti-cancer therapeutics: A review. 3 Biotech 2012, 2, 1–15. [Google Scholar] [CrossRef]
- Simmons, E.G. The International Mycological Association: Its history in brief with summaries of its International Mycological Congresses and diverse international relationships. IMA Fungus 2010, 1, 18–100. [Google Scholar] [CrossRef]
- Garbelotto, M.; Gonthier, P. Biology, epidemiology, and control of Heterobasidion species worldwide. Annu. Rev. Phytopathol. 2013, 51, 39–59. [Google Scholar] [CrossRef]
- Su, C.H.; Lai, M.N.; Lin, C.C.; Ng, L.T. Comparative characterization of physicochemical properties and bioactivities of polysaccharides from selected medicinal mushrooms. Appl. Microbiol. Biotechnol. 2016, 100, 4385–4393. [Google Scholar] [CrossRef]
- Chaturvedi, V.K.; Agarwal, S.; Gupta, K.K.; Ramteke, P.W.; Singh, M.P. Medicinal mushroom: Boon for therapeutic applications. 3 Biotech 2018, 8, 334. [Google Scholar] [CrossRef] [PubMed]
- Jiang, M.Y.; Feng, T.; Liu, J.K. N-containing compounds of macromycetes. Nat. Prod. Rep. 2011, 28, 783–808. [Google Scholar] [CrossRef] [PubMed]
- Keszthelyi, D.; Troost, F.J.; Masclee, A.A. Understanding the role of tryptophan and serotonin metabolism in gastrointestinal function. Neurogastroenterol. Motil. 2009, 21, 1239–1249. [Google Scholar] [CrossRef]
- Turner, E.H.; Loftis, J.M.; Blackwell, A.D. Serotonin a la carte: Supplementation with the serotonin precursor 5-hydroxytryptophan. Pharmacol. Ther. 2006, 109, 325–338. [Google Scholar] [CrossRef] [PubMed]
- Muszyńska, B.; Sułkowska-Ziaja, K.; Ekiert, H. Indole compounds in some culinary-medicinal higher basidiomycetes from Poland. Int. J. Med. Mushrooms 2011, 13, 449–454. [Google Scholar] [CrossRef] [PubMed]
- Muszyńska, B.; Sułkowska-Ziaja, K.; Ekiert, H. Indole compounds in fruiting bodies of some edible Basidiomycota species. Food Chem. 2011, 125, 1306–1308. [Google Scholar] [CrossRef]
- Sułkowska-Ziaja, K.; Maślanka, A.; Szewczyk, A.; Muszyńska, B. Physiologically Active Compounds in Four Species of Phellinus. Nat. Prod. Commun. 2017, 12, 363–366. [Google Scholar] [CrossRef]
- Muszyńska, B.; Sułkowska-Ziaja, K.; Ekiert, H. Indole compounds in fruiting bodies of some selected Macromycetes species and in their mycelia cultured in vitro. Pharmazie 2009, 64, 479–480. [Google Scholar]
- Ferreira, I.C.; Barros, L.; Abreu, R.M. Antioxidants in wild mushrooms. Curr. Med. Chem. 2009, 16, 1543–1560. [Google Scholar] [CrossRef]
- Pasinetti, G.M.; Singh, R.; Westfall, S.; Herman, F.; Faith, J.; Ho, L. The Role of the Gut Microbiota in the Metabolism of Polyphenols as Characterized by Gnotobiotic Mice. J. Alzheimers Dis. 2018, 63, 409–421. [Google Scholar] [CrossRef]
- Karaman, M.; Jovin, E.; Malbasa, R.; Matavuly, M.; Popović, M. Medicinal and edible lignicolous fungi as natural sources of antioxidative and antibacterial agents. Phytother. Res. 2010, 24, 1473–1481. [Google Scholar] [CrossRef]
- Ashidate, K.; Kawamura, M.; Mimura, D.; Tohda, H.; Miyazaki, S.; Teramoto, T.; Yamamoto, Y.; Hirata, Y. Gentisic acid, an aspirin metabolite, inhibits oxidation of low-density lipoprotein and the formation of cholesterol ester hydroperoxides in human plasma. Eur. J. Pharmacol. 2005, 513, 173–179. [Google Scholar] [CrossRef]
- Yokokawa, H. Fatty acid and sterol compositions in mushrooms of ten species of polyporaceae. Phytochemistry 1980, 19, 2615–2618. [Google Scholar] [CrossRef]
- Zhang, Y.; Mills, G.L.; Nair, M.G. Cyclooxygenase inhibitory and antioxidant compounds from the fruiting body of an edible mushroom, Agrocybe aegerita. Phytomedicine 2003, 10, 386–390. [Google Scholar] [CrossRef]
- Krzyczkowski, W.; Malinowska, E.; Suchocki, P.; Kleps, J.; Olejnik, M.; Herold, F. Isolation and quantitative determination of ergosterol peroxide in various edible mushroom species. Food Chem. 2009, 113, 351–355. [Google Scholar] [CrossRef]
- Kała, K.; Krakowska, A.; Gdula-Argasinska, J.; Opoka, W.; Muszyńska, B. Assessing the Bioavailability of Zinc and Indole Compounds from Mycelial Cultures of the Bay Mushroom Imleria badia (Agaricomycetes) Using In Vitro Models. Int. J. Med. Mushrooms 2019, 21, 343–352. [Google Scholar] [CrossRef]
- Muszyńska, B.; Kała, K.; Radović, J.; Sułkowska-Ziaja, K.; Krakowska, A.; Gdula-Argasińska, J.; Opoka, W.; Kudaković, T. Study of biological activity of Tricholoma equestre fruiting bodies and their safety for human. Eur. Food Res. Technol. 2018, 244, 2255–2264. [Google Scholar] [CrossRef]
- Lemieszek, M.; Rzeski, W. Anticancer properties of polysaccharides isolated from fungi of the Basidiomycetes class. Contemp. Oncol. 2012, 16, 285–289. [Google Scholar] [CrossRef]
- Li, Y.H.; Niu, Y.B.; Sun, Y.; Zhang, F.; Liu, C.X.; Fan, L.; Mei, Q.B. Role of phytochemicals in colorectal cancer prevention. World J. Gastroenterol. 2015, 21, 9262–9272. [Google Scholar] [CrossRef]
- Pandya, U.; Dhuldhaj, U.; Sahay, N.S. Bioactive mushroom polysaccharides as antitumor: An overview. Nat. Prod. Res. 2019, 33, 2668–2680. [Google Scholar] [CrossRef]
- Saljoughian, M. Adaptogenic or Medicinal Mushrooms. US Pharm. 2009, 34, 16–18. [Google Scholar]
- Chen, J.C.; Hsieh, Y.Y.; Lo, H.L.; Li, A.; Chou, C.J.; Yang, P.M. In Vitro and In Silico Mechanistic Insights into miR-21-5p-Mediated Topoisomerase Drug Resistance in Human Colorectal Cancer Cells. Biomolecules 2019, 9, 467. [Google Scholar] [CrossRef] [PubMed]
- Tomasi, S.; Lohézic-Le Dévéhat, F.; Sauleau, P.; Bézivin, C.; Boustie, J. Cytotoxic activity of methanol extracts from Basidiomycete mushrooms on murine cancer cell lines. Pharmazie 2004, 59, 290–293. [Google Scholar]
- Cordell, G.; Kinghorn, D.; Pezzuto, J. Separation, Structure Elucidation, and Bioassay of Cytotoxic Natural Products; CRC Press: Boca Raton, FL, USA, 1993. [Google Scholar]
- Swanton, C. Cell-cycle targeted therapies. Lancet Oncol. 2004, 5, 27–36. [Google Scholar] [CrossRef]
- Youn, M.J.; Kim, J.K.; Park, S.Y.; Kim, Y.; Kim, S.J.; Lee, J.S.; Chai, K.Y.; Kim, H.J.; Cui, M.X.; So, H.S.; et al. Chaga mushroom (Inonotus obliquus) induces G0/G1 arrest and apoptosis in human hepatoma HepG2 cells. World J. Gastroenterol. 2008, 14, 511–517. [Google Scholar] [CrossRef]
- Song, F.Q.; Liu, Y.; Kong, X.S.; Chang, W.; Song, G. Progress on understanding the anticancer mechanisms of medicinal mushroom: Inonotus obliquus. Asian Pac. J. Cancer Prev. 2013, 14, 1571–1578. [Google Scholar] [CrossRef]
- Ma, L.; Chen, H.; Dong, P.; Lu, X. Anti-inflammatory and anticancer activities of extracts and compounds from the mushroom Inonotus obliquus. Food Chem. 2013, 139, 503–508. [Google Scholar] [CrossRef]
- Lee, H.S.; Kim, E.J.; Kim, S.H. Ethanol extract of Innotus obliquus (Chaga mushroom) induces G1 cell cycle arrest in HT-29 human colon cancer cells. Nutr. Res. Pract. 2015, 9, 111–116. [Google Scholar] [CrossRef]
- Zhang, N.; Yin, Y.; Xu, S.J.; Chen, W.S. 5-Fluorouracil: Mechanisms of resistance and reversal strategies. Molecules 2008, 13, 1551–1569. [Google Scholar] [CrossRef]
- Li, G.; Yu, K.; Li, F.; Xu, K.; Li, J.; He, S.; Cao, S.; Tan, G. Anticancer potential of Hericium erinaceus extracts against human gastrointestinal cancers. J. Ethnopharmacol. 2014, 153, 521–530. [Google Scholar] [CrossRef]
- Kang, J.H.; Jang, J.E.; Mishra, S.K.; Lee, H.J.; Nho, C.W.; Shin, D.; Jin, M.; Kim, M.K.; Choi, C.; Oh, S.H. Ergosterol peroxide from Chaga mushroom (Inonotus obliquus) exhibits anti-cancer activity by down-regulation of the β-catenin pathway in colorectal cancer. J. Ethnopharmacol. 2015, 173, 303–312. [Google Scholar] [CrossRef]
- Fan, L.; Ding, S.; Ai, L.; Deng, K. Antitumor and immunomodulatory activity of water-soluble polysaccharide from Inonotus obliquus. Carbohydr. Polym. 2012, 90, 870–874. [Google Scholar] [CrossRef]
- Won, D.P.; Lee, J.S.; Kwon, D.S.; Lee, K.E.; Shin, W.C.; Hong, E.K. Immunostimulating activity by polysaccharides isolated from fruiting body of Inonotus obliquus. Mol. Cells. 2011, 31, 165–173. [Google Scholar] [CrossRef]
- Yang, L.; Wu, D.; Luo, K.; Wu, S.; Wu, P. Andrographolide enhances 5-fluorouracil-induced apoptosis via caspase-8-dependent mitochondrial pathway involving p53 participation in hepatocellular carcinoma (SMMC-7721) cells. Cancer Lett. 2009, 276, 180–188. [Google Scholar] [CrossRef]
- Wang, F.F.; Shi, C.; Yang, Y.; Fang, Y.; Sheng, L.; Li, N. Medicinal mushroom Phellinus igniarius induced cell apoptosis in gastric cancer SGC-7901 through a mitochondria-dependent pathway. Biomed. Pharmacother. 2018, 102, 18–25. [Google Scholar] [CrossRef]
- Opattova, A.; Horak, J.; Vodenkova, S.; Kostovcikova, K.; Cumova, A.; Macinga, P.; Galanova, N.; Rejhova, A.; Vodickova, L.; Kozics, K.; et al. Ganoderma Lucidum induces oxidative DNA damage and enhances the effect of 5-Fluorouracil in colorectal cancer in vitro and in vivo. Mutat. Res. 2019, 845, 403065. [Google Scholar] [CrossRef]
- Borges, M.; Azevedo, M.; Bonatelli, J.; Felipe, M.; Astolfi-Filho, S. A practical method for the preparation of total DNA from filamentous fungi. Fungal Genet. Rep. 1990, 37, 1–2. [Google Scholar] [CrossRef][Green Version]
- Altschul, S.F.; Madden, T.L.; Schäffer, A.A.; Zhang, J.; Zhang, Z.; Miller, W.; Lipman, D.J. Gapped BLAST and PSI-BLAST: A new generation of protein database search programs. Nucleic Acids Res. 1997, 25, 3389–3402. [Google Scholar] [CrossRef]
- Muszyńska, B.; Łojewski, M.; Sułkowska-Ziaja, K.; Szewczyk, A.; Gdula-Argasińska, J.; Hałaszuk, P. In vitro cultures of Bacopa monnieri and an analysis of selected groups of biologically active metabolites in their biomass. Pharm. Biol. 2016, 54, 2443–2453. [Google Scholar] [CrossRef]
- Yuan, J.P.; Kuang, H.C.; Wang, J.H.; Liu, X. Evaluation of ergosterol and its esters in the pileus, gill, and stipe tissues of agaric fungi and their relative changes in the comminuted fungal tissues. Appl. Microbiol. Biotechnol. 2008, 80, 459–465. [Google Scholar] [CrossRef]
- Evans, B.C.; Nelson, C.E.; Yu, S.S.; Beavers, K.R.; Kim, A.J.; Li, H.; Nelson, H.M.; Giorgio, T.D.; Duvall, C.L. Ex vivo red blood cell hemolysis assay for the evaluation of pH-responsive endosomolytic agents for cytosolic delivery of biomacromolecular drugs. J. Vis. Exp. 2013, 73, e50166. [Google Scholar] [CrossRef]
- Ates, G.; Vanhaecke, T.; Rogiers, V.; Rodrigues, R.M. Assaying Cellular Viability Using the Neutral Red Uptake Assay. Methods Mol. Biol. 2017, 1601, 19–26. [Google Scholar]
- Carmichael, J.; DeGraff, W.G.; Gazdar, A.F.; Minna, J.D.; Mitchell, J.B. Evaluation of a tetrazolium-based semiautomated colorimetric assay: Assessment of chemosensitivity testing. Cancer Res. 1987, 47, 936–942. [Google Scholar]
- Kumar, P.; Nagarajan, A.; Uchil, P.D. Analysis of Cell Viability by the Lactate Dehydrogenase Assay. Cold Spring Harb. Protoc. 2018, 1, 465–468. [Google Scholar] [CrossRef]

| Groups of Compounds | % of TIC |
| Total carbohydrates | 6.82 |
| Total sterols | 1.82 |
| Total carboxylic acid | 1.74 |
| Indole compounds | [mg/100 g d.w.] ± SD |
| 5-Hydroxy-L-tryptophan | 39.1 ± 1.4 |
| L-Tryptophan | 34.9 ± 2.4 |
| 6-Methyl-D,L-tryptophan | 1.1 ± 0.2 |
| Melatonin | * |
| Phenolic acids | [mg/100 g d.w.] ± SD |
| Protocatechuic acid | 2.2 ± 0,05 |
| Gentisic acid | 76.5 ± 0.7 |
| Sterol compounds | [mg/100 g d.w.]± SD |
| Ergosterol | 9.5 ± 0.07 |
| Ergosterol peroxide | 23.8 ± 0.3 |
| Bioelements | [mg/100 g d.w.] ± SD |
| Cu | 1.0 ± 0.06 |
| Fe | 14.2 ± 1.7 |
| Zn | 4.2 ± 0.3 |
| Mg | 186.6 ± 4.5 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sadowska, A.; Zapora, E.; Sawicka, D.; Niemirowicz-Laskowska, K.; Surażyński, A.; Sułkowska-Ziaja, K.; Kała, K.; Stocki, M.; Wołkowycki, M.; Bakier, S.; et al. Heterobasidion annosum Induces Apoptosis in DLD-1 Cells and Decreases Colon Cancer Growth in In Vivo Model. Int. J. Mol. Sci. 2020, 21, 3447. https://doi.org/10.3390/ijms21103447
Sadowska A, Zapora E, Sawicka D, Niemirowicz-Laskowska K, Surażyński A, Sułkowska-Ziaja K, Kała K, Stocki M, Wołkowycki M, Bakier S, et al. Heterobasidion annosum Induces Apoptosis in DLD-1 Cells and Decreases Colon Cancer Growth in In Vivo Model. International Journal of Molecular Sciences. 2020; 21(10):3447. https://doi.org/10.3390/ijms21103447
Chicago/Turabian StyleSadowska, Anna, Ewa Zapora, Diana Sawicka, Katarzyna Niemirowicz-Laskowska, Arkadiusz Surażyński, Katarzyna Sułkowska-Ziaja, Katarzyna Kała, Marcin Stocki, Marek Wołkowycki, Sławomir Bakier, and et al. 2020. "Heterobasidion annosum Induces Apoptosis in DLD-1 Cells and Decreases Colon Cancer Growth in In Vivo Model" International Journal of Molecular Sciences 21, no. 10: 3447. https://doi.org/10.3390/ijms21103447
APA StyleSadowska, A., Zapora, E., Sawicka, D., Niemirowicz-Laskowska, K., Surażyński, A., Sułkowska-Ziaja, K., Kała, K., Stocki, M., Wołkowycki, M., Bakier, S., Pawlik, A., Jaszek, M., Muszyńska, B., & Car, H. (2020). Heterobasidion annosum Induces Apoptosis in DLD-1 Cells and Decreases Colon Cancer Growth in In Vivo Model. International Journal of Molecular Sciences, 21(10), 3447. https://doi.org/10.3390/ijms21103447







