The Impact of Glycosylation of Osteopontin on Urinary Stone Formation
Abstract
1. Introduction
2. Results
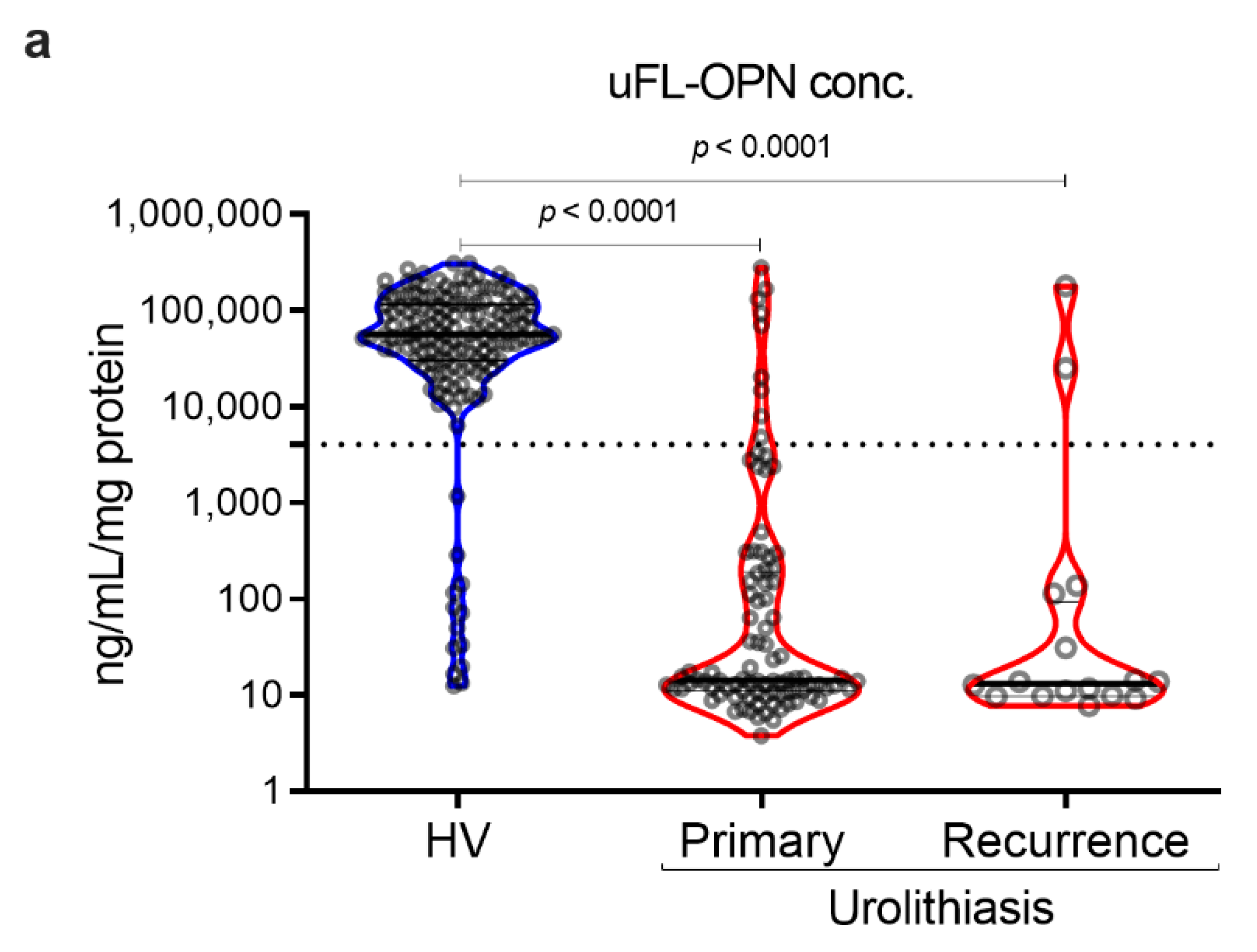
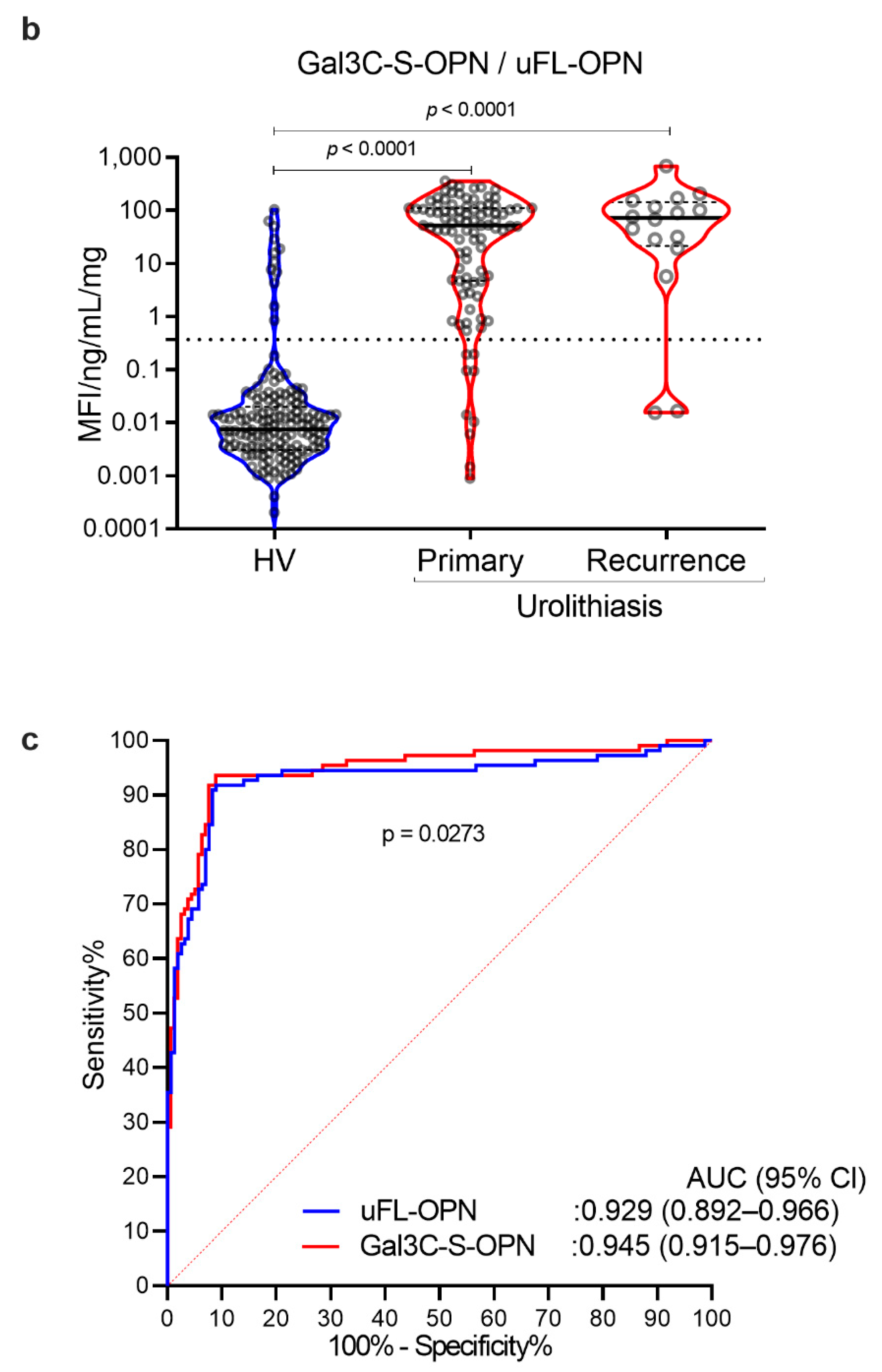
2.1. Identification of Urolithiasis-Related Aberrant Glycosylation Profile of Urine OPN in Retrospective Cohort Study
2.2. Immunoblotting and Lycopersicon Esculentum Lectin (LEL) Blotting Analysis
2.3. Longitudinal Follow-Up of the Urinary Gal3C-S-OPN and uFL-OPN Level in Urolithiasis Patients during Stone Treatment
3. Discussion
4. Materials and Methods
4.1. Study Design and Assessments
4.2. Quantification of uFL-OPN Concentration
4.3. Detection of Lectin-Reactive Glycosylated OPN in Urine by Using a Recombinant Lectin Array Chip
4.4. Immunoprecipitation and Immunoblotting and Lycopersicon Esculentum Lectin (LEL) Blotting Analysis
4.5. Purification of uFL-OPN from Pooled Urine of Urolithiasis Patients or Healthy Volunteers
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| OPN | Osteopontin |
| uFL-OPN | Urinary full-length osteopontin |
| URS | Ureteroscopic lithotripsy |
| MFI | Mean fluorescence intensity |
| HVs | Healthy volunteers |
| Gal3C-S-OPN | Gal3C-S reactive-osteopontin |
| polyLacNAcylated | Poly-N-acetyllactosamine glycosylated |
| IQR | Interquartile range |
| AUC | Area under the curve |
| LEL | Lycopersicon esculentum lectin |
| BSA | Bovine serum albumin |
| PBST | Phosphate-buffered saline |
| HRP | Horeseradish peroxidase |
| TBST | Tris-buffered saline with Tween20 |
| SDS | Sodium dodecyl sulfate |
| PAGE | Polyacrylamide gel electrophoresis |
| ROC | Receiver operating characteristics curve |
References
- Hesse, A.; Brandle, E.; Wilbert, D.; Kohrmann, K.U.; Alken, P. Study on the prevalence and incidence of urolithiasis in Germany comparing the years 1979 vs. 2000. Eur. Urol. 2003, 44, 709–713. [Google Scholar] [CrossRef]
- Romero, V.; Akpinar, H.; Assimos, D.G. Kidney stones: A global picture of prevalence, incidence, and associated risk factors. Rev. Urol. 2010, 12, e86–e96. [Google Scholar]
- Strohmaier, W.L. Course of calcium stone disease without treatment. What can we expect? Eur. Urol. 2000, 37, 339–344. [Google Scholar] [CrossRef]
- Uribarri, J.; Oh, M.S.; Carroll, H.J. The first kidney stone. Ann. Intern. Med. 1989, 111, 1006–1009. [Google Scholar] [CrossRef] [PubMed]
- Kohri, K.; Nomura, S.; Kitamura, Y.; Nagata, T.; Yoshioka, K.; Iguchi, M.; Yamate, T.; Umekawa, T.; Suzuki, Y.; Sinohara, H.; et al. Structure and expression of the mRNA encoding urinary stone protein (osteopontin). J. Biol. Chem. 1993, 268, 15180–15184. [Google Scholar] [PubMed]
- Okada, A.; Nomura, S.; Saeki, Y.; Higashibata, Y.; Hamamoto, S.; Hirose, M.; Itoh, Y.; Yasui, T.; Tozawa, K.; Kohri, K. Morphological conversion of calcium oxalate crystals into stones is regulated by osteopontin in mouse kidney. J. Bone Miner. Res. 2008, 23, 1629–1637. [Google Scholar] [CrossRef] [PubMed]
- Sodek, J.; Ganss, B.; McKee, M.D. Osteopontin. Crit. Rev. Oral Biol. Med. 2000, 11, 279–303. [Google Scholar] [CrossRef]
- Kazanecki, C.C.; Uzwiak, D.J.; Denhardt, D.T. Control of osteopontin signaling and function by post-translational phosphorylation and protein folding. J. Cell Biochem. 2007, 102, 912–924. [Google Scholar] [CrossRef]
- Kleinman, J.G.; Beshensky, A.; Worcester, E.M.; Brown, D. Expression of osteopontin, a urinary inhibitor of stone mineral crystal growth, in rat kidney. Kidney Int. 1995, 47, 1585–1596. [Google Scholar] [CrossRef] [PubMed]
- Wesson, J.A.; Johnson, R.J.; Mazzali, M.; Beshensky, A.M.; Stietz, S.; Giachelli, C.; Liaw, L.; Alpers, C.E.; Couser, W.G.; Kleinman, J.G.; et al. Osteopontin is a critical inhibitor of calcium oxalate crystal formation and retention in renal tubules. J. Am. Soc. Nephrol. 2003, 14, 139–147. [Google Scholar] [CrossRef]
- Liu, C.C.; Huang, S.P.; Tsai, L.Y.; Wu, W.J.; Juo, S.H.; Chou, Y.H.; Huang, C.H.; Wu, M.T. The impact of osteopontin promoter polymorphisms on the risk of calcium urolithiasis. Clin. Chim. Acta 2010, 411, 739–743. [Google Scholar] [CrossRef]
- Yasui, T.; Fujita, K.; Hayashi, Y.; Ueda, K.; Kon, S.; Maeda, M.; Uede, T.; Kohri, K. Quantification of osteopontin in the urine of healthy and stone-forming men. Urol. Res. 1999, 27, 225–230. [Google Scholar] [CrossRef]
- Hirose, M.; Tozawa, K.; Okada, A.; Hamamoto, S.; Higashibata, Y.; Gao, B.; Hayashi, Y.; Shimizu, H.; Kubota, Y.; Yasui, T.; et al. Role of osteopontin in early phase of renal crystal formation: Immunohistochemical and microstructural comparisons with osteopontin knock-out mice. Urol. Res. 2012, 40, 121–129. [Google Scholar] [CrossRef]
- Bautista, D.S.; Denstedt, J.; Chambers, A.F.; Harris, J.F. Low-molecular-weight variants of osteopontin generated by serine proteinases in urine of patients with kidney stones. J. Cell Biochem. 1996, 61, 402–409. [Google Scholar] [CrossRef]
- Christensen, B.; Klaning, E.; Nielsen, M.S.; Andersen, M.H.; Sorensen, E.S. C-terminal modification of osteopontin inhibits interaction with the αVβ3-integrin. J. Biol. Chem. 2012, 287, 3788–3797. [Google Scholar] [CrossRef]
- Tsuji, H.; Tohru, U.; Hirotsugu, U.; Masanori, I.; Yuji, H.; Takashi, K. Urinary concentration of osteopontin and association with urinary supersaturation and crystal formation. Int. J. Urol. 2007, 14, 630–634. [Google Scholar] [CrossRef]
- Icer, M.A.; Gezmen-Karadag, M.; Sozen, S. Can urine osteopontin levels, which may be correlated with nutrition intake and body composition, be used as a new biomarker in the diagnosis of nephrolithiasis? Clin. Biochem. 2018, 60, 38–43. [Google Scholar] [CrossRef]
- Icer, M.A.; Gezmen-Karadag, M. The multiple functions and mechanisms of osteopontin. Clin. Biochem. 2018, 59, 17–24. [Google Scholar] [CrossRef]
- Chagan-Yasutan, H.; Lacuesta, T.L.; Ndhlovu, L.C.; Oguma, S.; Leano, P.S.; Telan, E.F.; Kubo, T.; Morita, K.; Uede, T.; Dimaano, E.M.; et al. Elevated levels of full-length and thrombin-cleaved osteopontin during acute dengue virus infection are associated with coagulation abnormalities. Thromb. Res. 2014, 134, 449–454. [Google Scholar] [CrossRef]
- Bornstein, P. Matricellular proteins: An overview. J. Cell Commun. Signal. 2009, 3, 163–165. [Google Scholar] [CrossRef]
- Breyne, J.; Juthier, F.; Corseaux, D.; Marechaux, S.; Zawadzki, C.; Jeanpierre, E.; Ung, A.; Ennezat, P.V.; Susen, S.; Van Belle, E.; et al. Atherosclerotic-like process in aortic stenosis: Activation of the tissue factor-thrombin pathway and potential role through osteopontin alteration. Atherosclerosis 2010, 213, 369–376. [Google Scholar] [CrossRef]
- Kitagori, K.; Yoshifuji, H.; Oku, T.; Sasaki, C.; Miyata, H.; Mori, K.P.; Nakajima, T.; Ohmura, K.; Kawabata, D.; Yukawa, N.; et al. Cleaved form of osteopontin in urine as a clinical marker of lupus nephritis. PLoS ONE 2016, 11, e0167141. [Google Scholar] [CrossRef]
- Tateno, H.; Mori, A.; Uchiyama, N.; Yabe, R.; Iwaki, J.; Shikanai, T.; Angata, T.; Narimatsu, H.; Hirabayashi, J. Glycoconjugate microarray based on an evanescent-field fluorescence-assisted detection principle for investigation of glycan-binding proteins. Glycobiology 2008, 18, 789–798. [Google Scholar] [CrossRef]
- Hirabayashi, J.; Hashidate, T.; Arata, Y.; Nishi, N.; Nakamura, T.; Hirashima, M.; Urashima, T.; Oka, T.; Futai, M.; Muller, W.E.; et al. Oligosaccharide specificity of galectins: A search by frontal affinity chromatography. Biochim. Biophys. Acta Gen. Subj. 2002, 1572, 232–254. [Google Scholar] [CrossRef]
- Stowell, S.R.; Arthur, C.M.; Mehta, P.; Slanina, K.A.; Blixt, O.; Leffler, H.; Smith, D.F.; Cummings, R.D. Galectin-1, -2, and -3 exhibit differential recognition of sialylated glycans and blood group antigens. J. Biol. Chem. 2008, 283, 10109–10123. [Google Scholar] [CrossRef]
- Itakura, Y.; Nakamura-Tsuruta, S.; Kominami, J.; Tateno, H.; Hirabayashi, J. Sugar-binding profiles of chitin-binding lectins from the hevein family: A comprehensive study. Int. J. Mol. Sci. 2017, 18, 1160. [Google Scholar] [CrossRef]
- Kariya, Y.; Kanno, M.; Matsumoto-Morita, K.; Konno, M.; Yamaguchi, Y.; Hashimoto, Y. Osteopontin O-glycosylation contributes to its phosphorylation and cell-adhesion properties. Biochem. J. 2014, 463, 93–102. [Google Scholar] [CrossRef]
- Kato, K.; Jeanneau, C.; Tarp, M.A.; Benet-Pages, A.; Lorenz-Depiereux, B.; Bennett, E.P.; Mandel, U.; Strom, T.M.; Clausen, H. Polypeptide GalNAc-transferase T3 and familial tumoral calcinosis. Secretion of fibroblast growth factor 23 requires O-glycosylation. J. Biol. Chem. 2006, 281, 18370–18377. [Google Scholar] [CrossRef]
- Schjoldager, K.T.; Vester-Christensen, M.B.; Bennett, E.P.; Levery, S.B.; Schwientek, T.; Yin, W.; Blixt, O.; Clausen, H. O-glycosylation modulates proprotein convertase activation of angiopoietin-like protein 3: Possible role of polypeptide GalNAc-transferase-2 in regulation of concentrations of plasma lipids. J. Biol. Chem. 2010, 285, 36293–36303. [Google Scholar] [CrossRef]
- Rolain, T.; Bernard, E.; Beaussart, A.; Degand, H.; Courtin, P.; Egge-Jacobsen, W.; Bron, P.A.; Morsomme, P.; Kleerebezem, M.; Chapot-Chartier, M.P.; et al. O-glycosylation as a novel control mechanism of peptidoglycan hydrolase activity. J. Biol. Chem. 2013, 288, 22233–22247. [Google Scholar] [CrossRef]
- Gerken, T.A.; Butenhof, K.J.; Shogren, R. Effects of glycosylation on the conformation and dynamics of O-linked glycoproteins: Carbon-13 NMR studies of ovine submaxillary mucin. Biochemistry 1989, 28, 5536–5543. [Google Scholar] [CrossRef]
- Dwek, R.A. Glycobiology: “towards understanding the function of sugars”. Biochem. Soc. Trans. 1995, 23, 1–25. [Google Scholar] [CrossRef]
- Masuda, K.; Takahashi, N.; Tsukamoto, Y.; Honma, H.; Kohri, K. N-Glycan structures of an osteopontin from human bone. Biochem. Biophys. Res. Commun. 2000, 268, 814–817. [Google Scholar] [CrossRef]
- Trischler, M.; Koch-Brandt, C.; Ullrich, O. Apical transport of osteopontin is independent of N-glycosylation and sialylation. Mol. Membr. Biol. 2001, 18, 275–281. [Google Scholar] [CrossRef]
- Lee, N.; Wang, W.C.; Fukuda, M. Granulocytic differentiation of HL-60 cells is associated with increase of poly-N-acetyllactosamine in Asn-linked oligosaccharides attached to human lysosomal membrane glycoproteins. J. Biol. Chem. 1990, 265, 20476–20487. [Google Scholar]
- Renouf, D.V.; Hounsell, E.F. Conformational studies of the backbone (poly-N-acetyllactosamine) and the core region sequences of O-linked carbohydrate chains. Int. J. Biol. Macromol. 1993, 15, 37–42. [Google Scholar] [CrossRef]
- Wang, W.C.; Lee, N.; Aoki, D.; Fukuda, M.N.; Fukuda, M. The poly-N-acetyllactosamines attached to lysosomal membrane glycoproteins are increased by the prolonged association with the Golgi complex. J. Biol. Chem. 1991, 266, 23185–23190. [Google Scholar]
- Saitoh, O.; Wang, W.C.; Lotan, R.; Fukuda, M. Differential glycosylation and cell surface expression of lysosomal membrane glycoproteins in sublines of a human colon cancer exhibiting distinct metastatic potentials. J. Biol. Chem. 1992, 267, 5700–5711. [Google Scholar]



| Group | Urolithiasis | Healthy | p-Value 5 | |||
|---|---|---|---|---|---|---|
| Primary a (n = 94) | Recurrence b (n = 16) | Volunteers c (n = 157) | a vs. b | b vs. c | a vs. c | |
| Sex (male/female) | 55/39 | 6/10 | 57/100 | 0.3553 | >0.9999 | 0.019 |
| median (IQR 1) | median (IQR 1) | median (IQR 1) | ||||
| Age (years) | 67 (60–73) | 67 (49–71) | 46 (37–57) | 0.9333 | 0.0003 | <0.0001 |
| uFL-OPN 2 (ng/mL/mg protein) | 14.4 (11.1–191.1) | 13.3 (9.8–93.3) | 56,392 (30,271–115,517) | >0.9999 | <0.0001 | <0.0001 |
| Gal3C-S-OPN 3 /uFL-OPN 2 (MFI 4/uFL-OPN 2) | 52.3 (4.7–111.6) | 72.0 (21.5–144.2) | 0.007 (0.003–0.020) | >0.9999 | <0.0001 | <0.0001 |
| Group | Urolithiasis after URS | p-Value 5 | |
|---|---|---|---|
| Stone free (n = 14) | Residual stone (n = 7) | ||
| Sex (male/female) | 5/9 | 6/1 | 0.0635 |
| median (IQR 1) | median (IQR 1) | ||
| Age (years) | 61 (54–70) | 74 (57–77) | 0.3690 |
| uFL-OPN 2 (ng/mL/mg protein) | 120,003 (15.5–49,527) | 35.6 (8.8–18,322) | 0.0385 |
| Gal3C-S-OPN 3 /uFL-OPN 2 (MFI 4/uFL-OPN 2) | 0.02 (0.001–10.2) | 1.87 (0.026–88.6) | 0.0030 |
| Follow-up period (day) | 345 (213–407) | 206 (150–525) | 0.5815 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anan, G.; Yoneyama, T.; Noro, D.; Tobisawa, Y.; Hatakeyama, S.; Sutoh Yoneyama, M.; Yamamoto, H.; Imai, A.; Iwamura, H.; Kohada, Y.; et al. The Impact of Glycosylation of Osteopontin on Urinary Stone Formation. Int. J. Mol. Sci. 2020, 21, 93. https://doi.org/10.3390/ijms21010093
Anan G, Yoneyama T, Noro D, Tobisawa Y, Hatakeyama S, Sutoh Yoneyama M, Yamamoto H, Imai A, Iwamura H, Kohada Y, et al. The Impact of Glycosylation of Osteopontin on Urinary Stone Formation. International Journal of Molecular Sciences. 2020; 21(1):93. https://doi.org/10.3390/ijms21010093
Chicago/Turabian StyleAnan, Go, Tohru Yoneyama, Daisuke Noro, Yuki Tobisawa, Shingo Hatakeyama, Mihoko Sutoh Yoneyama, Hayato Yamamoto, Atsushi Imai, Hiromichi Iwamura, Yuki Kohada, and et al. 2020. "The Impact of Glycosylation of Osteopontin on Urinary Stone Formation" International Journal of Molecular Sciences 21, no. 1: 93. https://doi.org/10.3390/ijms21010093
APA StyleAnan, G., Yoneyama, T., Noro, D., Tobisawa, Y., Hatakeyama, S., Sutoh Yoneyama, M., Yamamoto, H., Imai, A., Iwamura, H., Kohada, Y., Mikami, J., Ito, J., Kaiho, Y., Yoneyama, T., Hashimoto, Y., Sato, M., & Ohyama, C. (2020). The Impact of Glycosylation of Osteopontin on Urinary Stone Formation. International Journal of Molecular Sciences, 21(1), 93. https://doi.org/10.3390/ijms21010093






