Superantigenic Activation of Human Cardiac Mast Cells
Abstract
:1. Introduction
2. Results
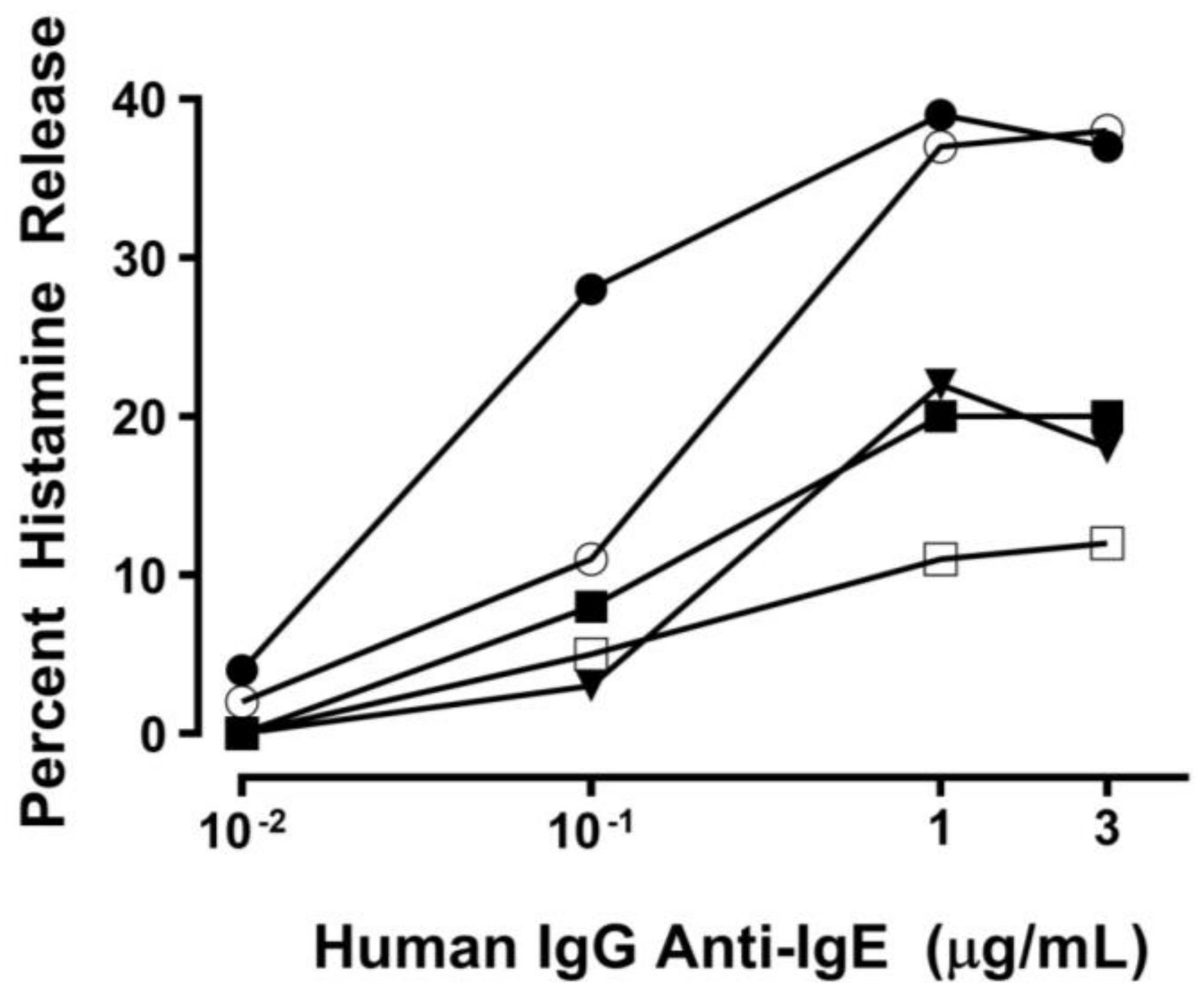
2.1. Effect of Human IgG Anti-IgE on Mediator Release from HHMCs
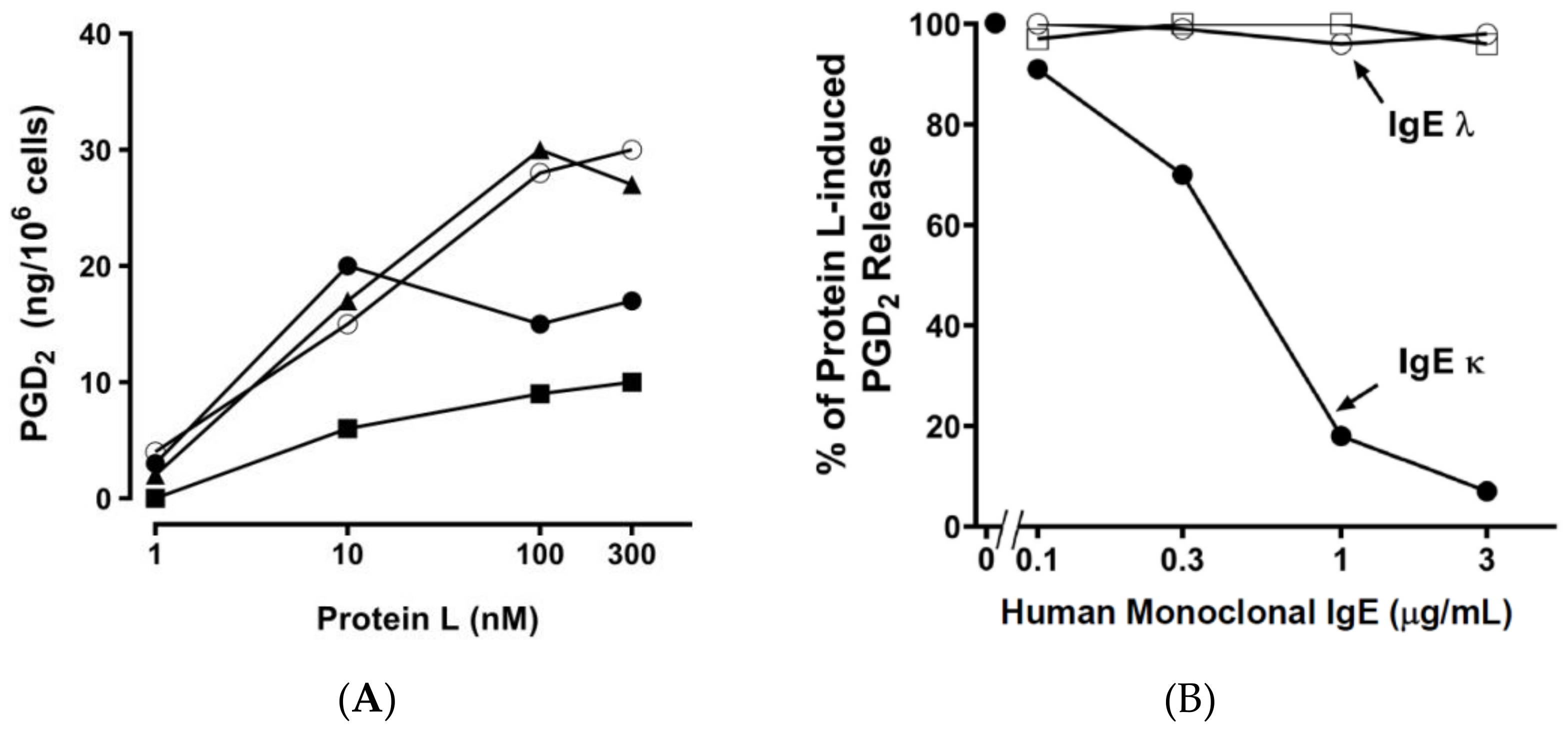
2.2. Effect of Bacterial Superantigens on Mediator Release from HHMCs
2.3. Effect of Viral Superantigens on Mediator Release from HHMCs
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Buffers
4.3. Human Monoclonal IgM and IgE and Human Polyclonal IgG
4.4. Isolation of HHMCs
4.5. Histamine Release Assay
4.6. Immunoassay of LTC4 and PGD2
4.7. VEGF-A and VEGF-C Release
4.8. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| S. aureus | Staphylococcus aureus |
| P. magnus | Peptostreptococcus magnus |
| LTC4 | Cysteinyl leukotriene C4 |
| HHMCs | Human heart mast cells |
| Ig | Immunoglobulin |
| FcεRI | High-affinity receptor for IgE |
| PGD2 | Prostaglandin D2 |
| gp120 | Glycoprotein 120 |
| VEGF | Vascular endothelial growth factor |
| SAg | Superantigen |
| APCs | Antigen-presenting cells |
| MHC | Major histocompatibility complex |
| TCR | T cell receptor |
| V | Variable |
| H | Heavy |
| L | Light |
| SE | Staphylococcus aureus enterotoxins |
| IL | Interleukin |
| HIV | Human immunodeficiency virus |
| FCS | Fetal calf serum |
| BSA | Bovine serum albumin |
| H-aIgE | Human IgG anti-IgE |
References
- White, J.; Herman, A.; Pullen, A.M.; Kubo, R.; Kappler, J.W.; Marrack, P. The V beta-specific superantigen staphylococcal enterotoxin B: Stimulation of mature T cells and clonal deletion in neonatal mice. Cell 1989, 56, 27–35. [Google Scholar] [CrossRef]
- Kotzin, B.L.; Leung, D.Y.; Kappler, J.; Marrack, P. Superantigens and their potential role in human disease. Adv. Immunol. 1993, 54, 99–166. [Google Scholar] [PubMed]
- Marone, G. Superantigens and superallergens. Chem. Immunol. Allergy 2007, 93. [Google Scholar] [CrossRef]
- Marrack, P.; Kappler, J. The staphylococcal enterotoxins and their relatives. Science 1990, 248, 705–711. [Google Scholar] [CrossRef]
- Bouvet, J.P.; Pires, R.; Lunel-Fabiani, F.; Crescenzo-Chaigne, B.; Maillard, P.; Valla, D.; Opolon, P.; Pillot, J. Protein F. A novel F(ab)-binding factor, present in normal liver, and largely released in the digestive tract during hepatitis. J. Immunol. 1990, 145, 1176–1180. [Google Scholar] [PubMed]
- Germain, R.N. Antigen presentation. The second class story. Nature 1991, 353, 605–607. [Google Scholar] [CrossRef] [PubMed]
- Fields, B.A.; Ober, B.; Malchiodi, E.L.; Lebedeva, M.I.; Braden, B.C.; Ysern, X.; Kim, J.K.; Shao, X.; Ward, E.S.; Mariuzza, R.A. Crystal structure of the V alpha domain of a T cell antigen receptor. Science 1995, 270, 1821–1824. [Google Scholar] [CrossRef]
- Li, H.; Llera, A.; Tsuchiya, D.; Leder, L.; Ysern, X.; Schlievert, P.M.; Karjalainen, K.; Mariuzza, R.A. Three-dimensional structure of the complex between a T cell receptor beta chain and the superantigen staphylococcal enterotoxin B. Immunity 1998, 9, 807–816. [Google Scholar] [CrossRef]
- Malchiodi, E.L.; Eisenstein, E.; Fields, B.A.; Ohlendorf, D.H.; Schlievert, P.M.; Karjalainen, K.; Mariuzza, R.A. Superantigen binding to a T cell receptor beta chain of known three-dimensional structure. J. Exp. Med. 1995, 182, 1833–1845. [Google Scholar] [CrossRef] [PubMed]
- Sundberg, E.J.; Li, H.; Llera, A.S.; McCormick, J.K.; Tormo, J.; Schlievert, P.M.; Karjalainen, K.; Mariuzza, R.A. Structures of two streptococcal superantigens bound to TCR beta chains reveal diversity in the architecture of T cell signaling complexes. Structure 2002, 10, 687–699. [Google Scholar] [CrossRef]
- Goodyear, C.S.; Silverman, G.J. B cell superantigens: A microbe’s answer to innate-like B cells and natural antibodies. Springer Semin. Immunopathol. 2005, 26, 463–484. [Google Scholar] [CrossRef] [PubMed]
- Viau, M.; Zouali, M. B-lymphocytes, innate immunity, and autoimmunity. Clin. Immunol. 2005, 114, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Bachert, C.; Gevaert, P.; Zhang, N.; van Zele, T.; Perez-Novo, C. Role of staphylococcal superantigens in airway disease. Chem. Immunol. Allergy 2007, 93, 214–236. [Google Scholar] [PubMed]
- Marone, G.; Rossi, F.W.; Detoraki, A.; Granata, F.; Genovese, A.; Spadaro, G. Role of superallergens in allergic disorders. Chem. Immunol. Allergy. 2007, 93, 195–213. [Google Scholar] [PubMed]
- Pastacaldi, C.; Lewis, P.; Howarth, P. Staphylococci and staphylococcal superantigens in asthma and rhinitis: A systematic review and meta-analysis. Allergy 2011, 66, 549–555. [Google Scholar] [CrossRef] [PubMed]
- Pascual, V.; Capra, J.D. B-cell superantigens? Curr. Biol. 1991, 1, 315–317. [Google Scholar] [CrossRef]
- Silverman, G.J.; Goodyear, C.S. A model B-cell superantigen and the immunobiology of B lymphocytes. Clin. Immunol. 2002, 102, 117–134. [Google Scholar] [CrossRef] [PubMed]
- Zouali, M. B-cell superantigens: Implications for selection of the human antibody repertoire. Immunol. Today 1995, 16, 399–405. [Google Scholar] [CrossRef]
- Thomas, D.; Chou, S.; Dauwalder, O.; Lina, G. Diversity in Staphylococcus aureus enterotoxins. Chem. Immunol. Allergy 2007, 93, 24–41. [Google Scholar] [PubMed]
- Friedrich, R.; Panizzi, P.; Fuentes-Prior, P.; Richter, K.; Verhamme, I.; Anderson, P.J.; Kawabata, S.; Huber, R.; Bode, W.; Bock, P.E. Staphylocoagulase is a prototype for the mechanism of cofactor-induced zymogen activation. Nature 2003, 425, 535–539. [Google Scholar] [CrossRef]
- Lowy, F.D. Staphylococcus aureus infections. N. Engl. J. Med. 1998, 339, 520–532. [Google Scholar] [CrossRef] [PubMed]
- Becker, S.; Frankel, M.B.; Schneewind, O.; Missiakas, D. Release of protein A from the cell wall of Staphylococcus aureus. Proc. Natl. Acad. Sci. USA 2014, 111, 1574–1579. [Google Scholar] [CrossRef]
- Forsgren, A.; Sjoquist, J. “Protein A” from S. aureus. I. Pseudo-immune reaction with human gamma-globulin. J. Immunol. 1966, 97, 822–827. [Google Scholar]
- Inganäs, M. Comparison of mechanisms of interaction between protein A from Staphylococcus aureus and human monoclonal IgG, IgA and IgM in relation to the classical FC gamma and the alternative F(ab’)2 epsilon protein A interactions. Scand. J. Immunol. 1981, 13, 343–352. [Google Scholar]
- Florio, G.; Petraroli, A.; Patella, V.; Triggiani, M.; Marone, G. The immunoglobulin superantigen-binding site of HIV-1 gp120 activates human basophils. AIDS 2000, 14, 931–938. [Google Scholar] [CrossRef]
- Patella, V.; Florio, G.; Petraroli, A.; Marone, G. HIV-1 gp120 induces IL-4 and IL-13 release from human Fc epsilon RI+ cells through interaction with the VH3 region of IgE. J. Immunol. 2000, 164, 589–595. [Google Scholar] [CrossRef]
- Killelea, B.K.; Chagpar, A.B.; Horowitz, N.R.; Lannin, D.R. Characteristics and treatment of human epidermal growth factor receptor 2 positive breast cancer: 43,485 cases from the National Cancer Database treated in 2010 and 2011. Am. J. Surg. 2017, 213, 426–432. [Google Scholar] [CrossRef]
- Kwong, P.D.; Wyatt, R.; Robinson, J.; Sweet, R.W.; Sodroski, J.; Hendrickson, W.A. Structure of an HIV gp120 envelope glycoprotein in complex with the CD4 receptor and a neutralizing human antibody. Nature 1998, 393, 648–659. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Broder, C.C.; Kennedy, P.E.; Berger, E.A. HIV-1 entry cofactor: functional cDNA cloning of a seven-transmembrane, G protein-coupled receptor. Science 1996, 272, 872–877. [Google Scholar] [CrossRef]
- Dragic, T.; Litwin, V.; Allaway, G.P.; Martin, S.R.; Huang, Y.; Nagashima, K.A.; Cayanan, C.; Maddon, P.J.; Koup, R.A.; Moore, J.P.; et al. HIV-1 entry into CD4+ cells is mediated by the chemokine receptor CC-CKR-5. Nature 1996, 381, 667–673. [Google Scholar] [CrossRef]
- Karray, S.; Zouali, M. Identification of the B cell superantigen-binding site of HIV-1 gp120. Proc. Natl. Acad. Sci. USA 1997, 94, 1356–1360. [Google Scholar] [CrossRef]
- Silverman, G.J. B-cell superantigens. Immunol. Today. 1997, 18, 379–386. [Google Scholar] [CrossRef]
- Zouali, M. B cell superantigens subvert innate functions of B cells. Chem. Immunol. Allergy 2007, 93, 92–105. [Google Scholar]
- Durand, M.; Chartrand-Lefebvre, C.; Baril, J.G.; Trottier, S.; Trottier, B.; Harris, M.; Walmsley, S.; Conway, B.; Wong, A.; Routy, J.P.; et al. The Canadian HIV and aging cohort study—Determinants of increased risk of cardio-vascular diseases in HIV-infected individuals: rationale and study protocol. BMC Infect. Dis. 2017, 17, 611. [Google Scholar] [CrossRef]
- Teer, E.; Essop, M.F. HIV and cardiovascular disease: Role of immunometabolic perturbations. Physiology (Bethesda) 2018, 33, 74–82. [Google Scholar] [CrossRef]
- Lakhrif, Z.; Pugniere, M.; Henriquet, C.; di Tommaso, A.; Dimier-Poisson, I.; Billiald, P.; Juste, M.O.; Aubrey, N. A method to confer Protein L binding ability to any antibody fragment. mAbs 2016, 8, 379–388. [Google Scholar] [CrossRef]
- Bjorck, L. Protein L. A novel bacterial cell wall protein with affinity for Ig L chains. J. Immunol. 1988, 140, 1194–1197. [Google Scholar]
- Myhre, E.B.; Erntell, M. A non-immune interaction between the light chain of human immunoglobulin and a surface component of a Peptococcus magnus strain. Mol. Immunol. 1985, 22, 879–885. [Google Scholar] [CrossRef]
- Patella, V.; Casolaro, V.; Bjorck, L.; Marone, G. Protein L. A bacterial Ig-binding protein that activates human basophils and mast cells. J. Immunol. 1990, 145, 3054–3061. [Google Scholar]
- Genovese, A.; Bouvet, J.P.; Florio, G.; Lamparter-Schummert, B.; Bjorck, L.; Marone, G. Bacterial immunoglobulin superantigen proteins A and L activate human heart mast cells by interacting with immunoglobulin E. Infect. Immun. 2000, 68, 5517–5524. [Google Scholar] [CrossRef]
- Genovese, A.; Borgia, G.; Bjorck, L.; Petraroli, A.; de Paulis, A.; Piazza, M.; Marone, G. Immunoglobulin superantigen protein L induces IL-4 and IL-13 secretion from human Fc epsilon RI+ cells through interaction with the kappa light chains of IgE. J. Immunol. 2003, 170, 1854–1861. [Google Scholar] [CrossRef]
- Nilson, B.H.; Solomon, A.; Bjorck, L.; Akerstrom, B. Protein L from Peptostreptococcus magnus binds to the kappa light chain variable domain. J. Biol. Chem. 1992, 267, 2234–2239. [Google Scholar]
- Ingason, A.B.; Mechmet, F.; Atacho, D.A.M.; Steingrimsson, E.; Petersen, P.H. Distribution of mast cells within the mouse heart and its dependency on Mitf. Mol. Immunol. 2018, 105, 9–15. [Google Scholar] [CrossRef]
- Aldi, S.; Robador, P.A.; Tomita, K.; Di Lorenzo, A.; Levi, R. IgE receptor-mediated mast-cell renin release. Am. J. Pathol. 2014, 184, 376–381. [Google Scholar] [CrossRef]
- Ponomaryov, T.; Payne, H.; Fabritz, L.; Wagner, D.D.; Brill, A. Mast cells granular contents are crucial for deep vein thrombosis in mice. Circ. Res. 2017, 121, 941–950. [Google Scholar] [CrossRef]
- Somasundaram, P.; Ren, G.; Nagar, H.; Kraemer, D.; Mendoza, L.; Michael, L.H.; Caughey, G.H.; Entman, M.L.; Frangogiannis, N.G. Mast cell tryptase may modulate endothelial cell phenotype in healing myocardial infarcts. J. Pathol. 2005, 205, 102–111. [Google Scholar]
- Frangogiannis, N.G.; Mendoza, L.H.; Lindsey, M.L.; Ballantyne, C.M.; Michael, L.H.; Smith, C.W.; Entman, M.L. IL-10 is induced in the reperfused myocardium and may modulate the reaction to injury. J. Immunol. 2000, 165, 2798–2808. [Google Scholar] [CrossRef]
- Patella, V.; Marino, I.; Lamparter, B.; Arbustini, E.; Adt, M.; Marone, G. Human heart mast cells. Isolation, purification, ultrastructure, and immunologic characterization. J. Immunol. 1995, 154, 2855–2865. [Google Scholar]
- Patella, V.; de Crescenzo, G.; Marino, I.; Genovese, A.; Adt, M.; Gleich, G.J.; Marone, G. Eosinophil granule proteins activate human heart mast cells. J. Immunol. 1996, 157, 1219–1225. [Google Scholar]
- Kaartinen, M.; Penttila, A.; Kovanen, P.T. Mast cells accompany microvessels in human coronary atheromas: Implications for intimal neovascularization and hemorrhage. Atherosclerosis 1996, 123, 123–131. [Google Scholar] [CrossRef]
- Bankl, H.C.; Radaszkiewicz, T.; Klappacher, G.W.; Glogar, D.; Sperr, W.R.; Grossschmidt, K.; Bankl, H.; Lechner, K.; Valent, P. Increase and redistribution of cardiac mast cells in auricular thrombosis. Possible role of kit ligand. Circulation 1995, 91, 275–283. [Google Scholar] [CrossRef] [PubMed]
- Varricchi, G.; Raap, U.; Rivellese, F.; Marone, G.; Gibbs, B.F. Human mast cells and basophils-How are they similar how are they different? Immunol. Rev. 2018, 282, 8–34. [Google Scholar] [CrossRef] [PubMed]
- Borriello, F.; Granata, F.; Varricchi, G.; Genovese, A.; Triggiani, M.; Marone, G. Immunopharmacological modulation of mast cells. Curr. Opin. Pharmacol. 2014, 17, 45–57. [Google Scholar] [CrossRef] [PubMed]
- Mukai, K.; Tsai, M.; Saito, H.; Galli, S.J. Mast cells as sources of cytokines, chemokines, and growth factors. Immunol. Rev. 2018, 282, 121–150. [Google Scholar] [CrossRef]
- Galli, S.J. The mast cell-IgE paradox: From homeostasis to anaphylaxis. Am. J. Pathol. 2016, 186, 212–224. [Google Scholar] [CrossRef]
- Bradding, P.; Arthur, G. Mast cells in asthma—State of the art. Clin. Exp. Allergy 2016, 46, 194–263. [Google Scholar] [CrossRef]
- Olivera, A.; Beaven, M.A.; Metcalfe, D.D. Mast cells signal their importance in health and disease. J. Allergy Clin. Immunol. 2018, 142, 381–393. [Google Scholar] [CrossRef]
- Piliponsky, A.M.; Romani, L. The contribution of mast cells to bacterial and fungal infection immunity. Immunol. Rev. 2018, 282, 188–197. [Google Scholar] [CrossRef]
- Marone, G.; Varricchi, G.; Loffredo, S.; Galdiero, M.R.; Rivellese, F.; de Paulis, A. Are basophils and mast cells masters in HIV Infection? Int. Arch. Allergy Immunol. 2016, 171, 158–165. [Google Scholar] [CrossRef]
- Suurmond, J.; Rivellese, F.; Dorjee, A.L.; Bakker, A.M.; Rombouts, Y.J.; Rispens, T.; Wolbink, G.; Zaldumbide, A.; Hoeben, R.C.; Huizinga, T.W.; et al. Toll-like receptor triggering augments activation of human mast cells by anti-citrullinated protein antibodies. Ann. Rheum. Dis. 2015, 74, 1915–1923. [Google Scholar] [CrossRef]
- Detoraki, A.; Staiano, R.I.; Granata, F.; Giannattasio, G.; Prevete, N.; de Paulis, A.; Ribatti, D.; Genovese, A.; Triggiani, M.; Marone, G. Vascular endothelial growth factors synthesized by human lung mast cells exert angiogenic effects. J. Allergy Clin. Immunol. 2009, 123, 1142–1149. [Google Scholar] [CrossRef]
- Varricchi, G.; Loffredo, S.; Galdiero, M.R.; Marone, G.; Cristinziano, L.; Granata, F. Innate effector cells in angiogenesis and lymphangiogenesis. Curr. Opin. Immunol. 2018, 53, 152–160. [Google Scholar] [CrossRef]
- Marone, G.; Varricchi, G.; Loffredo, S.; Granata, F. Mast cells and basophils in inflammatory and tumor angiogenesis and lymphangiogenesis. Eur. J. Pharmacol. 2016, 778, 146–151. [Google Scholar] [CrossRef]
- Abdel-Majid, R.M.; Marshall, J.S. Prostaglandin E2 induces degranulation-independent production of vascular endothelial growth factor by human mast cells. J. Immunol. 2004, 172, 1227–1236. [Google Scholar] [CrossRef] [PubMed]
- Theoharides, T.C.; Zhang, B.; Kempuraj, D.; Tagen, M.; Vasiadi, M.; Angelidou, A.; Alysandratos, K.D.; Kalogeromitros, D.; Asadi, S.; Stavrianeas, N.; et al. IL-33 augments substance P-induced VEGF secretion from human mast cells and is increased in psoriatic skin. Proc. Natl. Acad. Sci. USA 2010, 107, 4448–4453. [Google Scholar] [CrossRef]
- Varricchi, G.; Granata, F.; Loffredo, S.; Genovese, A.; Marone, G. Angiogenesis and lymphangiogenesis in inflammatory skin disorders. J. Am. Acad. Dermatol. 2015, 73, 144–153. [Google Scholar] [CrossRef] [PubMed]
- Rivellese, F.; Suurmond, J.; Habets, K.; Dorjee, A.L.; Ramamoorthi, N.; Townsend, M.J.; de Paulis, A.; Marone, G.; Huizinga, T.W.; Pitzalis, C.; et al. Ability of interleukin-33- and immune complex-triggered activation of human mast cells to down-regulate monocyte-mediated immune responses. Arthritis Rheumatol. 2015, 67, 2343–2353. [Google Scholar] [CrossRef] [PubMed]
- Rivellese, F.; Nerviani, A.; Rossi, F.W.; Marone, G.; Matucci-Cerinic, M.; de Paulis, A.; Pitzalis, C. Mast cells in rheumatoid arthritis: friends or foes? Autoimmun. Rev. 2017, 16, 557–563. [Google Scholar] [CrossRef] [PubMed]
- Rivellese, F.; Mauro, D.; Nerviani, A.; Pagani, S.; Fossati-Jimack, L.; Messemaker, T.; Kurreeman, F.A.S.; Toes, R.E.M.; Ramming, A.; Rauber, S.; et al. Mast cells in early rheumatoid arthritis associate with disease severity and support B cell autoantibody production. Ann. Rheum. Dis. 2018, 77, 1773–1781. [Google Scholar] [CrossRef]
- Visciano, C.; Liotti, F.; Prevete, N.; Cali, G.; Franco, R.; Collina, F.; de Paulis, A.; Marone, G.; Santoro, M.; Melillo, R.M. Mast cells induce epithelial-to-mesenchymal transition and stem cell features in human thyroid cancer cells through an IL-8-Akt-Slug pathway. Oncogene 2015, 34, 5175–5186. [Google Scholar] [CrossRef] [PubMed]
- Galdiero, M.R.; Varricchi, G.; Marone, G. The immune network in thyroid cancer. Oncoimmunology 2016, 5, e1168556. [Google Scholar] [CrossRef]
- Varricchi, G.; Galdiero, M.R.; Loffredo, S.; Marone, G.; Iannone, R.; Granata, F. Are mast cells MASTers in cancer? Front Immunol. 2017, 8, 424. [Google Scholar] [CrossRef]
- Varricchi, G.; Galdiero, M.R.; Marone, G.; Granata, F.; Borriello, F. Controversial role of mast cells in skin cancers. Exp. Dermatol. 2017, 26, 11–17. [Google Scholar] [CrossRef]
- Fairweather, D.; Frisancho-Kiss, S.; Gatewood, S.; Njoku, D.; Steele, R.; Barrett, M.; Rose, N.R. Mast cells and innate cytokines are associated with susceptibility to autoimmune heart disease following coxsackievirus B3 infection. Autoimmunity 2004, 37, 131–145. [Google Scholar] [CrossRef]
- Varricchi, G.; Galdiero, M.R.; Tocchetti, C.G. Cardiac toxicity of immune checkpoint inhibitors: Cardio-oncology meets immunology. Circulation 2017, 136, 1989–1992. [Google Scholar] [CrossRef]
- Ngkelo, A.; Richart, A.; Kirk, J.A.; Bonnin, P.; Vilar, J.; Lemitre, M.; Marck, P.; Branchereau, M.; Le Gall, S.; Renault, N.; et al. Mast cells regulate myofilament calcium sensitization and heart function after myocardial infarction. J. Exp. Med. 2016, 213, 1353–1374. [Google Scholar] [CrossRef]
- Patella, V.; Marino, I.; Arbustini, E.; Lamparter-Schummert, B.; Verga, L.; Adt, M.; Marone, G. Stem cell factor in mast cells and increased mast cell density in idiopathic and ischemic cardiomyopathy. Circulation 1998, 97, 971–978. [Google Scholar] [CrossRef]
- Shi, G.P.; Bot, I.; Kovanen, P.T. Mast cells in human and experimental cardiometabolic diseases. Nat. Rev. Cardiol. 2015, 12, 643–658. [Google Scholar] [CrossRef]
- De Paulis, A.; Prevete, N.; Fiorentino, I.; Rossi, F.W.; Staibano, S.; Montuori, N.; Ragno, P.; Longobardi, A.; Liccardo, B.; Genovese, A.; et al. Expression and functions of the vascular endothelial growth factors and their receptors in human basophils. J. Immunol. 2006, 177, 7322–7331. [Google Scholar] [CrossRef] [PubMed]
- Granata, F.; Frattini, A.; Loffredo, S.; Staiano, R.I.; Petraroli, A.; Ribatti, D.; Oslund, R.; Gelb, M.H.; Lambeau, G.; Marone, G.; et al. Production of vascular endothelial growth factors from human lung macrophages induced by group IIA and group X secreted phospholipases A2. J. Immunol. 2010, 184, 5232–5241. [Google Scholar] [CrossRef]
- Staiano, R.I.; Loffredo, S.; Borriello, F.; Iannotti, F.A.; Piscitelli, F.; Orlando, P.; Secondo, A.; Granata, F.; Lepore, M.T.; Fiorelli, A.; et al. Human lung-resident macrophages express CB1 and CB2 receptors whose activation inhibits the release of angiogenic and lymphangiogenic factors. J. Leukoc. Biol. 2016, 99, 531–540. [Google Scholar] [CrossRef]
- Loffredo, S.; Borriello, F.; Iannone, R.; Ferrara, A.L.; Galdiero, M.R.; Gigantino, V.; Esposito, P.; Varricchi, G.; Lambeau, G.; Cassatella, M.A.; et al. Group V secreted phospholipase A2 induces the release of proangiogenic and antiangiogenic factors by human neutrophils. Front. Immunol. 2017, 8, 443. [Google Scholar] [CrossRef]
- Benyon, R.C.; Lowman, M.A.; Church, M.K. Human skin mast cells: Their dispersion, purification, and secretory characterization. J. Immunol. 1987, 138, 861–867. [Google Scholar] [PubMed]
- Guhl, S.; Lee, H.H.; Babina, M.; Henz, B.M.; Zuberbier, T. Evidence for a restricted rather than generalized stimulatory response of skin-derived human mast cells to substance P. J. Neuroimmunol. 2005, 163, 92–101. [Google Scholar] [CrossRef]
- De Paulis, A.; Marino, I.; Ciccarelli, A.; de Crescenzo, G.; Concardi, M.; Verga, L.; Arbustini, E.; Marone, G. Human synovial mast cells. I. Ultrastructural in situ and in vitro immunologic characterization. Arthritis Rheum. 1996, 39, 1222–1233. [Google Scholar] [CrossRef]
- Marone, G.; Casolaro, V.; Paganelli, R.; Quinti, I. IgG anti-IgE from atopic dermatitis induces mediator release from basophils and mast cells. J. Investig. Dermatol. 1989, 93, 246–252. [Google Scholar] [CrossRef]
- Taimeh, Z.; Loughran, J.; Birks, E.J.; Bolli, R. Vascular endothelial growth factor in heart failure. Nat. Rev. Cardiol. 2013, 10, 519–530. [Google Scholar] [CrossRef]
- Varricchi, G.; Harker, J.; Borriello, F.; Marone, G.; Durham, S.R.; Shamji, M.H. T follicular helper (Tfh) cells in normal immune responses and in allergic disorders. Allergy 2016, 71, 1086–1094. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Cheng, X.; Xiang, M.X.; Alanne-Kinnunen, M.; Wang, J.A.; Chen, H.; He, A.; Sun, X.; Lin, Y.; Tang, T.T.; et al. IgE stimulates human and mouse arterial cell apoptosis and cytokine expression and promotes atherogenesis in Apoe-/-mice. J. Clin. Investig. 2011, 121, 3564–3577. [Google Scholar] [CrossRef] [PubMed]
- Szczeklik, A.; Sladek, K.; Szczerba, A.; Dropinski, J. Serum immunoglobulin E response to myocardial infarction. Circulation 1988, 77, 1245–1249. [Google Scholar] [CrossRef] [PubMed]
- Kovanen, P.T.; Manttari, M.; Palosuo, T.; Manninen, V.; Aho, K. Prediction of myocardial infarction in dyslipidemic men by elevated levels of immunoglobulin classes A, E, and G, but not M. Arch. Intern. Med. 1998, 158, 1434–1439. [Google Scholar] [CrossRef]
- Kareinen, I.; Baumann, M.; Nguyen, S.D.; Maaninka, K.; Anisimov, A.; Tozuka, M.; Jauhiainen, M.; Lee-Rueckert, M.; Kovanen, P.T. Chymase released from hypoxia-activated cardiac mast cells cleaves human apoA-I at Tyr(192) and compromises its cardioprotective activity. J. Lipid Res. 2018, 59, 945–957. [Google Scholar] [CrossRef]
- Theoharides, T.C.; Sismanopoulos, N.; Delivanis, D.A.; Zhang, B.; Hatziagelaki, E.E.; Kalogeromitros, D. Mast cells squeeze the heart and stretch the gird: their role in atherosclerosis and obesity. Trends Pharmacol. Sci. 2011, 32, 534–542. [Google Scholar] [CrossRef]
- Dell’Italia, L.J.; Collawn, J.F.; Ferrario, C.M. Multifunctional role of chymase in acute and chronic tissue injury and remodeling. Circ. Res. 2018, 122, 319–336. [Google Scholar]
- Uemura, K.; Kondo, H.; Ishii, Y.; Kobukata, M.; Haraguchi, M.; Imamura, T.; Otsubo, T.; Ikebe-Ebata, Y.; Abe, I.; Ayabe, R.; et al. Mast cells play an important role in the pathogenesis of hyperglycemia-induced atrial fibrillation. J Cardiovasc. Electrophysiol. 2016, 27, 981–989. [Google Scholar] [CrossRef] [PubMed]
- Shubin, N.J.; Glukhova, V.A.; Clauson, M.; Truong, P.; Abrink, M.; Pejler, G.; White, N.J.; Deutsch, G.H.; Reeves, S.R.; Vaisar, T.; et al. Proteome analysis of mast cell releasates reveals a role for chymase in the regulation of coagulation factor XIIIA levels via proteolytic degradation. J. Allergy Clin. Immunol. 2017, 139, 323–334. [Google Scholar] [CrossRef]
- Wroblewski, M.; Bauer, R.; Cubas Cordova, M.; Udonta, F.; Ben-Batalla, I.; Legler, K.; Hauser, C.; Egberts, J.; Janning, M.; Velthaus, J.; et al. Mast cells decrease efficacy of anti-angiogenic therapy by secreting matrix-degrading granzyme B. Nat. Commun. 2017, 8, 269. [Google Scholar] [CrossRef]
- Nascimento, C.R.; Andrade, D.; Carvalho-Pinto, C.E.; Serra, R.R.; Vellasco, L.; Brasil, G.; Ramos-Junior, E.S.; da Mota, J.B.; Almeida, L.N.; Andrade, M.V.; et al. Mast cell coupling to the kallikrein-kinin system fuels intracardiac parasitism and worsens heart pathology in experimental chagas disease. Front. Immunol. 2017, 8, 840. [Google Scholar] [CrossRef]
- Marone, G.; Borriello, F.; Varricchi, G.; Genovese, A.; Granata, F. Basophils: Historical reflections and perspectives. Chem. Immunol. Allergy. 2014, 100, 172–192. [Google Scholar]
- Sun, Y.; Vandenbriele, C.; Kauskot, A.; Verhamme, P.; Hoylaerts, M.F.; Wright, G.J. A human platelet receptor protein microarray identifies the high affinity immunoglobulin E receptor subunit alpha (FcepsilonR1alpha) as an activating platelet endothelium aggregation receptor 1 (PEAR1) ligand. Mol. Cell Proteom. 2015, 14, 1265–1274. [Google Scholar] [CrossRef] [PubMed]
- Nawata, Y.; Koike, T.; Hosokawa, H.; Tomioka, H.; Yoshida, S. Anti-IgE autoantibody in patients with atopic dermatitis. J. Immunol. 1985, 135, 478–482. [Google Scholar]
- Sabroe, R.A.; Seed, P.T.; Francis, D.M.; Barr, R.M.; Black, A.K.; Greaves, M.W. Chronic idiopathic urticaria: comparison of the clinical features of patients with and without anti-FcepsilonRI or anti-IgE autoantibodies. J. Am. Acad. Dermatol. 1999, 40, 443–450. [Google Scholar] [CrossRef]
- Gruber, B.L.; Baeza, M.L.; Marchese, M.J.; Agnello, V.; Kaplan, A.P. Prevalence and functional role of anti-IgE autoantibodies in urticarial syndromes. J. Investig. Dermatol. 1988, 90, 213–217. [Google Scholar] [CrossRef]
- Sanjuan, M.A.; Sagar, D.; Kolbeck, R. Role of IgE in autoimmunity. J. Allergy Clin. Immunol. 2016, 137, 1651–1661. [Google Scholar] [CrossRef]
- Marone, G.; Granata, F. Angiogenesis, lymphangiogenesis and clinical implications. Preface. Chem. Immunol. Allergy. 2014, 99, 11–12. [Google Scholar]
- Bry, M.; Kivela, R.; Holopainen, T.; Anisimov, A.; Tammela, T.; Soronen, J.; Silvola, J.; Saraste, A.; Jeltsch, M.; Korpisalo, P.; et al. Vascular endothelial growth factor-B acts as a coronary growth factor in transgenic rats without inducing angiogenesis, vascular leak, or inflammation. Circulation 2010, 122, 1725–1733. [Google Scholar] [CrossRef]
- Bosisio, D.; Ronca, R.; Salvi, V.; Presta, M.; Sozzani, S. Dendritic cells in inflammatory angiogenesis and lymphangiogenesis. Curr. Opin. Immunol. 2018, 53, 180–186. [Google Scholar] [CrossRef]
- Longo, V.; Tamma, R.; Brunetti, O.; Pisconti, S.; Argentiero, A.; Silvestris, N.; Ribatti, D. Mast cells and angiogenesis in pancreatic ductal adenocarcinoma. Clin. Exp. Med. 2018, 18, 319–323. [Google Scholar] [CrossRef]
- Albini, A.; Bruno, A.; Noonan, D.M.; Mortara, L. Contribution to tumor angiogenesis from innate immune cells within the tumor microenvironment: Implications for immunotherapy. Front. Immunol. 2018, 9, 527. [Google Scholar] [CrossRef]
- Wilson, A.M.; Shao, Z.; Grenier, V.; Mawambo, G.; Daudelin, J.F.; Dejda, A.; Pilon, F.; Popovic, N.; Boulet, S.; Parinot, C.; et al. Neuropilin-1 expression in adipose tissue macrophages protects against obesity and metabolic syndrome. Sci. Immunol. 2018, 3, eaan4626. [Google Scholar] [CrossRef]
- Miller, A.J. The grossly invisible and generally ignored lymphatics of the mammalian heart. Med. Hypotheses 2011, 76, 604–606. [Google Scholar] [CrossRef]
- Aspelund, A.; Robciuc, M.R.; Karaman, S.; Makinen, T.; Alitalo, K. Lymphatic system in cardiovascular medicine. Circ. Res. 2016, 118, 515–530. [Google Scholar] [CrossRef]
- Kholova, I.; Dragneva, G.; Cermakova, P.; Laidinen, S.; Kaskenpaa, N.; Hazes, T.; Cermakova, E.; Steiner, I.; Yla-Herttuala, S. Lymphatic vasculature is increased in heart valves, ischaemic and inflamed hearts and in cholesterol-rich and calcified atherosclerotic lesions. Eur. J. Clin. Investig. 2011, 41, 487–497. [Google Scholar] [CrossRef]
- Norman, S.; Riley, P.R. Anatomy and development of the cardiac lymphatic vasculature: Its role in injury and disease. Clin. Anat. 2016, 29, 305–315. [Google Scholar] [CrossRef]
- Machnik, A.; Neuhofer, W.; Jantsch, J.; Dahlmann, A.; Tammela, T.; Machura, K.; Park, J.K.; Beck, F.X.; Muller, D.N.; Derer, W.; et al. Macrophages regulate salt-dependent volume and blood pressure by a vascular endothelial growth factor-C-dependent buffering mechanism. Nat. Med. 2009, 15, 545–552. [Google Scholar] [CrossRef]
- Wiig, H.; Schroder, A.; Neuhofer, W.; Jantsch, J.; Kopp, C.; Karlsen, T.V.; Boschmann, M.; Goss, J.; Bry, M.; Rakova, N.; et al. Immune cells control skin lymphatic electrolyte homeostasis and blood pressure. J. Clin. Investig. 2013, 123, 2803–2815. [Google Scholar] [CrossRef]
- Chen, H.I.; Poduri, A.; Numi, H.; Kivela, R.; Saharinen, P.; McKay, A.S.; Raftrey, B.; Churko, J.; Tian, X.; Zhou, B.; et al. VEGF-C and aortic cardiomyocytes guide coronary artery stem development. J. Clin. Investig. 2014, 124, 4899–4914. [Google Scholar] [CrossRef]
- Mylonakis, E.; Calderwood, S.B. Infective endocarditis in adults. N. Engl. J. Med. 2001, 345, 1318–1330. [Google Scholar] [CrossRef]
- Shahreyar, M.; Fahhoum, R.; Akinseye, O.; Bhandari, S.; Dang, G.; Khouzam, R.N. Severe sepsis and cardiac arrhythmias. Ann. Transl. Med. 2018, 6, 6. [Google Scholar] [CrossRef]
- Ulloa-Morales, A.J.; Goodyear, C.S.; Silverman, G.J. Essential domain-dependent roles within soluble IgG for in vivo superantigen properties of staphylococcal Protein A: Resolving the B-Cell Superantigen paradox. Front. Immunol. 2018, 9, 2011. [Google Scholar] [CrossRef]
- Vigorito, C.; Giordano, A.; Cirillo, R.; Genovese, A.; Rengo, F.; Marone, G. Metabolic and hemodynamic effects of peptide leukotriene C4 and D4 in man. Int. J. Clin. Lab. Res. 1997, 27, 178–184. [Google Scholar] [CrossRef]
- Levi, R.; Malm, J.R.; Bowman, F.O.; Rosen, M.R. The arrhythmogenic actions of histamine on human atrial fibers. Circ. Res. 1981, 49, 545–550. [Google Scholar] [CrossRef]
- Vigorito, C.; Poto, S.; Picotti, G.B.; Triggiani, M.; Marone, G. Effect of activation of the H1 receptor on coronary hemodynamics in man. Circulation 1986, 73, 1175–1182. [Google Scholar] [CrossRef]
- Hattori, Y.; Levi, R. Effect of PGD2 on cardiac contractility: A negative inotropism secondary to coronary vasoconstriction conceals a primary positive inotropic action. J. Pharmacol. Exp. Ther. 1986, 237, 719–724. [Google Scholar]
- Gelderblom, H.R.; Hausmann, E.H.; Ozel, M.; Pauli, G.; Koch, M.A. Fine structure of human immunodeficiency virus (HIV) and immunolocalization of structural proteins. Virology 1987, 156, 171–176. [Google Scholar] [CrossRef]
- Tseng, Z.H.; Secemsky, E.A.; Dowdy, D.; Vittinghoff, E.; Moyers, B.; Wong, J.K.; Havlir, D.V.; Hsue, P.Y. Sudden cardiac death in patients with human immunodeficiency virus infection. J. Am. Coll. Cardiol. 2012, 59, 1891–1896. [Google Scholar] [CrossRef]
- Zaaqoq, A.M.; Khasawneh, F.A.; Smalligan, R.D. Cardiovascular complications of HIV-associated immune dysfunction. Cardiol. Res. Pract. 2015, 2015, 302638. [Google Scholar] [CrossRef]
- Theoharides, T.C.; Kavalioti, M. Stress, inflammation and natural treatments. J. Biol. Regul. Homeost Agents 2018, 32, 1345–1347. [Google Scholar]
- Kritas, S.K.; Gallenga, C.E.; Ronconi, G.; Caraffa, A.; Toniato, E.; Lauritano, D.; Conti, P. Impact of mold on mast cell-cytokine immune response. J. Biol. Regul. Homeost. Agents 2018, 32, 763–768. [Google Scholar]
- Gupta, K.; Idahosa, C.; Roy, S.; Lee, D.; Subramanian, H.; Dhingra, A.; Boesze-Battaglia, K.; Korostoff, J.; Ali, H. Differential regulation of mas-related G Protein-coupled receptor X2-mediated mast cell degranulation by antimicrobial host defense peptides and porphyromonas gingivalis Lipopolysaccharide. Infect. Immun. 2017, 85, e00246-17. [Google Scholar] [CrossRef]
- Sala, P.; Tonutti, E.; Ruscio, M.; Colle, R.; Antonutto, G.; Falconieri, G. IgE myeloma. Report of a new case and review of the literature. Haematologica 1981, 66, 787–795. [Google Scholar]
- Marone, G.; Tamburini, M.; Giudizi, M.G.; Biagiotti, R.; Almerigogna, F.; Romagnani, S. Mechanism of activation of human basophils by Staphylococcus aureus Cowan 1. Infect. Immun. 1987, 55, 803–809. [Google Scholar] [PubMed]
- Patella, V.; Giuliano, A.; Bouvet, J.P.; Marone, G. Endogenous superallergen protein Fv induces IL-4 secretion from human Fc epsilon RI+ cells through interaction with the VH3 region of IgE. J. Immunol. 1998, 161, 5647–5655. [Google Scholar] [PubMed]
- Siraganian, R.P. An automated continuous-flow system for the extraction and fluorometric analysis of histamine. Anal. Biochem. 1974, 57, 383–394. [Google Scholar] [CrossRef]
- De Paulis, A.; Cirillo, R.; Ciccarelli, A.; de Crescenzo, G.; Oriente, A.; Marone, G. Characterization of the anti-inflammatory effect of FK-506 on human mast cells. J. Immunol. 1991, 147, 4278–4285. [Google Scholar]
- Loffredo, S.; Ferrara, A.L.; Bova, M.; Borriello, F.; Suffritti, C.; Veszeli, N.; Petraroli, A.; Galdiero, M.R.; Varricchi, G.; Granata, F.; et al. Secreted phospholipases A2 in hereditary angioedema with C1-inhibitor deficiency. Front. Immunol. 2018, 9, 1721. [Google Scholar] [CrossRef] [PubMed]




© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Varricchi, G.; Loffredo, S.; Borriello, F.; Pecoraro, A.; Rivellese, F.; Genovese, A.; Spadaro, G.; Marone, G. Superantigenic Activation of Human Cardiac Mast Cells. Int. J. Mol. Sci. 2019, 20, 1828. https://doi.org/10.3390/ijms20081828
Varricchi G, Loffredo S, Borriello F, Pecoraro A, Rivellese F, Genovese A, Spadaro G, Marone G. Superantigenic Activation of Human Cardiac Mast Cells. International Journal of Molecular Sciences. 2019; 20(8):1828. https://doi.org/10.3390/ijms20081828
Chicago/Turabian StyleVarricchi, Gilda, Stefania Loffredo, Francesco Borriello, Antonio Pecoraro, Felice Rivellese, Arturo Genovese, Giuseppe Spadaro, and Gianni Marone. 2019. "Superantigenic Activation of Human Cardiac Mast Cells" International Journal of Molecular Sciences 20, no. 8: 1828. https://doi.org/10.3390/ijms20081828
APA StyleVarricchi, G., Loffredo, S., Borriello, F., Pecoraro, A., Rivellese, F., Genovese, A., Spadaro, G., & Marone, G. (2019). Superantigenic Activation of Human Cardiac Mast Cells. International Journal of Molecular Sciences, 20(8), 1828. https://doi.org/10.3390/ijms20081828






