Structure-Guided Immobilization of an Evolved Unspecific Peroxygenase
Abstract
1. Introduction
2. Results and Discussion
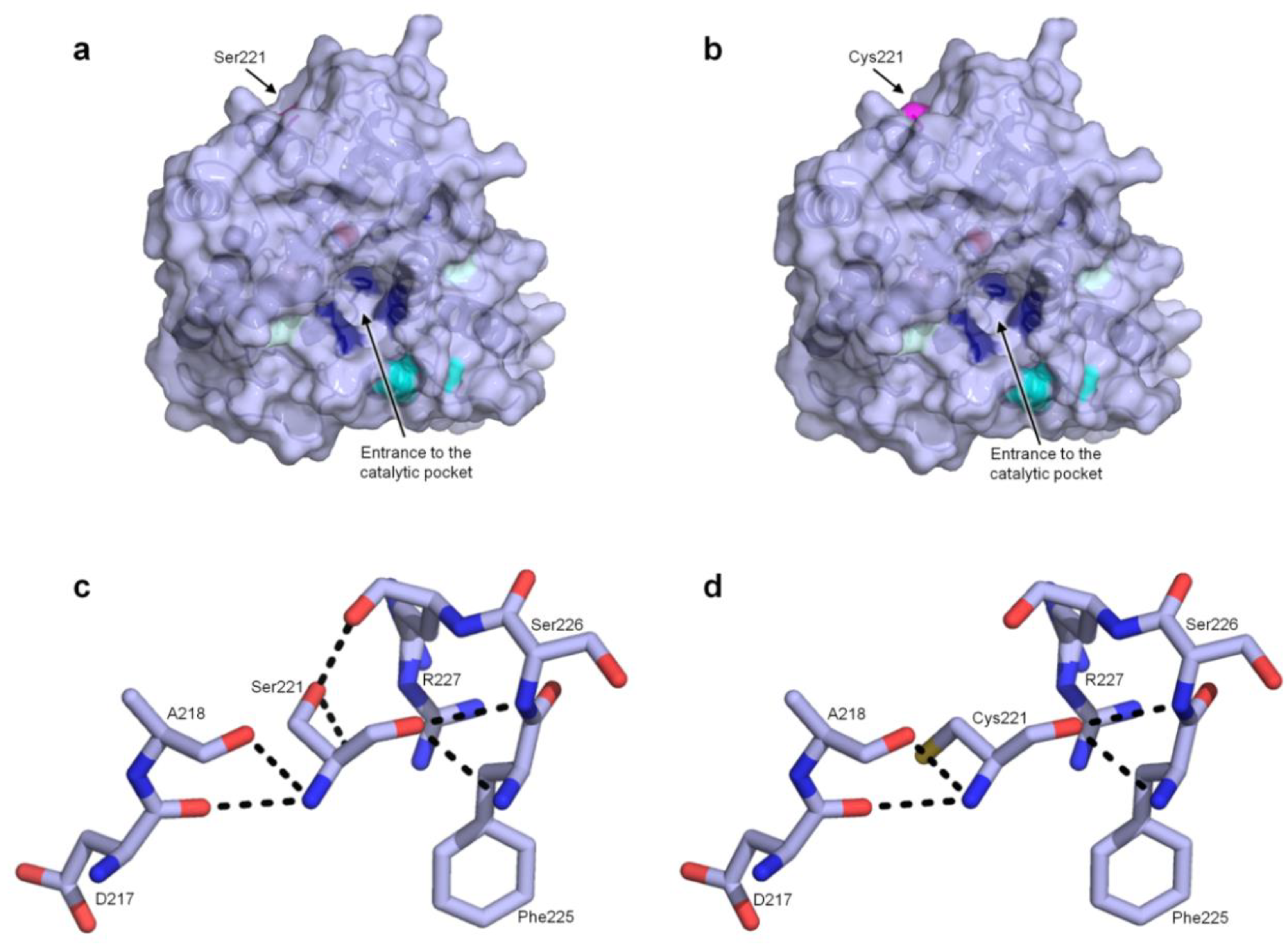
2.1. Unspecific Peroxygenase (UPO) Mutant Engineering (PaDa-I-Cys, S221C)
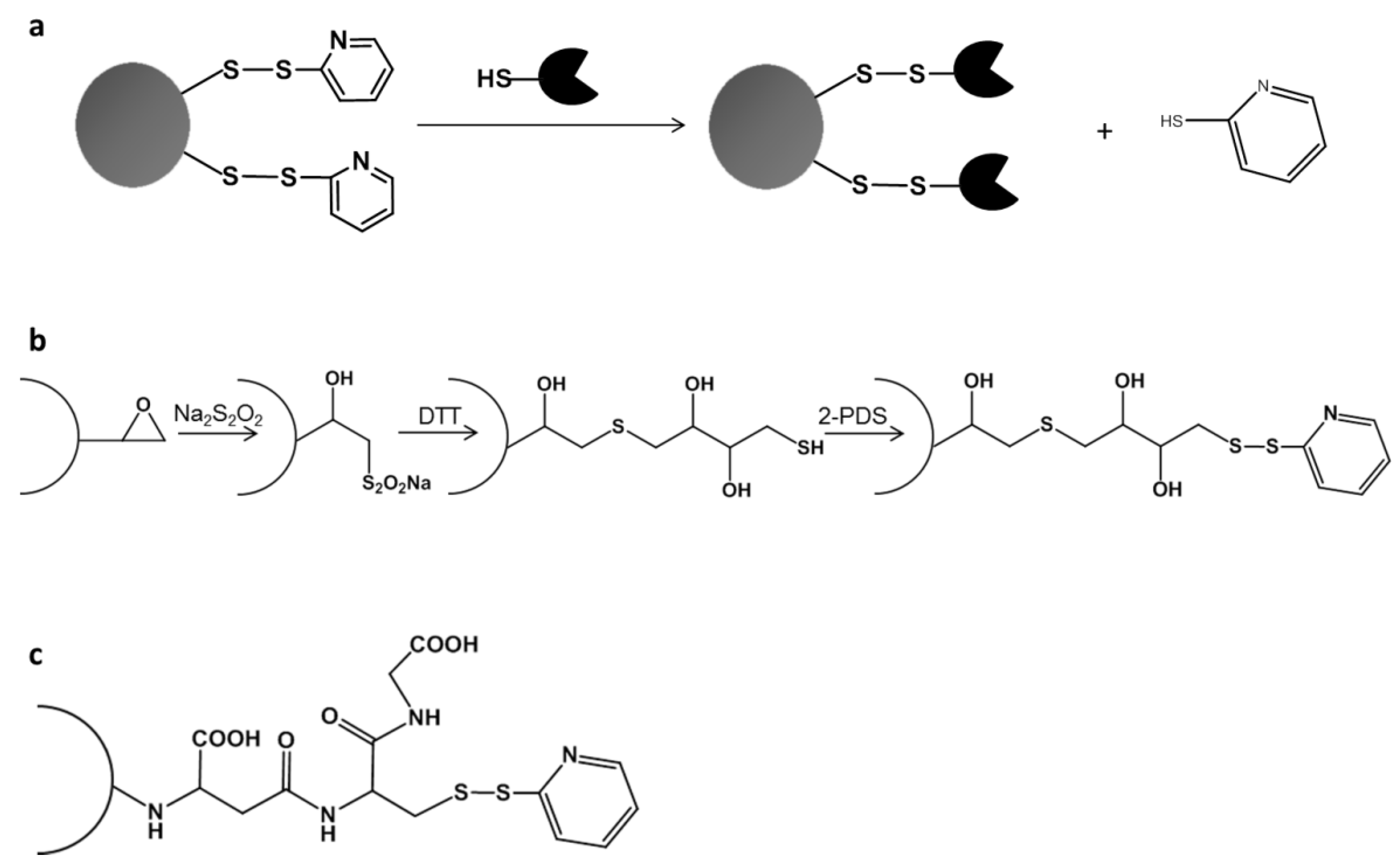
2.2. Directed Unique-Point Covalent Immobilization (DUCI) Strategy
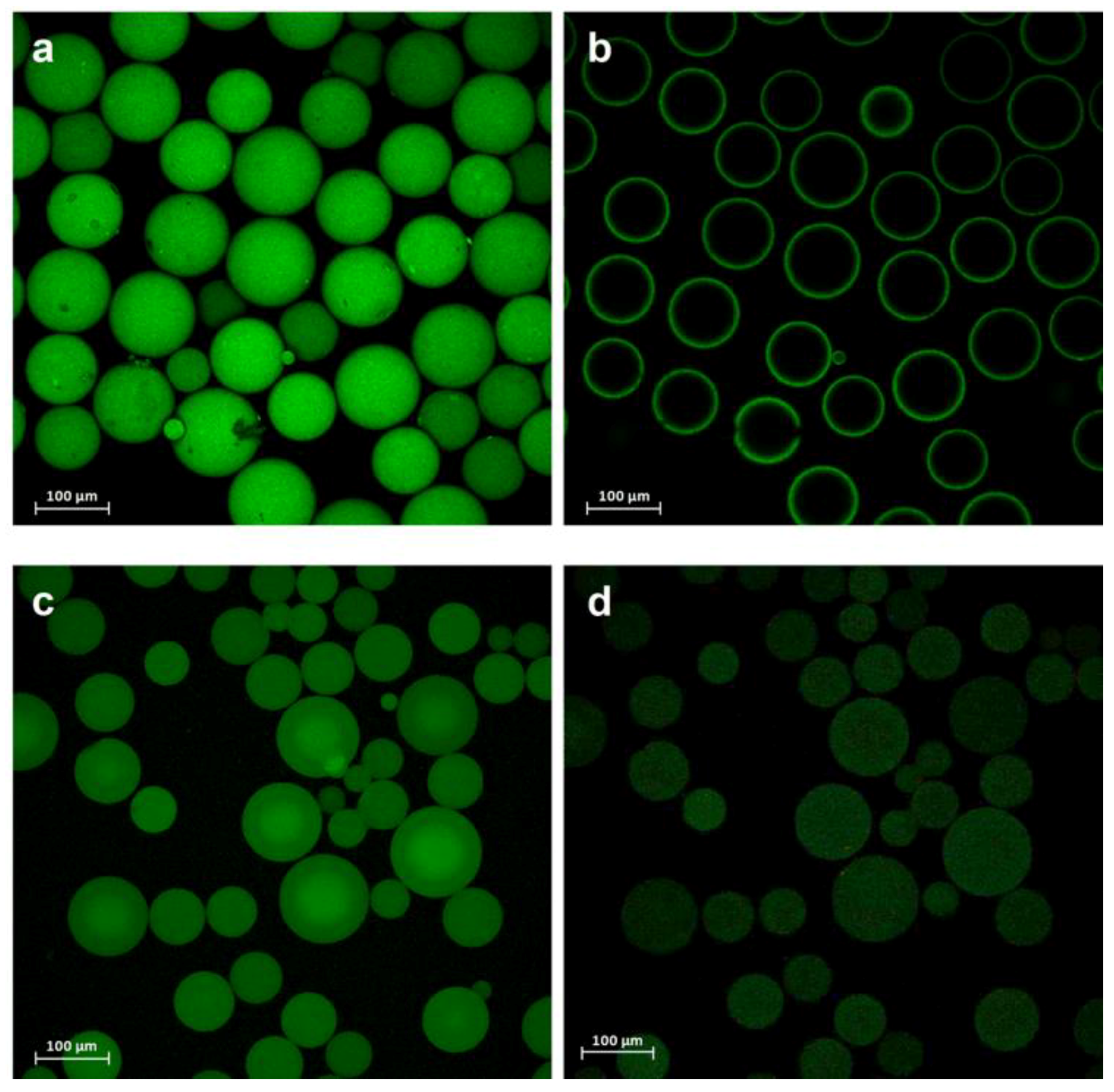
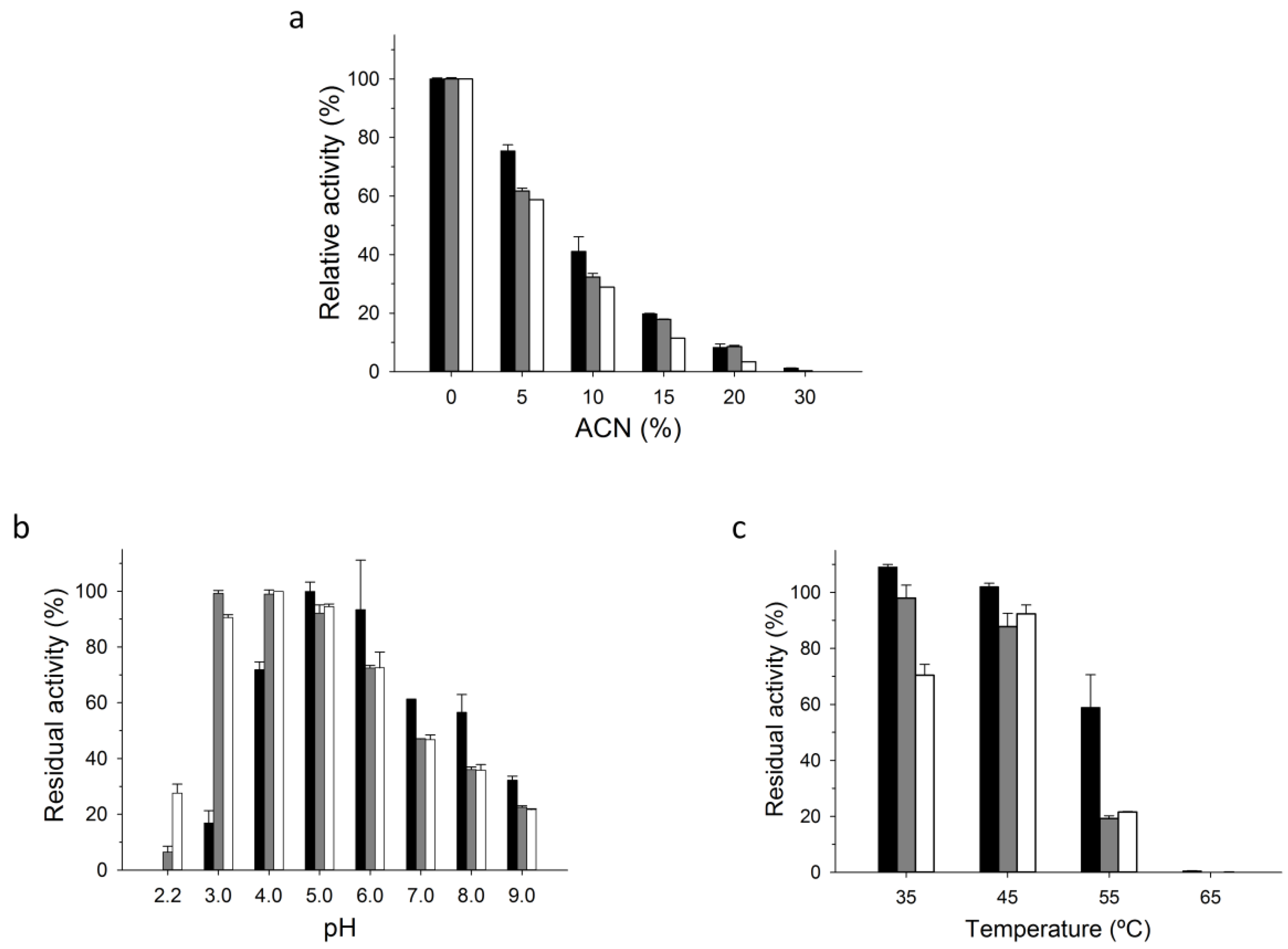
2.3. PaDa-I-Cys-DUCI Characterization
3. Materials and Methods
3.1. Reagents and Enzymes
3.2. Culture Media
3.3. Construction of the PaDa-I-Cys Variant
3.4. DNA Sequencing
3.5. Protein Modeling
3.6. Activation of Sepabeads with Disulfide Bonds
3.7. Directed Unique-Point Covalent Immobilization (DUCI)
3.8. Fluorescence Confocal Microscopy
3.9. Biochemical Characterization
3.10. Activity Colorimetric Assays
3.11. High-Performance Liquid Chromatography (HPLC) Analysis
4. Conclusions
5. Patents
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| 2-PDS | 2,2’-dithiodipyridine |
| ABTS | 2,2′-Azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) |
| ACN | Acetonitrile |
| AOBS | Acousto optical beam splitter |
| C-I | Sepabeads EC-EP203 |
| C-II | Thiol-Sepharose® |
| DTT | 1,4-Dithiothreitol |
| DUCI | Directed unique-point covalent immobilization |
| EDTA | Ethylenediaminetetraacetic acid |
| FITC | Fluorescein isothiocyanate |
| IVOE | In vivo overlap extension |
| NBD | 5-Nitro-1,3-benzodioxole |
| PaDa-I | Evolved mutant from the wild type UPO secreted by Agrocybe aegerita |
| PaDa-I-Cys | PaDa-I with the S221C mutation for DUCI immobilization |
| PDB | Protein data bank |
| UPO | Unspecific peroxygenase |
References
- Kakiuchi, F.; Chatani, N. Catalytic methods for C–H bond functionalization: Application in organic synthesis. Adv. Synth. Catal. 2003, 345, 1077–1101. [Google Scholar] [CrossRef]
- Bordeaux, M.; Galarneau, A.; Drone, J. Catalytic, mild, and selective oxyfunctionalization of linear alkanes: Current challenges. Angew. Chem. Int. Ed. 2012, 51, 10712–10723. [Google Scholar] [CrossRef]
- Munro, A.W.; Girvan, H.M.; Mason, A.E.; Dunford, A.J.; McLean, K.J. What makes a P450 tick? Trends Biochem. Sci. 2013, 38, 140–150. [Google Scholar] [CrossRef] [PubMed]
- Sakaki, T. Practical applications of cytochrome P450. Biol. Pharm. Bull. 2012, 35, 844–849. [Google Scholar] [CrossRef] [PubMed]
- Ullrich, R.; Hofrichter, M. Enzymatic hydroxylation of aromatic compounds. Cell. Mol. Life Sci. 2007, 64, 271–293. [Google Scholar] [CrossRef] [PubMed]
- Ullrich, R.; Nüske, J.; Scheibner, K.; Spanztzel, J.; Hofrichter, M. Novel haloperoxidase from the agaric basidiomycete Agrocybe aegerita oxidizes aryl alcohols and aldehydes. Appl. Environ. Microbiol. 2004, 70, 4575–4581. [Google Scholar] [CrossRef]
- Hofrichter, M.; Ullrich, R. Oxidations catalyzed by fungal peroxidases. Curr. Opin. Chem. Biol. 2014, 19, 116–125. [Google Scholar] [CrossRef] [PubMed]
- Hofrichter, M.; Ullrich, R. Heme-thiolate haloperoxidases: Versatile biocatalysts with biotechnological and environmental significance. Appl. Microbiol. Biotechnol. 2006, 71, 276–288. [Google Scholar] [CrossRef]
- Hofrichter, M.; Ullrich, R.; Pecyna, M.J.; Liers, C.; Lundell, T. New and classic families of secreted fungal heme peroxidases. Appl. Microbiol. Biotechnol. 2010, 87, 871–897. [Google Scholar] [CrossRef] [PubMed]
- Molina-Espeja, P.; Garcia-Ruiz, E.; Gonzalez-Perez, D.; Ullrich, R.; Hofrichter, M.; Alcalde, M. Directed Evolution of Unspecific Peroxygenase from Agrocybe aegerita. Appl. Environ. Microbiol. 2014, 80, 3496–3507. [Google Scholar] [CrossRef]
- Molina-Espeja, P.; Ma, S.; Mate, D.M.; Ludwig, R.; Alcalde, M. Tandem-yeast expression system for engineering and producing unspecific peroxygenase. Enzyme Microb. Technol. 2015, 73–74, 29–33. [Google Scholar] [CrossRef]
- Poraj-Kobielska, M.; Peter, S.; Leonhardt, S.; Ullrich, R.; Scheibner, K.; Hofrichter, M. Immobilization of unspecific peroxygenases (EC 1.11.2.1) in PVA/PEG gel and hollow fiber modules. Biochem. Eng. J. 2015, 98, 144–150. [Google Scholar] [CrossRef]
- Zhang, W.; Fernández-Fueyo, E.; Ni, Y.; van Schie, M.; Gacs, J.; Renirie, R.; Wever, R.; Mutti, F.G.; Rother, D.; Alcalde, M.; et al. Selective aerobic oxidation reactions using a combination of photocatalytic water oxidation and enzymatic oxyfunctionalizations. Nat. Catal. 2018, 1, 55–62. [Google Scholar] [CrossRef]
- Gómez de Segura, A.; Alcalde, M.; Yates, M.; Rojas-Cervantes, M.L.; López-Cortés, N.; Ballesteros, A.; Plou, F.J. Immobilization of dextransucrase from Leuconostoc mesenteroides NRRL B-512F on Eupergit C supports. Biotechol. Prog. 2004, 20, 1414–1420. [Google Scholar] [CrossRef]
- Berrio, J.; Plou, F.J.; Ballesteros, A.; Martinez, A.T.; Martinez, M.J. Immobilization of Pycnoporous coccineus laccase on Eupergit C: Stability increase and treatment of oil mill wastewaters. Biocatal. Biotransfor. 2007, 25, 130–134. [Google Scholar] [CrossRef]
- Kahar, U.M.; Sani, M.H.; Chan, K.G.; Goh, K.M. Immobilization of α-amylase from Anoxybacillus sp. SK3-4 on relizyme and immobead supports. Molecules 2016, 21, 1196. [Google Scholar] [CrossRef]
- Santos-Moriano, P.; Monsalve-Ledesma, L.; Ortega-Munoz, M.; Fernandez-Arrojo, L.; Ballesteros, A.O.; Santoyo-Gonzalez, F.; Plou, F.J. Vinyl sulfone-activated silica for efficient covalent immobilization of alkaline unstable enzymes: Application to levansucrase for fructooligosaccharide synthesis. RSC Adv. 2016, 6, 64175–64181. [Google Scholar] [CrossRef]
- Torres-Salas, P.; Monte-Martínez, A.; Cutiño-Avila, B.; Rodriguez-Colinas, B.; Alcalde, M.; Ballesteros, A.O.; Plou, F.J. Immobilized biocatalysts: Novel approaches and tools for binding enzymes to supports. Adv. Mater. 2011, 23, 5275–5282. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, K.; Fernandez-Lafuente, R. Control of protein immobilization: Coupling immobilization and site-directed mutagenesis to improve biocatalyst or biosensor performance. Enzyme Microb. Technol. 2011, 48, 107–122. [Google Scholar] [CrossRef] [PubMed]
- Simons, J.R.; Mosisch, M.; Torda, A.E.; Hilterhaus, L. Site directed immobilization of glucose-6-phosphate dehydrogenase via thiol-disulfide interchange: Influence on catalytic activity of cysteines introduced at different positions. J. Biotechnol. 2013, 167, 1–7. [Google Scholar] [CrossRef]
- Godoy, C.A.; Romero, O.; de las Rivas, B.; Mateo, C.; Fernandez-Lorente, G.; Guisan, J.M.; Palomo, J.M. Changes on enantioselectivity of a genetically modified thermophilic lipase by site-directed oriented immobilization. J. Mol. Catal. B Enzym. 2013, 87, 121–127. [Google Scholar] [CrossRef]
- Ramirez-Escudero, M.; Molina-Espeja, P.; Gomez de Santos, P.; Hofrichter, M.; Sanz-Aparicio, J.; Alcalde, M. Structural Insights into the Substrate Promiscuity of a Laboratory-Evolved Peroxygenase. ACS Chem. Biol. 2018, 13, 3259–3268. [Google Scholar] [CrossRef]
- Grazú, V.; Abian, O.; Mateo, C.; Batista-Viera, F.; Fernández-Lafuente, R.; Guisán, J.M. Novel bifunctional epoxy/thiol-reactive support to immobilize thiol containing proteins by the epoxy chemistry. Biomacromolecules 2003, 4, 1495–1501. [Google Scholar] [CrossRef] [PubMed]
- Torres, P.; Datla, A.; Rajasekar, V.W.; Zambre, S.; Ashar, T.; Yates, M.; Rojas-Cervantes, M.L.; Calero-Rueda, O.; Barba, V.; Martinez, M.J.; et al. Characterization and application of a sterol esterase immobilized on polyacrylate epoxy-activated carriers (DilbeadsTM). Catal. Commun. 2008, 9, 539–545. [Google Scholar] [CrossRef]
- Fernandez-Arrojo, L.; Rodriguez-Colinas, B.; Gutierrez-Alonso, P.; Fernandez-Lobato, M.; Alcalde, M.; Ballesteros, A.O.; Plou, F.J. Dried alginate-entrapped enzymes (DALGEEs) and their application to the production of fructooligosaccharides. Process Biochem. 2013, 48, 677–682. [Google Scholar] [CrossRef]
- Ghazi, I.; Gómez de Segura, A.; Fernández-Arrojo, L.; Alcalde, M.; Yates, M.; Rojas-Cervantes, M.L.; Plou, F.J.; Ballesteros, A. Immobilisation of fructosyltransferase from Aspergillus aculeatus on epoxy-activated Sepabeads EC for the synthesis of fructo-oligosaccharides. J. Mol. Catal. B Enzym. 2005, 35, 19–27. [Google Scholar] [CrossRef]
- Ullrich, R.; Hofrichter, M. The haloperoxidase of the agaric fungus Agrocybe aegerita hydroxylates toluene and naphthalene. FEBS Lett. 2005, 579, 6247–6250. [Google Scholar] [CrossRef] [PubMed]
- Kluge, M.; Ullrich, R.; Scheibner, K.; Hofrichter, M. Stereoselective benzylic hydroxylation of alkylbenzenes and epoxidation of styrene derivatives catalyzed by the peroxygenase of Agrocybe aegerita. Green Chem. 2012, 14, 440–446. [Google Scholar] [CrossRef]
- Willot, S.J.-P.; Fernandez-Fueyo, E.; Tieves, F.; Pesic, M.; Alcalde, M.; Arends, I.W.C.E.; Park, C.B.; Hollmann, F. Expanding the spectrum of light-driven peroxygenase reactions. ACS Catal. 2019, 9, 890–894. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Burek, B.O.; Fernandez-Fueyo, E.; Alcalde, M.; Bloh, J.Z.; Hollmann, F. Selective activation of C-H bonds by cascading photochemistry with biocatalysis. Angew. Chem. Int. Ed. 2017, 56, 15451–15455. [Google Scholar] [CrossRef]
- Alcalde, M. Mutagenesis protocols in Saccharomyces cerevisiae by In Vivo Overlap Extension. In Methods in Molecular Biology; Braman, J., Ed.; Humana Press: Totowa, NJ, USA, 2010; Volume 634, pp. 3–14. [Google Scholar] [CrossRef]
| PaDa-I | PaDa-I-Cys | |
|---|---|---|
| NBD/ABTS 1 | 0.14 | 0.16 |
| Total ABTS U in culture | 1673 | 1019 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Molina-Espeja, P.; Santos-Moriano, P.; García-Ruiz, E.; Ballesteros, A.; Plou, F.J.; Alcalde, M. Structure-Guided Immobilization of an Evolved Unspecific Peroxygenase. Int. J. Mol. Sci. 2019, 20, 1627. https://doi.org/10.3390/ijms20071627
Molina-Espeja P, Santos-Moriano P, García-Ruiz E, Ballesteros A, Plou FJ, Alcalde M. Structure-Guided Immobilization of an Evolved Unspecific Peroxygenase. International Journal of Molecular Sciences. 2019; 20(7):1627. https://doi.org/10.3390/ijms20071627
Chicago/Turabian StyleMolina-Espeja, Patricia, Paloma Santos-Moriano, Eva García-Ruiz, Antonio Ballesteros, Francisco J. Plou, and Miguel Alcalde. 2019. "Structure-Guided Immobilization of an Evolved Unspecific Peroxygenase" International Journal of Molecular Sciences 20, no. 7: 1627. https://doi.org/10.3390/ijms20071627
APA StyleMolina-Espeja, P., Santos-Moriano, P., García-Ruiz, E., Ballesteros, A., Plou, F. J., & Alcalde, M. (2019). Structure-Guided Immobilization of an Evolved Unspecific Peroxygenase. International Journal of Molecular Sciences, 20(7), 1627. https://doi.org/10.3390/ijms20071627








