Compositional Features of HDL Particles Interact with Albuminuria to Modulate Cardiovascular Disease Risk
Abstract
1. Introduction
2. Results
2.1. Study Group Characterization
2.2. CVD Risk Associations at Baseline Screening of UAE and HDL-Associated Parameters
2.3. Lipoprotein NMR Analyses
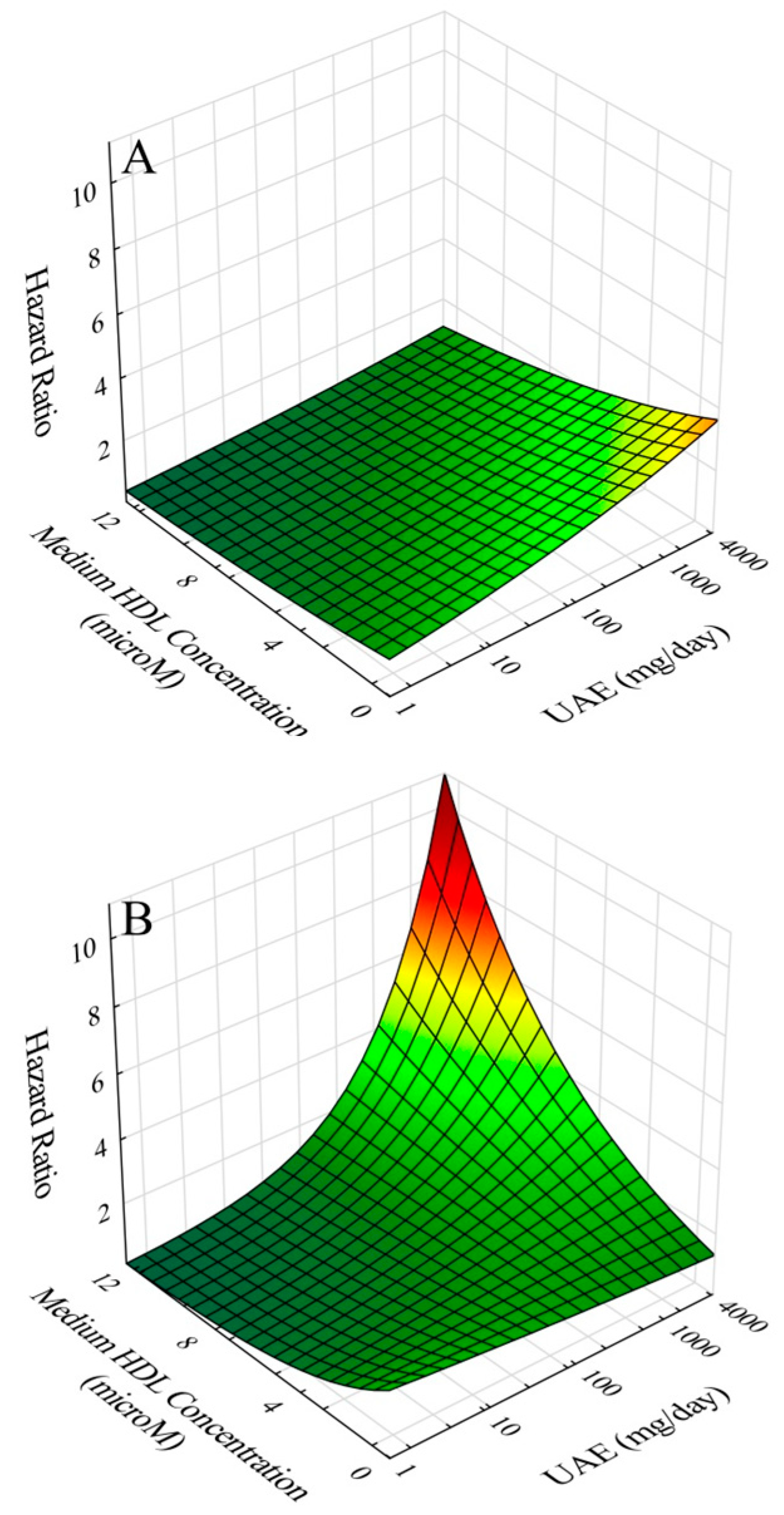
2.4. CVD Risk Associations at Second Screening of UAE and HDL-associated Parameters
2.5. Effects of Gender, Age, and Co-Morbidities on Outcomes and Interaction of UAE with ApoA-I/IHDL-P
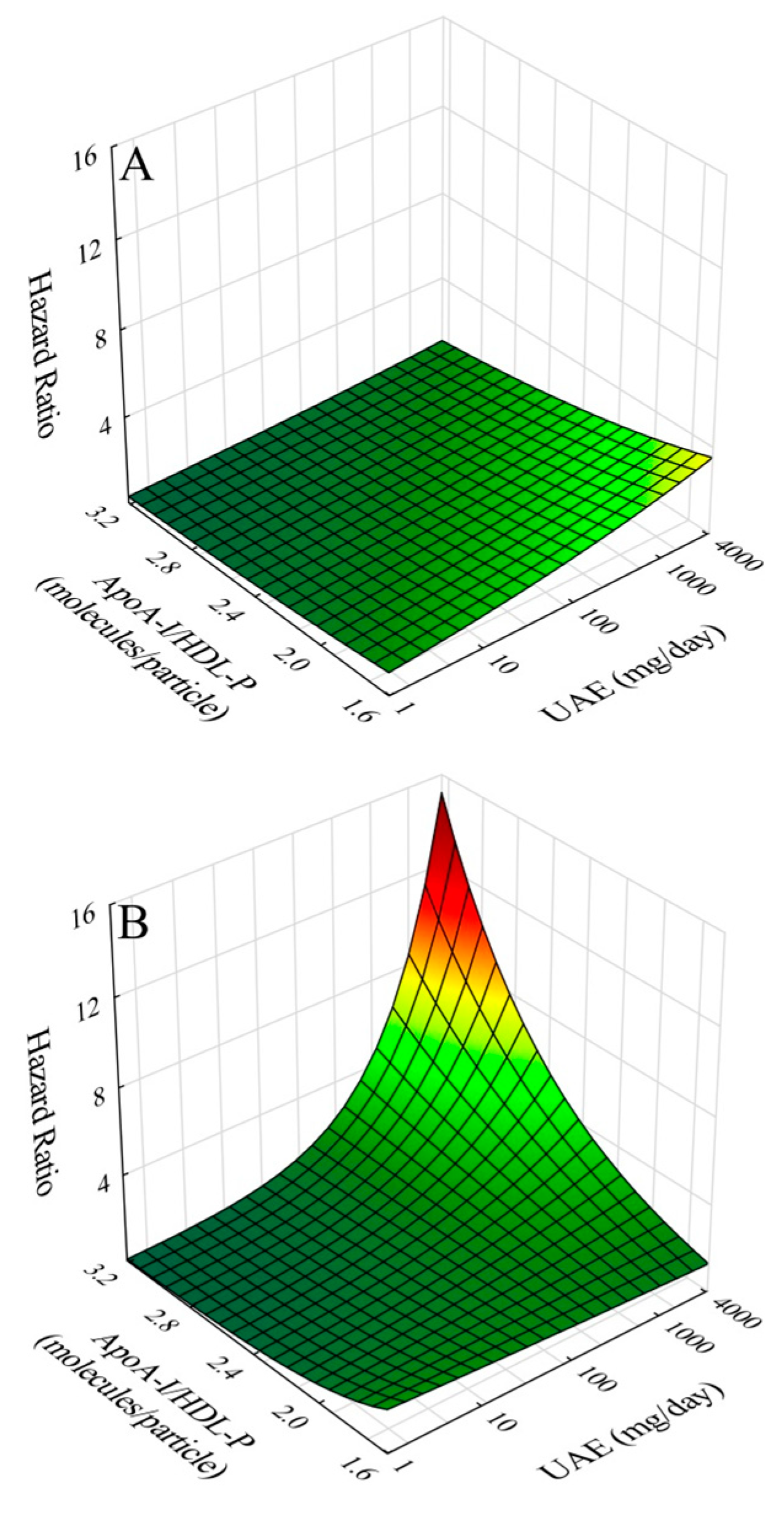
2.6. CVD Risk Relationships of ApoA-I/HDL-C and ApoA-I/IHDL-P
3. Discussion
4. Materials and Methods
4.1. Study Population
4.2. Clinical Parameters and Biomarkers
4.3. Outcome
4.4. Data Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A. Estimates of Mean Number of apoA-I Molecules per Particle and Mean apoA-I Concentrations of Large, Medium, and Small HDL Subfractions
Appendix B. Approach to Graphical Modeling of CVD Risk Contours without and with Interactions
Appendix C. Rationale for inverse correlation of apoA-I/HDL-P with apoA-I/HDL-C
References
- Stehouwer, C.D.A.; Smulders, Y.M. Microalbuminuria and risk for cardiovascular disease: Analysis of potential mechanisms. J. Am. Soc. Nephrol. 2006, 17, 2106–2111. [Google Scholar] [CrossRef] [PubMed]
- De Jong, P.E.; Gansevoort, R.T.; Bakker, S.J.L. Macroalbuminuria and microalbuminuria: Do both predict renal and cardiovascular events with similar strength. J. Nephrol. 2007, 20, 375–380. [Google Scholar] [PubMed]
- Smink, P.A.; Heerspink, H.J.L.; Gansevoort, R.T.; de Jong, P.E.; Hillege, H.L.; Bakker, S.J.; de Zeeuw, D. Albuminuria, estimated GFR. Traditional risk factors, and incident cardiovascular disease: The PREVEND (Prevention of Renal and Vascular Endstage Disease) Study. Am. J. Kidney Dis. 2012, 60, 804–811. [Google Scholar] [CrossRef] [PubMed]
- Hillege, H.L.; Janssen, W.M.; Bak, A.A.; Diercks, G.F.; Grobbee, D.E.; Crijns, H.J.; Van Gilst, W.H.; De Zeeuw, D.; De Jong, P.E.; Prevend Study Group. Microalbuminuria is common, also in a nondiabetic nonhypertensive population, and an independent indicator of cardiovascular risk factors and cardiovascular morbidity. J. Intern. Med. 2001, 249, 519–526. [Google Scholar] [CrossRef] [PubMed]
- Ordonez, J.D.; Hiatt, R.A.; Killebrew, E.J.; Fireman, B.H. The increased risk of coronary heart disease associated with nephrotic syndrome. Kidney Int. 1993, 44, 638–642. [Google Scholar] [CrossRef] [PubMed]
- Gerstein, H.C.; Mann, J.F.; Yi, Q.; Zinman, B.; Dinneen, S.F.; Hoogwerf, B.; Hallé, J.P.; Young, J.; Rashkow, A.; Joyce, C.; et al. Albuminuria and risk of cardiovascular events, death, and heart failure in diabetic and nondiabetic individuals. JAMA 2001, 286, 421–426. [Google Scholar] [CrossRef] [PubMed]
- Yuyun, M.F.; Khaw, K.T.; Luben, R.; Dinneen, S.F.; Hoogwerf, B.; Hallé, J.P.; Young, J.; Rashkow, A.; Joyce, C.; Nawaz, S.; et al. Microalbuminuria independently predicts all-cause and cardiovascular mortality in a British population: The European Prospective Investigation into Cancer in Norfolk (EPIC-Norfolk) population study. Int. J. Epidemiol. 2004, 33, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Tonelli, M.; Jose, P.; Curhan, G.; Sacks, F.; Braunwald, E.; Pfeffer, M. Proteinuria, impaired kidney function, and adverse outcomes in people with coronary disease: Analysis of a previously conducted randomized trial. BMJ 2006, 332, 1426. [Google Scholar] [CrossRef] [PubMed]
- de Boer, I.H.; Astor, B.C.; Kramer, H.; Palmas, W.; Rudser, K.; Seliger, S.L.; Shlipak, M.G.; Siscovick, D.S.; Tsai, M.Y.; Kestenbaum, B. Mild elevations of urine albumin excretion are associated with atherogenic lipoprotein abnormalities in the Multi-Ethnic Study of Atherosclerosis (MESA). Atherosclerosis 2008, 197, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Corsetti, J.P.; Gansevoort, R.T.; Bakker, S.J.L.; Sparks, C.E.; Vart, P.; Dullaart, R.P.F. Apolipoprotein B attenuates albuminuria-associated cardiovascular disease in Prevention of Renal and Vascular Endstage Disease (PREVEND) participants. J. Am. Soc. Nephrol. 2014, 25, 2906–2915. [Google Scholar] [CrossRef] [PubMed]
- Joven, J.; Villabona, C.; Vilella, E.; Masana, L.; Alberti, R.; Valles, M. Abnormalities of lipoprotein metabolism in patients with the nephritic syndrome. N. Engl. J. Med. 1990, 329, 579–584. [Google Scholar] [CrossRef] [PubMed]
- Warnick, G.L.; Caslake, M.J.; Boulton-Jones, J.M.; Dagen, M.; Packard, C.J.; Shepherd, J. Low-density lipoprotein metabolism in the nephritic syndrome. Metabolism 1990, 39, 187–192. [Google Scholar] [CrossRef]
- Warnick, G.L.; Packard, C.J.; Demant, T.; Bedford, D.K.; Boulton-Jones, J.M.; Shepherd, J. Metabolism of apolipoprotein B-containing lipoproteins in subjects with nephritic-range proteinuria. Kidney Int. 1991, 40, 129–138. [Google Scholar] [CrossRef]
- De Sain-van der Velden, M.G.; Kaysen, G.A.; Barrett, H.A.; Stellaard, F.; Gadellaa, M.M.; Voorbij, H.A.; Reijngoud, D.J.; Rabelink, T.J. Increased VLDL in nephritic patients results from a decreased catabolism while increased LDL results from increase synthesis. Kidney Int. 1998, 53, 994–1001. [Google Scholar] [CrossRef] [PubMed]
- Krikken, J.A.; Waanders, F.; Dallinga-Thie, G.M.; Dikkeschei, L.D.; Vogt, L.; Navis, G.J.; Dullaart, R.P. Antiproteinuric therapy decreases LDL-cholesterol as well as HDL-cholesterol in non-diabetic proteinuric patients: Relationships with cholesteryl ester transfer protein mass and adiponectin. Expert Opin. Ther. Targets 2009, 13, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Vaziri, N.D. HDL abnormalities in nephrotic syndrome and chronic kidney disease. Nat. Rev. Nephrol. 2016, 12, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Vaziri, N.D.; Liang, K.; Parks, J.S. Acquired lecithin:cholesterol acyltransferase (LCAT) deficiency in nephrotic syndrome. Am. J. Physiol. 2001, 49, F825–F829. [Google Scholar]
- Dullaart, R.P.F.; Gansevoort, R.T.; Dikkeschei, B.D.; de Zeeuw, D.; de Jong, P.E.; van Tol, A. Role of elevated lecithin: Cholesterol acyltransferase and cholesteryl ester transfer protein activities in ab normal lipoprotein from proteinuric patients. Kidney Int. 1993, 44, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Marcel, Y.L. Serum albumin is a significant intermediate in cholesterol transfer between cells and lipoproteins. Biochemistry 1996, 35, 7174–7180. [Google Scholar] [CrossRef] [PubMed]
- Moulin, P.; Appel, G.B.; Ginsberg, H.N.; Tall, A.R. Increased concentration of plasma cholesteryl ester transfer protein in nephrotic syndrome: Role in dyslipidemia. J. Lipid Res. 1992, 33, 1817–1822. [Google Scholar] [PubMed]
- Liang, K.; Vaziri, N.D. Downregulation of hepatic high-sensitivity lipoprotein receptor SR-B1 in nephrotic syndrome. Kidney Int. 1999, 56, 621–626. [Google Scholar] [CrossRef] [PubMed]
- Vaziri, N.D.; Gollapudi, P.; Han, S.; Farahmand, G.; Yuan, J.; Rahimi, A.; Moradi, H. Nephrotic syndrome causes upregulation of hepatic HDL endocytic receptor and PDZK-1 dependent downregulation of HDL docking receptor. Nephrol. Dial. Transplant. 2011, 26, 3118–3123. [Google Scholar] [CrossRef] [PubMed]
- Moradi, H.; Pahl, M.V.; Elahimehr, R.; Vaziri, N.D. Impaired antioxidant activity of high-density lipoprotein in chronic kidney disease. Transl. Res. 2900, 153, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Moradi, H.; Vaziri, N.D.; Kashyap, M.L.; Said, H.M.; Kalantar-Zadeh, K. Role of HDL dysfunction in end-stage renal disease: A double-edged sword. J. Ren. Nutr. 2013, 23, 203–206. [Google Scholar] [CrossRef] [PubMed]
- Vaziri, N.D. Role of dyslipisemia in impairment of energy metabolism, oxidative stress, inflammation and cardiovascular disease in chronic kidney disease. Clin. Exp. Nephrol. 2014, 18, 265–268. [Google Scholar] [CrossRef] [PubMed]
- Muls, E.; Rosseneu, M.; Daneels, R.; Schurgers, M.; Boelaert, J. Lipoprotein distribution and composition in the human nephrotic syndrome. Atherosclerosis 1985, 54, 225–237. [Google Scholar] [CrossRef]
- Pinto-Sietsma, S.J.; Janssen, W.M.; Hillege, H.L.; Navis, G.; De Zeeuw, D.; De Jong, P.E. Urinary albumin excretion is associated with renal functional abnormalities in a nondiabetic population. J. Am. Soc. Nephrol. 2000, 11, 1882–1888. [Google Scholar] [PubMed]
- Kappelle, P.J.W.H.; Gansevoort, R.T.; Hillege, J.L.; Wolffenbuttel, B.H.R.; Dullaart, R.P.F. Apolipoprotein B/A-I and total cholesterol/high-density lipoprotein cholesterol ratios both predict cardiovascular events in the general population independently of nonlipid risk factors, albuminuria and C-reactive protein. J. Intern. Med. 2011, 269, 232–242. [Google Scholar] [CrossRef] [PubMed]
- de Greeff, A.; Reggiori, F.; Shennan, A.H. Clinical assessment of the DINAMAP ProCare monitor in an adult population according to the British Hypertension Society Protocol. Blood Press. Monit. 2007, 12, 51–55. [Google Scholar] [CrossRef] [PubMed]
- Borggreve, S.E.; Hillege, H.L.; Wolffenbuttel, B.H.R.; de Jong, P.E.; Zuurman, M.W.; van der Steege, G.; van Tol, A.; Dullaart, R.P.; PREVEND Study Group. An increased coronary risk is paradoxically associated with common cholesteryl ester transfer protein gene variations that relate to higher high-density lipoprotein cholesterol: A population-based study. J. Clin. Endocrinol. Metab. 2006, 91, 3382–3388. [Google Scholar] [CrossRef] [PubMed]
- Corsetti, J.P.; Gansevoort, R.T.; Sparks, C.E.; Dullaart, R.P.F. HDL protection against primary cardiac risk is lost with inflammation. Eur. J. Clin. Investig. 2010, 40, 483–489. [Google Scholar] [CrossRef] [PubMed]
- Levey, A.S.; Stevens, L.A.; Schmid, C.H.; Zhang, Y.L.; Castro, A.F., 3rd; Feldman, H.I.; Kusek, J.W.; Eggers, P.; Van Lente, F.; Greene, T.; et al. A new equation to estimate glomerular filtration rate. Ann Intern Med 2009, 150, 604–612. [Google Scholar] [CrossRef] [PubMed]
- Jeyarajah, E.J.; Cromwell, W.C.; Otvos, J.D. Lipoprotein particle analysis by nuclear magnetic resonance spectroscopy. Clin. Lab. Med. 2006, 26, 847–870. [Google Scholar] [CrossRef] [PubMed]
- Matyus, S.P.; Braun, P.J.; Wolak-Dinsmore, J.; Jeyarajah, E.J.; Shalaurova, I.; Xu, Y.; Warner, S.M.; Clement, T.S.; Connelly, M.A.; Fischer, T.J. NMR measurement of LDL particle number using the Vantera Clinical Analyzer. Clin. Biochem. 2014, 47, 203–210. [Google Scholar]
| Parameter | Total Population | Normoalbuminuria (86.1% of Subjects) | Microalbuminuria | Macroalbuminuria | pa | pb | pc |
|---|---|---|---|---|---|---|---|
| Subjects (%) | N = 6286 | 87.4 | 11.5 | 1.1 | |||
| Age (yrs) | 48.7 ± 12.0 | 47.8 ± 11.8 | 54.6 ± 11.8 | 57.3 ± 12.4 | <0.001 | ||
| Females (%) | 51.0 | 53.2 | 36.8 | 29.6 | <0.001 | ||
| Cardiac history (%) | 3.0 | 2.1 | 7.2 | 10.3 | <0.001 | ||
| Diabetes (%) | 2.7 | 1.7 | 8.4 | 18.8 | <0.001 | ||
| Statins (%) | 3.7 | 3.0 | 7.3 | 13.2 | <0.001 | ||
| Anti-hypertensives (%) | 14.6 | 12.7 | 25.6 | 37.9 | <0.001 | ||
| Current Smoker (%) | 31.9 | 31.6 | 35.4 | 18.6 | 0 | ||
| Ethanol Use (%) | 25.5 | 25.2 | 27.4 | 24.3 | 0.46 | ||
| BMI (kg/m2) | 26.0 ± 4.1 | 25.7 ± 3.9 | 27.7 ± 4.6 | 29.5 ± 4.5 | <0.001 | <0.001 | <0.001 |
| Systolic BP (mmHg) | 128 ± 19 | 126 ± 18 | 141 ± 23 | 151 ± 24 | <0.001 | <0.001 | <0.001 |
| Diastolic BP (mmHg) | 74 ± 10 | 73 ± 9 | 79 ± 11 | 82 ± 10 | <0.001 | <0.001 | <0.001 |
| UAE (mg/24 h) | 8.93 (6.19–15.61) | 8.09 (5.92–11.93) | 53.0 (38.0–87.1) | 495 (347–1077) | <0.001 | <0.001 | <0.001 |
| Creatinine (μmol/L) | 83.4 ± 14.3 | 82.4 ± 13.3 | 88.2 ± 18.3 | 106.9 ± 33.9 | <0.001 | <0.001 | <0.001 |
| eGFR (mL/min/1.7 m2) | 84.4 ± 15.0 | 85.2 ± 14.5 | 79.7 ± 16.5 | 67.7 ± 20.2 | <0.001 | <0.001 | <0.001 |
| HDL-C (mmol/L) | 1.33 ± 0.40 | 1.35 ± 0.40 | 1.21 ± 0.38 | 1.15 ± 0.31 | <0.001 | <0.001 | <0.001 |
| ApoA-I (μmol/L) | 48.0 ± 9.8 | 48.2 ± 9.8 | 46.9 ± 9.8 | 45.8 ± 9.9 | <0.001 | 0.32 | 0.58 |
| ApoA-II (μmol/L) | 19.7 ± 3.7 | 19.7 ± 3.7 | 19.4 ± 3.9 | 18.1 ± 3.6 | <0.001 | 0.013 | 0.027 |
| ApoA-I/HDL-C (μmol/mmol) | 37.6 ± 10.2 | 37.1 ± 7.9 | 40.7 ± 9.1 | 41.7 ± 10.2 | <0.001 | <0.001 | <0.001 |
| ApoA-II/HDL-C (μmol/mmol) | 15.8 ± 4.6 | 15.6 ± 4.5 | 17.2 ± 5.1 | 16.7 ± 4.9 | <0.001 | <0.001 | <0.001 |
| Cholesterol (mmol/L) | 5.62 ± 1.13 | 5.58 ± 1.12 | 5.89 ± 1.13 | 6.09 ± 1.35 | <0.001 | 0.001 | 0.24 |
| NonHDL-C (mmol/L) | 4.28 ± 1.20 | 4.22 ± 1.19 | 4.67 ± 1.21 | 4.94 ± 1.39 | <0.001 | <0.001 | 0.005 |
| LDL-C (mmol/L) | 3.66 ± 1.04 | 3.62 ± 1.03 | 3.89 ± 1.04 | 3.98 ± 1.07 | <0.001 | 0.18 | 0.40 |
| Triglycerides (mmol/L) | 1.14 (0.83–1.66) | 1.11 (0.81–1.60) | 1.43 (1.00–2.20) | 1.49 (1.07–2.77) | <0.001 | <0.001 | <0.001 |
| ApoB (g/L) | 1.03 ± 0.30 | 1.01 ± 0.29 | 1.12 ± 0.32 | 1.15 ± 0.30 | <0.001 | <0.001 | 0.024 |
| Glucose (mmol/L) | 4.83 ± 1.08 | 4.75 ± 0.87 | 5.37 ± 1.81 | 5.80 ± 2.70 | <0.001 | <0.001 | <0.001 |
| CRP (mg/L) | 1.18 (0.53–2.77) | 1.07 (0.49–2.55) | 2.08 (0.96–4.32) | 2.57 (1.25–6.24) | <0.001 | <0.001 | <0.001 |
| Models | Model without Interaction | Model with Interaction | ||||
|---|---|---|---|---|---|---|
| Parameters | HR | 95% CI | p | HR | 95% CI | p |
| UAE | 1.11 | 1.00–1.23 | 0.036 | 1.16 | 1.02–1.31 | 0.023 |
| HDL-C | 0.62 | 0.53–0.74 | <10−6 | 0.61 | 0.51–0.73 | <10−6 |
| interaction | 1.07 | 0.94–1.22 | 0.34 | |||
| UAE | 1.15 | 1.04–1.27 | 0.005 | 1.16 | 1.04–1.29 | 0.006 |
| ApoA-I | 0.77 | 0.67–0.88 | <10−3 | 0.76 | 0.66–0.88 | <10−3 |
| interaction | 1.02 | 0.93–1.11 | 0.72 | |||
| UAE | 1.16 | 1.05–1.28 | 0.003 | 1.16 | 1.05–1.29 | 0.004 |
| ApoA-II | 0.89 | 0.78–1.01 | 0.067 | 0.88 | 0.77–1.01 | 0.068 |
| interaction | 1.01 | 0.93–1.11 | 0.76 | |||
| UAE | 1.11 | 1.01–1.23 | 0.036 | 1.17 | 1.05–1.30 | 0.003 |
| ApoA-I/HDL-C | 1.26 | 1.14–1.39 | <10−5 | 1.32 | 1.19–1.47 | <10−6 |
| interaction | 0.91 | 0.84–0.99 | 0.019 | |||
| UAE | 1.11 | 1.01–1.23 | 0.035 | 1.15 | 1.04–1.28 | 0.009 |
| ApoA-II/HDL-C | 1.34 | 1.20–1.51 | <10−6 | 1.40 | 1.24–1.58 | <10−6 |
| interaction | 0.93 | 0.85–1.01 | 0.10 | |||
| Models | Model without Interaction | Model with Interaction | ||||
|---|---|---|---|---|---|---|
| Parameters | HR | 95% CI | p | HR | 95% CI | p |
| UAE | 1.04 | 0.94–1.16 | 0.46 | 1.10 | 0.97–1.26 | 0.14 |
| HDL-C | 0.74 | 0.61–0.88 | <10−3 | 0.72 | 0.59–0.86 | <10−3 |
| interaction | 1.10 | 0.96–1.26 | 0.16 | |||
| UAE | 1.07 | 0.96–1.19 | 0.21 | 1.07 | 0.95–1.19 | 0.26 |
| ApoA-I | 0.73 | 0.62–0.85 | <10−4 | 0.73 | 0.662–0.86 | <10−3 |
| interaction | 1.00 | 0.89–1.12 | 0.96 | |||
| UAE | 1.06 | 0.96–1.18 | 0.26 | 1.07 | 0.96–1.19 | 0.22 |
| ApoA-II | 0.76 | 0.65–0.89 | <10−3 | 0.75 | 0.64–0.89 | <10−3 |
| interaction | 1.03 | 0.92–1.14 | 0.63 | |||
| UAE | 1.04 | 0.94–1.16 | 0.44 | 1.11 | 0.99–1.25 | 0.078 |
| ApoA-I/HDL-C | 1.10 | 0.96–1.26 | 0.16 | 1.16 | 1.01–1.34 | 0.036 |
| interaction | 0.90 | 0.81–0.99 | 0.025 | |||
| UAE | 1.05 | 0.94–1.16 | 0.41 | 1.07 | 0.96–1.20 | 0.23 |
| ApoA-II/HDL-C | 1.16 | 1.00–1.34 | 0.52 | 1.19 | 1.02–1.39 | 0.029 |
| interaction | 0.95 | 0.86–1.05 | 0.29 | |||
| Parameter | Total Population | Normoalbuminuria (86.1% of subjects) | Microalbuminuria | Macroalbuminuria | pa | pb | pc |
|---|---|---|---|---|---|---|---|
| HDL-P (μmol/L) | 21.14 ± 2.74 | 21.23 ± 2.71 | 20.57 ± 2.83 | 20.40 ± 3.23 | <0.001 | <0.001 | 0.003 |
| Large HDL-P (μmol/L) | 1.81 ± 1.29 | 1.86 ± 1.30 | 1.50 ± 1.21 | 1.49 ± 1.16 | <0.001 | 0.001 | 0.009 |
| Medium HDL-P (μmol/L) | 5.15 ± 2.19 | 5.25 ± 2.19 | 4.59 ± 2.11 | 4.06 ± 2.09 | <0.001 | <0.001 | 0.18 |
| Small HDL-P (μmol/L) | 14.18 ± 2.87 | 14.13 ± 2.88 | 14.48 ± 2.75 | 14.85 ± 3.06 | <0.001 | 0.65 | 0.53 |
| HDL Size (nm) | 8.97 ± 0.45 | 8.99 ± 0.45 | 8.84 ± 0.45 | 8.82 ± 0.45 | <0.001 | <0.001 | 0.005 |
| HDL-C (mmol/L) | 1.33 ± 0.32 | 1.34 ± 0.31 | 1.23 ± 0.32 | 1.21 ± 0.34 | <0.001 | <0.001 | 0.009 |
| ApoA-I (μmol/L) | 45.1 ± 7.8 | 45.4 ± 7.7 | 43.0 ± 7.9 | 42.5 ± 8.6 | <0.001 | <0.001 | 0.023 |
| ApoA-I/HDL-C (μmol/mmol) | 34.5 ± 2.9 | 34.4 ± 2.9 | 35.5 ± 3.0 | 35.9 ± 3.1 | <0.001 | <0.001 | <0.001 |
| ApoA-I/HDL-P (molecules/particle) | 2.13 ± 0.21 | 2.14 ± 0.21 | 2.09 ± 0.20 | 2.08 ± 0.21 | <0.001 | 0.25 | 0.38 |
| Models | Model without Interaction | Model with Interaction | ||||
|---|---|---|---|---|---|---|
| Parameters | HR | 95% CI | p | HR | 95% CI | p |
| UAE | 1.21 | 1.12–1.32 | <10−5 | 1.23 | 1.12–1.34 | <10−5 |
| HDL-P | 0.83 | 0.75–0.93 | 0.001 | 0.82 | 0.73–0.93 | 0.004 |
| interaction | 1.02 | 0.95–1.10 | 0.56 | |||
| UAE | 1.21 | 1.11–1.31 | <10−5 | 1.23 | 1.13–1.34 | <10−5 |
| Large HDL-P | 0.74 | 0.64–0.85 | <10−4 | 0.72 | 0.62–0.84 | <10−4 |
| interaction | 1.05 | 0.96–1.16 | 0.30 | |||
| UAE | 1.20 | 1.10–1.30 | <10−4 | 1.28 | 1.16–1.40 | <10−5 |
| Medium HDL-P | 0.74 | 0.65–0.84 | <10−5 | 0.70 | 0.61–0.81 | <10−5 |
| interaction | 1.11 | 1.01–1.22 | 0.029 | |||
| UAE | 1.22 | 1.13–1.33 | <10−5 | 1.24 | 1.14–1.35 | <10−6 |
| Small HDL-P | 1.11 | 0.99–1.24 | 0.07 | 1.15 | 1.02–1.30 | 0.024 |
| interaction | 0.93 | 0.86–1.01 | 0.10 | |||
| UAE | 1.20 | 1.10–1.30 | <10−4 | 1.24 | 1.13–1.36 | <10−5 |
| HDL Size | 0.70 | 0.61–0.80 | <10−6 | 0.67 | 0.58–0.77 | <10−6 |
| interaction | 1.08 | 0.99–1.18 | 0.10 | |||
| UAE | 1.19 | 1.10–1.29 | <10−4 | 1.29 | 1.14–1.37 | <10−5 |
| HDL-C | 0.69 | 0.60–0.79 | <10−6 | 0.66 | 0.57–0.76 | 0.004 |
| interaction | 1.08 | 1.00–1.18 | 0.06 | |||
| UAE | 1.20 | 1.11–1.30 | <10−4 | 1.24 | 1.14–1.37 | <10−5 |
| ApoA-I | 0.74 | 0.65–0.84 | <10−5 | 0.71 | 0.62–0.81 | <10−5 |
| interaction | 1.07 | 0.99–1.16 | 0.11 | |||
| UAE | 1.19 | 1.09–1.29 | <10−4 | 1.23 | 1.12–1.35 | <10−5 |
| ApoA-I/HDL-C | 1.37 | 1.22–1.55 | <10−6 | 1.43 | 1.25–1.62 | <10−6 |
| interaction | 0.94 | 0.86–1.02 | 0.11 | |||
| UAE | 1.21 | 1.11–1.31 | <10−5 | 1.26 | 1.15–1.37 | <10−6 |
| ApoA-I/HDL-P | 0.74 | 0.65–0.85 | <10−4 | 0.71 | 0.61–0.82 | <10−5 |
| interaction | 1.10 | 1.00–1.20 | 0.039 | |||
| Models | Model without Interaction | Model with Interaction | ||||
|---|---|---|---|---|---|---|
| Parameters | HR | 95% CI | p | HR | 95% CI | p |
| UAE | 1.13 | 1.05–1.24 | 0.003 | 1.16 | 1.076–1.27 | 0.002 |
| HDL-P | 0.88 | 0.79–0.99 | 0.030 | 0.86 | 0.77–0.968 | 0.018 |
| interaction | 1.04 | 0.96–1.12 | 0.36 | |||
| UAE | 1.14 | 1.05–1.24 | 0.003 | 1.16 | 1.06–1.27 | 0.001 |
| Large HDL-P | 0.86 | 0.74–1.01 | 0.060 | 0.84 | 0.72–0.99 | 0.037 |
| interaction | 1.05 | 0.96–1.16 | 0.28 | |||
| UAE | 1.13 | 1.04–1.243 | 0.006 | 1.21 | 1.09–1.33 | <10−3 |
| Medium HDL-P | 0.801 | 0.71–0.92 | 0.002 | 0.76 | 0.66–0.88 | <10−3 |
| interaction | 1.12 | 1.02–1.23 | 0.018 | |||
| UAE | 1.14 | 1.05–1.24 | 0.003 | 1.15 | 1.06–1.26 | 0.001 |
| Small HDL-P | 1.05 | 0.93–1.18 | 0.44 | 1.08 | 0.95–1.23 | 0.24 |
| interaction | 0.94 | 0.87–1.03 | 0.19 | |||
| UAE | 1.13 | 1.04–1.24 | 0.004 | 1.17 | 1.07–1.29 | <10−3 |
| HDL Size | 0.80 | 0.69–0.92 | 0.002 | 0.77 | 0.66–0.90 | <10−3 |
| interaction | 1.07 | 0.98–1.17 | 0.14 | |||
| UAE | 1.13 | 1.04–1.24 | 0.004 | 1.18 | 1.08–1.31 | <10−3 |
| HDL-C | 0.79 | 0.68–0.91 | 0.002 | 0.76 | 0.65–0.88 | <10−3 |
| interaction | 1.08 | 1.00–1.187 | 0.049 | |||
| UAE | 1.14 | 1.04–1.24 | 0.003 | 1.18 | 1.08–1.30 | <10−3 |
| ApoA-I | 0.83 | 0.73–0.95 | 0.006 | 0.80 | 0.70–0.92 | 0.002 |
| interaction | 1.08 | 0.99–1.17 | 0.08 | |||
| UAE | 1.13 | 1.04–1.23 | 0.006 | 1.17 | 1.06–1.29 | 0.002 |
| ApoA-I/HDL-C | 1.23 | 1.08–1.40 | 0.002 | 1.27 | 1.10–1.45 | <10−3 |
| interaction | 0.94 | 0.87–1.03 | 0.18 | |||
| UAE | 1.14 | 1.05–1.24 | 0.003 | 1.18 | 1.08–1.30 | <10−3 |
| ApoA-I/HDL-P | 0.86 | 0.74–1.00 | 0.044 | 0.81 | 0.69–0.95 | 0.011 |
| interaction | 1.09 | 1.00–1.20 | 0.048 | |||
| Parameters | HR | 95% CI | p |
|---|---|---|---|
| Gender | 2.16 | 1.65–2.83 | <10−6 |
| Age | 1.85 | 1.59–2.14 | <10−6 |
| ApoB Concentration | 1.22 | 1.08–1.37 | <10−3 |
| Diabetes | 1.61 | 1.08–2.40 | 0.020 |
| Past CVD History | 2.27 | 1.64–3.15 | <10−5 |
| CRP Concentration | 1.18 | 1.03–1.34 | 0.013 |
| eGFR | 0.88 | 0.78–1.00 | 0.053 |
| UAE | 1.18 | 1.08–1.30 | <10−3 |
| ApoA-I/HDL-C | 0.81 | 0.69–0.95 | 0.011 |
| UAE x ApoA-I/HDL-P | 1.09 | 1.00–1.20 | 0.048 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Corsetti, J.P.; Bakker, S.J.L.; Gansevoort, R.T.; Gruppen, E.G.; Connelly, M.A.; Sparks, C.E.; Dullaart, R.P.F. Compositional Features of HDL Particles Interact with Albuminuria to Modulate Cardiovascular Disease Risk. Int. J. Mol. Sci. 2019, 20, 977. https://doi.org/10.3390/ijms20040977
Corsetti JP, Bakker SJL, Gansevoort RT, Gruppen EG, Connelly MA, Sparks CE, Dullaart RPF. Compositional Features of HDL Particles Interact with Albuminuria to Modulate Cardiovascular Disease Risk. International Journal of Molecular Sciences. 2019; 20(4):977. https://doi.org/10.3390/ijms20040977
Chicago/Turabian StyleCorsetti, James P., Stephan J. L. Bakker, Ronald T. Gansevoort, Eke G. Gruppen, Margery A. Connelly, Charles E. Sparks, and Robin P. F. Dullaart. 2019. "Compositional Features of HDL Particles Interact with Albuminuria to Modulate Cardiovascular Disease Risk" International Journal of Molecular Sciences 20, no. 4: 977. https://doi.org/10.3390/ijms20040977
APA StyleCorsetti, J. P., Bakker, S. J. L., Gansevoort, R. T., Gruppen, E. G., Connelly, M. A., Sparks, C. E., & Dullaart, R. P. F. (2019). Compositional Features of HDL Particles Interact with Albuminuria to Modulate Cardiovascular Disease Risk. International Journal of Molecular Sciences, 20(4), 977. https://doi.org/10.3390/ijms20040977






