Decoding the Mechanism of Action of Rapid-Acting Antidepressant Treatment Strategies: Does Gender Matter?
Abstract
1. Introduction
1.1. Does Gender Matter in Major Depressive Disorder (MDD)?
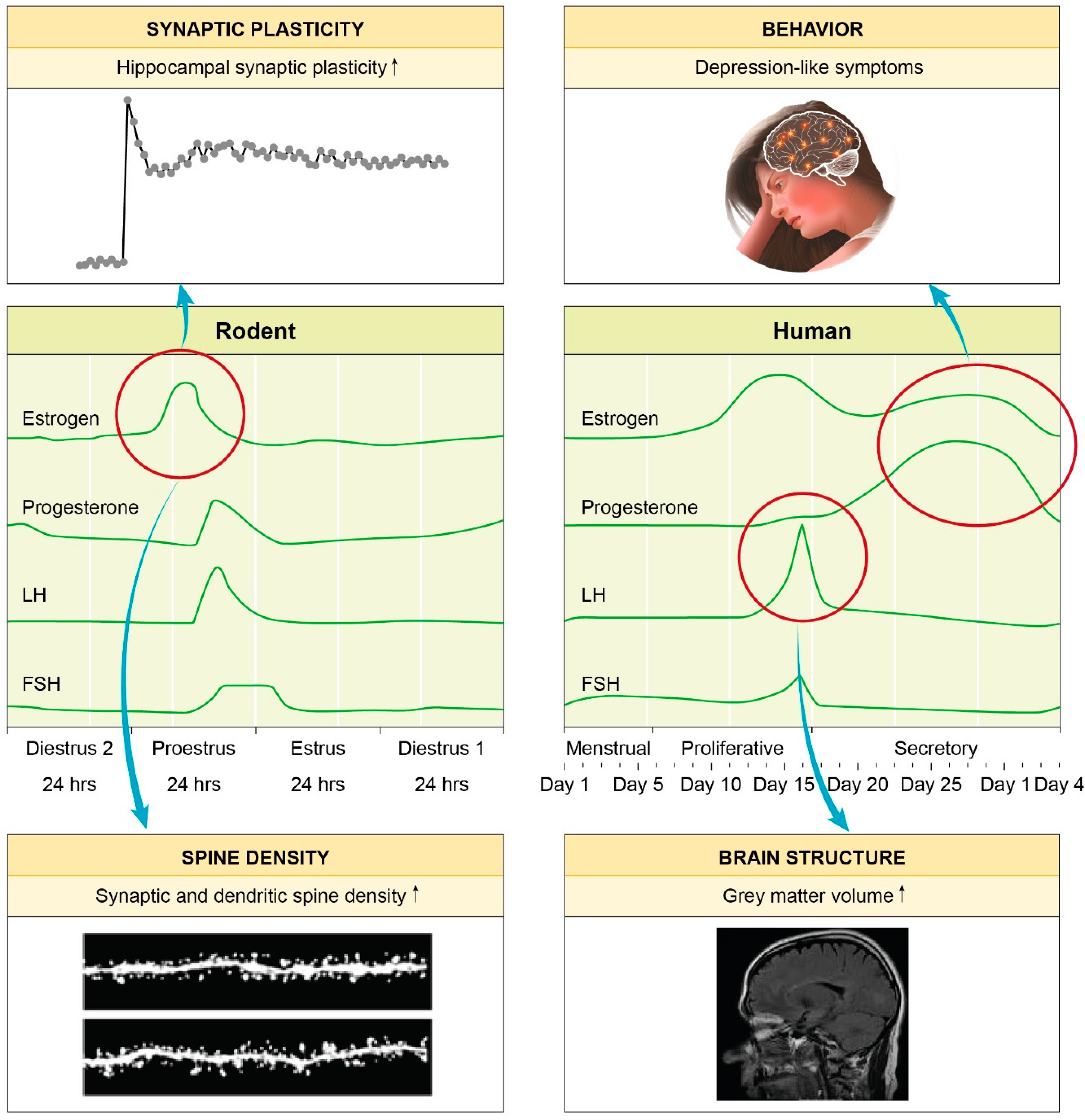
1.2. Genetics, Epigenetics and Hormones: Powerful Players Shaping Gender-Specificity of MDD
1.3. Gender-Specific Differences in MDD Therapy
1.4. Why We Need Antidepressant Treatment Approaches with a Rapid Onset of Action
2. Gender-Specific Differences in the Molecular Mechanisms of Rapid-Acting Antidepressant Drugs
2.1. Ketamine: Evidence of Gender-Specific Differences in the Effect on the Brain
2.2. Rapid Antidepressant-like Effects with Less Side Effects? Emerging Data on Ketamine Metabolites
2.3. Other Rapid-Acting Antidepressant Agents
3. Non-Pharmacological, Rapid-Acting Treatment for MDD: Electroconvulsive Therapy
3.1. Molecular Pathways Shaping the Effect of Stimulating the Brain
3.2. Clinical Efficacy and the Role of Gender
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Patel, V.; Chisholm, D.; Parikh, R.; Charlson, F.J.; Degenhardt, L.; Dua, T.; Ferrari, A.J.; Hyman, S.; Laxminarayan, R.; Levin, C.; et al. Addressing the burden of mental, neurological and substance use disorders: Key messages from Disease Control Priorities. Lancet 2016, 387, 1672–1685. [Google Scholar] [CrossRef]
- WHO. World Health Organization Factsheet: Depression. Available online: www.who.int/news-room/fact-sheets/detail/depression (accessed on 10 October 2018).
- Kessler, R.C.; Berglund, P.; Demler, O.; Jin, R.; Merikangas, K.R.; Walters, E.E. Lifetime prevalence and age-of-onset distributions of DSM-IV disorders in the National Comorbidity Survey Replication. Arch. Gen. Psychiatry 2005, 62, 593–602. [Google Scholar] [CrossRef] [PubMed]
- NICE. Depression in Adults: Recognition and Management—National Institute for Health and Care Excellence Guidelines [CG90]. Available online: www.nice.org.uk/guidance/cg90 (accessed on 2 January 2019).
- Freeman, E.W.; Sammel, M.D.; Boorman, D.W.; Zhang, R. Longitudinal pattern of depressive symptoms around natural menopause. JAMA Psychiatry 2014, 71, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Gillies, G.E.; McArthur, S. Estrogen actions in the brain and the basis for differential action in men and women: A case for sex-specific medicines. Pharmacol. Rev. 2010, 62, 155–198. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Xiao, G.M. The neuroprotective effects of progesterone on traumatic brain injury: Current status and future prospects. Acta Pharmacol. Sin. 2013, 34, 1485–1490. [Google Scholar] [CrossRef] [PubMed]
- Kessler, R.C.; McGonagle, K.A.; Swartz, M.; Blazer, D.G.; Nelson, C.B. Sex and depression in the National Comorbidity Survey. I: Lifetime prevalence, chronicity and recurrence. J. Affect. Disord. 1993, 29, 85–96. [Google Scholar] [CrossRef]
- Kornstein, S.G.; Schatzberg, A.F.; Thase, M.E.; Yonkers, K.A.; McCullough, J.P.; Keitner, G.I.; Gelenberg, A.J.; Ryan, C.E.; Hess, A.L.; Harrison, W.; et al. Gender differences in chronic major and double depression. J. Affect. Disord. 2000, 60, 1–11. [Google Scholar] [CrossRef]
- Marcus, S.M.; Young, E.A.; Kerber, K.B.; Kornstein, S.; Farabaugh, A.H.; Mitchell, J.; Wisniewski, S.R.; Balasubramani, G.K.; Trivedi, M.H.; Rush, A.J. Gender differences in depression: findings from the STAR*D study. J. Affect. Disord. 2005, 87, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Kundakovic, M. Sex-Specific Epigenetics: Implications for Environmental Studies of Brain and Behavior. Curr. Environ. Health Rep. 2017, 4, 385–391. [Google Scholar] [CrossRef]
- LeGates, T.A.; Kvarta, M.D.; Thompson, S.M. Sex differences in antidepressant efficacy. Neuropsychopharmacology 2018. [Google Scholar] [CrossRef]
- Marrocco, J.; McEwen, B.S. Sex in the brain: Hormones and sex differences. Dial. Clin. Neurosci. 2016, 18, 373–383. [Google Scholar]
- Sramek, J.J.; Murphy, M.F.; Cutler, N.R. Sex differences in the psychopharmacological treatment of depression. Dial. Clin. Neurosci. 2016, 18, 447–457. [Google Scholar]
- Kendler, K.S.; Thornton, L.M.; Prescott, C.A. Gender differences in the rates of exposure to stressful life events and sensitivity to their depressogenic effects. Am. J. Psychiatry 2001, 158, 587–593. [Google Scholar] [CrossRef] [PubMed]
- Bundy, J.L.; Vied, C.; Nowakowski, R.S. Sex differences in the molecular signature of the developing mouse hippocampus. BMC Genom. 2017, 18, 237. [Google Scholar] [CrossRef] [PubMed]
- Duclot, F.; Kabbaj, M. The estrous cycle surpasses sex differences in regulating the transcriptome in the rat medial prefrontal cortex and reveals an underlying role of early growth response 1. Genome Biol. 2015, 16, 256. [Google Scholar] [CrossRef] [PubMed]
- Labonte, B.; Engmann, O.; Purushothaman, I.; Menard, C.; Wang, J.; Tan, C.; Scarpa, J.R.; Moy, G.; Loh, Y.E.; Cahill, M.; et al. Sex-specific transcriptional signatures in human depression. Nat. Med. 2017, 23, 1102–1111. [Google Scholar] [CrossRef] [PubMed]
- Binder, E.B. Dissecting the molecular mechanisms of gene x environment interactions: Implications for diagnosis and treatment of stress-related psychiatric disorders. Eur. J. Psychotraumatol. 2017, 8, 1412745. [Google Scholar] [CrossRef] [PubMed]
- Nestler, E.J. Epigenetic mechanisms of depression. JAMA Psychiatry 2014, 71, 454–456. [Google Scholar] [CrossRef] [PubMed]
- Nugent, B.M.; Wright, C.L.; Shetty, A.C.; Hodes, G.E.; Lenz, K.M.; Mahurkar, A.; Russo, S.J.; Devine, S.E.; McCarthy, M.M. Brain feminization requires active repression of masculinization via DNA methylation. Nat. Neurosci. 2015, 18, 690–697. [Google Scholar] [CrossRef]
- Bale, T.L. Stress sensitivity and the development of affective disorders. Horm. Behav. 2006, 50, 529–533. [Google Scholar] [CrossRef]
- Fernandez-Guasti, A.; Fiedler, J.L.; Herrera, L.; Handa, R.J. Sex, stress and mood disorders: At the intersection of adrenal and gonadal hormones. Horm. Metab. Res. 2012, 44, 607–618. [Google Scholar] [CrossRef] [PubMed]
- Babb, J.A.; Masini, C.V.; Day, H.E.; Campeau, S. Stressor-specific effects of sex on HPA axis hormones and activation of stress-related neurocircuitry. Stress (Amsterdam, Netherlands) 2013, 16, 664–677. [Google Scholar] [CrossRef] [PubMed]
- Bohacek, J.; Manuella, F.; Roszkowski, M.; Mansuy, I.M. Hippocampal gene expression induced by cold swim stress depends on sex and handling. Psychoneuroendocrinology 2015, 52, 1–12. [Google Scholar] [CrossRef]
- Galea, L.A.; McEwen, B.S.; Tanapat, P.; Deak, T.; Spencer, R.L.; Dhabhar, F.S. Sex differences in dendritic atrophy of CA3 pyramidal neurons in response to chronic restraint stress. Neuroscience 1997, 81, 689–697. [Google Scholar] [CrossRef]
- McLaughlin, K.J.; Baran, S.E.; Wright, R.L.; Conrad, C.D. Chronic stress enhances spatial memory in ovariectomized female rats despite CA3 dendritic retraction: Possible involvement of CA1 neurons. Neuroscience 2005, 135, 1045–1054. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, A.; Kabbaj, M. Sex Differences in Effects of Ketamine on Behavior, Spine Density and Synaptic Proteins in Socially Isolated Rats. Biol. Psychiatry 2016, 80, 448–456. [Google Scholar] [CrossRef] [PubMed]
- McEwen, B.S.; De Kloet, E.R.; Rostene, W. Adrenal steroid receptors and actions in the nervous system. Physiol. Rev. 1986, 66, 1121–1188. [Google Scholar] [CrossRef] [PubMed]
- Woolley, C.S.; McEwen, B.S. Estradiol mediates fluctuation in hippocampal synapse density during the estrous cycle in the adult rat. J. Neurosci. 1992, 12, 2549–2554. [Google Scholar] [CrossRef] [PubMed]
- Warren, S.G.; Humphreys, A.G.; Juraska, J.M.; Greenough, W.T. LTP varies across the estrous cycle: Enhanced synaptic plasticity in proestrus rats. Brain Res. 1995, 703, 26–30. [Google Scholar] [CrossRef]
- Hagemann, G.; Ugur, T.; Schleussner, E.; Mentzel, H.J.; Fitzek, C.; Witte, O.W.; Gaser, C. Changes in brain size during the menstrual cycle. PLoS ONE 2011, 6, e14655. [Google Scholar] [CrossRef]
- Epperson, C.N.; Steiner, M.; Hartlage, S.A.; Eriksson, E.; Schmidt, P.J.; Jones, I.; Yonkers, K.A. Premenstrual dysphoric disorder: Evidence for a new category for DSM-5. Am. J. Psychiatry 2012, 169, 465–475. [Google Scholar] [CrossRef] [PubMed]
- Eiland, L.; Ramroop, J.; Hill, M.N.; Manley, J.; McEwen, B.S. Chronic juvenile stress produces corticolimbic dendritic architectural remodeling and modulates emotional behavior in male and female rats. Psychoneuroendocrinology 2012, 37, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Amin, Z.; Canli, T.; Epperson, C.N. Effect of estrogen-serotonin interactions on mood and cognition. Behav. Cogn. Neurosci. Rev. 2005, 4, 43–58. [Google Scholar] [CrossRef] [PubMed]
- Young, S.N.; Gauthier, S.; Anderson, G.M.; Purdy, W.C. Tryptophan, 5-hydroxyindoleacetic acid and indoleacetic acid in human cerebrospinal fluid: Interrelationships and the influence of age, sex, epilepsy and anticonvulsant drugs. J. Neurol. Neurosurg. Psychiatry 1980, 43, 438–445. [Google Scholar] [CrossRef] [PubMed]
- Staley, J.K.; Krishnan-Sarin, S.; Zoghbi, S.; Tamagnan, G.; Fujita, M.; Seibyl, J.P.; Maciejewski, P.K.; O’Malley, S.; Innis, R.B. Sex differences in [123I]beta-CIT SPECT measures of dopamine and serotonin transporter availability in healthy smokers and nonsmokers. Synapse 2001, 41, 275–284. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.H.; Kim, Y.K. The roles of BDNF in the pathophysiology of major depression and in antidepressant treatment. Psychiatry Investig. 2010, 7, 231–235. [Google Scholar] [CrossRef]
- McEwen, B.S.; Akama, K.T.; Spencer-Segal, J.L.; Milner, T.A.; Waters, E.M. Estrogen effects on the brain: Actions beyond the hypothalamus via novel mechanisms. Behav. Neurosci. 2012, 126, 4–16. [Google Scholar] [CrossRef]
- Keers, R.; Aitchison, K.J. Gender differences in antidepressant drug response. Int. Rev. Psychiatry 2010, 22, 485–500. [Google Scholar] [CrossRef]
- Wohlfarth, T.; Storosum, J.G.; Elferink, A.J.; van Zwieten, B.J.; Fouwels, A.; van den Brink, W. Response to tricyclic antidepressants: Independent of gender? Am. J. Psychiatry 2004, 161, 370–372. [Google Scholar] [CrossRef]
- Khan, A.; Brodhead, A.E.; Schwartz, K.A.; Kolts, R.L.; Brown, W.A. Sex differences in antidepressant response in recent antidepressant clinical trials. J. Clin. Psychopharmacol. 2005, 25, 318–324. [Google Scholar] [CrossRef]
- Kornstein, S.G.; Schatzberg, A.F.; Thase, M.E.; Yonkers, K.A.; McCullough, J.P.; Keitner, G.I.; Gelenberg, A.J.; Davis, S.M.; Harrison, W.M.; Keller, M.B. Gender differences in treatment response to sertraline versus imipramine in chronic depression. Am. J. Psychiatry 2000, 157, 1445–1452. [Google Scholar] [CrossRef] [PubMed]
- Gaynes, B.N.; Warden, D.; Trivedi, M.H.; Wisniewski, S.R.; Fava, M.; Rush, A.J. What did STAR*D teach us? Results from a large-scale, practical, clinical trial for patients with depression. Psychiatr. Serv. 2009, 60, 1439–1445. [Google Scholar] [CrossRef] [PubMed]
- Berlanga, C.; Flores-Ramos, M. Different gender response to serotonergic and noradrenergic antidepressants. A comparative study of the efficacy of citalopram and reboxetine. J. Affect. Disord. 2006, 95, 119–123. [Google Scholar] [CrossRef] [PubMed]
- Haykal, R.F.; Akiskal, H.S. The long-term outcome of dysthymia in private practice: Clinical features, temperament and the art of management. J. Clin. Psychiatry 1999, 60, 508–518. [Google Scholar] [CrossRef] [PubMed]
- Schneider, L.S.; Small, G.W.; Hamilton, S.H.; Bystritsky, A.; Nemeroff, C.B.; Meyers, B.S. Estrogen replacement and response to fluoxetine in a multicenter geriatric depression trial. Fluoxetine Collaborative Study Group. Am. J. Geriatr. Psychiatry 1997, 5, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Thase, M.E.; Entsuah, R.; Cantillon, M.; Kornstein, S.G. Relative antidepressant efficacy of venlafaxine and SSRIs: Sex-age interactions. J. Womens Health (Larchmt) 2005, 14, 609–616. [Google Scholar] [CrossRef] [PubMed]
- Cuijpers, P.; Weitz, E.; Twisk, J.; Kuehner, C.; Cristea, I.; David, D.; DeRubeis, R.J.; Dimidjian, S.; Dunlop, B.W.; Faramarzi, M.; et al. Gender as predictor and moderator of outcome in cognitive behavior therapy and pharmacotherapy for adult depression: An “individual patient data” meta-analysis. Depress. Anxiety 2014, 31, 941–951. [Google Scholar] [CrossRef]
- NIMH. RDoC Matrix. Available online: https://www.nimh.nih.gov/research-priorities/rdoc/constructs/rdoc-matrix.shtml (accessed on 13 February 2019).
- Gex-Fabry, M.; Balant-Gorgia, A.E.; Balant, L.P.; Garrone, G. Clomipramine metabolism. Model-based analysis of variability factors from drug monitoring data. Clin. Pharmacokinet. 1990, 19, 241–255. [Google Scholar] [CrossRef]
- Preskorn, S.H.; Mac, D.S. Plasma levels of amitriptyline: Effect of age and sex. J. Clin. Psychiatry 1985, 46, 276–277. [Google Scholar]
- Degli Esposti, L.; Piccinni, C.; Sangiorgi, D.; Fagiolini, A.; Buda, S. Patterns of antidepressant use in Italy: Therapy duration, adherence and switching. Clin. Drug Investig. 2015, 35, 735–742. [Google Scholar] [CrossRef]
- Fornaro, M.; Anastasia, A.; Monaco, F.; Novello, S.; Fusco, A.; Iasevoli, F.; De Berardis, D.; Veronese, N.; Solmi, M.; de Bartolomeis, A. Clinical and psychopathological features associated with treatment-emergent mania in bipolar-II depressed outpatients exposed to antidepressants. J. Affect. Disord. 2018, 234, 131–138. [Google Scholar] [CrossRef]
- Fornaro, M.; Anastasia, A.; Novello, S.; Fusco, A.; Pariano, R.; De Berardis, D.; Solmi, M.; Veronese, N.; Stubbs, B.; Vieta, E.; et al. The emergence of loss of efficacy during antidepressant drug treatment for major depressive disorder: An integrative review of evidence, mechanisms and clinical implications. Pharmacol. Res. 2019, 139, 494–502. [Google Scholar] [CrossRef] [PubMed]
- Labermaier, C.; Masana, M.; Muller, M.B. Biomarkers predicting antidepressant treatment response: How can we advance the field? Dis. Mark. 2013, 35, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Herzog, D.P.; Beckmann, H.; Lieb, K.; Ryu, S.; Müller, M.B. Understanding and Predicting Antidepressant Response: Using Animal Models to Move Toward Precision Psychiatry. Front. Psychiatry 2018, 9, 512. [Google Scholar] [CrossRef]
- Xu, Y.; Hackett, M.; Carter, G.; Loo, C.; Galvez, V.; Glozier, N.; Glue, P.; Lapidus, K.; McGirr, A.; Somogyi, A.A.; et al. Effects of Low-Dose and Very Low-Dose Ketamine among Patients with Major Depression: A Systematic Review and Meta-Analysis. Int. J. Neuropsychopharmacol. 2016, 19, pyv124. [Google Scholar] [CrossRef] [PubMed]
- Zarate, C.A., Jr.; Singh, J.B.; Carlson, P.J.; Brutsche, N.E.; Ameli, R.; Luckenbaugh, D.A.; Charney, D.S.; Manji, H.K. A randomized trial of an N-methyl-D-aspartate antagonist in treatment-resistant major depression. Arch. Gen. Psychiatry 2006, 63, 856–864. [Google Scholar] [CrossRef] [PubMed]
- De Berardis, D.; Fornaro, M.; Valchera, A.; Cavuto, M.; Perna, G.; Di Nicola, M.; Serafini, G.; Carano, A.; Pompili, M.; Vellante, F.; et al. Eradicating Suicide at Its Roots: Preclinical Bases and Clinical Evidence of the Efficacy of Ketamine in the Treatment of Suicidal Behaviors. Int. J. Mol. Sci. 2018, 19, 2888. [Google Scholar] [CrossRef] [PubMed]
- Tomasetti, C.; Iasevoli, F.; Buonaguro, E.F.; De Berardis, D.; Fornaro, M.; Fiengo, A.L.; Martinotti, G.; Orsolini, L.; Valchera, A.; Di Giannantonio, M.; et al. Treating the Synapse in Major Psychiatric Disorders: The Role of Postsynaptic Density Network in Dopamine-Glutamate Interplay and Psychopharmacologic Drugs Molecular Actions. Int. J. Mol. Sci. 2017, 18, 135. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Lee, B.; Liu, R.J.; Banasr, M.; Dwyer, J.M.; Iwata, M.; Li, X.Y.; Aghajanian, G.; Duman, R.S. mTOR-dependent synapse formation underlies the rapid antidepressant effects of NMDA antagonists. Science 2010, 329, 959–964. [Google Scholar] [CrossRef]
- Autry, A.E.; Adachi, M.; Nosyreva, E.; Na, E.S.; Los, M.F.; Cheng, P.F.; Kavalali, E.T.; Monteggia, L.M. NMDA receptor blockade at rest triggers rapid behavioural antidepressant responses. Nature 2011, 475, 91–95. [Google Scholar] [CrossRef]
- Franceschelli, A.; Sens, J.; Herchick, S.; Thelen, C.; Pitychoutis, P.M. Sex differences in the rapid and the sustained antidepressant-like effects of ketamine in stress-naive and “depressed” mice exposed to chronic mild stress. Neuroscience 2015, 290, 49–60. [Google Scholar] [CrossRef] [PubMed]
- Saland, S.K.; Kabbaj, M. Sex Differences in the Pharmacokinetics of Low-dose Ketamine in Plasma and Brain of Male and Female Rats. J. Pharmacol. Exp. Ther. 2018, 367, 393–404. [Google Scholar] [CrossRef] [PubMed]
- Ho, M.F.; Correia, C.; Ingle, J.N.; Kaddurah-Daouk, R.; Wang, L.; Kaufmann, S.H.; Weinshilboum, R.M. Ketamine and ketamine metabolites as novel estrogen receptor ligands: Induction of cytochrome P450 and AMPA glutamate receptor gene expression. Biochem. Pharmacol. 2018, 152, 279–292. [Google Scholar] [CrossRef] [PubMed]
- Dossat, A.M.; Wright, K.N.; Strong, C.E.; Kabbaj, M. Behavioral and biochemical sensitivity to low doses of ketamine: Influence of estrous cycle in C57BL/6 mice. Neuropharmacology 2018, 130, 30–41. [Google Scholar] [CrossRef]
- Kara, N.Z.; Agam, G.; Anderson, G.W.; Zitron, N.; Einat, H. Lack of effect of chronic ketamine administration on depression-like behavior and frontal cortex autophagy in female and male ICR mice. Behav. Brain Res. 2017, 317, 576–580. [Google Scholar] [CrossRef] [PubMed]
- Zanos, P.; Moaddel, R.; Morris, P.J.; Georgiou, P.; Fischell, J.; Elmer, G.I.; Alkondon, M.; Yuan, P.; Pribut, H.J.; Singh, N.S.; et al. NMDAR inhibition-independent antidepressant actions of ketamine metabolites. Nature 2016, 533, 481–486. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, J.I.; Toki, H.; Qu, Y.; Yang, C.; Koike, H.; Hashimoto, K.; Mizuno-Yasuhira, A.; Chaki, S. (2R,6R)-Hydroxynorketamine is not essential for the antidepressant actions of (R)-ketamine in mice. Neuropsychopharmacology 2018, 43, 1900–1907. [Google Scholar] [CrossRef]
- Zanos, P.; Thompson, S.M.; Duman, R.S.; Zarate, C.A., Jr.; Gould, T.D. Convergent Mechanisms Underlying Rapid Antidepressant Action. CNS Drugs 2018, 32, 197–227. [Google Scholar] [CrossRef]
- Chou, D.; Peng, H.Y.; Lin, T.B.; Lai, C.Y.; Hsieh, M.C.; Wen, Y.C.; Lee, A.S.; Wang, H.H.; Yang, P.S.; Chen, G.D.; et al. (2R,6R)-hydroxynorketamine rescues chronic stress-induced depression-like behavior through its actions in the midbrain periaqueductal gray. Neuropharmacology 2018, 139, 1–12. [Google Scholar] [CrossRef]
- Pham, T.H.; Defaix, C.; Xu, X.; Deng, S.X.; Fabresse, N.; Alvarez, J.C.; Landry, D.W.; Brachman, R.A.; Denny, C.A.; Gardier, A.M. Common Neurotransmission Recruited in (R,S)-Ketamine and (2R,6R)-Hydroxynorketamine-Induced Sustained Antidepressant-like Effects. Biol. Psychiatry 2018, 84, e3–e6. [Google Scholar] [CrossRef]
- Cavalleri, L.; Merlo Pich, E.; Millan, M.J.; Chiamulera, C.; Kunath, T.; Spano, P.F.; Collo, G. Ketamine enhances structural plasticity in mouse mesencephalic and human iPSC-derived dopaminergic neurons via AMPAR-driven BDNF and mTOR signaling. Mol. Psychiatry 2018, 23, 812–823. [Google Scholar] [CrossRef] [PubMed]
- Collo, G.; Cavalleri, L.; Chiamulera, C.; Merlo Pich, E. (2R,6R)-Hydroxynorketamine promotes dendrite outgrowth in human inducible pluripotent stem cell-derived neurons through AMPA receptor with timing and exposure compatible with ketamine infusion pharmacokinetics in humans. Neuroreport 2018, 29, 1425–1430. [Google Scholar] [CrossRef] [PubMed]
- Yao, N.; Skiteva, O.; Zhang, X.; Svenningsson, P.; Chergui, K. Ketamine and its metabolite (2R,6R)-hydroxynorketamine induce lasting alterations in glutamatergic synaptic plasticity in the mesolimbic circuit. Mol. Psychiatry 2017, 23, 2066–2077. [Google Scholar] [CrossRef] [PubMed]
- Shirayama, Y.; Hashimoto, K. Lack of Antidepressant Effects of (2R,6R)-Hydroxynorketamine in a Rat Learned Helplessness Model: Comparison with (R)-Ketamine. Int. J. Neuropsychopharmacol. 2018, 21, 84–88. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Qu, Y.; Abe, M.; Nozawa, D.; Chaki, S.; Hashimoto, K. (R)-Ketamine Shows Greater Potency and Longer Lasting Antidepressant Effects Than Its Metabolite (2R,6R)-Hydroxynorketamine. Biol. Psychiatry 2017, 82, e43–e44. [Google Scholar] [CrossRef] [PubMed]
- Thomas, A.M.; Duman, R.S. Novel rapid-acting antidepressants: Molecular and cellular signaling mechanisms. Neuronal Signal. 2017, 1. [Google Scholar] [CrossRef] [PubMed]
- Witkin, J.M.; Knutson, D.E.; Rodriguez, G.J.; Shi, S. Rapid-Acting Antidepressants. Curr. Pharm. Des. 2018, 24, 2556–2563. [Google Scholar] [CrossRef]
- Furey, M.L.; Drevets, W.C. Antidepressant efficacy of the antimuscarinic drug scopolamine: A randomized, placebo-controlled clinical trial. Arch. Gen. Psychiatry 2006, 63, 1121–1129. [Google Scholar] [CrossRef]
- Griffiths, R.; Richards, W.; Johnson, M.; McCann, U.; Jesse, R. Mystical-type experiences occasioned by psilocybin mediate the attribution of personal meaning and spiritual significance 14 months later. J. Psychopharmacol. 2008, 22, 621–632. [Google Scholar] [CrossRef]
- Moskal, J.R.; Burgdorf, J.S.; Stanton, P.K.; Kroes, R.A.; Disterhoft, J.F.; Burch, R.M.; Khan, M.A. The Development of Rapastinel (Formerly GLYX-13); A Rapid Acting and Long Lasting Antidepressant. Curr. Neuropharmacol. 2017, 15, 47–56. [Google Scholar] [CrossRef]
- Preskorn, S.; Macaluso, M.; Mehra, D.O.; Zammit, G.; Moskal, J.R.; Burch, R.M.; Group, G.-C.S. Randomized proof of concept trial of GLYX-13, an N-methyl-D-aspartate receptor glycine site partial agonist, in major depressive disorder nonresponsive to a previous antidepressant agent. J. Psychiatr. Pract. 2015, 21, 140–149. [Google Scholar] [CrossRef] [PubMed]
- Chaki, S.; Fukumoto, K. mGlu receptors as potential targets for novel antidepressants. Curr. Opin. Pharmacol. 2018, 38, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Chappell, M.D.; Li, R.; Smith, S.C.; Dressman, B.A.; Tromiczak, E.G.; Tripp, A.E.; Blanco, M.J.; Vetman, T.; Quimby, S.J.; Matt, J.; et al. Discovery of (1S,2R,3S,4S,5R,6R)-2-Amino-3-[(3,4-difluorophenyl)sulfanylmethyl]-4-hydroxy-bicy clo[3.1.0]hexane-2,6-dicarboxylic Acid Hydrochloride (LY3020371.HCl): A Potent, Metabotropic Glutamate 2/3 Receptor Antagonist with Antidepressant-Like Activity. J. Med. Chem. 2016, 59, 10974–10993. [Google Scholar] [CrossRef] [PubMed]
- Witkin, J.M.; Ornstein, P.L.; Mitch, C.H.; Li, R.; Smith, S.C.; Heinz, B.A.; Wang, X.S.; Xiang, C.; Carter, J.H.; Anderson, W.H.; et al. In vitro pharmacological and rat pharmacokinetic characterization of LY3020371, a potent and selective mGlu2/3 receptor antagonist. Neuropharmacology 2017, 115, 100–114. [Google Scholar] [CrossRef] [PubMed]
- Fischell, J.; Van Dyke, A.M.; Kvarta, M.D.; LeGates, T.A.; Thompson, S.M. Rapid Antidepressant Action and Restoration of Excitatory Synaptic Strength After Chronic Stress by Negative Modulators of Alpha5-Containing GABAA Receptors. Neuropsychopharmacology 2015, 40, 2499–2509. [Google Scholar] [CrossRef] [PubMed]
- Zanos, P.; Nelson, M.E.; Highland, J.N.; Krimmel, S.R.; Georgiou, P.; Gould, T.D.; Thompson, S.M. A Negative Allosteric Modulator for alpha5 Subunit-Containing GABA Receptors Exerts a Rapid and Persistent Antidepressant-Like Action without the Side Effects of the NMDA Receptor Antagonist Ketamine in Mice. eNeuro 2017, 4. [Google Scholar] [CrossRef] [PubMed]
- Sales, A.J.; Fogaca, M.V.; Sartim, A.G.; Pereira, V.S.; Wegener, G.; Guimaraes, F.S.; Joca, S.R.L. Cannabidiol Induces Rapid and Sustained Antidepressant-Like Effects Through Increased BDNF Signaling and Synaptogenesis in the Prefrontal Cortex. Mol. Neurobiol. 2018. [Google Scholar] [CrossRef]
- Sartim, A.G.; Guimaraes, F.S.; Joca, S.R. Antidepressant-like effect of cannabidiol injection into the ventral medial prefrontal cortex-Possible involvement of 5-HT1A and CB1 receptors. Behav. Brain Res. 2016, 303, 218–227. [Google Scholar] [CrossRef]
- Shbiro, L.; Hen-Shoval, D.; Hazut, N.; Rapps, K.; Dar, S.; Zalsman, G.; Mechoulam, R.; Weller, A.; Shoval, G. Effects of cannabidiol in males and females in two different rat models of depression. Physiol. Behav. 2018, 201, 59–63. [Google Scholar] [CrossRef]
- Shoval, G.; Shbiro, L.; Hershkovitz, L.; Hazut, N.; Zalsman, G.; Mechoulam, R.; Weller, A. Prohedonic Effect of Cannabidiol in a Rat Model of Depression. Neuropsychobiology 2016, 73, 123–129. [Google Scholar] [CrossRef]
- Ly, C.; Greb, A.C.; Cameron, L.P.; Wong, J.M.; Barragan, E.V.; Wilson, P.C.; Burbach, K.F.; Soltanzadeh Zarandi, S.; Sood, A.; Paddy, M.R.; et al. Psychedelics Promote Structural and Functional Neural Plasticity. Cell Rep. 2018, 23, 3170–3182. [Google Scholar] [CrossRef]
- Sur, C.; Fresu, L.; Howell, O.; McKernan, R.M.; Atack, J.R. Autoradiographic localization of alpha5 subunit-containing GABAA receptors in rat brain. Brain Res. 1999, 822, 265–270. [Google Scholar] [CrossRef]
- Guimaraes, F.S.; Chiaretti, T.M.; Graeff, F.G.; Zuardi, A.W. Antianxiety effect of cannabidiol in the elevated plus-maze. Psychopharmacology 1990, 100, 558–559. [Google Scholar] [CrossRef] [PubMed]
- Zanelati, T.V.; Biojone, C.; Moreira, F.A.; Guimaraes, F.S.; Joca, S.R. Antidepressant-like effects of cannabidiol in mice: Possible involvement of 5-HT1A receptors. Br. J. Pharmacol. 2010, 159, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Husain, S.S.; Kevan, I.M.; Linnell, R.; Scott, A.I. Electroconvulsive therapy in depressive illness that has not responded to drug treatment. J. Affect. Disord. 2004, 83, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Hoy, K.E.; Fitzgerald, P.B. Brain stimulation in psychiatry and its effects on cognition. Nat. Rev. Neurol. 2010, 6, 267–275. [Google Scholar] [CrossRef] [PubMed]
- NIMH. National Institute of Mental Health Information: Brain Stimulation Therapies. Available online: https://www.nimh.nih.gov/health/topics/brain-stimulation-therapies/brain-stimulation-therapies.shtml (accessed on 29 October 2018).
- The UK ECT Review Group. Efficacy and safety of electroconvulsive therapy in depressive disorders: A systematic review and meta-analysis. Lancet 2003, 361, 799–808. [Google Scholar] [CrossRef]
- Miller, A.H.; Maletic, V.; Raison, C.L. Inflammation and its discontents: The role of cytokines in the pathophysiology of major depression. Biol. Psychiatry 2009, 65, 732–741. [Google Scholar] [CrossRef] [PubMed]
- Nazimek, K.; Strobel, S.; Bryniarski, P.; Kozlowski, M.; Filipczak-Bryniarska, I.; Bryniarski, K. The role of macrophages in anti-inflammatory activity of antidepressant drugs. Immunobiology 2017, 222, 823–830. [Google Scholar] [CrossRef] [PubMed]
- Guloksuz, S.; Rutten, B.P.; Arts, B.; van Os, J.; Kenis, G. The immune system and electroconvulsive therapy for depression. J. ECT 2014, 30, 132–137. [Google Scholar] [CrossRef]
- Yrondi, A.; Sporer, M.; Peran, P.; Schmitt, L.; Arbus, C.; Sauvaget, A. Electroconvulsive therapy, depression, the immune system and inflammation: A systematic review. Brain Stimul. 2018, 11, 29–51. [Google Scholar] [CrossRef]
- Bouckaert, F.; Dols, A.; Emsell, L.; De Winter, F.L.; Vansteelandt, K.; Claes, L.; Sunaert, S.; Stek, M.; Sienaert, P.; Vandenbulcke, M. Relationship Between Hippocampal Volume, Serum BDNF and Depression Severity Following Electroconvulsive Therapy in Late-Life Depression. Neuropsychopharmacology 2016, 41, 2741–2748. [Google Scholar] [CrossRef]
- Albala, A.A.; Greden, J.F.; Tarika, J.; Carroll, B.J. Changes in serial dexamethasone suppression tests among unipolar depressive receiving electroconvulsive treatment. Biol. Psychiatry 1981, 16, 551–560. [Google Scholar]
- Yuuki, N.; Ida, I.; Oshima, A.; Kumano, H.; Takahashi, K.; Fukuda, M.; Oriuchi, N.; Endo, K.; Matsuda, H.; Mikuni, M. HPA axis normalization, estimated by DEX/CRH test but less alteration on cerebral glucose metabolism in depressed patients receiving ECT after medication treatment failures. Acta Psychiatr. Scand. 2005, 112, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Dored, G.; Stefansson, S.; d’Elia, G.; Kagedal, B.; Karlberg, E.; Ekman, R. Corticotropin, cortisol and beta-endorphin responses to the human corticotropin-releasing hormone during melancholia and after unilateral electroconvulsive therapy. Acta Psychiatr. Scand. 1990, 82, 204–209. [Google Scholar] [CrossRef] [PubMed]
- Saijo, T.; Takano, A.; Suhara, T.; Arakawa, R.; Okumura, M.; Ichimiya, T.; Ito, H.; Okubo, Y. Electroconvulsive therapy decreases dopamine D(2)receptor binding in the anterior cingulate in patients with depression: A controlled study using positron emission tomography with radioligand [(1)(1)C]FLB 457. J. Clin. Psychiatry 2010, 71, 793–799. [Google Scholar] [CrossRef] [PubMed]
- Madsen, T.M.; Treschow, A.; Bengzon, J.; Bolwig, T.G.; Lindvall, O.; Tingstrom, A. Increased neurogenesis in a model of electroconvulsive therapy. Biol. Psychiatry 2000, 47, 1043–1049. [Google Scholar] [CrossRef]
- Chen, F.; Madsen, T.M.; Wegener, G.; Nyengaard, J.R. Repeated electroconvulsive seizures increase the total number of synapses in adult male rat hippocampus. Eur. Neuropsychopharmacol. 2009, 19, 329–338. [Google Scholar] [CrossRef]
- Yrondi, A.; Peran, P.; Sauvaget, A.; Schmitt, L.; Arbus, C. Structural-functional brain changes in depressed patients during and after electroconvulsive therapy. Acta Neuropsychiatr. 2018, 30, 17–28. [Google Scholar] [CrossRef]
- Benson-Martin, J.J.; Stein, D.J.; Baldwin, D.S.; Domschke, K. Genetic mechanisms of electroconvulsive therapy response in depression. Hum. Psychopharmacol. 2016, 31, 247–251. [Google Scholar] [CrossRef]
- de Jong, J.O.; Arts, B.; Boks, M.P.; Sienaert, P.; van den Hove, D.L.; Kenis, G.; van Os, J.; Rutten, B.P. Epigenetic effects of electroconvulsive seizures. J. ECT 2014, 30, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Pusalkar, M.; Ghosh, S.; Jaggar, M.; Husain, B.F.; Galande, S.; Vaidya, V.A. Acute and Chronic Electroconvulsive Seizures (ECS) Differentially Regulate the Expression of Epigenetic Machinery in the Adult Rat Hippocampus. Int. J. Neuropsychopharmacol. 2016, 19, pyw040. [Google Scholar] [CrossRef] [PubMed]
- Bloch, Y.; Ratzoni, G.; Sobol, D.; Mendlovic, S.; Gal, G.; Levkovitz, Y. Gender differences in electroconvulsive therapy: A retrospective chart review. J. Affect. Disord. 2005, 84, 99–102. [Google Scholar] [CrossRef] [PubMed]
- Bolu, A.; Ozselek, S.; Akarsu, S.; Alper, M.; Balikci, A. Is There a Role of Gender in Electroconvulsive Therapy Response? Klinik Psikofarmakoloji Bülteni-Bull. Clin. Psychopharmacol. 2016, 26, 32–38. [Google Scholar] [CrossRef]
- Bousman, C.A.; Katalinic, N.; Martin, D.M.; Smith, D.J.; Ingram, A.; Dowling, N.; Ng, C.; Loo, C.K. Effects of COMT, DRD2, BDNF and APOE Genotypic Variation on Treatment Efficacy and Cognitive Side Effects of Electroconvulsive Therapy. J. ECT 2015, 31, 129–135. [Google Scholar] [CrossRef] [PubMed]
| Publication | Test Subject | Study Design | Antidepressant-Like Effect | Molecular Mechanism |
|---|---|---|---|---|
| Franceschelli et al., 2015 [64] | Male and female C57/BL6J mice | KET in naïve and CMS animals: female and male mice (FST) | KET effect: Female mice > male mice | Effects on excitatory amino acids (glutamate and aspartate), serotoninergic activity. |
| Saland et al., 2018 [65] | Male and female Sprague-Dawley rats | KET metabolism and distribution | ↑ level of KET and NK in both brain and plasma | |
| Ho et al., 2018 [66] | Human iPSC-derived astrocytes | Oestrogen + KET in vitro | Oestrogens augmented the effect of KET | ↑ level of AMPA receptor subunit and ERα. Oestrogens: ↑ level of CYP2A6 and CYP2B6. |
| Dossat et al., 2018 [67] | Male and female C57/BL6J mice | Oestrogen and Progesterone receptor agonist and KET (FST) | Female in proestrus + KET: sensitive to lower dose. | Proestrus female ↑ p-Akt and p-CaMKIIα. |
| Sarkar et al. 2016 [28] | Male and female Sprague-Dawley rats | KET and social isolation stress (behaviour and synaptic protein level) | IS: male depression like behaviour at 8 weeks while female at 11 weeks. KET rescued the phenotype. | Decline in spine density and synaptic proteins reversed by KET only in male but not female |
| Publication | Test Subject | Study Design | Antidepressant-Like Effect | Molecular Mechanism |
|---|---|---|---|---|
| Zanos et al. 2016 [69] | Male and female C57/BL6J mice | KET: female and male mice (FST) Ketamine metabolites: male mice (CSD, FST, ST) | KET: Female mice > male mice HNK: HNK > (2S,6S)-hydroxynorketamine HNK: lacks ketamine-related side effects | HNK-effects independent of NMDAR-signalling by AMPAR-signalling |
| Yamaguchi et al. 2018 [70] | Male C57/BL6 mice | LPS with (R)-ketamine and HNK (FST, TST) | (R)-ketamine > HNK Blocking CYP: (R)-ketamine effects ↑ | (R)-ketamine and HNK in plasma, brain, CSF |
| Chou et al. 2018 [72] | Male and female Sprague-Dawley rats | LH with ketamine metabolites (FST, SPT) | HNK: Male ≈ Female (2S,6S)-hydroxynorketamine: no effect | HNK: enhancement of AMPAR-signalling in vlPAG |
| Pham et al. 2018 [73] | Male BALB/cJ mice | Local (mPFC) and systemic injection of KET and HNK (FST) | Local injection: HNK ≈ KET Systemic injection: HNK ≈ KET | HNK+KET: extracellular 5-hydroxytryptamine (mPFC) ↑, extracellular glutamate (mPFC) ↑ KET: extracellular GABA ↑ |
| Cavalleri et al. 2018 [74] | Murine and human DA neurons | KET and HNK in vitro | - | HNK+KET: structural plasticity ↑ (arborization ↑, soma size ↑) |
| Collo et al. 2018 [75] | Human DA neurons (PSCs) | KET and HNK in vitro | - | HNK+KET: structural plasticity ↑ (dendrite length ↑ and number ↑) |
| Yao et al. 2017 [76] | Male C57/BL6 mice | KET and HNK tested ex vivo with electrophysiology | - | HNK+KET: lasting modulation of AMPAR and synaptic plasticity (NAc+VTA), potentiation ↓ and depression ↑ of GA synapses (NAc+VTA-DA neurons) |
| Shirayama et al. 2018 [77] | Male Sprague-Dawley rats | LH with ketamine metabolites (CAT) | KET: antidepressant-like effect HNK: no effect | - |
| Yang et al. 2017 [78] | Male C57/BL6 mice | LH and CSD with KET and HNK (FST, TST, SPT) | KET: antidepressant-like effect HNK: no effect | - |
| Agent | Molecular Target | Reference |
|---|---|---|
| Scopolamine | M1/2-antagonist, AMPAR↑, mTOR↑ | [81,82] |
| GLYX-13 | Partial agonist and modulator of NMDAR, AMPAR↑ | [83,84] |
| MGS0039, LY3020371 | mGlu2/3 antagonists, AMPAR↑ | [85,86,87] |
| L-655,708, MRK-016 | NAM of α5-GABAA-R, cortex & HC-specific | [88,89] |
| Cannabidiol | 5-HT1A-R↑, CB1↑, vmPFC | [90,91,92,93] |
| Psychedelics (LSD, DOI, DMT, MDMA) | TrkB→mTOR↑+BDNF↑, 5-HT2A-R↑, PFC | [94] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Herzog, D.P.; Wegener, G.; Lieb, K.; Müller, M.B.; Treccani, G. Decoding the Mechanism of Action of Rapid-Acting Antidepressant Treatment Strategies: Does Gender Matter? Int. J. Mol. Sci. 2019, 20, 949. https://doi.org/10.3390/ijms20040949
Herzog DP, Wegener G, Lieb K, Müller MB, Treccani G. Decoding the Mechanism of Action of Rapid-Acting Antidepressant Treatment Strategies: Does Gender Matter? International Journal of Molecular Sciences. 2019; 20(4):949. https://doi.org/10.3390/ijms20040949
Chicago/Turabian StyleHerzog, David P., Gregers Wegener, Klaus Lieb, Marianne B. Müller, and Giulia Treccani. 2019. "Decoding the Mechanism of Action of Rapid-Acting Antidepressant Treatment Strategies: Does Gender Matter?" International Journal of Molecular Sciences 20, no. 4: 949. https://doi.org/10.3390/ijms20040949
APA StyleHerzog, D. P., Wegener, G., Lieb, K., Müller, M. B., & Treccani, G. (2019). Decoding the Mechanism of Action of Rapid-Acting Antidepressant Treatment Strategies: Does Gender Matter? International Journal of Molecular Sciences, 20(4), 949. https://doi.org/10.3390/ijms20040949





