β-Galactosidase from Lactobacillus helveticus DSM 20075: Biochemical Characterization and Recombinant Expression for Applications in Dairy Industry
Abstract
1. Introduction
2. Results and Discussion
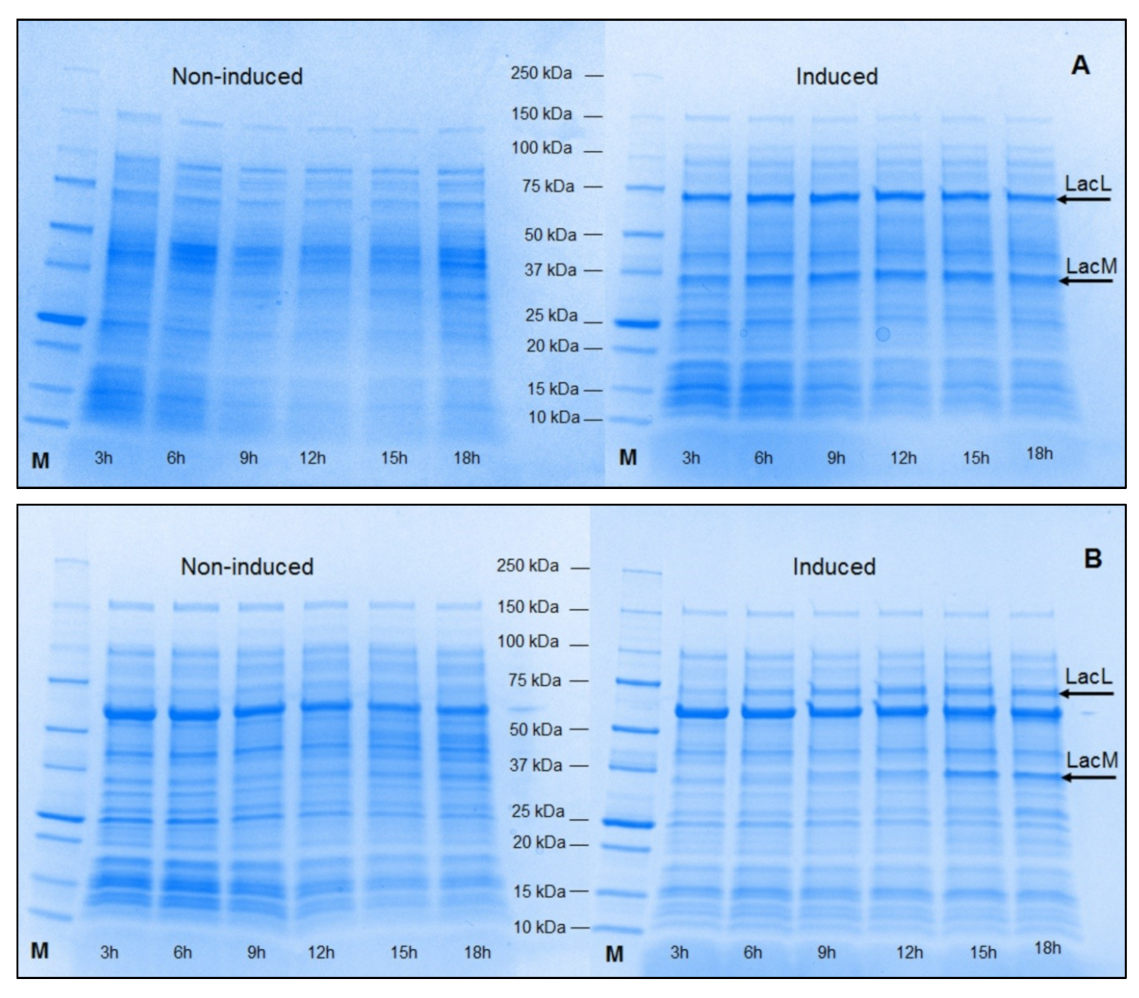
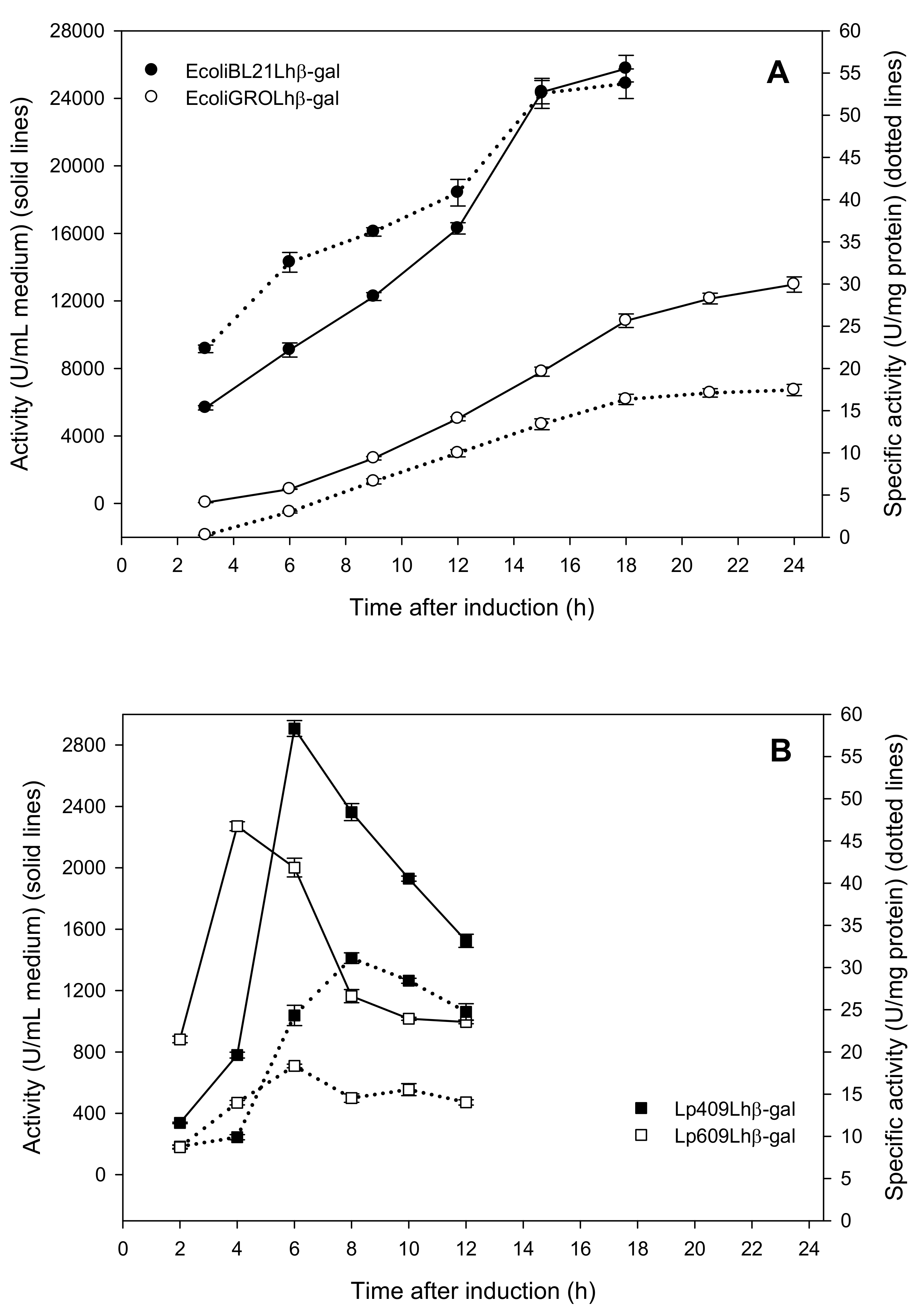
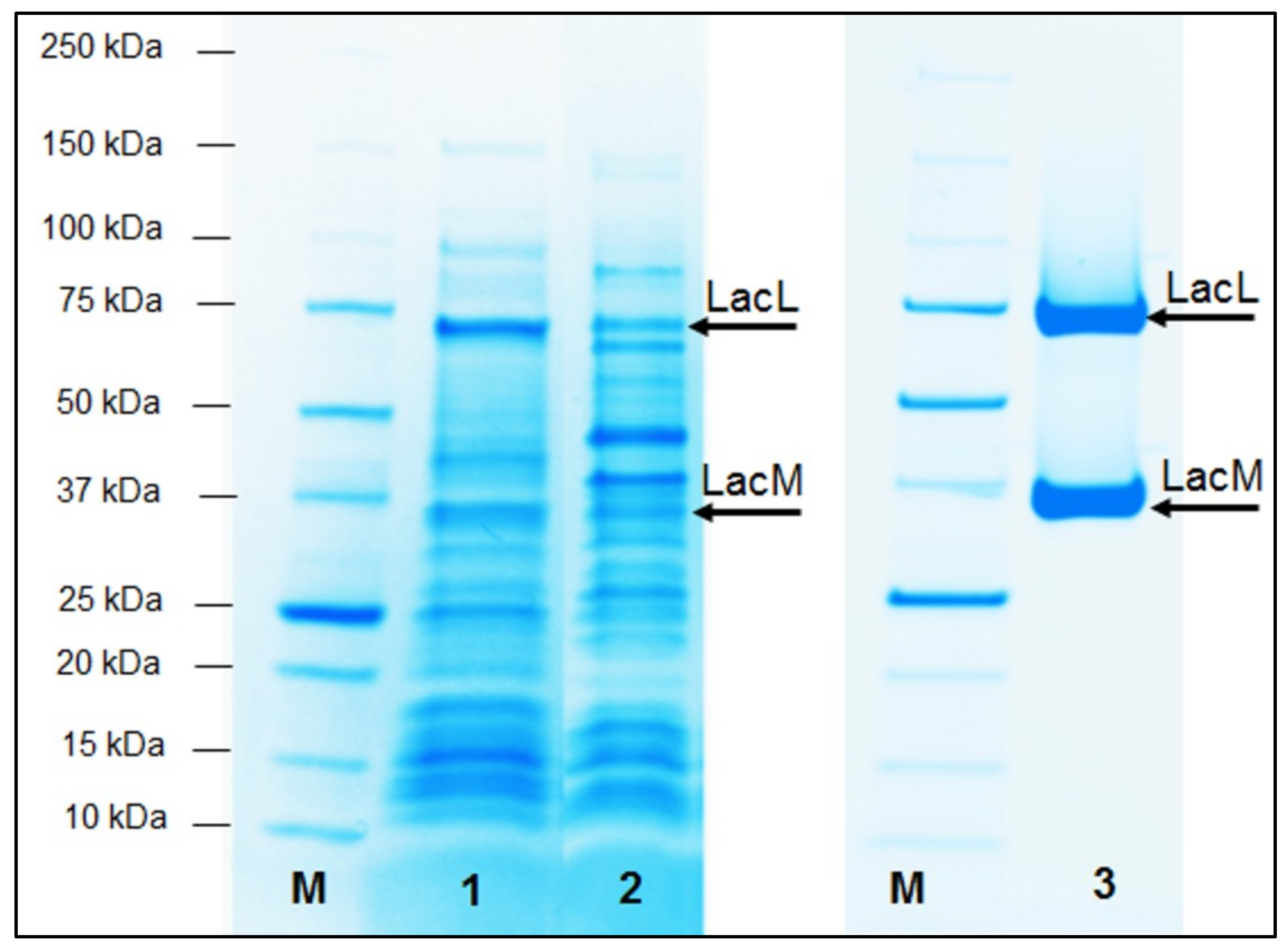
2.1. Production of Recombinant β-Galactosidases from L. helveticus in E. coli
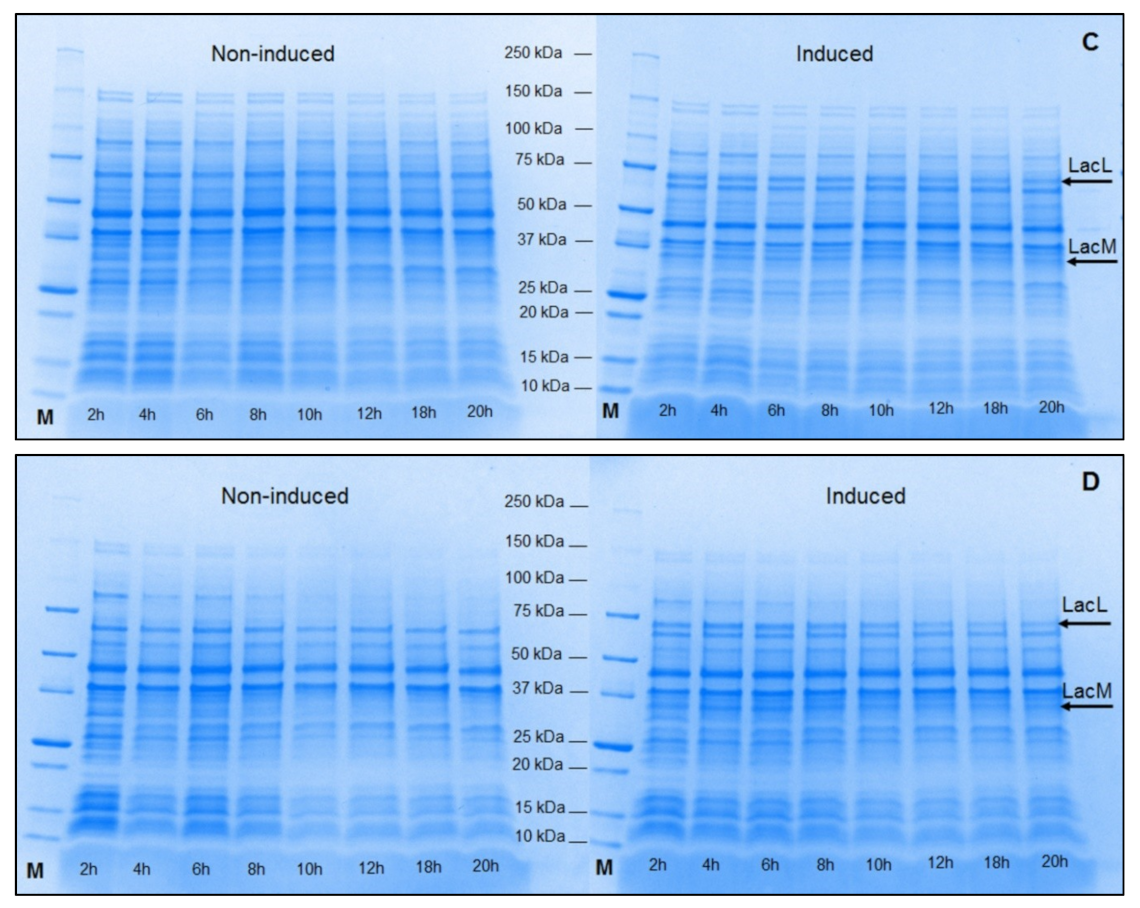
2.2. Production of Recombinant β-Galactosidases from L. helveticus in L. plantarum
2.3. Biochemical Characterization
2.3.1. Enzyme Kinetics
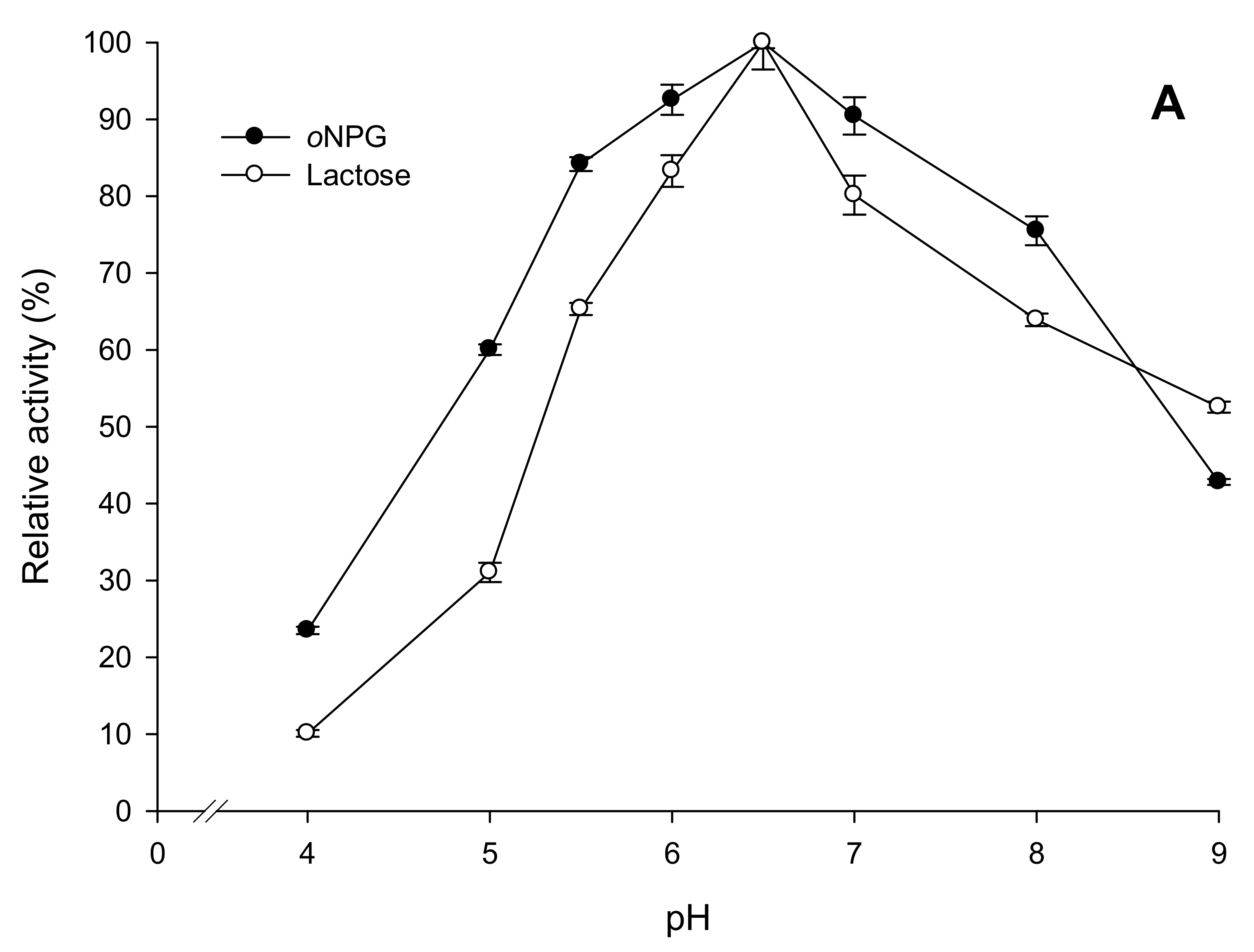
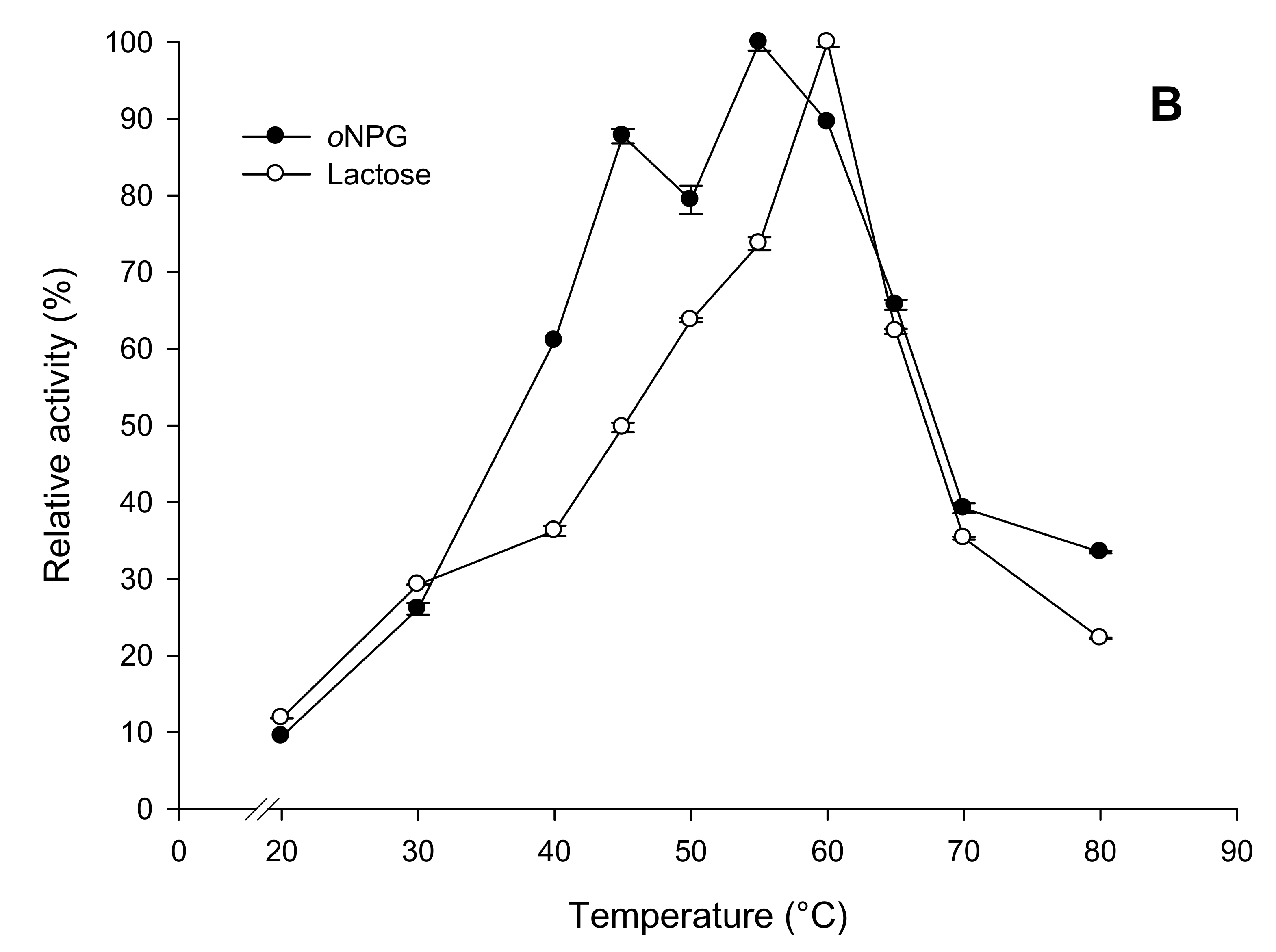
2.3.2. Effects of Temperature and pH on Enzyme Activity and Stability
2.3.3. Effects of Various Cations on Enzyme Activity
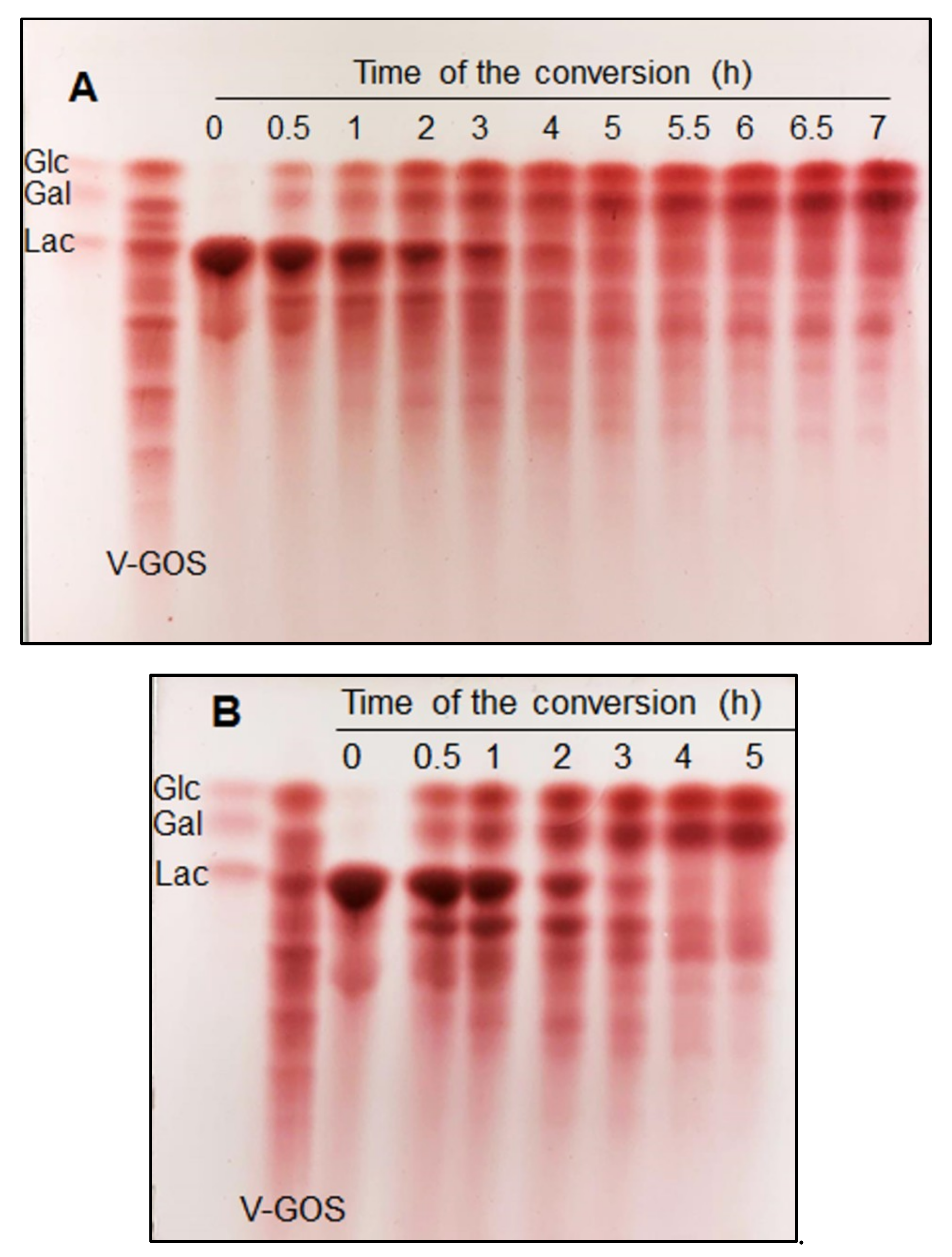
2.4. GOS Formation
3. Materials and Methods
3.1. Bacterial Strains and Culture Conditions
3.2. Chemicals and Enzymes
3.3. DNA Manipulation
3.4. Construction of Plasmids and Expression in E. coli
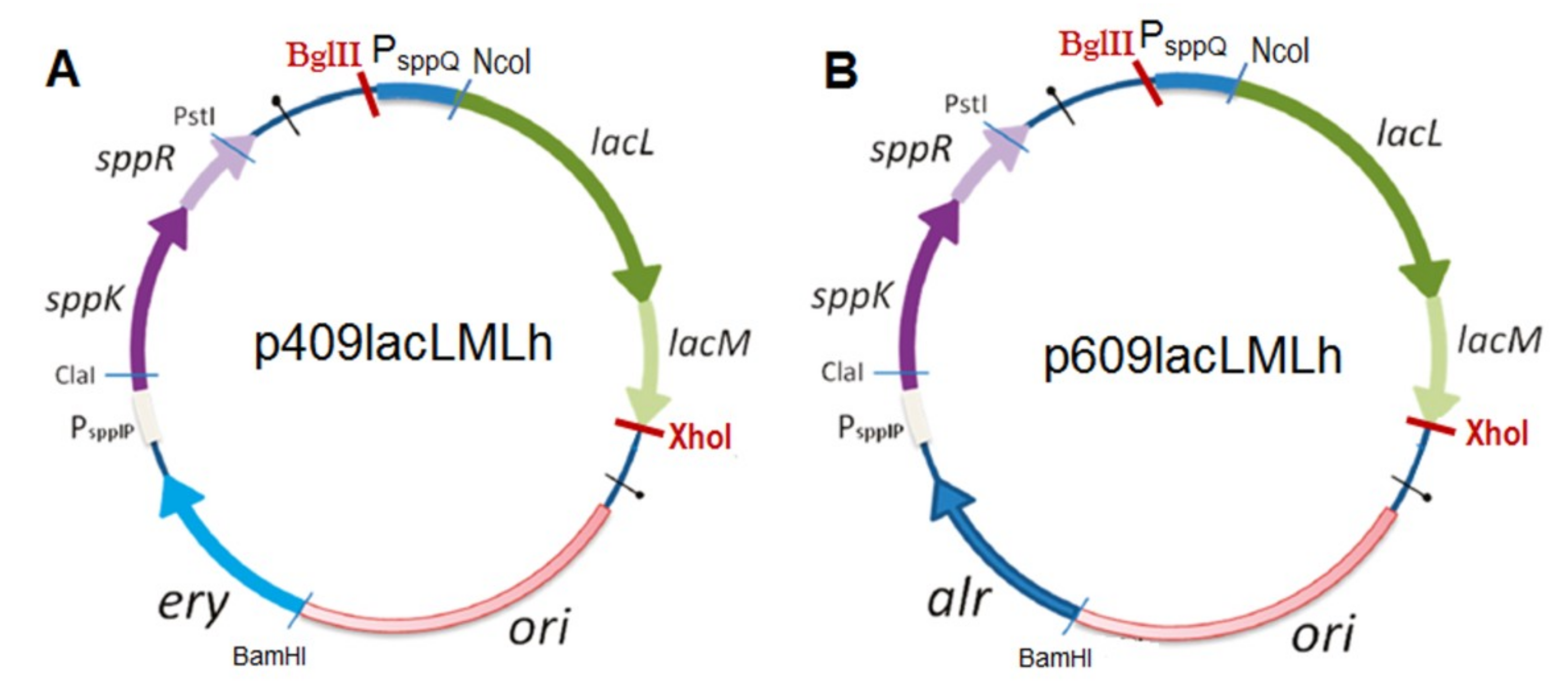
3.5. Construction of Plasmids and Expression in L. plantarum
3.6. Fermentation and Purification of Recombinant β-Galactosidase
3.7. Gel electrophoresis Analysis
3.8. β-Galactosidase Assays
3.9. Determination of Protein Concentration
3.10. Biochemical Characterization of Recombinant β-Galactosidases
3.11. Formation of Galacto-Oligosaccharides (GOS)
3.12. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ljungh, A.; Wadström, T. Lactic acid bacteria as probiotics. Curr. Issues Intest. Microbiol. 2006, 7, 73–89. [Google Scholar] [PubMed]
- Nagpal, R.; Kumar, A.; Kumar, M.; Behare, P.V.; Jain, S.; Yadav, H. Probiotics, their health benefits and applications for developing healthier foods: A review. FEMS Microbiol. Lett. 2012, 334, 1–15. [Google Scholar] [CrossRef]
- Gatti, M.; Trivisano, C.; Fabrizi, E.; Neviani, E.; Gardini, F. Biodiversity among Lactobacillus helveticus strains isolated from different natural whey starter cultures as revealed by classification trees. Appl. Environ. Microbiol. 2004, 70, 182–190. [Google Scholar] [CrossRef] [PubMed]
- Fortina, M.G.; Ricci, G.; Mora, D.; Guglielmetti, S.; Manachini, P.L. Unusual organization for lactose and galactose gene clusters in Lactobacillus helveticus. Appl. Environ. Microbiol. 2003, 69, 3238–3243. [Google Scholar] [CrossRef] [PubMed]
- de Macías, M.E.; Manca de Nadra, M.C.; Strasser de Saad, A.M.; Pesce de Ruiz Holgado, A.A.; Oliver, G. Isolation and properties of β-galactosidase of a strain of Lactobacillus helveticus isolated from natural whey starter. J. Appl. Biochem. 1983, 5, 275–281. [Google Scholar]
- Mollet, B.; Pilloud, N. Galactose utilization in Lactobacillus helveticus: Isolation and characterization of the galactokinase (galK) and galactose-1-phosphate uridyl transferase (galT) genes. J. Bacteriol. 1991, 173, 4464–4473. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.H.; Splechtna, B.; Yamabhai, M.; Haltrich, D.; Peterbauer, C. Cloning and expression of the β-galactosidase genes from Lactobacillus reuteri in Escherichia coli. J. Biotechnol. 2007, 129, 581–591. [Google Scholar] [CrossRef]
- Nguyen, T.H.; Splechtna, B.; Krasteva, S.; Kneifel, W.; Kulbe, K.D.; Divne, C.; Haltrich, D. Characterization and molecular cloning of a heterodimeric β-galactosidase from the probiotic strain Lactobacillus acidophilus R22. FEMS Microbiol. Lett. 2007, 269, 136–144. [Google Scholar] [CrossRef]
- Iqbal, S.; Nguyen, T.H.; Nguyen, T.T.; Maischberger, T.; Haltrich, D. β-galactosidase from Lactobacillus plantarum WCFS1: Biochemical characterization and formation of prebiotic galacto-oligosaccharides. Carbohydr. Res. 2010, 345, 1408–1416. [Google Scholar] [CrossRef]
- Maischberger, T.; Leitner, E.; Nitisinprasert, S.; Juajun, O.; Yamabhai, M.; Nguyen, T.H.; Haltrich, D. β-Galactosidase from Lactobacillus pentosus: Purification, characterization and formation of galacto-oligosaccharides. Biotechnol. J. 2010, 5, 838–847. [Google Scholar] [CrossRef]
- Iqbal, S.; Nguyen, T.H.; Nguyen, H.A.; Nguyen, T.T.; Maischberger, T.; Kittl, R.; Haltrich, D. Characterization of a heterodimeric GH2 β-galactosidase from Lactobacillus sakei Lb790 and formation of prebiotic galacto-oligosaccharides. J. Agric. Food Chem. 2011, 59, 3803–3811. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.T.; Nguyen, H.A.; Arreola, S.L.; Mlynek, G.; Djinović-Carugo, K.; Mathiesen, G.; Nguyen, T.H.; Haltrich, D. Homodimeric β-galactosidase from Lactobacillus delbrueckii subsp. bulgaricus DSM 20081: Expression in Lactobacillus plantarum and biochemical characterization. J. Agric. Food Chem. 2012, 60, 1713–1721. [Google Scholar] [CrossRef] [PubMed]
- Geiger, B.; Nguyen, H.M.; Wenig, S.; Nguyen, H.A.; Lorenz, C.; Kittl, R.; Mathiesen, G.; Eijsink, V.G.H.; Haltrich, D.; Nguyen, T.H. From by-product to valuable components: Efficient enzymatic conversion of lactose in whey using β-galactosidase from Streptococcus thermophilus. Biochem. Eng. J. 2016, 116, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Vaillancourt, K.; Moineau, S.; Frenette, M.; Lessard, C.; Vadeboncoeur, C. Galactose and lactose genes from the galactose-positive bacterium Streptococcus salivarius and the phylogenetically related galactose-negative bacterium Streptococcus thermophilus: Organization, sequence, transcription, and activity of the gal gene products. J. Bacteriol. 2002, 184, 785–793. [Google Scholar] [PubMed]
- Nakayama, T.; Amachi, T. β-Galactosidase, enzymology. In Encyclopedia of Bioprocess Technology: Fermentation, Biocatalysis, and Bioseparation; Flickinger, M.C., Drew, S.W., Eds.; John Willey and Sons: New York, NY, USA, 1999; Volume 3, pp. 1291–1305. [Google Scholar]
- Pivarnik, L.F.; Senegal, A.G.; Rand, A.G. Hydrolytic and transgalactosylic activities of commercial β-galactosidase (lactase) in food processing. Adv. Food Nutr. Res. 1995, 38, 1–102. [Google Scholar] [PubMed]
- Prenosil, J.E.; Stuker, E.; Bourne, J.R. Formation of oligosaccharises during enzymatic lactose hydrolysis: Part I: State of art. Biotechnol. Bioeng. 1987, 30, 1019–1025. [Google Scholar] [CrossRef] [PubMed]
- Nath, A.; Mondal, S.; Chakraborty, S.; Bhattacharjee, C.; Chowdhury, R. Production, purification, characterization, immobilization, and application of β-galactosidase: A review. Asia-Pac. J. Chem. Eng 2014, 9, 330–348. [Google Scholar] [CrossRef]
- Husain, Q. β-Galactosidases and their potential applications: A review. Crit. Rev. Biotechnol. 2010, 30, 41–62. [Google Scholar] [CrossRef]
- Petzelbauer, I.; Zeleny, R.; Reiter, A.; Kulbe, K.D.; Nidetzky, B. Development of an ultra-high-temperature process for the enzymatic hydrolysis of lactose: II. Oligosaccharide formation by two thermostable β-glycosidases. Biotechnol. Bioeng. 2000, 69, 140–149. [Google Scholar] [CrossRef]
- Roberfroid, M.; Gibson, G.R.; Hoyles, L.; McCartney, A.L.; Rastall, R.; Rowland, I.; Wolvers, D.; Watzl, B.; Szajewska, H.; Stahl, B.; et al. Prebiotic effects: Metabolic and health benefits. Br. J. Nutr. 2010, 104, S1–S63. [Google Scholar] [CrossRef]
- Gibson, G.R.; Probert, H.M.; Loo, J.V.; Rastall, R.A.; Roberfroid, M.B. Dietary modulation of the human colonic microbiota: Updating the concept of prebiotics. Nutr. Res. Rev. 2004, 17, 259–275. [Google Scholar] [CrossRef] [PubMed]
- Sangwan, V.; Tomar, S.K.; Singh, R.R.B.; Singh, A.K.; Ali, B. Galactooligosaccharides: Novel components of designer foods. J. Food Sci. 2011, 76, R103–R111. [Google Scholar] [CrossRef] [PubMed]
- De Man, J.C.; Rogosa, M.; Sharpe, M.E. A medium for the cultivation of lactobacilli. J. Appl. Bacteriol. 1960, 23, 130–135. [Google Scholar] [CrossRef]
- Arreola, S.L.; Intanon, M.; Suljic, J.; Kittl, R.; Pham, N.H.; Kosma, P.; Haltrich, D.; Nguyen, T.H. Two β-galactosidases from the human isolate Bifidobacterium breve DSM 20213: Molecular cloning and expression, biochemical characterization and synthesis of galacto-oligosaccharides. PLoS ONE 2014, 9, e104056. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Watzlawick, H.; Fridjonsson, O.; Hreggvidsson, G.; Altenbuchner, J. Improved soluble expression of the gene encoding amylolytic enzyme Amo45 by fusion with the mobile-loop-region of co-chaperonin GroES in Escherichia coli. Biocatal. Biotransfor. 2013, 31, 335–342. [Google Scholar] [CrossRef]
- Klaenhammer, T.R. Functional activities of Lactobacillus probiotics: Genetic mandate. Int. Dairy. J. 1998, 8, 497–505. [Google Scholar] [CrossRef]
- De Vos, W.M. Gene expression systems for lactic acid bacteria. Curr. Opin. Microbiol. 1999, 2, 289–295. [Google Scholar] [CrossRef]
- Kuipers, O.P.; De Ruyter, P.G.G.A.; Kleerebezem, M.; De Vos, W.M. Controlled overproduction of proteins by lactic acid bacteria. Trends Biotechnol. 1997, 15, 135–140. [Google Scholar] [CrossRef]
- Sørvig, E.; Mathiesen, G.; Naterstad, K.; Eijsink, V.G.H.; Axelsson, L. High-level, inducible gene expression in Lactobacillus sakei and Lactobacillus plantarum using versatile expression vectors. Microbiology 2005, 151, 2439–2449. [Google Scholar] [CrossRef]
- Halbmayr, E.; Mathiesen, G.; Nguyen, T.H.; Maischberger, T.; Peterbauer, C.K.; Eijsink, V.G.H.; Haltrich, D. High-level expression of recombinant β-galactosidases in Lactobacillus plantarum and Lactobacillus sakei using a sakacin p-based expression system. J. Agric. Food Chem. 2008, 56, 4710–4719. [Google Scholar] [CrossRef]
- Nguyen, T.T.; Mathiesen, G.; Fredriksen, L.; Kittl, R.; Nguyen, T.H.; Eijsink, V.G.H.; Haltrich, D.; Peterbauer, C.K. A food-grade system for inducible gene expression in Lactobacillus plantarum using an alanine racemase-encoding selection marker. J. Agric. Food Chem. 2011, 59, 5617–5624. [Google Scholar] [CrossRef] [PubMed]
- Sørvig, E.; Grönqvist, S.; Naterstad, K.; Mathiesen, G.; Eijsink, V.G.H.; Axelsson, L. Construction of vectors for inducible gene expression in Lactobacillus sakei and L. plantarum. FEMS Microbiol. Lett. 2003, 229, 119–126. [Google Scholar] [CrossRef]
- Staudigl, P.; Haltrich, D.; Peterbauer, C.K. L-Arabinose isomerase and D-xylose isomerase from Lactobacillus reuteri: Characterization, coexpression in the food grade host Lactobacillus plantarum, and application in the conversion of D-galactose and D-glucose. J. Agric. Food Chem. 2014, 62, 1617–1624. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.H.; Splechtna, B.; Steinböck, M.; Kneifel, W.; Lettner, H.P.; Kulbe, K.D.; Haltrich, D. Purification and characterization of two novel β-galactosidases from Lactobacillus reuteri. J. Agric. Food Chem. 2006, 54, 4989–4998. [Google Scholar] [CrossRef] [PubMed]
- Juers, D.H.; Rob, B.; Dugdale, M.L.; Rahimzadeh, N.; Giang, C.; Lee, M.; Matthews, B.W.; Huber, R.E. Direct and indirect roles of His-418 in metal binding and in the activity of β-galactosidase (E. coli). Protein Sci. 2009, 18, 1281–1292. [Google Scholar] [CrossRef] [PubMed]
- Juers, D.H.; Jacobson, R.H.; Wigley, D.; Zhang, X.J.; Huber, R.E.; Tronrud, D.E.; Matthews, B.W. High resolution refinement of β-galactosidase in a new crystal form reveals multiple metal-binding sites and provides a structural basis for alpha-complementation. Protein Sci. 2000, 9, 1685–1699. [Google Scholar] [CrossRef] [PubMed]
- Edwards, R.A.; Jacobson, A.L.; Huber, R.E. Thermal denaturation of β-galactosidase and of two site-specific mutants. Biochemistry 1990, 29, 11001–11008. [Google Scholar] [CrossRef] [PubMed]
- Kleerebezem, M.; Boekhorst, J.; van Kranenburg, R.; Molenaar, D.; Kuipers, O.P.; Leer, R.; Tarchini, R.; Peters, S.A.; Sandbrink, H.M.; Fiers, M.W.; et al. Complete genome sequence of Lactobacillus plantarum WCFS1. Proc. Natl. Acad. Sci. USA 2003, 100, 1990–1995. [Google Scholar] [CrossRef]
- Strych, U.; Penland, R.L.; Jimenez, M.; Krause, K.L.; Benedik, M.J. Characterization of the alanine racemases from two Mycobacteria. FEMS Microbiol. Lett. 2001, 196, 93–98. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
| Substrate | Method for Determination of β-Galactosidase Activity | Kinetic Parameter | EcoliBL21Lhβ-Gal |
|---|---|---|---|
| oNPG | Release of oNP | vmax,oNP (μmol min−1 mg−1) | 476 ± 66 |
| Km,oNPG (mM) | 1.40 ± 0.30 | ||
| kcat (s−1) | 865 ± 120 | ||
| kcat/Km (mM−1 s−1) | 618 | ||
| Ki,s (mM) | 3.6 ± 0.8 | ||
| Lactose | Release of D-glucose | vmax,Glucose (μmol min−1 mg−1) | 11.1 ± 0.2 |
| Km,Lactose (mM) | 15.7 ± 1.3 | ||
| kcat (s−1) | 20.2 ± 0.3 | ||
| kcat/Km (mM−1 s−1) | 1.29 |
| (A) | pH | τ1/2c (h) | (B) | Temperature (°C) | τ1/2 (h) |
|---|---|---|---|---|---|
| 4.0 | 1 | 4 | 6 months | ||
| 5.0 | 8 | 20 | 2 months | ||
| 5.5 | 816 | 30 | 216 | ||
| 6.0 | 552 | 40 | 5 | ||
| 6.5 | 144 | 50 | 0.33 | ||
| 7.0 | 48 | 40 (+1 mM MgCl2) | 72 | ||
| 8.0 | 1 | 50 (+1 mM MgCl2) | 3 | ||
| 9.0 | 0.5 |
| Cation | Relative Activity (%) | ||
|---|---|---|---|
| 1 mM | 10 mM | 100 mM | |
| None | 100 | 100 | 100 |
| Na+ | 160 | 142 | 109 |
| K+ | 118 | 96 | 68 |
| K+ a,c | 204 | 204 | nd |
| Mn2+ a,c | 143 | 105 | nd |
| Mg2+ a,c | 152 | 117 | nd |
| Ca2+ a,c | 140 | 80 | nd |
| Cu2+ b,c | 17 | 7 | nd |
| Zn2+ a,c | 120 | 78 | nd |
| Strain or Plasmid | Relevant Characteristics | Reference |
|---|---|---|
| Strains | ||
| L. helveticus DSM 20075 | source of lacLM genes | DSMZ (Braunschweig, Germany) |
| L. plantarum WCFS1 | wild-type, expression host | [39] |
| L. plantarum TLG02 | Δalr, D-alanine auxotroph, expression host | [32] |
| E. coli NEB5α | cloning host | New England Biolabs (Ipswich, MA) |
| E. coli MB2159 | D-alanine auxotroph, cloning host | [40] |
| E. coli BL21 Star DE3 | expression host | Invitrogen (Carlsbad, CA) |
| E. coli T7 Express | expression host | Novagen (Darmstadt, Germany) |
| Plasmids | ||
| pET-21d(+) | T7 Promoter, Ampr, C-terminal His-Tag | Novagen (Darmstadt, Germany) |
| pGRO7 | plasmid encoding chaperones GroES and GroEL, Cmr | Takara (Shiga, Japan) |
| pSIP409 | spp-based expression vector, Ermr, gusA controlled by PsppQ | [30] |
| pSIP609 | derivative of pSIP409, erm replaced by alr | [32] |
| pET21lacLMLh | lacLM from L. helveticus in pET-21d(+) | current study |
| p409lacLMLh | pSIP409 derivative, containing lacLM from L. helveticus | current study |
| P609lacLMLh | pSIP609 derivative, containing lacLM from L. helveticus | current study |
| Primer | Sequence (5’-3’) | Reference Accession Number |
|---|---|---|
| FwdNcoI | GGCCCCATGGAAGCAAATATCAATTG | GenBank: AJ512877 |
| Rev1XhoI | GGCCCTCGAGTGAATTTAGAATAAATGAAAATTC | GenBank: AJ512877 |
| Rev2XhoI | GGCCCTCGAGTTAATTTAGAATAAATGAAAATTC | GenBank: AJ512877 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kittibunchakul, S.; Pham, M.-L.; Tran, A.-M.; Nguyen, T.-H. β-Galactosidase from Lactobacillus helveticus DSM 20075: Biochemical Characterization and Recombinant Expression for Applications in Dairy Industry. Int. J. Mol. Sci. 2019, 20, 947. https://doi.org/10.3390/ijms20040947
Kittibunchakul S, Pham M-L, Tran A-M, Nguyen T-H. β-Galactosidase from Lactobacillus helveticus DSM 20075: Biochemical Characterization and Recombinant Expression for Applications in Dairy Industry. International Journal of Molecular Sciences. 2019; 20(4):947. https://doi.org/10.3390/ijms20040947
Chicago/Turabian StyleKittibunchakul, Suwapat, Mai-Lan Pham, Anh-Minh Tran, and Thu-Ha Nguyen. 2019. "β-Galactosidase from Lactobacillus helveticus DSM 20075: Biochemical Characterization and Recombinant Expression for Applications in Dairy Industry" International Journal of Molecular Sciences 20, no. 4: 947. https://doi.org/10.3390/ijms20040947
APA StyleKittibunchakul, S., Pham, M.-L., Tran, A.-M., & Nguyen, T.-H. (2019). β-Galactosidase from Lactobacillus helveticus DSM 20075: Biochemical Characterization and Recombinant Expression for Applications in Dairy Industry. International Journal of Molecular Sciences, 20(4), 947. https://doi.org/10.3390/ijms20040947







