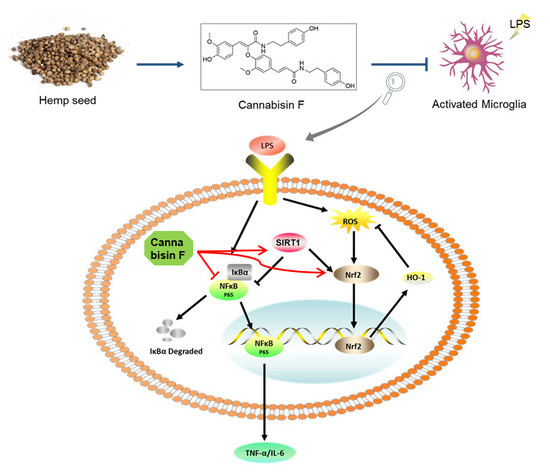
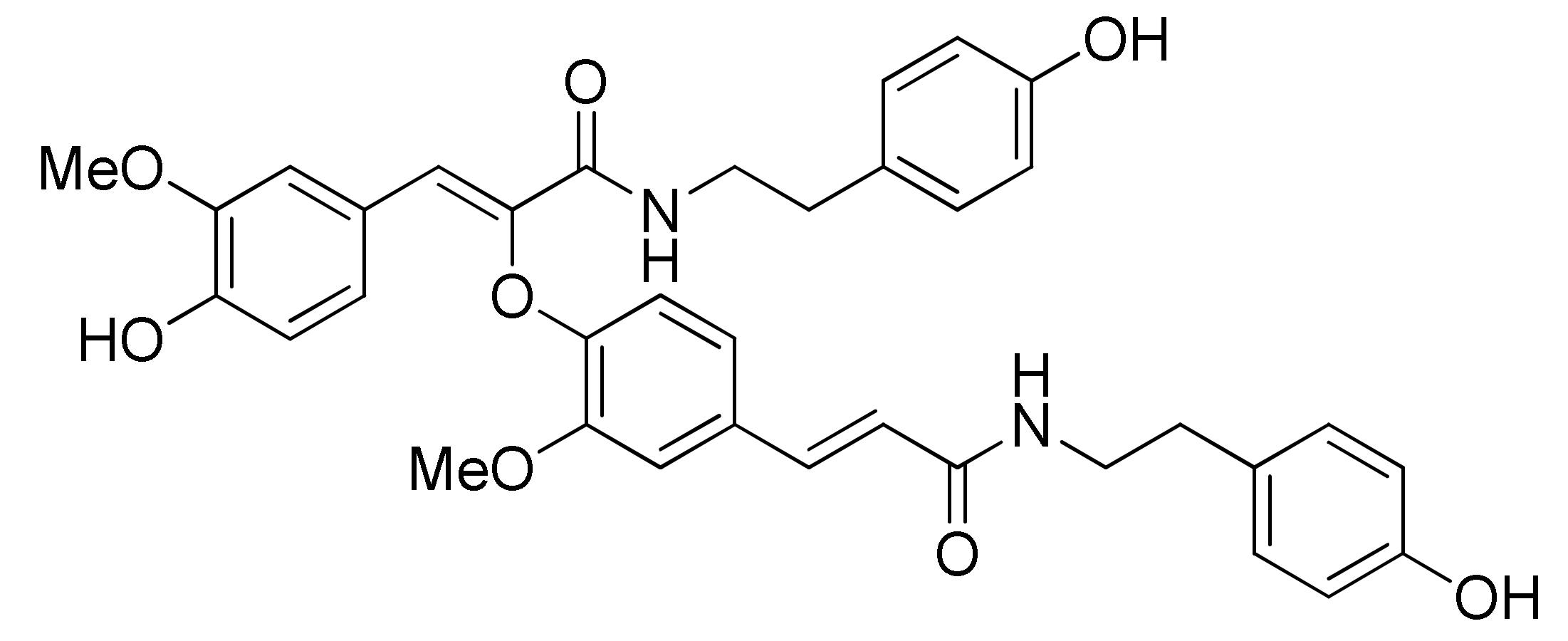
Cannabisin F from Hemp (Cannabis sativa) Seed Suppresses Lipopolysaccharide-Induced Inflammatory Responses in BV2 Microglia as SIRT1 Modulator
Abstract
1. Introduction
2. Results
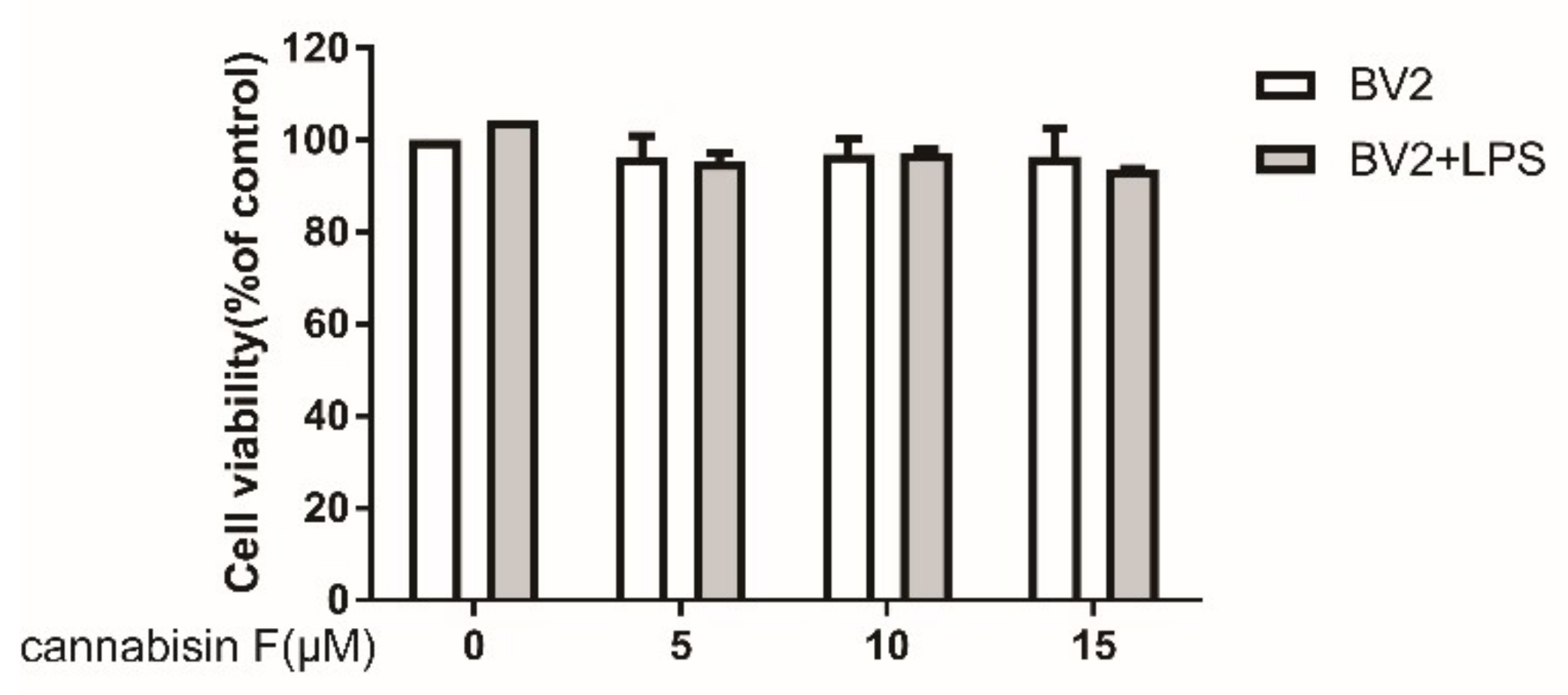
2.1. Cannabisin F Did Not Affect the Viability of BV2 Microglia
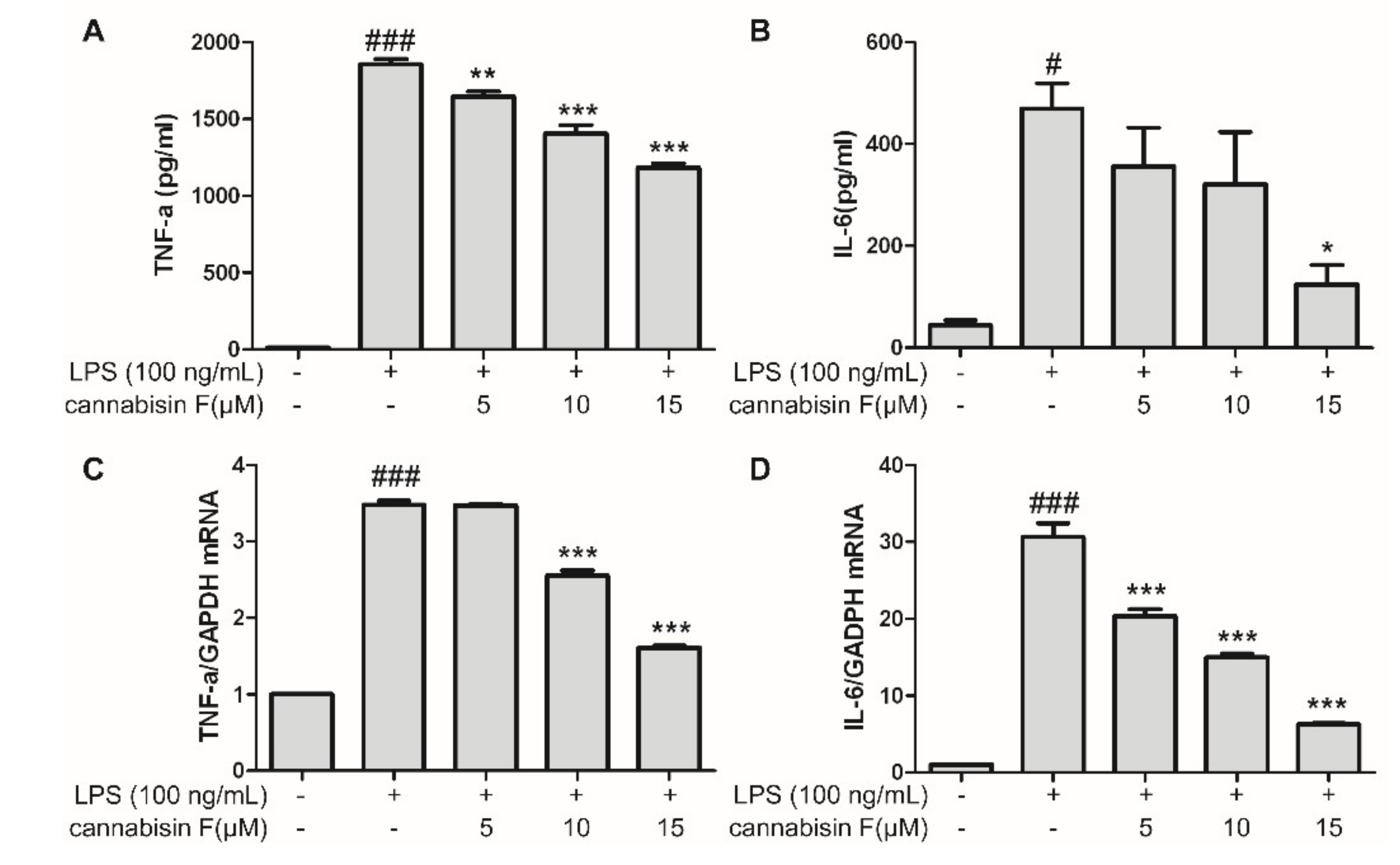
2.2. Cannabisin F Prevented the Production of IL-6 and TNF-α
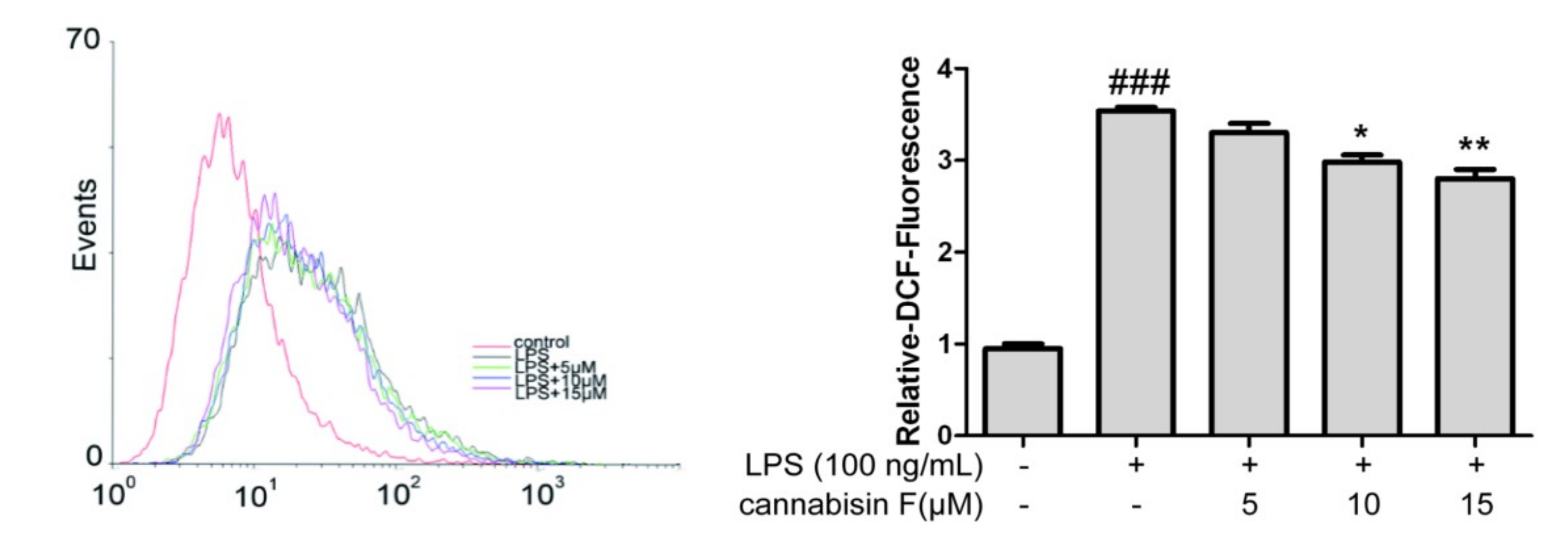
2.3. Cannabisin F Suppressed the Production of Intracellular ROS
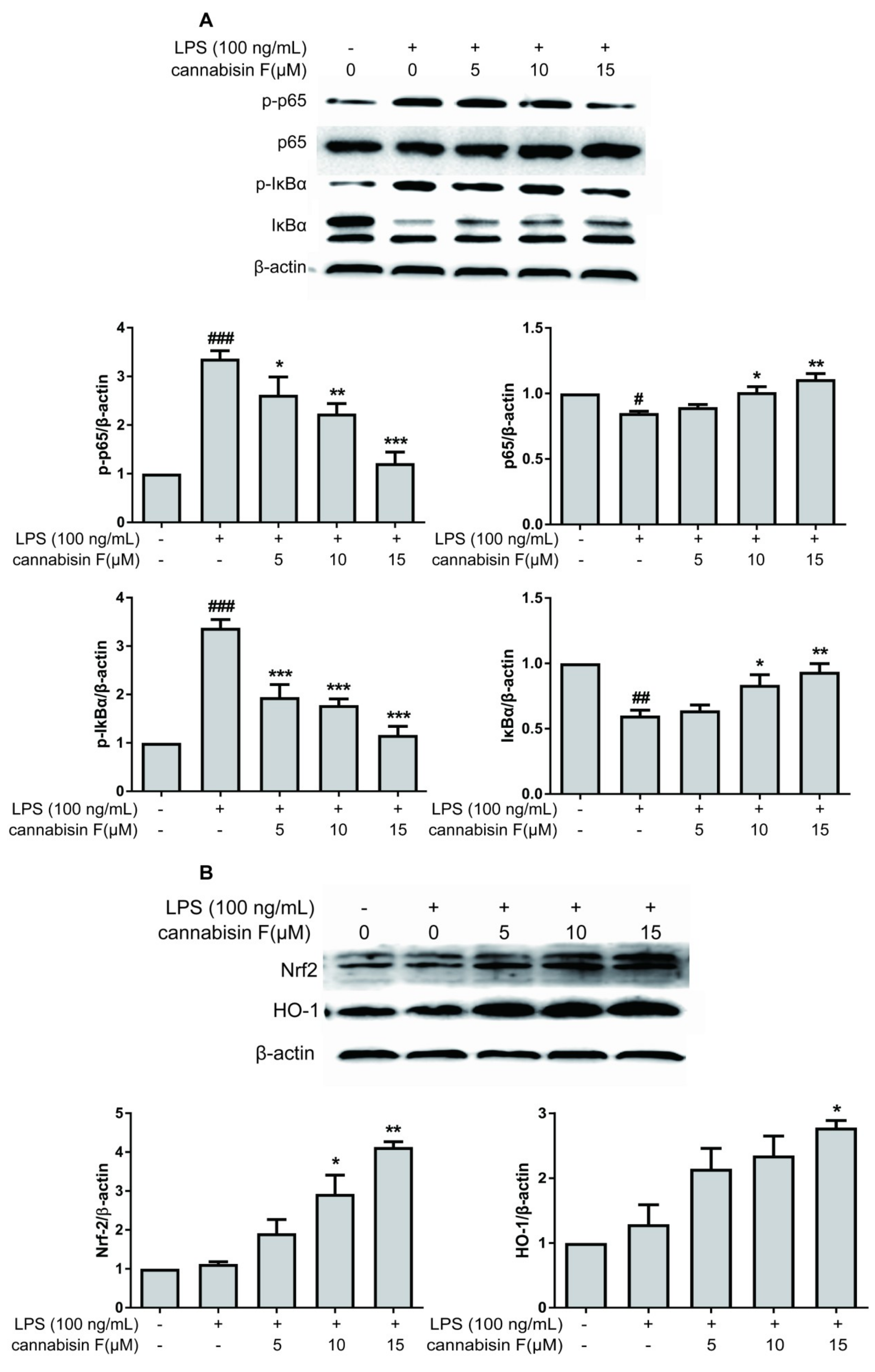
2.4. Cannabisin F Prevented NF-κB Activation and Promoted the Expression of Antioxidant Protein Nrf2/HO-1 in LPS-Stimulated BV2 Microglia Cells
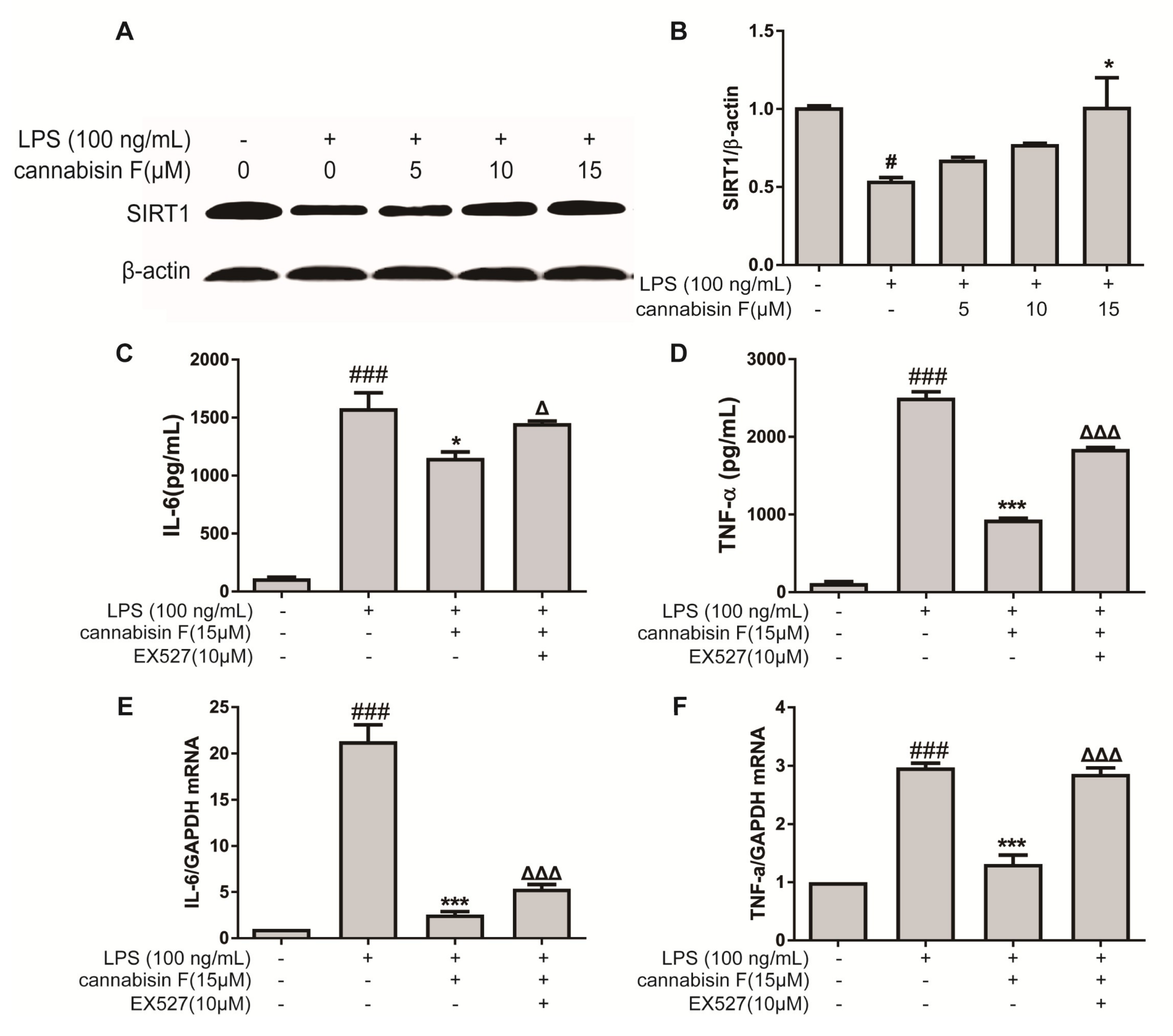
2.5. Cannabisin F Increased the Expression of SIRT1 and SIRT1 Inhibitor EX527 Reversed Anti-Inflammatory Action of Cannabisin F
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Cell Culture and Drug Treatment
4.3. Cell Viability Assay
4.4. Enzyme-Linked Immunosorbent Assay (ELISA)
4.5. Quantitative real-time PCR (RT-qPCR)
4.6. Measurement of Intracellular Reactive Oxygen Species (ROS)
4.7. Protein Extraction and Western Blot Analysis
4.8. Statistical Analyses
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ARE | Antioxidant response elements |
| AD | Alzheimer’s disease |
| CCK-8 | Cell Counting Kit-8 |
| CNS | Central nervous system |
| ECL | Chemiluminescent substrate |
| ELISA | Enzyme-linked immunosorbent assay |
| FBS | Fetal bovine serum |
| HO-1 | Heme Oxygenase-1 |
| IκB | Inhibit proteins of nuclear factor kappaB |
| IL-6 | Interleukin-6 |
| LPS | Lipopolysaccharide |
| MyD88 | Myeloid differentiation factor 88 |
| NAD+ | Nicotinamide adenine dinucleotide |
| NF-κB | Nuclear factor kappa B |
| NMR | Nuclear magnetic resonance |
| Nrf2 | Nuclear factor erythroid-2 related factor 2 |
| RIPA | Radio Immunoprecipitation Assay |
| ROS | Reactive oxygen species |
| PBS | Phosphate buffer solution |
| PD | Parkinson’s disease |
| PMSF | Phenylmethanesulfonyl fluoride |
| qRT-PCR | Quantitative real-time PCR |
| SDS-PAGE | Sodium dodecyl sulfate polyacrylamide gels |
| SIRT1 | Silent information regulator 1 |
| TLR4 | Toll-like receptor 4 |
| TNF-α | Tumor necrosis factor α |
References
- Ransohoff, R.M.; Perry, V.H. Microglial physiology: Unique stimuli, specialized responses. Annu. Rev. Immunol. 2009, 27, 119–145. [Google Scholar] [CrossRef] [PubMed]
- Callaway, J.C. Hempseed as a nutritional resource: An overview. Euphytica 2004, 140, 65–72. [Google Scholar] [CrossRef]
- Montserrat-de La Paz, S.; Marín-Aguilar, F.; García-Giménez, M.D.; Fernández-Arche, M.A. Hemp (Cannabis sativa L.) seed oil: Analytical and phytochemical characterization of the unsaponifiable fraction. J. Agric. Food Chem. 2014, 62, 1105–1110. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.; Ten, Z.; Wang, X.; Yang, X. Physicochemical and functional properties of hemp (Cannabis sativa L.) protein isolate. J. Agric. Food Chem. 2006, 54, 8945–8950. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.W.; Bian, Z.X.; Zhu, L.X.; Wu, J.C.Y.; Sung, J.J.Y. Efficacy of a chinese herbal proprietary medicine (Hemp Seed Pill) for functional constipation. Am. J. Gastroenterol. 2011, 106, 120–129. [Google Scholar] [CrossRef] [PubMed]
- Richard, M.N.; Ganguly, R.; Steigerwald, S.N.; Al-Khalifa, A.; Pierce, G.N. Dietary hempseed reduces platelet aggregation. J. Thromb. Haemost. 2010, 5, 424–425. [Google Scholar] [CrossRef]
- Rivest, S. Cannabinoids in microglia: A new trick for immune surveillance and neuroprotection. Neuron 2006, 49, 4–8. [Google Scholar] [CrossRef]
- Đolić, M.B.; Rajaković-Ognjanović, V.N.; Marković, J.P.; Janković-Mandić, L.J.; Mitrić, M.N.; Onjia, A.E.; Rajaković, L.V. The effect of different extractants on lead desorption from a natural mineral. Appl. Surf. Sci. 2015, 324, 221–231. [Google Scholar] [CrossRef]
- Luo, J.; Yin, J.H.; Wu, H.Z.; Wei, Q. Extract from Fructus cannabis activating calcineurin improved learning and memory in mice with chemical drug-induced dysmnesia. Acta Pharmacol. Sin. 2003, 24, 1137–1142. [Google Scholar]
- Yan, X.; Tang, J.; Dos Santos Passos, C.; Nurisso, A.; Simões-Pires, C.A.; Ji, M.; Lou, H.; Fan, P. Characterization of lignanamides from hemp (Cannabis sativa L.) seed and their antioxidant and acetylcholinesterase inhibitory activities. J. Agric. Food Chem. 2015, 63, 10611–10619. [Google Scholar] [CrossRef]
- Zhou, Y.; Wang, S.; Lou, H.; Fan, P. Chemical constituents of hemp (Cannabis sativa L.) seed with potential anti-neuroinflammatory activity. Phytochem. Lett. 2018, 23, 57–61. [Google Scholar] [CrossRef]
- Cai, P.; Xun, F.U.; Deng, A.G.; Zhan, X.J.; Cai, G.M.; Shun-Xiang, L.I. Anti-aging effect of hemp seed oil, protein and lignanamide of bama on old mice. Cent. South Pharm. 2010, 8, 165. [Google Scholar]
- Lin, X.; Zhen, M.; Chen, L. The effect of different extractants from Hemp seed in mice with experimental Alzheimer’s disease. Pharm. Clin. Chin. Mater. Med. 2016, 32, 130. [Google Scholar]
- Frassinetti, S.; Moccia, E.; Caltavuturo, L.; Gabriele, M.; Longo, V.; Bellani, L.; Giorgi, G.; Giorgetti, L. Nutraceutical potential of hemp (Cannabis sativa L.) seeds and sprouts. Food Chem. 2018, 262, 56–66. [Google Scholar] [CrossRef] [PubMed]
- Wu, N.; Shen, Q.; Cai, G.M.; Zhao, Y.L.; He, Q.; Wang, F. Identification and free radical scavenging activity of lignanamide extract from Fructus cannabis of Bama. Acta Chim. Sin. 2009, 67, 700–704. [Google Scholar]
- Luo, Q.; Yan, X.; Bobrovskaya, L.; Ji, M.; Yuan, H.; Lou, H.; Fan, P. Anti-neuroinflammatory effects of grossamide from hemp seed via suppression of TLR-4-mediated NF-κB signaling pathways in lipopolysaccharide-stimulated BV2 microglia cells. Mol. Cell. Biochem 2017, 428, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Perry, V.H.; Holmes, C. Microglial priming in neurodegenerative disease. Nat. Rev. Neurol. 2014, 10, 217. [Google Scholar] [CrossRef] [PubMed]
- Kreutzberg, G.W. Microglia: A sensor for pathological events in the CNS. Trends Neurosci. 1996, 19, 312–318. [Google Scholar] [CrossRef]
- Hanisch, U.; Kettenmann, H. Microglia: Active sensor and versatile effector cells in the normal and pathologic brain. Nat. Neurosci. 2007, 10, 1387. [Google Scholar] [CrossRef]
- Lu, Y.; Yeh, W.; Ohashi, P.S. LPS/TLR4 signal transduction pathway. Cytokine 2008, 42, 145–151. [Google Scholar] [CrossRef]
- Taylor, J.M.; Main, B.S.; Crack, P.J. Neuroinflammation and oxidative stress: Co-conspirators in the pathology of Parkinson’s disease. Neurochem. Int. 2013, 62, 803–819. [Google Scholar] [CrossRef] [PubMed]
- Witte, M.E.; Geurts, J.J.G.; de Vries, H.E.; van der Valk, P.; van Horssen, J. Mitochondrial dysfunction: A potential link between neuroinflammation and neurodegeneration? Mitochondrion 2010, 10, 411–418. [Google Scholar] [CrossRef] [PubMed]
- De Vries, H.E.; Witte, M.; Hondius, D.; Rozemuller, A.J.M.; Drukarch, B.; Hoozemans, J.; van Horssen, J. Nrf2-induced antioxidant protection: A promising target to counteract ROS-mediated damage in neurodegenerative disease? Free Radic. Biol. Med. 2008, 45, 1375–1383. [Google Scholar] [CrossRef] [PubMed]
- Hybertson, B.M.; Gao, B.; Bose, S.K.; McCord, J.M. Oxidative stress in health and disease: The therapeutic potential of Nrf2 activation. Mol. Asp. Med. 2011, 32, 234–246. [Google Scholar] [CrossRef]
- Foresti, R.; Bains, S.K.; Pitchumony, T.S.; de Castro Brás, L.E.; Drago, F.; Dubois-Randé, J.; Bucolo, C.; Motterlini, R. Small molecule activators of the Nrf2-HO-1 antioxidant axis modulate heme metabolism and inflammation in BV2 microglia cells. Pharmacol. Res. 2013, 76, 132–148. [Google Scholar] [CrossRef]
- Zhang, T.; Berrocal, J.G.; Frizzell, K.M.; Gamble, M.J.; DuMond, M.E.; Krishnakumar, R.; Yang, T.; Sauve, A.A.; Kraus, W.L. Enzymes in the NAD+ salvage pathway regulate SIRT1 activity at target gene promoters. J. Biol. Chem. 2009, 284, 20408. [Google Scholar] [CrossRef] [PubMed]
- Rajendrasozhan, S.; Yang, S.; Kinnula, V.L.; Rahman, I. SIRT1, an Anti-inflammatory and antiaging protein, is decreased in lungs of patients with chronic obstructive pulmonary disease. Am. J. Resp. Crit. Care 2008, 177, 861–870. [Google Scholar] [CrossRef] [PubMed]
- Salminen, A.; Huuskonen, J.; Ojala, J.; Kauppinen, A.; Kaarniranta, K.; Suuronen, T. Activation of innate immunity system during aging: NF-κB signaling is the molecular culprit of inflamm-aging. Ageing Res. Rev. 2008, 7, 83–105. [Google Scholar] [CrossRef]
- Chong, Z.Z.; Shang, Y.C.; Wang, S.; Maiese, K. SIRT1: New avenues of discovery for disorders of oxidative stress. Expert Opin. Ther. Targets 2012, 16, 167–178. [Google Scholar] [CrossRef]
- Wun, Z.; Lin, C.; Huang, W.; Huang, Y.; Xu, P.; Chang, W.; Wu, S.; Liou, C. Anti-inflammatory effect of sophoraflavanone G isolated from Sophora flavescens in lipopolysaccharide-stimulated mouse macrophages. Food Chem. Toxicol. 2013, 62, 255–261. [Google Scholar] [CrossRef]
- Catorce, M.N.; Gevorkian, G. LPS-induced murine neuroinflammation model: Main features and suitability for pre-clinical assessment of nutraceuticals. Curr. Neuropharmacol. 2016, 14, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Michaud, M.; Balardy, L.; Moulis, G.; Gaudin, C.; Peyrot, C.; Vellas, B.; Cesari, M.; Nourhashemi, F. Proinflammatory cytokines, aging, and age-related diseases. J. Am. Med. Dir. Assoc. 2013, 14, 877–882. [Google Scholar] [CrossRef] [PubMed]
- Yakovleva, T.; Bazov, I.; Watanabe, H.; Hauser, K.F.; Bakalkin, G. Transcriptional control of maladaptive and protective responses in alcoholics: A role of the NF-κB system. Brain Behav. Immun. 2011, 25, S29–S38. [Google Scholar] [CrossRef] [PubMed]
- Vallabhapurapu, S.; Karin, M. Regulation and function of NF-kappaB transcription factors in the immune system. Annu. Rev. Immunol. 2009, 27, 693–733. [Google Scholar] [CrossRef] [PubMed]
- Shinozaki, S.; Chang, K.; Sakai, M.; Shimizu, N.; Yamada, M.; Tanaka, T.; Nakazawa, H.; Ichinose, F.; Yamada, Y.; Ishigami, A.; et al. Inflammatory stimuli induce inhibitory S-nitrosylation of the deacetylase SIRT1 to increase acetylation and activation of p53 and p65. Sci. Signal 2014, 7, a106. [Google Scholar] [CrossRef] [PubMed]
- Kauppinen, A.; Suuronen, T.; Ojala, J.; Kaarniranta, K.; Salminen, A. Antagonistic crosstalk between NF-κB and SIRT1 in the regulation of inflammation and metabolic disorders. Cell. Signal. 2013, 25, 1939–1948. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.A.; Khan, M.; Jo, M.H.; Jo, M.G.; Amin, F.U.; Kim, M.O. Melatonin stimulates the SIRT1/Nrf2 signaling pathway counteracting lipopolysaccharide (LPS)-induced oxidative stress to rescue postnatal rat brain. CNS Neurosci. Ther. 2017, 23, 33–44. [Google Scholar] [CrossRef]
- Lim, J.L.; Wilhelmus, M.M.M.; de Vries, H.E.; Drukarch, B.; Hoozemans, J.J.M.; van Horssen, J. Antioxidative defense mechanisms controlled by Nrf2: State-of-the-art and clinical perspectives in neurodegenerative diseases. Arch. Toxicol. 2014, 88, 1773–1786. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, S.; Luo, Q.; Fan, P. Cannabisin F from Hemp (Cannabis sativa) Seed Suppresses Lipopolysaccharide-Induced Inflammatory Responses in BV2 Microglia as SIRT1 Modulator. Int. J. Mol. Sci. 2019, 20, 507. https://doi.org/10.3390/ijms20030507
Wang S, Luo Q, Fan P. Cannabisin F from Hemp (Cannabis sativa) Seed Suppresses Lipopolysaccharide-Induced Inflammatory Responses in BV2 Microglia as SIRT1 Modulator. International Journal of Molecular Sciences. 2019; 20(3):507. https://doi.org/10.3390/ijms20030507
Chicago/Turabian StyleWang, Shanshan, Qian Luo, and Peihong Fan. 2019. "Cannabisin F from Hemp (Cannabis sativa) Seed Suppresses Lipopolysaccharide-Induced Inflammatory Responses in BV2 Microglia as SIRT1 Modulator" International Journal of Molecular Sciences 20, no. 3: 507. https://doi.org/10.3390/ijms20030507
APA StyleWang, S., Luo, Q., & Fan, P. (2019). Cannabisin F from Hemp (Cannabis sativa) Seed Suppresses Lipopolysaccharide-Induced Inflammatory Responses in BV2 Microglia as SIRT1 Modulator. International Journal of Molecular Sciences, 20(3), 507. https://doi.org/10.3390/ijms20030507