Toll-Like Receptor Mediated Activation of Natural Autoantibody Producing B Cell Subpopulations in an Autoimmune Disease Model
Abstract
1. Introduction
2. Results
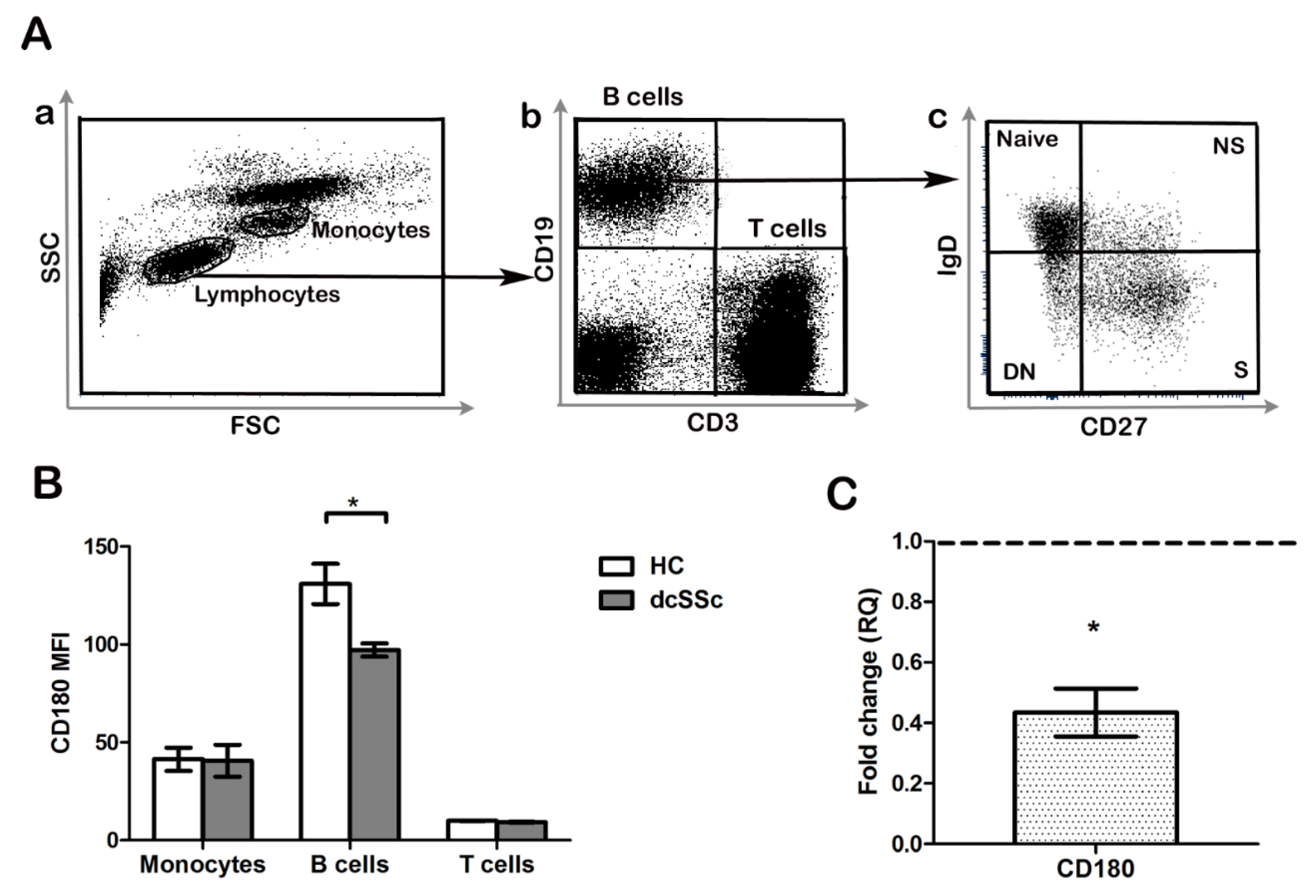
2.1. CD180 Expression Is Decreased in dcSSc B Cells
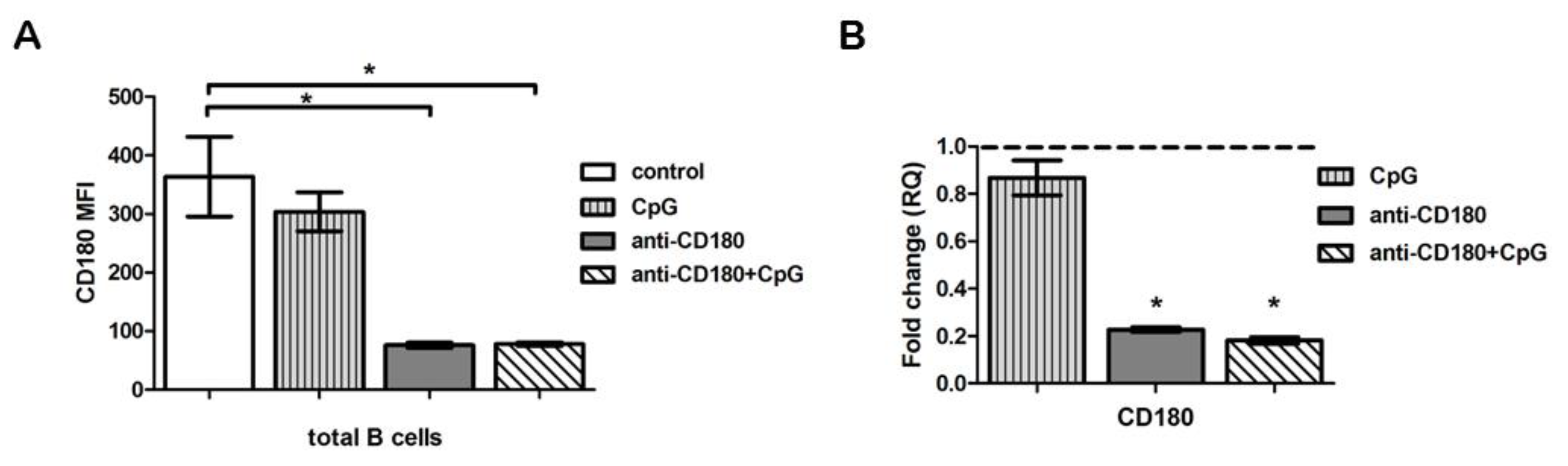
2.2. TLR Ligation Results in Reduced CD180 mRNA and Protein Expression of B Cells
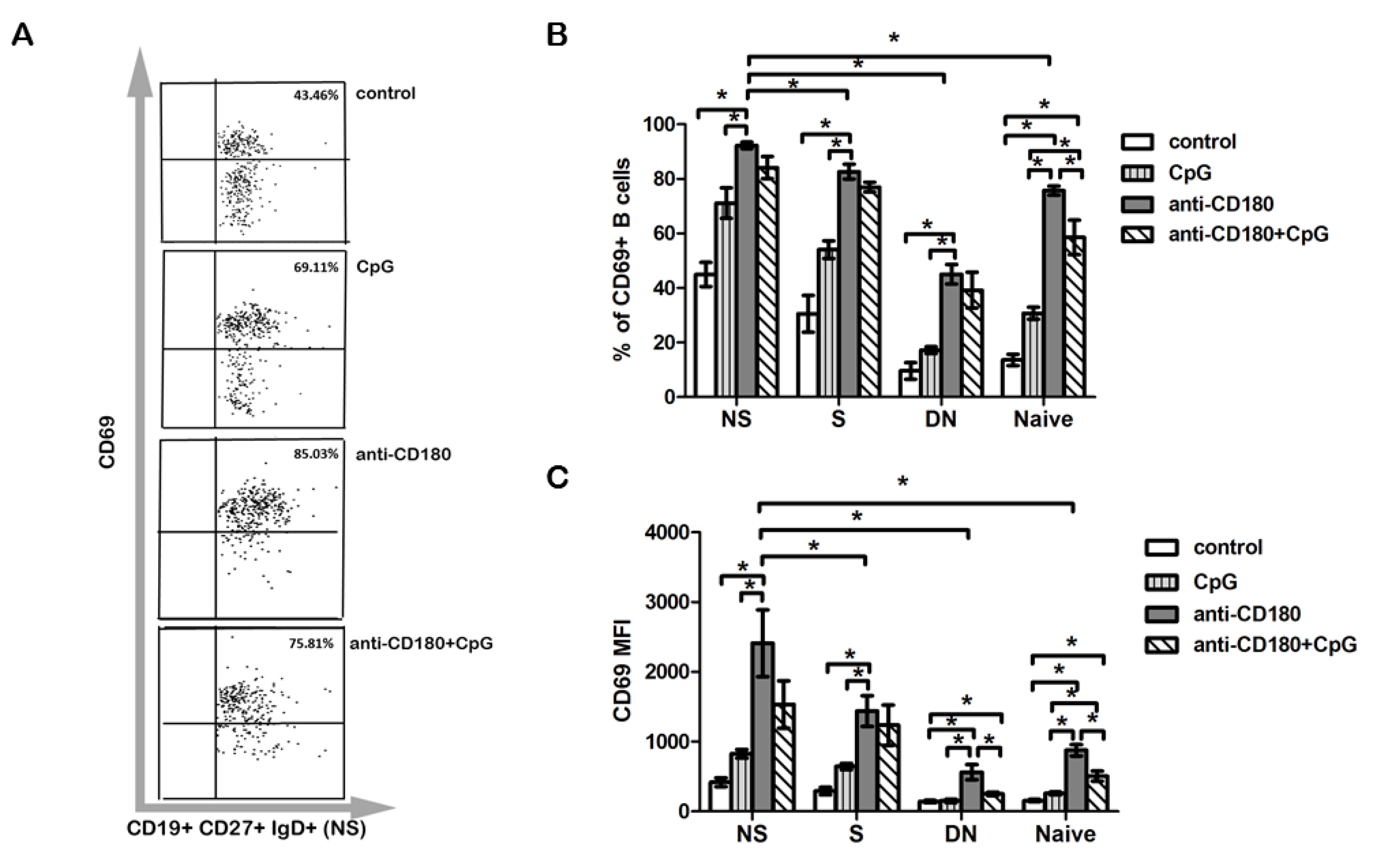
2.3. The Frequency of CD180+ Cells Is the Highest in the Non-Switched Memory B Cell Subset
2.4. Anti-CD180 Stimulation Resulted in Activation of B Cell Subsets
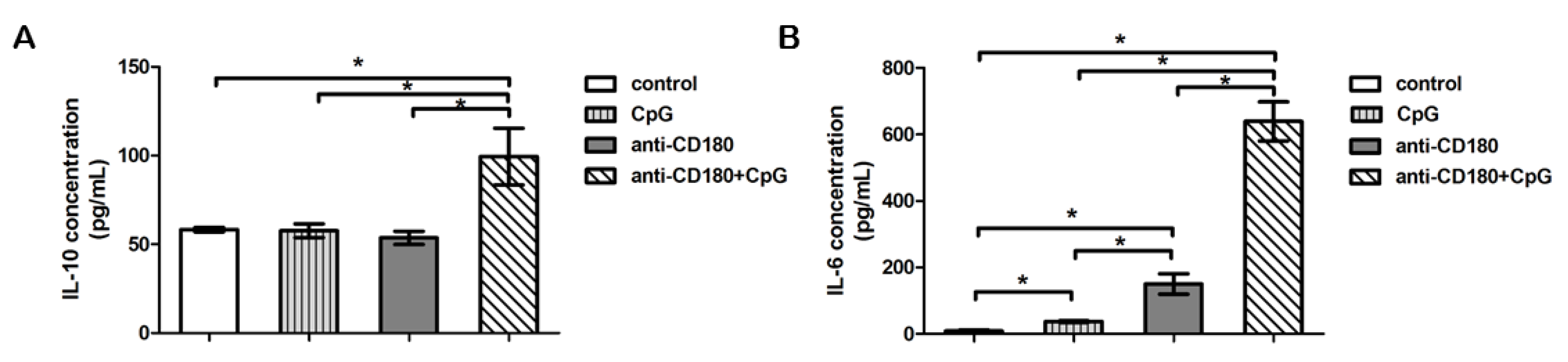
2.5. TLR Stimulation Differentially Affects IL-6 and IL-10 Production of B Cells
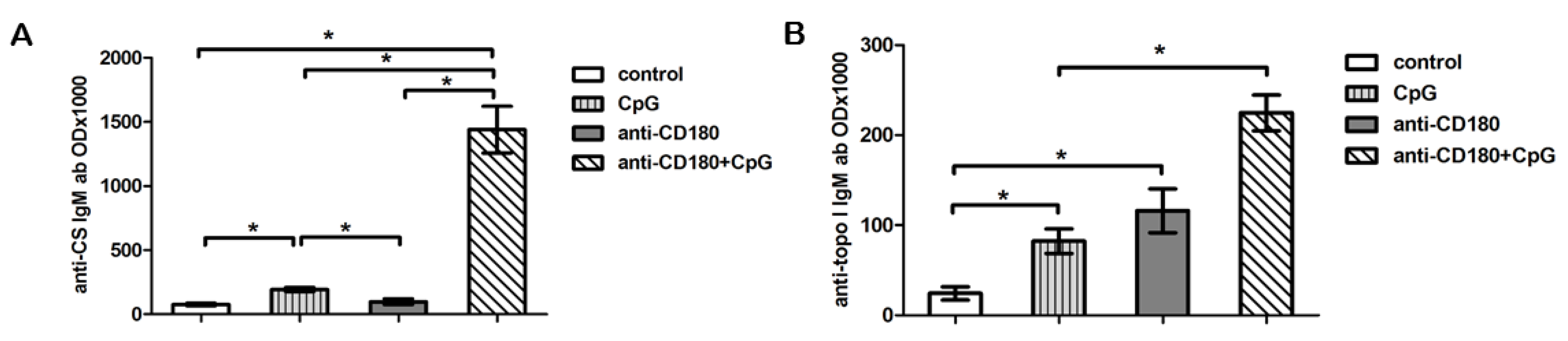
2.6. CD180 Stimulation Induces Natural Autoantibody Production
3. Discussion
4. Materials and Methods
4.1. Patients
4.2. Mononuclear Cell Isolation, B Cell Separation
4.3. Cell Stimulation
4.4. RNA Isolation, cDNA Synthesis, and qPCR
4.5. Flow Cytometric Analysis
4.6. IL-10 and IL-6 ELISA
4.7. Measurement of Natural and Pathologic Autoantibodies
4.8. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ANA | Antinuclear antibodies |
| Breg | Regulatory B cell |
| CpG | 5’—C—phosphate—G—3’ dinucleotide |
| CS | Citrate synthase |
| dcSSc | Diffuse cutaneous systemic sclerosis |
| DN B | Double negative B cell |
| ELISA | Enzyme-linked immunosorbent assay |
| FSC | Forward scatter |
| GAPDH | Glyceraldehyde 3-phosphate dehydrogenase |
| HC | Healthy control |
| Hu | Human |
| LEAF | Low endotoxin, azide-free |
| MFI | Mean fluorescence intensity |
| Ms | Mouse |
| MZ | Marginal zone |
| NS B | Non-switched B cell |
| PBMC | Peripheral blood mononuclear cells |
| PFA | Paraformaldehyde |
| qPCR | Quantitative polymerase chain reaction |
| RQ | Relative quantification |
| S B | Switched B cell |
| SLE | Systemic lupus erythematosus |
| SSc | Systemic sclerosis |
| SSC | Side scatter |
| TLR | Toll-like receptor |
| TMB | 3,3′,5,5′-Tetramethylbenzidine |
| Topo I | Topoisomerase I |
References
- Varga, J.; Trojanowska, M.; Kuwana, M. Pathogenesis of systemic sclerosis: Recent insights of molecular and cellular mechanisms and therapeutic opportunities. J. Scleroderma Relat. Disord. 2017, 2, 137–152. [Google Scholar] [CrossRef]
- Cabral-Marques, O.; Riemekasten, G. Functional autoantibodies directed against cell surface receptors in systemic sclerosis. J. Scleroderma Relat. Disord. 2017, 2, 160–168. [Google Scholar] [CrossRef]
- Johnson, M.E.; Grassetti, A.V.; Taroni, J.N.; Lyons, S.M.; Schweppe, D.; Gordon, J.K.; Spiera, R.F.; Lafyatis, R.; Anderson, P.J.; Gerber, S.A.; et al. Stress granules and RNA processing bodies are novel autoantibody targets in systemic sclerosis. Arthritis Res. 2016, 18, 27. [Google Scholar] [CrossRef] [PubMed]
- Senécal, J.; Hoa, S.; Yang, R.; Koenig, M. Pathogenic roles of autoantibodies in systemic sclerosis: Current understandings in pathogenesis. J. Scleroderma Relat. Disord. 2019. [Google Scholar] [CrossRef]
- Mannoor, K.; Xu, Y.; Chen, C. Natural autoantibodies and associated B cells in immunity and autoimmunity. Autoimmunity 2013, 46, 138–147. [Google Scholar] [CrossRef]
- Schultz, T.E.; Blumenthal, A. The RP105/MD-1 complex: Molecular signaling mechanisms and pathophysiological implications. J. Leukoc. Biol. 2017, 101, 183–192. [Google Scholar] [CrossRef]
- Valentine, M.A.; Clark, E.A.; Shu, G.L.; Norris, N.A.; Ledbetter, J.A. Antibody to a novel 95-kDa surface glycoprotein on human B cells induces calcium mobilization and B cell activation. J. Immunol. 1988, 140, 4071–4078. [Google Scholar]
- Chaplin, J.W.; Kasahara, S.; Clark, E.A.; Ledbetter, J.A. Anti-CD180 (RP105) activates B cells to rapidly produce polyclonal Ig via a T cell and MyD88-independent pathway. J. Immunol. 2011, 187, 4199–4209. [Google Scholar] [CrossRef]
- Divanovic, S.; Trompette, A.; Atabani, S.F.; Madan, R.; Golenbock, D.T.; Visintin, A.; Finberg, R.W.; Tarakhovsky, A.; Vogel, S.N.; Belkaid, Y.; et al. Negative regulation of Toll-like receptor 4 signaling by the Toll-like receptor homolog RP105. Nat. Immunol. 2005, 6, 571–578. [Google Scholar] [CrossRef]
- Fujita, K.; Akasaka, Y.; Kuwabara, T.; Wang, B.; Tanaka, K.; Kamata, I.; Yokoo, T.; Kinoshita, T.; Iuchi, A.; Akishima-Fukasawa, Y.; et al. Pathogenesis of lupus-like nephritis through autoimmune antibody produced by CD180-negative B lymphocytes in NZBWF1 mouse. Immunol. Lett. 2012, 144, 1–6. [Google Scholar] [CrossRef]
- Koarada, S.; Tada, Y.; Ushiyama, O.; Morito, F.; Suzuki, N.; Ohta, A.; Miyake, K.; Kimoto, M.; Nagasawa, K. B cells lacking RP105, a novel B cell antigen, in systemic lupus erythematosus. Arthritis Rheum. 1999, 42, 2593–2600. [Google Scholar] [CrossRef]
- Kikuchi, Y.; Koarada, S.; Nakamura, S.; Yonemitsu, N.; Tada, Y.; Haruta, Y.; Morito, F.; Ohta, A.; Miyake, K.; Horiuchi, T.; et al. Increase of RP105-lacking activated B cells in the peripheral blood and salivary glands in patients with Sjögren’s syndrome. Clin. Exp. Rheumatol. 2008, 26, 5–12. [Google Scholar] [PubMed]
- Eriksen, A.B.; Indrevær, R.L.; Holm, K.L.; Landskron, J.; Blomhoff, H.K. TLR9-signaling is required for turning retinoic acid into a potent stimulator of RP105 (CD180)-mediated proliferation and IgG synthesis in human memory B cells. Cell Immunol. 2012, 279, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Chaplin, J.W.; Chappell, C.P.; Clark, E.A. Targeting antigens to CD180 rapidly induces antigen-specific IgG, affinity maturation, and immunological memory. J. Exp. Med. 2013, 210, 2135–2146. [Google Scholar] [CrossRef] [PubMed]
- Koarada, S.; Tada, Y. RP105-negative B cells in systemic lupus erythematosus. Clin. Dev. Immunol. 2012, 2012, 259186. [Google Scholar] [CrossRef] [PubMed][Green Version]
- You, M.; Dong, G.; Li, F.; Ma, F.; Ren, J.; Xu, Y.; Yue, H.; Tang, R.; Ren, D.; Hou, Y. Ligation of CD180 inhibits IFN-α signaling in a Lyn-PI3K-BTK-dependent manner in B cells. Cell Mol. Immunol. 2017, 14, 192–202. [Google Scholar] [CrossRef]
- Rosser, E.C.; Mauri, C. Regulatory B cells: Origin, phenotype, and function. Immunity 2015, 42, 607–612. [Google Scholar] [CrossRef]
- Blair, P.A.; Noreña, L.Y.; Flores-Borja, F.; Rawlings, D.J.; Isenberg, D.A.; Ehrenstein, M.R.; Mauri, C. CD19+CD24hiCD38hi B cells exhibit regulatory capacity in healthy individuals but are functionally impaired in systemic Lupus Erythematosus patients. Immunity 2010, 32, 129–140. [Google Scholar] [CrossRef]
- Iwata, Y.; Matsushita, T.; Horikawa, M.; Dilillo, D.J.; Yanaba, K.; Venturi, G.M.; Szabolcs, P.M.; Bernstein, S.H.; Magro, C.M.; Williams, A.D.; et al. Characterization of a rare IL-10-competent B-cell subset in humans that parallels mouse regulatory B10 cells. Blood 2011, 117, 530–541. [Google Scholar] [CrossRef]
- Arkatkar, T.; Du, S.W.; Jacobs, H.M.; Dam, E.M.; Hou, B.; Buckner, J.H.; Rawlings, D.J.; Jackson, S.W. B cell-derived IL-6 initiates spontaneous germinal center formation during systemic autoimmunity. J. Exp. Med. 2017, 214, 3207–3217. [Google Scholar] [CrossRef]
- Weller, S.; Braun, M.C.; Tan, B.K.; Rosenwald, A.; Cordier, C.; Conley, M.E.; Plebani, A.; Kumararatne, D.S.; Bonnet, D.; Tournilhac, O.; et al. Human blood IgM “memory” B cells are circulating splenic marginal zone B cells harboring a prediversified immunoglobulin repertoire. Blood 2004, 104, 3647–3654. [Google Scholar] [CrossRef] [PubMed]
- Czömpöly, T.; Olasz, K.; Nyárády, Z.; Simon, D.; Bovári, J.; Németh, P. Detailed analyses of antibodies recognizing mitochondrial antigens suggest similar or identical mechanism for production of natural antibodies and natural autoantibodies. Autoimmun Rev. 2008, 7, 463–467. [Google Scholar] [CrossRef] [PubMed]
- Czömpöly, T.; Olasz, K.; Simon, D.; Nyárády, Z.; Pálinkás, L.; Czirják, L.; Berki, T.; Németh, P. A possible new bridge between innate and adaptive immunity: Are the anti-mitochondrial citrate synthase autoantibodies components of the natural antibody network? Mol. Immunol. 2006, 43, 1761–1768. [Google Scholar] [CrossRef] [PubMed]
- Simon, D.; Czömpöly, T.; Berki, T.; Minier, T.; Peti, A.; Tóth, E.; Czirják, L.; Németh, P. Naturally occurring and disease-associated auto-antibodies against topoisomerase I: A fine epitope mapping study in systemic sclerosis and systemic lupus erythematosus. Int. Immunol. 2009, 21, 415–422. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Roe, K.; Shu, G.L.; Draves, K.E.; Giordano, D.; Pepper, M.; Clark, E.A. Targeting Antigens to CD180 but Not CD40 Programs Immature and Mature B Cell Subsets to Become Efficient APCs. J. Immunol. 2019, 203, 1715–1729. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, C.; Cheng, P.; Zhang, X.; Li, X.; Hu, Y.; Xu, F.; Hong, F.; Dong, G.; Xiong, H. CD180 Ligation Inhibits TLR7- and TLR9-Mediated Activation of Macrophages and Dendritic Cells Through the Lyn-SHP-1/2 Axis in Murine Lupus. Front. Immunol. 2018, 9, 2643. [Google Scholar] [CrossRef] [PubMed]
- Simon, D.; Balogh, P.; Bognár, A.; Kellermayer, Z.; Engelmann, P.; Németh, P.; Farkas, N.; Minier, T.; Lóránd, V.; Czirják, L.; et al. Reduced non-switched memory B cell subsets cause imbalance in B cell repertoire in systemic sclerosis. Clin. Exp. Rheumatol. 2016, 34, 30–36. [Google Scholar] [PubMed]
- Ehrenstein, M.R.; Notley, C.A. The importance of natural IgM: Scavenger, protector and regulator. Nat. Rev. Immunol. 2010, 10, 778–786. [Google Scholar] [CrossRef]
- Vani, J.; Elluru, S.; Negi, V.S.; Lacroix-Desmazes, S.; Kazatchkine, M.D.; Bayry, J.; Bayary, J.; Kaveri, S.V. Role of natural antibodies in immune homeostasis: IVIg perspective. Autoimmun. Rev. 2008, 7, 440–444. [Google Scholar] [CrossRef]
- Baleva, M.; Nikolov, K. The role of intravenous immunoglobulin preparations in the treatment of systemic sclerosis. Int. J. Rheumatol. 2011, 2011, 829751. [Google Scholar] [CrossRef]
- Sato, S.; Hasegawa, M.; Takehara, K. Serum levels of interleukin-6 and interleukin-10 correlate with total skin thickness score in patients with systemic sclerosis. J. Derm. Sci. 2001, 27, 140–146. [Google Scholar] [CrossRef]
- Chun, H.Y.; Chung, J.W.; Kim, H.A.; Yun, J.M.; Jeon, J.Y.; Ye, Y.M.; Kim, S.H.; Park, H.S.; Suh, C.H. Cytokine IL-6 and IL-10 as biomarkers in systemic lupus erythematosus. J. Clin. Immunol. 2007, 27, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Cassese, G.; Arce, S.; Hauser, A.E.; Lehnert, K.; Moewes, B.; Mostarac, M.; Muehlinghaus, G.; Szyska, M.; Radbruch, A.; Manz, R.A. Plasma cell survival is mediated by synergistic effects of cytokines and adhesion-dependent signals. J. Immunol. 2003, 171, 1684–1690. [Google Scholar] [CrossRef] [PubMed]
- Jego, G.; Bataille, R.; Pellat-Deceunynck, C. Interleukin-6 is a growth factor for nonmalignant human plasmablasts. Blood 2001, 97, 1817–1822. [Google Scholar] [CrossRef]
- Kitani, A.; Hara, M.; Hirose, T.; Harigai, M.; Suzuki, K.; Kawakami, M.; Kawaguchi, Y.; Hidaka, T.; Kawagoe, M.; Nakamura, H. Autostimulatory effects of IL-6 on excessive B cell differentiation in patients with systemic lupus erythematosus: Analysis of IL-6 production and IL-6R expression. Clin. Exp. Immunol. 1992, 88, 75–83. [Google Scholar] [CrossRef]
- Mavropoulos, A.; Simopoulou, T.; Varna, A.; Liaskos, C.; Katsiari, C.G.; Bogdanos, D.P.; Sakkas, L.I. Breg Cells Are Numerically Decreased and Functionally Impaired in Patients With Systemic Sclerosis. Arthritis Rheumatol. 2016, 68, 494–504. [Google Scholar] [CrossRef]
- Gantner, F.; Hermann, P.; Nakashima, K.; Matsukawa, S.; Sakai, K.; Bacon, K.B. CD40-dependent and -independent activation of human tonsil B cells by CpG oligodeoxynucleotides. Eur. J. Immunol. 2003, 33, 1576–1585. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Erdő-Bonyár, S.; Rapp, J.; Minier, T.; Ráth, G.; Najbauer, J.; Czirják, L.; Németh, P.; Berki, T.; Simon, D. Toll-Like Receptor Mediated Activation of Natural Autoantibody Producing B Cell Subpopulations in an Autoimmune Disease Model. Int. J. Mol. Sci. 2019, 20, 6152. https://doi.org/10.3390/ijms20246152
Erdő-Bonyár S, Rapp J, Minier T, Ráth G, Najbauer J, Czirják L, Németh P, Berki T, Simon D. Toll-Like Receptor Mediated Activation of Natural Autoantibody Producing B Cell Subpopulations in an Autoimmune Disease Model. International Journal of Molecular Sciences. 2019; 20(24):6152. https://doi.org/10.3390/ijms20246152
Chicago/Turabian StyleErdő-Bonyár, Szabina, Judit Rapp, Tünde Minier, Gábor Ráth, József Najbauer, László Czirják, Péter Németh, Timea Berki, and Diána Simon. 2019. "Toll-Like Receptor Mediated Activation of Natural Autoantibody Producing B Cell Subpopulations in an Autoimmune Disease Model" International Journal of Molecular Sciences 20, no. 24: 6152. https://doi.org/10.3390/ijms20246152
APA StyleErdő-Bonyár, S., Rapp, J., Minier, T., Ráth, G., Najbauer, J., Czirják, L., Németh, P., Berki, T., & Simon, D. (2019). Toll-Like Receptor Mediated Activation of Natural Autoantibody Producing B Cell Subpopulations in an Autoimmune Disease Model. International Journal of Molecular Sciences, 20(24), 6152. https://doi.org/10.3390/ijms20246152



