Multipotent Neurotrophic Effects of Hepatocyte Growth Factor in Spinal Cord Injury
Abstract
1. Introduction
2. HGF/c-Met Expression within Injury Site after SCI
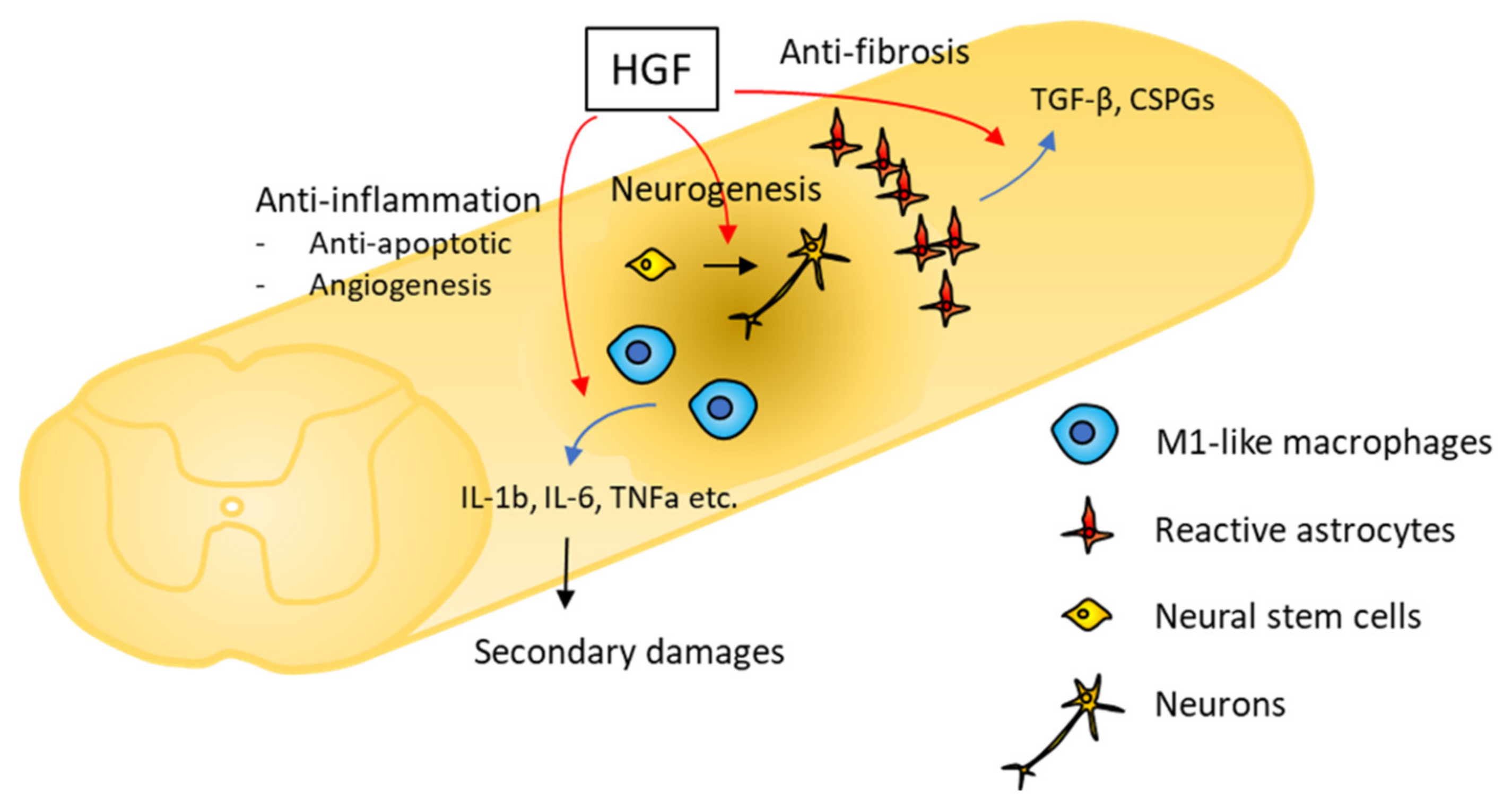
3. Neurotrophic Activities of HGF in SCI
3.1. Anti-Inflammatory Effects
3.2. Anti-Apoptotic Effects
3.3. Angiogenic Properties
3.4. Anti-Fibrotic Effects
3.5. Neurogenic and Oligogliogenic Effects
4. Recent Advances towards Clinical Applications in SCI Recovery
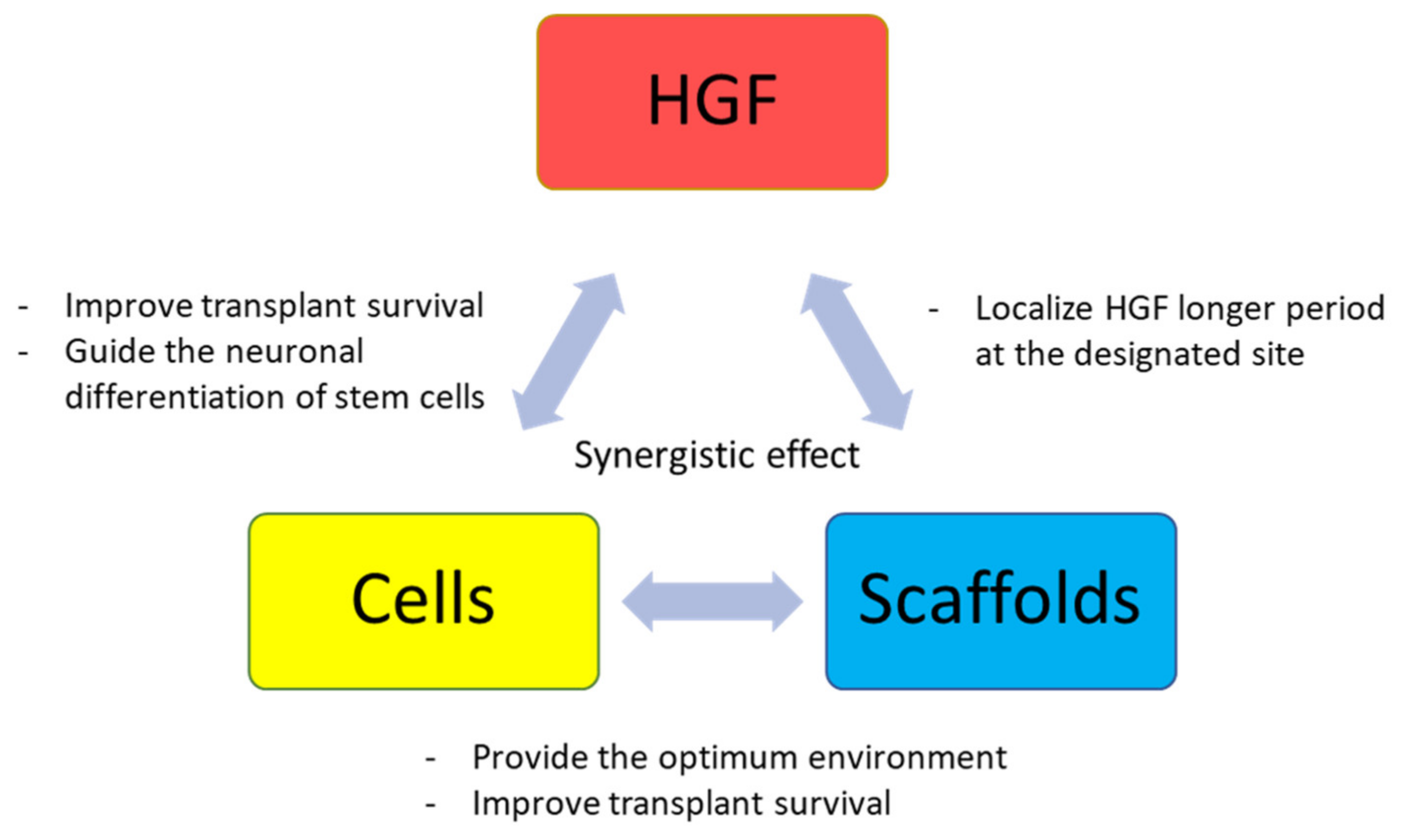
4.1. Single Modality Approach
4.2. Combination Therapy Approach
5. Summary
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AIF | apoptosis-inducing factor |
| ATF3 | activating transcription factor 3 |
| BMSCs | bone marrow stromal stem cells |
| CBD | collagen-binding domain |
| CSF1 | colony stimulating factor 1 |
| CSPG | chondroitin sulfate proteoglycan |
| DRGs | dorsal root ganglions |
| GSK-3b | glycogen synthase kinase-3 beta |
| HGF | hepatocyte growth factor |
| IL-1 | interleukin-1 |
| MAG | myelin-associated glycoprotein |
| MSCs | mesenchymal stem cells |
| NF-κB | nuclear factor kappa B |
| NSCs | neural stem cells |
| OMgp | oligodendrocyte-myelin glycoprotein |
| SCI | spinal cord injury |
| TNF-α | tumor necrosis factor-alpha |
| TGF-β | transforming growth factor beta |
References
- Readdy, W.J.; Chan, A.K.; Matijakovich, D.J.; Dhall, S.D. A review and update on the guidelines for the acute non-operative management of cervical spinal cord injury. J. Neurosurg. Sci. 2015, 59, 119–128. [Google Scholar] [PubMed]
- Yue, J.K.; Upadhyayula, P.S.; Chan, A.K.; Winkler, E.A.; Burke, J.F.; Readdy, W.J.; Sharma, S.; Deng, H.; Dhall, S.S. A review and update on the current and emerging clinical trials for the acute management of cervical spine and spinal cord injuries—Part III. J. Neurosurg. Sci. 2016, 60, 529–542. [Google Scholar] [PubMed]
- Lee, J.K.; Zheng, B. Role of myelin-associated inhibitors in axonal repair after spinal cord injury. Exp. Neurol. 2012, 235, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, T.; Nawa, K.; Ichihara, A. Partial purification and characterization of hepatocyte growth factor from serum of hepatectomized rats. Biochem. Biophys. Res. Commun. 1984, 122, 1450–1459. [Google Scholar] [CrossRef]
- Russell, W.E.; McGowan, J.A.; Bucher, N.L. Partial characterization of a hepatocyte growth factor from rat platelets. J. Cell Physiol. 1984, 119, 183–192. [Google Scholar] [CrossRef]
- Kitamura, K.; Iwanami, A.; Nakamura, M.; Yamane, J.; Watanabe, K.; Suzuki, Y.; Miyazawa, D.; Shibata, S.; Funakoshi, H.; Miyatake, S.; et al. Hepatocyte growth factor promotes endogenous repair and functional recovery after spinal cord injury. J. Neurosci. Res. 2007, 85, 2332–2342. [Google Scholar] [CrossRef]
- Yamane, K.; Mazaki, T.; Shiozaki, Y.; Yoshida, A.; Shinohara, K.; Nakamura, M.; Yoshida, Y.; Zhou, D.; Kitajima, T.; Tanaka, M.; et al. Collagen-Binding Hepatocyte Growth Factor (HGF) alone or with a Gelatin- furfurylamine Hydrogel Enhances Functional Recovery in Mice after Spinal Cord Injury. Sci. Rep. 2018, 8, 917. [Google Scholar] [CrossRef]
- Jeong, S.R.; Kwon, M.J.; Lee, H.G.; Joe, E.H.; Lee, J.H.; Kim, S.S.; Suh-Kim, H.; Kim, B.G. Hepatocyte growth factor reduces astrocytic scar formation and promotes axonal growth beyond glial scars after spinal cord injury. Exp. Neurol. 2012, 233, 312–322. [Google Scholar] [CrossRef]
- Kitamura, K.; Fujiyoshi, K.; Yamane, J.; Toyota, F.; Hikishima, K.; Nomura, T.; Funakoshi, H.; Nakamura, T.; Aoki, M.; Toyama, Y.; et al. Human hepatocyte growth factor promotes functional recovery in primates after spinal cord injury. PLoS ONE 2011, 6, e27706. [Google Scholar] [CrossRef]
- Bottaro, D.P.; Rubin, J.S.; Faletto, D.L.; Chan, A.M.; Kmiecik, T.E.; Vande Woude, G.F.; Aaronson, S.A. Identification of the hepatocyte growth factor receptor as the c-met proto-oncogene product. Science 1991, 251, 802–804. [Google Scholar] [CrossRef]
- Naldini, L.; Vigna, E.; Narsimhan, R.P.; Gaudino, G.; Zarnegar, R.; Michalopoulos, G.K.; Comoglio, P.M. Hepatocyte growth factor (HGF) stimulates the tyrosine kinase activity of the receptor encoded by the proto-oncogene c-MET. Oncogene 1991, 6, 501–504. [Google Scholar] [PubMed]
- Nakamura, T.; Sakai, K.; Nakamura, T.; Matsumoto, K. Hepatocyte growth factor twenty years on: Much more than a growth factor. J. Gastroenterol. Hepatol. 2011, 1, 188–202. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, K.; Nakamura, T. Hepatocyte growth factor (HGF) as a tissue organizer for organogenesis and regeneration. Biochem. Biophys. Res. Commun. 1997, 239, 639–644. [Google Scholar] [CrossRef] [PubMed]
- Yanagita, K.; Matsumoto, K.; Sekiguchi, K.; Ishibashi, H.; Niho, Y.; Nakamura, T. Hepatocyte growth factor may act as a pulmotrophic factor on lung regeneration after acute lung injury. J. Biol. Chem. 1993, 268, 21212–21217. [Google Scholar] [PubMed]
- Igawa, T.; Matsumoto, K.; Kanda, S.; Saito, Y.; Nakamura, T. Hepatocyte growth factor may function as a renotropic factor for regeneration in rats with acute renal injury. Am. J. Physiol. 1993, 265, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Shimamura, M.; Sato, N.; Sata, M.; Wakayama, K.; Ogihara, T.; Morishita, R. Expression of hepatocyte growth factor and c-Met after spinal cord injury in rats. Brain Res. 2007, 1151, 188–194. [Google Scholar] [CrossRef]
- Takano, M.; Kawabata, S.; Shibata, S.; Yasuda, A.; Nori, S.; Tsuji, O.; Nagoshi, N.; Iwanami, A.; Ebise, H.; Horiuchi, K.; et al. Enhanced Functional Recovery from Spinal Cord Injury in Aged Mice after Stem Cell Transplantation through HGF Induction. Stem Cell Rep. 2017, 8, 509–518. [Google Scholar] [CrossRef]
- Popovich, P.G.; Guan, Z.; Wei, P.; Huitinga, I.; van Rooijen, N.; Stokes, B.T. Depletion of hematogenous macrophages promotes partial hindlimb recovery and neuroanatomical repair after experimental spinal cord injury. Exp. Neurol. 1999, 158, 351–365. [Google Scholar] [CrossRef]
- Giannopoulou, M.; Dai, C.; Tan, X.; Wen, X.; Michalopoulos, G.K.; Liu, Y. Hepatocyte growth factor exerts its anti-inflammatory action by disrupting nuclear factor-kappaB signaling. Am. J. Pathol. 2008, 173, 30–41. [Google Scholar] [CrossRef]
- Kamimoto, M.; Mizuno, S.; Nakamura, T. Reciprocal regulation of IL-6 and IL-10 balance by HGF via recruitment of heme oxygenase-1 in macrophages for attenuation of liver injury in a mouse model of endotoxemia. Int. J. Mol. Med. 2009, 24, 161–170. [Google Scholar]
- Kamimoto, M.; Mizuno, S.; Matsumoto, K.; Nakamura, T. Hepatocyte growth factor prevents multiple organ injuries in endotoxemic mice through a heme oxygenase-1-dependent mechanism. Biochem. Biophys. Res. Commun. 2009, 380, 333–337. [Google Scholar] [CrossRef] [PubMed]
- Coudriet, G.M.; He, J.; Trucco, M.; Mars, W.M.; Piganelli, J.D. Hepatocyte growth factor modulates interleukin-6 production in bone marrow derived macrophages: Implications for inflammatory mediated diseases. PLoS ONE 2010, 5, e15384. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, S.; Nakamura, T. Prevention of neutrophil extravasation by hepatocyte growth factor leads to attenuations of tubular apoptosis and renal dysfunction in mouse ischemic kidneys. Am. J. Pathol. 2005, 166, 1895–1905. [Google Scholar] [CrossRef]
- Niimura, M.; Takagi, N.; Takagi, K.; Mizutani, R.; Ishihara, N.; Matsumoto, K.; Funakoshi, H.; Nakamura, T.; Takeo, S. Prevention of apoptosis-inducing factor translocation is a possible mechanism for protective effects of hepatocyte growth factor against neuronal cell death in the hippocampus after transient forebrain ischemia. J. Cereb. Blood Flow Metab. 2006, 26, 1354–1365. [Google Scholar] [CrossRef] [PubMed]
- Olianas, M.C.; Dedoni, S.; Onali, P. Inhibition of TNF-α-induced neuronal apoptosis by antidepressants acting through the lysophosphatidic acid receptor LPA1. Apoptosis 2019, 24, 478–498. [Google Scholar] [CrossRef] [PubMed]
- Schizas, N.; Perry, S.; Andersson, B.; Wählby, C.; Kullander, K.; Hailer, N.P. Differential Neuroprotective Effects of Interleukin-1 Receptor Antagonist on Spinal Cord Neurons after Excitotoxic Injury. Neuroimmunomodulation 2017, 24, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Akuzawa, S.; Kazui, T.; Shi, E.; Yamashita, K.; Bashar, A.H.; Terada, H. Interleukin-1 receptor antagonist attenuates the severity of spinal cord ischemic injury in rabbits. J. Vasc. Surg. 2008, 48, 694–700. [Google Scholar] [CrossRef]
- Nakamura, T.; Mizuno, S. The discovery of hepatocyte growth factor (HGF) and its significance for cell biology, life sciences and clinical medicine. Proc. Jpn. Acad. Ser. B Phys. Biol. Sci. 2010, 86, 588–610. [Google Scholar] [CrossRef]
- Potente, M.; Gerhardt, H.; Carmeliet, P. Basic and therapeutic aspects of angiogenesis. Cell 2011, 146, 873–887. [Google Scholar] [CrossRef]
- Yamamoto, K.; Morishita, R.; Hayashi, S.; Matsushita, H.; Nakagami, H.; Moriguchi, A.; Matsumoto, K.; Nakamura, T.; Kaneda, Y.; Ogihara, T. Contribution of Bcl-2, but not Bcl-xL and Bax, to antiapoptotic actions of hepatocyte growth factor in hypoxia-conditioned human endothelial cells. Hypertension 2001, 37, 1341–1348. [Google Scholar] [CrossRef]
- Birukova, A.A.; Alekseeva, E.; Mikaelyan, A.; Birukov, K.G. HGF attenuates thrombin-induced endothelial permeability by Tiam1-mediated activation of the Rac pathway and by Tiam1/Rac-dependent inhibition of the Rho pathway. FASEB J. 2007, 21, 2776–2786. [Google Scholar] [CrossRef] [PubMed]
- Lutton, C.; Young, Y.W.; Williams, R.; Meedeniya, A.C.; Mackay-Sim, A.; Goss, B. Combined VEGF and PDGF treatment reduces secondary degeneration after spinal cord injury. J. Neurotrauma 2012, 29, 957–970. [Google Scholar] [CrossRef] [PubMed]
- Maldonado-Lasunción, I.; Verhaagen, J.; Oudega, M. Mesenchymal Stem Cell-Macrophage Choreography Supporting Spinal Cord Repair. Neurotherapeutics 2018, 15, 578–587. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Liu, Y. Delayed administration of hepatocyte growth factor reduces renal fibrosis in obstructive nephropathy. Am. J. Physiol. Renal. Physiol. 2003, 284, 349–357. [Google Scholar] [CrossRef]
- Mizuno, S.; Matsumoto, K.; Li, M.Y.; Nakamura, T. HGF reduces advancing lung fibrosis in mice: A potential role for MMP-dependent myofibroblast apoptosis. FASEB J. 2005, 19, 580–582. [Google Scholar] [CrossRef]
- Tada, T.; Zhan, H.; Tanaka, Y.; Hongo, K.; Matsumoto, K.; Nakamura, T. Intraventricular administration of hepatocyte growth factor treats mouse communicating hydrocephalus induced by transforming growth factor beta1. Neurobiol. Dis. 2006, 21, 576–586. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, J. Hepatocyte growth factor: New arsenal in the fights against renal fibrosis? Kidney Int. 2006, 70, 238–240. [Google Scholar] [CrossRef]
- Nakamura, T.; Sakata, R.; Ueno, T.; Sata, M.; Ueno, H. Inhibition of transforming growth factor beta prevents progression of liver fibrosis and enhances hepatocyte regeneration in dimethylnitrosamine-treated rats. Hepatology 2000, 32, 247–255. [Google Scholar] [CrossRef]
- Gesteira, T.F.; Coulson-Thomas, Y.M.; Coulson-Thomas, V.J. Anti-inflammatory properties of the glial scar. Neural. Regen. Res. 2016, 11, 1742–1743. [Google Scholar]
- Dalamagkas, K.; Tsintou, M.; Seifalian, A.M. Stem cells for spinal cord injuries bearing translational potential. Neural. Regen. Res. 2018, 13, 35–42. [Google Scholar]
- Götz, M.; Sirko, S.; Beckers, J.; Irmler, M. Reactive astrocytes as neural stem or progenitor cells: In vivo lineage, In vitro potential, and Genome-wide expression analysis. Glia 2015, 63, 1452–1468. [Google Scholar] [CrossRef] [PubMed]
- Hofstetter, C.P.; Holmström, N.A.; Lilja, J.A.; Schweinhardt, P.; Hao, J.; Spenger, C.; Wiesenfeld-Hallin, Z.; Kurpad, S.N.; Frisén, J.; Olson, L. Allodynia limits the usefulness of intraspinal neural stem cell grafts; directed differentiation improves outcome. Nat. Neurosci. 2005, 8, 346–353. [Google Scholar] [CrossRef] [PubMed]
- Kokuzawa, J.; Yoshimura, S.; Kitajima, H.; Shinoda, J.; Kaku, Y.; Iwama, T.; Morishita, R.; Shimazaki, T.; Okano, H.; Kunisada, T.; et al. Hepatocyte growth factor promotes proliferation and neuronal differentiation of neural stem cells from mouse embryos. Mol. Cell Neurosci. 2003, 24, 190–197. [Google Scholar] [CrossRef]
- Kato, M.; Yoshimura, S.; Kokuzawa, J.; Kitajima, H.; Kaku, Y.; Iwama, T.; Shinoda, J.; Kunisada, T.; Sakai, N. Hepatocyte growth factor promotes neuronal differentiation of neural stem cells derived from embryonic stem cells. Neuroreport 2004, 15, 5–8. [Google Scholar] [CrossRef]
- Hu, Z.; Li, T.; Zhang, X.; Chen, Y. Hepatocyte growth factor enhances the generation of high-purity oligodendrocytes from human embryonic stem cells. Differentiation 2009, 78, 177–184. [Google Scholar] [CrossRef]
- Ma, Q.; Cai, M.; Shang, J.W.; Yang, J.; Gu, X.Y.; Liu, W.B.; Yang, Q. In vitro neural differentiation of bone marrow stromal cells induced by hepatocyte growth factor and glial cell derived neurotrophic factor. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 4654–4663. [Google Scholar]
- Zheng, B.; Wang, C.; He, L.; Xu, X.; Qu, J.; Hu, J.; Zhang, H. Neural differentiation of mesenchymal stem cells influences chemotactic responses to HGF. J. Cell Physiol. 2013, 228, 149–162. [Google Scholar] [CrossRef]
- Kitamura, K.; Nagoshi, N.; Tsuji, O.; Matsumoto, M.; Okano, H.; Nakamura, M. Application of Hepatocyte Growth Factor for Acute Spinal Cord Injury: The Road from Basic Studies to Human Treatment. Int. J. Mol. Sci. 2019, 20, E1054. [Google Scholar] [CrossRef]
- Ajroud-Driss, S.; Christiansen, M.; Allen, J.A.; Kessler, J.A. Phase 1/2 open-label dose-escalation study of plasmid DNA expressing two isoforms of hepatocyte growth factor in patients with painful diabetic peripheral neuropathy. Mol. Ther. 2013, 21, 1279–1286. [Google Scholar] [CrossRef]
- Kessler, J.A.; Smith, A.G.; Cha, B.S.; Choi, S.H.; Wymer, J.; Shaibani, A.; Ajroud-Driss, S.; Vinik, A. VM202 DPN-II Study Group. Double-blind, placebo-controlled study of HGF gene therapy in diabetic neuropathy. Ann. Clin. Transl. Neurol. 2015, 2, 465–478. [Google Scholar] [CrossRef]
- Warita, H.; Kato, M.; Asada, R.; Yamashita, A.; Hayata, D.; Adachi, K.; Aoki, M. Safety, Tolerability, and Pharmacodynamics of Intrathecal Injection of Recombinant Human HGF (KP-100) in Subjects with Amyotrophic Lateral Sclerosis: A Phase I Trial. J. Clin. Pharmacol. 2019, 59, 677–687. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, K.; Funakoshi, H.; Takahashi, H.; Sakai, K. HGF-Met Pathway in Regeneration and Drug Discovery. Biomedicines 2014, 2, 275–300. [Google Scholar] [CrossRef] [PubMed]
- Lu, P.; Tuszynski, M.H. Growth factors and combinatorial therapies for CNS regeneration. Exp. Neurol. 2008, 209, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Straley, K.S.; Foo, C.W.; Heilshorn, S.C. Biomaterial design strategies for the treatment of spinal cord injuries. J. Neurotrauma 2010, 27, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Kitajima, T.; Terai, H.; Ito, Y. A fusion protein of hepatocyte growth factor for immobilization to collagen. Biomaterials 2007, 28, 1989–1997. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yamane, K.; Misawa, H.; Takigawa, T.; Ito, Y.; Ozaki, T.; Matsukawa, A. Multipotent Neurotrophic Effects of Hepatocyte Growth Factor in Spinal Cord Injury. Int. J. Mol. Sci. 2019, 20, 6078. https://doi.org/10.3390/ijms20236078
Yamane K, Misawa H, Takigawa T, Ito Y, Ozaki T, Matsukawa A. Multipotent Neurotrophic Effects of Hepatocyte Growth Factor in Spinal Cord Injury. International Journal of Molecular Sciences. 2019; 20(23):6078. https://doi.org/10.3390/ijms20236078
Chicago/Turabian StyleYamane, Kentaro, Haruo Misawa, Tomoyuki Takigawa, Yoshihiro Ito, Toshifumi Ozaki, and Akihiro Matsukawa. 2019. "Multipotent Neurotrophic Effects of Hepatocyte Growth Factor in Spinal Cord Injury" International Journal of Molecular Sciences 20, no. 23: 6078. https://doi.org/10.3390/ijms20236078
APA StyleYamane, K., Misawa, H., Takigawa, T., Ito, Y., Ozaki, T., & Matsukawa, A. (2019). Multipotent Neurotrophic Effects of Hepatocyte Growth Factor in Spinal Cord Injury. International Journal of Molecular Sciences, 20(23), 6078. https://doi.org/10.3390/ijms20236078




