Emerging Anticancer Potentials of Selenium on Osteosarcoma
Abstract
1. Introduction
2. Literature Search
3. Evidence from In Vitro Study
3.1. Selenium Exerts Cytotoxicity, Anti-Proliferation and Pro-Apoptotic Activities on Osteosarcoma Cells
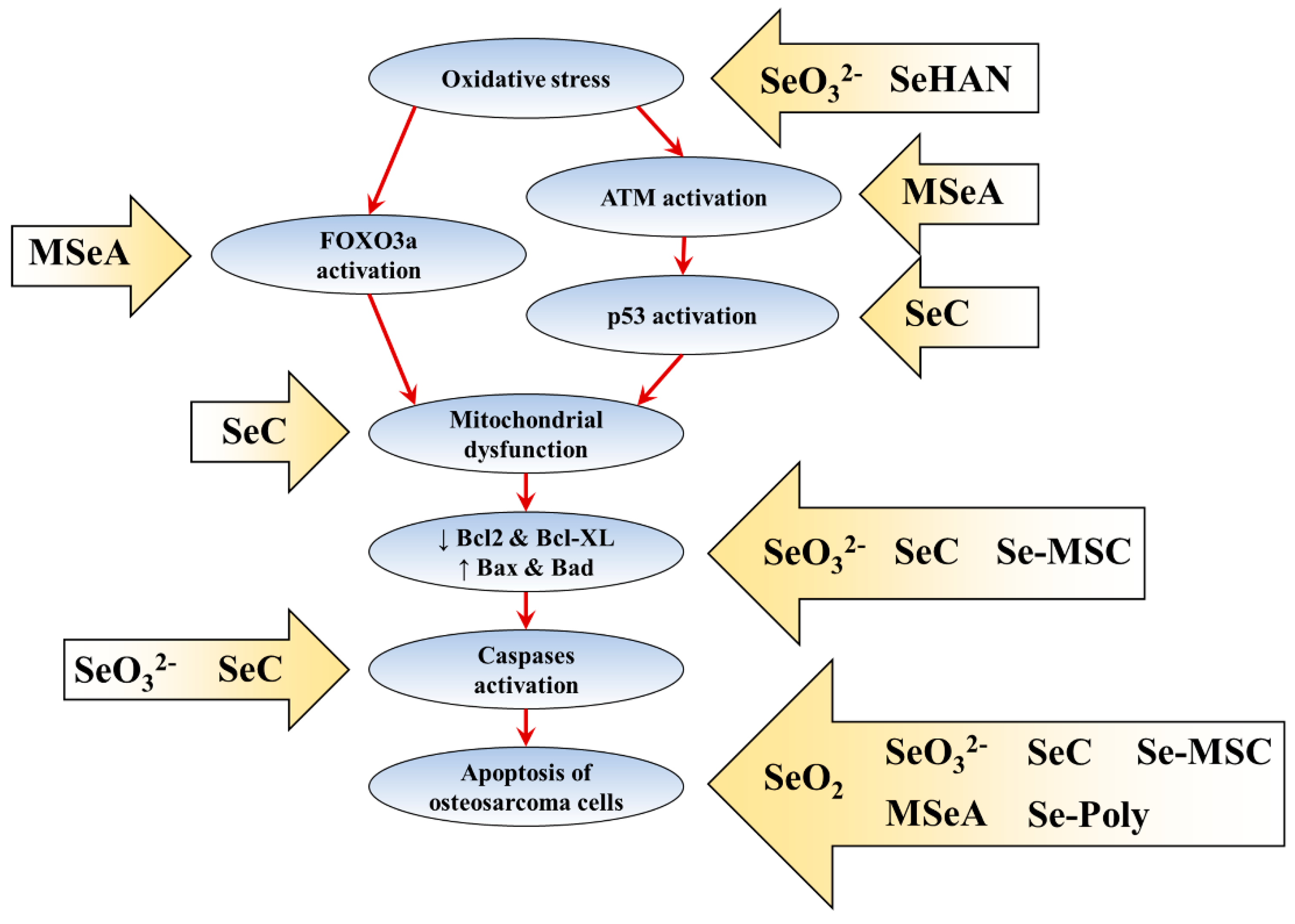
3.2. Molecular Mechanism of Selenium-Induced Osteosarcoma Cell Death
3.3. Role of Selenium in the Tumour Microenvironment
3.4. Selenium as a Bone Implant Material
4. Evidence from Animal and Human Studies
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AMPK | 5’ adenosine monophosphate-activated protein kinase |
| ATM | Ataxia-telangiectasia mutated kinase |
| Bad | Bcl-2−associated death promoter protein |
| BAX | Bcl-2−associated x gene |
| Bcl-2 | B-cell lymphoma 2 |
| Bcl-XL | B-cell lymphoma-extra large |
| BGLAP3 | Bone γ-carboxyglutamate protein 3 |
| BMSCs | Bone marrow stem cells |
| BRCA1 | Breast cancer type 1 susceptibility gene |
| CASP3 | Caspase-3 gene |
| CASP9 | Caspase-9 gene |
| CDK-2 | Cyclin-dependent kinase-2 |
| FOXO3a | Forkhead Box O3a protein |
| HA | Hydroxyapatite |
| MMP-9 | Matrix metallopeptidase-9 |
| MnTMPyP | Manganese(iii) tetrakis(1-methyl-4-pyridyl)porphyrin |
| MSeA | Methyseleninic acid |
| NO | Nitric oxide |
| NPC | Nutritional Prevention of Cancer trial |
| Nrf2 | Nuclear factor erythroid 2−related factor 2 |
| O2−• | Superoxide anion |
| PARP | Poly(ADP-ribose) polymerase |
| PLLA | Poly-l-lactic acid |
| pTEN | Tumour suppressor protein phosphatase and tensin homolog |
| ROS | Reactive oxygen species |
| RSeOH | Seleninic acid |
| SeC | Selenocysteine |
| SeHA | Seo32−-containing HA |
| SeHA/ALG | Selenium-containing Hydroxyapatite/Alginate |
| SeHA/ALG RIS I | Seha/AIG granules with risedronate sodium during formation |
| SeHA/ALG RIS II | Seha/AIG granules with risedronate sodium and potassium chloride during formation |
| SeHAN | Selenite-substituted HA nanoparticles |
| SeMet | Selenomethionine |
| Se-MSC | Selenium-methylselenocysteine |
| SeNP-PLLA | Selenium-coated poly-l-lactic acid nanocomposites |
| SeO2 | Selenium dioxide |
| SeO32− | Selenite |
| SeO42− | Selenate |
| Se-Poly | Selenium-polysaccharide |
| Ser | Serine |
| tBid | Truncated BH3 interacting-domain death agonist protein |
| WRN | Werner syndrome protein |
| γH2Ax | Phosphorylated H2A histone family member x |
References
- Cheng, W.-H.; Lei, X.G. Selenium: Basic nutritional aspects. In Molecular, Genetic, and Nutritional Aspects of Major and Trace Minerals; Collins, J.F., Ed.; Academic Press: Cambridge, MA, USA, 2017; pp. 449–461. [Google Scholar]
- Tan, H.W.; Mo, H.Y.; Lau, A.T.Y.; Xu, Y.M. Selenium Species: Current Status and Potentials in Cancer Prevention and Therapy. Int. J. Mol. Sci. 2018, 20. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Perez, M.; Ali, W.; Marc, M.A.; Handzlik, J.; Dominguez-Alvarez, E. Selenides and Diselenides: A Review of Their Anticancer and Chemopreventive Activity. Molecules 2018, 23. [Google Scholar] [CrossRef] [PubMed]
- Bartolini, D.; Sancineto, L.; de Bem, A.F.; Tew, K.D.; Santi, C.; Radi, R.; Toquato, P.; Galli, F. Selenocompounds in Cancer Therapy: An Overview. Adv. Cancer Res. 2017, 136, 259–302. [Google Scholar] [CrossRef] [PubMed]
- Kieliszek, M.; Lipinski, B.; Blazejak, S. Application of Sodium Selenite in the Prevention and Treatment of Cancers. Cells 2017, 6. [Google Scholar] [CrossRef]
- Reilly, C. Selenium in Foods. In Selenium in Food and Health, 2nd ed.; Springer: Salmon Tower Building, NY, USA, 2006; pp. 158–172. [Google Scholar]
- Navarro-Alarcon, M.; Cabrera-Vique, C. Selenium in food and the human body: A review. Sci. Total Environ. 2008, 400, 115–141. [Google Scholar] [CrossRef]
- Fairweather-Tait, S.J.; Collings, R.; Hurst, R. Selenium bioavailability: Current knowledge and future research requirements. Am. J. Clin. Nutr. 2010, 91, 1484–1491. [Google Scholar] [CrossRef]
- Rayman, M.P.; Infante, H.G.; Sargent, M. Food-chain selenium and human health: Spotlight on speciation. Br. J. Nutr. 2008, 100, 238–253. [Google Scholar] [CrossRef]
- Nakayama, S.F.; Iwai-Shimada, M.; Oguri, T.; Isobe, T.; Takeuchi, A.; Kobayashi, Y.; Michikawa, T.; Yamazaki, S.; Nitta, H.; Kawamoto, T.; et al. Blood mercury, lead, cadmium, manganese and selenium levels in pregnant women and their determinants: The Japan Environment and Children’s Study (JECS). J. Expo. Sci. Environ. Epidemiol. 2019, 29, 633–647. [Google Scholar] [CrossRef]
- Monsen, E.R. Dietary Reference Intakes for the antioxidant nutrients: Vitamin C, vitamin E, selenium, and carotenoids. J. Am. Diet. Assoc. 2000, 100, 637–640. [Google Scholar] [CrossRef]
- Department of Health. Dietary reference values for food energy and nutrients for the United Kingdom. Report of the Panel on Dietary Reference Values of the Committee on Medical Aspects of Food Policy, 8th ed.; Stationery Office: London, UK, 1991; Volume 41, pp. 1–210. [Google Scholar]
- Al-Ahmary, K.M. Selenium content in selected foods from the Saudi Arabia market and estimation of the daily intake. Arab. J. Chem. 2009, 2, 95–99. [Google Scholar] [CrossRef]
- Al-Othman, A.M.; Al-Othman, Z.A.; El-Desoky, G.E.; Aboul-Soud, M.A.; Habila, M.A.; Giesy, J.P. Daily intake of selenium and concentrations in blood of residents of Riyadh City, Saudi Arabia. Environ. Geochem Health 2012, 34, 417–431. [Google Scholar] [CrossRef] [PubMed]
- Sunde, R.A.; Paterson, E.; Evenson, J.K.; Barnes, K.M.; Lovegrove, J.A.; Gordon, M.H. Longitudinal selenium status in healthy British adults: Assessment using biochemical and molecular biomarkers. Br. J. Nutr. 2008, 99 (Suppl.3), 37–47. [Google Scholar] [CrossRef] [PubMed]
- Stoffaneller, R.; Morse, N.L. A review of dietary selenium intake and selenium status in Europe and the Middle East. Nutrients 2015, 7, 1494–1537. [Google Scholar] [CrossRef]
- Schrauzer, G.N. Selenomethionine: A Review of Its Nutritional Significance, Metabolism and Toxicity. J. Nutr. 2000, 130, 1653–1656. [Google Scholar] [CrossRef]
- Roman, M.; Jitaru, P.; Barbante, C. Selenium biochemistry and its role for human health. Metallomics 2014, 6, 25–54. [Google Scholar] [CrossRef] [PubMed]
- Tinggi, U. Selenium: Its role as antioxidant in human health. Environ. Health Prev. Med. 2008, 13, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Tapiero, H.; Townsend, D.M.; Tew, K.D. The antioxidant role of selenium and seleno-compounds. Biomed. Pharmacother. 2003, 57, 134–144. [Google Scholar] [CrossRef]
- Gladyshev, V.N.; Hatfield, D.L. Selenocysteine-containing proteins in mammals. J. Biomed. Sci. 1999, 6, 151–160. [Google Scholar] [CrossRef]
- Moreno-Reyes, R.; Egrise, D.; Nève, J.; Pasteels, J.L.; Schoutens, A. Selenium deficiency-induced growth retardation is associated with an impaired bone metabolism and osteopenia. J. Bone Min. Res. 2001, 16, 1556–1563. [Google Scholar] [CrossRef]
- Badr, D.M.; Hafez, H.F.; Agha, A.M.; Shouman, S.A. The Combination of alpha-Tocopheryl Succinate and Sodium Selenite on Breast Cancer: A Merit or a Demerit? Oxidative Med. Cell Longev. 2016, 2016, 4741694. [Google Scholar] [CrossRef]
- Berggren, M.; Sittadjody, S.; Song, Z.; Samira, J.L.; Burd, R.; Meuillet, E.J. Sodium selenite increases the activity of the tumor suppressor protein, PTEN, in DU-145 prostate cancer cells. Nutr. Cancer 2009, 61, 322–331. [Google Scholar] [CrossRef] [PubMed]
- Li, G.X.; Lee, H.J.; Wang, Z.; Hu, H.; Liao, J.D.; Watts, J.C.; Combs, G.F., Jr.; Lu, J. Superior in vivo inhibitory efficacy of methylseleninic acid against human prostate cancer over selenomethionine or selenite. Carcinogenesis 2008, 29, 1005–1012. [Google Scholar] [CrossRef] [PubMed]
- Schroterova, L.; Kralova, V.; Voracova, A.; Haskova, P.; Rudolf, E.; Cervinka, M. Antiproliferative effects of selenium compounds in colon cancer cells: Comparison of different cytotoxicity assays. Toxicol. in Vitro 2009, 23, 1406–1411. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, M.; Endo, M.; Shinohara, F.; Echigo, S.; Rikiishi, H. Differential apoptotic response of human cancer cells to organoselenium compounds. Cancer Chemother. Pharmacol. 2010, 66, 475–484. [Google Scholar] [CrossRef]
- Qi, Y.; Schoene, N.W.; Lartey, F.M.; Cheng, W.H. Selenium compounds activate ATM-dependent DNA damage response via the mismatch repair protein hMLH1 in colorectal cancer cells. J. Biol. Chem. 2010, 285, 33010–33017. [Google Scholar] [CrossRef] [PubMed]
- Cai, Z.; Dong, L.; Song, C.; Zhang, Y.; Zhu, C.; Zhang, Y.; Ling, Q.; Hoffmann, P.R.; Li, J.; Huang, Z.; et al. Methylseleninic Acid Provided at Nutritional Selenium Levels Inhibits Angiogenesis by Down-regulating Integrin beta3 Signaling. Sci. Rep. 2017, 7, 9445. [Google Scholar] [CrossRef]
- Kao, R.H.; Lai, G.M.; Chow, J.M.; Liao, C.H.; Zheng, Y.M.; Tsai, W.L.; Hsia, S.; Lai, I.C.; Lee, H.L.; Chuang, S.E.; et al. Opposite Regulation of CHOP and GRP78 and Synergistic Apoptosis Induction by Selenium Yeast and Fish Oil via AMPK Activation in Lung Adenocarcinoma Cells. Nutrients 2018, 10. [Google Scholar] [CrossRef]
- Ren, H.; Mu, J.; Ma, J.; Gong, J.; Li, J.; Wang, J.; Gao, T.; Zhu, P.; Zheng, S.; Xie, J.; et al. Selenium Inhibits Homocysteine-Induced Endothelial Dysfunction and Apoptosis via Activation of AKT. Cell. Physiol. Biochem. Int. J. Exp. Cell Physiol. Biochem. Pharmacol. 2016, 38, 871–882. [Google Scholar] [CrossRef]
- Sakalli Cetin, E.; Naziroglu, M.; Cig, B.; Ovey, I.S.; Aslan Kosar, P. Selenium potentiates the anticancer effect of cisplatin against oxidative stress and calcium ion signaling-induced intracellular toxicity in MCF-7 breast cancer cells: Involvement of the TRPV1 channel. J. Recept. Signal. Transduct. Res. 2017, 37, 84–93. [Google Scholar] [CrossRef]
- Nicastro, H.L.; Dunn, B.K. Selenium and prostate cancer prevention: Insights from the selenium and vitamin E cancer prevention trial (SELECT). Nutrients 2013, 5, 1122–1148. [Google Scholar] [CrossRef]
- Clark, L.C.; Combs, G.F., Jr.; Turnbull, B.W.; Slate, E.H.; Chalker, D.K.; Chow, J.; Davis, L.S.; Glover, R.A.; Graham, G.F.; Gross, E.G.; et al. Effects of Selenium Supplementation for Cancer Prevention in Patients With Carcinoma of the Skin. A Randomized Controlled Trial. JAMA 1996, 276, 1957–1963. [Google Scholar] [CrossRef] [PubMed]
- Vinceti, M.; Filippini, T.; Cilloni, S.; Crespi, C.M. The Epidemiology of Selenium and Human Cancer. In Selenium and Selenoproteins in Cancer; Tew, K.D., Galli, F., Eds.; Academic Press: Cambridge, MA, USA, 2017; Volume 136, pp. 1–48. [Google Scholar]
- Wei, W.-Q.; Abnet, C.C.; Qiao, Y.-L.; Dawsey, S.M.; Dong, Z.-W.; Sun, X.-D.; Fan, J.-H.; Gunter, E.W.; Taylor, P.R.; Mark, S.D. Prospective study of serum selenium concentrations and esophageal and gastric cardia cancer, heart disease, stroke, and total death. Am. J. Clin. Nutr. 2004, 79, 80–85. [Google Scholar] [CrossRef]
- Ward, E.; DeSantis, C.; Robbins, A.; Kohler, B.; Jemal, A. Childhood and adolescent cancer statistics, 2014. CA Cancer J. Clin. 2014, 64, 83–103. [Google Scholar] [CrossRef] [PubMed]
- Bishop, M.W.; Janeway, K.A.; Gorlick, R. Future directions in the treatment of osteosarcoma. Curr. Opin. Pediatrics 2016, 28, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Misaghi, A.; Goldin, A.; Awad, M.; Kulidjian, A.A. Osteosarcoma: A comprehensive review. Sicot-J. 2018, 4, 12. [Google Scholar] [CrossRef]
- Ritter, J.; Bielack, S.S. Osteosarcoma. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2010, 21, 320–325. [Google Scholar] [CrossRef]
- Stiller, C. Incidence of Childhood Leukaemia. Available online: http://www.euro.who.int/__data/assets/pdf_file/0005/97016/4.1.-Incidence-of-childhood-leukaemia-EDITED_layouted.pdf (accessed on 3 October 2019).
- Kager, L.; Tamamyan, G.; Bielack, S. Novel insights and therapeutic interventions for pediatric osteosarcoma. Future Oncol. 2017, 13, 357–368. [Google Scholar] [CrossRef]
- Moore, D.D.; Luu, H.H. Osteosarcoma. Cancer Treat. Res. 2014, 162, 65–92. [Google Scholar] [CrossRef]
- Daw, N.C.; Chou, A.J.; Jaffe, N.; Rao, B.N.; Billups, C.A.; Rodriguez-Galindo, C.; Meyers, P.A.; Huh, W.W. Recurrent osteosarcoma with a single pulmonary metastasis: A multi-institutional review. Br. J. Cancer 2015, 112, 278–282. [Google Scholar] [CrossRef]
- Anderson, M.E. Update on Survival in Osteosarcoma. Orthop. Clin. North. Am. 2016, 47, 283–292. [Google Scholar] [CrossRef]
- Huang, Y.-M.; Hou, C.-H.; Hou, S.-M.; Yang, R.-S. The Metastasectomy and Timing of Pulmonary Metastases on the Outcome of Osteosarcoma Patients. Clin. Med. Oncol. 2009, 3, 99–105. [Google Scholar] [CrossRef]
- Beukhof, C.M.; Medici, M.; van den Beld, A.W.; Hollenbach, B.; Hoeg, A.; Visser, W.E.; de Herder, W.W.; Visser, T.J.; Schomburg, L.; Peeters, R.P. Selenium Status Is Positively Associated with Bone Mineral Density in Healthy Aging European Men. PLoS ONE 2016, 11, e0152748. [Google Scholar] [CrossRef] [PubMed]
- Zeng, H.; Cao, J.J.; Combs, G.F., Jr. Selenium in bone health: Roles in antioxidant protection and cell proliferation. Nutrients 2013, 5, 97–110. [Google Scholar] [CrossRef] [PubMed]
- Hiraoka, K.; Komiya, S.; Hamada, T.; Zenmyo, M.; Inoue, A. Osteosarcoma cell apoptosis induced by selenium. J. Orthop Res. 2001, 19, 809–814. [Google Scholar] [CrossRef]
- Chen, X.J.; Duan, F.D.; Zhang, H.H.; Xiong, Y.; Wang, J. Sodium selenite-induced apoptosis mediated by ROS attack in human osteosarcoma U2OS cells. Biol. Trace Elem. Res. 2012, 145, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chen, J.; Zhang, D.; Zhang, Y.; Wen, Y.; Li, L.; Zheng, L. Tumoricidal effects of a selenium (Se)-polysaccharide from Ziyang green tea on human osteosarcoma U-2 OS cells. Carbohydr Polym 2013, 98, 1186–1190. [Google Scholar] [CrossRef] [PubMed]
- Cheng, W.H.; Wu, R.T.; Wu, M.; Rocourt, C.R.; Carrillo, J.A.; Song, J.; Bohr, C.T.; Tzeng, T.J. Targeting Werner syndrome protein sensitizes U-2 OS osteosarcoma cells to selenium-induced DNA damage response and necrotic death. Biochem Biophys Res. Commun. 2012, 420, 24–28. [Google Scholar] [CrossRef]
- Tarrado-Castellarnau, M.; Cortes, R.; Zanuy, M.; Tarrago-Celada, J.; Polat, I.H.; Hill, R.; Fan, T.W.; Link, W.; Cascante, M. Methylseleninic acid promotes antitumour effects via nuclear FOXO3a translocation through Akt inhibition. Pharmacol. Res. 2015, 102, 218–234. [Google Scholar] [CrossRef]
- Huang, G.; Yong, B.C.; Xu, M.H.; Li, J.C.; Guo, H.H.; Shen, J.N. Analysis of Selenium Levels in Osteosarcoma Patients and the Effects of Se-Methylselenocysteine on Osteosarcoma Cells In Vitro. Nutr. Cancer 2015, 67, 847–856. [Google Scholar] [CrossRef]
- Wang, W.; Meng, F.B.; Wang, Z.X.; Li, X.; Zhou, D.S. Selenocysteine inhibits human osteosarcoma cells growth through triggering mitochondrial dysfunction and ROS-mediated p53 phosphorylation. Cell Biol. Int. 2018, 42, 580–588. [Google Scholar] [CrossRef]
- Tran, P.A.; Sarin, L.; Hurt, R.H.; Webster, T.J. Titanium surfaces with adherent selenium nanoclusters as a novel anticancer orthopedic material. J. Biomed. Mater. Res. A 2010, 93, 1417–1428. [Google Scholar] [CrossRef] [PubMed]
- Uskokovic, V.; Iyer, M.A.; Wu, V.M. One Ion to Rule Them All: Combined Antibacterial, Osteoinductive and Anticancer Properties of Selenite-Incorporated Hydroxyapatite. J. Mater. Chem. B 2017, 5, 1430–1445. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Valencia, C.; Freixeiro, P.; Serra, J.; Ferreiros, C.M.; Gonzalez, P.; Lopez-Alvarez, M. In vitro evaluation of the antibacterial and osteogenic activity promoted by selenium-doped calcium phosphate coatings. Biomed. Mater. 2017, 12, 015028. [Google Scholar] [CrossRef] [PubMed]
- Stolzoff, M.; Webster, T.J. Reducing bone cancer cell functions using selenium nanocomposites. J. Biomed. Mater. Res. A 2016, 104, 476–482. [Google Scholar] [CrossRef]
- Wang, Y.; Ma, J.; Zhou, L.; Chen, J.; Liu, Y.; Qiu, Z.; Zhang, S. Dual functional selenium-substituted hydroxyapatite. Interface Focus 2012, 2, 378–386. [Google Scholar] [CrossRef]
- Abul-Hassan, K.S.; Lehnert, B.E.; Guant, L.; Walmsley, R. Abnormal DNA repair in selenium-treated human cells. Mutat. Res. 2004, 565, 45–51. [Google Scholar] [CrossRef]
- Kolmas, J.; Pajor, K.; Pajchel, L.; Przekora, A.; Ginalska, G.; Oledzka, E.; Sobczak, M. Fabrication and physicochemical characterization of porous composite microgranules with selenium oxyanions and risedronate sodium for potential applications in bone tumors. Int. J. Nanomed. 2017, 12, 5633–5642. [Google Scholar] [CrossRef]
- Wojewoda, M.; Duszynski, J.; Szczepanowska, J. Antioxidant defence systems and generation of reactive oxygen species in osteosarcoma cells with defective mitochondria: Effect of selenium. Biochim Biophys Acta. 2010, 1797, 890–896. [Google Scholar] [CrossRef]
- Wojewoda, M.; Duszynski, J.; Wieckowski, M.; Szczepanowska, J. Effect of selenite on basic mitochondrial function in human osteosarcoma cells with chronic mitochondrial stress. Mitochondrion 2012, 12, 149–155. [Google Scholar] [CrossRef]
- Wojewoda, M.; Walczak, J.; Duszynski, J.; Szczepanowska, J. Selenite activates the ATM kinase-dependent DNA repair pathway in human osteosarcoma cells with mitochondrial dysfunction. Biochem. Pharmacol. 2015, 95, 170–176. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, J.; Hao, H.; Cai, M.; Wang, S.; Ma, J.; Li, Y.; Mao, C.; Zhang, S. In Vitro and in Vivo Mechanism of Bone Tumor Inhibition by Selenium-Doped Bone Mineral Nanoparticles. ACS Nano 2016, 10, 9927–9937. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Simon, H.-U. A novel link between p53 and ROS. Cell Cycle 2013, 12, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Chen, Y.; St Clair, D.K. ROS and p53: A versatile partnership. Free Radic. Biol. Med. 2008, 44, 1529–1535. [Google Scholar] [CrossRef] [PubMed]
- Renault, V.M.; Thekkat, P.U.; Hoang, K.L.; White, J.L.; Brady, C.A.; Kenzelmann Broz, D.; Venturelli, O.S.; Johnson, T.M.; Oskoui, P.R.; Xuan, Z.; et al. The pro-longevity gene FoxO3 is a direct target of the p53 tumor suppressor. Oncogene 2011, 30, 3207–3221. [Google Scholar] [CrossRef] [PubMed]
- Jiang, P.; Du, W.; Heese, K.; Wu, M. The Bad guy cooperates with good cop p53: Bad is transcriptionally up-regulated by p53 and forms a Bad/p53 complex at the mitochondria to induce apoptosis. Mol. Cell Biol 2006, 26, 9071–9082. [Google Scholar] [CrossRef]
- Stambolic, V.; MacPherson, D.; Sas, D.; Lin, Y.; Snow, B.; Jang, Y.; Benchimol, S.; Mak, T.W. Regulation of PTEN transcription by p53. Mol. Cell 2001, 8, 317–325. [Google Scholar] [CrossRef]
- Boulares, A.H.; Yakovlev, A.G.; Ivanova, V.; Stoica, B.A.; Wang, G.; Iyer, S.; Smulson, M. Role of Poly(ADP-ribose) Polymerase (PARP) Cleavage in Apoptosis. Caspase 3-Resistant PARP Mutant Increases Rates of Apoptosis in Transfected Cells. J. Biol. Chem. 1999, 274, 22932–22940. [Google Scholar] [CrossRef]
- Chaitanya, G.V.; Steven, A.J.; Babu, P.P. PARP-1 cleavage fragments: Signatures of cell-death proteases in neurodegeneration. Cell Commun. Signal. CCS 2010, 8, 31. [Google Scholar] [CrossRef]
- Alfranca, A.; Martinez-Cruzado, L.; Tornin, J.; Abarrategi, A.; Amaral, T.; de Alava, E.; Menendez, P.; Garcia-Castro, J.; Rodriguez, R. Bone microenvironment signals in osteosarcoma development. Cell. Mol. Life Sci. CMLS 2015, 72, 3097–3113. [Google Scholar] [CrossRef]
- Boyce, B.F.; Xing, L. Functions of RANKL/RANK/OPG in bone modeling and remodeling. Arch. Biochem Biophys 2008, 473, 139–146. [Google Scholar] [CrossRef]
- Branstetter, D.; Rohrbach, K.; Huang, L.Y.; Soriano, R.; Tometsko, M.; Blake, M.; Jacob, A.P.; Dougall, W.C. RANK and RANK ligand expression in primary human osteosarcoma. J. Bone Oncol. 2015, 4, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Navet, B.; Ando, K.; Vargas-Franco, J.W.; Brion, R.; Amiaud, J.; Mori, K.; Yagita, H.; Mueller, C.G.; Verrecchia, F.; Dumars, C.; et al. The Intrinsic and Extrinsic Implications of RANKL/RANK Signaling in Osteosarcoma: From Tumor Initiation to Lung Metastases. Cancers 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Trinidad, E.M.; Gonzalez-Suarez, E. RANKL inhibitors for osteosarcoma treatment: Hope and caution. Ann. Transl. Med. 2016, 4, 534. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Di Grappa, M.A.; Molyneux, S.D.; McKee, T.D.; Waterhouse, P.; Penninger, J.M.; Khokha, R. RANKL blockage prevents and treats aggressive osteosarcomas. Sci. Transl. Med. 2015, 7, 317ra197. [Google Scholar] [CrossRef] [PubMed]
- Moon, H.J.; Ko, W.K.; Han, S.W.; Kim, D.S.; Hwang, Y.S.; Park, H.K.; Kwon, I.K. Antioxidants, like coenzyme Q10, selenite, and curcumin, inhibited osteoclast differentiation by suppressing reactive oxygen species generation. Biochem Biophys Res. Commun. 2012, 418, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Nadiminty, N.; Liao, H.; Cao, A.C. Selenium inhibits the expression of osteogenic genes associated with bone remodeling. In Proceedings of the AACR Annual Meeting, Los Angeles, CA, USA, 14–18 April 2007; American Association for Cancer Research: Philadephia, PA, USA, 2007; p. 2192. [Google Scholar]
- Zheng, Y.; Wang, G.; Chen, R.; Hua, Y.; Cai, Z. Mesenchymal stem cells in the osteosarcoma microenvironment: Their biological properties, influence on tumor growth, and therapeutic implications. Stem Cell Res. Ther. 2018, 9, 22. [Google Scholar] [CrossRef]
- Liu, H.; Bian, W.; Liu, S.; Huang, K. Selenium protects bone marrow stromal cells against hydrogen peroxide-induced inhibition of osteoblastic differentiation by suppressing oxidative stress and ERK signaling pathway. Biol. Trace Elem. Res. 2012, 150, 441–450. [Google Scholar] [CrossRef]
- Zheng, C.; Wang, J.; Liu, Y.; Yu, Q.; Liu, Y.; Deng, N.; Liu, J. Functional Selenium Nanoparticles Enhanced Stem Cell Osteoblastic Differentiation through BMP Signaling Pathways. Adv. Funct. Mater. 2014, 24, 6872–6883. [Google Scholar] [CrossRef]
- Ahmed, H.H.; Aglan, H.A.; Mabrouk, M.; Abd-Rabou, A.A.; Beherei, H.H. Enhanced mesenchymal stem cell proliferation through complexation of selenium/titanium nanocomposites. J. Mater. Sci. Mater. Med. 2019, 30, 24. [Google Scholar] [CrossRef]
- Margulies, B.S.; Damron, T.A.; Allen, M.J. The differential effects of the radioprotectant drugs amifostine and sodium selenite treatment in combination with radiation therapy on constituent bone cells, Ewing’s sarcoma of bone tumor cells, and rhabdomyosarcoma tumor cells in vitro. J. Orthop Res. 2008, 26, 1512–1519. [Google Scholar] [CrossRef]
- Tran, P.A.; Sarin, L.; Hurt, R.H.; Webster, T.J. Differential effects of nanoselenium doping on healthy and cancerous osteoblasts in coculture on titanium. Int. J. Nanomed. 2010, 5, 351–358. [Google Scholar]
- Ebert, R.; Ulmer, M.; Zeck, S.; Meissner-Weigl, J.; Schneider, D.; Stopper, H.; Schupp, N.; Kassem, M.; Jakob, F. Selenium supplementation restores the antioxidative capacity and prevents cell damage in bone marrow stromal cells in vitro. Stem Cells 2006, 24, 1226–1235. [Google Scholar] [CrossRef] [PubMed]
- Park, S.H.; Kim, J.H.; Nam, S.W.; Kim, B.W.; Kim, G.Y.; Kim, W.J.; Choi, Y.H. Selenium improves stem cell potency by stimulating the proliferation and active migration of 3T3-L1 preadipocytes. Int. J. Oncol. 2014, 44, 336–342. [Google Scholar] [CrossRef] [PubMed]
- Kuzmenko, V. Influence of silver and selenium nanoparticles on mesenchymal stromal cells. Environ. Res. Eng. Manag. 2019, 74. [Google Scholar] [CrossRef]
- Burgess, D.J. Anticancer Drugs: Selective oxycution? Nat. Rev. Drug Discov. 2011, 10, 658. [Google Scholar] [CrossRef]
- Liu, J.; Wang, Z. Increased Oxidative Stress as a Selective Anticancer Therapy. Oxidative Med. Cell. Longev. 2015, 2015, 294303. [Google Scholar] [CrossRef]
- Carrle, D.; Bielack, S.S. Current strategies of chemotherapy in osteosarcoma. Int. Orthop. (SICOT) 2006, 30, 445–451. [Google Scholar] [CrossRef]
- Spina, A.; Sorvillo, L.; Di Maiolo, F.; Esposito, A.; D’Auria, R.; Di Gesto, D.; Chiosi, E.; Naviglio, S. Inorganic phosphate enhances sensitivity of human osteosarcoma U2OS cells to doxorubicin via a p53-dependent pathway. J. Cell. Physiol. 2013, 228, 198–206. [Google Scholar] [CrossRef]
- Clement, M.V.; Pervaiz, S. Intracellular superoxide and hydrogen peroxide concentrations: A critical balance that determines survival or death. Redox Rep. Commun. Free Radic. Res. 2001, 6, 211–214. [Google Scholar] [CrossRef]
- Wang, J.; Yi, J. Cancer cell killing via ROS: To increase or decrease, that is the question. Cancer Biol. Ther. 2014, 7, 1875–1884. [Google Scholar] [CrossRef]
- Wang, Y.; Hao, H.; Liu, H.; Wang, Y.; Li, Y.; Yang, G.; Ma, J.; Mao, C.; Zhang, S. Selenite-Releasing Bone Mineral Nanoparticles Retard Bone Tumor Growth and Improve Healthy Tissue Functions In Vivo. Adv. Healthc. Mater. 2015, 4, 1813–1818. [Google Scholar] [CrossRef] [PubMed]
- Bierke, P.; Svedenstal, B.M. The influence of selenium, vitamin E, and oestrogen on the development of tumours in mice exposed to 90Sr. Acta Oncol. (Stockh. Swed.) 1994, 33, 963–967. [Google Scholar] [CrossRef] [PubMed]
- Zeng, H.; Combs, G.F., Jr. Selenium as an anticancer nutrient: Roles in cell proliferation and tumor cell invasion. J. Nutr. Biochem. 2008, 19, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Amaral, A.F.; Cantor, K.P.; Silverman, D.T.; Malats, N. Selenium and bladder cancer risk: A meta-analysis. Cancer Epidemiol Biomark. Prev. 2010, 19, 2407–2415. [Google Scholar] [CrossRef]
- Klein, E.A.; Thompson, I.M., Jr.; Tangen, C.M.; Lucia, M.S.; Goodman, P.J.; Minasian, L.M.; Ford, L.G.; Parnes, H.L.; Gaziano, J.M.; Karp, D.D.; et al. Vitamin E and the Risk of Prostate Cancer. The Selenium and Vitamin E Cancer Prevention Trial (SELECT). JAMA 2011, 306, 1549–1556. [Google Scholar] [CrossRef]
- WHO. Diet. Nutrition and the Prevention of Chronic Diseases; World Health Organization: Geneva, Switzerland, 2003; pp. 1–149. [Google Scholar]
- Vinceti, M.; Filippini, T.; del Giovane, C.; Dennert, G.; Zwahlen, M.; Brinkman, M.; Zeegers, M.P.; Horneber, M.; D’Amico, R.; Crespi, C.M. Selenium for preventing cancer. Cochrane Database Syst Rev. 2018, 1. [Google Scholar] [CrossRef]
- Kristal, A.R.; Darke, A.K.; Morris, J.S.; Tangen, C.M.; Goodman, P.J.; Thompson, I.M.; Meyskens, F.L., Jr.; Goodman, G.E.; Minasian, L.M.; Parnes, H.L.; et al. Baseline selenium status and effects of selenium and vitamin e supplementation on prostate cancer risk. J. Natl Cancer Inst. 2014, 106, djt456. [Google Scholar] [CrossRef]
- Lener, M.R.; Gupta, S.; Scott, R.J.; Tootsi, M.; Kulp, M.; Tammesoo, M.-L.; Viitak, A.; Metspalu, A.; Serrano-Fernández, P.; Kładny, J.; et al. Can selenium levels act as a marker of colorectal cancer risk? BMC Cancer 2013, 13, 214. [Google Scholar] [CrossRef]
- Thompson, P.A.; Ashbeck, E.L.; Roe, D.J.; Fales, L.; Buckmeier, J.; Wang, F.; Bhattacharyya, A.; Hsu, C.H.; Chow, H.H.; Ahnen, D.J.; et al. Selenium Supplementation for Prevention of Colorectal Adenomas and Risk of Associated Type 2 Diabetes. J. Natl Cancer Inst. 2016, 108. [Google Scholar] [CrossRef]
- Vinceti, M.; Dennert, G.; Crespi, C.M.; Zwahlen, M.; Brinkman, M.; Zeegers, M.P.A.; Horneber, M.; D’Amico, R.; del Giovance, C. Selenium for preventing cancer. Cochrane Database Syst Rev. 2014. [Google Scholar] [CrossRef]
| Selenium Types | In vitro Models | Treatment Condition | Results | Ref |
|---|---|---|---|---|
| SeO2 | Human osteosarcoma MG-63, U-2 OS and Saos-2 cells | 0.02−20 µg/mL (equivalent to 0.18–180.25 µM) for 24 h |
| [49] |
| SeO32− | Primary rat growth plate chondrocytes and non-tumourigenic mouse osteoblast MC3T3 cells | Treatment with SeO32− (0.005–25 µM). Treatment time not indicated. |
| [86] |
| Pretreatment with 0.05 µM SeO32−. Treatment time not indicated. |
| |||
| Human osteosarcoma U-2 OS cells | Pretreatment of SeO32− (0.01–10 µM) for 1 h, followed by 24 h cisplatin treatment |
| [61] | |
| Human blood lymphocytes | Pretreatment of SeO32− (0.01–10 µM) for 1 h, followed by 150 cGy γ irradiation |
| ||
| Human osteosarcoma U-2 OS cells, non-tumourigenic human embryonic kidney 293 cells and rat skeletal muscle L6 cells | Treated (5–40 µM) for 24–48 h |
| [50] | |
| Se-Poly | Human osteosarcoma U-2 OS cells | Treated with 25–200 µg/mL of purified Se-Poly for 24 to 72 h |
| [51] |
| MSeA | Human osteosarcoma U-2 OS cells | Treated with MSeA (0–10 µM) for 48 h with or without the knockdown of WRN |
| [52] |
| Treated with 1.5 and 5 µM MSeA up to 72 h |
| [53] | ||
| Se-MSC | Human osteosarcoma MG-63 and U-2 OS cellsDrug-resistant human osteosarcoma Saos-2/MTX300 cells | Treated with Se-MSC (0–150 µM) up to 96 h |
| [54] |
| Treated with Se-MSC (0–250 µM) up to 96 h |
| |||
| SeC | Human osteosarcoma MG-63 cells | Treated with SeC (0–20 µM) up to 72 h |
| [55] |
| SeO32−-doping titanium substrate | Mouse osteosarcoma K7M2−pCl Neo cells and non-tumourigenic human osteoblast hFOB 1.19 cells | Cultured on selenite-doping titanium surface for 4, 24 and/or 72 h |
| [56] |
| Cultured K7M2−pCl Neo cells and hFOB 1.19 cells with acellular media that collected from selenium-doping orthopaedic implant material (3 days incubation) for 3 days |
| |||
| Mouse osteosarcoma osteoblast from ATCC (unknown cell types) and normal primary human calvarial osteoblast | Cultured on SeO32−-doping titanium surface for 3 days |
| [87] | |
| Both cancerous and normal osteoblast were co-cultured on the uncoated or SeO32−-doping titanium surfaces for 4–65 h |
| |||
| Selenium-doped calcium phosphate coating | Human osteosarcoma MG-63 cells and non-tumourigenic preosteoblast MC3T3-E1 cells | Cultured the cells with selenium-doped coatings (0.6 and 2.7 at% selenium) for 24 h or up to 21 days incubation |
| [58] |
| SeNP-PLLA nanocomposites | Human osteosarcoma MG-63 cells and non-tumourigenic foetal osteoblast hFOB cells | Incubated with SeNP-PLLA (0.025M SeO32−) for 48 h |
| [59] |
| SeHAN | Human osteosarcoma MG-63 cells and normal human BMSCs | SeHAN with different preparation concentration and treatment concentration (50–200 µg/mL) was added into culture medium with a monolayer of osteosarcoma cells for 3 days |
| [60] |
| Human osteosarcoma MNNG/HOS cells | SeHANs (3, 6 and 10% molar ratio of selenium and phosphate) in pH 5.0 and pH 7.4 were added to a monolayer of osteosarcoma cells at 50 µg/mL up to 24 h (2 µM sodium SeO32− as control) |
| [66] | |
| SeHA | Mouse osteosarcoma K7M2 cells and normal primary mouse lung fibroblast (isolated from C57B/6/J mouse lungs) | SeHA (0.102−3.0 wt%) in 2 mg/mL were added to monolayer cells for 48 h |
| [57] |
| Non-tumourigenic mouse calvarial preosteoblastic MC3T3-E1 subclone 4 cells | SeHA (0.102−3.0 wt%) in 5 mg/mL were added to fibroblast for 72 h |
| ||
| SeHA/ALG | Human osteosarcoma Saos-2 cells and non-tumourigenic hFOB 1.19 cells | Treated with supernatant extracts (50 and 100%) from SeHA/ALG, SeHA/ALG/RIS I and II for 24 h |
| [62] |
| Selenium Types | In vivo Models and Treatment | Positive Results | Negative Results | Ref |
| SeO2 | BALB/c nu/nu mice with subcutaneous human osteosarcoma KOS xenograft were fed with SeO2−containing drinking water (0.2 and 2 µg/mL) until day 44 after inoculation |
|
| [49] |
| SeO32− | α-tocopherol acetate (0.5 mg) with or without SeO32− (10 µg) was injected intraperitoneally into 90Sr-induced osteosarcoma mice every 2 weeks, starting from day 105 after exposure until 14-month. The regime changed to 30-day intervals for the rest of the life-span. |
| [98] | |
| Se-Poly | U-2 OS xenograft (100 mm3) BALB/c nude mice were orally treated with Se-Poly from Ziyang green tea (100, 200 and 400 mg/kg) daily for 28 days. |
| [51] | |
| SeC | Nude mice with MG-63 xenograft (50 mm3) was treated with intravenous injection of SeC (5 and 10 mg/kg/day) every other day for 2 weeks |
| [55] | |
| SeHAN | Nude mice with an orthotopic intrafemorally injection of SOSP-9607 xenograft were treated with intratumoural injection of SeHAN for 30 days |
| [97] | |
| BALB/c nude mice with osteosarcoma xenograft (100 mm3) were treated with intratumoural injection of 10%SeHAN every 3 days for 30 days |
| [66] | ||
| Selenium levels | Nine osteosarcoma patients and nine non-osteosarcoma patients |
| [54] | |
| Paired osteosarcoma and normal bone tissues from 14 osteosarcoma patients |
|
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pang, K.-L.; Chin, K.-Y. Emerging Anticancer Potentials of Selenium on Osteosarcoma. Int. J. Mol. Sci. 2019, 20, 5318. https://doi.org/10.3390/ijms20215318
Pang K-L, Chin K-Y. Emerging Anticancer Potentials of Selenium on Osteosarcoma. International Journal of Molecular Sciences. 2019; 20(21):5318. https://doi.org/10.3390/ijms20215318
Chicago/Turabian StylePang, Kok-Lun, and Kok-Yong Chin. 2019. "Emerging Anticancer Potentials of Selenium on Osteosarcoma" International Journal of Molecular Sciences 20, no. 21: 5318. https://doi.org/10.3390/ijms20215318
APA StylePang, K.-L., & Chin, K.-Y. (2019). Emerging Anticancer Potentials of Selenium on Osteosarcoma. International Journal of Molecular Sciences, 20(21), 5318. https://doi.org/10.3390/ijms20215318






