The Main (Glyco) Phospholipid (MPL) of Thermoplasma acidophilum
Abstract
1. Introduction
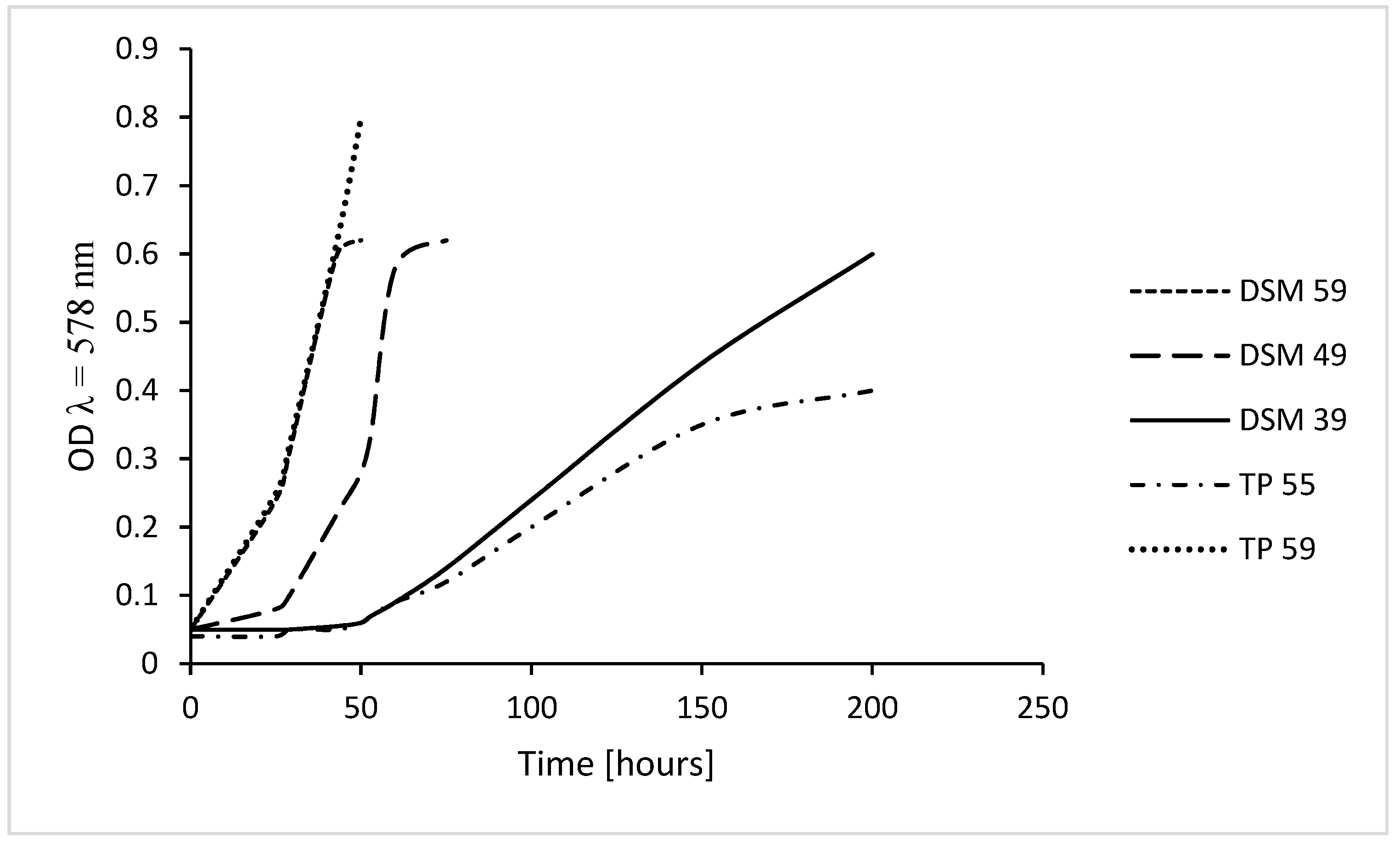
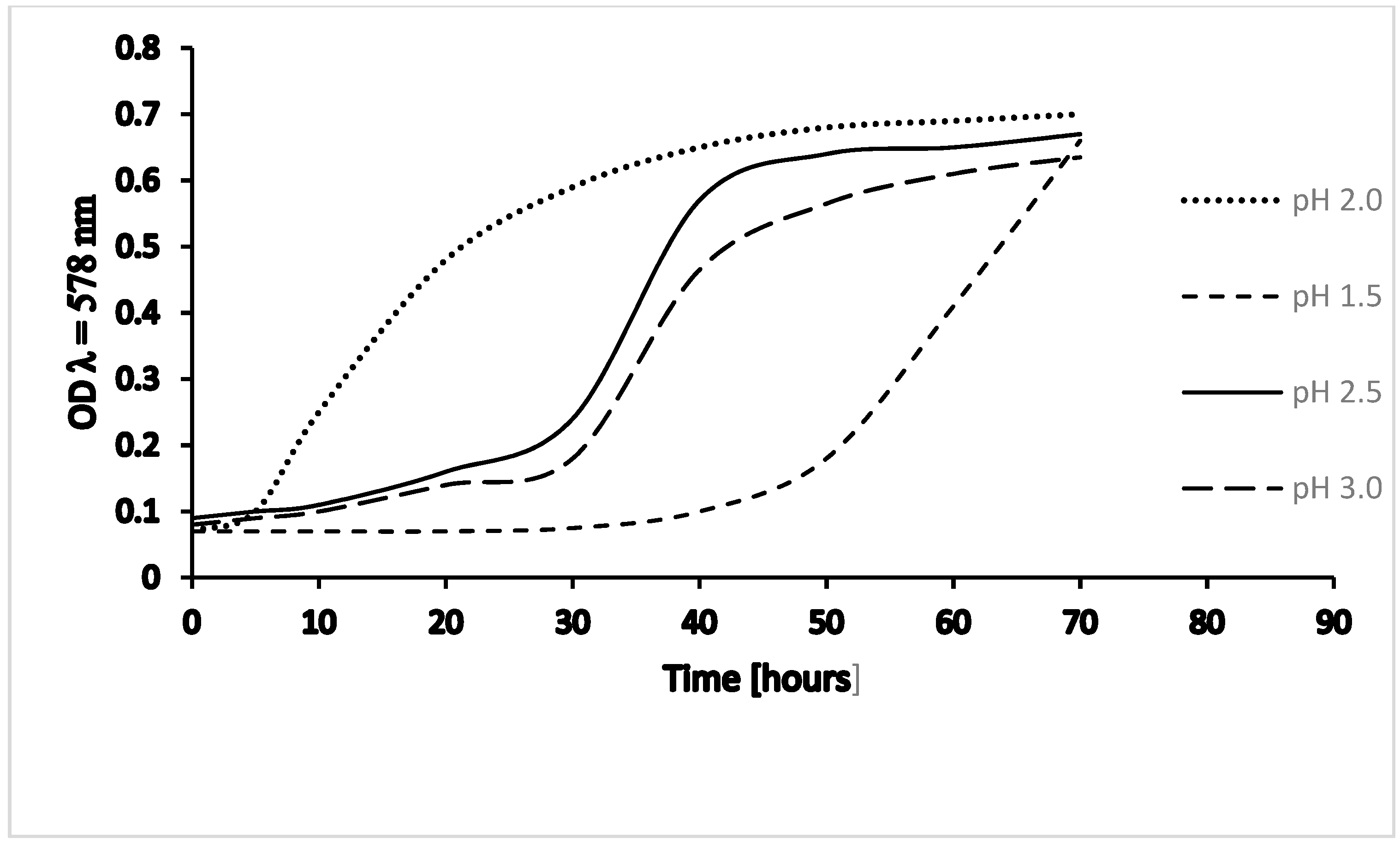
2. Growth of Thermoplasma
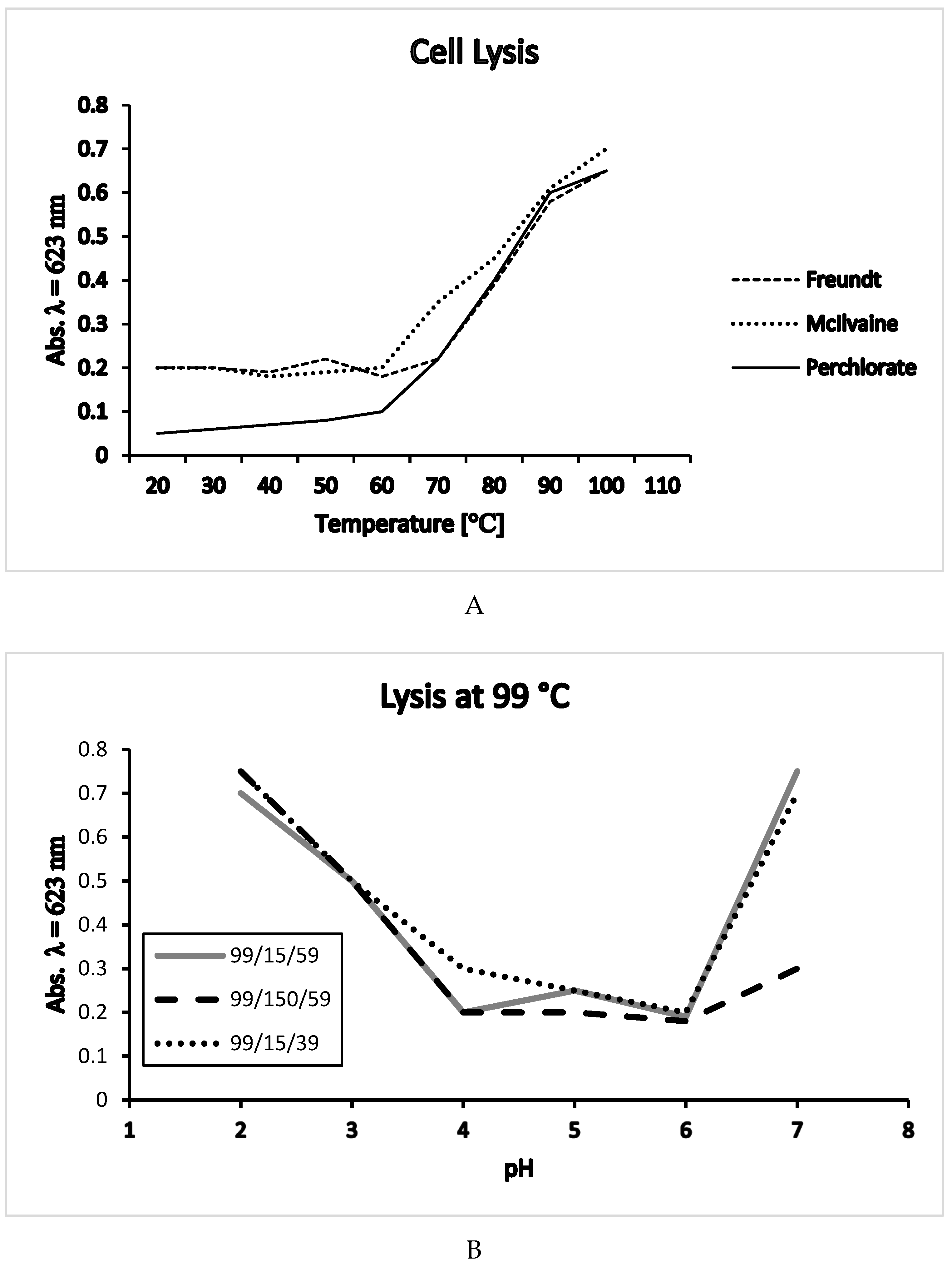
3. Cell Lysis Experiments
4. Composition of Membrane Lipids
5. Differential Thermal Analysis (DTA) and Differential Scanning Calorimetry (DSC)
5.1. Cryoprotectants
5.2. Mixture of MPL with Other Lipids
5.3. Mixture with Cholesterol
5.4. Mixture with Pore-forming Peptides and Ionophores
6. Model Membranes
6.1. Black Lipid Membranes
6.2. Monomolecular Films at Water/Air Interface
6.3. MPL Liposomes
6.4. Mixed Liposomes
6.5. Proton Permeability and Light-Driven Proton Pumping
6.5.1. Proton Permeability
6.5.2. Reconstitution of Bacteriorhodopsin (BR) and Light-Driven Proton Pumping
6.5.3. Light-Driven Liposomal ATP Synthesis
7. Applications
Funding
Patents
Conflicts of Interest
Abbreviations
| AFM | atomic force microscope |
| ATP | adenosine-triphosphate |
| BLM | black lipid membrane |
| BR | bacteriorhodopsin |
| CF | carboxyfluorescein |
| DHMG | 1,2-dihexadecyl-3-O-ß-D-maltosyl-sn-glycerol |
| DMPC | dimyristoylphosphatidylcholine |
| DMSO | dimethyl-sulfoxide |
| DOC | deoxycholate |
| DOPC | dioleoylphosphatidylcholine |
| DPGG | dipalmitylglucosylglycerol |
| DPhG | diphytanylglycerol |
| DPhGG | diphytanylglucosylglycerol |
| DPhPC | diphytanylphosphatidylcholine |
| DPPC | dipalmitoylphosphatidylcholine |
| DPPG | dipalmitoylphosphatidylglycerol |
| DSPC | distearoylphosphatidycholine |
| DSM | Deutsche Sammlung von Mikroorganismen |
| DSC | differential scanning calorimetry |
| DTA | differential thermoanalysis |
| EL | egg (yolk) lecithin |
| EM | electron microscopy |
| EPR/ESR | electron paramagnetic resonance/electron spin resonance |
| FCCP | carbonylcyanide-4-(trifluoromethoxy)phenylhydrazone |
| GDNT | glycerol-dialkyl-nonitol-tetraether |
| GL | glycolipid |
| LPS | lipopolysaccharide |
| MGL | main glycolipid |
| ML | Micrococcus luteus lipid extract |
| MPL | main glycophospholipid from Thermoplasma acidophilum |
| OD | optical density |
| Palm | palmitic acid |
| PBFI | potassium-binding (benzofuran) fluorescence indicator |
| RT | room temperature |
| SBL | soybean lecithin |
| SDS | sodium dodecyl-sulfate |
| TDOC | taurodeoxycholate |
| TEL | tetraether lipid |
| T.a. | Thermoplasma acidophilum |
| TPLF | total polar lipid fraction |
| TPLF | Ta total polar lipid fraction from T.a. |
| Val | valinomycin |
References
- Darland, G.; Brock, T.D.; Samsonoff, W.; Conti, S.F. A thermophilic, acidophilic mycoplasm isolated from a coal refuse pile. Science 1970, 170, 1416–1418. [Google Scholar] [CrossRef] [PubMed]
- Langworthy, T.A.; Smith, P.F.; Mayberry, W.R. Lipids of Thermoplasma acidophilum. J. Bacteriol. 1972, 112, 1193–1200. [Google Scholar] [PubMed]
- Woese, C.R.; Magrum, L.J.; Fox, G.E. Archaebacteria. J. Mol. Evol. 1978, 11, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Woese, C.R.; Maniloff, J.; Zablen, B. Phylogenetic analysis of the mycoplasmas. Proc. Natl. Acad. Sci. USA 1980, 77, 494–498. [Google Scholar] [CrossRef] [PubMed]
- Segerer, A.; Langworthy, T.A.; Stetter, K.O. Thermoplasma acidophilum and Thermoplasma volcanium sp. nov. from Solfatara Fields. Syst. Appl. Microbiol. 1988, 10, 161–171. [Google Scholar] [CrossRef]
- Freisleben, H.-J. The archaeon Thermoplasma acidophilum. In Proceedings of the Annual Meeting of the Indonesian Society of Microbiology, Manado, Indonesia, 19 July 1994. [Google Scholar]
- Freisleben, H.-J.; Deisinger, B. Technical and medical applications of archaeal tetraether lipid. In Proceedings of the 3RD International Symposium on Advanced and Aerospace Science and Technology in Indonesia, Jakarta, Indonesia, 31 August–3 September 1998; pp. XV–XXIV. [Google Scholar]
- Freisleben, H.-J. Thermoplasma Species and Archaeal Tetraether Lipids. Hayati Indones. 1999, 6, 51–55. [Google Scholar]
- Huber, G.; Huber, R.; Jones, B.E.; Lauerer, G.; Neuner, A.; Segerer, A.; Stetter, K.O.; Degens, E.T. Hyperthermophilic Archaea and Bacteria Occurring within Indonesian Hydrothermal Areas. Syst. Appl. Microbiol. 1991, 14, 397–404. [Google Scholar] [CrossRef]
- Handayani, S.; Santoso, I.; Freisleben, H.-J.; Huber, H.; Andi; Ardiansyah, F.; Mulyanto, C.; Luthfa, Z.; Saleh, R.; Freisleben, S.K.U.; et al. Archaeal life on Tangkuban Perahu—Sampling and culture growth in Indonesian laboratories. Hayati J. Biosci. 2012, 19, 150–154. [Google Scholar] [CrossRef]
- Malik, A.; Santoso, I.; Yehuda, A.; Freisleben, S.K.U.; Wanandi, S.I.; Huber, H.; Luthfa, Z.; Saleh, R.; Freisleben, H.-J. Characterization of Thermoplasma species cultured from sampling on Tangkuban Perahu, Indonesia. Microbiol. Indones. 2014, 8, 16–23. [Google Scholar] [CrossRef]
- Freisleben, H.-J.; Henkel, L.; Gutermann, R.; Rudolph, P.; John, G.; Sternberg, B.; Winter, S.; Ring, K. Fermentor cultivation of Thermoplasma acidophilum for the production of cell mass and of the main phospholipid fraction. Appl. Microbiol. Biotechnol. 1994, 40, 745–752. [Google Scholar] [CrossRef]
- Rudolph, P. Herstellung und Charakterisierung von Liposomen aus Tetraetherlipid des Archaebakteriums Thermoplasma acidophilum. [Fabrication and Characterization of Liposomes from Tetraether Lipid of the Archaebacterium Thermoplasma acidophilum]. Ph.D. Thesis, Goethe-Universität, Frankfurt am Main, Germany, 1993. [Google Scholar]
- Mehlhorn, R.J.; Candau, P.; Packer, L. Measurements of Volumes and Electrochemical Gradients with spin Probes in Membrane Vesicles. In Methods in Enzymology; Packer, L., Ed.; Academic Press: New York, NY, USA, 1982; pp. 751–762. [Google Scholar]
- Lomax, T.L.; Mehlhorn, R.J.; Briggs, W.R. Active auxin uptake by Zucchini membrane vesicles: Quantitation using ESR volume and ΔpH determinations. Proc. Natl. Acad. Sci. USA 1985, 82, 6541–6545. [Google Scholar] [CrossRef] [PubMed]
- Efstathiu, G. Einfluss der Temperatur und des extrazellulären pH auf die Membranstabilität von Thermoplasma acidophilum. [The Influence of Temperature and Extracelluar pH on Membrane Stability of Thermoplasma acidophilum]. Master’s Thesis, Goethe-Universität, Frankfurt am Main, Germany, 1987. [Google Scholar]
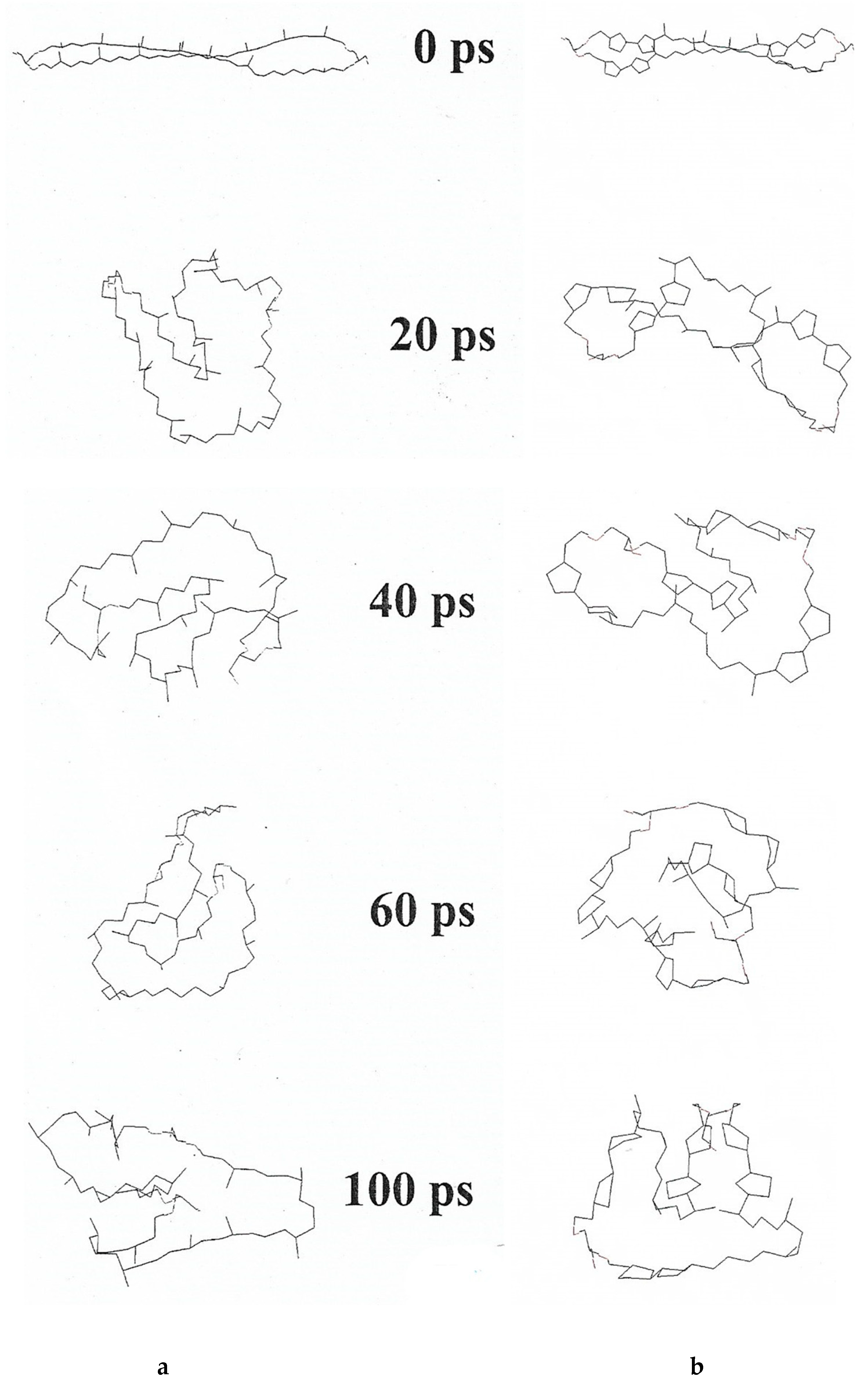
- Luthfa, Z.; Freisleben, H.-J.; Saleh, R. Temperature and pH-dependent molecular dynamics of Thermoplasma acidophilum tetraether lipid membrane in a computer-simulated model. Int. J. Mater. Eng. Technol. 2015, 13, 161–185. [Google Scholar] [CrossRef]
- Shimada, H.; Nemoto, N.; Shida, Y.; Oshima, T.; Yamagishi, A. Complete polar lipid composition of Thermoplasma acidophilum HO-62 determined by high performance liquid chromatography with evaporative light-scattering detection. J. Bacteriol. 2002, 184, 556–563. [Google Scholar] [CrossRef] [PubMed]
- Chong, P.L.-G. Archaebacterial bipolar tetraether lipids: Physico-chemical and membrane properties. Chem. Phys. Lipids 2010, 163, 253–265. [Google Scholar] [CrossRef]
- Uda, I.; Sugai, A.; Itoh, Y.H.; Itoh, T. Variation in molecular species of polar lipids from Thermoplasma acidophilum depends on growth temperature. Lipids 2001, 36, 103–105. [Google Scholar] [CrossRef]
- Shimada, H.; Nemoto, N.; Shida, Y.; Oshima, T.; Yamagishi, A. Effects of pH and Temperature on the Composition of Polar Lipids in Thermoplasma acidophilum HO-62. J. Bacteriol. 2008, 190, 5404–5411. [Google Scholar] [CrossRef]
- Ernst, M.; Freisleben, H.-J.; Antonopoulos, E.; Henkel, L.; Mlekusch, W.; Reibnegger, G. Calorimetry of archaeal tetraether lipid—Indication of a novel metastable thermotropic phase in the main phosphotetraether lipid from Thermoplasma acidophilum cultured at 59 °C. Chem. Phys. Lipids 1998, 94, 1–12. [Google Scholar] [CrossRef]
- Swain, M.; Brisson, J.-R.; Sprott, G.D.; Cooper, F.P.; Patel, C.B. Identification of ß-L-glucose as the sugar moiety of the main polar lipid from Thermoplasma acidophilum. Biochim. Biophys. Acta 1997, 1345, 56–64. [Google Scholar] [CrossRef]
- Antonopoulos, E.; Freisleben, H.-J.; Mulyanto, C.; Krisnamurti, D.G.B.; Estuningtyas, A.; Ridwan, R.; Freisleben, S.K.U. Fractionation and purification of membrane lipids from the archaeon Thermoplasma acidophilum DSM 1728/10217. Separ. Purific. Technol. 2013, 110, 119–126. [Google Scholar] [CrossRef]
- Langworthy, T.A. Lipids of Thermoplasma. Methods Enzymol. 1982, 88, 396–406. [Google Scholar]
- Blöcher, D.; Gutermann, R.; Henkel, B.; Ring, K. Physicochemical characterization of tetraether lipids from Thermoplasma acidophilum. Differential scanning calorimetry studies on glycolipids and glycophospholipids. Biochim. Biophys. Acta 1984, 778, 74–80. [Google Scholar] [CrossRef]
- Blöcher, D.; Six, L.; Gutermann, R.; Henkel, B.; Ring, K. Physicochemical characterization of tetraether lipids from Thermoplasma acidophilum. Calorimetric studies on miscibility with diether model lipids carrying branched and unbranched alkyl chains. Biochim. Biophys. Acta 1985, 818, 333–342. [Google Scholar] [CrossRef]
- Blöcher, D.; Gutermann, R.; Henkel, B.; Ring, K. Physicochemical characterization of tetraether lipids from Thermoplasma acidophilum. V. Evidence for the existence of a metastable phase in lipids with uncyclizated hydrocarbon chains. Biochim. Biophys. Acta 1990, 1024, 54–60. [Google Scholar] [CrossRef]
- Blöcher, D.; Ring, K. Mixtures of tetraether lipids from Thermoplasma acidophilum with varying degree of cyclization show a kinetic effect for a metastable phase. Chem. Phys. Lipids 1991, 58, 233–239. [Google Scholar] [CrossRef]
- Blöcher, D. Modulation of calorimetric data by cryoprotectants. Studia Biophys. 1990, 137, 99–106. [Google Scholar]
- Blöcher, D.; Gutermann, R.; Henkel, B.; Ring, K. Physicochemical characterization of tetraether lipids from Thermoplasma acidophilum. IV. Calorimetric studies on the miscibility of tetraether lipids with dipalmitoylphosphatidylcholine and dipalmitoylphosphatidylglycerol. Naturforschung 1985, 40c, 606–611. [Google Scholar] [CrossRef]
- Blöcher, D.; Freisleben, H.-J.; Ring, K. Thermotropic properties of dispersions of cholesterol with tetraether lipids from Thermoplasma acidophilum. Arch. Biochem. Biophys. 1991, 290, 224–228. [Google Scholar] [CrossRef]
- Blöcher, D.; Freisleben, H.-J.; Becker, G.; Jung, G.; Ring, K. Incorporation of synthetic peptide helices in membranes of tetraether lipids from Thermoplasma acidophilum. A calorimetric study. Biochim. Biophys. Acta 1991, 1065, 160–166. [Google Scholar] [CrossRef]
- Freisleben, H.-J.; Blöcher, D.; Ring, K. Calorimetry of tetraether lipids from Thermoplasma acidophilum. Incorporation of alamethicin, melittin, valinomycin and nonactin. Arch. Biochem. Biophys. 1992, 294, 418–426. [Google Scholar] [CrossRef]
- Garidel, P.; Kaconis, Y.; Heinbockel, L.; Wulf, M.; Gerber, S.; Munk, A.; Vill, V.; Brandenburg, K. Self-Organisation, Thermotropic and Lyotropic Properties of Glycolipids Related to their Biological Implications. Open Biochem. J. 2015, 9, 49–72. [Google Scholar]
- Yeagle, P.L. Cholesterol and the cell membrane. Biochim. Biophys. Acta 1985, 822, 267–287. [Google Scholar] [CrossRef]
- Finean, J.B. Interaction between cholesterol and phospholipid in hydrated bilayers. Chem. Phys. Lipids 1990, 54, 147–156. [Google Scholar] [CrossRef]
- Davis, P.J.; Keough, K.M.W. Differential scanning calorimetric studies of aqueous dispersions of mixtures of cholesterol with some mixed-acid and single-acid phosphatidylcholines. Biochemistry 1983, 22, 6334–6340. [Google Scholar] [CrossRef]
- Ruocco, M.J.; Shipley, G.G. Interaction of cholesterol with galactocerebroside and galactocerebroside-phosphatidylcholine bilayer membranes. Biophys. J. 1984, 46, 695–707. [Google Scholar] [CrossRef]
- Papahadjopoulos, D.; Jacobson, K.; Poste, G.; Shepherd, G. effects of local anaesthetics on membrane properties. 1. Changes in the fluidity of phospholipid bilayers. Biochim. Biophys. Acta 1975, 394, 504–519. [Google Scholar] [CrossRef]
- Mueller, P.; Rudin, D.O.; Tien, H.T.; Wescott, W.C. Reconstitution of cell membrane structure in vitro and its transformation into an excitable system. Nature 1962, 194, 979–980. [Google Scholar] [CrossRef] [PubMed]
- Stern, J.; Freisleben, H.-J.; Janku, S.; Ring, K. Black lipid membranes of tetraether lipids from Thermoplasma acidophilum. Biochim. Biophys. Acta 1992, 1128, 227–236. [Google Scholar] [CrossRef]
- Gliozzi, A.; Paoli, G.; Rolandi, R.; De Rosa, M.; Gambacorta, A. Structure and Transport Properties of Artificial Bipolar Lipid Membranes. Bioelectrochem. Bioenerg. 1982, 141, 591–601. [Google Scholar] [CrossRef]
- Benz, R.; Fröhlich, O.; Läuger, P.; Montal, M. Electrical Capacity of Black Lipid Films and of Lipid Bilayers Made from Monolayers. Biochim. Biophys. Acta 1975, 394, 323–334. [Google Scholar] [CrossRef]
- Montal, M.; Mueller, P. Formation of Bimolecular Membranes from Lipid Monolayers and a Study of Their Electrical Properties. Proc. Nat. Acad. Sci. USA 1972, 69, 3561–3566. [Google Scholar] [CrossRef]
- White, S.H.; Petersen, D.C.; Simon, S.; Yafuso, M. Formation of Planar Bilayer Membranes from Lipid Monolayers—A Critique. Biophys. J. 1976, 16, 481–489. [Google Scholar] [CrossRef]
- White, S.H. Formation of “Solvent-Free” Black Lipid Bilayer Membranes from Glyceryl Monooleate Dispersed in Squalene. Biophys. J. 1978, 23, 337–347. [Google Scholar] [CrossRef]
- Gliozzi, A.; Rolandi, R.; De Rosa, M.; Gambacorta, A. Artificial Black Membranes from Bipolar Lipids of Thermophilic Archaebacteria. Biophys. J. 1982, 37, 563–566. [Google Scholar] [CrossRef]
- Strobl, C.; Six, L.; Heckmann, K.; Henkel, B.; Ring, K. Physicochemical characterization of tetraether lipids from Thermoplasma acidophilum. II. Film balance studies on the monomolecular organization of the main glycophospholipid in monofilms. Z. Naturforsch. C 1985, 40, 219–222. [Google Scholar] [CrossRef][Green Version]
- Phillips, M.C.; Chapman, D. Monolayer characteristics of saturated 1,2-diacylphosphatidyl-cholines (lecithins) and phosphatidylethanolamines at the air-water interface. Biochim. Biophys. Acta 1968, 163, 301–303. [Google Scholar] [CrossRef]
- Six, L.; Ruess, K.-P.; Liefländer, M. An efficient and stereoselective synthesis of 1,2-O-dialkyl-3-O-ß-D-glucosyl-sn-glycerols. Tetrahedron Lett. 1983, 24, 1229–1232. [Google Scholar] [CrossRef]
- Elferinck, M.G.L.; de Wit, J.G.; Demel, R.; Driessen, A.J.M.; Konings, W.N. Functional reconstitution of membrane proteins in monolayer liposomes from bipolar lipids of Sulfolobus acidocaldarius. J. Biol. Chem. 1992, 267, 1375–1381. [Google Scholar]
- Yamauchi, K.; Moriya, A.; Kinoshita, M. Peculiar membrane morphologies of archaebacterial lipid models: 1,1%-polymethylene bis (2-alkyl-sn-glycero-3-phosphocholine). Biochim. Biophys. Acta 1989, 1003, 151–160. [Google Scholar] [CrossRef]
- Bakowsky, U.; Rothe, U.; Antonopoulos, E.; Martini, T.; Henkel, L.; Freisleben, H.-J. Monomolecular organization of the main tetraether lipid from Thermoplasma acidophilum at the water-air interface. Chem. Phys. Lipids 2000, 105, 31–42. [Google Scholar] [CrossRef]
- Freisleben, H.-J. Tetraether Lipid Liposomes. In Membrane Structure in Disease and Drug Therapy; Zimmer, G., Ed.; Marcel Dekker, Inc.: New York, NY, USA, 2000; pp. 127–152. [Google Scholar]
- Freisleben, H.-J. Archaeosomes and Tetraether Lipid Liposomes. Maj. Ilmu Kefarmasian Indones. 2012, 9, 53–65. [Google Scholar]
- Rudolph, P.; Wiesner, H.; Engelhardt, M.; Freisleben, H.-J. Liposomes of the main phospholipid (MPL) from the archaebacterium Thermoplasma acidophilum—Size and stability. Biol. Chem. Hoppe Seyler 1993, 374, 145. [Google Scholar]
- Gulik, A.; Luzzati, V.; De Rosa, M.; Gambacorta, A. Structure and Polymorphism of Bipolar Isopranyl Ether Lipids from Archaebacteria. J. Mol. Biol. 1985, 182, 131–149. [Google Scholar] [CrossRef]
- Gulik, A.; Luzzati, V.; De Rosa, M.; Gambacorta, A. Structure and Polymorphism of Tetraether Lipids from Sulfolobus solfataricus. I. Crystallographic Analysis. Syst. Appl. Microbiol. 1986, 7, 258–261. [Google Scholar] [CrossRef]
- Gliozzi, A.; Paoli, G.; Pisani, D.; Gliozzi, F.; De Rosa, M.; Gambacorta, A. Phase transitions of bipolar lipids of thermophilic archaebacteria. Biochim. Biophys. Acta 1986, 861, 420–428. [Google Scholar] [CrossRef]
- Sternberg, B.; Rudolph, P.; Freisleben, H.-J. Morphology of Liposomes Made of Bipolar and Membrane-Spanning Lipids from Thermoplasma acidophilum; Liposome Workshop: Leyden, The Netherlands, 1992. [Google Scholar]
- New, R.C.C. (Ed.) Liposomes, a Practical Approach; IRL Press: Oxford, UK, 1990. [Google Scholar]
- Bangham, A.D.; Standish, M.M.; Watkins, J.C. Diffusion of univalent ions across the lamellae of swollen phospholipids. J. Mol. Biol. 1965, 13, 238–252. [Google Scholar] [CrossRef]
- Huang, C. Studies of phosphatidylcholine vesicles formation and physical characteristics. Biochemistry 1969, 8, 344–352. [Google Scholar] [CrossRef]
- Weder, H.-G.; Zumbühl, O. The preparation of variably sized homogenous liposomes for laboratory, clinical, and industrial use by controlled detergent dialysis. In Liposome Technology; Gregoriadis, G., Ed.; CRC Press: Boca Raton, FL, USA, 1984; pp. 79–107. [Google Scholar]
- Barenholtz, Y.; Amselem, S.; Lichtenberg, D. A new method for preparation of phospholipid vesicles (liposomes)—French press. FEBS Lett. 1979, 99, 210–214. [Google Scholar] [CrossRef]
- Olsen, F.; Heath, C.A.; Szoka, F.C.; Vail, W.; Mayhew, E.; Papahadjopoulos, D. Preparation of unilamellar liposomes of intermediate size (0.1–0.2 µM) by a combination of reverse phase evaporation and extrusion through polycarbonate membranes. Biochim. Biophys. Acta 1980, 601, 559–571. [Google Scholar]
- Schivelbusch, O. Der Einfluss von Gallensäuren auf Größe und Membranfluidität von Eilecithin-Liposomen und Liposomen aus dem Hauptphospholipid von Thermoplasma acidophilum—Eine ESR-spektroskopische Studie. [The Influence of Bile Acids on the Size and Membrane Fluidity of Liposomes from Egg Lecithin and from the Main Phospholipid of Thermoplasma acidophilum—An ESR-Spectroscopic Study]. Ph.D. Thesis, Goethe-Universität, Frankfurt am Main, Germany, 1996. [Google Scholar]
- Wiesner, H.M. Herstellung stabiler Liposomen definierter Größe unter 600 nm aus dem Hauptphospholipid von Thermoplasma acidophilum unter Verwendung von Eilecithin. [Preparation of Stable Liposomes of Defined Size under 600 nm from the Main Phospholipid of Thermoplasma acidophilum in Combination with Egg Lecithin]. Ph.D. Thesis, Goethe-Universität, Frankfurt am Main, Germany, 1995. [Google Scholar]
- Ring, K.; Henkel, B.; Valenteijn, A.; Gutermann, R. Studies on the permeability and stability of liposomes derived from a membrane-spanning bipolar archaebacterial lipid. In Liposomes as Drug Carriers; Schmidt, K.H., Ed.; G. Thieme-Verlag: Stuttgart, Germany, 1986; pp. 100–123. [Google Scholar]
- Oertl, A.; Antonopoulos, E.; Freisleben, S.U.; Freisleben, H.-J. Stable archaeal tetraether lipid liposomes for photodynamic application: Transfer of carboxyfluorescein to cultured T84 tumor cells. Med. J. Indones. 2016, 25, 196–206. [Google Scholar] [CrossRef]
- Freisleben, H.-J.; Zwicker, K.; Jezek, P.; John, G.; Bettin-Bogutzki, A.; Ring, K.; Nawroth, T. Reconstitution of bacteriorhodopsin and ATP synthase from Micrococcus luteus into liposomes of the purified main tetraether lipid from Thermoplasma acidophilum: Proton conductance and light-driven ATP synthesis. Chem. Phys. Lipids 1995, 78, 137–147. [Google Scholar] [CrossRef]
- Elferink, M.G.L.; de Wit, J.G.; Driessen, A.J.M.; Konings, W.N. Stability and proton-permeability of liposomes composed of archaeal tetraetherlipids. Biochim. Biophys. Acta 1994, 1193, 247–254. [Google Scholar] [CrossRef]
- Komatsu, H.; Chong, P.L.-G. Low Permeability of Liposomal Membranes Composed of Bipolar Tetraether Lipids from Thermoacidophilic Archaebacterium Sulfolobus acidocaldarius. Biochemistry 1998, 37, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Bettin-Bogutzki, A. Einlagerung von Proteinen in Liposomen aus atypischen Lipiden. [Incorporation of Proteins into Liposomes of Atypical Lipids]. Master’s Thesis, Goethe-Universität, Frankfurt am Main, Germany, 1985. [Google Scholar]
- Hellingwerf, K.J.; Scholte, B.J.; van Dam, K. BR-vesicles, an outline of the requirement for light-dependent H+-pumping. Biochim. Biophys. Acta 1978, 513, 66–77. [Google Scholar] [CrossRef]
- Nawroth, T.; Conrad, H.; Vienken, J.; Dose, K. Neutron small angle scattering of matched proteoliposomes with incorporated F0F1 ATPase complex from Rhodosperillium rubrum FR 1. Biol. Chem. Hoppe Seyler 1983, 364, 923–931. [Google Scholar] [CrossRef]
- Wagner, N.; Gutweiler, M.; Pabst, R.; Dose, K. Coreconstitution of bacterial ATP-synthase with monomeric bacteriorhodopsin into liposomes. A comparison between the efficiency of monomeric bacteriorhodopsin and purple membrane patches in coreconstitution experiments. Eur. J. Biochem. 1987, 165, 177–183. [Google Scholar] [CrossRef]
- Nawroth, T.; Zwicker, K.; Eul, U.; Dose, K. Isolation of ATP—Synthase from Micrococcus luteus by FPLC and co-reconstitution in preformed liposomes with bacteriorhodopsin. Biol. Chem. Hoppe Seyler 1990, 371, 806–807. [Google Scholar]
- Zwicker, K. Isolation der ATP-Synthase aus Micrococcus luteus und funktionelle Rekonstitution in Modellmembranen. [Isolation of ATP-Synthase from Micrococcus luteus and Functional Reconstitution into Model Membranes]. Ph.D. Thesis, J. Gutenberg-Universität, Mainz, Germany, 1992. [Google Scholar]
- Patel, G.B.; Sprott, G.D. Archaeobacterial ether lipid liposomes (archaeosomes) as novel vaccine and drug delivery. Crit. Rev. Biotechnol. 1999, 19, 317–357. [Google Scholar] [CrossRef]
- Balakireva, L.A.; Balakirev, M.Y. Transfection of Eukaryotic Cells with Bipolar Cationic Derivatives of Tetraether Lipid. In Membrane Structure in Disease and Drug Therapy; Zimmer, G., Ed.; Marcel Dekker, Inc.: New York, NY, USA, 2000; pp. 153–166. [Google Scholar]
- Deisinger, B.; Nawroth, T.; Zwicker, K.; Matuschka, S.; John, G.; Zimmer, G.; Freisleben, H.-J. Purification of ATP synthase from beef heart mitochondria (F0F1) and co-reconstitution with monomeric bacteriorhodopsin into liposomes capable of light-driven ATP synthesis. Eur. J. Biochem. 1993, 218, 377–383. [Google Scholar] [CrossRef]
- Matuschka, S.; Zwicker, K.; Nawroth, T.; Zimmer, G. ATP synthesis by purified ATP-synthase from beef heart mitochondria after co-reconstitution with bacteriorhodopsin. Arch. Biochem. Biophys. 1995, 322, 135–142. [Google Scholar] [CrossRef]
- Berhanu, S.; Ueda, T.; Kuruma, Y. Artificial photosynthetic cell producing energy for protein synthesis. Nat. Commun. 2019, 10, 1325. [Google Scholar] [CrossRef]
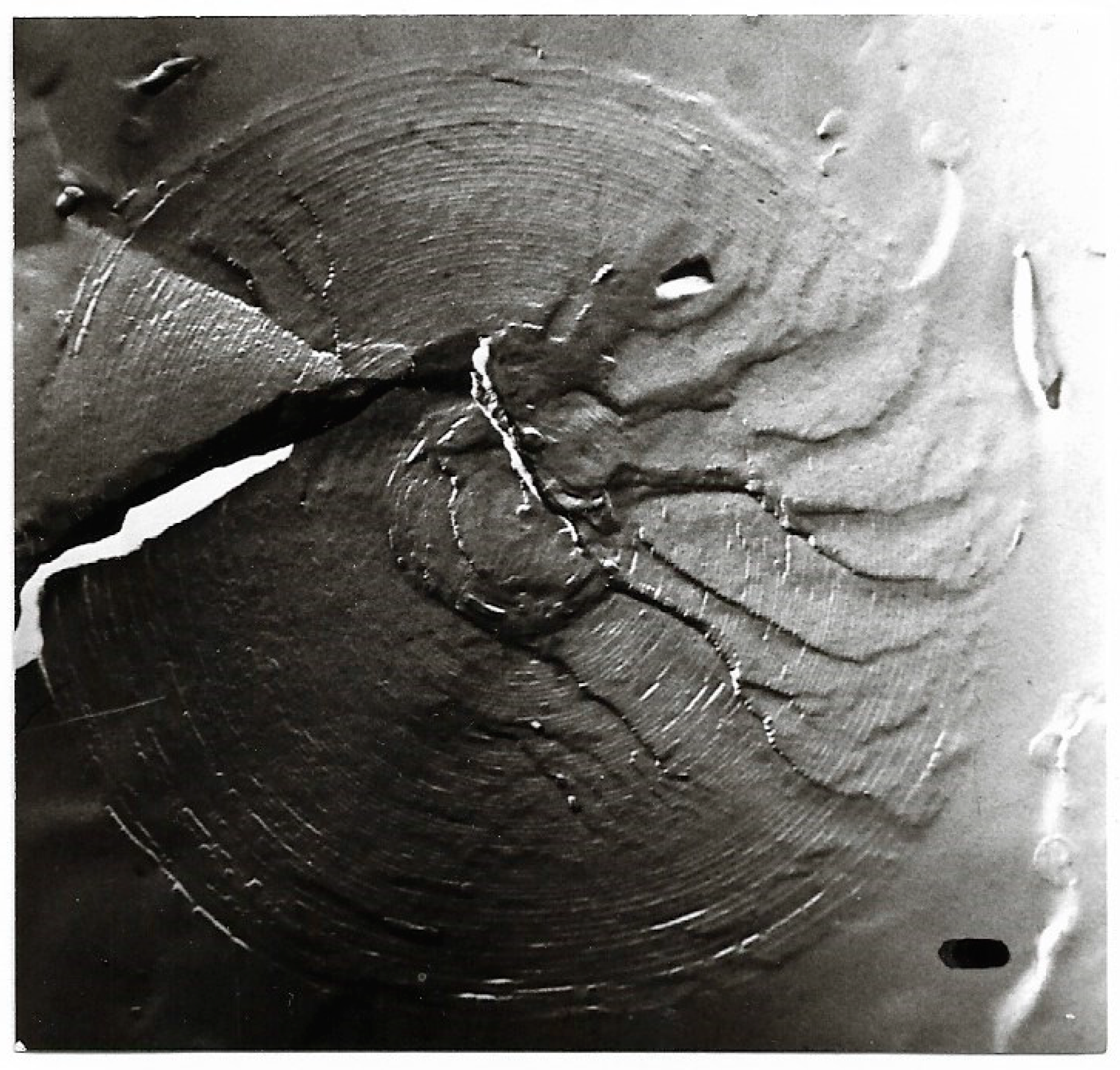
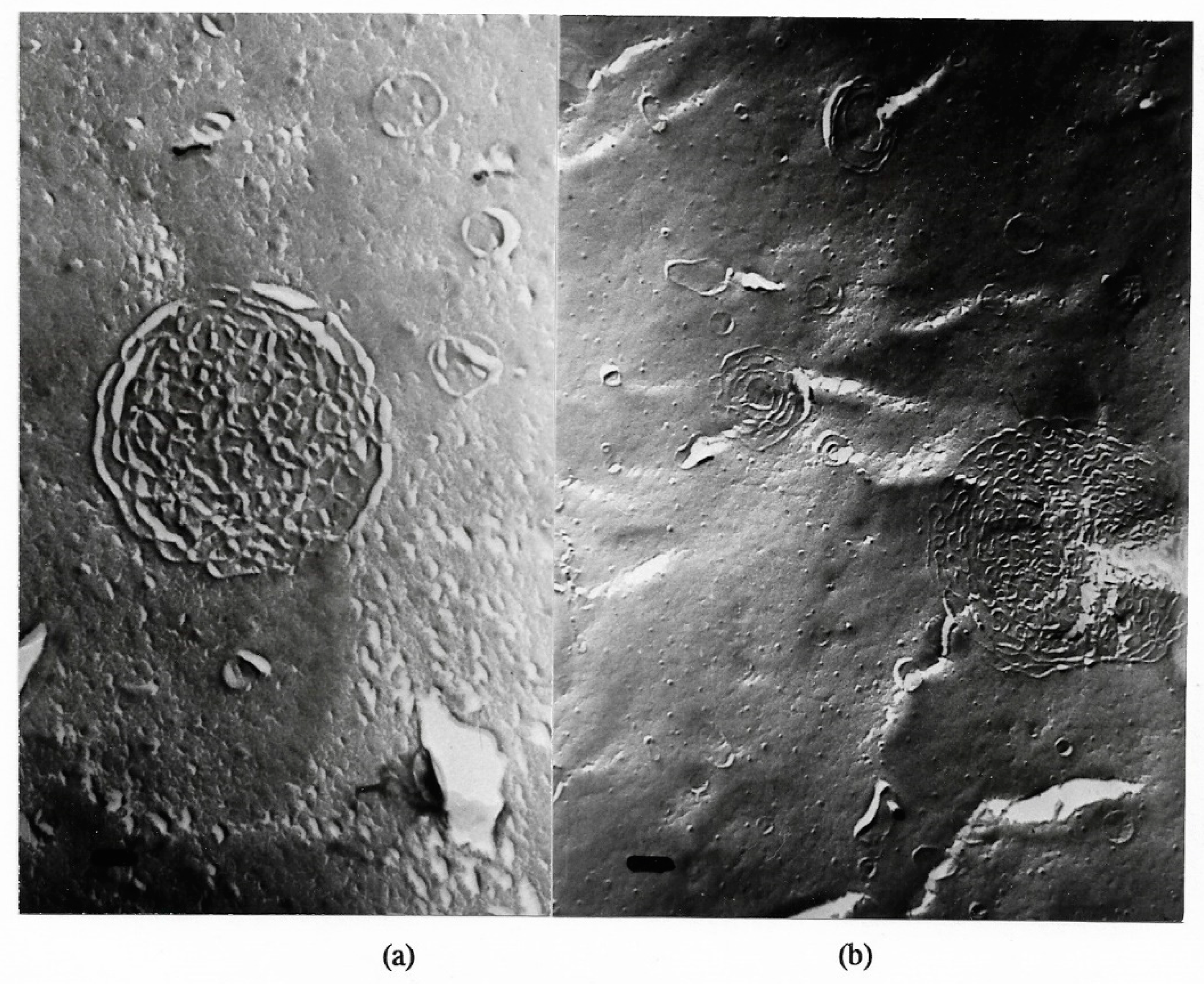

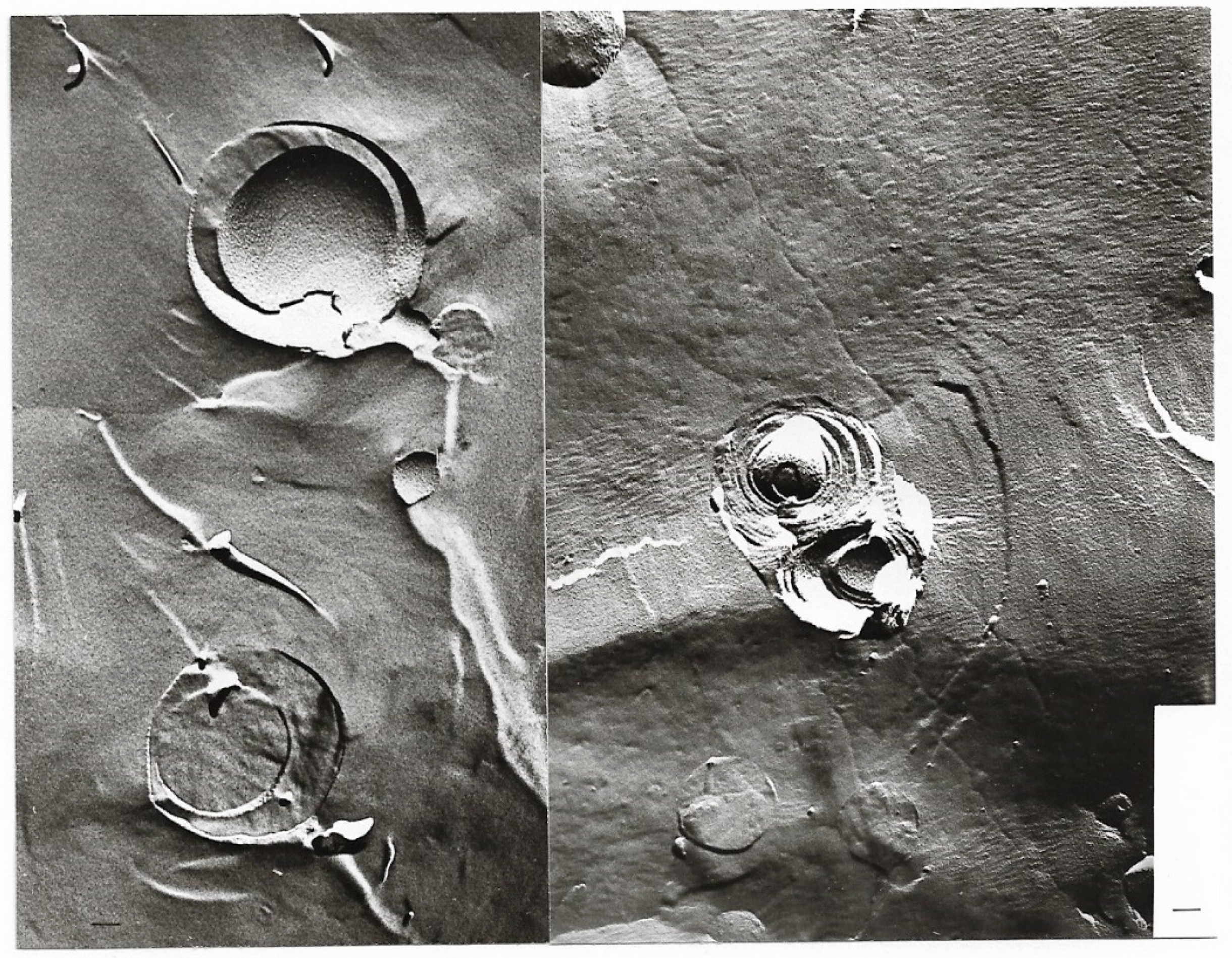
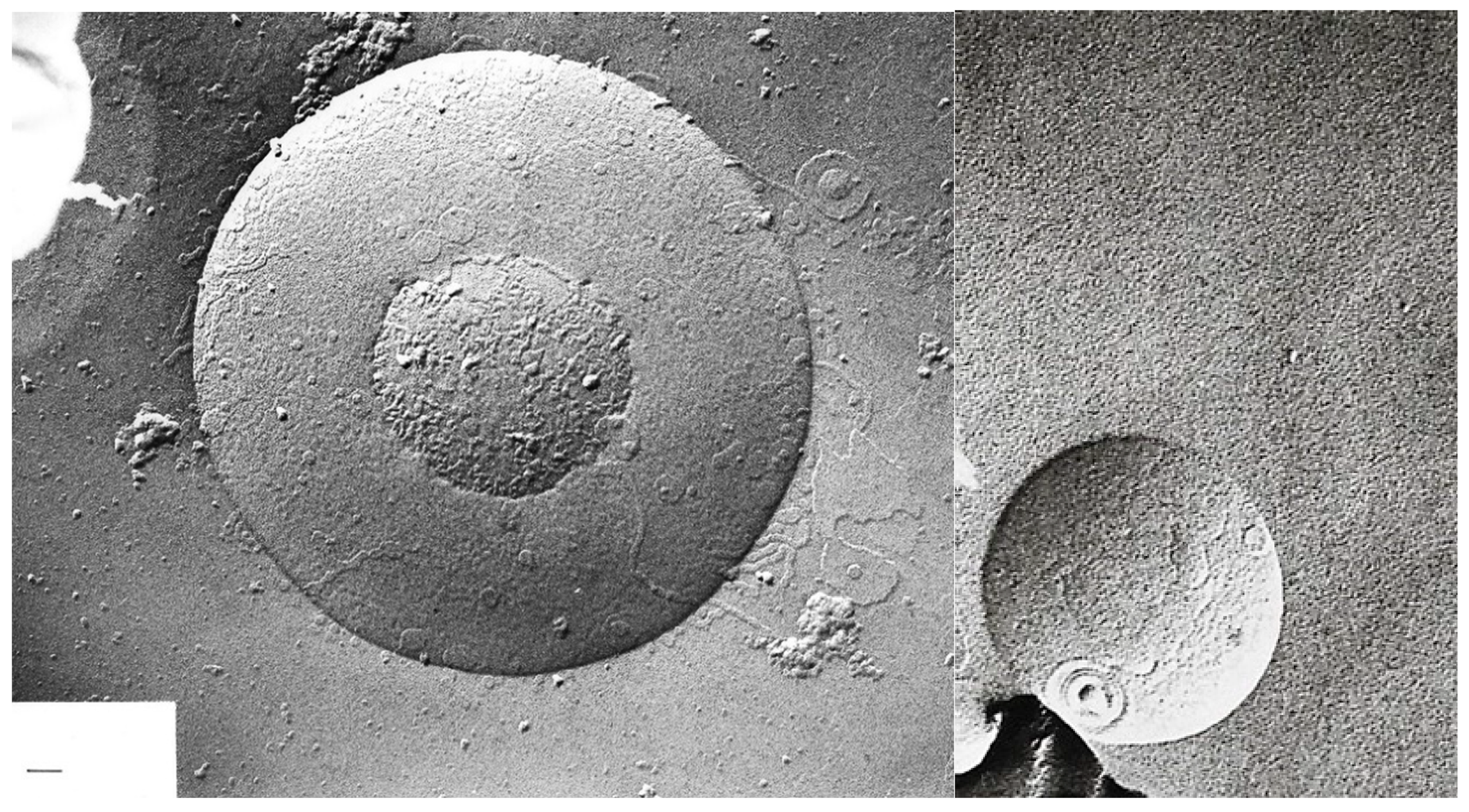

| pH | 1.2 | 1.5 | 1.8 | 2.4 | 3.0 |
|---|---|---|---|---|---|
| Growth Temperature | |||||
| 39 °C | 2.8 [22,24] | ||||
| 40 °C | 3.2* [20] | ||||
| 45 °C | 3.6 [21] | ||||
| 50 °C | 3.8* [20]; 3.9 [21] | ||||
| 55 °C | 4.0 [21] | 2.8 [18]; 4.1 [21] | 4.8 [21] | 5.1 [21] | |
| 59 °C | 3.8 [22,24] | ||||
| 60 °C | 4.2* [20]; 4.5 [21] |
| Growth at 59 °C; Lipid Fraction From T.a. | Glass Transition Range [°C] | Metastable Phase Transition Range [°C] | Main Phase Transition Range [°C] | Tm [°C] | ΔH [J/g] |
|---|---|---|---|---|---|
| Total lipid | −95/−50 | −40/−10 | −16.5 | 5.0 | |
| Phospholipid | −95/−50 | −34/−8 | −14.3 | 8.9 | |
| MPL | −95/−50 | −32/−7 | −14.0 | 8.9 | |
| Glycolipid | −95/−65 | −65/−30 | −30/−13 | −17.7 | 9.0 |
| MPL | 39 °C | 49 °C | 59 °C | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Scan | dT/dt [K/s] phase | °C | endo-/exo-therm | dT/dt [K/s); phase | °C | endo-/exo-therm | dT/dt [K/s]; phase | °C | endo-/exo- therm |
| heating | 0.02–0.13 | −30 → +30 | endo-exo-endo | 0.017–0.083 | −30 → +20 | endo-exo-endo | |||
| Tm | A→C | −15 | endo | A→C | −15 | endo | A→C | −15 | endo |
| A/C→B’ | 0 | exo | A/C→B’ | ~ 0 | small exo | ||||
| B’→C | 20 | endo | B’→C | 20 | small endo | +17 | endo | ||
| ΔH | depends on dT/dt | depends on dT/dt | widely indep-endent | ||||||
| cooling | 0.08 C→A | −20 → −45 | exo | 0.033 C→A | −30 → −50 | exo | 0.033 C→A | exo | |
| Tm | −27 | exo | |||||||
| Phase Transition | From | to | Mixture | ||||
|---|---|---|---|---|---|---|---|
| Tm [°C] range | J/mol | kJ/mol K | |||||
| MPL | Glass transition | −90/−50 | 1.26 | Rigid glass | Gel analogue | ||
| MPL | Main transition | −30 | −14 | Gel analogue | Liquid crystalline | ||
| DPhGG | Glass transition | −84/−61 | 0.74 | Rigid glass | Liquid crystalline | Unlimited, no phase separation | |
| DPGG | sharp | 62 | −98 | Limited; only >62 °C; <62 °C phase separation with DPGG-rich and DPGG-poor domains | |||
| DPPG | Transition and mixture behavior similar to DPGG | ||||||
| DPPC | weak pretr, sharp ph.tr. | 35/ 42 | Limited, phase separation with DPPC-rich (ph.tr. >0 °C) and DPPC-poor (ph.tr. at −13 °C) domains | ||||
| Shift of Tm [°K] | MPL/Ionophore Ratio at ΔH = 0 | Times 2# | DPPC/Ionophore Ratio at ΔH = 0 | ||
|---|---|---|---|---|---|
| MPL | DPPC/DHMG* | ||||
| Alamethicin | −4.7 | −4.2 | 0.5 | 1.0 | 1.5 |
| Melittin | −15.8 | −10 to −20 | 2–2.5 | 4–5 | 6–8 |
| Valinomycin | >0 | −0.6 | 0.5 | 1.0 | 0 |
| Nonactin | −3.4 | −6.7/−2.7* | ~0.3 | ~0.6 | ΔH increases |
| Lipid | Solvent | Cm [µF/cm2] | td [nm] | Stability | Conductance [µS/cm2] | ||
|---|---|---|---|---|---|---|---|
| Valinomycin 10−7 M | Nonactin 10−6 M | Gramicidin 5 × 10−11 M | |||||
| MPL | n-decane or squalene | 0.744 | 2.5 (3.0) | + | 200–250 | 300 | 60 |
| MGL | 2.5 (3.0) | +/(+)* | 700 (1000)* | 110 | |||
| GDNT | ~0.7 | 2.5–3.0 | + | 1200 | n.d. | 130 | |
| DPhGG | n-decane | 0.412 | ~4.5 | (+) | 125 | ||
| squalene | ~0.7 | 2.6–3.0 | − | 900 | |||
| DPhPC | n-decane | 0.412 | 4.72 5.68 | + | 185 85 | 50 | 40/double conc. 100 |
| squalene | ~0.7 | ~3.0 | (+) | 480 | 1100 | ||
| Preparation | Mean Diameter [nm] ± SD | Dispersity Index 0-1 | Reference [62] |
|---|---|---|---|
| Hand-shaken | 2500 to > 7500 | 0.9 | [63] |
| Sonication | 600 ± 40 | 0.5 | [64] |
| Detergent dialysis | 370 ± 35 | 0.5 | [65] |
| French pressure cell | 151 ± 22 | 0.4 | [66] |
| Polycarbonate filter extrusion 200 nm | 221 ± 63 | 0.6 | [67] |
| Polycarbonate filter extrusion 100 nm | 120 ± 40 |
| Spin Probe | Lecithin Liposomes | MPL Liposomes |
|---|---|---|
| 3-Doxyl-5α-cholestane | 0.655 | 0.932 |
| 5-Doxylstearic acid | 0.644 | 0.781 |
| 16-Doxylstearic acid | 0.201 | 0.258 |
| 5-Doxyldecane | 0.130 | 0.187 |
| di-tert-Butylnitroxide | 0.117 | 0.091 |
| Liposomes | Egg Lecithin | MPL | ||||
|---|---|---|---|---|---|---|
| Original | Bile Acid Salts | Original | Bile Acid Salts | |||
| 10 mM | 30 mM | 10 mM | 30 mM | |||
| size | 159.8 nm | 47.2 nm | 1898.4 nm | 131.5 nm | 107.9 nm | 114.8 nm |
| Lipid/Detergent Ratio | MPL | MPL/EL 75:25 (% g/g) | MPL/EL 50:50 (% g/g) | MPL/EL 25:75 (% g/g) | Egg Lecithin |
|---|---|---|---|---|---|
| 0.05 | 372 ± 98 | 558 | − | 490 | − |
| 0.1 | 446 (396–495) | 396 (199–550) | 728 (359–778) | 484 (422–645) | − |
| 0.15 | n.d. | 263 (169–307) | 431 (386–664) | 471 (453–489) | − |
| 0.2 | 444 (421–466) | 461 (480–792) | 818 | 413 (325–510) | 153 ± 5 (145–162) |
| 0.3 | − | − | − | − | 144 ± 9 (137–150) |
| Days | MPL | MPL/EL 75:25 (%g/g) | MPL/EL 50:50 (%g/g) | MPL/EL 25:75 (%g/g) | Egg Lecithin | |
|---|---|---|---|---|---|---|
| 0–2 | 466 | 372 | 408 | 386 | 159 | |
| 5–10 | 450 | 394 | 396 | 146 | ||
| 15–38 | 358 | 695 (day 38) | 395 | 411 | 149 | |
| 49–70 | 446 | − | 366 (day 49) | 405 | 149 (day 66) | |
| 80–90 | − | − | 446 (day 89) | − | ||
| 128 | 434 | − | − | - | − | |
| 746 | 498 | − | − | - | − | |
| Lipid | 4 °C | 30 °C | 50 °C |
|---|---|---|---|
| Egg lecithin | 9% | 41% | 100% |
| DPPC | 6% | 32% | 100% |
| DPPC/MPL molar ratio 2:1 | 5% | 8% | 62% |
| DPPC/MPL molar ratio 1:2 | 3% | 5% | 15% |
| MPL | 2.5% | 3% | 8% |
| Liposomes | MPL | Egg Lecithin | Remarks |
|---|---|---|---|
| + 5µM Val | + 5µM Val | ||
| Valinomycin | 4.5 × 10−6 × cm sec−1 | 10−5-10−4 × cm sec−1 | Proton permeability coefficient |
| + Val | − | + | excess valinomycin |
| + Palm | − | ++ | |
| + FCCP | +++ | +++ | |
| 2.7 × 10−5 nmol H+ sec−1 cm−2 | 1.2 × 10−4 nmol H+ sec−1 cm−2 | Proton permeability |
| Addition | Concentration | Initial Pump Rate | Proton Efflux [ng H+ x min−1] | |
|---|---|---|---|---|
| Calculated per mg MPL | Calculated per mg BR | |||
| nil | 1.4 | 0.6 | 0.1 | |
| valinomycin | 0.24 µM | 41.6 | 1.2 | 0.15 |
| Val + gramicidin | 0.24 µM + 1 µM | 15.8 | 0.05 | 0.008 |
| Val + gramicidin | 0.24 µM + 5 µM | 6.2 | 0.02 | 0.003 |
| Val + FCCP | 0.24 µM + 1 µM | 15.5 | 0.21 | 0.035 |
| Val + FCCP | 0.24 µM + 6 µM | 11.6 | 0.58 | 0.1 |
| Val + FCCP | 0.24 µM + 15 µM | 6.3 | 0.58 | 0.1 |
| Lipid | Liposomes Size [nm] | nmol ATP (mg × min)−1 | Detergent/Method | Reference |
|---|---|---|---|---|
| MPL | 132 | 13.4 | Octylglucoside det-dialys/ French press | [65,66,72] |
| SBL | 52 | 77.1 | Taurodeoxycholate (TDOC) | [77,78,79,80] |
| DOPC | 86 | 40.5 | ||
| ML | 123 | 2.2 | ||
| DMPC | 120 | 0 |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Freisleben, H.-J. The Main (Glyco) Phospholipid (MPL) of Thermoplasma acidophilum. Int. J. Mol. Sci. 2019, 20, 5217. https://doi.org/10.3390/ijms20205217
Freisleben H-J. The Main (Glyco) Phospholipid (MPL) of Thermoplasma acidophilum. International Journal of Molecular Sciences. 2019; 20(20):5217. https://doi.org/10.3390/ijms20205217
Chicago/Turabian StyleFreisleben, Hans-Joachim. 2019. "The Main (Glyco) Phospholipid (MPL) of Thermoplasma acidophilum" International Journal of Molecular Sciences 20, no. 20: 5217. https://doi.org/10.3390/ijms20205217
APA StyleFreisleben, H.-J. (2019). The Main (Glyco) Phospholipid (MPL) of Thermoplasma acidophilum. International Journal of Molecular Sciences, 20(20), 5217. https://doi.org/10.3390/ijms20205217





