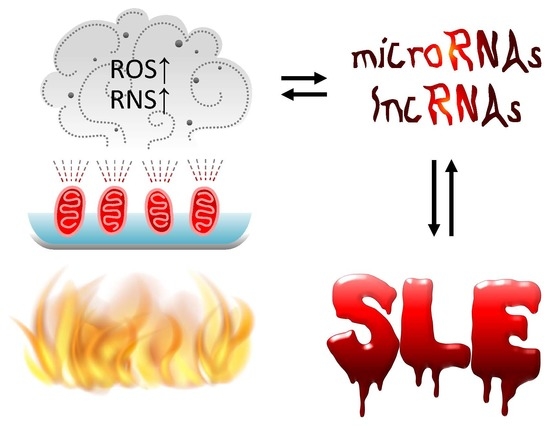
Cross-Talk between Mitochondrial Dysfunction-Provoked Oxidative Stress and Aberrant Noncoding RNA Expression in the Pathogenesis and Pathophysiology of SLE
Abstract
:1. Introduction
2. Epigenetic Regulations of Gene Expression/Silencing in Physiological Conditions
2.1. Abnormal DNA Methylation/Demethylation in SLE
2.2. Abnormal Histone Modification in SLE
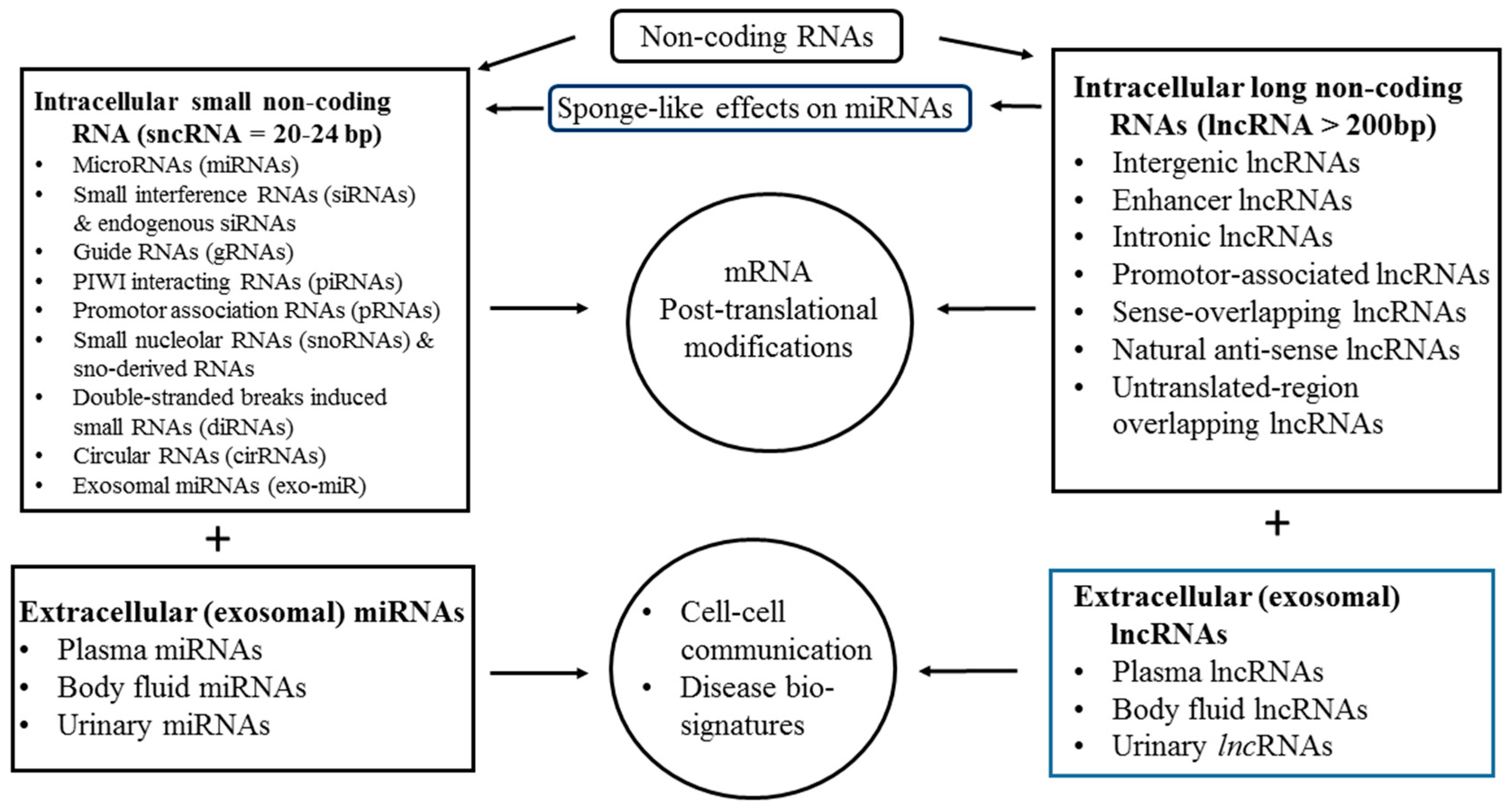
2.3. Physiological Functions of ncRNAs
2.4. Aberrant Intracellular and Extracellular Exosomal ncRNA Expression in Association with Pathological Changes in Patients with SLE
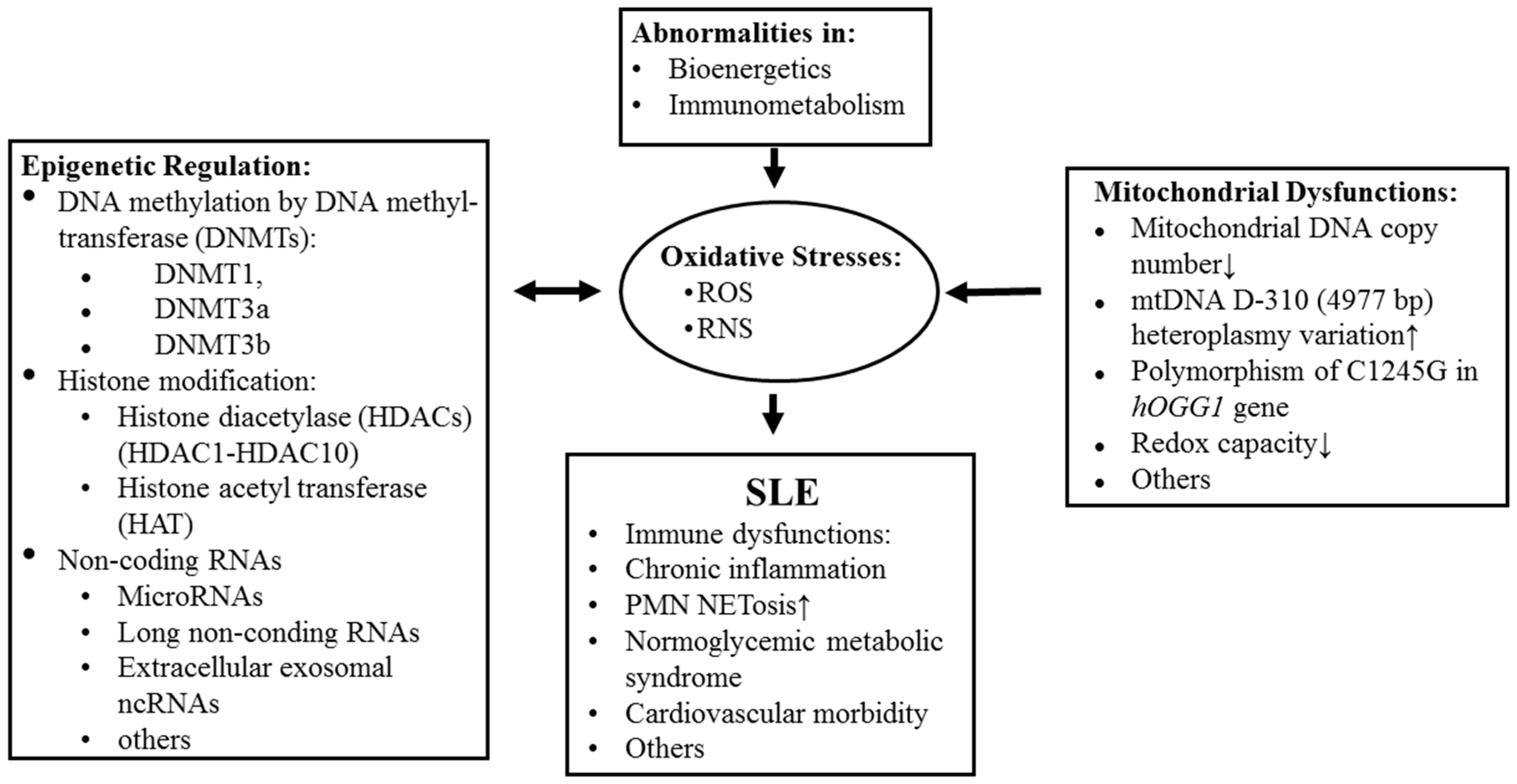
3. Increased Oxidative Stress in Patients with SLE
3.1. Causes of Excessive Oxidative Stress in SLE
3.2. Effects of Excessive Oxidative Stress on the Pathogenesis and Pathophysiology in SLE Patients
4. Cross-Talk Between Oxidative Stress and ncRNAs in Physiological Condition
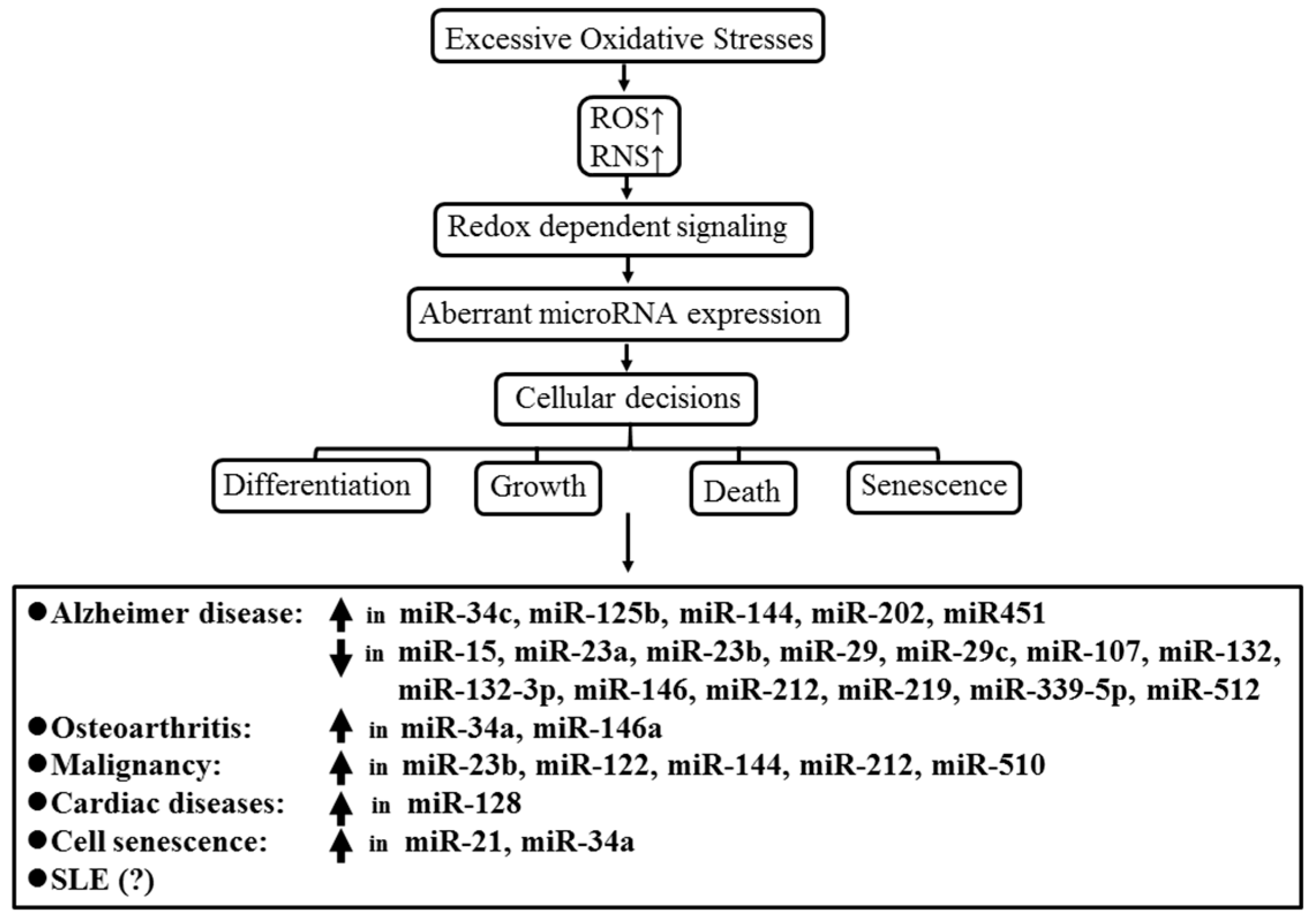
4.1. Excessive Oxidative Stress May Influence ncRNA Expression in Various Diseases
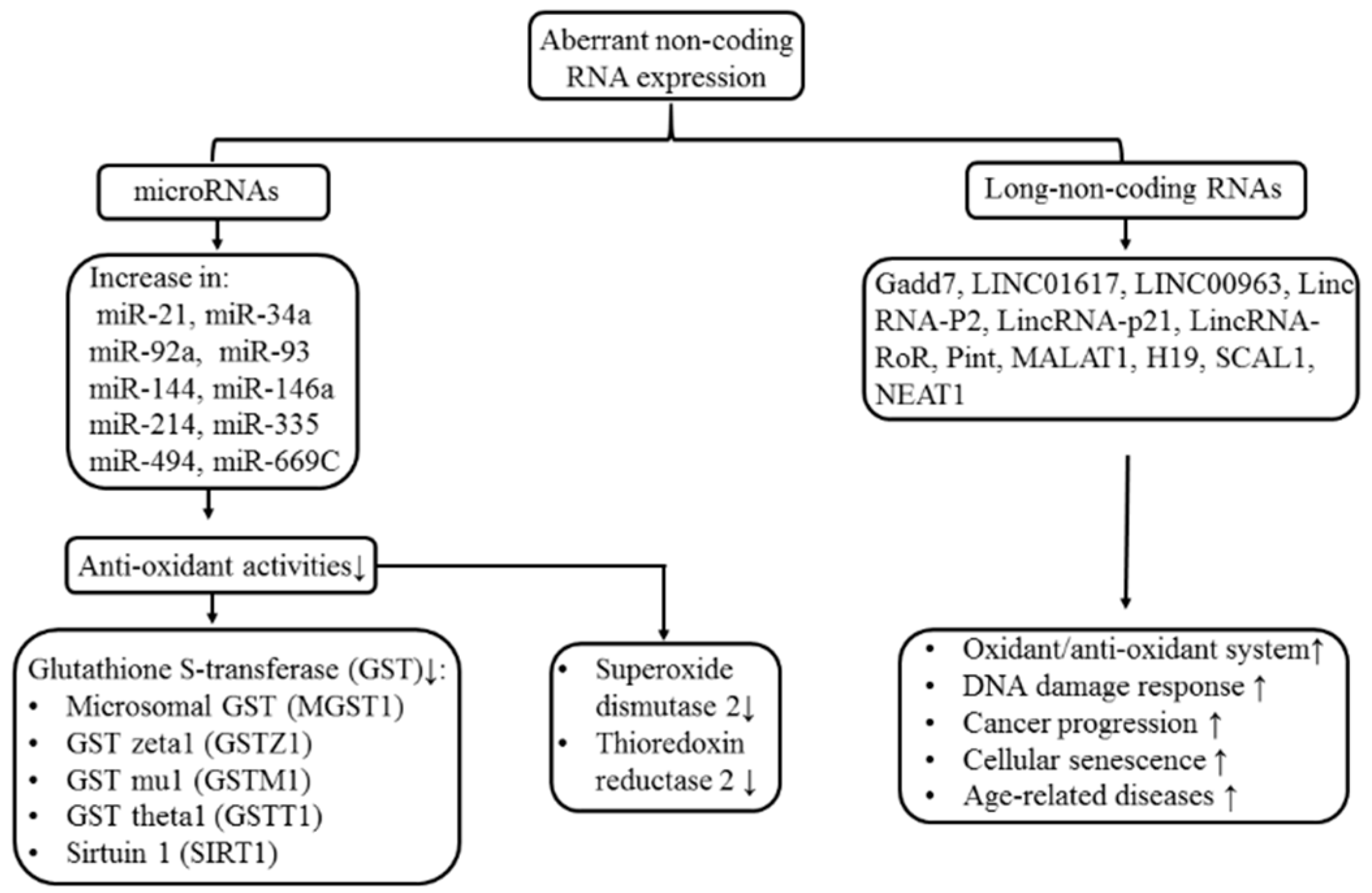
4.2. Aberrant ncRNA Expression Induces Oxidant/Antioxidant Imbalance in Different Pathological Processes
5. Antioxidant Therapy and Manipulation of Epigenetic Expression to Treat Patients with SLE
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| C.V | cardiovascular |
| DNA | deoxyribonucleic acid |
| DNMT | DNA methyltransfersase |
| FcγR | Immunoglobulin G Fragment C-gamma receptor |
| GLUT | glucose transporter |
| GSH | reduced form glutathione |
| GPx | glutathione peroxidase |
| GST | glutathione S-transferase |
| HAT | histone acetyltransferase |
| HDAC | histone deacetylase |
| IFN | interferon |
| IL | interleukin |
| LN | lupus nephritis |
| lncRNA | long noncoding ribonucleic acid |
| MAPK | mitogen-activated protein kinase |
| MHC | major histocompatibility complex |
| miR | microRNA |
| mtDNA | mitochondrial DNA |
| mTOR | mammalian target of rapamycin |
| NAC | N-acetylcysteine |
| ncRNA | non-coding RNA |
| NET | neutrophil extracellular trap |
| Ras | rat sarcoma protein, a superfamily of small GTPase |
| RNS | reactive nitrogen species |
| ROS | reactive oxygen species |
| SIRT1 | sirtuin 1 |
| SLE | systemic lupus erythematosus |
| SLEDAI | SLE disease activity index |
| SLEDAI-2K | SLEDAI in 2000 year |
| SOD | superoxide dismutase |
| TET | ten-eleven translocation DNA dioxygenase |
| Th | helper T cell |
| Treg | regulatory T cell |
References
- Wang, L.; Mohan, C.; Li, Q.Z. Arraying autoantibodies in SLE-lessons learned. Curr. Mol. Med. 2015, 15, 456–461. [Google Scholar] [CrossRef]
- Yaniv, G.; Twig, G.; Shor, D.B.; Furer, A.; Sherer, Y.; Mozes, O.; Kimisar, O.; Slonimsky, E.; Klang, E.; Lotan, E.; et al. A volcano explosion of autoantibodies in systemic lupus erythematosus: A diversity of 180 different antibodies found in SLE patients. Autoimmun. Rev. 2015, 14, 75–79. [Google Scholar] [CrossRef]
- Kahlenberg, J.M.; Kaplan, M.J. The inflammasome and lupus—Another innate immune mechanism contributing to disease pathogenesis? Curr. Opin. Rheumatol. 2014, 26, 475–481. [Google Scholar] [CrossRef]
- Weidenbusch, M.; Kulkarni, O.P.; Anders, H.J. The innate immune system in human systemic lupus erythematosus. Clin. Sci. 2017, 131, 625–634. [Google Scholar] [CrossRef]
- Tsai, C.Y.; Li, K.J.; Hsieh, S.C.; Liao, H.T.; Yu, C.L. What’s wrong with neutrophils in lupus? Clin. Exp. Rheumatol. 2019, 37, 684–693. [Google Scholar] [PubMed]
- Zharkova, O.; Celhar, T.; Crarens, P.D.; Satherthwaite, A.B.; Fairhurst, A.W.; Davis, L.S. Pathways leading to an immunological diseases: Systemic lupus erythematosus. Rheumatology 2017, 56, i55–i66. [Google Scholar] [CrossRef] [PubMed]
- Harley, I.T.W.; Kaufman, K.M.; Langefeld, C.D.; Haraey, J.B.; Kelly, J.A. Genetic susceptibility to SLE: New insights from fine mapping and genome-wide association studies. Nat. Rev. Genet. 2009, 10, 285–290. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Davidson, A. Taming lupus—A new understanding of pathogenesis is leading to clinical advances. Nat. Med. 2012, 18, 870–882. [Google Scholar] [CrossRef]
- Ghodke-Puranick, Y.; Niewold, T.T. Immunogenetics of systemic lupus erythematosus: A comprehensive review. J. Autoimmun. 2015, 64, 125–136. [Google Scholar] [CrossRef] [Green Version]
- Teruel, M.; Alacon-Riguelme, M.E. The genetic basis of systemic lupus erythematosus: What are the risk factors and what have we learned. J. Autoimmun. 2016, 74, 161–175. [Google Scholar] [CrossRef]
- Iwamoto, T.; Niewold, T.B. Genetics of human lupus nephritis. Clin. Immunol. 2017, 185, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Hiraki, L.T.; Silverman, E.D. Genomics of systemic lupus erythematosus: Insights gained by studying monogenic young-onset systemic lupus erythematosus. Rheum. Dis. N. Am. 2017, 43, 415–434. [Google Scholar] [CrossRef] [PubMed]
- Saeed, M. Lupus pathology based on genomics. Immunogenetics 2017, 69, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Goulielmos, G.N.; Zervou, M.I.; Vazgiourakis, V.M.; Ghodke-Puranik, Y.; Garyballos, A.; Niewold, T.B. The genetics and molecular pathogenesis of systemic lupus erythematosus (SLE) in populations of different ancestry. Gene 2018, 668, 59–72. [Google Scholar] [CrossRef] [PubMed]
- Javinani, A.; Ashraf-Ganjouei, A.; Asloni, S.; Janshidi, A.; Mahmoudi, M. Exploring the etiopathogenesis of systemic lupus erythematosus: A genetic perspective. Immunogenetics 2019, 71, 283–297. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Zhao, M.; Chang, C.; Lu, Q. The real culprit in systemic lupus erythematosus: Abnormal epigenetic regulation. Int. J. Mol. Sci. 2015, 16, 11013–11033. [Google Scholar] [CrossRef] [PubMed]
- Miceli-Richard, C. Epigenetics and lupus. Jt. Bone Spine 2015, 82, 90–93. [Google Scholar] [CrossRef]
- Hedrick, C.M.; Mabert, K.; Rouen, T.; Tsokos, G.C. DNA methylation in systemic lupus erythematosus. Epigenomics 2017, 9, 505–525. [Google Scholar] [CrossRef]
- Zhan, Y.; Guo, Y.; Lu, Q. Aberrant epigenetic regulation in the pathogenesis of systemic lupus erythematosus and its implications in precision medicine. Cytogent. Genome Res. 2016, 149, 141–155. [Google Scholar] [CrossRef]
- Wang, Z.; Chang, C.; Peng, M.; Lu, Q. Translating epigenetics into clinic: Focus on lupus. Clin. Epigenet. 2017, 9, 78. [Google Scholar] [CrossRef]
- Ren, J.; Panther, E.; Liao, X.; Grammer, A.C.; Lipsky, P.E.; Reilly, C.M. The impact of protein acetylation/deacetylation on systemic lupus erythematosus. Int. J. Mol. Sci. 2018, 19, 4007. [Google Scholar] [CrossRef] [PubMed]
- Long, H.; Yin, H.; Wang, L.; Gershwin, M.E.; Lu, Q. The critical role of epigenetics in systemic lupus erythematosus and autoimmunity. J. Autoimmun. 2016, 74, 118–138. [Google Scholar] [CrossRef] [PubMed]
- Lai, N.-S.; Koo, M.; Yu, C.-L.; Lu, M.C. Immunopathogenesis of systemic lupus erythematosus and rheumatoid arthritis: The role of aberrant expression of non-coding RNAs in T cells. Clin. Exp. Immunol. 2017, 187, 327–336. [Google Scholar] [CrossRef] [PubMed]
- Zununi Vahed, S.; Nakhjarvani, M.; Etemadi, J.; Jamashidi, N.; Pourlak, T.; Abedizar, S. Altered levels of immune-regulatory microRNAs in plasma samples of patients with lupus nephritis. Bioimpacts 2018, 8, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Honarpisheh, M.; Kohler, P.; von Rauchhaupt, E.; Lech, M. The involvement of microRNAs in modulation of innate and adaptive immunity in systemic lupus erythematosus and lupus nephritis. J. Immunol. Res. 2018, 4126106. [Google Scholar] [CrossRef] [PubMed]
- McMurray, R.W. Sex-hormones in the pathogenesis in systemic lupus erythematosus. Front Biosci. 2001, 6, E193–E206. [Google Scholar] [CrossRef] [PubMed]
- Khan, D.; Dai, R.; Ahmed, S.A. Sex differences and estrogen regulation of miRNA in lupus, a prototypical autoimmune disease. Cell Immunol. 2015, 294, 70–79. [Google Scholar] [CrossRef]
- Assad, S.; Khan, H.H.; Ghazanfar, H.; Khan, Z.-H.; Mansor, S.; Rahman, M.A.; Khan, G.H.; Zafar, B.; Tariz, U.; Malik, S.A. Role of sex-hormone levels and psychological stress in the pathogenesis of autoimmune diseases. Cureus 2017, 9, E1315. [Google Scholar] [CrossRef]
- Christou, E.A.A.; Banos, A.; Kosmara, D.; Bertsias, G.K.; Boumpas, D.T. Sexual dimorphism in SLE; above and beyond sex hormones. Lupus 2019, 28, 3–10. [Google Scholar] [CrossRef]
- Sari-Puttini, P.; Atzeni, F.; Laccarino, L.; Doria, A. Environment and systemic lupus erythematosus: An overview. Autoimmunity 2005, 38, 465–472. [Google Scholar] [CrossRef]
- Parks, C.G.; de Souza Espinodola Santos, A.; Barbhaiya, M.; Costenbader, K.H. Understanding the role of environmental factors in the development of systemic lupus erythematosus. Best Pract. Res. Clin. Rheumatol. 2017, 31, 306–320. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.C. Lupus erythematosus and nutrition: A review of the literature. J. Ren. Nutr. 2000, 10, 170–183. [Google Scholar] [CrossRef] [PubMed]
- Minami, Y.; Sasaki, T.; Arai, Y.; Kurisu, Y.; Hisamichi, S. Diet and systemic lupus erythematosus: A 4 year prospective study of Japanese patients. J. Rheumatol. 2003, 30, 747–754. [Google Scholar] [PubMed]
- Hsieh, C.-C.; Lin, B.-F. Dietary factors regulate cytokines in murine models of systemic lupus erythematosus. Autoimmun. Rev. 2011, 11, 22–27. [Google Scholar] [CrossRef]
- Klack, K.; Bonfa, E.; Borba Neto, E.F. Diet and nutritional aspects in systemic lupus erythematosus. Rev. Bras. Rheumatol. 2012, 52, 384–408. [Google Scholar]
- Lee, H.-T.; Lin, C.-S.; Chen, W.-S.; Liao, H.-T.; Tsai, C.-Y.; Wei, Y.-H. Leukocyte mitochondrial DNA alteration in systemic lupus erythematosus and its relevance to the susceptibility to lupus nephritis. Int. J. Mol. Sci. 2012, 13, 8853–8868. [Google Scholar] [CrossRef]
- Lee, H.-T.; Wu, T.-H.; Lin, C.-S.; Lee, C.-S.; Wei, Y.-H.; Tsai, C.-Y.; Chang, D.-M. The pathogenesis of systemic lupus erythematosus—From the viewpoint of oxidative stress and mitochondrial dysfunction. Mitochondrion 2016, 30, 1–7. [Google Scholar] [CrossRef]
- Lee, H.-T.; Wu, T.-H.; Lin, C.-S.; Lee, C.-S.; Pan, S.-C.; Chang, D.-M.; Wei, Y.-H.; Tsai, C.-Y. Oxidative DNA and mitochondrial DNA change in patients with SLE. Front Biosci. Landmark. 2017, 22, 493–503. [Google Scholar] [Green Version]
- Lee, H.-T.; Lin, C.-S.; Pan, S.-C.; Wu, T.-H.; Lee, C.-S.; Chang, D.-M.; Tsai, C.-Y.; Wei, Y.-H. Alterations of oxygen consumption and extracellular acidification rates by glutamine in PBMCs of SLE patients. Mitochondrion 2019, 44, 65–74. [Google Scholar] [CrossRef]
- Marion, T.N.; Postlethwaite, A.E. Chance, genetics, and the heterogeneity of disease and pathogenesis in systemic lupus erythematosus. Sem. Immunopathol. 2014, 36, 495–517. [Google Scholar] [CrossRef]
- Leishangthem, B.D.; Sharma, A.; Bhatnagar, A. Role of altered mitochondria functions in the pathogenesis of systemic lupus erythematosus. Lupus 2016, 25, 272–281. [Google Scholar] [CrossRef] [PubMed]
- Kuren, B.T.; Scofield, R.H. Lipid peroxidation in systemic lupus erythematosus. Indian J. Exp. Biol. 2006, 44, 349–356. [Google Scholar]
- Das, U.N. Oxidative, anti-oxidants, essential fatty acids, eicosanoids, cytokines, gene/oncogene expression and apoptosis in systemic lupus erythematosus. J. Assoc. Physicians India 1998, 46, 630–634. [Google Scholar]
- Dandekar, A.; Mendez, R.; Zhang, K. Cross talk between ER stress, oxidative stress, and inflammation in health and disease. Methods Mol. Biol. 2015, 1292, 205–214. [Google Scholar]
- Hussain, T.; Tan, B.; Yin, Y.; Blachier, F.; Tossou, M.C.; Rahu, N. Oxidative stress and inflammation: What polyphenols can do for us? Oxidative Med. Cell Longev. 2016, 2016, 7432797. [Google Scholar] [CrossRef]
- Guzik, T.J.; Touyz, R.M. Oxidative stress, inflammation, and vascular aging in hypertension. Hypertension 2017, 70, 660–667. [Google Scholar] [CrossRef]
- Plotnikov, E.; Losenkov, I.; Epimakhova, E.; Bohan, N. Protective effects of pyruvic acid salt against lithium toxicity and oxidative damage in human blood mononuclear cells. Adv. Pharm. Bull. 2019, 9, 302–306. [Google Scholar] [CrossRef]
- Lee, D.; Lee, S.H.; Noh, I.; Oh, E.; Ryu, H.; Ha, J.; Jeong, S.; Yoo, J.; Jeon, T.J.; Yun, C.O.; et al. A helical polypeptide-based potassium ionophore induces endoplasmic reticulum stress-mediated apoptosis by perturbing ion homeostasis. Adv. Sci. (Weinheim) 2019, 6, 1801995. [Google Scholar] [CrossRef]
- Das, U.N. Current and emerging strategies for the treatment and management of systemic lupus erythematosus based on molecular signatures of acute and chronic inflammation. J. Inflamm. Res. 2010, 3, 143–170. [Google Scholar] [CrossRef] [Green Version]
- Perl, A. Oxidative stress in the pathology and treatment of systemic lupus erythematosus. Nat. Rev. Rheumatol. 2013, 9, 674–686. [Google Scholar] [CrossRef] [Green Version]
- Su, Y.J.; Cheng, T.T.; Chen, C.J.; Chiu, W.C.; Chang, W.N.; Tsai, N.W.; Kung, C.T.; Lin, W.C.; Huang, C.C.; Chang, Y.T.; et al. The association among antioxidant enzymes, autoantibodies, and disease severity score in systemic lupus erythematosus: Comparison of neuropsychiatric and nonneuropsychiatric groups. BioMed Res. Int. 2014, 2014, 137231. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, W.; Liu, C.; Yan, M.; Raman, I.; Du, Y.; Fang, X.; Zhou, X.J.; Mohan, C.; Li, Q.Z. Delivering oxidation resistance-1 (OXR1) to mouse kidney by genetic modified mesenchymal stem cells exhibited enhanced protection against nephrotoxic serum induced renal injury and lupus nephritis. J. Stem Cell Res. Ther. 2014, 4, 231. [Google Scholar] [PubMed]
- Jafari, S.M.; Salimi, S.; Nakhaee, A.; Kalani, H.; Tavallaie, S.; Farajian-Mashhadi, F.; Zakeri, Z.; Sandoughi, M. Prooxidant-antioxidant balance in patients with systemic lupus erythematosus and its relationship with clinical and laboratory findings. Autoimmune Dis. 2016, 2016, 4343514. [Google Scholar] [CrossRef] [PubMed]
- Shah, D.; Mahajan, N.; Sah, S.; Nath, S.K.; Paudyal, B. Oxidative stress and its biomarkers in systemic lupus erythematosus. J. Biomed. Sci. 2014, 21, 23. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.-W.; Huang, K.; Yang, C.; Kang, C.-S. Non-coding RNAs as regulators in epigenetics. Oncol. Rep. 2017, 37, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Liu, C. Coding or noncoding, the converging concepts of RNAs. Front. Genet. 2019, 10, 496. [Google Scholar] [CrossRef]
- Banerjee, J.; Khanna, S.; Bhattacharya, A. MicroRNA regulation of oxidative stress. Oxid. Med. Cell Longev. 2017, 2872156. [Google Scholar] [CrossRef]
- Bu, H.; Wedel, S.; Cavinato, M.; Jansen-Dürr, P. MicroRNA regulation of oxidative stress-induced cellular senescence. Oxid. Med. Cell Longev. 2017, 2017, 2398696. [Google Scholar] [CrossRef]
- Wang, X.; Shen, C.; Zhu, J.; Shen, G.; Li, Z.; Dong, J. Long non-coding RNAs in the regulation of oxidative stress. Oxid. Med. Cell Longev. 2019, 1318795. [Google Scholar] [CrossRef]
- Bayoumi, A.S.; Sayed, A.; Broskova, Z.; Teoh, J.-P.; Wilson, J.; Su, H.; Tang, Y.-L.; Kim, I. Crosstalk between long noncoding RNAs and microRNAs in health and disease. Int. J. Mol. Sci. 2016, 17, 356. [Google Scholar] [CrossRef]
- Yamamura, S.; Imai-Sumida, M.; Tanaka, Y.; Dahiya, R. Interaction and cross-talk between non-coding RNAs. Cell Mol. Life Sci. 2018, 75, 467–484. [Google Scholar] [CrossRef] [PubMed]
- Deng, C.; Yang, J.; Scott, J.; Hanash, S.; Richardson, B.C. Role of the ras-MAPK signaling pathway in the DNA methyltransferase response to DNA hypomethylation. Biol. Chem. 1998, 379, 1113–1120. [Google Scholar] [CrossRef] [PubMed]
- Sawalha, A.H.; Jeffries, M.; Webb, R.; Lu, Q.; Gorelik, G.; Ray, D.; Osban, J.; Knowlion, N.; Johnson, K.; Richardsonm, B. Defective T cell ERK signaling induces interferon-regulated gene expression and overexpression of methylation sensitive genes similar to lupus patients. Genes Immun. 2008, 9, 368–378. [Google Scholar] [CrossRef] [PubMed]
- Lu, Q.; Wu, A.; Tesmer, L.; Ray, D.; Yousif, N.; Richardson, B. Demethylation of CD40LG on the inactive X in T cells from women with lupus. J. Immunol. 2007, 179, 6352–6358. [Google Scholar] [CrossRef] [PubMed]
- Hu, N.; Qiu, X.; Luo, Y.; Yuan, J.; Li, Y.; Lei, W.; Zhang, G.; Zhou, Y.; Su, Y.; Lu, Q. Abnormal histone modification patterns in lupus CD4+T cells. J. Rheumatol. 2008, 35, 804–810. [Google Scholar] [PubMed]
- Zhou, Y.; Qiu, X.; Luo, Y.; Yuan, J.; Li, Y.; Zhong, Q.; Zhao, M.; Lu, Q. Histone modifications and methyl-CpG binding domain protein levels at the TNFSF7 (CD70) promoter in SLE CD4+ T cells. Lupus 2011, 20, 1365–1371. [Google Scholar] [CrossRef] [PubMed]
- Hedrich, C.M.; Tsokos, G.C. Epigenetic mechanisms in systemic lupus erythematosus and other autoimmune diseases. Trends Mol. Med. 2011, 17, 714–724. [Google Scholar] [CrossRef] [Green Version]
- Dai, Y.; Zhang, L.; Hu, C.; Zhang, Y. Genome-wide analysis of histone H3 lysine 4 trimethylation by ChIP-chip in peripheral blood mononuclear cells of systemic lupus erythematosus patients. Clin. Exp. Rheumatol. 2010, 28, 158–168. [Google Scholar]
- Zhang, Z.; Song, L.; Maurer, K.; Petri, M.A.; Sullivan, K.E. Global H4 acetylation analysis by ChIP-chip in SLE monocytes. Genes Immun. 2010, 11, 124–133. [Google Scholar] [CrossRef]
- Apostolidis, S.A.; Rauen, T.; Hedrich, C.M.; Tsokos, G.C.; Crispin, J.C. Protein phosphatase 2A enables expression of interleukin 17 (IL-17) through chromatin remodeling. J. Biol. Chem. 2013, 288, 26775–26784. [Google Scholar] [CrossRef]
- Hedrich, C.M.; Raurex, J.; Apostolidis, S.A.; Grammatikos, A.P.; Rodriguez Rodrigues, N.; Ioannidis, C.; Kyttaris, V.C.; Crispin, J.C.; Tsokos, G.C. Stat3 promotes IL-10 expression in lupus T cells through trans-activation and chromatin remodeling. Proc. Natl. Acad. Sci. USA 2014, 111, 13457–13462. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, K.E.; Suriano, A.; Dietzmann, K.; Lin, J.; Goldman, D.; Petri, M.A. The TNF-alpha locus is altered in monocytes from patients with systemic lupus erythematosus. Clin. Immunol. 2007, 123, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Hu, N.; Long, H.; Zhao, M.; Yin, H.; Lu, Q. Aberrant expression pattern of histone acetylation modifier and mitigation of lupus by SIRT1-siRNA in MRL/lpr mice. Scand. J. Rheumatol. 2009, 38, 464–471. [Google Scholar] [CrossRef] [PubMed]
- Javierre, B.M.; Fernandez, A.F.; Richter, J.; Al-Shahrour, F.; Martin-Subero, J.I.; Rodriguez-Ubreva, J.; Berdasco, M.; Fraga, M.F.; O’Hanlon, T.P.; Rider, L.G.; et al. Changes in the pattern of DNA methylation associate with twin discordance in systemic lupus erythematosus. Genome Res. 2010, 20, 170–179. [Google Scholar] [CrossRef]
- Ruvkun, G. Molecular biology. Glimpses of a tiny RNA world. Science 2001, 294, 797–799. [Google Scholar] [CrossRef]
- Dai, R.; Ahmed, S.A. MicroRNA, a new paradigm for understanding immunoregulation, inflammation, and autoimmune diseases. Transl. Res. 2011, 157, 163–179. [Google Scholar] [CrossRef] [Green Version]
- Qu, B.; Shen, N. miRNAs in the pathogenesis of systemic lupus erythematosus. Int. J. Mol. Sci. 2015, 16, 9557–9572. [Google Scholar] [CrossRef]
- Chen, J.-Q.; Papp, G.; Szodoray, P.; Zeher, M. The role of microRNAs in the pathogenesis of autoimmune diseases. Autoimmun. Rev. 2016, 15, 1171–1180. [Google Scholar] [CrossRef] [Green Version]
- Le, X.; Yu, X.; Shen, N. Novel insights of microRNAs in the development of systemic lupus erythematosus. Curr. Opin. Rheumatol. 2017, 29, 450–457. [Google Scholar] [CrossRef]
- Long, H.; Wang, X.; Chen, Y.; Wang, L.; Zhao, M.; Lu, Q. Dysregulation of microRNAs in autoimmune diseases: Pathogenesis, biomarkers and potential therapeutic targets. Cancer Lett. 2018, 428, 90–103. [Google Scholar] [CrossRef]
- Lu, M.C.; Lai, N.S.; Chen, H.C.; Yu, H.C.; Huang, K.Y.; Tung, C.H.; Huang, H.B.; Yu, C.L. Decreased microRNA (miR)-145 and increased miR-224 expression in T cells from patients with systemic lupus erythematosus involved in lupus immunopathogenesis. Clin. Exp. Immunol. 2013, 171, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.C.; Yu, C.L.; Chen, H.C.; Yu, H.C.; Huang, H.B.; Lai, N.S. Aberrant T cell expression of Ca2+ influx-regulated miRNA in patients with systemic lupus erythematosus promotes lupus pathogenesis. Rheumatology (Oxford) 2015, 54, 343–348. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.Y.; Hsieh, S.-C.; Lu, M.-C.; Yu, C.-L. Aberrant non-coding RNA expression profiles as biomarker/biosignature in autoimmune and inflammatory rheumatic diseases. J. Lab. Preci. Med. 2018, 3, 51. [Google Scholar] [CrossRef]
- Su, X.; Ye, L.; Chen, X.; Zhang, H.; Zhou, Y.; Ding, X.; Chen, D.; Lin, Q.; Chen, C. MiR-199-3p promotes ERK-mediated IL-10 production by targeting poly(ADP-ribose)polymerase-1 in patients with systemic lupus eruthematosus. Chemo-Biol. Interact. 2019, 306, 110–116. [Google Scholar] [CrossRef]
- Heegaard, N.H.H.; Carlsen, A.L.; Skovgaard, K.; Heegaard, P.M.H. Circulating extracellular microRNA in systemic autoimmunity. Exp. Suppl. 2015, 106, 171–195. [Google Scholar]
- Turpin, D.; Truchetet, M.E.; Faustin, B.; Augusto, J.F.; Contin-Bordes, C.; Brisson, A.; Blanco, P.; Duffau, P. Role of extracellular vesicles in autoimmune diseases. Autoimmun. Rev. 2016, 15, 174–183. [Google Scholar] [CrossRef]
- Carlsen, A.L.; Schetter, A.J.; Nielsen, C.T.; Lood, C.; Knudsen, S.; Voss, A.; Harris, C.C.; Hellmark, T.; Segelmark, M.; Jacobsen, S.; et al. Circulating microRNA expression profiles associated with systemic lupus erythematosus. Arthritis Rheum. 2013, 65, 1324–1334. [Google Scholar] [CrossRef]
- Kim, B.-S.; Jung, J.-Y.; Jeon, J.-Y.; Kim, H.-A.; Suh, C.-H. Circulating hsa-miR-30e-5p, hsa-miR-92a-3p, and hsa-miR-223-3p may be novel biomarkers in systemic lupus erythematosus. HLA 2016, 88, 187–193. [Google Scholar] [CrossRef]
- Ishibe, Y.; Kusaoi, M.; Murayama, G.; Nemoto, T.; Kon, T.; Ogasawara, M.; Kempe, K.; Yamaji, K.; Tamura, N. Changes in the expression of Circulating microRNAs in systemic lupus erythematosus patient blood plasma after passing through a plasma absorption membrane. Ther. Apher. Dial. 2018, 22, 278–289. [Google Scholar] [CrossRef]
- Natasha, G.; Gundogan, B.; Tan, A.; Farhatnia, Y.; Wu, W.; Rajadas, J.; Seifalian, A.M. Exosomes as immunotheranostic nanoparticles. Clin. Ther. 2014, 36, 820–829. [Google Scholar] [CrossRef]
- Tan, L.; Wu, H.; Liu, Y.; Zhao, M.; Li, D.; Lu, Q. Recent advances of exosomes in immune modulation and autoimmune diseases. Autoimmunity 2016, 49, 357–365. [Google Scholar] [CrossRef] [PubMed]
- Rekker, K.; Saare, M.; Roost, A.M.; Kubo, A.-L.; Zarovni, N.; Chiesi, A.; Salumets, A.; Peters, M. Comparison of serum exosome isolation methods for microRNA profiling. Clin. Biochem. 2014, 47, 135–138. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, S.C.; Tsai, C.Y.; Yu, C.L. Potential serum and urine biomarkers in patients with lupus nephritis and the unsolved problems. Open Access Rheumatol. 2016, 8, 81–91. [Google Scholar] [PubMed]
- Tsai, C.Y.; Lu, M.C.; Yu, C.L. Can urinary exosomal micro-RNA detection become a diagnostic and prognostic gold standard for patients with lupus nephritis and diabetic nephropathy? J. Lab. Precis. Med. 2017, 2, 91. [Google Scholar] [CrossRef]
- Perez-Hernandez, J.; Redon, J.; Cortes, R. Extracellular vesicles as therapeutic agents in systemic lupus erythemaotusus. Int. J. Mol. Sci. 2017, 18, E717. [Google Scholar] [CrossRef]
- Xu, H.; Jia, S.; Xu, H. Potential therapeutic applications of exosomes in different autoimmune diseases. Clin. Immunol. 2019, 205, 116–124. [Google Scholar] [CrossRef]
- Kelemen, E.; Danis, J.; Göblös, A.; Bata-Csörgö, Z.; Szell, M. Exosomal long non-coding RNAs as biomarkers in human diseases. J. Int. Fed. Clin. Chem. Lab. Med. 2019, 30, 224–236. [Google Scholar]
- Wu, G.C.; Hu, Y.; Guan, S.Y.; Ye, D.Q.; Pan, H.F. Differential plasma expression profiles of long non-coding RNAs, reveal potential biomarkers for systemic lupus erythematosus. Biomolecules 2019, 9, E206. [Google Scholar] [CrossRef]
- Gloss, B.S.; Dinger, M.E. The specificity of long noncoding RNA expression. Biochim. Biophys. Acta 2016, 1859, 16–22. [Google Scholar] [CrossRef]
- Derrien, T.; Johnson, R.; Bussotti, G.; Tanzer, A.; Djebali, S.; Tilgner, H.; Guemec, G.; Martin, D.; Merkel, A.; Knowles, D.G.; et al. The GENCODE v7 catalog of human lung noncoding RNAs: Analysis of their gene structure, evolution, and expression. Genome Res. 2012, 22, 1775–1789. [Google Scholar] [CrossRef]
- Cabili, M.N.; Trapnell, C.; Goff, L.; Koziol, M.; Tazon-Vega, B.; Regev, A.; Rinn, J.L. Integrative annotation of human large intergenic noncoding RNAs reveals global properties and specific subclasses. Genes Dev. 2011, 25, 1915–1927. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hadjicharalambous, M.R.; Lindsay, M.A. Long non-coding RNAs and the innate immune response. Noncoding RNA 2019, 5, E34. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chen, S.; Chen, S.; Du, J.; Lin, J.; Qin, H.; Wang, J.; Liang, J.; Xu, J. Long noncoding RNA expression profile and association with SLEDAI score in monocyte-derived dendritic cells from patients with systemic lupus erythematosus. Arthritis Res. Ther. 2018, 20–138. [Google Scholar] [CrossRef]
- Mathy, N.W.; Chen, X.-M. Long non-coding RNAs (lncRNAs) and their transcriptional control of inflammatory responses. J. Biol. Chem. 2017, 292, 12375–12382. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luo, Q.; Li, X.; Xu, C.; Zeng, L.; Ye, J.; Guo, Y.; Huang, Z.; Li, J. Integrative analysis of long non-coding RNAs and messenger RNA expression profiles in systemic lupus erythematosus. Mol. Med. Rep. 2018, 17, 3489–3496. [Google Scholar] [PubMed]
- Zhao, C.N.; Mao, Y.M.; Liu, L.N.; Li, X.M.; Wang, D.G.; Pan, H.F. Emerging role of lncRNAs in systemic lupus erythematosus. Biomed. Pharm. 2018, 106, 584–592. [Google Scholar] [CrossRef]
- Simchovitz, A.; Hanan, M.; Niederhoffer, N.; Madrer, N.; Yayon, N.; Bennett, E.R.; Greenberg, D.S.; Kadener, S.; Soreq, H. NEAT1 is overexpressed in Parkinson’s disease substantia nigra and confers drug-inducible neuroprotection from oxidative stress. FASEB J. 2019, 33, 11223–11234. [Google Scholar] [CrossRef]
- Li, K.J.; Wu, C.H.; Hsieh, S.C.; Lu, M.C.; Tsai, C.Y.; Yu, C.L. Deranged bioenergetics and defective redox capacity in T-lymphocytes and neutrophils are related to cellular dysfunction and increased oxidative stress in patients with active systemic lupus erythematosus. Clin. Dev. Immunol. 2012, 2012, 548516. [Google Scholar] [CrossRef]
- Lee, H.-T.; Lin, C.-S.; Lee, C.-S.; Tsai, C.-Y.; Wei, Y.-H. Increase 8-hydroxy-2′-deoxyguanosine in plasma and decreased mRNA expression of human 8-oxoguanine DNA glycosylase 1, anti-oxidant enzymes, mitochondrial biogenesis-related proteins and glycolytic enzymes in leukocytes in patients with systemic lupus erythematosus. Clin. Exp. Immunol. 2014, 176, 66–77. [Google Scholar]
- Yang, S.K.; Zhang, H.R.; Shi, S.P.; Zhu, Y.Q.; Song, N.; Dai, Q.; Zhang, W.; Gui, M.; Zhang, H. The role of mitochondria in systemic lupus erythematosus: A glimpse of various pathogenetic mechanisms. Curr. Med. Chem. 2018. [Google Scholar] [CrossRef]
- Tsai, C.Y.; Shen, C.Y.; Liao, H.T.; Li, K.J.; Lee, H.T.; Lu, C.S.; Wu, C.H.; Kuo, Y.M.; Hsieh, S.C.; Yu, C.L. Molecular and cellular bases of immunosenescence, inflammation, and cardiovascular complications mimicking “inflammaging” in patients with systemic lupus erythematosus. Int. J. Mol. Sci. 2019, 20, 3878. [Google Scholar] [CrossRef] [PubMed]
- Vlassopoulos, A.; Lean, M.E.J.; Combet, E. Oxidative stress, protein glycation and nutrition-interactions relevant to health and disease throughout the lifecycle. Proc. Nutr. Soc. 2014, 73, 430–438. [Google Scholar] [CrossRef] [PubMed]
- McGuire, P.J. Mitochondrial dysfunction and the aging immune system. Biology (Basel) 2019, 8, E26. [Google Scholar] [CrossRef] [PubMed]
- Ye, B.; Hou, N.; Xiao, L.; Xu, Y.; Xu, H.; Li, F. Dynamic monitoring of oxidative DNA double strand break and repair in cardiomyocytes. Cardiovasc. Pathol. 2016, 25, 93–100. [Google Scholar] [CrossRef] [PubMed]
- de Leeuw, K.; Graaff, R.; de Vries, R.; Dullaart, R.P.; Smit, A.J.; Kallenberg, C.G.; Bjil, M. Accumulation of advanced glycation endproducts in patients with systemic lupus erythematosus. Rheumatology 2007, 46, 1551–1556. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Jiang, M.; Chen, W.; Zhao, T.; Wei, Y. Cancer and ER stress: Mutual crosstalk between autophagy, oxidative stress and inflammatory response. Biomed. Pharmacother. 2019, 118, 109249. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Du, Y.; Le, W.; Wang, K.; Kieffer, N.; Zhang, J. Redox control of the survival of healthy and diseased cells. Antioxid. Redox Signal. 2011, 15, 2867–2908. [Google Scholar] [CrossRef]
- Cao, S.S.; Kaufman, R.J. Endoplasmic reticulum stress and oxidative stress in cell fate decision and human disease. Antioxid. Redox Signal. 2014, 21, 396–413. [Google Scholar] [CrossRef]
- Pervaiz, S. Redox dichotomy in cell fate decision: Evasive mechanism or Achilles heel? Antioxid. Redox Signal. 2018, 29, 1191–1195. [Google Scholar] [CrossRef]
- Prasad, K.N. Oxidative stress and pro-inflammatory cytokines may act as one of the signals for regulating microRNAs expression in Alzheimer’s disease. Mech. Ageing Dev. 2017, 162, 63–71. [Google Scholar] [CrossRef]
- Prasad, K.N. Oxidative stress, pro-inflammatory cytokines, and antioxidants regulate expression levels of microRNAs in Parkinson’s disease. Curr. Aging Sci. 2017, 10, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Prasad, K.N.; Bondy, S.C. MicroRNAs in hearing disorders: Their regulation by oxidative stress, inflammation and antioxidants. Front Cell Neurosci. 2017, 11, 276. [Google Scholar] [CrossRef] [PubMed]
- Guillaumet-Adkins, A.; Yańez, Y.; Peris-Diaz, M.D.; Calabria, I.; Palanca-Ballester, C.; Sandoval, J. Epigenetics and oxidative stress in aging. Oxid. Med. Cell Longev. 2017, 2017, 9175806. [Google Scholar] [CrossRef]
- Cheleschi, S.; De Palma, A.; Pascarelli, N.A.; Giordano, N.; Galeazzi, M.; Tenti, S.; Fioravanti, A. Could oxidative stress regulate the expression of microRNA-146a and microRNA-34a in human osteoarthritic chondrocyte cultures? Int. J. Mol. Sci. 2017, 18, E2660. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Xu, W.; Feng, Y.; Zhou, X. Non-coding RNA involvement in the pathogenesis of diabetic cardiomyopathy. J. Cell. Mol. Med. 2019, 23, 5859–5867. [Google Scholar] [CrossRef]
- Lan, J.; Huang, Z.; Han, J.; Shao, J.; Huang, C. Redox regulation of microRNAs in cancer. Cancer Lett. 2018, 418, 250–259. [Google Scholar] [CrossRef]
- Esposti, D.D.; Aushev, V.N.; Lee, E.; Cros, M.-P.; Zhu, J.; Herceg, Z.; Chen, J.; Hernandez-Vargas, H. miR-500a-5p regulates oxidative stress response genes in breast cancer and predicts cancer survival. Sci. Rep. 2017, 7, 15966. [Google Scholar] [CrossRef]
- Sangokoya, C.; Telen, M.J.; Chi, J.-T. microRNA miR-144 modulates oxidative stress tolerance and associates with anemia severity in sickle cell disease. Blood 2010, 116, 4338–4348. [Google Scholar] [CrossRef]
- Kim, C.; Kang, D.; Lee, E.K.; Lee, J.S. Long noncoding RNAs and RNA binding proteins in oxidative stress, cellular senescence, and age-related diseases. Oxid. Med. Cell Longev. 2017, 2017, 2062384. [Google Scholar] [CrossRef]
- Tehrani, S.S.; Karimian, A.S.; Parsian, H.; Majidinia, M.; Yousefi, G. Multiple functions of long non-coding RNAs in oxidative stress, DNA damage response and cancer progression. J. Cell. Biochem. 2018, 119, 223–236. [Google Scholar] [CrossRef]
- Mohan, I.K.; Das, U.N. Oxidant stress, anti-oxidants and essential fatty acids in systemic lupus erythematosus. Prostaglandins Leukot. Essent Fatty Acids 1997, 56, 193–198. [Google Scholar] [CrossRef]
- Kudaravalli, J. Improvement in endothelial dysfunction in patients with systemic lupus erythematosus with N-acetylcysteine and atorvastatin. Ind. J. Pharmacol. 2011, 43, 311–315. [Google Scholar] [CrossRef] [PubMed]
- Lai, Z.W.; Hanczko, R.; Bonilla, E.; Caza, T.N.; Clair, B.; Bartos, A.; Miklossy, G.; Jimah, J.; Deherty, E.; Tily, H.; et al. N-aceylcysteine reduces disease activity by blocking mammalian target of rapamycin in T cells from systemic lupus erythematosus patients: A randomized double blind, placebo-controlled trial. Arthritis Rheum. 2012, 64, 2937–2946. [Google Scholar] [CrossRef] [PubMed]
- Tzang, B.S.; Hsu, T.C.; Kuo, C.Y.; Chen, T.Y.; Chiang, S.Y.; Li, S.L.; Kao, S.H. Cytamine attenuates lupus-associated apoptosis of ventricular tissue by suppressing both intrinsic and extrinsic pathways. J. Cell. Mol. Med. 2012, 16, 2104–2111. [Google Scholar] [CrossRef] [PubMed]
- Portal-Núňez, S.; Esbrit, P.; Alcaraz, M.J.; Largo, R. Oxidative stress, autophagy, epigenetic changes and regulation by miRNAs as potential therapeutic targets in osteoarthritis. Biochem. Pharmacol. 2016, 108, 1–10. [Google Scholar] [CrossRef]
- Dong, D.; Zhang, Y.; Reece, E.A.; Wang, L.; Harman, C.R.; Yang, P. microRNA expression profiling and functional annotation analysis of their targets modulated by oxidative stress during embryonic heart development in diabetic mice. Reprod. Toxicol. 2016, 65, 365–374. [Google Scholar] [CrossRef] [Green Version]


|
| SLE | lnc RNA Expression | Target mRNA | Pathological Processes |
|---|---|---|---|
| Intracellular [103,104,105,106] | |||
| NEAT1↑* | IL-6↑, IFN↑, CXCL10↑ | DNA hypomethylation | |
| MALAT1↑ | IL-21↑, SIRT1↑ | SLEDAI-2K↑ | |
| Linc0597↑ | TNF-α↑, IL-6↑ | ESR↑, CRP↑, C3 ↓, | |
| Linc DC↑ | STAT3↑ | Th1↑ | |
| ENST00000604411.1↑ | XIST | SLEDAI score↑ | |
| ENST000005011222↑ | NEAT1 | ||
| Linc 0949↓ | TNF-α↑, IL-6↑ | Inflammation↑ | |
| Linc-HSFY2-3:3↓ | - | SLEDAI score↑ | |
| Linc-SERPIN139-1:2↓ | - | ||
| Gas 5↓ | Apoptotic gene↓ | T cell apoptosis↓ | |
| Circulating plasma exosomal [98] | |||
| Linc0597↑ | TNF-α↑, IL-6↑ | MAPK signaling↑ | |
| Lnc0640↑ | Phosphatase 4 (DUSP4)↑ | Lupus pathogenesis | |
| Lnc5150↑ | Arrestin β2 (ARRB2)↑ | ||
| Ribosomal protein S6 kinase A5 (RPS6KA5)↑ | |||
| Gas 5↓ | Apoptotic gene↓ | T cell apoptosis↓ | |
| Lnc 7074↓ | |||
| SLE | miRNA | Target mRNA | Pathological Process | |
|---|---|---|---|---|
| Intracellular [82,83,84,85,86] | ● Increase in: | |||
| miR-21* | Arylamide small nucleotide inhibiors | DNA hypomethylation↑ | ||
| miR-524-5p | Jagged-1, Hes-1 | IFN-γ↑, SLEDAI↑ | ||
| miR-126 | KRAS | |||
| miR-148a | PTEN | |||
| ● Decrease in: | ||||
| miR-142-3p | HMGB-1 | T and B activation↑ | ||
| miR-142-5p | PD-L1 | |||
| miR-146a* | IRF-5, STAF-1 | Innate immune response↑, lupus nephritis↑ | ||
| miR-224↑ | API5 | Type 1, IFN↑ | ||
| miR199-3p↑ | PARP-1 | IL-10↑ | ||
| ● Decrease in: | ||||
| miR-31 | RhoA | Cell apoptosis↑ | ||
| miR-142-3p | HMGB-1 | |||
| miR410 | STAT3 | |||
| miR-125a | STAT3, hexokinase 2, NEDDG | IL-10↑ | ||
| miR-125b* | Claudin 2, cingulin, SYVN1 | |||
| mi-1273e | Th17/Treg ratio↑ | |||
| miR-3201 | ||||
| Circulating plasma [87,88,89,90,91,92,93,94] | ● Increase in: | |||
| miR-142-3p | IL-1β | |||
| miR-181a | FoxO1 | |||
| hsa-miR-30e-5p hsa-miR-92a-3p | Oral ulcer and lupus anticoagulant | |||
| hsa-miR-223-3p | ||||
| miR-16-5p | p38MAPK, NF-κB | |||
| miR-223-3p | Voltage-gated K+ channel KV4.2 | |||
| miR-451 | LKB1/AMPK | |||
| ● Decrease in: | ||||
| miR-106a | THBS2 | |||
| miR-17 | JAB1/CSN5 | |||
| miR-20a | IkBβ | |||
| miR-203 | ZEB1 | |||
| miR-92a | p63 | |||
| miR-146a | JAK2/STAT3 | |||
| miR-1202 | cyclin dependent kinase 14 | |||
| Urinary exosomal (lupus Nephritis) [95,96] | ● Increase in: | |||
| miR-125a | STAT3, hexokinase 2, NEDDG | Glomerulonephritis | ||
| miR-146* | NF-κB | |||
| miR-150 | Akt3 | |||
| miR-155 | PTEN, Wnt/β-catenin | |||
| ● Decrease in: | ||||
| miR-141 | Tram1, GL/2, TGF-β | Glomerulonephritis | ||
| miR-192 | nin one binding protein | |||
| miR-200a | HMGB1/RAGE | |||
| miR-200c | ZEB1, Notch 1 | |||
| miR-221 | BIM-Bax/Bak, TIMP3 | |||
| miR-222 | PPP2R2A/Akt/mTOR, PCSK9 | |||
| miR-429 | TRAF6, DLC-1, HIF-1α | |||
| ● Decrease in: | ||||
| miR-3201 | Endocapillary glomerular inflammation | |||
| miR-1273e | ||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsai, C.-Y.; Hsieh, S.-C.; Lu, C.-S.; Wu, T.-H.; Liao, H.-T.; Wu, C.-H.; Li, K.-J.; Kuo, Y.-M.; Lee, H.-T.; Shen, C.-Y.; et al. Cross-Talk between Mitochondrial Dysfunction-Provoked Oxidative Stress and Aberrant Noncoding RNA Expression in the Pathogenesis and Pathophysiology of SLE. Int. J. Mol. Sci. 2019, 20, 5183. https://doi.org/10.3390/ijms20205183
Tsai C-Y, Hsieh S-C, Lu C-S, Wu T-H, Liao H-T, Wu C-H, Li K-J, Kuo Y-M, Lee H-T, Shen C-Y, et al. Cross-Talk between Mitochondrial Dysfunction-Provoked Oxidative Stress and Aberrant Noncoding RNA Expression in the Pathogenesis and Pathophysiology of SLE. International Journal of Molecular Sciences. 2019; 20(20):5183. https://doi.org/10.3390/ijms20205183
Chicago/Turabian StyleTsai, Chang-Youh, Song-Chou Hsieh, Cheng-Shiun Lu, Tsai-Hung Wu, Hsien-Tzung Liao, Cheng-Han Wu, Ko-Jen Li, Yu-Min Kuo, Hui-Ting Lee, Chieh-Yu Shen, and et al. 2019. "Cross-Talk between Mitochondrial Dysfunction-Provoked Oxidative Stress and Aberrant Noncoding RNA Expression in the Pathogenesis and Pathophysiology of SLE" International Journal of Molecular Sciences 20, no. 20: 5183. https://doi.org/10.3390/ijms20205183
APA StyleTsai, C.-Y., Hsieh, S.-C., Lu, C.-S., Wu, T.-H., Liao, H.-T., Wu, C.-H., Li, K.-J., Kuo, Y.-M., Lee, H.-T., Shen, C.-Y., & Yu, C.-L. (2019). Cross-Talk between Mitochondrial Dysfunction-Provoked Oxidative Stress and Aberrant Noncoding RNA Expression in the Pathogenesis and Pathophysiology of SLE. International Journal of Molecular Sciences, 20(20), 5183. https://doi.org/10.3390/ijms20205183