Extracellular Matrix in Regulation of Contractile System in Cardiomyocytes
Abstract
1. Introduction
2. Literature Overview
3. Actin-Based Contractile Systems in Muscle and Non-Muscle Cells
3.1. Actin Cytoskeleton
3.2. Actin Filaments in Myofibrillar Apparatus
3.3. Actin Isoforms
4. Dynamics of Contractile Apparatus in Cardiomyocytes
4.1. Myofibrillogenesis
4.2. Actin Isoform Switching during Differentiation of Cardiomyocytes
4.3. Rearrangements of Contractile Apparatus in Cardiomyocytes in Culture
5. Extracellular Matrix
5.1. Cardiac Extracellular Matrix
5.2. Extracellular Matrix in Heart Development
5.3. Extracellular Matrix in Cardiogenic Differentiation Methods
5.4. Culturing of Cardiomyocytes in the Presence of Extracellular Matrix
5.5. Extracellular Matrix Production by Cardiomyocytes in Culture
6. Integrins in Cardiomyocytes
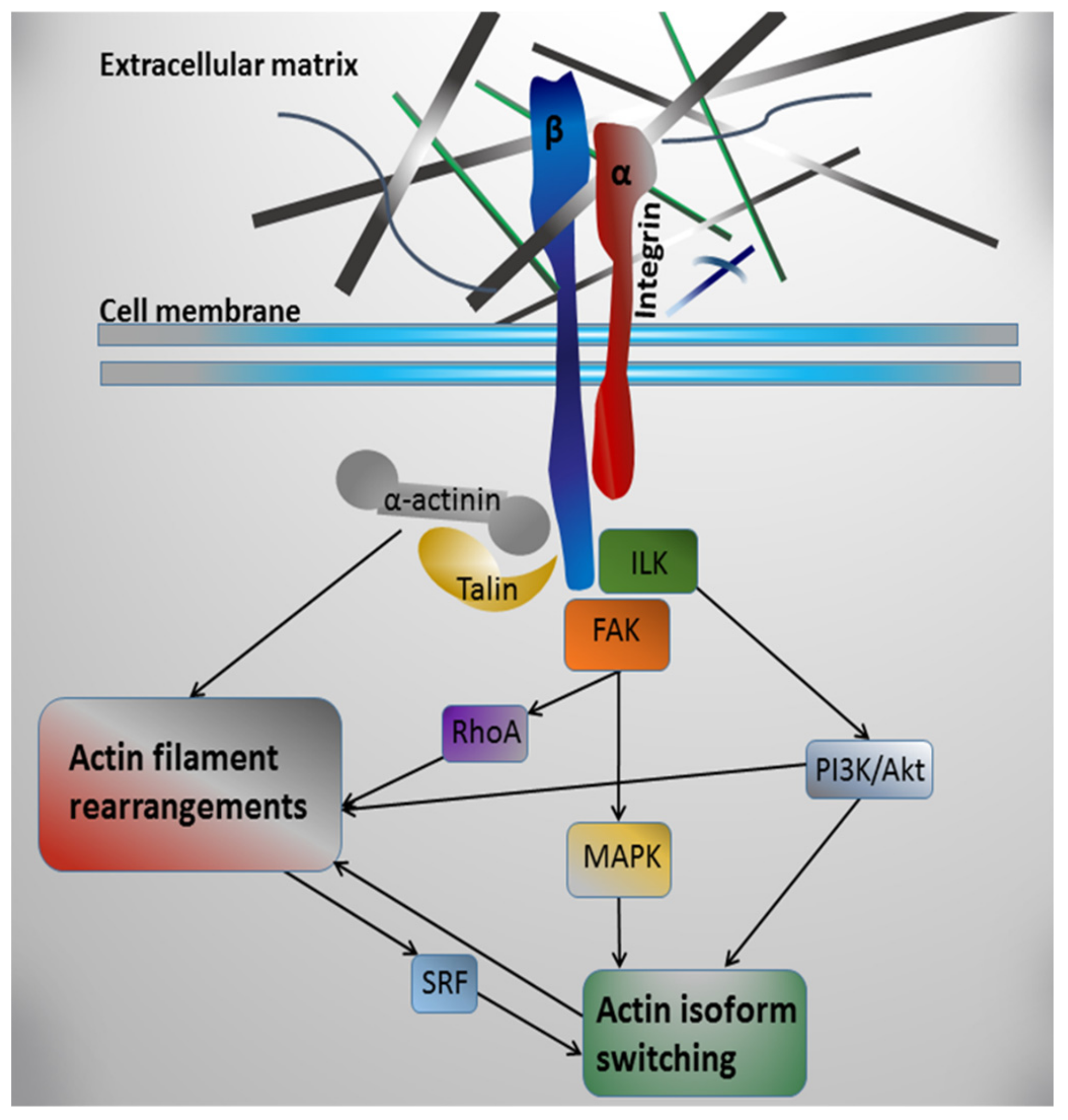
7. Possible Mechanisms of Integrin-Mediated Regulation of Actin Contractile System in Cardiomyocytes
8. Conclusions
Funding
Conflicts of Interest
Abbreviations
| CMs | Cardiomyocytes |
| ECM | Extracellular matrix |
| SMA | Smooth muscle actin |
| ILK | Integrin-linked kinase |
| FAK | Focal adhesion kinase |
| SRF | Serum response factor |
References
- Vandekerckhove, J.; Bugaisky, G.; Buckingham, M. Simultaneous expression of skeletal muscle and heart actin proteins in various striated muscle tissues and cells. J. Biol. Chem. 1986, 261, 1838–1843. [Google Scholar] [PubMed]
- Van Bilsen, M.; Chien, K.R. Growth and hypertrophy of the heart: Toward an understanding of cardiac specific and inducible gene expression. Cardiovasc. Res. 1993, 27, 1140–1149. [Google Scholar] [CrossRef] [PubMed]
- Ampe, C.; Van Troys, M. Mammalian Actins: Isoform-Specific Functions and Diseases. Handb. Exp. Pharmacol. 2017, 235, 1–37. [Google Scholar] [PubMed]
- Clément, S.; Stouffs, M.; Bettiol, E.; Kampf, S.; Krause, K.H.; Chaponnier, C.; Jaconi, M.J. Expression and function of alpha–smooth muscle actin during embryonic-stem-cell-derived cardiomyocyte differentiation. J. Cell Sci. 2007, 120, 229–238. [Google Scholar] [PubMed]
- Potta, S.P.; Liang, H.; Winkler, J.; Doss, M.X.; Chen, S.; Wagh, V.; Pfannkuche, K.; Hescheler, J.; Sachinidis, A. Isolation and functional characterization of alpha-smooth muscle actin expressing cardiomyocytes from embryonic stem cells. Cell. Physiol. Biochem. 2010, 25, 595–604. [Google Scholar] [PubMed]
- Ruzicka, D.L.; Schwartz, R.J. Sequential activation of alpha-actin genes during avian cardiogenesis: Vascular smooth muscle alpha-actin gene transcripts mark the onset of cardiomyocyte differentiation. J. Cell Biol. 1988, 107, 2575–2586. [Google Scholar] [PubMed]
- Handel, S.E.; Greaser, M.L.; Schultz, E.; Wang, S.M.; Bulinski, J.C.; Lin, J.J.; Lessard, J.L. Chicken cardiac myofibrillogenesis studied with antibodies specific for titin and the muscle and nonmuscle isoforms of actin and tropomyosin. Cell Tissue Res. 1991, 263, 419–430. [Google Scholar] [PubMed]
- Eppenberger-Eberhardt, M.; Flamme, I.; Kurer, V.; Eppenberger, H.M. Reexpression of α-smooth muscle actin isoform in cultured adult rat cardiomyocytes. Dev. Biol. 1990, 139, 269–278. [Google Scholar] [PubMed]
- Schaub, M.C.; Hefti, M.A.; Harder, B.A.; Eppenberger, H.M. Various hypertrophic stimuli induce distinct phenotypes in cardiomyocytes. J. Mol. Med. 1997, 75, 901–920. [Google Scholar] [PubMed]
- Harder, B.A.; Hefti, M.A.; Eppenberger, H.M.; Schaub, M.C. Differential protein localization in sarcomeric and nonsarcomeric contractile structures of cultured cardiomyocytes. J. Struct. Biol. 1998, 122, 162–175. [Google Scholar] [PubMed]
- Bildyug, N.; Bozhokina, E.; Khaitlina, S. Contribution of α-smooth muscle actin and extracellular matrix to the in vitro reorganization of cardiomyocyte contractile system. Cell Biol. Int. 2016, 40, 472–477. [Google Scholar] [PubMed]
- Sanchez-Quintana, D.; Garcia-Martinez, V.; Macias, D.; Hurle, J.M. Structural arrangement of the extracellular matrix network during myocardial development in the chick embryo heart. Anat. Embryol. (Berl.) 1991, 184, 451–460. [Google Scholar] [PubMed]
- Lockhart, M.; Wirrig, E.; Phelps, A.; Wessels, A. Extracellular Matrix and Heart Development. Birth Defects Res. A Clin. Mol. Teratol. 2011, 91, 535–550. [Google Scholar] [PubMed]
- Bildiug, N.B.; Pinaev, G.P. Extracellular matrix dependence of the cardiomyocyte contractile apparatus organization. Tsitologiia 2013, 55, 713–724. [Google Scholar]
- Bil’diug, N.B.; Iudintseva, N.M.; Pinaev, G.P. Contractile apparatus organization of cardiomyocytes upon their cultivation in collagen gels. Tsitologiia 2014, 56, 822–827. [Google Scholar] [PubMed]
- Kelly, D.E. Myofibrillogenesis and Z-band differentiation. Anat. Rec. 1969, 163, 403–425. [Google Scholar] [PubMed]
- Fischman, D.A. The synthesis and assembly of myofibrils in embryonic muscle. Curr. Top. Dev. Biol. 1970, 5, 235–280. [Google Scholar] [PubMed]
- Isobe, Y.; Shimada, Y. Myofibrillogenesis in vitro as seen with the scanning electron microscope. Cell Tissue Res. 1983, 231, 481–494. [Google Scholar] [PubMed]
- Lichnovský, V. Early stages of myofibrillogenesis in human embryonic and fetal heart. Acta Univ. Palacki. Olomuc. Fac. Med. 1983, 104, 151–166. [Google Scholar] [PubMed]
- Holtzer, H.; Sasse, J.; Horwitz, A.; Antin, P.; Pacifici, M. Myogenic lineages and myofibrillogenesis. Bibl. Anat. 1986, 29, 109–125. [Google Scholar]
- Sanger, J.M.; Mittal, B.; Pochapin, M.B.; Sanger, J.W. Myofibrillogenesis in living cells microinjected with fluorescently labeled α-actinin. J. Cell Biol. 1986, 102, 2053–2066. [Google Scholar] [PubMed]
- Imanaka-Yoshida, K. Myofibrillogenesis in precardiac mesoderm explant culture. Cell Struct. Funct. 1997, 22, 45–49. [Google Scholar] [PubMed]
- Du, A.; Sanger, J.M.; Sanger, J.W. Cardiac myofibrillogenesis inside intact embryonic hearts. Dev. Biol. 2008, 318, 236–246. [Google Scholar] [PubMed]
- Sanger, J.W.; Kang, S.; Siebrands, C.C.; Freeman, N.; Du, A.; Wang, J.; Stout, A.L.; Sanger, J.M. How to build a myofibril. J. Muscle Res. Cell Motil. 2005, 26, 343–354. [Google Scholar] [PubMed]
- Sanger, J.W.; Wang, J.; Fan, Y.; White, J.; Mi-Mi, L.; Dube, D.K.; Sanger, J.M.; Pruyne, D. Assembly and Maintenance of Myofibrils in Striated Muscle. Handb. Exp. Pharmacol. 2017, 235, 39–75. [Google Scholar] [PubMed]
- Maĭer, R.; David, K.; Vasilev, G.; Guski, G. Ultrastructural-morphometric study of cardiac myocytes in the cell culture. Biulleten Eksp. Biol. Med. 1979, 88, 492–494. [Google Scholar]
- Dlugosz, A.A.; Antin, P.B.; Nachmias, V.T.; Holtzer, H. The relationship between stress fiber-like structures and nascent myofibrils in cultured cardiac myocytes. J. Cell Biol. 1984, 99, 2268–2278. [Google Scholar] [PubMed]
- Borisov, A.B.; Goncharova, E.I.; Pinaev, G.P.; Rumiantsev, P.P. Changes in α-actinin localization and myofibrillogenesis in rat cardiomyocytes under cultivation. Tsitologiia 1989, 31, 642–646. [Google Scholar]
- Messerli, J.; Eppenberger-Eberhardt, M.; Rutishauser, B.; Schwarb, P.; von Arx, P.; Koch-Schneidemann, S.; Eppenberger, H.; Perriard, J. Remodelling of cardiomyocyte cytoarchitecture visualized by three-dimensional (3D) confocal microscopy. Histochemistry 1993, 100, 193–202. [Google Scholar] [PubMed]
- Nag, A.C.; Lee, M.L.; Sarkar, F.H. Remodelling of adult cardiac muscle cells in culture: Dynamic process of disorganization and reorganization of myofibrils. J. Muscle Res. Cell Motil. 1996, 17, 313–334. [Google Scholar]
- LoRusso, S.M.; Rhee, D.; Sanger, J.M.; Sanger, J.W. Premyofibrils in spreading adult cardiomyocytes in tissue culture: Evidence for reexpression of the embryonic program for myofibrillogenesis in adult cells. Cell Motil. Cytoskelet. 1997, 37, 183–198. [Google Scholar]
- Hilenski, L.L.; Terracio, L.; Sawyer, R.; Borg, T.K. Effects of extracellular matrix on cytoskeletal and myofibrillar organization in vitro. Scanning Microsc. 1989, 3, 535–548. [Google Scholar] [PubMed]
- Geisse, N.A.; Sheehy, S.P.; Parker, K.K. Control of myocyte remodeling in vitro with engineered substrates. In Vitro Cell. Dev. Biol. Anim. 2009, 45, 343–350. [Google Scholar] [PubMed]
- Yang, H.; Borg, T.K.; Liu, H.; Gao, B.Z. Interactive relationship between basement-membrane development and sarcomerogenesis in single cardiomyocytes. Exp. Cell Res. 2015, 330, 222–232. [Google Scholar] [PubMed]
- Pinkert, M.A.; Hortensius, R.A.; Ogle, B.M.; Eliceiri, K.W. Imaging the Cardiac Extracellular Matrix. Adv. Exp. Med. Biol. 2018, 1098, 21–44. [Google Scholar] [PubMed]
- Hacker, T.A. Animal Models and Cardiac Extracellular Matrix Research. Adv. Exp. Med. Biol. 2018, 1098, 45–58. [Google Scholar] [PubMed]
- Corda, S.; Samuel, J.L.; Rappaport, L. Extracellular matrix and growth factors during heart growth. Heart Fail. Rev. 2000, 5, 119–130. [Google Scholar] [PubMed]
- Little, C.D.; Rongish, B.J. The extracellular matrix during heart development. Experientia 1995, 51, 873–882. [Google Scholar] [PubMed]
- Kwak, H.B. Aging, exercise, and extracellular matrix in the heart. J. Exerc. Rehabil. 2013, 9, 338–347. [Google Scholar]
- Fomovsky, G.M.; Thomopoulos, S.; Holmes, J.W. Contribution of Extracellular Matrix to the Mechanical Properties of the Heart. J. Mol. Cell. Cardiol. 2010, 48, 490–496. [Google Scholar]
- Jourdan-Lesaux, C.; Zhang, J.; Lindsey, M.L. Extracellular Matrix Roles During Cardiac Repair. Life Sci. 2010, 87, 391–400. [Google Scholar] [CrossRef] [PubMed]
- Mishra, P.K.; Givvimani, S.; Chavali, V.; Tyagi, S.C. Cardiac matrix: A clue for future therapy. Biochim. Biophys. Acta 2013, 1832, 2271–2276. [Google Scholar] [CrossRef] [PubMed]
- Takawale, A.; Sakamuri, S.S.; Kassiri, Z. Extracellular Matrix Communication and Turnover in Cardiac Physiology and Pathology. Compr. Physiol. 2015, 5, 687–719. [Google Scholar] [PubMed]
- Hall, M.L.; Ogle, B.M. Cardiac Extracellular Matrix Modification as a Therapeutic Approach. Adv. Exp. Med. Biol. 2018, 1098, 131–150. [Google Scholar] [PubMed]
- Ross, R.S.; Borg, T.K. Integrins and the myocardium. Circ. Res. 2001, 88, 1112–1119. [Google Scholar] [CrossRef] [PubMed]
- Parker, K.K.; Ingber, D.E. Extracellular matrix, mechanotransduction and structural hierarchies in heart tissue engineering. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2007, 362, 1267–1279. [Google Scholar] [CrossRef] [PubMed]
- Borg, T.K.; Baudino, T.A. Dynamic interactions between the cellular components of the heart and the extracellular matrix. Pflug. Arch. 2011, 462, 69–74. [Google Scholar] [CrossRef]
- Valiente-Alandi, I.; Schafer, A.E.; Blaxall, B.C. Extracellular matrix-mediated cellular communication in the heart. J. Mol. Cell. Cardiol. 2016, 91, 228–237. [Google Scholar] [CrossRef]
- Israeli-Rosenberg, S.; Manso, A.M.; Okada, H.; Ross, R.S. Integrins and Integrin-Associated Proteins in the Cardiac Myocyte. Circ. Res. 2014, 114, 572–586. [Google Scholar] [CrossRef]
- Sparrow, J.C.; Schöck, F. The initial steps of myofibril assembly: Integrins pave the way. Nat. Rev. Mol. Cell Biol. 2009, 10, 293–298. [Google Scholar] [CrossRef]
- Letort, G.; Ennomani, H.; Gressin, L.; Théry, M.; Blanchoin, L. Dynamic reorganization of the actin cytoskeleton. F1000Research 2015, 4. [Google Scholar] [CrossRef]
- Small, J.V. The actin cytoskeleton. Electron. Microsc. Rev. 1988, 1, 155–174. [Google Scholar] [CrossRef]
- Small, J.V.; Rottner, K.; Kaverina, I.; Anderson, K.I. Assembling an actin cytoskeleton for cell attachment and movement. Biochim. Biophys. Acta 1998, 16, 271–281. [Google Scholar] [CrossRef]
- Heggeness, M.; Wang, K.; Singer, S. Intracellular distributions of mechanochemical proteins in cultured fibroblasts. Proc. Natl Acad. Sci. USA 1977, 74, 3883–3887. [Google Scholar] [CrossRef] [PubMed]
- Lazarides, E. Immunofluorescence studies on the structure of actin filaments in tissue culture cells. J. Histochem. Cytochem. 1975, 23, 507–528. [Google Scholar] [CrossRef] [PubMed]
- Lazarides, E.; Burridge, K. α-Actinin: Immunofluorescent localization of a muscle structural protein in nonmuscle cells. Cell 1975, 6, 289–298. [Google Scholar] [CrossRef]
- Tucker, R.; Sanford, K.; Frankel, R. Tubulin and actin in paired nonneoplastic and spontaneously transformed neoplastic cell lines in vitro: Fluorescent antibody studies. Cell 1978, 13, 629–642. [Google Scholar] [CrossRef]
- Bretscher, A.; Lynch, W. Identification and localization of immunoreactive forms of caldesmon in smooth and nonmuscle cells: A comparison with the distributions of tropomyosin and α-actinin. J. Cell Biol. 1985, 100, 1656–1663. [Google Scholar] [CrossRef]
- Rönty, M.; Taivainen, A.; Moza, M.; Otey, C.A.; Carpén, O. Molecular analysis of the interaction between palladin and α-actinin. FEBS Lett. 2004, 566, 30–34. [Google Scholar] [CrossRef]
- Gordon, W.E., III. Immunofluorescent and ultrastructural studies of “sarcomeric” units in stress fibers of cultured non-muscle cells. Exp. Cell Res. 1978, 117, 253–260. [Google Scholar] [CrossRef]
- Isenberg, G.; Rathke, P.; Hulsmann, N.; Franke, W.; Wohlfarth-Bottermann, K. Cytoplasmic actomyosin fibrils in tissue culture cells: Direct proof of contractility by visualization of ATP–induced contraction in fibrils isolated by laser micro-beam dissection. Cell Tissue Res. 1976, 27, 427–443. [Google Scholar]
- Kreis, T.; Birchmeier, W. Stress fiber sarcomeres of fibroblasts are contractile. Cell 1980, 22, 555–561. [Google Scholar] [CrossRef]
- Peterson, L.J.; Rajfur, Z.; Maddox, A.S.; Freel, C.D.; Chen, Y.; Edlund, M.; Otey, C.; Burridge, K. Simultaneous stretching and contraction of stress fibers in vivo. Mol. Biol. Cell 2004, 15, 3497–3508. [Google Scholar] [CrossRef] [PubMed]
- Chu, M.; Gregorio, C.C.; Pappas, C.T. Nebulin, a multi-functional giant. J. Exp. Biol. 2016, 219, 146–152. [Google Scholar] [CrossRef] [PubMed]
- Pepe, I.M. Electron spin resonance studies of ultraviolet irradiation effects on some proteins. Radiat. Environ. Biophys. 1971, 7, 115–119. [Google Scholar] [CrossRef]
- Granger, B.L.; Lazarides, E. Desmin and vimentin coexist at the periphery of the myofibril Z disc. Cell 1979, 18, 1053–1063. [Google Scholar] [CrossRef]
- Lazarides, E.; Gard, D.L.; Granger, B.L.; O’Connor, C.M.; Breckler, J.; Danto, S.I. Regulation of the assembly of the Z-disc in muscle cells. Prog. Clin. Biol. Res. 1982, 85, 317–340. [Google Scholar] [PubMed]
- Burgoyne, T.; Morris, E.P.; Luther, P.K. Three-dimensional structure of vertebrate muscle Z-band: The small-square lattice Z-band in rat cardiac muscle. J. Mol. Biol. 2015, 427, 3527–3537. [Google Scholar]
- Vandekerckhove, J.; Weber, K. At least six different actins are expressed in a higher mammal: An analysis based on the amino acid sequence of the amino-terminal tryptic peptide. J. Mol. Biol. 1978, 126, 783–802. [Google Scholar] [CrossRef]
- McHugh, K.M.; Crawford, K.; Lessard, J.L. A comprehensive analysis of the developmental and tissue-specific expression of the isoactin multigene family in the rat. Dev. Biol. 1991, 148, 442–458. [Google Scholar] [CrossRef]
- Kabsch, W.; Vandekerckhove, J. Structure and function of actin. Annu. Rev. Biophys. Biomol. Struct. 1992, 21, 49–76. [Google Scholar] [CrossRef] [PubMed]
- Perrin, B.J.; Ervasti, J.M. The actin gene family: Function follows isoform. Cytoskeleton (Hoboken) 2010, 67, 630–634. [Google Scholar] [CrossRef] [PubMed]
- Hautmann, M.B.; Madsen, C.S.; Mack, C.P.; Owens, G.K. Substitution of the degenerate smooth muscle (SM) α-actin CC(A/T–rich)6GG elements with c-fos serum response elements results in increased basal expression but relaxed SM cell specificity and reduced angiotensin II inducibility. J. Biol. Chem. 1998, 273, 8398–8406. [Google Scholar] [CrossRef] [PubMed]
- Garrels, J.; Gibson, W. Identification and characterization of multiple forms of actin. Cell 1976, 9, 793–805. [Google Scholar] [PubMed]
- Whalen, R.; Butler-Browne, G.; Gros, F. Protein synthesis and actin heterogeneity in calf muscle cells in culture. Proc. Natl. Acad. Sci. USA 1976, 73, 2018–2022. [Google Scholar] [CrossRef] [PubMed]
- Rubenstein, P.; Spudich, J. Actin microheterogeneity in chick embryo fibroblasts. Proc. Natl. Acad. Sci. USA 1977, 74, 120–123. [Google Scholar] [PubMed]
- Khaitlina, S.Y. Functional specificity of actin isoforms. Int. Rev. Cytol. 2001, 202, 35–98. [Google Scholar]
- Sheterline, P.; Clayton, J.; Sparrow, J. Actin. Protein Profile 1995, 2, 1–103. [Google Scholar]
- Chaponnier, C.; Goethals, M.; Janmey, P.A.; Gabbiani, F.; Gabbiani, G.; Vandekerckhove, J. The specific NH2-terminal sequence Ac-EEED of alpha-smooth muscle actin plays a role in polymerization in vitro and in vivo. J. Cell Biol. 1995, 130, 887–895. [Google Scholar] [CrossRef]
- Rubenstein, P. The functional importance of multiple actin isoforms. Bioessays 1990, 12, 309–315. [Google Scholar] [CrossRef]
- Costa, M.L. Cytoskeleton and Adhesion in Myogenesis. ISRN Dev. Biol. 2014, 713631. [Google Scholar] [CrossRef]
- Tondeleir, D.; Vandamme, D.; Vandekerckhove, J.; Ampe, C.; Lambrechts, A. Actin isoform expression patterns during mammalian development and in pathology: Insights from mouse models. Cell Motil. Cytoskelet. 2009, 66, 798–815. [Google Scholar] [CrossRef] [PubMed]
- Joseph, W.; Sanger, J.V.; Wang, J.; Fan, Y.; White, J.; Sanger, J.M. Assembly and Dynamics of Myofibrils. J. Biomed. Biotechnol. 2010, 2010, 858606. [Google Scholar]
- Sanger, J.W.; Chowrashi, P.; Shanerm, N.C.; Spalthoffm, S.; Wangm, J.; Freeman, N.L.; Sanger, J.M. Myofibrillogenesis in skeletal muscle cells. Clin. Orthop. Relat. Res. 2002, 403, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Shaner, N.; Mittal, B.; Zhou, Q.; Chen, J.; Sanger, J.M.; Sanger, J.W. Dynamics of Z-band based proteins in developing skeletal muscle cells. Cell Motil. Cytoskeleton. 2005, 61, 34–48. [Google Scholar] [CrossRef] [PubMed]
- Rhee, D.; Sanger, J.M.; Sanger, J.W. The premyofibril: Evidence for its role in myofibrillogenesis. Cell Motil. Cytoskeleton. 1994, 28, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Dabiri, G.A.; Turnacioglu, K.K.; Sanger, J.M.; Sanger, J.W. Myofibrillogenesis visualized in living embryonic cardiomyocytes. Proc. Natl. Acad. Sci. USA 1997, 99, 9493–9498. [Google Scholar] [CrossRef] [PubMed]
- Golson, M.L.; Sanger, J.M.; Sanger, J.W. Inhibitors arrest myofibrillogenesis in skeletal muscle cells at early stages of assembly. Cell Motil. Cytoskeleton. 2004, 59, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Shao, Y.; Qin, W.; Runyan, R.B.; Xu, M.; Ma, Z.; Borg, T.K.; Markwald, R.; Gao, B.Z. Myosin filament assembly onto myofibrils in live neonatal cardiomyocytes observed by TPEF-SHG microscopy. Cardiovasc. Res. 2013, 97, 262–270. [Google Scholar] [CrossRef]
- Sawtell, N.M.; Lessard, J.L. Cellular distribution of smooth muscle actins during mammalian embryogenesis: Expression of the alpha–vascular but not the gamma-enteric isoform in differentiating striated myocytes. J. Cell Biol. 1989, 109, 2929–2937. [Google Scholar] [CrossRef]
- Schultheiss, T.; Lin, Z.X.; Lu, M.H.; Murray, J.; Fischman, D.A.; Weber, K.; Masaki, T.; Imamura, M.; Holtzer, H. Differential distribution of subsets of myofibrillar proteins in cardiac nonstriated and striated myofibrils. J. Cell Biol. 1990, 110, 1159–1172. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.H.; DiLullo, C.; Schultheiss, T.; Holtzer, S.; Murray, J.M.; Choi, J.; Fischman, D.A.; Holtzer, H. The vinculin/sarcomeric α-actinin/alpha-actin nexus in cultured cardiac myocytes. J. Cell Biol. 1993, 117, 1007–1022. [Google Scholar] [CrossRef] [PubMed]
- Ausma, J.; Litjens, N.; Lenders, M.H.; Duimel, H.; Mast, F.; Wouters, L.; Ramaekers, F.; Allessie, M.; Borgers, M. Time course of atrial fibrillation-induced cellular structural remodeling in atria of the goat. J. Mol. Cell. Cardiol. 2001, 33, 2083–2094. [Google Scholar] [CrossRef] [PubMed]
- Dispersyn, G.D.; Mesotten, L.; Meuris, B.; Maes, A.; Mortelmans, L.; Flameng, W.; Ramaekers, F.; Borgers, M. Dissociation of cardiomyocyte apoptosis and dedifferentiation in infarct border zones. Eur. Heart J. 2002, 23, 849–857. [Google Scholar] [CrossRef] [PubMed]
- Antin, P.B.; Ordahl, C.P. Isolation and characterization of an avian myogenic cell line. Dev. Biol. 1991, 143, 111–121. [Google Scholar] [CrossRef]
- Von Arx, P.; Bantle, S.; Soldati, T.; Perriard, J.C. Dominant negative effect of cytoplasmic actin isoproteins on cardiomyocytes cytoarchitecture and function. J. Cell Biol. 1995, 131, 1759–1773. [Google Scholar] [CrossRef] [PubMed]
- Kaech, S.; Fischer, M.; Doll, T.; Matus, A. Isoform specificity in the relationship of actin to dendritic spines. J. Neurosci. 1997, 17, 9565–9572. [Google Scholar] [CrossRef]
- Kumar, A.; Crafword, K.; Close, L.; Madison, M.; Lorenz, J.; Doetcshman, T.; Pawlowski, S.; Duffy, J.; Neumann, J.; Robbins, J.; et al. Rescue of cardiac α-actin-deficient mice by enteric smooth muscle-actin. Proc. Natl. Acad. Sci. USA 1997, 94, 4406–4411. [Google Scholar] [CrossRef] [PubMed]
- Mounier, N.; Perriard, J.-C.; Gabbiani, G.; Chaponnier, C. Transfected muscle and nonmuscle actins are differentially sorted by cultured smooth muscle and nonmuscle cells. J. Cell Sci. 1997, 110, 839–846. [Google Scholar]
- Fyrberg, E.A.; Fyrberg, C.C.; Biggs, J.R.; Saville, D.; Beall, C.J.; Ketchum, A. Functional nonequivalence of Drosophila actin isoforms. Biochem. Genet. 1998, 36, 271–287. [Google Scholar] [CrossRef]
- Martin, A.F.; Phillips, R.M.; Kumar, A.; Crawford, K.; Abbas, Z.; Lessard, J.L.; de Tombe, P.; Solaro, R.J. Ca2+ activation and tension cost in myofilaments from mouse hearts ectopically expressing enteric γ-actin. Am. J. Physiol. Heart Circ. Physiol. 2002, 283, 642–649. [Google Scholar] [CrossRef]
- Wang, J.; Zohar, R.; McCulloch, C.A. Multiple roles of alpha-smooth muscle actin in mechanotransduction. Exp. Cell Res. 2006, 312, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Driesen, R.B.; Nagaraju, C.K.; Abi-Char, J.; Coenen, T.; Lijnen, P.J.; Fagard, R.H.; Sipido, K.R.; Petrov, V.V. Reversible and irreversible differentiation of cardiac fibroblasts. Cardiovasc. Res. 2014, 101, 411–422. [Google Scholar] [CrossRef] [PubMed]
- Bildyug, N.B.; Khaitlina, S.Y. Redistribution of Sarcomeric Myosin and α-Actinin in Cardiomyocytes in Culture upon the Rearrangement of their Contractile Apparatus. Cell Tissue Biol. 2019, 13, 360–365. [Google Scholar]
- Namba, Y.; Ito, M.; Zu, Y.; Shigesada, K.; Maruyama, K. Human T cell Lplastin bundles actin filaments in a calcium-dependent manner. J. Biochem. 1992, 112, 503–507. [Google Scholar] [CrossRef] [PubMed]
- Weber, A.; Nachmias, V.T.; Pennise, C.R.; Pringm, M.; Safer, D. Interaction of thymosin beta 4 with muscle and platelet actin: Implications for actin sequestration in resting platelets. Biochemistry 1992, 31, 6179–6185. [Google Scholar] [CrossRef]
- Shuster, C.B.; Lin, A.Y.; Nayak, R.; Herman, I.M. β cap73: A novel beta actin-specific binding protein. Cell Motil. Cytoskeleton. 1996, 35, 175–187. [Google Scholar] [CrossRef]
- Yao, X.; Cheng, L.; Forte, J.G. Biochemical characterization of ezrin-actin interaction. J. Biol. Chem. 1996, 271, 7224–7229. [Google Scholar] [CrossRef]
- De La Cruz, E.M. Cofilin binding to muscle and non-muscle actin filaments: Isoform-dependent cooperative interactions. J. Mol. Biol. 2005, 346, 557–564. [Google Scholar] [CrossRef]
- Lechuga, S.; Baranwal, S.; Li, C.; Naydenov, N.G.; Kuemmerle, J.F.; Dugina, V.; Chaponnier, C.; Ivanov, A.I. Loss of γ-cytoplasmic actin triggers myofibroblast transition of human epithelial cells. Mol. Biol. Cell 2014, 25, 3133–3146. [Google Scholar] [CrossRef]
- Yamashiro, S.; Gokhin, D.S.; Sui, Z.; Bergeron, S.E.; Rubenstein, P.A.; Fowler, V.M. Differential actin-regulatory activities of Tropomodulin1 and Tropomodulin3 with diverse tropomyosin and actin isoforms. J. Biol. Chem. 2014, 289, 11616–11629. [Google Scholar] [CrossRef] [PubMed]
- Dugina, V.; Khromova, N.; Rybko, V.; Blizniukov, O.; Shagieva, G.; Chaponnier, C.; Kopnin, B.; Kopnin, P. Tumor promotion by gamma and suppression by beta non-muscle actin isoforms. Oncotarget 2015, 6, 14556–14571. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.A.; Oztug Durer, Z.A.; van Loon, A.P.; Bremer, K.V.; Quinlan, M.E. Drosophila and human FHOD family formin proteins nucleate actin filaments. J. Biol. Chem. 2018, 293, 532–540. [Google Scholar] [CrossRef] [PubMed]
- Hynes, R.O. Stretching the boundaries of extracellular matrix research. Nat. Rev. Mol. Cell Boil. 2014, 15, 761–763. [Google Scholar] [CrossRef] [PubMed]
- Byron, A.; Humphries, J.D.; Humphries, M.J. Defining the extracellular matrix using proteomics. Int. J. Exp. Pathol. 2013, 94, 75–92. [Google Scholar] [CrossRef] [PubMed]
- Hansen, N.U.; Genovese, F.; Leeming, D.J.; Karsdal, M.A. The importance of extracellular matrix for cell function and in vivo likeness. Exp. Mol. Pathol. 2015, 98, 286–294. [Google Scholar] [CrossRef] [PubMed]
- Scott-Burden, T. Extracellular matrix: The cellular environment. Physiology 1994, 9, 110–115. [Google Scholar] [CrossRef]
- Curtis, M.W.; Russell, B. Micromechanical regulation in cardiac myocytes and fibroblasts: Implications for tissue remodeling. Pflug. Arch. 2011, 462, 105–117. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Goldsmith, E.C.; Borg, T.K. The dynamic interaction of the extracellular matrix in cardiac remodeling. J. Card. Fail. 2002, 8, 314–318. [Google Scholar] [CrossRef] [PubMed]
- Bowers, S.L.; Baudino, T.A. Cardiac myocyte-fibroblast interactions and the coronary vasculature. J. Cardiovasc. Trans. Res. 2012, 5, 783–793. [Google Scholar] [CrossRef] [PubMed]
- Jugdutt, B.I. Remodeling of the myocardium and potential targets in the collagen degradation and synthesis pathways. Curr. Drug Targets Cardiovasc. Haematol. Disord. 2003, 3, 1–30. [Google Scholar] [CrossRef] [PubMed]
- Kassiri, Z.; Khokha, R. Myocardial extracellular matrix and its regulation by metalloproteinases and their inhibitors. Thromb. Haemost. 2005, 93, 212–219. [Google Scholar] [CrossRef] [PubMed]
- DeSouza, R.R. Aging of myocardial collagen. Biogerontology 2002, 3, 325–335. [Google Scholar] [CrossRef]
- Borg, T.K.; Goldsmith, E.C.; Price, R.; Carver, W.; Terracio, L.; Samarel, A.M. Specialization at the Z line of cardiac myocytes. Cardiovasc. Res. 2000, 46, 277–285. [Google Scholar] [CrossRef]
- Tuzlakoglu, K.; Santos, M.I.; Neves, N.; Reis, R.L. Design of nano- and microfiber combined scaffolds by electrospinning of collagen onto starch-based fiber meshes: A man-made equivalent of natural extracellular matrix. Tissue Eng. Part A 2011, 17, 463–473. [Google Scholar] [CrossRef] [PubMed]
- Marijianowski, M.M.H.; van der Loos, C.M.; Mohrschladt, M.F.; Becker, A.E. The neonatal heart has a relatively high content of total collagen and type I collagen, a condition that may explain the less compliant state. J. Am. Coll. Cardiol. 1994, 23, 1204–1208. [Google Scholar] [CrossRef]
- Wagenseil, J.E.; Ciliberto, C.H.; Knutsen, R.H.; Levy, M.A.; Kovacs, A.; Mecham, R.P. The importance of elastin to aortic development in mice. Am. J. Physiol. Heart Circ. Physiol. 2010, 299, 257–264. [Google Scholar] [CrossRef]
- Langer, G.A.; Frank, J.S.; Nudd, L.M.; Seraydarian, K. Sialic acid: Effect of removal on calcium exchangeability of cultured heart cells. Science 1976, 193, 1013–1015. [Google Scholar] [CrossRef]
- Lundgren, E.; Gullberg, D.; Rubin, K.; Borg, T.K.; Terracio, M.J.; Terracio, L. In vitro studies on adult cardiac myocytes: Attachment and biosynthesis of collagen type IV and laminin. J. Cell. Physiol. 1988, 136, 43–53. [Google Scholar] [CrossRef]
- Farhadian, F.; Contard, F.; Sabri, A.; Samuel, J.L.; Rappaport, L. Fibronectin and basement membrane in cardiovascular organogenesis and disease pathogenesis. Cardiovasc. Res. 1996, 32, 433–442. [Google Scholar] [CrossRef]
- Yurchenco, P.D.; Amenta, P.S.; Patton, B.L. Basement membrane assembly, stability and activities observed through a developmental lens. Matrix Biol. 2004, 22, 521–538. [Google Scholar] [CrossRef] [PubMed]
- Leivo, I.; Vaheri, A.; Timpl, R.; Wartiovaara, J. Appearance and distribution of collagens and laminin in the early mouse embryo. Dev. Biol. 1980, 76, 100–114. [Google Scholar] [CrossRef]
- Eghbali, M. Cardiac fibroblasts: Function, regulation of gene expression, and phenotypic modulation. Basic Res. Cardiol. 1992, 87, 183–189. [Google Scholar] [PubMed]
- Kurazumi, H.; Kubo, M.; Ohshima, M.; Yamamoto, Y.; Takemoto, Y.; Suzuki, R.; Ikenaga, S.; Mikamo, A.; Udo, K.; Hamano, K.; et al. The effects of mechanical stress on the growth, differentiation, and paracrine factor production of cardiac stem cells. PLoS ONE 2011, 6, 28890. [Google Scholar] [CrossRef]
- Nakagawa, M.; Terracio, L.; Carver, W.; Birkedal-Hanse, H.; Borg, T.K. Expression of collagenase and IL-la in developing rat hearts. Dev. Dyn. 1992, 195, 87–99. [Google Scholar] [CrossRef]
- Weis, S.M.; Emery, J.L.; Becker, K.D.; McBride, D.J.; Omens, J.H.; McCulloch, A.D. Myocardial Mechanics and Collagen Structure in the Osteogenesis Imperfecta Murine (OIM). Circ. Res. 2000, 87, 663–669. [Google Scholar] [CrossRef]
- Liu, X.; Wu, H.; Byrne, M.; Krane, S.; Jaenisch, R. Type III collagen is crucial for collagen I fibrillogenesis and for normal cardiovascular development. Proc. Natl. Acad. Sci. USA 1997, 94, 1852–1856. [Google Scholar] [CrossRef]
- George, E.L.; Georges-Labouesse, E.N.; Patel-King, R.S.; Rayburn, H.; Hynes, R.O. Defects in mesoderm, neural tube and vascular development in mouse embryos lacking fibronectin. Development 1993, 119, 1079–1091. [Google Scholar]
- Li, D.Y.; Brooke, B.; Davis, E.C.; Mecham, R.P.; Sorensen, L.K.; Boak, B.B.; Eichwald, E.; Keating, M.T. Elastin is an essential determinant of arterial morphogenesis. Nature 1998, 393, 276–280. [Google Scholar] [CrossRef]
- Pöschl, E.; Schlötzer-Schrehardt, U.; Brachvogel, B.; Saito, K.; Ninomiya, Y.; Mayer, U. Collagen IV is essential for basement membrane stability but dispensable for initiation of its assembly during early development. Development 2004, 131, 1619–1628. [Google Scholar] [CrossRef]
- Ichikawa-Tomikawa, N.; Ogawa, J.; Douet, V.; Xu, Z.; Kamikubo, Y.; Sakurai, T.; Kohsaka, S.; Chiba, H.; Hattori, N.; Yamada, Y.; et al. Laminin α1 is essential for mouse cerebellar development. Matrix Biol. 2012, 31, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Thyboll, J.; Kortesmaa, J.; Cao, R.; Soininen, R.; Wang, L.; Iivanainen, A.; Sorokin, L.; Risling, M.; Cao, Y.; Tryggvason, K. Deletion of the laminin alpha4 chain leads to impaired microvessel maturation. Mol. Cell. Biol. 2002, 22, 1194–1202. [Google Scholar] [CrossRef] [PubMed]
- Marjamaa, J.; Tulamo, R.; Abo-Ramadan, U.; Hakovirta, H.; Frösen, J.; Rahkonen, O.; Niemelä, M.; Bornstein, P.; Penttinen, R.; Kangasniemi, M. Mice with a deletion in the first intron of the Col1a1 gene develop dissection and rupture of aorta in the absence of aneurysms: High-resolution magnetic resonance imaging, at 4.7 T, of the aorta and cerebral arteries. Magn. Reson. Med. 2006, 55, 592–597. [Google Scholar] [CrossRef]
- Rahkonen, O.; Su, M.; Hakovirta, H.; Koskivirta, I.; Hormuzdi, S.G.; Vuorio, E.; Bornstein, P.; Penttinen, R. Mice with a deletion in the first intron of the Col1a1 gene develop age-dependent aortic dissection and rupture. Circ. Res. 2004, 94, 83–90. [Google Scholar] [CrossRef]
- Lohler, J.; Timpl, R.; Jaenisch, R. Embryonic lethal mutation in mouse collagen I gene causes rupture of blood vessels and is associated with erythropoietic and mesenchymal cell death. Cell 1984, 38, 597–607. [Google Scholar] [CrossRef]
- Wenstrup, R.J.; Florer, J.B.; Brunskill, E.W.; Bell, S.M.; Chervoneva, I.; Birk, D.E. Type V collagen controls the initiation of collagen fibril assembly. J. Biol. Chem. 2004, 279, 53331–53337. [Google Scholar] [CrossRef]
- Lincoln, J.; Florer, J.B.; Deutsch, G.H.; Wenstrup, R.J.; Yutzey, K.E. ColVa1 and ColXIa1 are required for myocardial morphogenesis and heart valve development. Dev. Dyn. 2006, 235, 3295–3305. [Google Scholar] [CrossRef]
- Eklund, L.; Piuhola, J.; Komulainen, J.; Sormunen, R.; Ongvarrasopone, C.; Fassler, R.; Muona, A.; Ilves, M.; Ruskoaho, H.; Takala, T.E.; et al. Lack of type XV collagen causes a skeletal myopathy and cardiovascular defects in mice. Proc. Natl. Acad. Sci. USA 2001, 98, 1194–1199. [Google Scholar] [CrossRef]
- Muona, A.; Eklund, L.; Vaisanen, T.; Pihlajaniemi, T. Developmentally regulated expression of type XV collagen correlates with abnormalities in Col15a1−/− mice. Matrix Biol. 2002, 21, 89–102. [Google Scholar] [CrossRef]
- Utriainen, A.; Sormunen, R.; Kettunen, M.; Carvalhaes, L.S.; Sajanti, E.; Eklund, L.; Kauppinen, R.; Kitten, G.T.; Pihlajaniemi, T. Structurally altered basement membranes and hydrocephalus in a type XVIII collagen deficient mouse line. Hum. Mol. Genet. 2004, 3, 2089–2099. [Google Scholar] [CrossRef]
- Georges-Labouesse, E.N.; George, E.L.; Rayburn, H.; Hynes, R.O. Mesodermal development in mouse embryos mutant for fibronectin. Dev. Dyn. 1996, 207, 145–156. [Google Scholar] [CrossRef]
- Astrof, S.; Crowley, D.; George, E.L.; Fukuda, T.; Sekiguchi, K.; Hanahan, D.; Hynes, R.O. Direct test ofpotential roles of EIIIA and EIIIB alternatively spliced segments of fibronectin in physiological and tumor angiogenesis. Mol. Cell. Biol. 2004, 24, 8662–8670. [Google Scholar] [CrossRef] [PubMed]
- Astrof, S.; Crowley, D.; Hynes, R.O. Multiple cardiovascular defects caused by the absence of alternatively spliced segments of fibronectin. Dev. Biol. 2007, 311, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Faury, G.; Pezet, M.; Knutsen, R.H.; Boyle, W.A.; Heximer, S.P.; MacLean, S.E.; Minkes, R.K.; Blumer, K.J.; Kovacs, A.; Kelly, D.P.; et al. Developmental adaptation of the mouse cardiovascular system to elastin haploinsufficiency. J. Clin. Investig. 2003, 112, 1419–1428. [Google Scholar] [CrossRef] [PubMed]
- Alpy, F.; Jivkov, I.; Sorokin, L.; Klein, A.; Arnold, C.; Huss, Y.; Kedinger, M.; Simon-Assmann, P.; Lefebvre, O. Generation of a conditionally null allele of the laminin α1 gene. Genesis 2005, 43, 59–70. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Hoshijima, M.; Lam, J.; Zhou, Z.; Jokiel, A.; Dalton, N.D.; Hultenby, K.; Ruiz-Lozano, P.; Ross, J., Jr.; Tryggvason, K.; et al. Cardiomyopathy associated with microcirculation dysfunction in laminin α4 chain-deficient mice. J. Biol. Chem. 2006, 281, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Borg, T.K.; Thompson, R.R.; Fitzharris, T.P.; Terracio, L.; Carver, W. Extracellular matrix: Nature vs. nurture in cardiac development. In Developmental Cardiology: Morphogenesis and Function; Clark, E., Takao, A., Eds.; Futura Press: New York, NY, USA, 1999. [Google Scholar]
- Farhadian, F.; Barrieux, A.; Lortet, S.; Marotte, F.; Oliviero, P.; Rappaport, L.; Samuel, J.L. Differential splicing of fibronectin pre-messenger ribonucleic acid during cardiac ontogeny and development of hypertrophy in the rat. Lab. Investig. 1994, 71, 552–559. [Google Scholar]
- Dixon, I.M.C.; Ju, H. The cardiac extracellular matrix during development. In The Developing Heart; Ostadal, B., Nagano, M., Takeda, N., Dhalla, N.S., Eds.; Lippincott-Raven: Philadelphia, PA, USA, 1997; pp. 81–90. [Google Scholar]
- Samuel, J.L.; Farhadian, F.; Sabri, A.; Marotte, F.; Roberts, V.; Rappaport, L. Expression of fibronectin during fetal aorta and postnatal development: An in situ hybridization and immunohistochemical study. Cardiovasc. Res. 1994, 28, 1653–1661. [Google Scholar] [CrossRef] [PubMed]
- Hanson, K.P.; Jung, J.P.; Tran, Q.A.; Hsu, S.-P.P.; Iida, R.; Ajeti, V.; Campagnola, P.J.; Eliceiri, K.W.; Squirrell, J.M.; Lyons, G.E.; et al. Spatial and temporal analysis of extracellular matrix proteins in the developing murine heart: A blueprint for regeneration. Tissue Eng. Part A 2013, 19, 1132–1143. [Google Scholar] [CrossRef]
- Thompson, R.P.; Fitzharris, T.P.; Denslow, S.; LeRoy, E.C. Collagen synthesis in the developing chick heart. Tex. Rep. Biol. Med. 1979, 39, 305–319. [Google Scholar]
- Borg, T.K. Development of the connective tissue network in the neonatal hamster heart. Anat. Rec. 1982, 165, 435–444. [Google Scholar] [CrossRef] [PubMed]
- Engelmann, G.L. Coordinate gene expression during neonatal rat heartdevelopment. A possible role for the myocyte in extracellular matrix biogenesis andcapillary angiogenesis. Cardiovasc. Res. 1993, 27, 1598–1605. [Google Scholar] [CrossRef] [PubMed]
- González, A.; López, B.; Ravassa, S.; José, G.S.; Díez, J. The complex dynamics of myocardial interstitial fibrosis in heart failure. Focus on collagen cross-linking. BBA Mol. Cell. Res. 2019, 1866, 1421–1432. [Google Scholar]
- Majkut, S.; Idema, T.; Swift, J.; Krieger, C.; Liu, A.; Discher, D.E. Heart-specific stiffening in early embryos parallels matrix and myosin expression to optimize beating. Curr. Biol. 2013, 23, 2434–2439. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Yoon, C.S.; Kim, H.; Rah, B. Expression of extracellular matrix components fibronectin and laminin in the human fetal heart. Cell Struct. Funct. 1999, 24, 19–26. [Google Scholar] [CrossRef]
- Price, R.L.; Nakagawa, M.; Terracio, L.; Borg, T.K. Ultrastructural localization of laminin on in vivo embryonic, neonatal, and adult rat cardiac myocytes and in early rat embryos raised in whole embryo culture. J. Histochem. Cytochem. 1992, 40, 1373–1381. [Google Scholar] [CrossRef]
- Tan, G.; Shim, W.; Gu, Y.; Qian, L.; Chung, Y.Y.; Lim, S.Y.; Yong, P.; Sim, E.; Wong, P. Differential effect of myocardial matrix and integrins on cardiac differentiation of human mesenchymal stem cells. Differentiation 2010, 79, 260–271. [Google Scholar] [CrossRef]
- Baharvand, H.; Azarnia, M.; Parivar, K.; Ashtiani, S.K. The effect of extracellular matrix on embryonic stem cell–derived cardiomyocytes. J. Mol. Cell. Cardiol. 2005, 38, 495–503. [Google Scholar] [CrossRef]
- Higuchi, S.; Lin, Q.; Wang, J.; Lim, T.K.; Joshi, S.B.; Anand, G.S.; Chung, M.C.; Sheetz, M.P.; Fujita, H. Heart extracellular matrix supports cardiomyocyte differentiation of mouse embryonic stem cells. J. Biosci. Bioeng. 2013, 115, 320–325. [Google Scholar] [CrossRef]
- Baghalishahi, M.; Efthekhar-Vaghefi, S.H.; Piryaei, A.; Nematolahi-Mahani, S.N.; Mollaei, H.R.; Sadeghi, Y. Cardiac extracellular matrix hydrogel together with or without inducer cocktail improves human adipose tissue-derived stem cells differentiation into cardiomyocyte-like cells. Biochem. Biophys. Res. Commun. 2018, 502, 215–225. [Google Scholar] [CrossRef]
- Fong, A.H.; Romero-López, M.; Heylman, C.M.; Keating, M.; Tran, D.; Sobrino, A.; Tran, A.Q.; Pham, H.H.; Fimbres, C.; Gershon, P.D.; et al. Three-Dimensional Adult Cardiac Extracellular Matrix Promotes Maturation of Human Induced Pluripotent Stem Cell-Derived Cardiomyocytes. Tissue Eng. Part A 2016, 22, 1016–1025. [Google Scholar] [CrossRef] [PubMed]
- Kong, Y.P.; Carrion, B.; Singh, R.K.; Putnam, A.J. Matrix identity and tractional forces influence indirect cardiac reprogramming. Sci. Rep. 2013, 3, 3474. [Google Scholar] [CrossRef] [PubMed]
- Vanwinkle, W.B.; Snuggs, M.B.; Buja, L.M. Cardiogel: A biosynthetic extracellular matrix for cardiomyocyte culture. In Vitro Cell. Dev. Biol. Anim. Soc. 1996, 32, 478–485. [Google Scholar] [CrossRef]
- Santhakumar, R.; Vidyasekar, P.; Verma, R.S. Cardiogel: A nano-matrix scaffold with potential application in cardiac regeneration using mesenchymal stem cells. PLoS ONE 2014, 9, 114697. [Google Scholar] [CrossRef] [PubMed]
- Guyette, J.P.; Charest, J.M.; Mills, R.W.; Jank, B.J.; Moser, P.T.; Gilpin, S.E.; Gershlak, J.R.; Okamoto, T.; Gonzalez, G.; Milan, D.J.; et al. Bioengineering Human Myocardium on Native Extracellular Matrix. Circ. Res. 2016, 118, 56–72. [Google Scholar] [CrossRef] [PubMed]
- Duan, Y.; Liu, Z.; O’Neill, J.; Wan, L.Q.; Freytes, D.O.; Vunjak-Novakovic, G. Hybrid gel composed of native heart matrix and collagen induces cardiacdifferentiation of human embryonic stem cells without supplemental growth factors. J. Cardiovasc. Trans. Res. 2011, 4, 605–615. [Google Scholar] [CrossRef] [PubMed]
- Subbiah, R.; Hwang, M.P.; Du, P.; Suhaeri, M.; Hwang, J.-H.; Hong, J.H.; Park, K. Tunable crosslinked cell-derived extracellular matrix guides cell fate. Macromol. Biosci. 2016, 16, 1723–1734. [Google Scholar] [CrossRef]
- Williams, C.; Budina, E.; Stoppel, W.L.; Sullivan, K.E.; Emani, S.; Emani, S.M.; Black, L.D., III. Cardiac extracellular matrix-fibrin hybrid scaffolds with tunable properties for cardiovascular tissue engineering. Acta Biomater. 2015, 14, 84–95. [Google Scholar] [CrossRef]
- Grinnell, F. Cellular adhesiveness and extracellular substrata. Int. Rev. Cytol. 1978, 53, 65–144. [Google Scholar] [PubMed]
- Petukhova, O.A.; Turoverova, L.V.; Kropacheva, I.V.; Pinaev, G.P. Morphology of epidermoid carcinoma A431 cells spread on immobilized ligands. Tsitologiia 2004, 46, 5–15. [Google Scholar]
- Aré, A.F.; Pospelova, T.V.; Pinaev, G.P. The characteristics of actin cytoskeleton structure and its rearrangements by extracellular matrix proteins in normal, immortalized and transformed rat fibroblasts. Tsitologiia 1999, 41, 707–715. [Google Scholar]
- Starke, J.; Wehrle-Haller, B. Plasticity of the actin cytoskeleton in response to extracellular matrix nanostructure and dimensionality. Biochem. Soc. Trans. 2014, 42, 1356–1366. [Google Scholar] [CrossRef]
- Claycomb, W.C.; Delcarpio, J.B.; Guice, S.E.; Moses, R.L. Culture and characterization of fetal human atrial and ventricular cardiac muscle cells. In Vitro Cell. Dev. Boil. 1989, 25, 1114–1120. [Google Scholar] [CrossRef]
- Bick, R.J.; Snuggs, M.B.; Poindexter, B.J.; Buja, L.M.; Van Winkle, W.B. Physical, contractile and calcium handling properties of neonatal cardiac myocytes cultured on different matrices. Cell Adhes. Commun. 1998, 6, 301–310. [Google Scholar] [CrossRef]
- Borg, T.K.; Rubin, K.; Lundgren, E.; Borg, K.; Obrink, B. Recognition of extracellular matrix components by neonatal and adult cardiac myocytes. Dev. Biol. 1984, 104, 86–96. [Google Scholar] [CrossRef]
- Lundgren, E.; Terracio, L.; Mårdh, S.; Borg, T.K. Extracellular matrix components influence the survival of adult cardiac myocytes in vitro. Exp. Cell Res. 1985, 158, 371–381. [Google Scholar] [CrossRef]
- Terracio, L.; Rubin, K.; Gullberg, D.; Balog, E.; Carver, W.; Jyring, R.; Borg, T. Expression of collagen binding integrins during cardiac development and hypertrophy. Circ. Res. 1991, 68, 734–744. [Google Scholar] [CrossRef]
- Borg, T.K.; Terracio, L. Cellular adhesion to artificial substrates and long term culture of adult cardiac myocytes. In Biology of Isolated Adult Cardiac Myocytes; Clark, W.A., Decker, R.S., Borg, T.K., Eds.; Elsevier Science Publishing Co, Inc.: New York, NY, USA, 1988; pp. 14–24. [Google Scholar]
- Borg, T.K.; Johnson, L.D.; Lill, P.H. Specific attachment of collagen to cardiac myocytes: In vivo and in vitro. Dev. Biol. 1983, 97, 417–423. [Google Scholar] [CrossRef]
- Engler, A.J.; Carag-Krieger, C.; Johnson, C.P.; Raab, M.; Tang, H.Y.; Speicher, D.W.; Sanger, J.W.; Sanger, J.M.; Discher, D.E. Embryonic cardiomyocytes beat best on a matrix with heartlike elasticity: Scar-like rigidity inhibits beating. J. Cell Sci. 2008, 121, 3794–3802. [Google Scholar] [CrossRef]
- Bhana, B.; Iyer, R.K.; Chen, W.L.; Zhao, R.; Sider, K.L.; Likhitpanichkul, M.; Simmons, C.A.; Radisic, M. Influence of substrate stiffness on the phenotype of heart cells. Biotechnol. Bioeng. 2010, 105, 1148–1160. [Google Scholar] [CrossRef]
- Jacot, J.G.; McCulloch, A.D.; Omens, J.H. Substrate stiffness affects the functional maturation of neonatal rat ventricular myocytes. Biophys. J. 2008, 95, 3479–3487. [Google Scholar] [CrossRef]
- Hynes, R.O. Integrins: Versatility, modulation, and signaling in cell adhesion. Cell 1992, 69, 11–25. [Google Scholar] [CrossRef]
- Borg, T.K.; Rubin, K.; Carver, W.; Samarel, A.M.; Terracio, L. The cell biology of the cardiac interstitium. Trends Cardiovasc. Med. 1996, 6, 65–70. [Google Scholar] [CrossRef]
- Hemler, M. Integrin associated proteins. Curr. Opin. Biol. 1999, 10, 585–587. [Google Scholar] [CrossRef]
- Ko, K.S.; McCulloch, C.A. Intercellular mechanotransduction: Cellular circuits that coordinate tissue responses to mechanical loading. Biochem. Biophys. Res. Commun. 2001, 285, 1077–1083. [Google Scholar] [CrossRef]
- Huang, H.; Kamm, R.D.; Lee, R.T. Cell mechanics and mechanotransduction: Pathways, probes, and physiology. Am. J. Physiol. Cell Physiol. 2004, 287, 1–11. [Google Scholar] [CrossRef]
- Clark, E.A.; Brugge, J.S. Integrins and signal transduction pathways: The road taken. Science 1995, 268, 233–239. [Google Scholar] [CrossRef]
- Schoenwaelder, S.M.; Burridge, K. Bidirectional signaling between the cytoskeleton and integrins. Curr. Opin. Cell Boil. 1999, 11, 274–286. [Google Scholar] [CrossRef]
- Liu, S.; Calderwood, D.A.; Ginsberg, M.H. Integrin cytoplasmic domain binding proteins. J. Cell Sci. 2000, 113, 3563–3571. [Google Scholar]
- Millward-Sadler, S.J.; Wright, M.O.; Lee, H.; Nishida, K.; Caldwell, H.; Nuki, G.; Salter, D.M. Integrin-regulated secretion of interleukin 4: A novel pathway of mechanotransduction in human articular chondrocytes. J. Cell Biol. 1999, 145, 183–189. [Google Scholar] [CrossRef]
- Gupta, V.; Grande-Allen, K.J. Effects of static and cyclic loading in regulating extracellular matrix synthesis by cardiovascular cells. Cardiovasc. Res. 2006, 72, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Holledge, M.M.; Millward-Sadler, S.J.; Nuki, G.; Salter, D.M. Mechanical regulation of proteoglycan synthesis in normal and osteoarthritic human articular chondrocytes—Roles for α5 and αVβ5 integrins. Biorheology 2008, 45, 275–288. [Google Scholar] [PubMed]
- Eble, D.; Spinale, F. Contractile and cytoskeletal content, structure, and mRNA levels with tachycardia–induced cardiomyopathy. Am. J. Physiol. 1995, 268, 2426–2439. [Google Scholar] [CrossRef] [PubMed]
- Janmey, P. The cytoskeleton and cell signalling: Component localization and mechanical coupling. Physiol. Rev. 1998, 78, 763–781. [Google Scholar] [CrossRef] [PubMed]
- Belkin, A.M.; Zhidkova, N.; Balzac, F.; Altruda, F.; Tomatis, D.; Maier, A.; Tarone, G.; Koteliansky, V.E.; Burridge, K. β1D integrin displaces the B1A isoform in striated muscles: Localization at junctional structures and signaling potential in non-muscle cells. J. Cell Biol. 1996, 132, 211–266. [Google Scholar] [CrossRef] [PubMed]
- Fassler, R.; Rohwedel, J.; Maltsev, V.; Bloch, W.; Lentini, S.; Guan, K.; Gullberg, D.; Hescheler, J.; Addicks, K.; Wobus, A.M. Differentiation and integrity of cardiac muscle cells are impaired in the absence of β1 integrin. J. Cell Sci. 1996, 109, 2989–2999. [Google Scholar] [PubMed]
- Belkin, A.M.; Retta, F.; Pletjushkina, O.Y.; Balzac, F.; Silengo, L.; Fassler, R.; Kotenliansky, V.E.; Burridge, K.; Tarone, G. Muscle β1D integrin reinforces the cytoskeleton-matrix link: Modulation of integrin adhesive function by alternative splicing. J. Cell Biol. 1997, 139, 1583–1595. [Google Scholar] [CrossRef] [PubMed]
- Soto-Ribeiro, M.; Kastberger, B.; Bachmann, M.; Azizi, L.; Fouad, K.; Jacquier, M.C.; Boettiger, D.; Bouvard, D.; Bastmeyer, M.; Hytönen, V.P.; et al. β1D integrin splice variant stabilizes integrin dynamics and reduces integrin signaling by limiting paxillin recruitment. J. Cell Sci. 2019, 132. [Google Scholar] [CrossRef]
- McCain, M.L.; Parker, K.K. Mechanotransduction: The role of mechanical stress, myocyte shape, and cytoskeletal architecture on cardiac function. Pflug. Arch. 2011, 462, 89–104. [Google Scholar] [CrossRef] [PubMed]
- Van der Flier, A.; Gaspar, A.C.; Thorsteinsdottir, S.; Baudoin, C.; Groeneveld, E.; Mummery, C.L.; Sonnenberg, A. Spatial and temporal expression of the beta1d integrin during mouse development. Dev. Dyn. 1997, 210, 472–486. [Google Scholar] [CrossRef]
- Brancaccio, M.; Cabodi, S.; Belkin, A.M.; Collo, G.; Koteliansky, V.E.; Tomatis, D.; Altruda, F.; Silengo, L.; Tarone, G. Differential onset of expression of α7 and β1d integrins during mouse heart and skeletal muscle development. Cell Adhes. Commun. 1998, 5, 193–205. [Google Scholar] [CrossRef]
- Collo, G.; Domanico, S.Z.; Klier, G.; Quaranta, V. Gradient of integrin α6A distribution in the myocardium during early heart development. Cell Adhes. Commun. 1995, 3, 101–113. [Google Scholar] [CrossRef]
- Hierck, B.P.; Poelmann, R.E.; van Iperen, L.; Brouwer, A.; Gittenberger-De Groot, A.C. Differential expression of α6 and other subunits of laminin binding integrins during development of the murine heart. Dev. Dyn. 1996, 206, 100–111. [Google Scholar] [CrossRef]
- Hilenski, L.L. The role of β1 integrin in spreading and myofibrillogenesis in neonatal rat cardiomyocytes in vitro. Cell Motil. Cytoskeleton. 1992, 21, 87–100. [Google Scholar] [CrossRef]
- Tadokoro, S.; Shattil, S.J.; Eto, K.; Tai, V.; Liddington, R.C.; de Pereda, J.M.; Ginsberg, M.H.; Calderwood, D.A. Talin binding to integrin beta tails: A final common step in integrin activation. Science 2003, 302, 103–106. [Google Scholar] [CrossRef]
- Otey, C.A.; Pavalko, F.M.; Burridge, K. An interaction between α-actinin and the β1 integrin subunit in vitro. J. Cell Biol. 1990, 111, 721–729. [Google Scholar] [CrossRef]
- Katsumi, A.; Orr, A.W.; Tzima, E.; Schwartz, M.A. Integrins in mechanotransduction. J. Biol. Chem. 2004, 279, 12001–12004. [Google Scholar] [CrossRef]
- Craig, S.W.; Johnson, R.P. Assembly of focal adhesions: Progress, paradigms, and portents. Curr. Opin. Cell Biol. 1996, 8, 74–85. [Google Scholar] [CrossRef]
- Juliano, R.L.; Haskill, S. Signal transduction from the extracellular matrix. J. Cell Biol. 1993, 120, 577–585. [Google Scholar] [CrossRef]
- Guan, J.L.; Shalloway, D. Regulation of focal adhesion-associated protein tyrosine kinase by both cellular adhesion and oncogenic transformation. Nature 1992, 358, 690–692. [Google Scholar] [CrossRef]
- Wary, K.K.; Mainiero, F.; Isakoff, S.J.; Marcantonio, E.E.; Giancotti, F.G. The adaptor protein Shc couples a class of integrins to the control of cell cycle progression. Cell 1996, 87, 733–743. [Google Scholar] [CrossRef]
- Dedhar, S.; Williams, B.; Hannigan, G. Integrin-linked kinase (ILK), a regulator of integrin and growth-factor signaling. Trends Cell Biol. 1999, 9, 319–323. [Google Scholar] [CrossRef]
- Qian, Y.; Zhong, X.; Flynn, D.C.; Zheng, J.Z.; Qiao, M.; Wu, C.; Dedhar, S.; Shi, X.; Jiang, B.H. ILK mediates actin filament rearrangements and cell migration and invasion through PI3K/Akt/Rac1 signaling. Oncogene 2005, 24, 3154–3165. [Google Scholar] [CrossRef]
- Niu, H.; Nie, L.; Liu, M.; Chi, Y.; Zhang, T.; Li, Y. Benazepril affects integrin-linked kinase and smooth muscle α–actin expression in diabetic rat glomerulus and cultured mesangial cells. BMC Nephrol. 2014, 15, 135. [Google Scholar] [CrossRef]
- Li, G.; Li, Y.Y.; Sun, J.E.; Lin, W.H.; Zhou, R.X. ILK-PI3K/AKT pathway participates in cutaneous wound contraction by regulating fibroblast migration and differentiation to myofibroblast. Lab. Investig. 2016, 96, 741–751. [Google Scholar] [CrossRef]
- Bildyug, N. Dynamics of integrin-linked kinase during the rearrangement of contractile apparatus in rat neonatal cardiomyocytes. In Proceedings of the 44th FEBS Congress, Krakow, Poland, 6–11 July 2019; pp. 17–21. [Google Scholar]
- Yano, Y.; Geibel, J.; Sumpio, B.E. Tyrosine phosphorylation of pp125FAK and paxillin in aortic endothelial cells induced by mechanical strain. Am. J. Physiol. 1996, 271, 635–649. [Google Scholar] [CrossRef]
- Ali, M.H.; Mungai, P.T.; Schumacker, P.T. Stretch-induced phosphorylation of focal adhesion kinase in endothelial cells: Role of mitochondrial oxidants. Am. J. Physiol. Lung Cell. Mol. Physiol. 2006, 291, 38–45. [Google Scholar] [CrossRef]
- Young, S.R.; Gerard-O’Riley, R.; Kim, J.B.; Pavalko, F.M. Focal adhesion kinase is important for fluid shear stress–induced mechanotransduction in osteoblasts. J. Bone Miner. Res. 2009, 24, 411–424. [Google Scholar] [CrossRef]
- Lal, H.; Verma, S.K.; Smith, M.; Guleria, R.S.; Lu, G.; Foster, D.M.; Dostal, D.E. Stretch-induced MAP kinase activation in cardiac myocytes: Differential regulation through β1-integrin and focal adhesion kinase. J. Mol. Cell. Cardiol. 2007, 43, 137–147. [Google Scholar] [CrossRef]
- Higashita, R.; Li, L.; Van Putten, V.; Yamamura, Y.; Zarinetchi, F.; Heasley, L.; Nemenoff, R.A. Galpha16 mimics vasoconstrictor action to induce smooth muscle α-actin in vascular smooth muscle cells through a Jun-NH2-terminal kinase-dependent pathway. J. Biol. Chem. 1997, 272, 25845–25850. [Google Scholar] [CrossRef]
- Wang, J.; Chen, H.; Seth, A.; McCulloch, C.A. Mechanical force regulation of myofibroblast differentiation in cardiac fibroblasts. Am. J. Physiol. Heart Circ. Physiol. 2003, 285, 1871–1881. [Google Scholar] [CrossRef]
- Zamir, E.; Geiger, B. Molecular complexity and dynamics of cell-matrix adhesions. J. Cell Sci. 2001, 114, 3583–3590. [Google Scholar]
- Yamada, K.M.; Pankov, R.; Cukierman, E. Dimensions and dynamics in integrin function. Braz. J. Med. Biol. Res. 2003, 36, 959–966. [Google Scholar] [CrossRef]
- Mack, C.P.; Somlyo, A.V.; Hautmann, M.; Somlyo, A.P.; Owens, G.K. Smooth muscle differentiation marker gene expression is regulated by RhoA–mediated actin polymerization. J. Biol. Chem. 2001, 276, 341–347. [Google Scholar] [CrossRef]
- Hall, A. Small GTP-binding proteins and the regulation of the actin cytoskeleton. Annu. Rev. Cell Biol. 1994, 10, 31–54. [Google Scholar] [CrossRef]
- Van Aelst, L.; D’Souza-Schorey, C. Rho GTPases and signaling networks. Genes Dev. 1997, 11, 2295–2322. [Google Scholar] [CrossRef]
- Wei, L.; Wang, L.; Carson, J.A.; Agan, J.E.; Imanaka-Yoshida, K.; Schwartz, R.J. β1 integrin and organized actin filaments facilitates cardiomyocyte-specific RhoA-dependent activation of the skeletal α-actin promoter. FASEB J. 2001, 15, 785–797. [Google Scholar] [CrossRef]
- Haller, K.; Rambaldi, I.; Daniels, E.; Featherstone, M. Subcellular localization of multiple PREP2 isoforms is regulated by actin, tubulin, and nuclear export. J. Biol. Chem. 2004, 279, 49384–49394. [Google Scholar] [CrossRef]
- Németh, Z.H.; Deitch, E.A.; Davidson, M.T.; Szabó, C.; Vizi, E.S.; Haskó, G. Disruption of the actin cytoskeleton results in nuclear factor-kappaB activation and inflammatory mediator production in cultured human intestinal epithelial cells. J. Cell. Physiol. 2004, 200, 71–81. [Google Scholar] [CrossRef]
- Miralles, F.; Posern, G.; Zaromytidou, A.I.; Treisman, R. Actin dynamics control SRF activity by regulation of its coactivator MAL. Cell 2003, 113, 329–342. [Google Scholar] [CrossRef]
- Boxer, L.M.; Prywes, R.; Roeder, R.G.; Kedes, L. The sarcomeric actin CArG-binding factor is indistinguishable from the c-fos serum response factor. Mol. Cell. Biol. 1989, 9, 515–522. [Google Scholar] [CrossRef] [PubMed]
- Chow, K.L.; Schwartz, R.J. A combination of closely associated positive and negative cis-acting promoter elements regulates transcription of the skeletal α-actin gene. Mol. Cell. Biol. 1990, 10, 528–538. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Muscat, G.E.; Kedes, L. Multiple 5′-flanking regions of the human a-skeletal actin gene synergistically modulate muscle-specific expression. Mol. Cell. Biol. 1987, 7, 4089–4099. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Su, M.; Fan, J.; Seth, A.; McCulloch, C.A. Transcriptional regulation of a contractile gene by mechanical forces applied through integrins in osteoblasts. J. Biol. Chem. 2002, 277, 22889–22895. [Google Scholar] [CrossRef] [PubMed]
| Extracellular Matrix Protein | Model | Effect on Cardiovascular System | Reference |
|---|---|---|---|
| Collagen I | Col1a2-deficient mice | impaired heart development, decreased heart weight, altered mechanical and structural properties of the ventricular myocardium | [136] |
| Col1a1-deficient mice | vascular abnormalities, age-dependent aortic dissection and rupture | [143,144] | |
| Col1a1−/− mice | normal development up to embryonic day 12, lethality between embryonic days 12-14 due to rupture of major blood vessels | [145] | |
| Collagen III | Col3a1−/− mice | abnormal cardiac development, life-shortening due to rupture of major blood vessels | [137] |
| Collagen IV | Col4a1/2−/− mice | structural defects in the basement membrane, lethality between embryonic days 10.5-11.5 due to pericardial bleeding and rupture of major blood vessels | [140] |
| Collagen V | Col5a1−/− mice | lethality at embryonic day 10 due to cardiovascular insufficiency | [146] |
| Collagen XI | Col11a1–/– mice | lethality at birth, thickening of the interventricular septum and atrioventricular valve leaflets, significant changes in the heart shape | [147] |
| Col XV | Col15a1−/− mice | defects in vessel architecture, impaired microvascular hemodynamics, defects in heart structure and function | [148,149] |
| Collagen XVIII | Col18a1−/− mice | significant thickening of the endothelial basement membrane in the atrioventricular valves of the heart | [150] |
| Fibronectin | Fn1−/− mice | multiple developmental abnormalities at embryonic day 8, lethality at embryonic day 10 due to cardiac and vascular defects | [138,151] |
| EIIIA−/− or EIIIB−/− mice | normal phenotype, viability, and fertility without defects in angiogenesis | [152] | |
| EIIIA−/− EIIIB−/− mice | severe cardiovascular defects by embryonic day 9.5, including vascular hemorrhage, impaired angiogenesis and heart defects, lethal at embryonic day 10.5 | [153] | |
| Elastin | Eln−/− mice | lethality at day 4.5 of postnatal development due to obstructive arterial disease | [139] |
| Eln+/− mice | changes in the arterial wall structure, high blood pressure | [154] | |
| Laminin | Lama1−/− mice | lethality after embryonic day 6.5 due to defects in the extraembryonic basement membrane | [155] |
| Lama4−/− mice | hemorrhages during the embryonic and neonatal development, impaired microvessel maturation, ischemic cardiac phenotype | [142,156] |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bildyug, N. Extracellular Matrix in Regulation of Contractile System in Cardiomyocytes. Int. J. Mol. Sci. 2019, 20, 5054. https://doi.org/10.3390/ijms20205054
Bildyug N. Extracellular Matrix in Regulation of Contractile System in Cardiomyocytes. International Journal of Molecular Sciences. 2019; 20(20):5054. https://doi.org/10.3390/ijms20205054
Chicago/Turabian StyleBildyug, Natalya. 2019. "Extracellular Matrix in Regulation of Contractile System in Cardiomyocytes" International Journal of Molecular Sciences 20, no. 20: 5054. https://doi.org/10.3390/ijms20205054
APA StyleBildyug, N. (2019). Extracellular Matrix in Regulation of Contractile System in Cardiomyocytes. International Journal of Molecular Sciences, 20(20), 5054. https://doi.org/10.3390/ijms20205054





