Ultrafine Particles from Residential Biomass Combustion: A Review on Experimental Data and Toxicological Response
Abstract
:1. Introduction
2. Characteristics of UFPs Generated by Residential Biomass Combustion
2.1. Ambient Measurements
2.2. Laboratory Scale Experiments
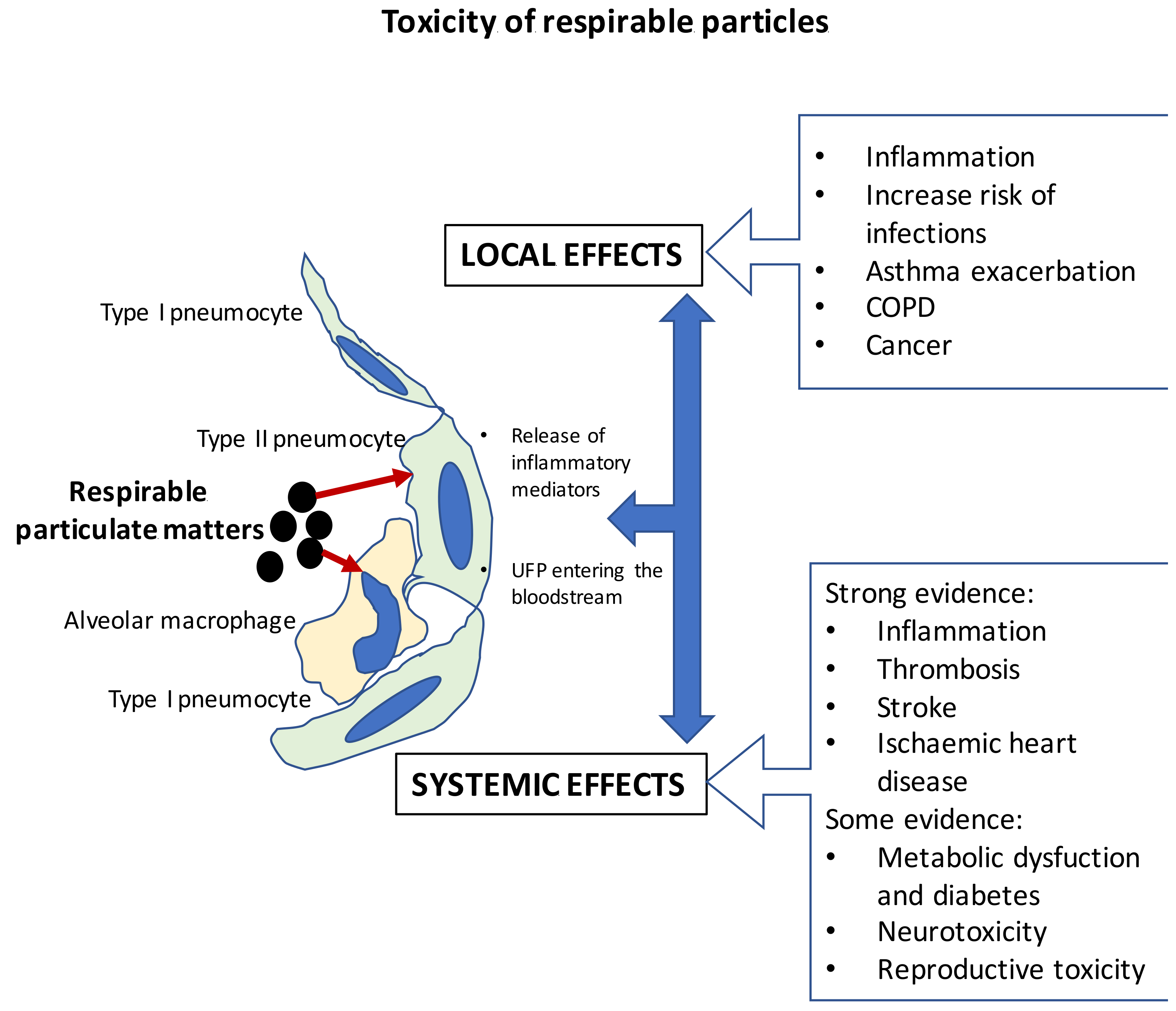
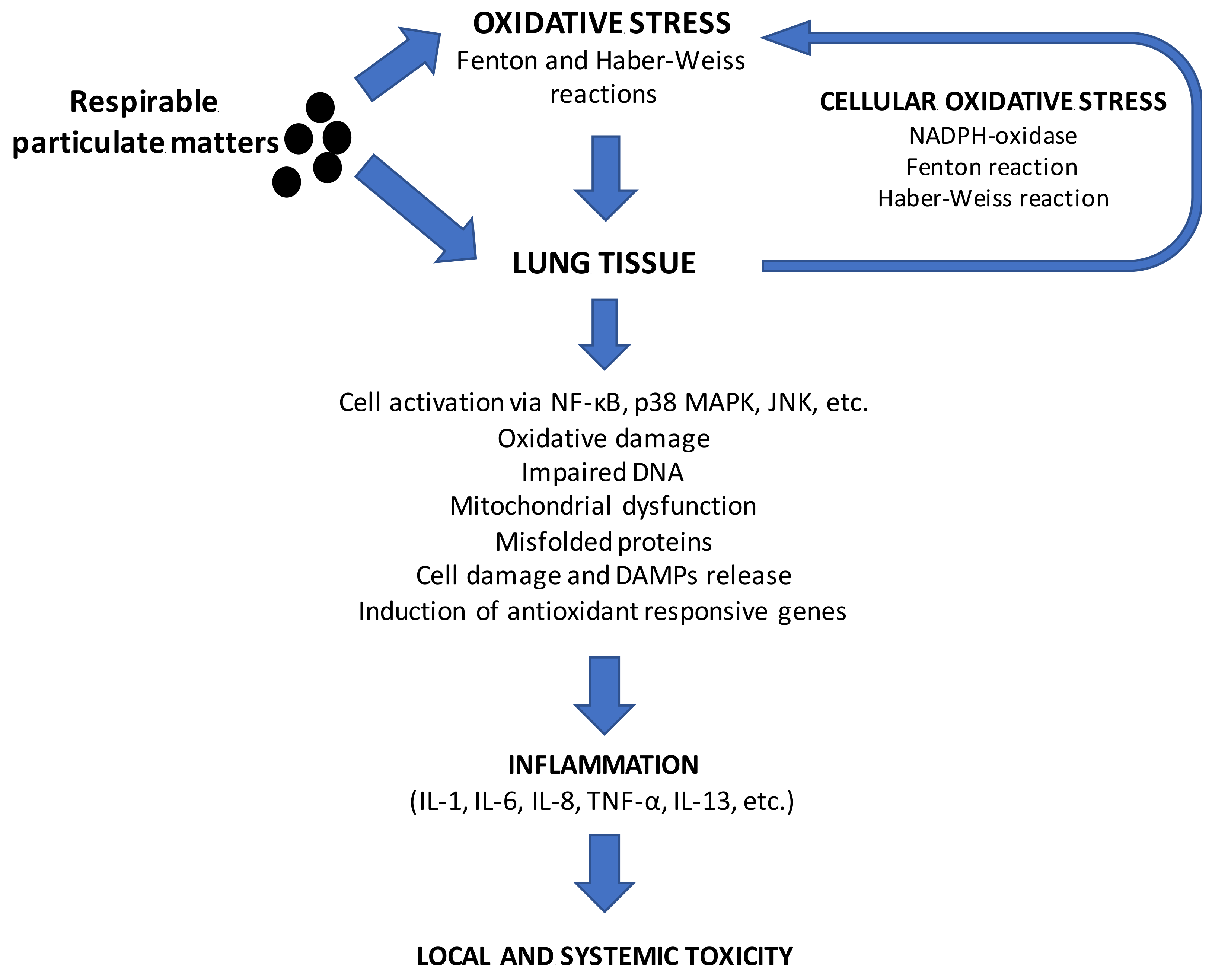
3. Interactions between Particulate Matter Exposure from Biomass and Human Health: In Vivo and In Vitro Effects
Mode of Action
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| IL | interleukin |
| PM | particulate matter |
| UFP | ultrafine particles |
References
- Lelieveld, J.; Münzel, T. Air pollution, chronic smoking, and mortality. Eur. Heart J. 2019, 40, 1590–1596. [Google Scholar] [CrossRef]
- Sigsgaard, T.; Forsberg, B.; Annesi-Maesano, I.; Blomberg, A.; Bølling, A.; Boman, C.; Bønløkke, J.; Brauer, M.; Bruce, N.; Héroux, M.E.; et al. Health impacts of anthropogenic biomass burning in the developed world. Eur. Respir. J. 2015, 46, 1577–1588. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Balmes, J.R. Household air pollution from domestic combustion of solid fuels and health. J. Allergy Clin. Immunol. 2019, 143, 1979–1987. [Google Scholar] [CrossRef]
- Smith, K.R.; Pillarisetti, A. Household Air Pollution from Solid Cookfuels and Its Effects on Health. In Injury Prevention and Environmental Health, 3rd ed.; Mock, C.N., Nugent, R., Kobusingye, O., Smith, K.R., Eds.; The International Bank for Reconstruction and Development/The World Bank: Washington, DC, USA, 2017; Chapter 7. [Google Scholar]
- Vecchi, R.; Bernardoni, V.; Valentini, S.; Piazzalunga, A.; Fermo, P.; Valli, G. Assessment of light extinction at a European. Environ. Pollut. 2018, 233, 679–689. [Google Scholar] [CrossRef]
- Perrino, C.; Tofful, L.; Dalla Torre, S.; Sargolini, T.; Canepari, S. Biomass burning contribution to PM10 concentration in Rome (Italy): Seasonal, daily and two-hourly variations. Chemosphere 2019, 222, 839–848. [Google Scholar] [CrossRef] [PubMed]
- Muntean, M.; Janssens-Maenhout, G.; Guizzardi, D.; Crippa, M.; Schaaf, E.; Poljanac, M.; Logar, M.; Cristea-Gassler, C. Impact Evaluation of Biomass Used in Small Combustion Activities Sector on Air Emissions: Analyses of Emissions From Alpine, Adriatic-Ionian and Danube Eu Macro-Regions by Using the Edgar Emissions Inventory, EUR 29033 EN; Publications Office of the European Union: Luxembourg, 2017; ISBN 978-92-79-77359-4. JRC109332; Available online: https://ec.europa.eu/jrc/en/publication/impact-evaluation-biomass-used-small-combustion-activities-sector-air-emissions-analyses-emissions (accessed on 10 September 2019). [CrossRef]
- Denier van der Gon, H.A.C.; Bergström, R.; Fountoukis, C.; Johansson, C.; Pandis, S.N.; Simpson, D.; Visschedijk, A.J.H. Particulate emissions from residential wood combustion in Europe –revised estimates and an evaluation. Atmos. Chem. Phys. 2015, 15, 6503–6519. [Google Scholar] [CrossRef]
- Amann, M.; Bertok, I.; Cofala, J.; Gyarfas, F.; Zbigniew Klimont, C.-H.; Schöpp, W.; Winiwarter, W. Baseline Scenarios for the Clean Air for Europe (CAFE) Programme; International Institute for Applied Systems Analysis: Laxenburg, Austria, 2005; Available online: http://pure.iiasa.ac.at/id/eprint/7656/ (accessed on 1 August 2019).
- Amann, M. The Final Policy Scenarios of the EU Clean Air Policy Package; International Institute for Applied Systems Analysis: Laxenburg, Austria, 2014; Available online:http://pure.iiasa.ac.at/id/eprint/11153/ (accessed on 1 August 2019).
- Kim, Y.H.; King, C.; Krantz, T.; Hargrove, M.M.; George, I.J.; McGee, J.; Copeland, L.; Hays, M.D.; Landis, M.S.; Higuchi, M.; et al. The role of fuel type and combustion phase on the toxicity of biomass smoke following inhalation exposure in mice. Arch. Toxicol. 2019, 93, 1501–1513. [Google Scholar] [CrossRef] [PubMed]
- Corsini, E.; Budello, S.; Marabini, L.; Galbiati, V.; Piazzalunga, A.; Barbieri, P.; Cozzutto, S.; Marinovich, M.; Pitea, D.; Galli, C.L. Comparison of wood smoke PM2.5 obtained from the combustion of FIR and beech pellets on inflammation and DNA damage in A549 and THP-1 human cell lines. Arch. Toxicol. 2013, 87, 2187–2199. [Google Scholar] [CrossRef] [PubMed]
- Corsini, E.; Ozgen, S.; Papale, A.; Galbiati, V.; Lonati, G.; Fermo, P.; Corbella, L.; Valli, G.; Bernardoni, V.; Dell’Acqua, M.; et al. Insights on wood combustion generated proinflammatory ultrafine particles (UFP). Toxicol. Lett. 2017, 15, 74–84. [Google Scholar] [CrossRef] [PubMed]
- Park, M.; Joo, H.S.; Lee, K.; Jang, M.; Kim, S.D.; Kim, I.; Borlaza, L.J.S.; Lim, H.; Shin, H.; Chung, K.H.; et al. Differential toxicities of fine particulate matters from various sources. Sci. Rep. 2018, 8, 17007. [Google Scholar] [CrossRef] [PubMed]
- Marchetti, S.; Longhin, E.; Bengalli, R.; Avino, P.; Stabile, L.; Buonanno, G.; Colombo, A.; Camatini, M.; Mantecca, P. In vitro lung toxicity of indoor PM10 from a stove fueled with different biomasses. Sci. Total Environ. 2019, 649, 1422–1433. [Google Scholar] [CrossRef] [PubMed]
- Schwarze, P.E.; Øvrevik, J.; Hetland, R.B.; Becher, R.; Cassee, F.R.; Låg, M.; Løvik, M.; Dybing, E.; Refsnes, M. Importance of size and composition of particles for effects on cells in vitro. Inhal Toxicol. 2007, 19, 17–22. [Google Scholar] [CrossRef]
- Gualtieri, M.; Grollino, M.G.; Consales, C.; Costabile, F.; Manigrasso, M.; Avino, P.; Aufderheide, M.; Cordelli, E.; Di Liberto, L.; Petralia, E.; et al. Is it the time to study air pollution effects under environmental conditions? A case study to support the shift of in vitro toxicology from the bench to the field. Chemosphere 2018, 207, 552–564. [Google Scholar] [CrossRef] [PubMed]
- Sippula, O.; Huttunen, K.; Hokkinen, J.; Kärki, S.; Suhonen, H.; Kajolinna, T.; Kortelainen, M.; Karhunen, T.; Jalava, P.; Uski, O.; et al. Emissions from a fast-pyrolysis bio-oil fired boiler: Comparison of health-related characteristics of emissions from bio-oil, fossil oil and wood. Environ. Pollut. 2019, 248, 888–897. [Google Scholar] [CrossRef] [PubMed]
- Naeher, L.P.; Brauer, M.; Lipsett, M.; Zelikoff, J.T.; Simpson, C.D.; Koenig, J.Q.; Smith, K.R. Woodsmoke health effects: A review. Inhal. Toxicol. 2007, 19, 67–106. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Review of Evidence on Health Aspects of Air Pollution—Revihaap Project Technical Report; WHO Regional Office for Europe: Copenhagen, Denmark, 2013. [Google Scholar]
- DeCarlo, P.F.; Kimmel, J.R.; Trimborn, A.; Northway, M.J.; Jayne, J.T.; Aiken, A.C.; Gonin, M.; Fuhrer, K.; Horvath, T.; Docherty, K.S.; et al. Field-deployable, high-resolution, time-of-flight aerosol mass spectrometer. Anal. Chem. 2006, 78, 8281–8289. [Google Scholar] [CrossRef] [PubMed]
- Kulmala, M.; Petäjä, T.; Ehn, M.; Thornton, J.; Sipilä, M.; Worsnop, D.R.; Kerminen, V.M. Advances on precursor characterization and atmospheric cluster composition in connection with atmospheric New particle formation. Annu. Rev. Phys. Chem. 2014, 65, 21–37. [Google Scholar] [CrossRef] [PubMed]
- Dall’Osto, M.; Beddows, D.C.S.; Asmi, A.; Poulain, L.; Hao, L.; Freney, E.; Allan, J.D.; Canagaratna, M.; Crippa, M.; Bianchi, F.; et al. Novel insights on new particle formation derived from a pan-european observing system. Sci. Rep. 2018, 8, 1482. [Google Scholar] [CrossRef] [PubMed]
- Manigrasso, M.; Protano, C.; Vitali, M.; Avino, P. Where do ultrafine particles and nano-sized particles come from? J. Alzheimeir’s Dis. 2019, 68, 1371–1390. [Google Scholar] [CrossRef]
- Schäfer, K.; Thomas, W.; Peters, A.; Ries, L.; Obleitner, F.; Schnelle-Kreis, J.; Birmili, W.; Diemer, J.; Fricke, W.; Junkermann, W.; et al. Influences of the 2010 Eyjafjallajökull volcanic plume on air qualityin the northern Alpine region. Atmos. Chem. Phys. 2011, 11, 8555–8575. [Google Scholar] [CrossRef]
- Morawska, L.; Ristovski, Z.; Jayaratne, E.R.; Keough, D.U.; Ling, X. Ambient nano and ultrafine particles from motor vehicle emissions: Characteristics, ambient processing and implications on human exposure. Atmos. Environ. 2008, 42, 8113–8138. [Google Scholar] [CrossRef] [Green Version]
- Kumar, P.; Morawska, L.; Birmili, W.; Paasonen, P.; Hug, M.; Kulmala, M.; Harrison, R.M.; Norfordj, L.; Britter, R. Ultrafine particles in cities. Environ. Int. 2014, 66, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, P.; Pirjola, L.; Ketzel, M.; Harrison, R.M. Nanoparticle emissions from11 non-vehicle exhaust sources – A review. Atmos. Environ. 2013, 67, 252–277. [Google Scholar] [CrossRef]
- Stacey, B. Measurement of ultrafine particles in airports: A review. Atmos. Environ. 2019, 198, 463–477. [Google Scholar] [CrossRef]
- Murphy, S.M.; Agrawal, H.; Sorooshian, A.; Padró, L.T.; Gates Hersey, S.; Welch, W.A.; Lung, H.; Miller, J.W. Comprehensive simultaneous shipboard and airborne characterization of exhaust from a modern container ship at sea. Environ. Sci. Technol. 2009, 43, 4626–4640. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Industrial emissions of nanomaterials and ultrafine particles. Final Report. 2011. Available online: https://publications.europa.eu/en/publication-detail/-/publication/f5002bc6-ddaa-48cb-9033-a9d12574a32e (accessed on 10 September 2019).
- Riffault, V.; Arndt, J.; Marris, H.; Mbengue, S.; Setyan, A.; Alleman, L.Y.; Deboudt, K.; Flament, P.; Augustin, P.; Delbarre, H.; et al. Fine and ultrafine particles in the vicinity of industrial activities: A review. Environ. Sci. Techn. 2015, 45, 2305–2356. [Google Scholar] [CrossRef]
- Fernandez-Camacho, R.; Rodriguez, S.; De la Rosa, J.D.; Sanchez de la Campa, A.M.; Alastuey, A.; Querol, X.; Gonzalez-Castanedo, Y.; Garcia-Orellana, I.; Nava, S. Ultrafine particle and fine trace metal (As, Cd, Cu, Pb and Zn) pollution episodes induced by industrial emissions in Huelva, SW Spain. Atmos. Environ. 2012, 61, 507–517. [Google Scholar] [CrossRef] [Green Version]
- Jones, A.M.; Harrison, R.M. Emission of ultrafine particles from the incineration of municipal solid waste: A review. Atmos. Environ. 2016, 140, 519–528. [Google Scholar] [CrossRef]
- Buonanno, G.; Ficco, G.; Stabile, L. Size distribution and number concentration of particles at the stack of a municipal waste incinerator. Waste Manag. 2009, 29, 749–755. [Google Scholar] [CrossRef]
- Buonanno, G.; Stabile, L.; Avino, P.; Belluso, E. Chemical, dimensional and morphological ultrafine particle characterization from a waste-to-energy plant. Waste Manag. 2011, 31, 2253–2262. [Google Scholar] [CrossRef]
- Sanderson, P.; Delgado-Saborit, J.M.; Harrison, R.M. A review of chemical and physical characterisation of atmospheric metallic nanoparticles. Atmos. Environ. 2014, 94, 353–365. [Google Scholar] [CrossRef] [Green Version]
- Cernuschi, S.; Giugliano, M.; Ozgen, S.; Consonni, S. Number concentration and chemical composition of ultrafine and nanoparticles from WTE (waste to energy) plants. Sci. Total Environ. 2012, 420, 319–326. [Google Scholar] [CrossRef] [PubMed]
- Morawska, L.; Ayoko, G.A.; Bae, G.N.; Buonanno, G.; Chao, C.Y.H.; Clifford, S.; Fu, S.C.; Hänninen, O.; He, C.; Isaxon, C.; et al. Airborne particles in indoor environment of homes, schools, offices and aged care facilities: The main routes of exposure. Environ. Intern. 2017, 108, 75–83. [Google Scholar] [CrossRef]
- Manigrasso, M.; Vitali, M.; Protano, C.; Avino, P. Temporal evolution of ultrafine particles and of alveolar deposited surface area from main indoor combustion and non-combustion sources in a model room. Sci. Total Environ. 2017, 598, 1015–1026. [Google Scholar] [CrossRef]
- Reid, J.S.; Koppmann, R.; Eck, T.F.; Eleuterio, D.P. A review of biomass burning emissions part II: Intensive physical properties of biomass burning particles. Atmos. Chem. Phys. 2005, 5, 799–825. [Google Scholar] [CrossRef]
- Reid, J.S.; Eck, T.F.; Christopher, S.A.; Koppmann, R.; Dubovik, O.; Eleuterio, D.P.; Holben, B.N.; Reid, E.A.; Zhang, J. A review of biomass burning emissions part III: Intensive optical properties of biomass burning particles. Atmos. Chem. Phys. 2005, 5, 827–849. [Google Scholar] [CrossRef]
- Hinds, W.C. Aerosol Technology: Properties, Behavior, and Measurement of Airborne Particles, 2nd ed.; Wiley: New York, NY, USA, 1999. [Google Scholar]
- Corsini, E.; Vecchi, R.; Marabini, L.; Fermo, P.; Becagli, S.; Bernardoni, V.; Caruso, D.; Corbella, L.; Dell’Acqua, M.; Galli, C.L.; et al. The chemical composition of ultrafine particles and associated biological effects at an alpine town impacted by wood burning. Sci. Total Environ. 2017, 587–588, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Lee, M.L.; Eatough, D.J. Review of recent advances in detection of organic markers in fine particulate matter and their use for source apportionment. J. Air Waste Manag. Assoc. 2010, 60, 3–25. [Google Scholar] [CrossRef]
- Ozgen, S.; Becagli, S.; Bernardoni, V.; Caserini, S.; Caruso, D.; Corbella, L.; Dell’Acqua, M.; Fermo, P.; Gonzalez, R.; Lonati, G.; et al. Analysis of the chemical composition of ultrafine particles from two domestic solid biomass fired room heaters under simulated real-world use. Atmos. Environ. 2017, 150, 87–97. [Google Scholar] [CrossRef] [Green Version]
- Viana, M.; Kuhlbusch, T.A.J.; Querol, X.; Alastuey, A.; Harrison, R.M.; Hopke, P.K.; Winiwarter, W.; Vallius, M.; Szidat, S.; Prévôt, A.S.H.; et al. Source apportionment of particulate matter in Europe: A review of methods and results. J. Aerosol Sci. 2008, 39, 827–849. [Google Scholar] [CrossRef]
- Belis, C.A.; Favez, O.; Mircea, M.; Diapouli, E.; Manousakas, M.I.; Vratolis, S.; Gilardoni, S.; Paglione, M.; Decesari, S.; Mocnik, G.; et al. European Guide on Air Pollution Source Apportionment with Receptor Models—Revised Version; EUR 29816 EN; Publications Office of the European Union: Luxembourg, 2019; ISBN 978-92-76-09001-4. JRC117306; Available online:https://source-apportionment.jrc.ec.europa.eu/downloads.aspx (accessed on 10 September 2019). [CrossRef]
- Bernardoni, V.; Elser, M.; Valli, G.; Valentini, S.; Bigi, A.; Fermo, P.; Piazzalunga, A.; Vecchi, R. Size-segregated aerosol in a hot-spot pollution urban area: Chemical composition and three-way source apportionment. Environ. Poll. 2017, 231, 601–611. [Google Scholar] [CrossRef] [PubMed]
- Ulbrich, I.M.; Canagaratna, M.R.; Cubison, M.J.; Zhang, Q.; Ng, N.L.; Aiken, A.C.; Jimenez, J.L. Three-dimensional factorization of size-resolved organic aerosol mass spectra from Mexico City. Atmos. Meas. Tech. 2012, 5, 195–224. [Google Scholar] [CrossRef] [Green Version]
- Xue, J.; Xue, W.; Sowlat, M.H.; Sioutas, C.; Lolinco, A.; Hasson, A.; Kleeman, M.J. Seasonal and Annual Source Appointment of Carbonaceous Ultrafine Particulate Matter (PM0.1) in Polluted California Cities. Environ. Sci. Technol. 2019, 53, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Kuwayama, T.; Ruehl, C.R.; Kleeman, M.J. Daily Trends and Source Apportionment of Ultrafine Particulate Mass (PM0.1) over an Annual Cycle in a Typical California City. Environ. Sci. Technol. 2013, 47, 13957–13966. [Google Scholar] [CrossRef] [PubMed]
- Kleeman, M.J.; Riddle, S.G.; Robert, M.A.; JAkober, C.A.; Fine, P.M.; Hays, M.D.; Schauer, J.J.; Hannigan, M.P. Source apportionment of fine (PM1.8) and ultrafine (PM0.1) airborne particulate matter during a severe winter pollution episode. Environ. Sci. Technol. 2009, 43, 272–279. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.; Pitz, M.; Schnelle-Kreis, J.; Diemer, J.; Reller, A.; Zimmermann, R.; Soentgen, J.; Stoelzel, M.; Wichmann, H.-E.; Peters, A.; et al. Source apportionment of ambient particles: Comparison of positive matrix factorization analysis applied to particle size distribution and chemical composition data. Atmos. Environ. 2011, 45, 1849–1857. [Google Scholar] [CrossRef]
- Krecl, P.; Hedberg Larsson, E.; Ström, J.; Johansson, C. Contribution of residential wood combustion and other sources to hourly winter aerosol in Northern Sweden determined by positive matrix factorization. Atmos. Chem. Phys. 2008, 8, 3639–3653. [Google Scholar] [CrossRef] [Green Version]
- Johansson, L.S.; Leckner, B.; Gustavsson, L.; Cooper, D.; Tullina, C.; Potter, A. Emission characteristics of modern and old-type residential boilers fired with wood logs and wood pellets. Atmos. Environ. 2004, 38, 4183–4195. [Google Scholar] [CrossRef]
- Tissari, J.; Lyyranen, J.; Hytonen, K.; Sippula, O.; Tapper, U.; Frey, A.; Saarnio, K.; Pennanen, A.S.; Hillamo, R.; Salonen, R.O.; et al. Fine particle and gaseous emissions from normal and smouldering wood combustion in a conventional masonry heater. Atmos. Environ. 2008, 42, 7862–7873. [Google Scholar] [CrossRef]
- Torvela, T.; Tissari, J.; Sippula, O.; Kaivosoja, T.; Leskinen, J.; Virén, A.; Lähde, A.; Jokiniemi, J. Effect of wood combustion conditions on the morphology of freshly emitted fine particles. Atmos. Environ. 2014, 87, 65–76. [Google Scholar] [CrossRef]
- Park, S.S.; Sim, S.Y.; Bae, M.S.; Schauer, J.J. Size distribution of water-soluble components in particulate matter emitted from biomass burning. Atmos. Environ. 2013, 73, 62–72. [Google Scholar] [CrossRef]
- Grilli, A.; Bengalli, R.; Longhin, E.; Capasso, L.; Proverbio, M.C.; Forcato, M.; Bicciato, S.; Gualtieri, M.; Battaglia, C.; Camatini, M. Transcirptional profiling of human bronchial epithelial cell BEAS-2B exposed to diesel and biomass ultrafine particles. BMC Genomics 2018, 19, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Kleeman, M.J.; Robert, M.A.; Riddle, S.G.; Fine, P.M.; Hays, M.D.; Schauer, J.J.; Hannigan, M.P. Size distribution of trace organic species emitted from biomass combustion and meat charbroiling. Atmos. Environ. 2008, 42, 3059–3075. [Google Scholar] [CrossRef]
- Salthammer, T.; Schripp, T.; Wientzek, S.; Wensing, M. Impact of operating wood-burning fireplace ovens on indoor air quality. Chemosphere 2014, 103, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, R.L.; Jensen, O.M.; Afshari, A.; Bergsøe, N.C. Wood-burning stoves in low-carbon dwellings. Energy Build. 2013, 59, 244–251. [Google Scholar] [CrossRef]
- De Gennaro, G.; Dambruoso, P.R.; Di Gilio, A.; Di Palma, V.; Marzocca, A.; Tutino, M. Discontinuous and Continuous Indoor Air Quality Monitoring in Homes with Fireplaces or Wood Stoves as Heating System. Int. J. Environ. Res. Public Health 2016, 13, 78. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Household air pollution and health; World Health Organization: Geneva, Switzerland; Available online:https://www.who.int/news-room/fact-sheets/detail/household-air-pollution-and-health (accessed on 20 August 2019).
- Wong, G.W.; Brunekreef, B.; Ellwood, P.; Anderson, H.R.; Asher, M.I.; Crane, J.; Lai, C.K. ISAAC Phase Three Study Group. Cooking fuels and prevalence of asthma: A global analysis of phase three of the International Study of Asthma and Allergies in Childhood (ISAAC). Lancet Respir. Med. 2013, 1, 386–394. [Google Scholar] [CrossRef]
- Sumpter, C.; Chandramohan, D. Systematic review and meta-analysis of the associations between indoor air pollution and tuberculosis. Trop. Med. Int. Health 2013, 18, 101–108. [Google Scholar] [CrossRef]
- Kelly, F.J.; Fussell, J.C. Linking ambient particulate matter pollution effects with oxidative biology and immune responses. Ann. N. Y. Acad. Sci. 2015, 1340, 84–94. [Google Scholar] [CrossRef]
- Azike, J.E. A review of the history, epidemiology and treatment of squamous cell carcinoma of the scrotum. Rare Tumors 2009, 1, e17. [Google Scholar] [CrossRef]
- Tena, A.F.; Clarà, P.C. Deposition of inhaled particles in the lungs. Arch. Bronconeumol. 2012, 48, 240–246. [Google Scholar] [CrossRef]
- Vu, T.V.; Ondracek, J.; Zdímal, V.; Schwarz, J.; Delgado-Saborit, J.M.; Harrison, R.M. Physical properties and lung deposition of particles emitted from five major indoor sources. Air Qual. Atmos. Health 2017, 10, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Linehan, S.A.; Martinez-Pomares, L.; Gordon, S. Mannose receptor and scavenger receptor: Two macrophage pattern recognition receptors with diverse functions in tissue homeostasis and host defense. Adv. Exp. Med. Biol. 2000, 479, 1–14. [Google Scholar] [PubMed]
- Yue, H.; Wei, W.; Yue, Z.; Lv, P.; Wang, L.; Ma, G.; Su, Z. Particle size affects the cellular response in macrophages. Eur. J. Pharm. Sci. 2010, 41, 650–657. [Google Scholar] [CrossRef]
- Guarieiro Lefol Nani, L.; Guarieiro Lefol Nani, A. Vehicle Emissions: What Will Change with Use of Biofuel? Biofuels—Economy, Environment and Sustainability, Zhen Fang, IntechOpen. Available online: https://www.intechopen.com/books/biofuels-economy-environment-and-sustainability/vehicle-emissions-what-will-change-with-use-of-biofuel- (accessed on 1 August 2019). [CrossRef]
- Kurmi, O.P.; Dunster, C.; Ayres, J.G.; Kelly, F.J. Oxidative potential of smoke from burning wood and mixed biomass fuels. Free Radic. Res. 2013, 47, 829–835. [Google Scholar] [CrossRef] [PubMed]
- Estrella, B.; Naumova, E.N.; Cepeda, M.; Voortman, T.; Katsikis, P.D.; Drexhage, H.A. Effects of Air Pollution on Lung Innate Lymphoid Cells: Review of In Vitro and In Vivo Experimental Studies. Int. J. Environ. Res. Public Health 2019, 16, 2347. [Google Scholar] [CrossRef] [PubMed]
- Hamra, G.B.; Guha, N.; Cohen, A.; Laden, F.; Raaschou-Nielsen, O.; Samet, J.M.; Vineis, P.; Forastiere, F.; Saldiva, P.; Yorifuji, T.; et al. Outdoor particulate matter exposure and lung cancer: A systematic review and meta-analysis. Environ. Health Perspect. 2014, 122, 906–911. [Google Scholar] [CrossRef]
- Huang, F.; Pan, B.; Wu, J.; Chen, E.; Chen, L. Relationship between exposure to PM2.5 and lung cancer incidence and mortality: A meta-analysis. Oncotarget 2017, 8, 43322–43331. [Google Scholar] [CrossRef] [Green Version]
- IARC working group on the evaluation of carcinogenic risks to humans. Household use of solid fuels and high-temperature frying: Evaluation of carcinogenic risks to humans. IARC Monogr. Eval. Carcinog. Risks Hum. 2010, 95, 1–430. [Google Scholar]
- Harada, A.; Sekido, N.; Akahoshi, T.; Wada, T.; Mukaida, N.; Matsushima, K. Essential involvement of interleukin-8 (IL-8) in acute inflammation. J. Leukoc. Biol. 1994, 56, 559–564. [Google Scholar] [CrossRef]
- Chung, K.F. Inflammatory mediators in chronic obstructive pulmonary disease. Curr. Drug Targets Inflamm. Allergy 2005, 4, 619–625. [Google Scholar] [CrossRef]
- Marabini, L.; Ozgen, S.; Turacchi, S.; Aminti, S.; Arnaboldi, F.; Lonati, G.; Fermo, P.; Corbella, L.; Valli, G.; Bernardoni, V.; et al. Ultrafine particles (UFPs) from domestic wood stoves: Genotoxicity in human lung carcinoma A549 cells. Mutag. Res. 2017, 820, 39–46. [Google Scholar] [CrossRef]
- Henneman, L.R.F.; Choirat, C.; Zigler, A.C.M. Accountability assessment of health improvements in the United States associated with reduced coal emissions between 2005 and 2012. Epidemiology 2019, 30, 477–485. [Google Scholar] [CrossRef]
- Li, Y.; Boraschi, D. Endotoxin contamination: A key element in the interpretation of nanosafety studies. Nanomedicine 2016, 11, 269–287. [Google Scholar] [CrossRef] [PubMed]
- Yoda, Y.; Tamura, K.; Shima, M. Airborne endotoxin concentrations in indoor and outdoor particulate matter and their predictors in an urban city. Indoor Air 2017, 27, 955–964. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pope, D.; Bruce, N.; Dherani, M.; Jagoe, K.; Rehfuess, E. Real-life effectiveness of ‘improved’ stoves and clean fuels in reducing PM2.5 and CO: Systematic review and meta-analysis. Environ. Int. 2017, 101, 7–18. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Corsini, E.; Marinovich, M.; Vecchi, R. Ultrafine Particles from Residential Biomass Combustion: A Review on Experimental Data and Toxicological Response. Int. J. Mol. Sci. 2019, 20, 4992. https://doi.org/10.3390/ijms20204992
Corsini E, Marinovich M, Vecchi R. Ultrafine Particles from Residential Biomass Combustion: A Review on Experimental Data and Toxicological Response. International Journal of Molecular Sciences. 2019; 20(20):4992. https://doi.org/10.3390/ijms20204992
Chicago/Turabian StyleCorsini, Emanuela, Marina Marinovich, and Roberta Vecchi. 2019. "Ultrafine Particles from Residential Biomass Combustion: A Review on Experimental Data and Toxicological Response" International Journal of Molecular Sciences 20, no. 20: 4992. https://doi.org/10.3390/ijms20204992





