The Effect of Psyllium Husk on Intestinal Microbiota in Constipated Patients and Healthy Controls
Abstract
1. Introduction
2. Results
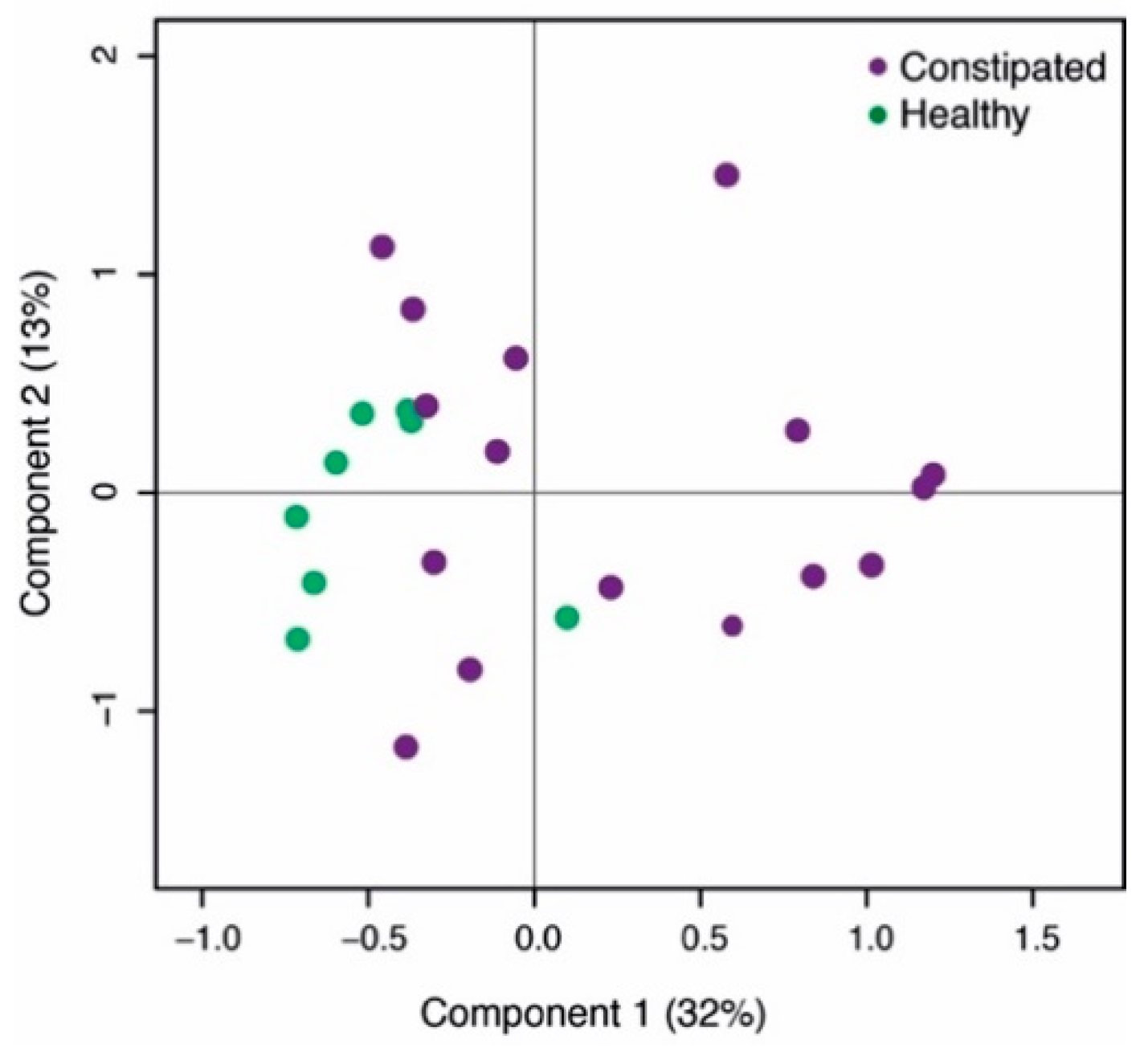
2.1. Differences Between Healthy and Constipated Patients at Baseline
2.2. Psyllium Supplement Has a Small but Significant Effect on the Microbial Composition of Healthy Adults, but no Effect on SCFAs
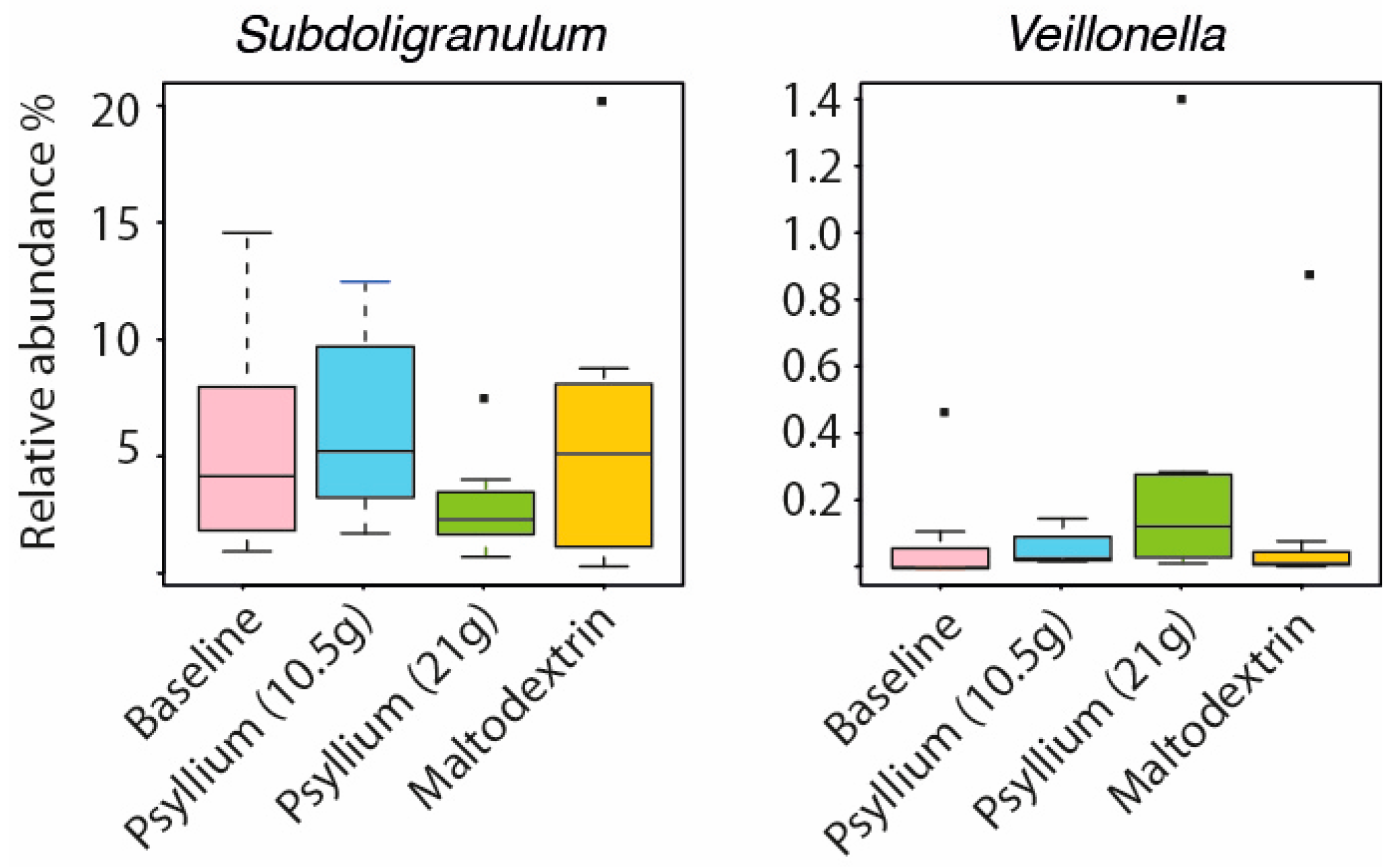
2.3. Intestinal Microbiota and SCFAs of Constipated Patients Were Altered With Psyllium Supplementation
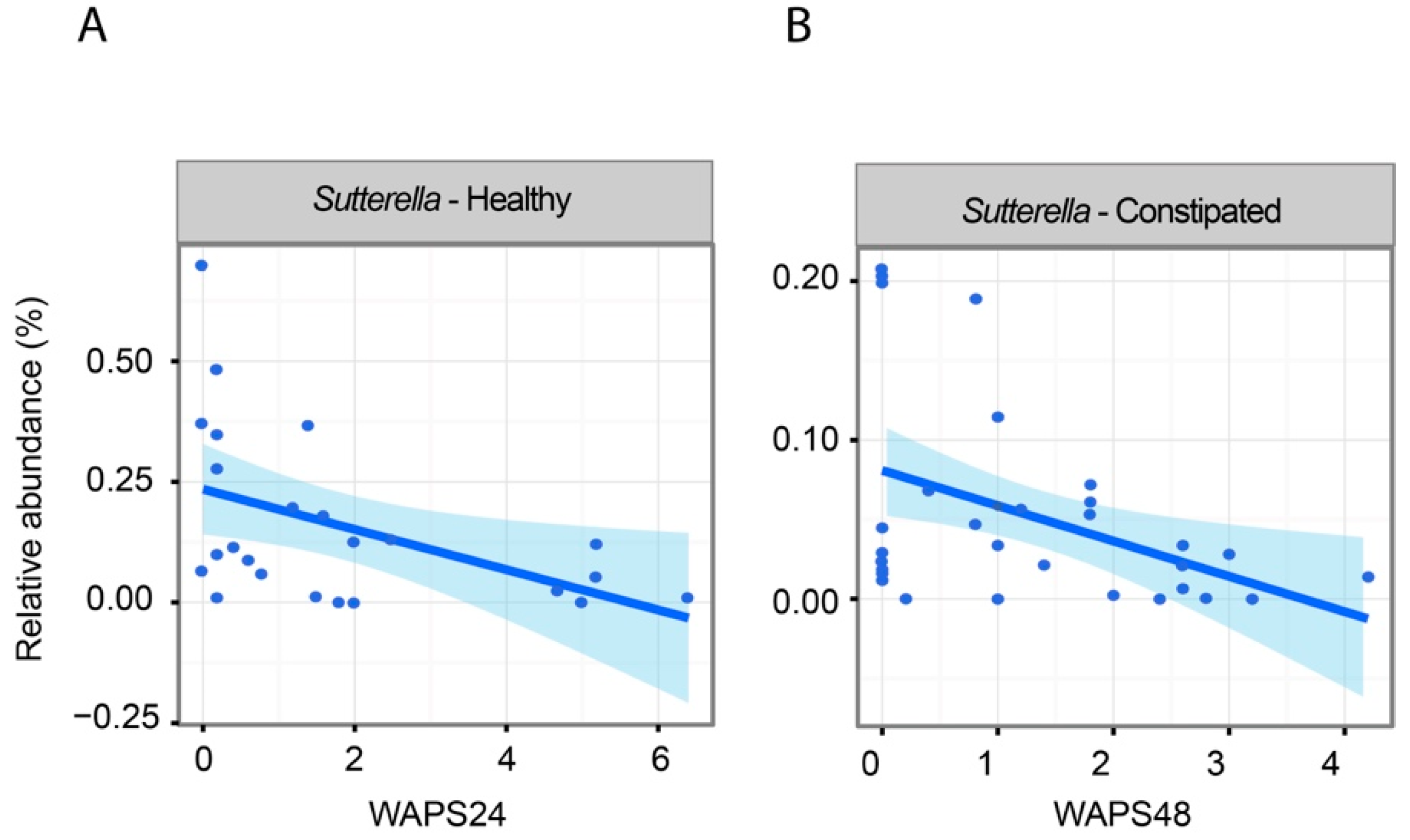
2.4. The Impact of Increased Transit on Intestinal Microbiota
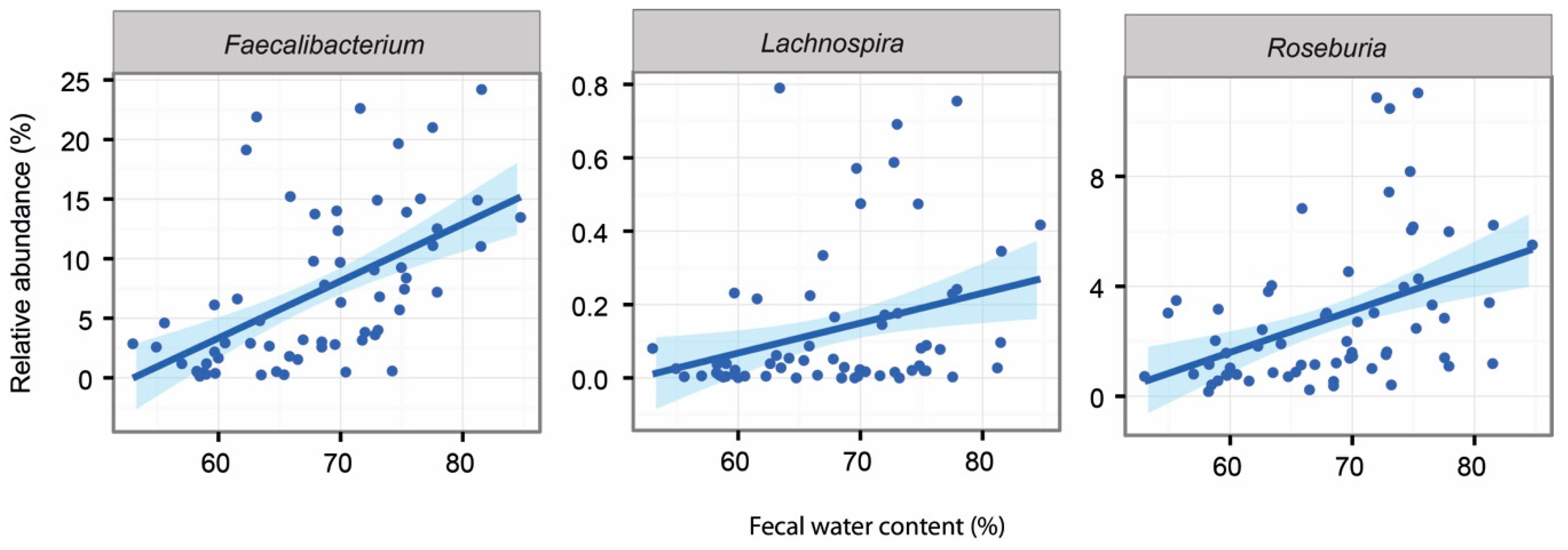
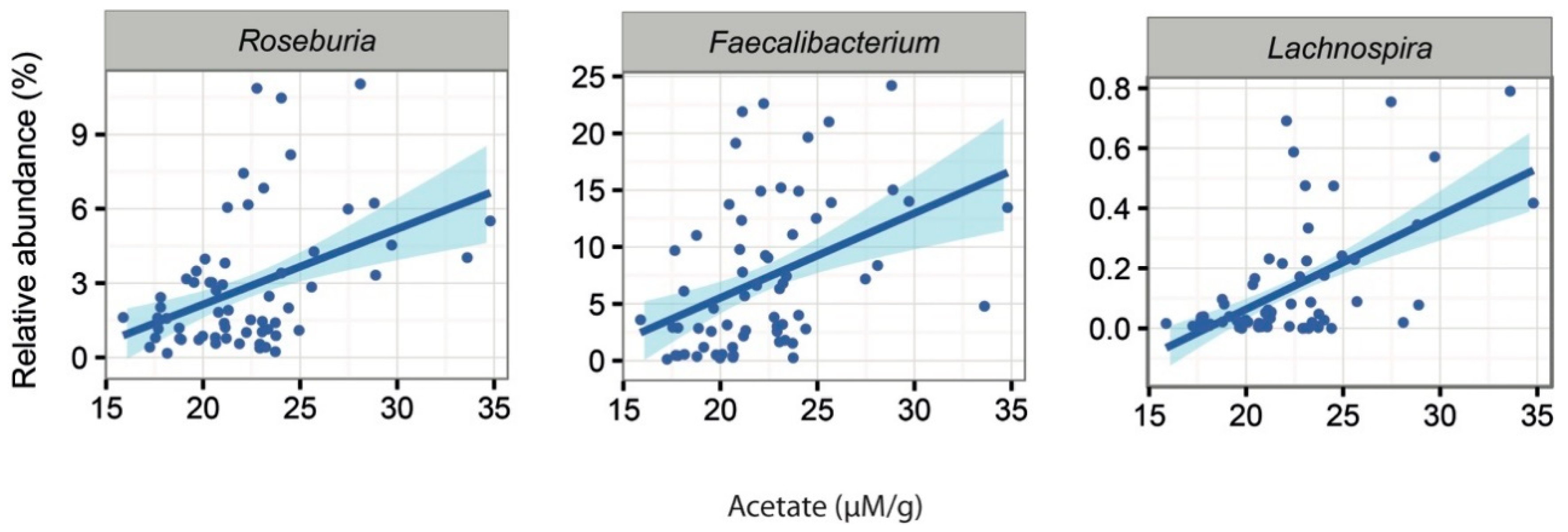
2.5. Associations Between Intestinal Microbiota and Faecal Water Content and SCFAs
3. Discussion
4. Materials and Methods
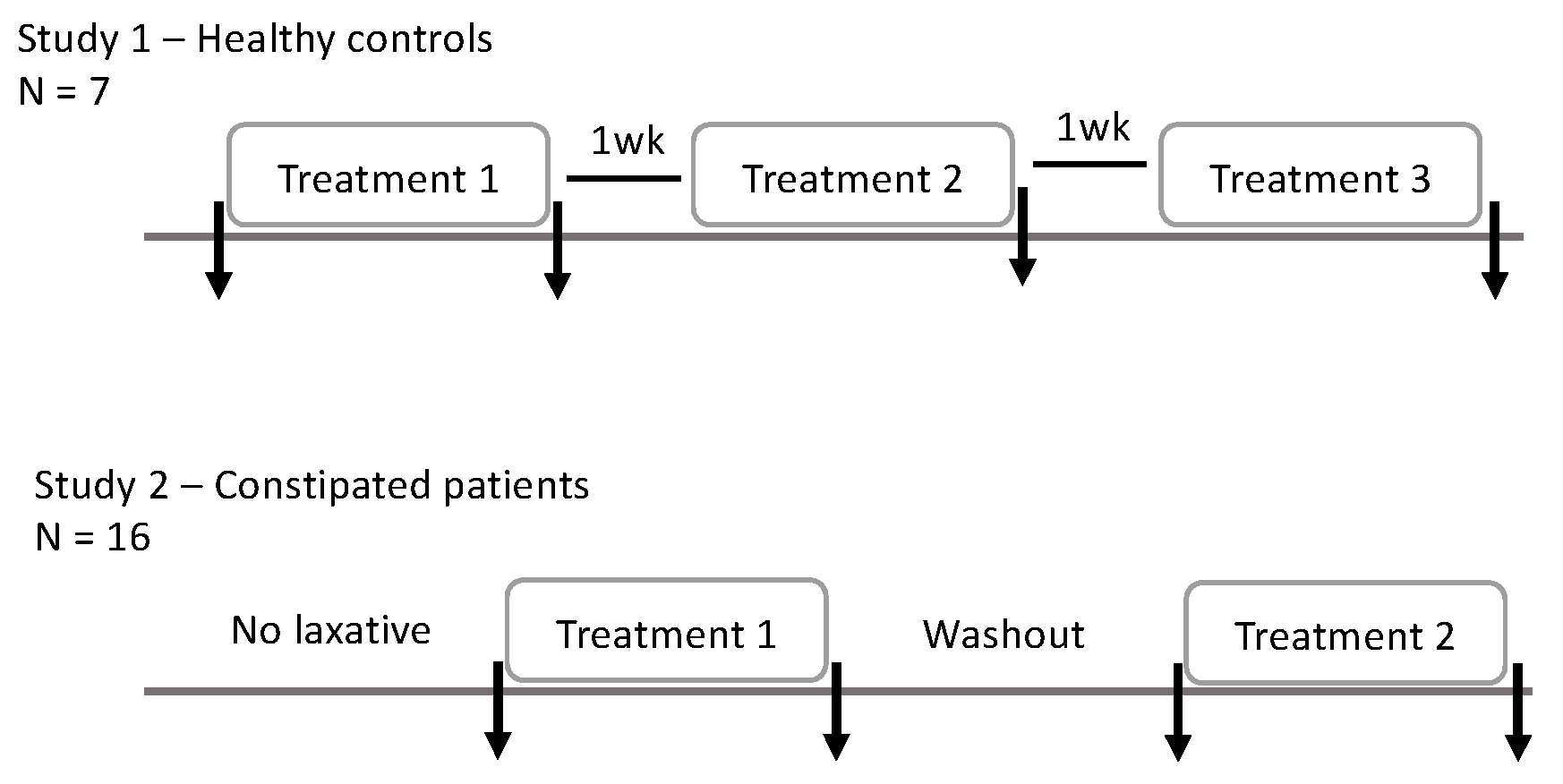
4.1. Study Design
4.2. Measurements of Transit (WAPS)
4.3. Analysis from Faecal Samples
4.3.1. Quantification of SCFA by Gas Chromatography–Mass Spectrometry (GC-MS)
4.3.2. Faecal Water
4.3.3. Extraction of the Faecal Microbiota DNA
4.3.4. Analysis of the Intestinal Microbiota
4.4. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| SCFA | Short-chain fatty acid |
| MRI | Magnetic Resonance Imaging |
| WAPS | Weighted Average Position Score |
References
- Pollet, A.; Van Craeyveld, V.; Van de Wiele, T.; Verstraete, W.; Delcour, J.A.; Courtin, C.M. In Vitro Fermentation of Arabinoxylan Oligosaccharides and Low Molecular Mass Arabinoxylans with Different Structural Properties from Wheat (Triticum aestivum L.) Bran and Psyllium (Plantago ovata Forsk) Seed Husk. J. Agric. Food Chem. 2012, 60, 946–954. [Google Scholar] [CrossRef] [PubMed]
- Al-Khaldi, S.F.; Martin, S.A.; Prakash, L. Fermentation of fenugreek fiber, psyllium husk, and wheat bran by Bacteroides ovatus V975. Curr. Microbiol. 1999, 39, 231–232. [Google Scholar] [CrossRef] [PubMed]
- Elli, M.; Cattivelli, D.; Soldi, S.; Bonatti, M.; Morelli, L. Evaluation of prebiotic potential of refined psyllium (Plantago ovata) fiber in healthy women. J. Clin. Gastroenterol. 2008, 42 Pt 2 (Suppl. 3), S174–S176. [Google Scholar] [CrossRef]
- Campbell, J.M.; Fahey, G.C. Psyllium and methylcellulose fermentation properties in relation to insoluble and soluble fiber standards. Nutr. Res. 1997, 17, 619–629. [Google Scholar] [CrossRef]
- Mendis, M.; Leclerc, E.; Simsek, S. Arabinoxylans, gut microbiota and immunity. Carbohyd. Polym. 2016, 139, 159–166. [Google Scholar] [CrossRef]
- Cremon, C.; Barbaro, M.R.; Ventura, M.; Barbara, G. Pre- and probiotic overview. Curr. Opin. Pharmacol. 2018, 43, 87–92. [Google Scholar] [CrossRef]
- Yang, J.; Wang, H.P.; Zhou, L.; Xu, C.F. Effect of dietary fiber on constipation: A meta analysis. World J. Gastroenterol. 2012, 18, 7378–7383. [Google Scholar] [CrossRef]
- Erdogan, A.; Rao, S.S.C.; Thiruvaiyaru, D.; Lee, Y.Y.; Adame, E.C.; Valestin, J.; O’Banion, M. Randomised clinical trial: Mixed soluble/insoluble fibre vs. psyllium for chronic constipation. Aliment. Pharmacol. Ther. 2016, 44, 35–44. [Google Scholar] [CrossRef]
- Suares, N.C.; Ford, A.C. Systematic review: The effects of fibre in the management of chronic idiopathic constipation. Aliment. Pharmacol. Ther. 2011, 33, 895–901. [Google Scholar] [CrossRef] [PubMed]
- Gamage, H.K.A.H.; Tetu, S.G.; Chong, R.W.W.; Bucio-Noble, D.; Rosewarne, C.P.; Kautto, L.; Ball, M.S.; Molloy, M.P.; Packer, N.H.; Paulsen, I.T. Fiber Supplements Derived From Sugarcane Stem, Wheat Dextrin and Psyllium Husk Have Different In Vitro Effects on the Human Gut Microbiota. Front. Microbiol. 2018, 9. [Google Scholar] [CrossRef]
- Llewellyn, S.R.; Britton, G.J.; Contijoch, E.J.; Vennaro, O.H.; Mortha, A.; Colombel, J.F.; Grinspan, A.; Clemente, J.C.; Merad, M.; Faith, J.J. Interactions Between Diet and the Intestinal Microbiota Alter Intestinal Permeability and Colitis Severity in Mice. Gastroenterology 2018, 154, 1037–1046. [Google Scholar] [CrossRef] [PubMed]
- Major, G.; Murray, K.; Singh, G.; Nowak, A.; Hoad, C.L.; Marciani, L.; Silos-Santiago, A.; Kurtz, C.B.; Johnston, J.M.; Gowland, P.; et al. Demonstration of differences in colonic volumes, transit, chyme consistency, and response to psyllium between healthy and constipated subjects using magnetic resonance imaging. Neurogastroenterol. Motil. Off. J. Eur. Gastrointest. Motil. Soc. 2018, 30, e13400. [Google Scholar] [CrossRef] [PubMed]
- Jalanka-Tuovinen, J.; Salonen, A.; Nikkila, J.; Immonen, O.; Kekkonen, R.; Lahti, L.; Palva, A.; de Vos, W.M. Intestinal microbiota in healthy adults: Temporal analysis reveals individual and common core and relation to intestinal symptoms. PLoS ONE 2011, 6, e23035. [Google Scholar] [CrossRef] [PubMed]
- Vandeputte, D.; Falony, G.; Vieira-Silva, S.; Wang, J.; Sailer, M.; Theis, S.; Verbeke, K.; Raes, J. Prebiotic inulin-type fructans induce specific changes in the human gut microbiota. Gut 2017, 66, 1968–1974. [Google Scholar] [CrossRef] [PubMed]
- Sloan, T.J.; Jalanka, J.; Major, G.A.D.; Krishnasamy, S.; Pritchard, S.; Abdelrazig, S.; Korpela, K.; Singh, G.; Mulvenna, C.; Hoad, C.L.; et al. A low FODMAP diet is associated with changes in the microbiota and reduction in breath hydrogen but not colonic volume in healthy subjects. PLoS ONE 2018, 13, e0201410. [Google Scholar] [CrossRef] [PubMed]
- Bliss, D.Z.; Weimer, P.J.; Jung, H.J.; Savik, K. In vitro degradation and fermentation of three dietary fiber sources by human colonic bacteria. J. Agric. Food Chem. 2013, 61, 4614–4621. [Google Scholar] [CrossRef]
- Ritz, N.L.; Lin, D.M.; Wilson, M.R.; Barton, L.L.; Lin, H.C. Sulfate-reducing bacteria slow intestinal transit in a bismuth-reversible fashion in mice. Neurogastroenterol. Motil. Off. J. Eur. Gastrointest. Motil. Soc. 2017, 29. [Google Scholar] [CrossRef]
- Wang, R. Physiological implications of hydrogen sulfide: A whiff exploration that blossomed. Physiol. Rev. 2012, 92, 791–896. [Google Scholar] [CrossRef]
- Jimenez, M.; Gil, V.; Martinez-Cutillas, M.; Mane, N.; Gallego, D. Hydrogen sulphide as a signalling molecule regulating physiopathological processes in gastrointestinal motility. Br. J. Pharmacol. 2017, 174, 2805–2817. [Google Scholar] [CrossRef]
- Canani, R.B.; Costanzo, M.D.; Leone, L.; Pedata, M.; Meli, R.; Calignano, A. Potential beneficial effects of butyrate in intestinal and extraintestinal diseases. World J. Gastroenterol. 2011, 17, 1519–1528. [Google Scholar] [CrossRef]
- Rasmussen, H.E.; Hamaker, B.; Rajan, K.B.; Mutlu, E.; Green, S.J.; Brown, M.; Kaur, A.; Keshavarzian, A. Starch-entrapped microsphere fibers improve bowel habit but do not exhibit prebiotic capacity in those with unsatisfactory bowel habits: A phase I, randomized, double-blind, controlled human trial. Nutr. Res. 2017, 44, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Lozupone, C.A.; Stombaugh, J.I.; Gordon, J.I.; Jansson, J.K.; Knight, R. Diversity, stability and resilience of the human gut microbiota. Nature 2012, 489, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Van den Bogert, B.; Erkus, O.; Boekhorst, J.; de Goffau, M.; Smid, E.J.; Zoetendal, E.G.; Kleerebezem, M. Diversity of human small intestinal Streptococcus and Veillonella populations. FEMS Microbiol. Ecol. 2013, 85, 376–388. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.; Kalliomaki, M.; Heilig, H.G.; Palva, A.; Lahteenoja, H.; de Vos, W.M.; Salojarvi, J.; Satokari, R. Duodenal microbiota composition and mucosal homeostasis in pediatric celiac disease. BMC Gastroenterol. 2013, 13, 113. [Google Scholar] [CrossRef] [PubMed]
- Wexler, H.M. Genus VIII. Sutterella. In Bergey’s Manual of Systematic Bacteriology; Brenner, D.J., Kreig, N., Staley, J.T., Garrity, G.M., Eds.; Springer-Verlag: New York, NY, USA, 2005; Volume 2. [Google Scholar]
- Hiippala, K.; Kainulainen, V.; Kalliomaki, M.; Arkkila, P.; Satokari, R. Mucosal Prevalence and Interactions with the Epithelium Indicate Commensalism of Sutterella spp. Front. Microbiol. 2016, 7, 1706. [Google Scholar] [CrossRef]
- Wu, F.; Guo, X.; Zhang, J.; Zhang, M.; Ou, Z.; Peng, Y. Phascolarctobacterium faecium abundant colonization in human gastrointestinal tract. Exp. Ther. Med. 2017, 14, 3122–3126. [Google Scholar] [CrossRef]
- Ferreira-Halder, C.V.; Faria, A.V.S.; Andrade, S.S. Action and function of Faecalibacterium prausnitzii in health and disease. Best Pract. Res. Clin. Gastroenterol. 2017, 31, 643–648. [Google Scholar] [CrossRef]
- Hooda, S.; Boler, B.M.; Serao, M.C.; Brulc, J.M.; Staeger, M.A.; Boileau, T.W.; Dowd, S.E.; Fahey, G.C., Jr.; Swanson, K.S. 454 pyrosequencing reveals a shift in fecal microbiota of healthy adult men consuming polydextrose or soluble corn fiber. J. Nutr. 2012, 142, 1259–1265. [Google Scholar] [CrossRef]
- Parthasarathy, G.; Chen, J.; Chen, X.F.; Chia, N.; O’Connor, H.M.; Wolf, P.G.; Gaskins, H.R.; Bharucha, A.E. Relationship Between Microbiota of the Colonic Mucosa vs Feces and Symptoms, Colonic Transit, and Methane Production in Female Patients With Chronic Constipation. Gastroenterology 2016, 150, 367–379.e1. [Google Scholar] [CrossRef]
- Tigchelaar, E.F.; Bonder, M.J.; Jankipersadsing, S.A.; Fu, J.; Wijmenga, C.; Zhernakova, A. Gut microbiota composition associated with stool consistency. Gut 2016, 65, 540–542. [Google Scholar] [CrossRef]
- Chaddock, G.; Lam, C.; Hoad, C.L.; Costigan, C.; Cox, E.F.; Placidi, E.; Thexton, I.; Wright, J.; Blackshaw, P.E.; Perkins, A.C.; et al. Novel MRI tests of orocecal transit time and whole gut transit time: Studies in normal subjects. Neurogastroenterol. Motil. Off. J. Eur. Gastrointest. Motil. Soci. 2014, 26, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Dore, J.; Ehrlich, S.D.; Levenez, F.; Pelletier, E.; Alberti, A.; Bertrand, L.; Bork, P.; Costea, P.I.; Sunagawa, S.; Guarner, F.; et al. IHMS Consortium IHMS_SOP 06 V1: Standard Operating Procedure for Fecal Samples DNA Extraction, Protocol Q; International Human Microbiome Standards, 2015; Available online: http://www.microbiome-standards.org (accessed on 21 March 2017).
- Salonen, A.; Nikkilä, J.; Jalanka-Tuovinen, J.; Immonen, O.; Rajilic-Stojanovic, M.; Kekkonen, R.A.; Palva, A.; de Vos, W.M. Comparative analysis of fecal DNA extraction methods with phylogenetic microarray: Effective recovery of bacterial and archaeal DNA using mechanical cell lysis. J. Microbiol. Methods 2010, 81, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Katri, K. Microbiota Analysis in R Easily; GitHub: San Francisco, CA, USA, 2016. [Google Scholar]
- Edgar, R.C. UPARSE: Highly accurate OTU sequences from microbial amplicon reads. Nat. Methods 2013, 10, 996–998. [Google Scholar] [CrossRef] [PubMed]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glockner, F.O. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2013, 41, D590–D596. [Google Scholar] [CrossRef] [PubMed]
- Nadkarni, M.A.; Martin, F.E.; Jacques, N.A.; Hunter, N. Determination of bacterial load by real-time PCR using a broad-range (universal) probe and primers set. Microbiology 2002, 148, 257–266. [Google Scholar] [CrossRef] [PubMed]
- Johnston, C.; Ufnar, J.A.; Griffith, J.F.; Gooch, J.A.; Stewart, J.R. A real-time qPCR assay for the detection of the nifH gene of Methanobrevibacter smithii, a potential indicator of sewage pollution. J. Appl. Microbiol. 2010, 109, 1946–1956. [Google Scholar] [CrossRef]
- Storey, J.D. A direct approach to false discovery rates. J. R. Stat.Soc. Ser. B 2002, 64, 479–498. [Google Scholar] [CrossRef]
| Demographics | Healthy | Constipated | p-Value |
|---|---|---|---|
| Age (y) | 25.75 (4.16) | 41.00 (15.75) | 0.02 |
| Gender (m/f) | 6/2 | 2/14 | 1.50 × 10−9 |
| Weight (kg) | 73.05 (12.49) | 72.16 (15.65) | 0.76 |
| Stool water content (%) | 71.13 (5.99) | 65.16 (6.55) | 0.07 |
| Acetate (µM/g wet faeces) | 6.90 (3.72) | 21.65 (3.64) | 1.5 × 10−5 |
| Propanoate (µM/g wet faeces) | 5.49 (4.24) | 4.70 (3.44) | 0.58 |
| Butyrate (µM/g wet faeces) | 5.88 (4.39) | 5.32 (4.78) | 0.78 |
| Total bacterial load (per µL of template log10) | 10.47 (0.12) | 10.37 (0.28) | 0.36 |
| Methanogenic archaea (per µL of template, log10) | 4.66 (2.5) | 6.21 (2.05) | 0.16 |
| Microbial richness | 177.75 (74.82) | 201.19 (57.35) | 0.35 |
| Microbial diversity | 8.26(2.71) | 10.25 (6.23) | 0.40 |
| Taxa | Constipated | Healthy | Fc | q-Value |
|---|---|---|---|---|
| Actinobacteria; Actinobacteria; Actinomycetales; Actinomycetaceae; Actinomyces | 0.14% | 0.02% | 5.65 | 0.01 |
| Actinobacteria; Actinobacteria; Corynebacteriales; Nocardiaceae; Rhodococcus | 0.03% | 0.00% | 6.44 | 0.03 |
| Actinobacteria; Coriobacteriia; Coriobacteriales; Coriobacteriaceae; Adlercreutzia | 0.10% | 0.01% | 9.44 | 0.00 |
| Actinobacteria; Coriobacteriia; Coriobacteriales; Coriobacteriaceae; uncultured | 0.79% | 0.17% | 4.52 | 0.04 |
| Firmicutes; Clostridia; Clostridiales; Christensenellaceae; Christensenella | 0.13% | 0.02% | 6.55 | 0.0002 |
| Firmicutes; Clostridia; Clostridiales; Family XIII; Incertae Sedis | 0.58% | 0.16% | 3.52 | 0.01 |
| Firmicutes; Clostridia; Clostridiales; Lachnospiraceae; Lachnospira | 0.13% | 0.58% | 0.22 | 0.01 |
| Firmicutes; Erysipelotrichia; Erysipelotrichales; Erysipelotrichaceae; Incertae Sedis | 0.46% | 0.06% | 7.15 | 0.01 |
| Proteobacteria; Betaproteobacteria; Burkholderiales; Alcaligenaceae; Sutterella | 0.03% | 0.23% | 0.15 | 0.00 |
| Proteobacteria; Deltaproteobacteria; Desulfovibrionales; Desulfovibrionaceae; Desulfovibrio | 0.11% | 0.01% | 14.00 | 0.01 |
| Taxa | Baseline | Psyllium | Wash-Out | Maltodextrin |
|---|---|---|---|---|
| Actinobacteria; Coriobacteriia; Coriobacteriales; Coriobacteriaceae; uncultured | 0.80% | 0.34% | 0.56% | 0.88% |
| Firmicutes; Clostridia; Clostridiales; Christensenellaceae; Christensenella | 0.13% | 0.07% | 0.08% | 0.08% |
| Firmicutes; Clostridia; Clostridiales; Lachnospiraceae; Lachnospira | 0.11% | 0.20% | 0.09% | 0.13% |
| Firmicutes; Clostridia; Clostridiales; Ruminococcaceae; Faecalibacterium | 3.48% | 9.43% | 8.53% | 7.47% |
| Firmicutes; Negativicutes; Selenomonadales; Acidaminococcaceae; Phascolarctobacterium | 0.54% | 1.95% | 0.30% | 0.60% |
| Firmicutes; Negativicutes; Selenomonadales; Veillonellaceae; Veillonella | 0.05% | 0.11% | 0.04% | 0.06% |
| Proteobacteria; Betaproteobacteria; Burkholderiales; Alcaligenaceae; Sutterella | 0.03% | 0.07% | 0.02% | 0.03% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jalanka, J.; Major, G.; Murray, K.; Singh, G.; Nowak, A.; Kurtz, C.; Silos-Santiago, I.; Johnston, J.M.; de Vos, W.M.; Spiller, R. The Effect of Psyllium Husk on Intestinal Microbiota in Constipated Patients and Healthy Controls. Int. J. Mol. Sci. 2019, 20, 433. https://doi.org/10.3390/ijms20020433
Jalanka J, Major G, Murray K, Singh G, Nowak A, Kurtz C, Silos-Santiago I, Johnston JM, de Vos WM, Spiller R. The Effect of Psyllium Husk on Intestinal Microbiota in Constipated Patients and Healthy Controls. International Journal of Molecular Sciences. 2019; 20(2):433. https://doi.org/10.3390/ijms20020433
Chicago/Turabian StyleJalanka, Jonna, Giles Major, Kathryn Murray, Gulzar Singh, Adam Nowak, Caroline Kurtz, Inmaculada Silos-Santiago, Jeffrey M. Johnston, Willem M. de Vos, and Robin Spiller. 2019. "The Effect of Psyllium Husk on Intestinal Microbiota in Constipated Patients and Healthy Controls" International Journal of Molecular Sciences 20, no. 2: 433. https://doi.org/10.3390/ijms20020433
APA StyleJalanka, J., Major, G., Murray, K., Singh, G., Nowak, A., Kurtz, C., Silos-Santiago, I., Johnston, J. M., de Vos, W. M., & Spiller, R. (2019). The Effect of Psyllium Husk on Intestinal Microbiota in Constipated Patients and Healthy Controls. International Journal of Molecular Sciences, 20(2), 433. https://doi.org/10.3390/ijms20020433





