Main Chain Polysulfoxides as Active ‘Stealth’ Polymers with Additional Antioxidant and Anti-Inflammatory Behaviour
Abstract
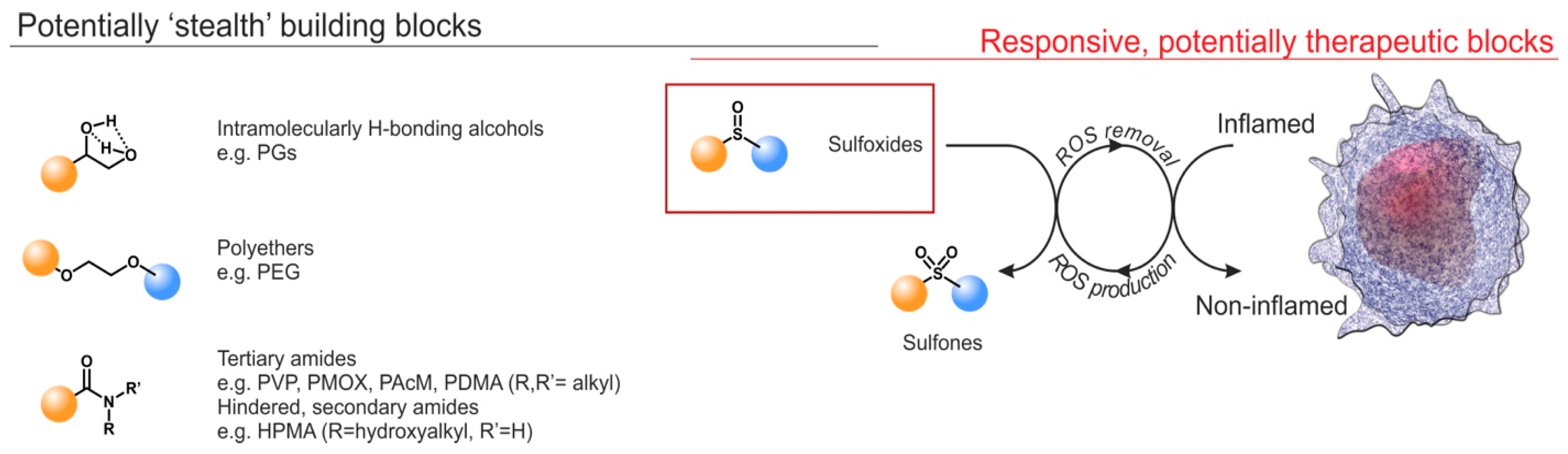
1. Introduction
2. Results and Discussion
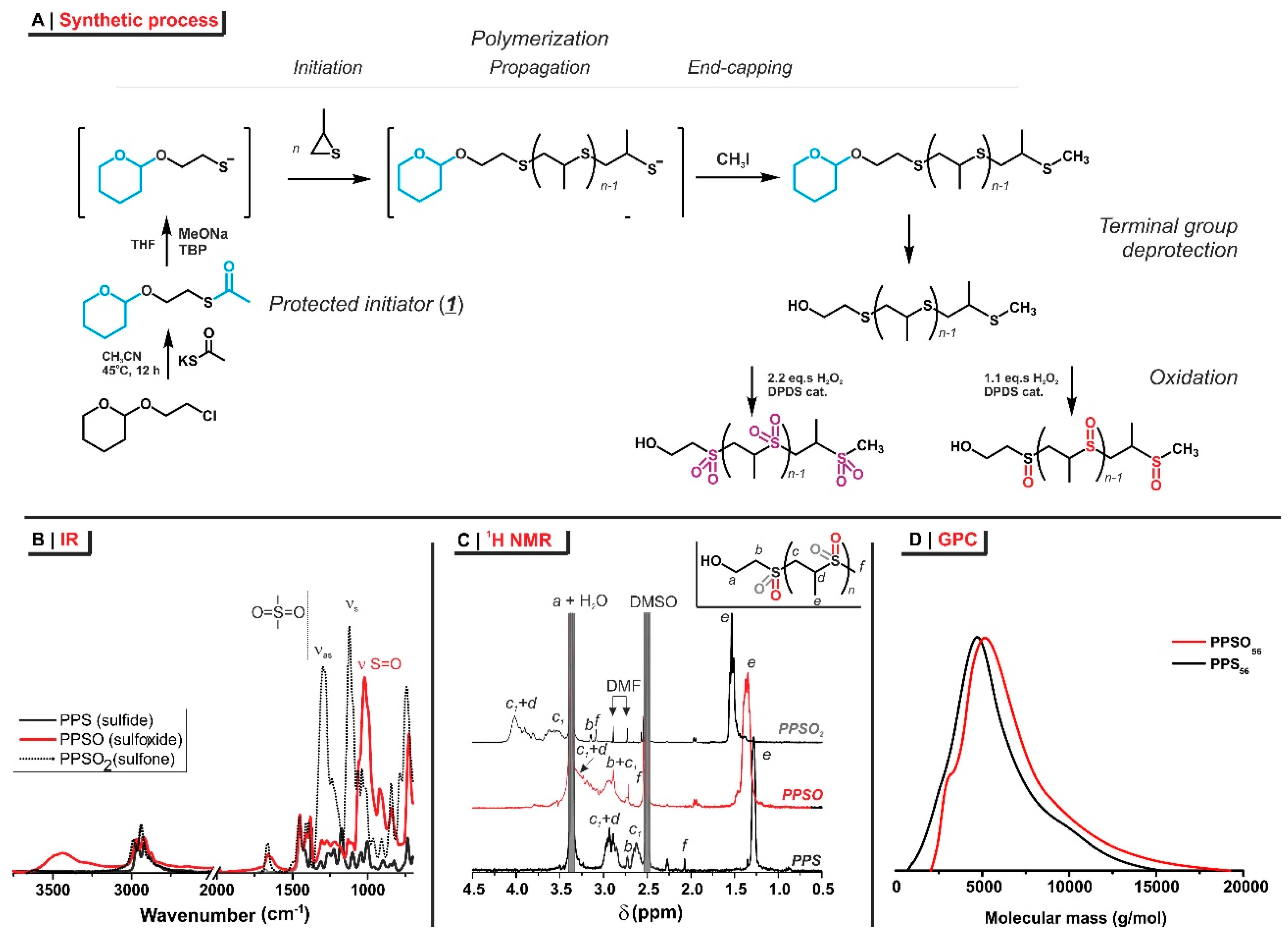
2.1. Polymer Synthesis
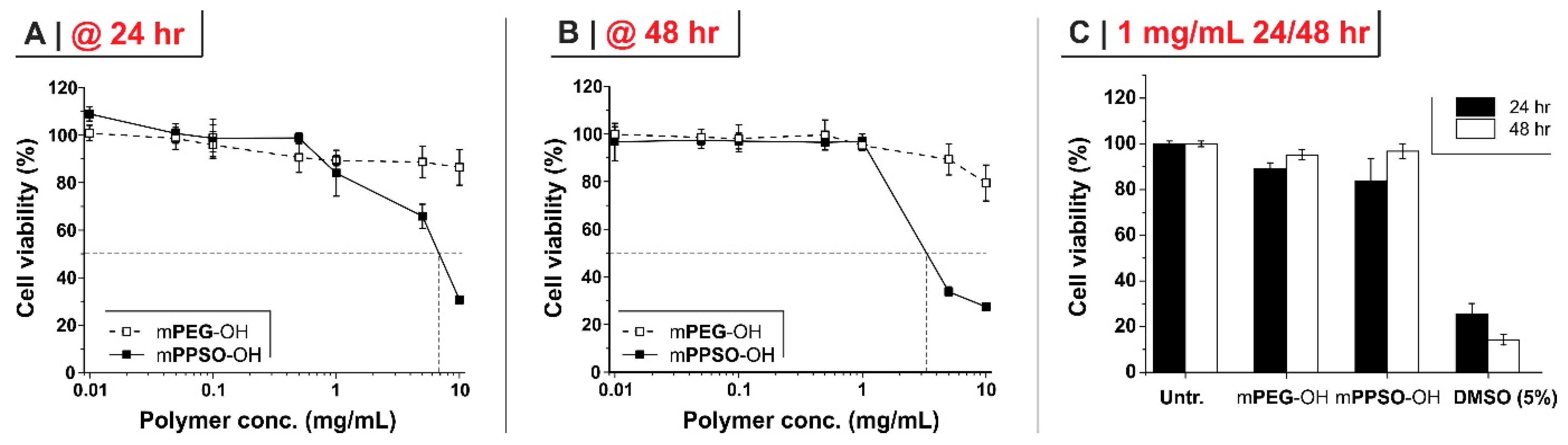
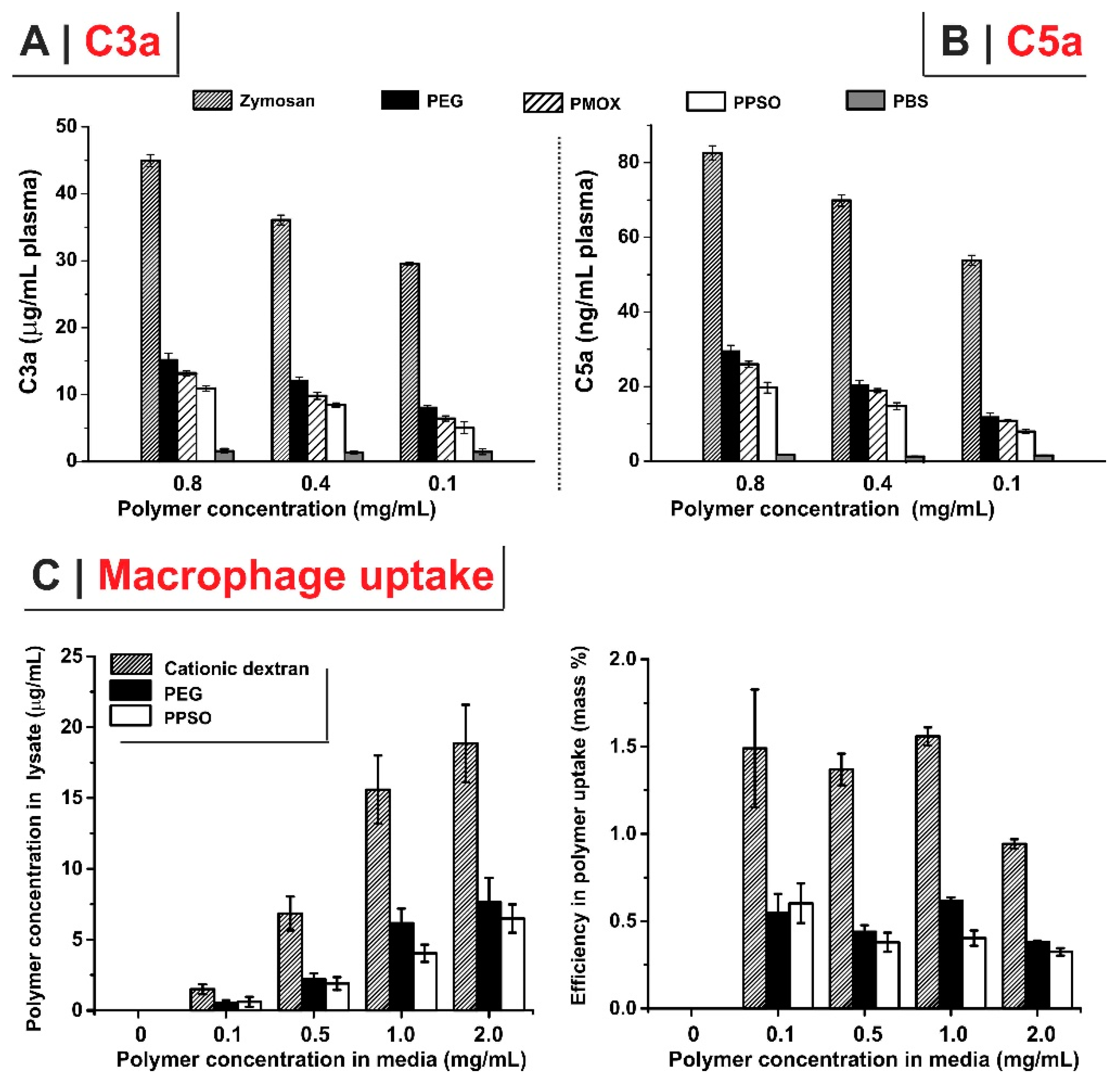
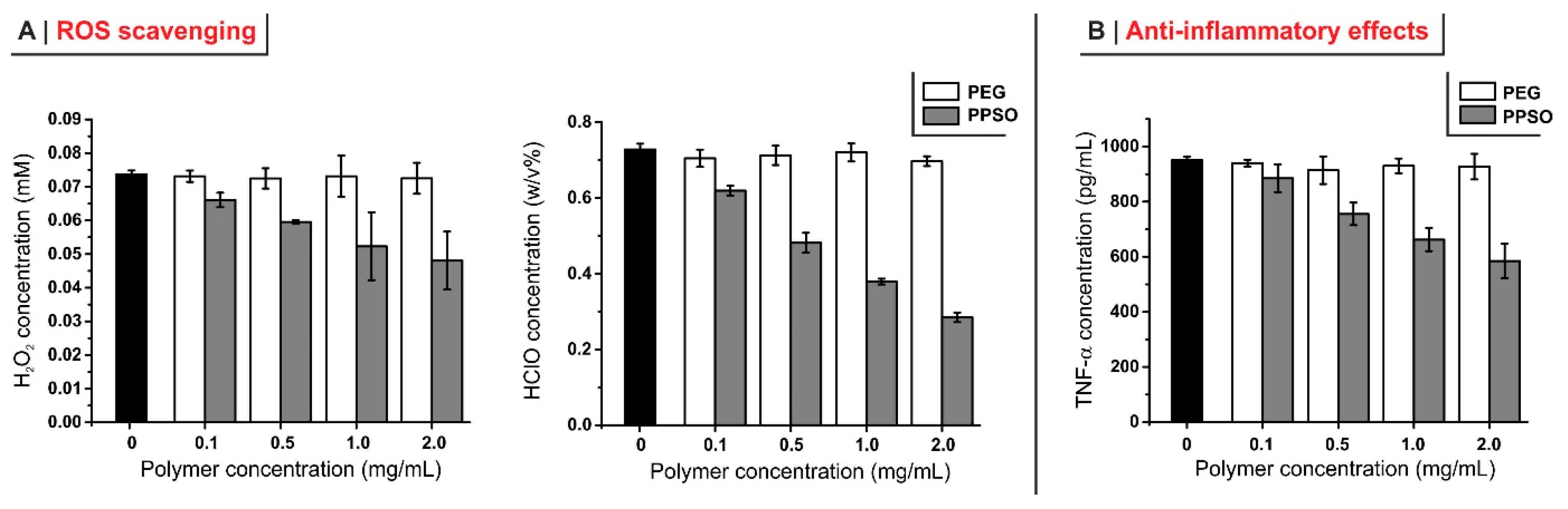
2.2. Potentially “Stealth” Character and Anti-Inflammatory Properties
3. Materials and Methods
3.1. Materials, (Bio)Chemicals, and Cells
3.2. Physico-Chemical Characterization
3.3. Preparative Operations
3.3.1. S-(2-((Tetrahydro-2H-pyran-2-yl)oxy)ethyl) ethanethioate (1)
3.3.2. Monohydroxy-Terminated Poly(Propylene Sulfide) (PPS)56
3.3.3. Poly(Propylene Sulfoxide) mPPSO-OH and Poly(Propylene Sulfone) mPPSO2-OH
3.3.4. FITC-Labelled Polymers
3.4. Biochemical/Biological Assays
3.4.1. Cell Viability
3.4.2. Complement Activation
3.4.3. Polymer Uptake
3.4.4. ROS-Scavenging Effects of PPSO (Analysis of H2O2 and ClO−)
3.4.5. Anti-Inflammatory Effect of PPSO (Analysis of TNF-α)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Allen, T.M.; Cullis, P.R. Drug delivery systems: Entering the mainstream. Science 2004, 303, 1818–1822. [Google Scholar] [CrossRef] [PubMed]
- Swierczewska, M.; Lee, K.C.; Lee, S. What is the future of PEGylated therapies? Expert Opin. Emerg. Drugs 2015, 20, 531–536. [Google Scholar] [CrossRef]
- D’arcy, R.; Tirelli, N. Fishing for fire: Strategies for biological targeting and criteria for material design in anti-inflammatory therapies. Polym. Adv. Technol. 2014, 25, 478–498. [Google Scholar] [CrossRef]
- Ekladious, I.; Colson, Y.L.; Grinstaff, M.W. Polymer–drug conjugate therapeutics: Advances, insights and prospects. Nat. Rev. Drug Discov. 2019, 18, 273–294. [Google Scholar] [CrossRef] [PubMed]
- Papavasiliou, G.; Sokic, S.; Turturro, M. Synthetic PEG Hydrogels as Extracellular Matrix Mimics for Tissue Engineering Applications. In Biotechnology-Molecular Studies and Novel Applications for Improved Quality of Human Life; InTech: London, UK, 2012. [Google Scholar]
- Webster, R.; Elliott, V.; Park, B.K.; Walker, D.; Hankin, M.; Taupin, P. PEG and PEG conjugates toxicity: Towards an understanding of the toxicity of PEG and its relevance to PEGylated biologicals. In Pegylated Protein Drugs: Basic Science and Clinical Applications; Springer: Basel, Switzerland, 2009. [Google Scholar]
- Hatakeyama, H.; Akita, H.; Harashima, H. The Polyethyleneglycol Dilemma: Advantage and Disadvantage of PEGylation of Liposomes for Systemic Genes and Nucleic Acids Delivery to Tumors. Biol. Pharm. Bull. 2013, 36, 892–899. [Google Scholar] [CrossRef] [PubMed]
- Abuchowski, A.; Vanes, T.; Palczuk, N.C.; Davis, F.F. Alteration of Immunological Properties of Bovine Serum-Albumin by Covalent Attachment of Polyethylene-Glycol. J. Biol. Chem. 1977, 252, 3578–3581. [Google Scholar] [PubMed]
- Duncan, R. Development of HPMA copolymer-anticancer conjugates: Clinical experience and lessons learnt. Adv. Drug Deliv. Rev. 2009, 61, 1131–1148. [Google Scholar] [CrossRef]
- Duncan, R.; Vicent, M.J. Do HPMA copolymer conjugates have a future as clinically useful nanomedicines? A critical overview of current status and future opportunities. Adv. Drug Deliv. Rev. 2010, 62, 272–282. [Google Scholar] [CrossRef]
- Francini, N.; Cochrane, D.; Illingworth, S.; Purdie, L.; Mantovani, G.; Fisher, K.; Seymour, L.W.; Spain, S.G.; Alexander, C. Polyvalent Diazonium Polymers Provide Efficient Protection of Oncolytic Adenovirus Enadenotucirev from Neutralizing Antibodies while Maintaining Biological Activity In Vitro and In Vivo. Bioconjug. Chem. 2019, 30, 1244–1257. [Google Scholar] [CrossRef]
- Immordino, M.L.; Dosio, F.; Cattel, L. Stealth liposomes: Review of the basic science, rationale, and clinical applications, existing and potential. Int. J. Nanomed. 2006, 1, 297–315. [Google Scholar]
- Zelikin, A.N.; Such, G.K.; Postma, A.; Caruso, F. Poly(vinylpyrrolidone) for bioconjugation and surface ligand immobilization. Biomacromolecules 2007, 8, 2950–2953. [Google Scholar] [CrossRef] [PubMed]
- Gaertner, F.C.; Luxenhofer, R.; Blechert, B.; Jordan, R.; Essler, M. Synthesis, biodistribution and excretion of radiolabeled poly(2-alkyl-2-oxazoline)s. J. Control. Release 2007, 119, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Zalipsky, S.; Hansen, C.B.; Oaks, J.M.; Allen, T.M. Evaluation of blood clearance rates and biodistribution of poly(2-oxazoline)-grafted liposomes. J. Pharm. Sci. 1996, 85, 133–137. [Google Scholar] [CrossRef]
- Torchilin, V.P.; Trubetskoy, V.S.; Whiteman, K.R.; Caliceti, P.; Ferruti, P.; Veronese, F.M. New Synthetic Amphiphilic Polymers for Steric Protection of Liposomes in-Vivo. J. Pharm. Sci. 1995, 84, 1049–1053. [Google Scholar] [CrossRef] [PubMed]
- Drotleff, S.; Lungwitz, U.; Breunig, M.; Dennis, A.; Blunk, T.; Tessmar, J.; Gopferich, A. Biomimetic polymers in pharmaceutical and biomedical sciences. Eur. J. Pharm. Biopharm. 2004, 58, 385–407. [Google Scholar] [CrossRef] [PubMed]
- Kohori, F.; Sakai, K.; Aoyagi, T.; Yokoyama, M.; Yamato, M.; Sakurai, Y.; Okano, T. Control of adriamycin cytotoxic activity using thermally responsive polymeric micelles composed of poly(N-isopropylacrylamide-co-N,N-dimethylacrylamide)-b-poly(D,L-lactide). Colloid Surf. B 1999, 16, 195–205. [Google Scholar] [CrossRef]
- Lila, A.S.A.; Kiwada, H.; Ishida, T. The accelerated blood clearance (ABC) phenomenon: Clinical challenge and approaches to manage. J. Control. Release 2013, 172, 38–47. [Google Scholar] [CrossRef]
- Ishida, T.; Kiwada, H. Accelerated blood clearance (ABC) phenomenon upon repeated injection of PEGylated liposomes. Int. J. Pharm. 2008, 354, 56–62. [Google Scholar] [CrossRef]
- Dams, E.T.M.; Laverman, P.; Oyen, W.J.G.; Storm, G.; Scherphof, G.L.; Van der Meer, J.W.M.; Corstens, F.H.M.; Boerman, O.C. Accelerated blood clearance and altered biodistribution of repeated injections of sterically stabilized liposomes. J. Pharm. Exp. Ther. 2000, 292, 1071–1079. [Google Scholar]
- Ishida, T.; Ichihara, M.; Wang, X.; Yamamoto, K.; Kimura, J.; Majima, E.; Kiwada, H. Injection of PEGylated liposomes in rats elicits PEG-specific IgM, which is responsible for rapid elimination of a second dose of PEGylated liposomes. J. Control. Release 2006, 112, 15–25. [Google Scholar] [CrossRef]
- Wang, X.Y.; Ishida, T.; Kiwada, H. Anti-PEG IgM elicited by injection of liposomes is involved in the enhanced blood clearance of a subsequent dose of PEGylated liposomes. J. Control. Release 2007, 119, 236–244. [Google Scholar] [CrossRef] [PubMed]
- Hamada, I.; Hunter, A.C.; Szebeni, J.; Moghimi, S.M. Poly(ethylene glycol)s generate complement activation products in human serum through increased alternative pathway turnover and a MASP-2-dependent process. Mol. Immunol. 2008, 46, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Moghimi, S.M.; Hunter, A.C.; Dadswell, C.M.; Savay, S.; Alving, C.R.; Szebeni, J. Causative factors behind poloxamer 188 (pluronic F68, Flocor (TM))-induced complement activation in human sera. A protective role against poloxamer-mediated complement activation by elevated serum lipoprotein levels. BBA Mol. Basis Dis. 2004, 1689, 103–113. [Google Scholar] [CrossRef]
- Ishida, T.; Kashima, S.; Kiwada, H. The contribution of phagocytic activity of liver macrophages to the accelerated blood clearance (ABC) phenomenon of PEGylated liposomes in rats. J. Control. Release 2008, 126, 162–165. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, Y.; Maeda, H. A New Concept for Macromolecular Therapeutics in Cancer-Chemotherapy-Mechanism of Tumoritropic Accumulation of Proteins and the Antitumor Agent Smancs. Cancer Res. 1986, 46, 6387–6392. [Google Scholar] [PubMed]
- Kierstead, P.H.; Okochi, H.; Venditto, V.J.; Chuong, T.C.; Kivimae, S.; Frechet, J.M.J.; Szoka, F.C. The effect of polymer backbone chemistry on the induction of the accelerated blood clearance in polymer modified liposomes. J. Control. Release 2015, 213, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Li, S.P.; Chung, H.S.; Simakova, A.; Wang, Z.Y.; Park, S.; Fu, L.Y.; Cohen-Karni, D.; Averick, S.; Matyjaszewski, K. Biocompatible Polymeric Analogues of DMSO Prepared by Atom Transfer Radical Polymerization. Biomacromolecules 2017, 18, 475–482. [Google Scholar] [CrossRef]
- Carampin, P.; Lallana, E.; Laliturai, J.; Carroccio, S.C.; Puglisi, C.; Tirelli, N. Oxidant-Dependent REDOX Responsiveness of Polysulfides. Macromol. Chem. Phys. 2012, 213, 2052–2061. [Google Scholar] [CrossRef]
- Pitha, J.; Szente, L.; Greenberg, J. Poly-L-Methionine Sulfoxide-a Biologically Inert Analog of Dimethylsulfoxide with Solubilizing Potency. J. Pharm. Sci. 1983, 72, 665–668. [Google Scholar] [CrossRef]
- Hofman, V.; Ringsdorf, H.; Muacevic, G. Pharmakologisch aktive Polymere, 8*. Poly[2-(rnethylsulfinyl)athylacrylat]e und ihre verrnittelnde Wirkung auf die transkutane Resorption von Pharrnaka. Makromol. Chem. 1975, 176, 1929–1943. [Google Scholar] [CrossRef]
- Sun, Y.; Lathwal, S.; Wang, Y.; Fu, L.Y.; Olszewski, M.; Fantin, M.; Enciso, A.E.; Szczepaniak, G.; Das, S.; Matyjaszewski, K. Preparation of Well-Defined Polymers and DNA-Polymer Bioconjugates via Small-Volume eATRP in the Presence of Air. ACS Macro Lett. 2019, 8, 603–609. [Google Scholar] [CrossRef]
- Navarro, L.A.; Enciso, A.E.; Matyjaszewski, K.; Zauscher, S. Enzymatically Degassed Surface-Initiated Atom Transfer Radical Polymerization with Real-Time Monitoring. J. Am. Chem. Soc. 2019, 141, 3100–3109. [Google Scholar] [CrossRef] [PubMed]
- Fu, C.K.; Zhang, C.; Peng, H.; Han, F.; Baker, C.; Wu, Y.; Ta, H.; Whittaker, A.K. Enhanced Performance of Polymeric F-19 MRI Contrast Agents through Incorporation of Highly Water-Soluble Monomer MSEA. Macromolecules 2018, 51, 5875–5882. [Google Scholar] [CrossRef]
- Li, S.P.; Omi, M.; Cartieri, F.; Konkolewicz, D.; Mao, G.; Gao, H.F.; Averick, S.E.; Mishina, Y.; Matyjaszewski, K. Cationic Hyperbranched Polymers with Biocompatible Shells for siRNA Delivery. Biomacromolecules 2018, 19, 3754–3765. [Google Scholar] [CrossRef] [PubMed]
- McKim, A.S.; Strub, R. Advances in the Regulated Pharmaceutical Use of Dimethyl Sulfoxide USP, Ph.Eur. Pharm. Technol. 2016, 28, s30–s35. [Google Scholar]
- Yi, X.Y.; Liu, M.X.; Luo, Q.; Zhuo, H.L.; Cao, H.; Wang, J.X.; Han, Y. Toxic effects of dimethyl sulfoxide on red blood cells, platelets, and vascular endothelial cells in vitro. FEBS Open Bio 2017, 7, 485–494. [Google Scholar] [CrossRef] [PubMed]
- Costa, L.D.; Ottoni, M.H.F.; dos Santos, M.G.; Meireles, A.B.; de Almeida, V.G.; Pereira, W.D.; de Avelar-Freitas, B.A.; Brito-Melo, G.E.A. Dimethyl Sulfoxide (DMSO) Decreases Cell Proliferation and TNF-alpha, IFN-gamma, and IL-2 Cytokines Production in Cultures of Peripheral Blood Lymphocytes. Molecules 2017, 22, 1789. [Google Scholar] [CrossRef]
- Elisia, I.; Nakamura, H.; Lam, V.; Hofs, E.; Cederberg, R.; Cait, J.; Hughes, M.R.; Lee, L.; Jia, W.; Adomat, H.H.; et al. DMSO Represses Inflammatory Cytokine Production from Human Blood Cells and Reduces Autoimmune Arthritis. PLoS ONE 2016, 11, e0152538. [Google Scholar] [CrossRef]
- Chen, Q.; Olashaw, N.; Wu, J. Participation of reactive oxygen species in the lysophosphatidic acid-stimulated mitogen-activated protein kinase kinase activation pathway. J. Biol. Chem. 1995, 270, 28499–58502. [Google Scholar] [CrossRef]
- El-Mohtadi, F.; d’arcy, R.; Tirelli, N. Oxidation-Responsive Materials: Biological Rationale, State of the Art, Multiple Responsiveness, and Open Issues. Macromol. Rapid Commun. 2019, 40, 1800699. [Google Scholar] [CrossRef]
- Khutoryanskiy, V.V.; Tirelli, N. Oxidation-responsiveness of nanomaterials for targeting inflammatory reactions. Pure Appl. Chem. 2008, 80, 1703–1718. [Google Scholar] [CrossRef]
- Rajkovic, O.; Gourmel, C.; d’Arcy, R.; Wong, R.; Rajkovic, I.; Tirelli, N.; Pinteaux, E. Reactive Oxygen Species-Responsive Nanoparticles for the Treatment of Ischemic Stroke. Adv. Ther. 2019, 2, 1900038. [Google Scholar] [CrossRef]
- Jeanmaire, D.; Laliturai, J.; Almalik, A.; Carampin, P.; d’Arcy, R.; Lallana, E.; Evans, R.; Winpenny, R.E.P.; Tirelli, N. Chemical specificity in REDOX-responsive materials: The diverse effects of different Reactive Oxygen Species (ROS) on polysulfide nanoparticles. Polym. Chem. 2014, 5, 1393–1404. [Google Scholar] [CrossRef]
- Wang, L.; Kilcher, G.; Tirelli, N. Avoiding Disulfides: Improvement of Initiation and End-Capping Reactions in the Synthesis of Polysulfide Block Copolymers. Macromol. Chem. Phys. 2009, 210, 447–456. [Google Scholar] [CrossRef]
- D’arcy, R.; Siani, A.; Lallana, E.; Tirelli, N. Influence of Primary Structure on Responsiveness. Oxidative, Thermal, and Thermo-Oxidative Responses in Polysulfides. Macromolecules 2015, 48, 8108–8120. [Google Scholar]
- Page, P.C.B.; Buckley, B.R.; Elliott, C.; Chan, Y.; Dreyfus, N.; Marken, F. Chemoselective Oxidation of Sulfides to Sulfoxides with Urea-Hydrogen Peroxide Complex Catalysed by Diselenide. Synlett 2016, 27, 80–82. [Google Scholar] [CrossRef]
- Fanucchi, M.; Glaspy, J.; Crawford, J.; Garst, J.; Figlin, R.; Sheridan, W.; Menchaca, D.; Tomita, D.; Ozer, H.; Harker, L. Effects of polyethylene glycol-conjugated recombinant human megakaryocyte growth and development factor on platelet counts after chemotherapy for lung cancer. N. Engl. J. Med. 1997, 336, 404–409. [Google Scholar] [CrossRef]
- Wang, Q.; Jiang, J.Y.; Chen, W.F.; Jiang, H.; Zhang, Z.R.; Sun, X. Targeted delivery of low-dose dexamethasone using PCL-PEG micelles for effective treatment of rheumatoid arthritis. J. Control Release 2016, 230, 64–72. [Google Scholar] [CrossRef]
- Poljsak, B.; Suput, D.; Milisav, I. Achieving the Balance between ROS and Antioxidants: When to Use the Synthetic Antioxidants. Oxid. Med. Cell. Longev. 2013, 956792. [Google Scholar] [CrossRef]
- Woffindin, C.; Hoenich, N.A.; Matthews, J.N.S. Cellulose-biased hemodialysis membranes-Biocompatibility and functional performance compared. Nephrol. Dial. Transplant. 1992, 7, 340–345. [Google Scholar] [CrossRef]
- Payne, M.S.; Horbett, T.A. Complement Activation by Hydroxyethylmethacrylate-Ethylmethacrylate Copolymers. J. Biomed. Mater. Res. 1987, 21, 843–859. [Google Scholar] [CrossRef] [PubMed]
- Lamba, N.M.K.; Courtney, J.M.; Gaylor, J.D.S.; Lowe, G.D.O. In vitro investigation of the blood response to medical grade PVC and the effect of heparin on the blood response. Biomaterials 2000, 21, 89–96. [Google Scholar] [CrossRef]
- Passirani, C.; Barratt, G.; Devissaguet, J.P.; Labarre, D. Interactions of nanoparticles bearing heparin or dextran covalently bound to poly(methyl methacrylate) with the complement system. Life Sci. 1998, 62, 775–785. [Google Scholar] [CrossRef]
- Gaucher, G.; Asahina, K.; Wang, J.H.; Leroux, J.C. Effect of Poly(N-vinyl-pyrrolidone)-block-poly(D,L-lactide) as Coating Agent on the Opsonization, Phagocytosis, and Pharmacokinetics of Biodegradable Nanoparticles. Biomacromolecules 2009, 10, 408–416. [Google Scholar] [CrossRef] [PubMed]
- Tauhardt, L.; Pretzel, D.; Kempe, K.; Gottschaldt, M.; Pohlers, D.; Schubert, U.S. Zwitterionic poly(2-oxazoline)s as promising candidates for blood contacting applications. Polym. Chem. 2014, 5, 5751–5764. [Google Scholar] [CrossRef]
- Mosqueira, V.C.F.; Legrand, P.; Gulik, A.; Bourdon, O.; Gref, R.; Labarre, D.; Barratt, G. Relationship between complement activation, cellular uptake and surface physicochemical aspects of novel PEG-modified nanocapsules. Biomaterials 2001, 22, 2967–2979. [Google Scholar] [CrossRef]
- Yu, J.; Lamba, N.M.K.; Courtney, J.M.; Whateley, T.L.; Gaylor, J.D.S.; Lowe, G.D.O.; Ishihara, K.; Nakabayashi, N. Polymeric Biomaterials-Influence of Phosphorylcholine Polar Groups on Protein Adsorption and Complement Activation. Int. J. Artif. Organs 1994, 17, 499–504. [Google Scholar] [CrossRef] [PubMed]
- Xiao, H.D.; Xin, K.; Dou, H.F.; Yin, G.; Quan, Y.W.; Wang, R.Y. A fast-responsive mitochondria-targeted fluorescent probe detecting endogenous hypochlorite in living RAW 264.7 cells and nude mouse. Chem. Commun. 2015, 51, 1442–1445. [Google Scholar] [CrossRef]
- D’arcy, R.; Tirelli, N. Mitsunobu Reaction: A Versatile Tool for PEG End Functionalization. Macromol. Rapid Commun. 2015, 36, 1829–1835. [Google Scholar] [CrossRef]
| (g/mol)/ | Đ b | End-Capping Yield (mol %) c | |||
|---|---|---|---|---|---|
| Theor. | 1H NMR a | GPC b | |||
| mPPS56-OH | 4315/56 | 4390/57 | 3850/≈49 | 1.26 | 98 |
| mPPSO56-OH | 5211/56 | 5300/57 | 4400/≈47 | 1.21 | not applicable |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
El Mohtadi, F.; d’Arcy, R.; Yang, X.; Turhan, Z.Y.; Alshamsan, A.; Tirelli, N. Main Chain Polysulfoxides as Active ‘Stealth’ Polymers with Additional Antioxidant and Anti-Inflammatory Behaviour. Int. J. Mol. Sci. 2019, 20, 4583. https://doi.org/10.3390/ijms20184583
El Mohtadi F, d’Arcy R, Yang X, Turhan ZY, Alshamsan A, Tirelli N. Main Chain Polysulfoxides as Active ‘Stealth’ Polymers with Additional Antioxidant and Anti-Inflammatory Behaviour. International Journal of Molecular Sciences. 2019; 20(18):4583. https://doi.org/10.3390/ijms20184583
Chicago/Turabian StyleEl Mohtadi, Farah, Richard d’Arcy, Xiaoye Yang, Zulfiye Yesim Turhan, Aws Alshamsan, and Nicola Tirelli. 2019. "Main Chain Polysulfoxides as Active ‘Stealth’ Polymers with Additional Antioxidant and Anti-Inflammatory Behaviour" International Journal of Molecular Sciences 20, no. 18: 4583. https://doi.org/10.3390/ijms20184583
APA StyleEl Mohtadi, F., d’Arcy, R., Yang, X., Turhan, Z. Y., Alshamsan, A., & Tirelli, N. (2019). Main Chain Polysulfoxides as Active ‘Stealth’ Polymers with Additional Antioxidant and Anti-Inflammatory Behaviour. International Journal of Molecular Sciences, 20(18), 4583. https://doi.org/10.3390/ijms20184583







