BTLA Expression on Th1, Th2 and Th17 Effector T-Cells of Patients with Systemic Lupus Erythematosus Is Associated with Active Disease
Abstract
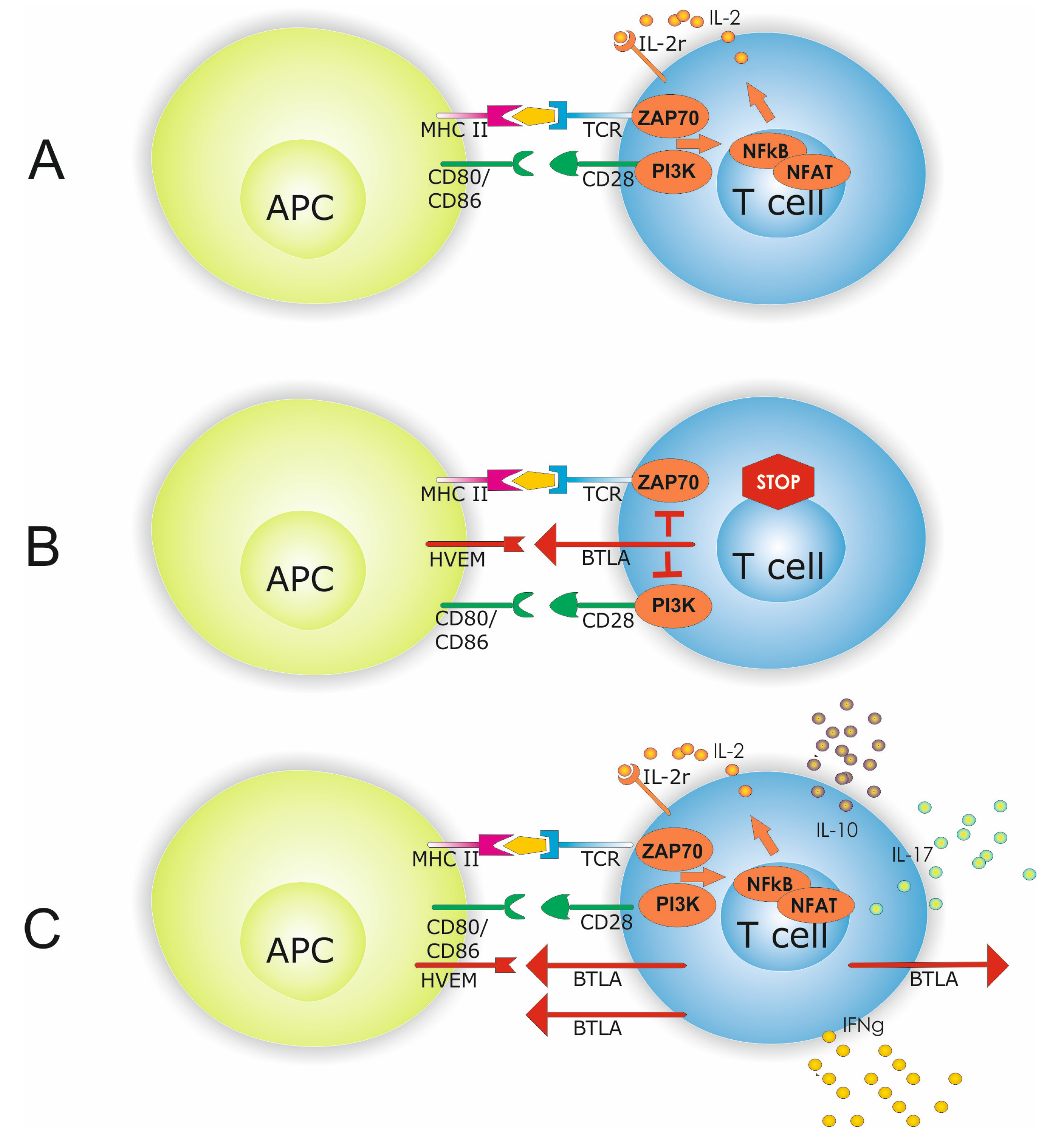
:1. Introduction
2. Results
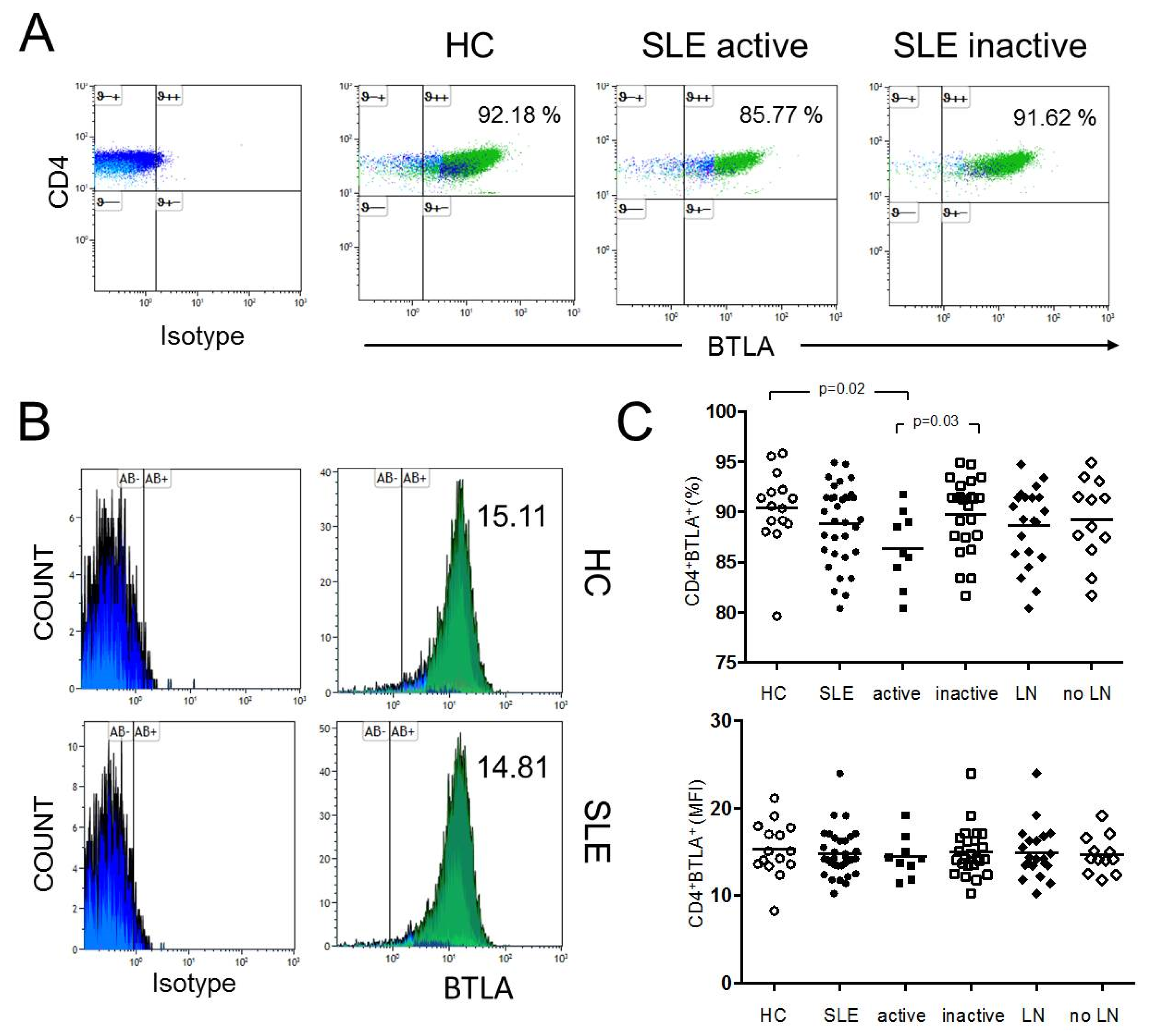
2.1. The Percentages of CD4+ BTLA+ T-cells are Significantly Decreased in Active SLE
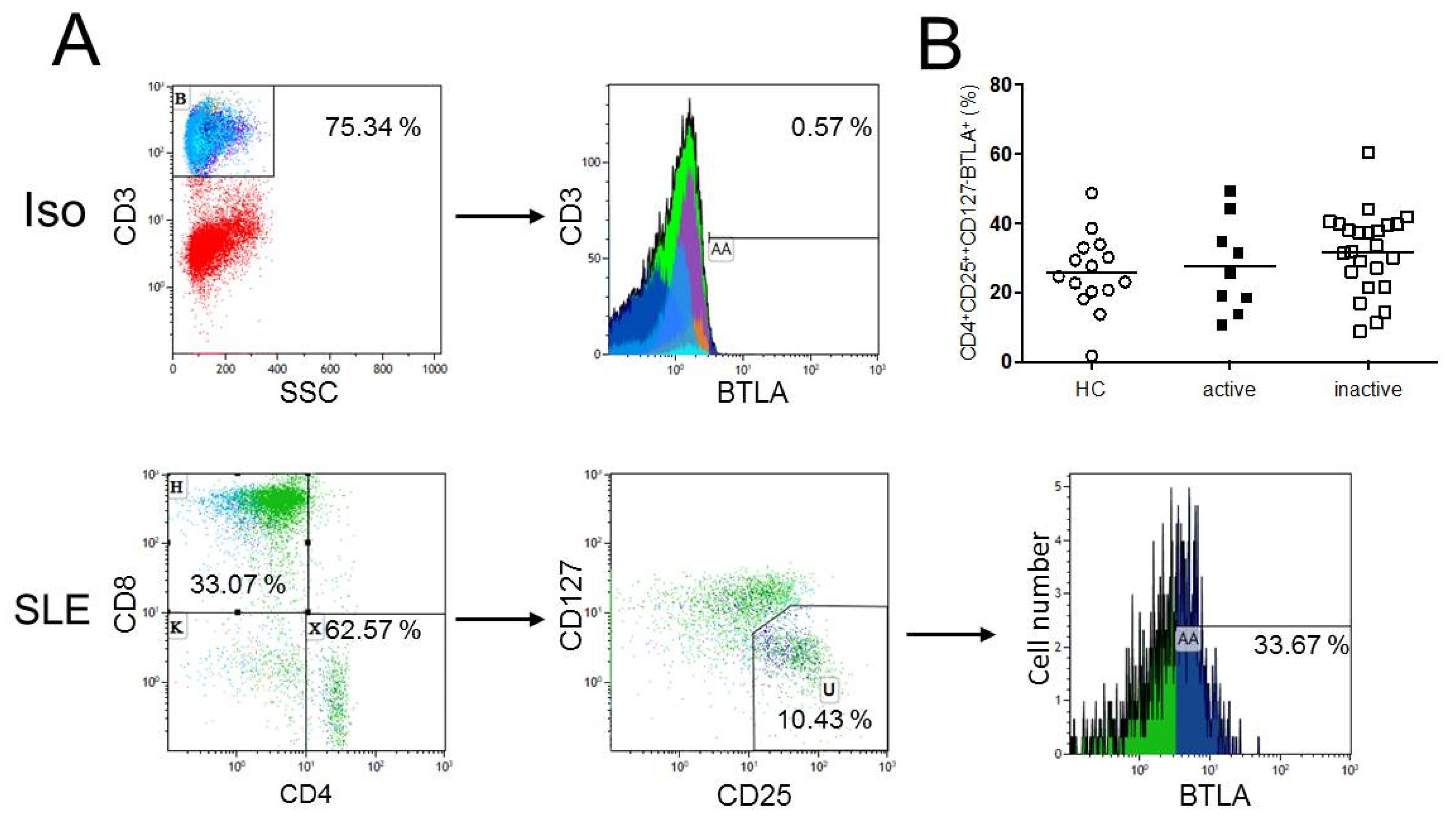
2.2. Expression of BTLA is not Significantly Different on CD4+CD25++CD127− Regulatory T-cells
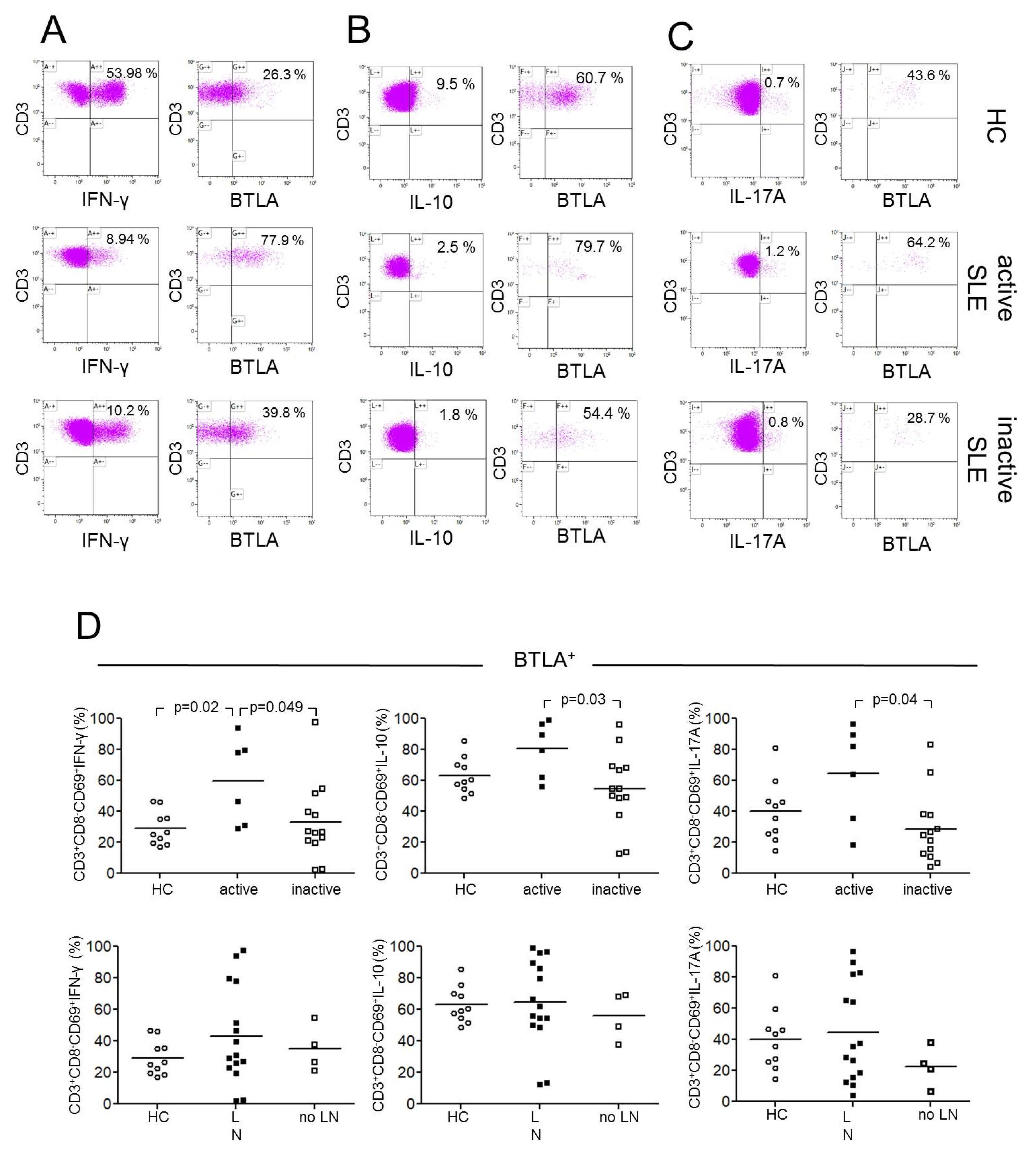
2.3. BTLA Expression on Th1, Th2 and Th17 Effector T-cells
2.4. BTLA Expression on Th1, Th2 and Th17 Effector T-cells in Lupus Nephritis
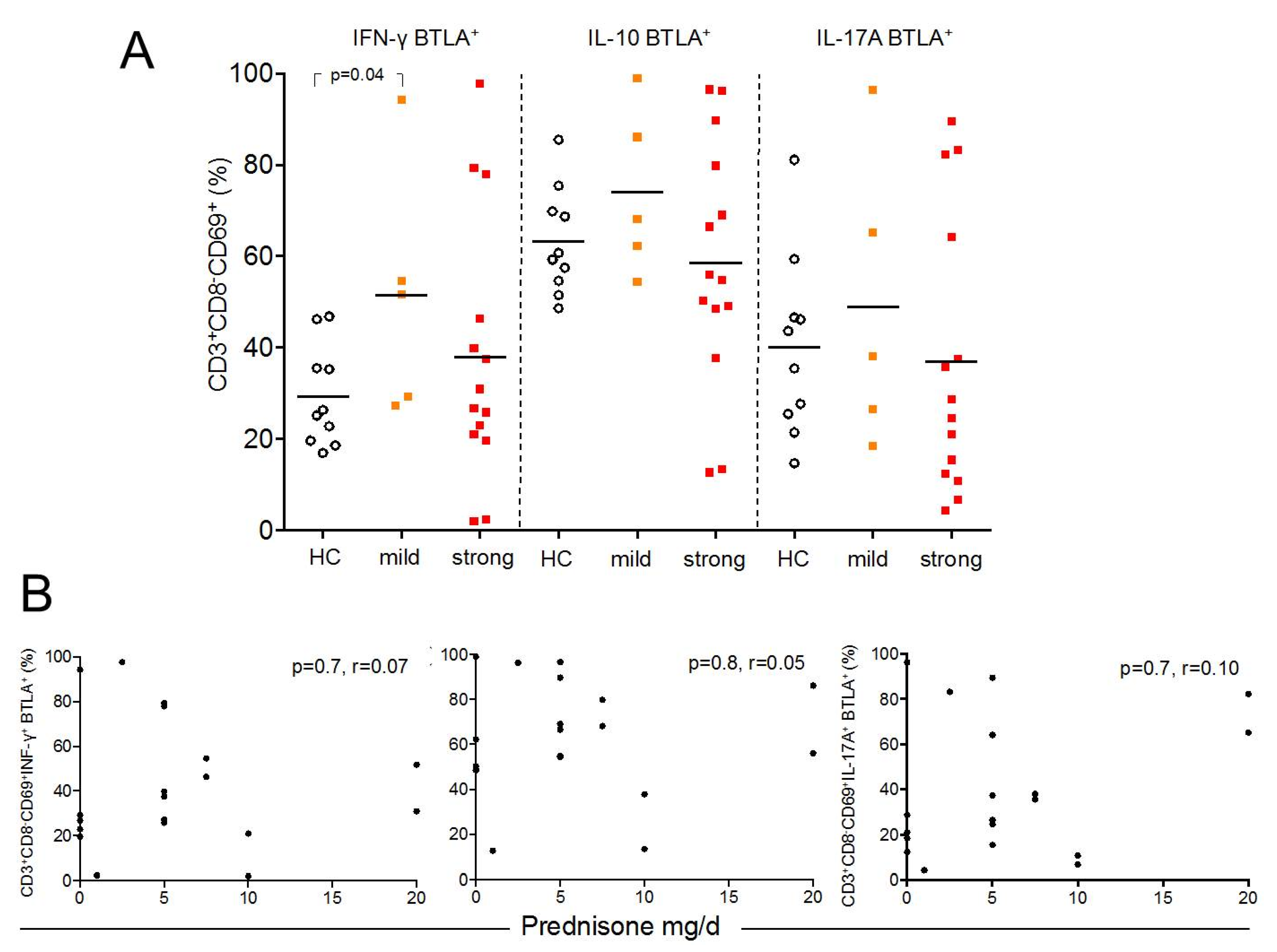
2.5. BTLA Expression on Th1, Th2, and Th17 Effector T-cells is Independent of Treatment
3. Discussion
4. Materials and Methods
4.1. Antibodies
4.2. Immunophenotyping
4.3. Cell Isolation and Ex Vivo Stimulation
4.4. Staining
4.5. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Dolff, S.; Witzke, O.; Wilde, B. Th17 cells in renal inflammation and autoimmunity. Autoimmun. Rev. 2019, 18, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Salomon, B.; Bluestone, J.A. Complexities of CD28/B7: CTLA-4 costimulatory pathways in autoimmunity and transplantation. Annu. Rev. Immunol. 2001, 19, 225–252. [Google Scholar] [CrossRef]
- Wells, A.D.; Walsh, M.C.; Bluestone, J.A.; Turka, L.A. Signaling through CD28 and CTLA-4 controls two distinct forms of T cell anergy. J. Clin. Investig. 2001, 108, 895–903. [Google Scholar] [CrossRef]
- Le Mercier, I.; Lines, J.L.; Noelle, R.J. Beyond CTLA-4 and PD-1, the Generation Z of Negative Checkpoint Regulators. Front. Immunol. 2015, 6, 418. [Google Scholar] [CrossRef] [PubMed]
- Van den Brom, R.R.; Abdulahad, W.H.; Rutgers, A.; Kroesen, B.J.; Roozendaal, C.; de Groot, D.J.; Schroder, C.P.; Hospers, G.A.; Brouwer, E. Rapid granulomatosis with polyangiitis induced by immune checkpoint inhibition. Rheumatology 2016, 55, 1143–1145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bertsias, G.K.; Nakou, M.; Choulaki, C.; Raptopoulou, A.; Papadimitraki, E.; Goulielmos, G.; Kritikos, H.; Sidiropoulos, P.; Tzardi, M.; Kardassis, D.; et al. Genetic, immunologic, and immunohistochemical analysis of the programmed death 1/programmed death ligand 1 pathway in human systemic lupus erythematosus. Arthritis Rheum. 2009, 60, 207–218. [Google Scholar] [CrossRef] [PubMed]
- Wilde, B.; Hua, F.; Dolff, S.; Jun, C.; Cai, X.; Specker, C.; Feldkamp, T.; Kribben, A.; Cohen Tervaert, J.W.; Witzke, O. Aberrant expression of the negative costimulator PD-1 on T cells in granulomatosis with polyangiitis. Rheumatology 2012, 51, 1188–1197. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dolff, S.; Quandt, D.; Feldkamp, T.; Jun, C.; Mitchell, A.; Hua, F.; Specker, C.; Kribben, A.; Witzke, O.; Wilde, B. Increased percentages of PD-1 on CD4+ T cells is associated with higher INF-gamma production and altered IL-17 production in patients with systemic lupus erythematosus. Scand. J. Rheumatol. 2014, 43, 307–313. [Google Scholar] [CrossRef] [PubMed]
- Sedy, J.R.; Gavrieli, M.; Potter, K.G.; Hurchla, M.A.; Lindsley, R.C.; Hildner, K.; Scheu, S.; Pfeffer, K.; Ware, C.F.; Murphy, T.L.; et al. B and T lymphocyte attenuator regulates T cell activation through interaction with herpesvirus entry mediator. Nat. Immunol. 2005, 6, 90–98. [Google Scholar] [CrossRef] [PubMed]
- Han, P.; Goularte, O.D.; Rufner, K.; Wilkinson, B.; Kaye, J. An inhibitory Ig superfamily protein expressed by lymphocytes and APCs is also an early marker of thymocyte positive selection. J. Immunol. 2004, 172, 5931–5939. [Google Scholar] [CrossRef]
- Zhang, X.; Lindwall, E.; Gauthier, C.; Lyman, J.; Spencer, N.; Alarakhia, A.; Fraser, A.; Ing, S.; Chen, M.; Webb-Detiege, T.; et al. Circulating CXCR5+CD4+helper T cells in systemic lupus erythematosus patients share phenotypic properties with germinal center follicular helper T cells and promote antibody production. Lupus 2015, 24, 909–917. [Google Scholar] [CrossRef] [PubMed]
- Oya, Y.; Watanabe, N.; Kobayashi, Y.; Owada, T.; Oki, M.; Ikeda, K.; Suto, A.; Kagami, S.; Hirose, K.; Kishimoto, T.; et al. Lack of B and T lymphocyte attenuator exacerbates autoimmune disorders and induces Fas-independent liver injury in MRL-lpr/lpr mice. Int. Immunol. 2011, 23, 335–344. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Testi, R.; Phillips, J.H.; Lanier, L.L. Leu 23 induction as an early marker of functional CD3/T cell antigen receptor triggering. Requirement for receptor cross-linking, prolonged elevation of intracellular [Ca++] and stimulation of protein kinase C. J. Immunol. 1989, 142, 1854–1860. [Google Scholar] [PubMed]
- Abdulahad, W.H.; Stegeman, C.A.; Limburg, P.C.; Kallenberg, C.G. Skewed distribution of Th17 lymphocytes in patients with Wegener’s granulomatosis in remission. Arthritis Rheum. 2008, 58, 2196–2205. [Google Scholar] [CrossRef] [PubMed]
- Sawaf, M.; Fauny, J.D.; Felten, R.; Sagez, F.; Gottenberg, J.E.; Dumortier, H.; Monneaux, F. Defective BTLA functionality is rescued by restoring lipid metabolism in lupus CD4+ T cells. Jci Insight 2018, 3, e99711. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Alexiou, M.; Martin-Orozco, N.; Chung, Y.; Nurieva, R.I.; Ma, L.; Tian, Q.; Kollias, G.; Lu, S.; Graf, D.; et al. Cutting edge: A critical role of B and T lymphocyte attenuator in peripheral T cell tolerance induction. J. Immunol. 2009, 182, 4516–4520. [Google Scholar] [CrossRef] [PubMed]
- Shui, J.W.; Steinberg, M.W.; Kronenberg, M. Regulation of inflammation, autoimmunity, and infection immunity by HVEM-BTLA signaling. J. Leukoc. Biol. 2011, 89, 517–523. [Google Scholar] [CrossRef]
- Shah, K.; Lee, W.W.; Lee, S.H.; Kim, S.H.; Kang, S.W.; Craft, J.; Kang, I. Dysregulated balance of Th17 and Th1 cells in systemic lupus erythematosus. Arthritis Res. Ther. 2010, 12, R53. [Google Scholar] [CrossRef]
- Dolff, S.; Bijl, M.; Huitema, M.G.; Limburg, P.C.; Kallenberg, C.G.; Abdulahad, W.H. Disturbed Th1, Th2, Th17 and T(reg) balance in patients with systemic lupus erythematosus. Clin. Immunol. 2011, 141, 197–204. [Google Scholar] [CrossRef]
- Dolff, S.; Quandt, D.; Wilde, B.; Feldkamp, T.; Hua, F.; Cai, X.; Specker, C.; Kribben, A.; Kallenberg, C.G.; Witzke, O. Increased expression of costimulatory markers CD134 and CD80 on interleukin-17 producing T cells in patients with systemic lupus erythematosus. Arthritis Res. Ther. 2010, 12, R150. [Google Scholar] [CrossRef]
- Cerovec, M.; Anic, B.; Padjen, I.; Cikes, N. Prevalence of the American College of Rheumatology classification criteria in a group of 162 systemic lupus erythematosus patients from Croatia. Croat. Med J. 2012, 53, 149–154. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hochberg, M.C. Updating the American College of Rheumatology revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum. 1997, 40, 1725. [Google Scholar] [CrossRef] [PubMed]
- Dolff, S.; Abdulahad, W.H.; Westra, J.; Doornbos-van der Meer, B.; Limburg, P.C.; Kallenberg, C.G.; Bijl, M. Increase in IL-21 producing T-cells in patients with systemic lupus erythematosus. Arthritis Res. Ther. 2011, 13, R157. [Google Scholar] [CrossRef] [PubMed]
- Pelchen-Matthews, A.; Parsons, I.J.; Marsh, M. Phorbol ester-induced downregulation of CD4 is a multistep process involving dissociation from p56lck, increased association with clathrin-coated pits, and altered endosomal sorting. J. Exp. Med. 1993, 178, 1209–1222. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | SLE Patients | HC | p-value |
|---|---|---|---|
| Total Number | 41 | 21 | |
| Women/Men | 39/2 | 19/2 | ns |
| Age (years, mean ± SD) | 39.7 ± 13.5 | 40.7 ± 13.9 | ns |
| SLEDAI (median (range)) | 2 (0–26) | ||
| Active Patients (SLEDAI > 4) | 12 | ||
| Inactive Patients (SLEDAI ≤ 4) | 29 | ||
| Lupus Nephritis ISN/RPS Classification, n | 28 | ||
| Class II | 8 | ||
| Class III | 4 | ||
| Class IV | 10 | ||
| Class V | 5 | ||
| Not Classified | 1 | ||
| Treatment, n | 21 | ||
| None | 5 | ||
| Glucocorticoids, n | 30 | ||
| median dose (range), dose (mg/day) | 5 (1–80) | ||
| Immunosuppressive/Immunomodulating, n | |||
| Hydroxychloroquine | 17 | ||
| median dose (range), users (mg/day) | 400 (200–400) | ||
| Methotrexate | 1 | ||
| median dose (range), users (mg/week) | 15 | ||
| Azathioprine | 6 | ||
| median dose (range), users (mg/day) | 75 (50–150) | ||
| MMF | 14 | ||
| median dose (range), users (mg/day) | 1750 (500–2000) | ||
| Cyclosporine A | 2 | ||
| median dose (range), users (mg/day) | 287.5 (250–325) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oster, C.; Wilde, B.; Specker, C.; Sun, M.; Kribben, A.; Witzke, O.; Dolff, S. BTLA Expression on Th1, Th2 and Th17 Effector T-Cells of Patients with Systemic Lupus Erythematosus Is Associated with Active Disease. Int. J. Mol. Sci. 2019, 20, 4505. https://doi.org/10.3390/ijms20184505
Oster C, Wilde B, Specker C, Sun M, Kribben A, Witzke O, Dolff S. BTLA Expression on Th1, Th2 and Th17 Effector T-Cells of Patients with Systemic Lupus Erythematosus Is Associated with Active Disease. International Journal of Molecular Sciences. 2019; 20(18):4505. https://doi.org/10.3390/ijms20184505
Chicago/Turabian StyleOster, Christoph, Benjamin Wilde, Christof Specker, Ming Sun, Andreas Kribben, Oliver Witzke, and Sebastian Dolff. 2019. "BTLA Expression on Th1, Th2 and Th17 Effector T-Cells of Patients with Systemic Lupus Erythematosus Is Associated with Active Disease" International Journal of Molecular Sciences 20, no. 18: 4505. https://doi.org/10.3390/ijms20184505
APA StyleOster, C., Wilde, B., Specker, C., Sun, M., Kribben, A., Witzke, O., & Dolff, S. (2019). BTLA Expression on Th1, Th2 and Th17 Effector T-Cells of Patients with Systemic Lupus Erythematosus Is Associated with Active Disease. International Journal of Molecular Sciences, 20(18), 4505. https://doi.org/10.3390/ijms20184505






