Streptococcal Pyrogenic Exotoxin A-Stimulated Monocytes Mediate Regulatory T-Cell Accumulation through PD-L1 and Kynurenine
Abstract
1. Introduction
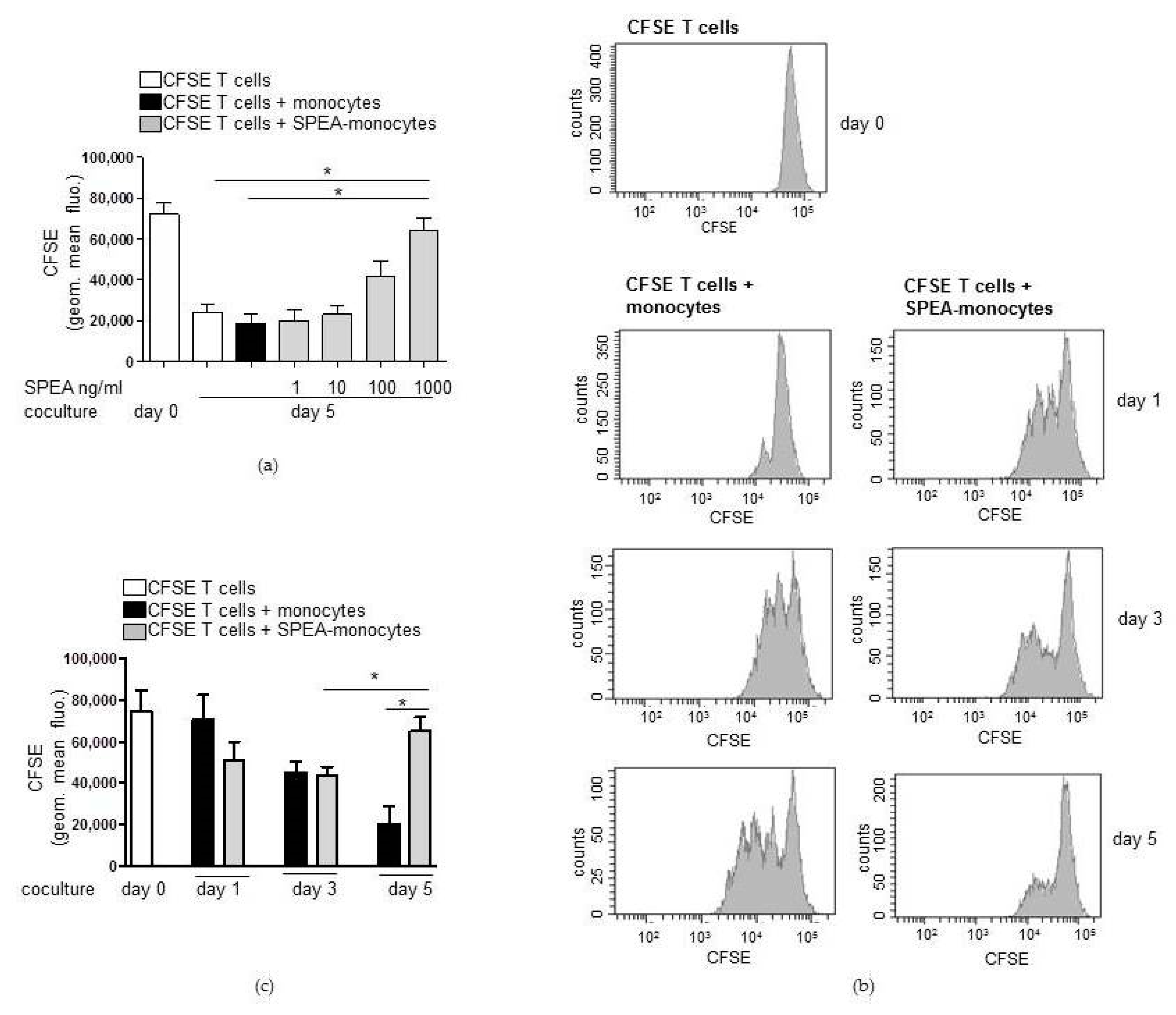
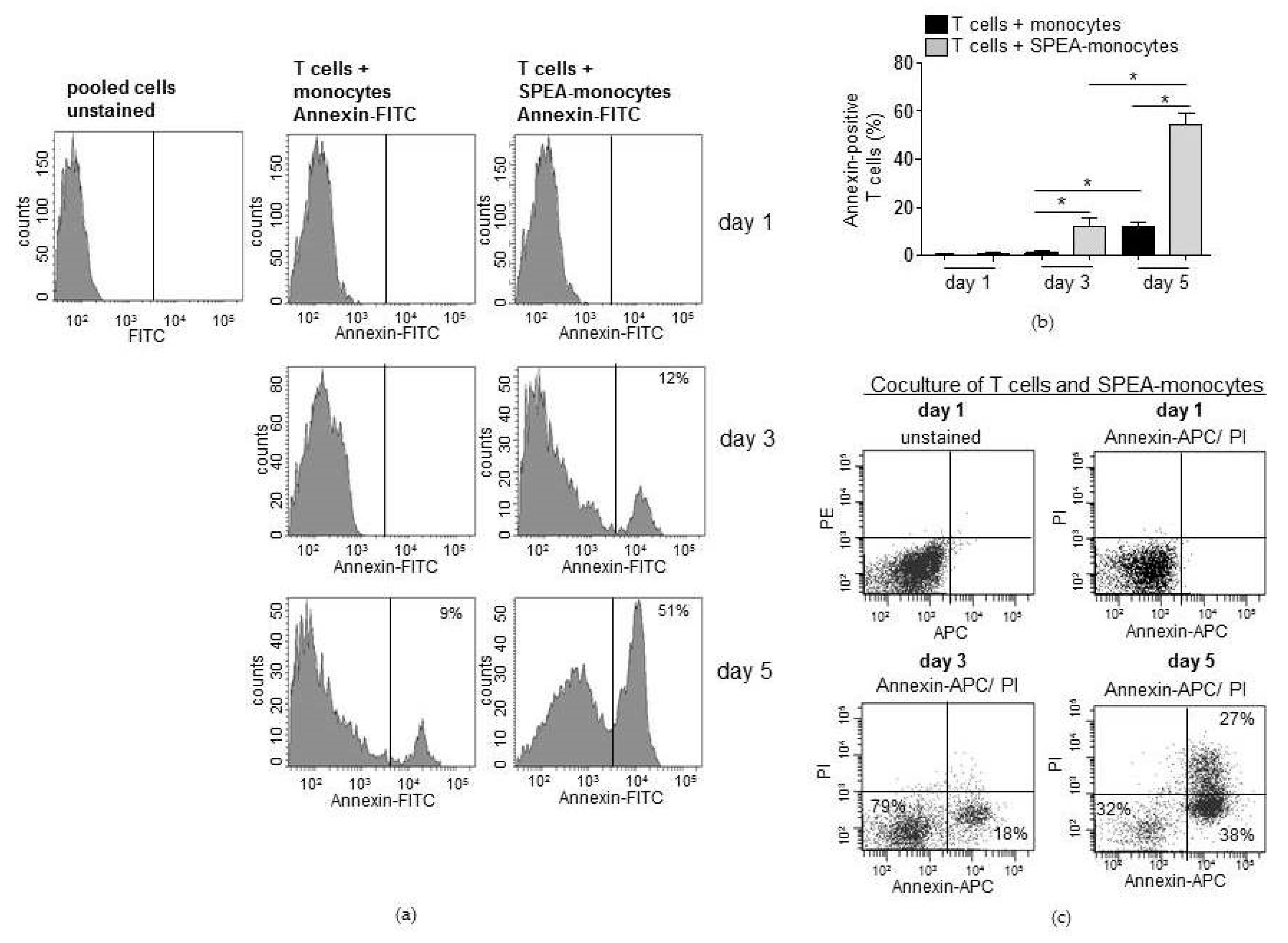
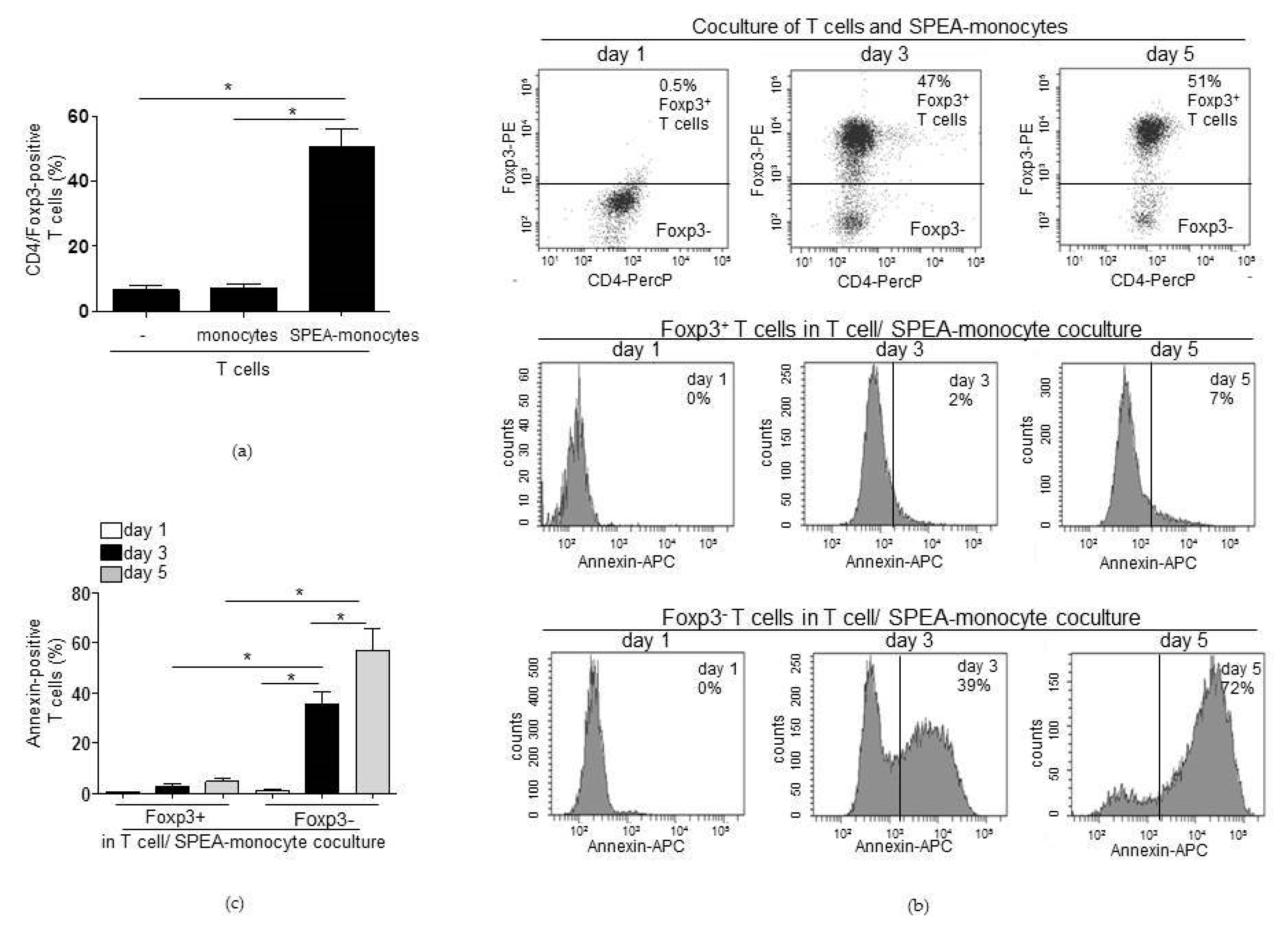
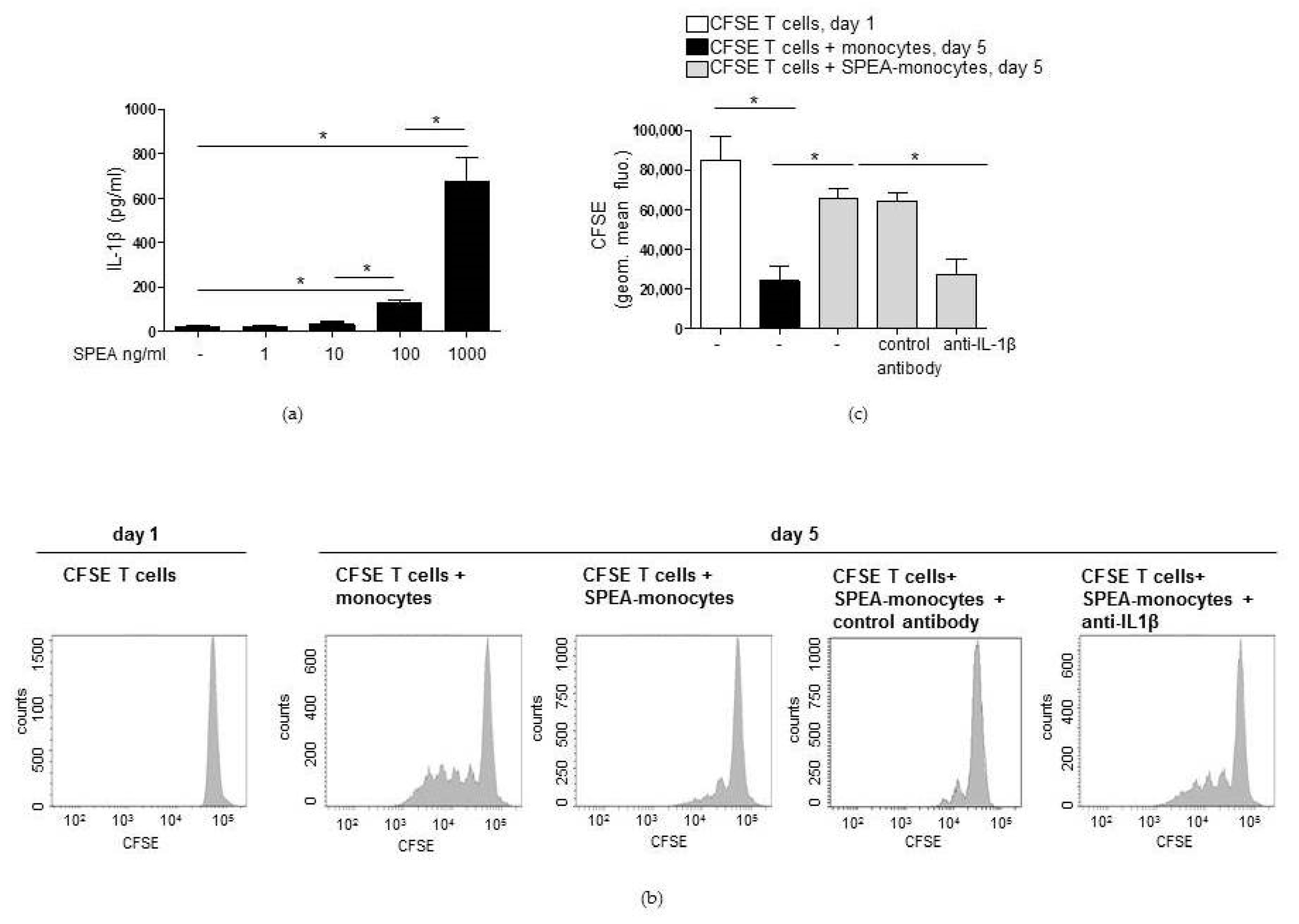
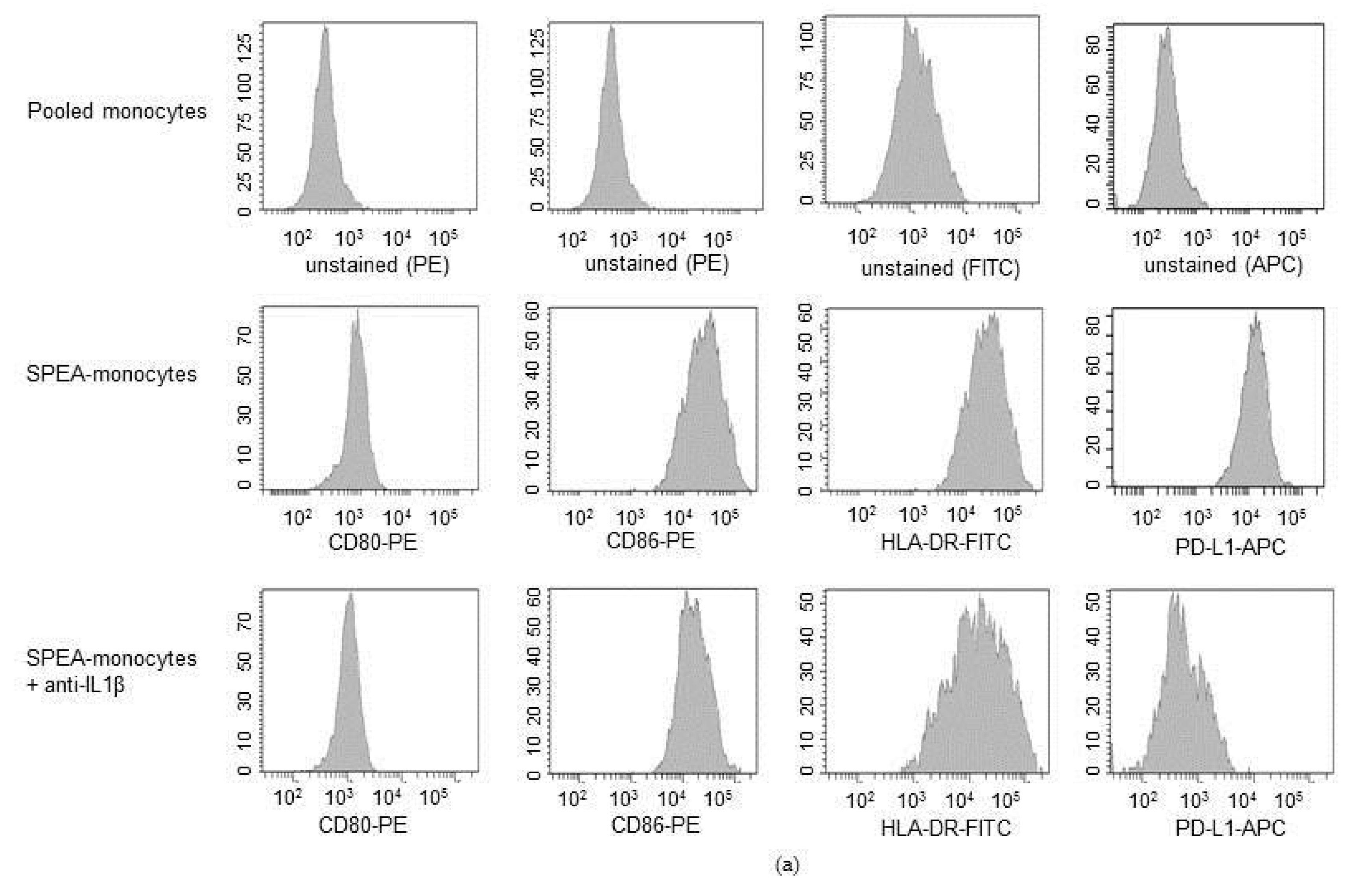
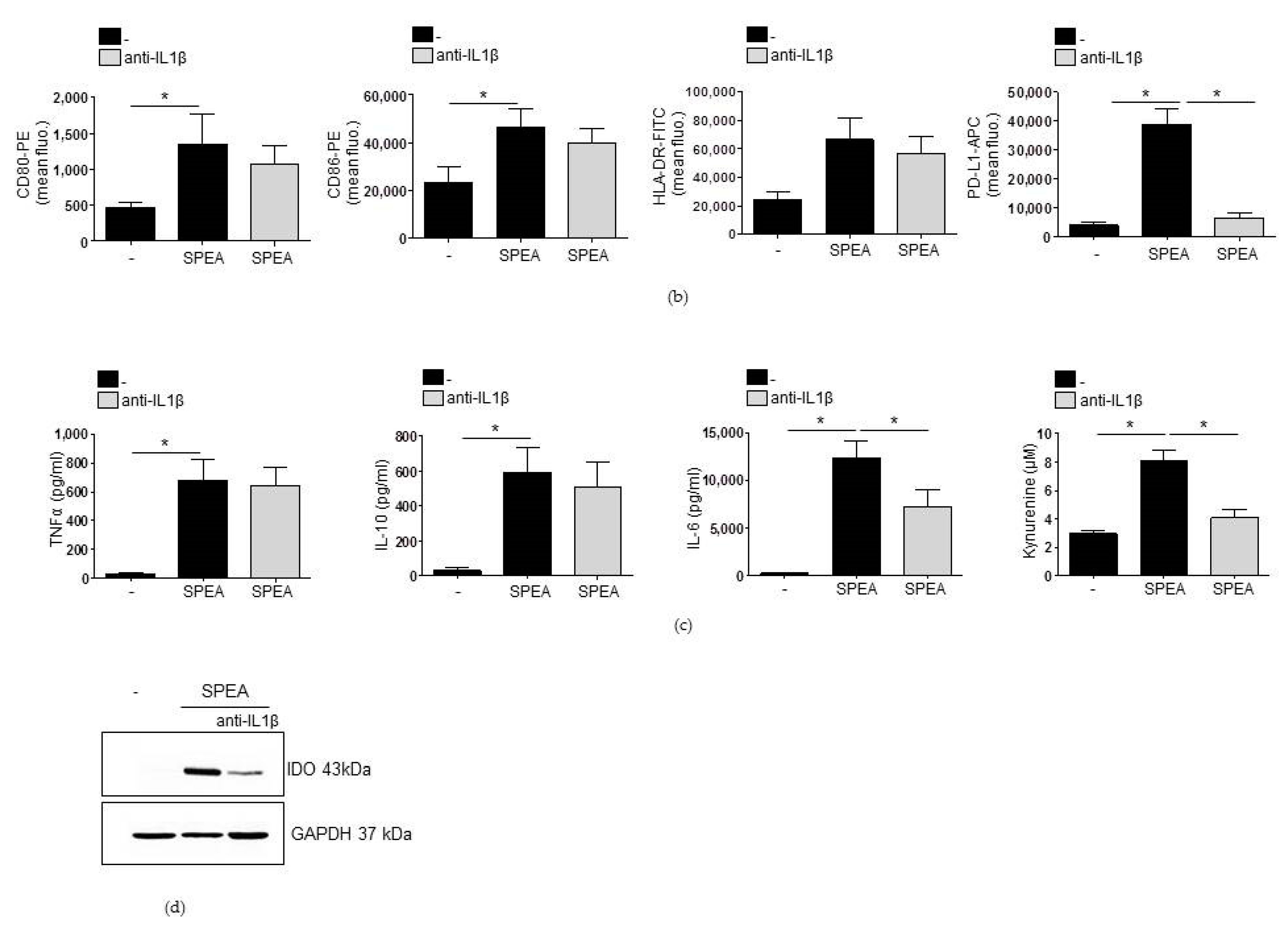
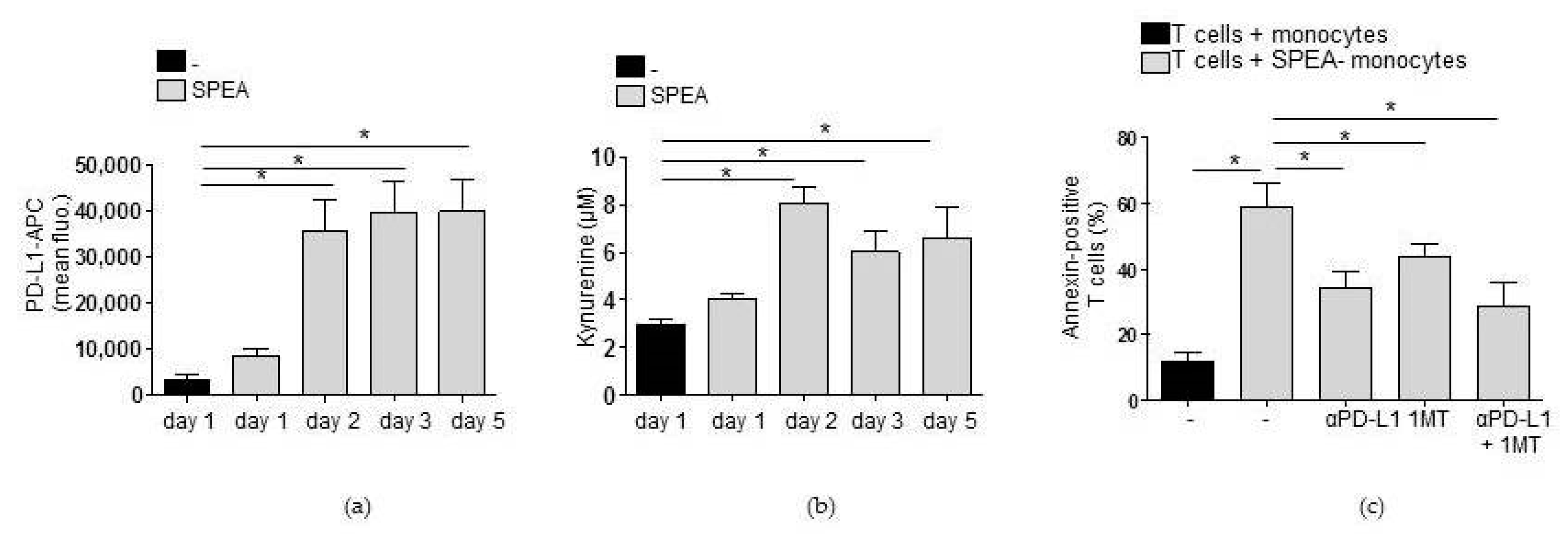
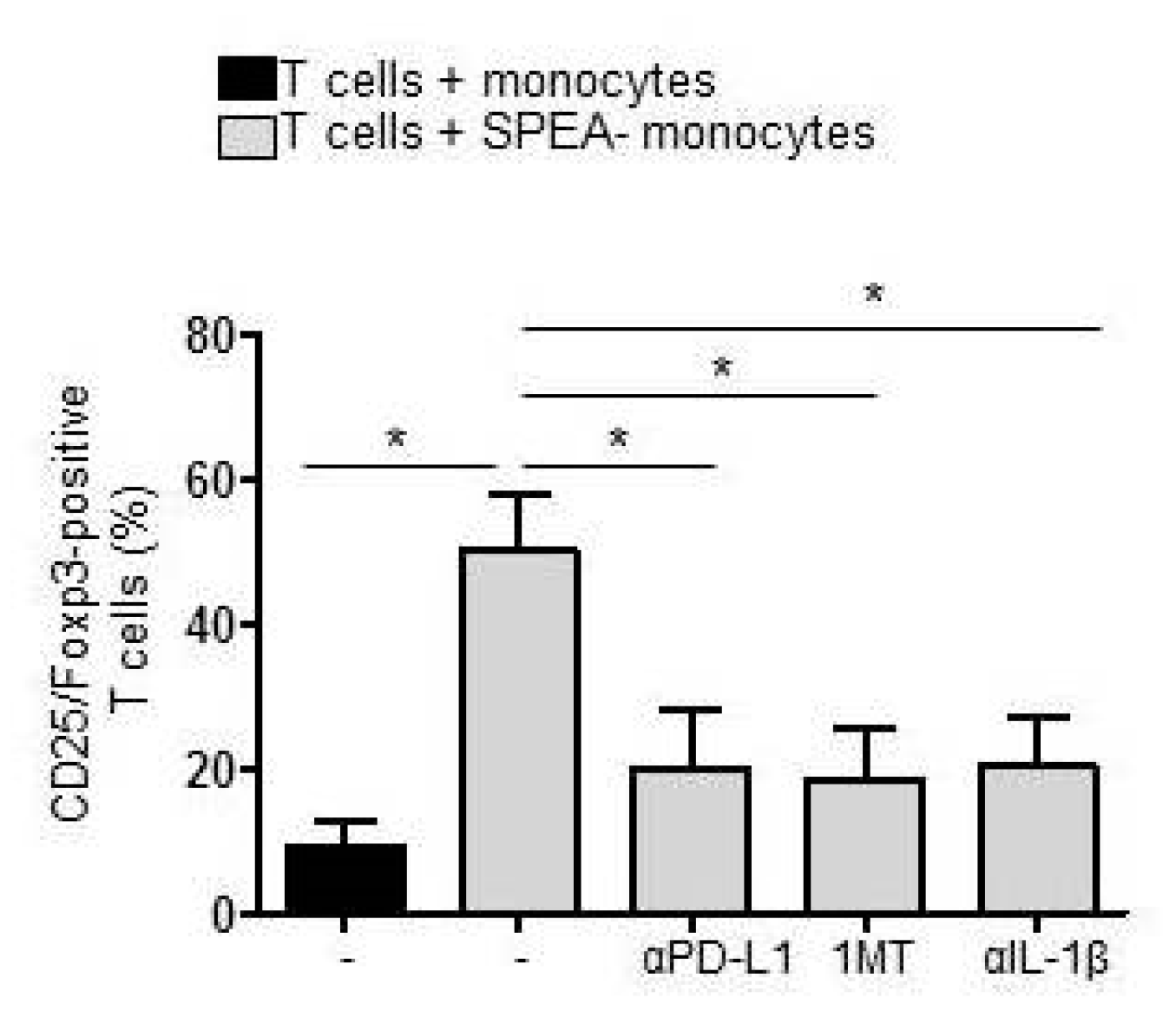
2. Results
3. Discussion
4. Materials and Methods
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Carapetis, J.R.; Steer, A.C.; Mulholland, E.K.; Weber, M. The global burden of group A streptococcal diseases. Lancet Infect. Dis. 2005, 5, 685–694. [Google Scholar] [CrossRef]
- Parks, T.; Smeesters, P.R.; Steer, A.C. Streptococcal skin infection and rheumatic heart disease. Curr. Opin. Infect. Dis. 2012, 25, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Lappin, E.; Ferguson, A.J. Gram-positive toxic shock syndromes. Lancet Infect. Dis. 2009, 9, 281–290. [Google Scholar] [CrossRef]
- Smeesters, P.R.; Dreze, P.A.; Perez-Morga, D.; Biarent, D.; Van Melderen, L.; Vergison, A. Group A streptococcus virulence and host factors in two toddlers with rheumatic fever following toxic shock syndrome. Int. J. Infect. Dis. 2010, 14, E403–E409. [Google Scholar] [CrossRef] [PubMed]
- Johansson, L.; Thulin, P.; Low, D.E.; Norrby-Teglund, A. Getting under the skin: The immunopathogenesis of streptococcus pyogenes deep tissue infections. Clin. Infect. Dis. 2010, 51, 58–65. [Google Scholar] [CrossRef] [PubMed]
- McCormick, J.K.; Yarwood, J.M.; Schlievert, P.M. Toxic shock syndrome and bacterial superantigens: An update. Annu. Rev. Microbiol. 2001, 55, 77–104. [Google Scholar] [CrossRef] [PubMed]
- Commons, R.J.; Smeesters, P.R.; Proft, T.; Fraser, J.D.; Robins-Browne, R.; Curtis, N. Streptococcal superantigens: Categorization and clinical associations. Trends Mol. Med. 2014, 20, 48–62. [Google Scholar] [CrossRef] [PubMed]
- Kasper, K.J.; Zeppa, J.J.; Wakabayashi, A.T.; Xu, S.X.; Mazzuca, D.M.; Welch, I.; Baroja, M.L.; Kotb, M.; Cairns, E.; Cleary, P.P.; et al. Bacterial superantigens promote acute nasopharyngeal infection by streptococcus pyogenes in a human mhc class ii-dependent manner. PLoS Pathog. 2014, 10, e1004155. [Google Scholar] [CrossRef]
- Zeppa, J.J.; Kasper, K.J.; Mohorovic, I.; Mazzuca, D.M.; Haeryfar, S.M.M.; McCormick, J.K. Nasopharyngeal infection by streptococcus pyogenes requires superantigen-responsive Vβ-specific T cells. Proc. Natl. Acad. Sci. USA 2017, 114, 10226–10231. [Google Scholar] [CrossRef]
- Miethke, T.; Duschek, K.; Wahl, C.; Heeg, K.; Wagner, H. Pathogenesis of the toxic shock syndrome: T cell mediated lethal shock caused by the superantigen TSST-1. Eur. J. Immunol. 1993, 23, 1494–1500. [Google Scholar] [CrossRef]
- Fraser, J.D.; Proft, T. The bacterial superantigen and superantigen-like proteins. Immunol. Rev. 2008, 225, 226–243. [Google Scholar] [CrossRef]
- Bohach, G.A.; Fast, D.J.; Nelson, R.D.; Schlievert, P.M. Staphylococcal and streptococcal pyrogenic toxins involved in toxic shock syndrome and related illnesses. Crit. Rev. Microbiol. 1990, 17, 251–272. [Google Scholar] [CrossRef] [PubMed]
- Miethke, T.; Wahl, C.; Holzmann, B.; Heeg, K.; Wagner, H. Bacterial superantigens induce rapid and T cell receptor V beta-selective down-regulation of L-selectin (gp90Mel-14) in vivo. J. Immunol. 1993, 151, 6777–6782. [Google Scholar] [PubMed]
- Spaulding, A.R.; Salgado-Pabon, W.; Kohler, P.L.; Horswill, A.R.; Leung, D.Y.; Schlievert, P.M. Staphylococcal and streptococcal superantigen exotoxins. Clin. Microbiol. Rev. 2013, 26, 422–447. [Google Scholar] [CrossRef] [PubMed]
- Michie, C.; Scott, A.; Cheesbrough, J.; Beverley, P.; Pasvol, G. Streptococcal toxic shock-like syndrome: Evidence of superantigen activity and its effects on T lymphocyte subsets in vivo. Clin. Exp. Immunol. 1994, 98, 140–144. [Google Scholar] [CrossRef] [PubMed]
- Miethke, T.; Wahl, C.; Heeg, K.; Echtenacher, B.; Krammer, P.H.; Wagner, H. T cell-mediated lethal shock triggered in mice by the superantigen staphylococcal enterotoxin b: Critical role of tumor necrosis factor. J. Exp. Med. 1992, 175, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Carlsson, R.; Sjogren, H.O. Kinetics of IL-2 and interferon-gamma production, expression of IL-2 receptors, and cell proliferation in human mononuclear cells exposed to staphylococcal enterotoxin a. Cell. Immunol. 1985, 96, 175–183. [Google Scholar] [CrossRef]
- Choi, Y.W.; Herman, A.; DiGiusto, D.; Wade, T.; Marrack, P.; Kappler, J. Residues of the variable region of the t-cell-receptor beta-chain that interact with s. Aureus toxin superantigens. Nature 1990, 346, 471–473. [Google Scholar] [CrossRef]
- Kappler, J.; Kotzin, B.; Herron, L.; Gelfand, E.W.; Bigler, R.D.; Boylston, A.; Carrel, S.; Posnett, D.N.; Choi, Y.; Marrack, P. V beta-specific stimulation of human T cells by staphylococcal toxins. Science 1989, 244, 811–813. [Google Scholar] [CrossRef]
- Heeg, K.; Gaus, H.; Griese, D.; Bendigs, S.; Miethke, T.; Wagner, H. Superantigen-reactive T cells that display an anergic phenotype in vitro appear functional in vivo. Int. Immunol. 1995, 7, 105–114. [Google Scholar] [CrossRef]
- Herrmann, T.; Baschieri, S.; Lees, R.K.; MacDonald, H.R. In vivo responses of CD4+ and CD8+ cells to bacterial superantigens. Eur. J. Immunol. 1992, 22, 1935–1938. [Google Scholar] [CrossRef]
- Huang, L.; Crispe, I.N. Superantigen-driven peripheral deletion of T cells. Apoptosis occurs in cells that have lost the alpha/beta T cell receptor. J. Immunol. 1993, 151, 1844–1851. [Google Scholar]
- Lee, W.T.; Vitetta, E.S. Memory T cells are anergic to the superantigen staphylococcal enterotoxin B. J. Exp. Med. 1992, 176, 575–579. [Google Scholar] [CrossRef]
- MacDonald, H.R.; Lees, R.K.; Baschieri, S.; Herrmann, T.; Lussow, A.R. Peripheral T-cell reactivity to bacterial superantigens in vivo: The response/anergy paradox. Immunol. Rev. 1993, 133, 105–117. [Google Scholar] [CrossRef]
- Feunou, P.; Poulin, L.; Habran, C.; Le Moine, A.; Goldman, M.; Braun, M.Y. CD4+CD25+ and CD4+CD25- T cells act respectively as inducer and effector T suppressor cells in superantigen-induced tolerance. J. Immunol. 2003, 171, 3475–3484. [Google Scholar] [CrossRef]
- Ivars, F. Superantigen-induced regulatory T cells in vivo. Chem. Immunol. Allergy 2007, 93, 137–160. [Google Scholar]
- Papiernik, M. Natural CD4+ CD25+ regulatory T cells. Their role in the control of superantigen responses. Immunol. Rev. 2001, 182, 180–189. [Google Scholar] [CrossRef]
- Taylor, A.L.; Cross, E.L.; Llewelyn, M.J. Induction of contact-dependent CD8(+) regulatory T cells through stimulation with staphylococcal and streptococcal superantigens. Immunology 2012, 135, 158–167. [Google Scholar] [CrossRef]
- Taylor, A.L.; Llewelyn, M.J. Superantigen-induced proliferation of human CD4+CD25- T cells is followed by a switch to a functional regulatory phenotype. J. Immunol. 2010, 185, 6591–6598. [Google Scholar] [CrossRef]
- Rink, L.; Nicklas, W.; Alvarez-Ossorio, L.; Fagin, U.; Kirchner, H. Microbial superantigens stimulate T cells by the superantigen bridge and independently by a cytokine pathway. J. Interferon Cytokine Res. 1997, 17, 489–499. [Google Scholar] [CrossRef]
- Mehindate, K.; Thibodeau, J.; Dohlsten, M.; Kalland, T.; Sekaly, R.P.; Mourad, W. Cross-linking of major histocompatibility complex class II molecules by staphylococcal enterotoxin a superantigen is a requirement for inflammatory cytokine gene expression. J. Exp. Med. 1995, 182, 1573–1577. [Google Scholar] [CrossRef]
- Palkama, T.; Hurme, M. Signal transduction mechanisms of HLA-DR-mediated interleukin-1 beta production in human monocytes. Role of protein kinase c and tyrosine kinase activation. Hum. Immunol. 1993, 36, 259–267. [Google Scholar] [CrossRef]
- Trede, N.S.; Castigli, E.; Geha, R.S.; Chatila, T. Microbial superantigens induce NF-kappa B in the human monocytic cell line THP-1. J. Immunol. 1993, 150, 5604–5613. [Google Scholar]
- Espel, E.; Garcia-Sanz, J.A.; Aubert, V.; Menoud, V.; Sperisen, P.; Fernandez, N.; Spertini, F. Transcriptional and translational control of tnf-alpha gene expression in human monocytes by major histocompatibility complex class II ligands. Eur. J. Immunol. 1996, 26, 2417–2424. [Google Scholar] [CrossRef]
- Kim, J.; Urban, R.G.; Strominger, J.L.; Wiley, D.C. Toxic shock syndrome toxin-1 complexed with a class II major histocompatibility molecule HLA-DR1. Science 1994, 266, 1870–1874. [Google Scholar] [CrossRef]
- Jardetzky, T.S.; Brown, J.H.; Gorga, J.C.; Stern, L.J.; Urban, R.G.; Chi, Y.I.; Stauffacher, C.; Strominger, J.L.; Wiley, D.C. Three-dimensional structure of a human class II histocompatibility molecule complexed with superantigen. Nature 1994, 368, 711–718. [Google Scholar] [CrossRef]
- Thibodeau, J.; Cloutier, I.; Lavoie, P.M.; Labrecque, N.; Mourad, W.; Jardetzky, T.; Sekaly, R.P. Subsets of HLA-DR1 molecules defined by SEB and TSST-1 binding. Science 1994, 266, 1874–1878. [Google Scholar] [CrossRef]
- Tiedemann, R.E.; Urban, R.J.; Strominger, J.L.; Fraser, J.D. Isolation of HLA-DR1.(staphylococcal enterotoxin a)2 trimers in solution. Proc. Natl. Acad. Sci. USA 1995, 92, 12156–12159. [Google Scholar] [CrossRef]
- Sahr, A.; Former, S.; Hildebrand, D.; Heeg, K. T-cell activation or tolerization: The yin and yang of bacterial superantigens. Front. Microbiol. 2015, 6, 1153. [Google Scholar] [CrossRef]
- Wolfle, S.J.; Strebovsky, J.; Bartz, H.; Sahr, A.; Arnold, C.; Kaiser, C.; Dalpke, A.H.; Heeg, K. PD-L1 expression on tolerogenic apcs is controlled by STAT-3. Eur. J. Immunol. 2011, 41, 413–424. [Google Scholar] [CrossRef]
- Francisco, L.M.; Sage, P.T.; Sharpe, A.H. The PD-1 pathway in tolerance and autoimmunity. Immunol. Rev. 2010, 236, 219–242. [Google Scholar] [CrossRef]
- Keir, M.E.; Butte, M.J.; Freeman, G.J.; Sharpe, A.H. PD-1 and its ligands in tolerance and immunity. Annu. Rev. Immunol. 2008, 26, 677–704. [Google Scholar] [CrossRef]
- Francisco, L.M.; Salinas, V.H.; Brown, K.E.; Vanguri, V.K.; Freeman, G.J.; Kuchroo, V.K.; Sharpe, A.H. PD-L1 regulates the development, maintenance, and function of induced regulatory T cells. J. Exp. Med. 2009, 206, 3015–3029. [Google Scholar] [CrossRef]
- Freeman, G.J.; Long, A.J.; Iwai, Y.; Bourque, K.; Chernova, T.; Nishimura, H.; Fitz, L.J.; Malenkovich, N.; Okazaki, T.; Byrne, M.C.; et al. Engagement of the PD-1 immunoinhibitory receptor by a novel B7 family member leads to negative regulation of lymphocyte activation. J. Exp. Med. 2000, 192, 1027–1034. [Google Scholar] [CrossRef]
- Riley, J.L. PD-1 signaling in primary T cells. Immunol. Rev. 2009, 229, 114–125. [Google Scholar] [CrossRef]
- Keir, M.E.; Liang, S.C.; Guleria, I.; Latchman, Y.E.; Qipo, A.; Albacker, L.A.; Koulmanda, M.; Freeman, G.J.; Sayegh, M.H.; Sharpe, A.H. Tissue expression of PD-L1 mediates peripheral T cell tolerance. J. Exp. Med. 2006, 203, 883–895. [Google Scholar] [CrossRef]
- Litzenburger, U.M.; Opitz, C.A.; Sahm, F.; Rauschenbach, K.J.; Trump, S.; Winter, M.; Ott, M.; Ochs, K.; Lutz, C.; Liu, X.; et al. Constitutive ido expression in human cancer is sustained by an autocrine signaling loop involving IL-6, STAT3 and the AHR. Oncotarget 2014, 5, 1038–1051. [Google Scholar] [CrossRef]
- Taylor, M.W.; Feng, G.S. Relationship between interferon-gamma, indoleamine 2,3-dioxygenase, and tryptophan catabolism. FASEB J. 1991, 5, 2516–2522. [Google Scholar] [CrossRef]
- Munn, D.H.; Shafizadeh, E.; Attwood, J.T.; Bondarev, I.; Pashine, A.; Mellor, A.L. Inhibition of T cell proliferation by macrophage tryptophan catabolism. J. Exp. Med. 1999, 189, 1363–1372. [Google Scholar] [CrossRef]
- Hill, M.; Tanguy-Royer, S.; Royer, P.; Chauveau, C.; Asghar, K.; Tesson, L.; Lavainne, F.; Remy, S.; Brion, R.; Hubert, F.X.; et al. IDO expands human CD4+CD25high regulatory T cells by promoting maturation of LPS-treated dendritic cells. Eur. J. Immunol. 2007, 37, 3054–3062. [Google Scholar] [CrossRef]
- Mellor, A.L.; Munn, D.H. IDO expression by dendritic cells: Tolerance and tryptophan catabolism. Nat. Rev. Immunol. 2004, 4, 762–774. [Google Scholar] [CrossRef]
- Puccetti, P.; Grohmann, U. IDO and regulatory T cells: A role for reverse signalling and non-canonical NF-kappab activation. Nat. Rev. Immunol. 2007, 7, 817–823. [Google Scholar] [CrossRef]
- Terness, P.; Bauer, T.M.; Rose, L.; Dufter, C.; Watzlik, A.; Simon, H.; Opelz, G. Inhibition of allogeneic T cell proliferation by indoleamine 2,3-dioxygenase-expressing dendritic cells: Mediation of suppression by tryptophan metabolites. J. Exp. Med. 2002, 196, 447–457. [Google Scholar] [CrossRef]
- Giesbrecht, K.; Eberle, M.E.; Wolfle, S.J.; Sahin, D.; Sahr, A.; Oberhardt, V.; Menne, Z.; Bode, K.A.; Heeg, K.; Hildebrand, D. IL-1beta as mediator of resolution that reprograms human peripheral monocytes toward a suppressive phenotype. Front. Immunol. 2017, 8, 899. [Google Scholar] [CrossRef]
- Ganem, M.B.; De Marzi, M.C.; Fernandez-Lynch, M.J.; Jancic, C.; Vermeulen, M.; Geffner, J.; Mariuzza, R.A.; Fernandez, M.M.; Malchiodi, E.L. Uptake and intracellular trafficking of superantigens in dendritic cells. PLoS ONE 2013, 8, e66244. [Google Scholar] [CrossRef]
- Hopkins, P.A.; Fraser, J.D.; Pridmore, A.C.; Russell, H.H.; Read, R.C.; Sriskandan, S. Superantigen recognition by HLA class II on monocytes up-regulates toll-like receptor 4 and enhances proinflammatory responses to endotoxin. Blood 2005, 105, 3655–3662. [Google Scholar] [CrossRef]
- Khan, A.A.; Martin, S.; Saha, B. Seb-induced signaling in macrophages leads to biphasic TNF-alpha. J. Leukoc. Biol. 2008, 83, 1363–1369. [Google Scholar] [CrossRef]
- Miethke, T.; Wahl, C.; Heeg, K.; Wagner, H. Bacterial superantigens induce T cell unresponsiveness in B cell-deficient mice. Eur. J. Immunol. 1995, 25, 3187–3190. [Google Scholar] [CrossRef]
- Wahl, C.; Miethke, T.; Heeg, K.; Wagner, H. Clonal deletion as direct consequence of an in vivo T cell response to bacterial superantigen. Eur. J. Immunol. 1993, 23, 1197–1200. [Google Scholar] [CrossRef]
- Van Parijs, L.; Ibraghimov, A.; Abbas, A.K. The roles of costimulation and Fas in T cell apoptosis and peripheral tolerance. Immunity 1996, 4, 321–328. [Google Scholar] [CrossRef]
- Mogil, R.J.; Radvanyi, L.; Gonzalez-Quintial, R.; Miller, R.; Mills, G.; Theofilopoulos, A.N.; Green, D.R. Fas (CD95) participates in peripheral T cell deletion and associated apoptosis in vivo. Int. Immunol. 1995, 7, 1451–1458. [Google Scholar] [CrossRef]
- Strasser, A.; Harris, A.W.; Huang, D.C.; Krammer, P.H.; Cory, S. Bcl-2 and Fas/APO-1 regulate distinct pathways to lymphocyte apoptosis. EMBO J. 1995, 14, 6136–6147. [Google Scholar] [CrossRef]
- Desbarats, J.; Duke, R.C.; Newell, M.K. Newly discovered role for fas ligand in the cell-cycle arrest of CD4+ T cells. Nat. Med. 1998, 4, 1377–1382. [Google Scholar] [CrossRef]
- Hildeman, D.A.; Zhu, Y.; Mitchell, T.C.; Bouillet, P.; Strasser, A.; Kappler, J.; Marrack, P. Activated T cell death in vivo mediated by proapoptotic bcl-2 family member bim. Immunity 2002, 16, 759–767. [Google Scholar] [CrossRef]
- Gonzalo, J.A.; Tarazona, R.; Schuurman, H.J.; Uytdehaag, F.; Wick, G.; Martinez, C.; Kroemer, G. A single injection of staphylococcus aureus enterotoxin B reduces autoimmunity in MRL/lpr mice. Clin. Immunol. Immunopathol. 1994, 71, 176–182. [Google Scholar] [CrossRef]
- Miethke, T.; Vabulas, R.; Bittlingmaier, R.; Heeg, K.; Wagner, H. Mechanisms of peripheral T cell deletion: Anergized T cells are Fas resistant but undergo proliferation-associated apoptosis. Eur. J. Immunol. 1996, 26, 1459–1467. [Google Scholar] [CrossRef]
- Scott, D.E.; Kisch, W.J.; Steinberg, A.D. Studies of T cell deletion and T cell anergy following in vivo administration of SEB to normal and lupus-prone mice. J. Immunol. 1993, 150, 664–672. [Google Scholar]
- Chao, D.T.; Korsmeyer, S.J. BCL-2 family: Regulators of cell death. Annu. Rev. Immunol. 1998, 16, 395–419. [Google Scholar] [CrossRef]
- Newton, K.; Strasser, A. The Bcl-2 family and cell death regulation. Curr. Opin. Genet. Dev. 1998, 8, 68–75. [Google Scholar] [CrossRef]
- Mitchell, T.; Kappler, J.; Marrack, P. Bystander virus infection prolongs activated T cell survival. J. Immunol. 1999, 162, 4527–4535. [Google Scholar]
- Parry, R.V.; Chemnitz, J.M.; Frauwirth, K.A.; Lanfranco, A.R.; Braunstein, I.; Kobayashi, S.V.; Linsley, P.S.; Thompson, C.B.; Riley, J.L. CTLA-4 and PD-1 receptors inhibit T-cell activation by distinct mechanisms. Mol. Cell. Biol. 2005, 25, 9543–9553. [Google Scholar] [CrossRef]
- Dagenais-Lussier, X.; Aounallah, M.; Mehraj, V.; El-Far, M.; Tremblay, C.; Sekaly, R.P.; Routy, J.P.; van Grevenynghe, J. Kynurenine reduces memory CD4 T-cell survival by interfering with interleukin-2 signaling early during HIV-1 infection. J. Virol. 2016, 90, 7967–7979. [Google Scholar] [CrossRef]
- Fife, B.T.; Bluestone, J.A. Control of peripheral T-cell tolerance and autoimmunity via the CTLA-4 and PD-1 pathways. Immunol. Rev. 2008, 224, 166–182. [Google Scholar] [CrossRef]
- Pierson, W.; Cauwe, B.; Policheni, A.; Schlenner, S.M.; Franckaert, D.; Berges, J.; Humblet-Baron, S.; Schonefeldt, S.; Herold, M.J.; Hildeman, D.; et al. Antiapoptotic Mcl-1 is critical for the survival and niche-filling capacity of Foxp3(+) regulatory T cells. Nat. Immunol. 2013, 14, 959–965. [Google Scholar] [CrossRef]
- Gabriel, S.S.; Bon, N.; Chen, J.; Wekerle, T.; Bushell, A.; Fehr, T.; Cippa, P.E. Distinctive expression of Bcl-2 factors in regulatory T cells determines a pharmacological target to induce immunological tolerance. Front. Immunol. 2016, 7, 73. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giesbrecht, K.; Förmer, S.; Sähr, A.; Heeg, K.; Hildebrand, D. Streptococcal Pyrogenic Exotoxin A-Stimulated Monocytes Mediate Regulatory T-Cell Accumulation through PD-L1 and Kynurenine. Int. J. Mol. Sci. 2019, 20, 3933. https://doi.org/10.3390/ijms20163933
Giesbrecht K, Förmer S, Sähr A, Heeg K, Hildebrand D. Streptococcal Pyrogenic Exotoxin A-Stimulated Monocytes Mediate Regulatory T-Cell Accumulation through PD-L1 and Kynurenine. International Journal of Molecular Sciences. 2019; 20(16):3933. https://doi.org/10.3390/ijms20163933
Chicago/Turabian StyleGiesbrecht, Katharina, Sandra Förmer, Aline Sähr, Klaus Heeg, and Dagmar Hildebrand. 2019. "Streptococcal Pyrogenic Exotoxin A-Stimulated Monocytes Mediate Regulatory T-Cell Accumulation through PD-L1 and Kynurenine" International Journal of Molecular Sciences 20, no. 16: 3933. https://doi.org/10.3390/ijms20163933
APA StyleGiesbrecht, K., Förmer, S., Sähr, A., Heeg, K., & Hildebrand, D. (2019). Streptococcal Pyrogenic Exotoxin A-Stimulated Monocytes Mediate Regulatory T-Cell Accumulation through PD-L1 and Kynurenine. International Journal of Molecular Sciences, 20(16), 3933. https://doi.org/10.3390/ijms20163933



