Zr-89 Immuno-PET Targeting Ectopic ATP Synthase Enables In-Vivo Imaging of Tumor Angiogenesis
Abstract
1. Introduction
2. Results
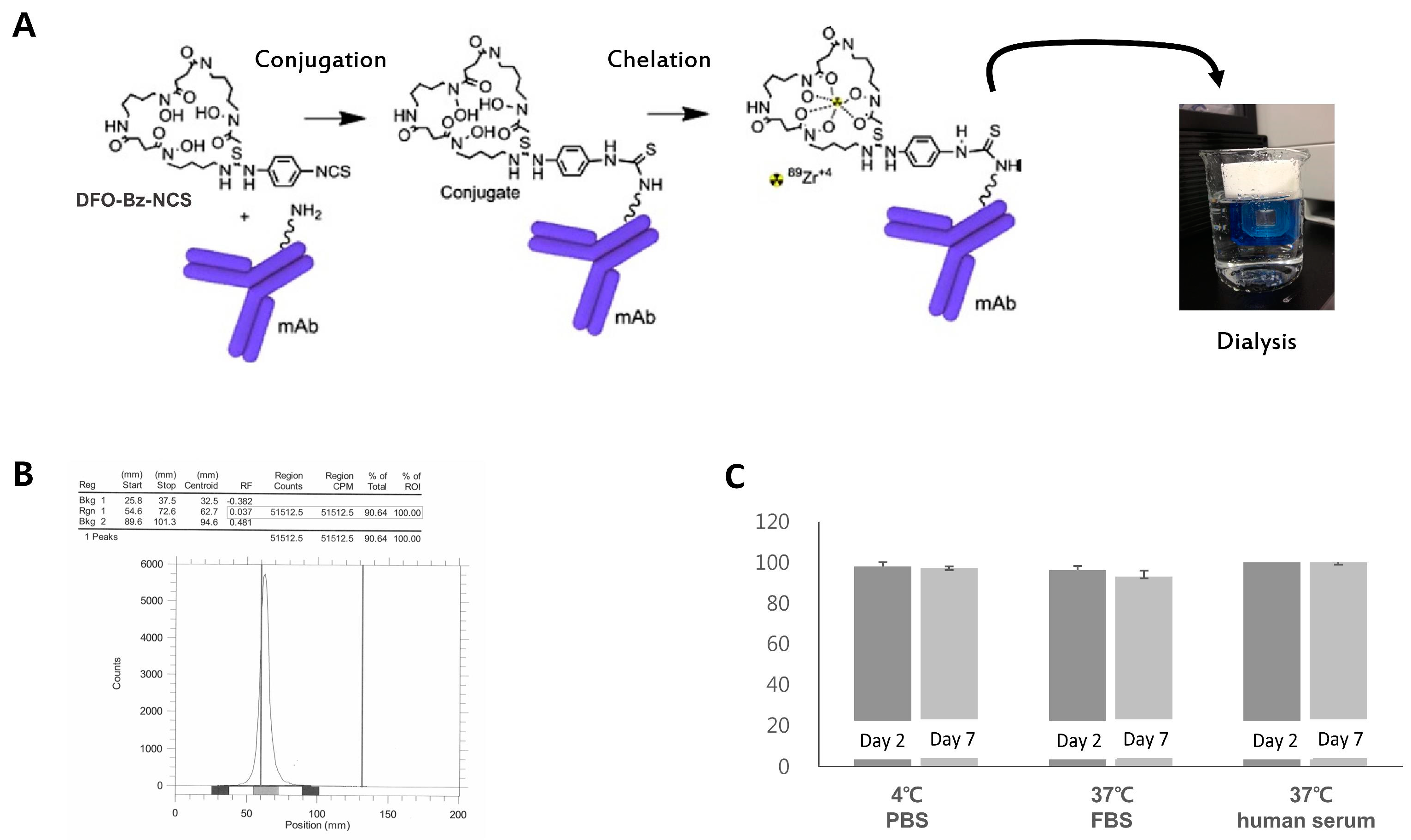
2.1. Radiosynthesis of Zr-89 ATPS mAb
2.2. Western Blotting and Confocal Microscopy for ATPS Expression
2.3. Cell Uptake and Saturation Binding Assay
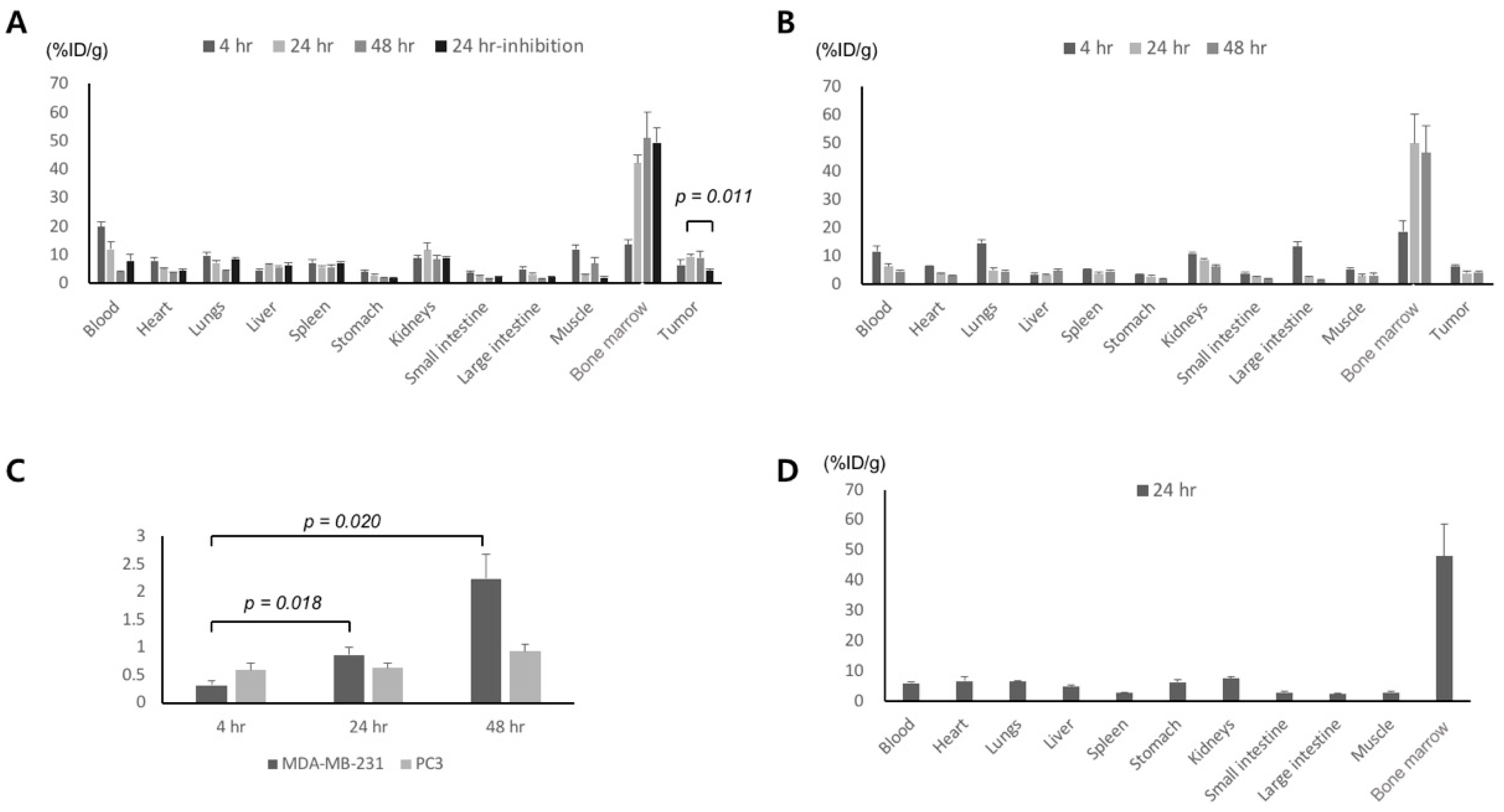
2.4. Biodistribution/Inhibition Study
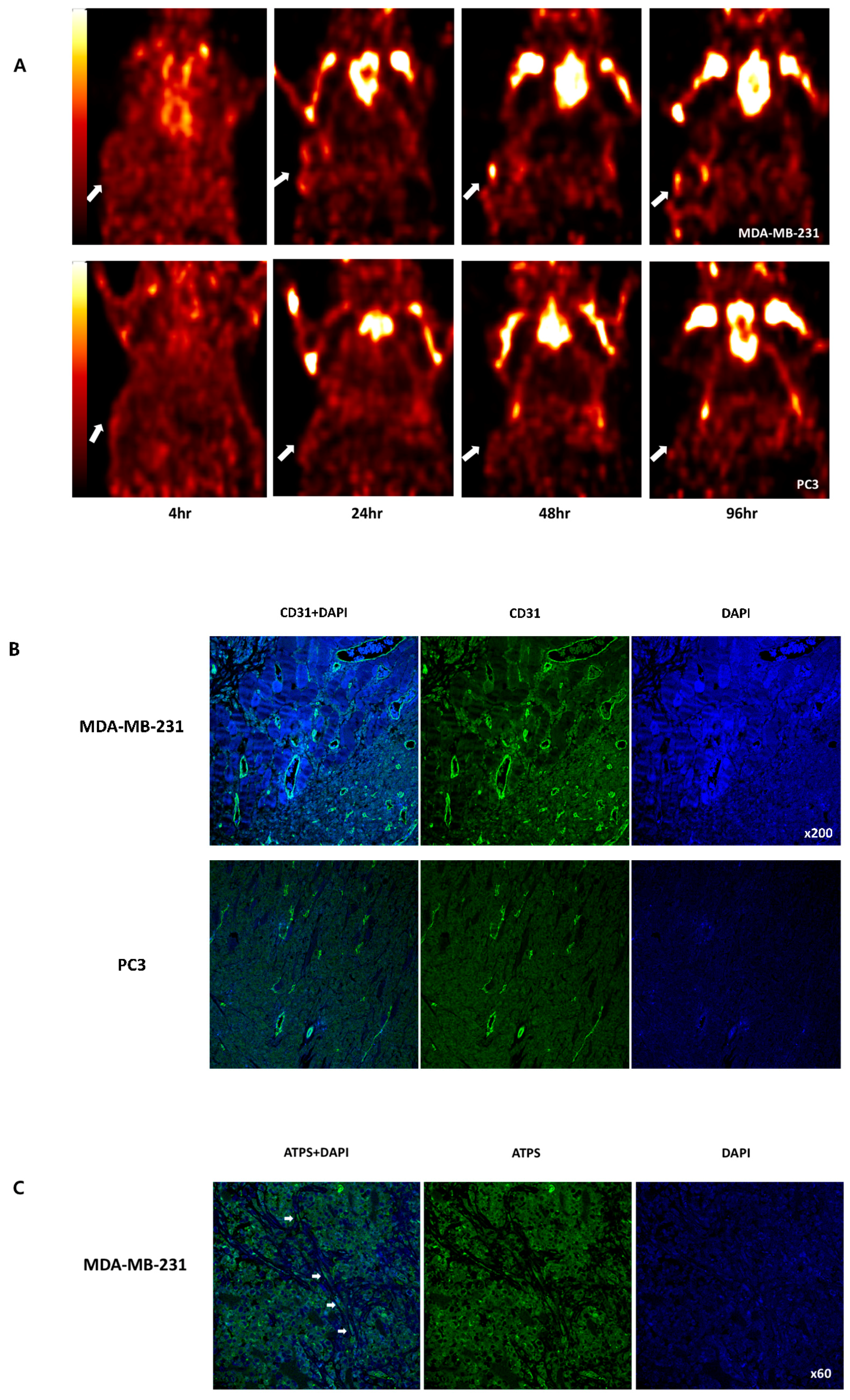
2.5. PET and confocal imaging
3. Discussion
4. Materials and Methods
4.1. Radiosynthesis of Zr-89-Labeled ATPS mAb
4.2. Cancer Cell Lines
4.3. Western Blotting and Confocal Microscopy for ATPS Expression in Cancer Cell Lines
4.4. Cellular Uptake and Saturation Binding Assay
4.5. Xenograft Tumor Model and Biodistribution Analysis
4.6. PET Imaging in Tumor-Bearing Mice
4.7. Confocal Microscopy for Tumor Angiogenesis
4.8. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| VEGF | Vascular endothelial growth factor |
| ATPS | Adenosine Triphosphate Synthase |
| PET | Positron emission tomography |
| FDG | Fluorodeoxyglucose |
| mAb | Monoclonal antibody |
| Her2 | Human epidermal growth factor receptor 2 |
| PSMA | Prostate-Specific Membrane Antigen |
| PBS | Phosphate buffered saline |
| FBS | Fetal bovine serum |
| ID/g | injected dose/gram tissue |
| Df-Bz-NCS | p-isothiocyanatobenzyl-desferrioxamine |
| SUVmax | Maximum standardized uptake value |
References
- Folkman, J. Role of angiogenesis in tumor growth and metastasis. Semin. Oncol. 2002, 29, 15–18. [Google Scholar] [CrossRef] [PubMed]
- Moser, T.L.; Stack, M.S.; Asplin, I.; Enghild, J.J.; Hojrup, P.; Everitt, L.; Hubchak, S.; Schnaper, H.W.; Pizzo, S.V. Angiostatin binds ATP synthase on the surface of human endothelial cells. Proc. Natl. Acad. Sci. USA 1999, 96, 2811–2816. [Google Scholar] [CrossRef] [PubMed]
- Moser, T.L.; Kenan, D.J.; Ashley, T.A.; Roy, J.A.; Goodman, M.D.; Misra, U.K.; Cheek, D.J.; Pizzo, S.V. Endothelial cell surface F1-F0 ATP synthase is active in ATP synthesis and is inhibited by angiostatin. Proc. Natl. Acad. Sci. USA 2001, 98, 6656–6661. [Google Scholar] [CrossRef] [PubMed]
- Chi, S.L.; Wahl, M.L.; Mowery, Y.M.; Shan, S.; Mukhopadhyay, S.; Hilderbrand, S.C.; Kenan, D.J.; Lipes, B.D.; Johnson, C.E.; Marusich, M.F.; et al. Angiostatin-like activity of a monoclonal antibody to the catalytic subunit of F1F0 ATP synthase. Cancer Res. 2007, 67, 4716–4724. [Google Scholar] [CrossRef] [PubMed]
- Hustinx, R.; Benard, F.; Alavi, A. Whole-body FDG-PET imaging in the management of patients with cancer. Semin. Nucl. Med. 2002, 32, 35–46. [Google Scholar] [CrossRef] [PubMed]
- van de Watering, F.C.; Rijpkema, M.; Perk, L.; Brinkmann, U.; Oyen, W.J.; Boerman, O.C. Zirconium-89 labeled antibodies: A new tool for molecular imaging in cancer patients. Biomed. Res. Int. 2014, 2014, 203601. [Google Scholar] [CrossRef] [PubMed]
- Holland, J.P.; Williamson, M.J.; Lewis, J.S. Unconventional nuclides for radiopharmaceuticals. Mol. Imaging 2010, 9, 1–20. [Google Scholar] [CrossRef]
- Fischer, G.; Seibold, U.; Schirrmacher, R.; Wangler, B.; Wangler, C. (89)Zr, a radiometal nuclide with high potential for molecular imaging with PET: Chemistry, applications and remaining challenges. Molecules 2013, 18, 6469–6490. [Google Scholar] [CrossRef] [PubMed]
- Park, B.N.; Lee, S.J.; Roh, J.H.; Lee, K.H.; An, Y.S.; Yoon, J.K. Radiolabeled Anti-Adenosine Triphosphate Synthase Monoclonal Antibody as a Theragnostic Agent Targeting Angiogenesis. Mol. Imaging 2017, 16. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Cao, M.; Liu, Y.; He, Y.; Wang, Y.; Yang, C.; Wang, W.; Du, Y.; Zhou, M.; Gao, F. Mitochondrial F1Fo-ATP synthase translocates to cell surface in hepatocytes and has high activity in tumor-like acidic and hypoxic environment. Acta Biochim. Biophys. Sin. (Shanghai) 2010, 42, 530–537. [Google Scholar] [CrossRef] [PubMed]
- Meijs, W.E.; Herscheid, J.D.; Haisma, H.J.; Pinedo, H.M. Evaluation of desferal as a bifunctional chelating agent for labeling antibodies with Zr-89. Int. J. Rad. Appl. Instrum. A 1992, 43, 1443–1447. [Google Scholar] [CrossRef]
- Chang, A.J.; Desilva, R.; Jain, S.; Lears, K.; Rogers, B.; Lapi, S. 89Zr-Radiolabeled Trastuzumab Imaging in Orthotopic and Metastatic Breast Tumors. Pharmaceuticals (Basel) 2012, 5, 79–93. [Google Scholar] [CrossRef] [PubMed]
- Janjigian, Y.Y.; Viola-Villegas, N.; Holland, J.P.; Divilov, V.; Carlin, S.D.; Gomes-DaGama, E.M.; Chiosis, G.; Carbonetti, G.; de Stanchina, E.; Lewis, J.S. Monitoring afatinib treatment in HER2-positive gastric cancer with 18F-FDG and 89Zr-trastuzumab PET. J. Nucl. Med. 2013, 54, 936–943. [Google Scholar] [CrossRef] [PubMed]
- Schuster, D.M.; Nanni, C.; Fanti, S. PET Tracers Beyond FDG in Prostate Cancer. Semin. Nucl. Med. 2016, 46, 507–521. [Google Scholar] [CrossRef] [PubMed]
- Osborne, J.R.; Green, D.A.; Spratt, D.E.; Lyashchenko, S.; Fareedy, S.B.; Robinson, B.D.; Beattie, B.J.; Jain, M.; Lewis, J.S.; Christos, P.; et al. A prospective pilot study of (89)Zr-J591/prostate specific membrane antigen positron emission tomography in men with localized prostate cancer undergoing radical prostatectomy. J. Urol. 2014, 191, 1439–1445. [Google Scholar] [CrossRef] [PubMed]
- Jodal, L.; Le Loirec, C.; Champion, C. Positron range in PET imaging: Non-conventional isotopes. Phys. Med. Biol. 2014, 59, 7419–7434. [Google Scholar] [CrossRef] [PubMed]
- Severin, G.W.; Jorgensen, J.T.; Wiehr, S.; Rolle, A.M.; Hansen, A.E.; Maurer, A.; Hasenberg, M.; Pichler, B.; Kjaer, A.; Jensen, A.I. The impact of weakly bound (8)(9)Zr on preclinical studies: Non-specific accumulation in solid tumors and aspergillus infection. Nucl. Med. Biol. 2015, 42, 360–368. [Google Scholar] [CrossRef] [PubMed]
- Abou, D.S.; Ku, T.; Smith-Jones, P.M. In vivo biodistribution and accumulation of 89Zr in mice. Nucl. Med. Biol. 2011, 38, 675–681. [Google Scholar] [CrossRef]
- Bradshaw, T.J.; Voorbach, M.J.; Reuter, D.R.; Giamis, A.M.; Mudd, S.R.; Beaver, J.D. Image quality of Zr-89 PET imaging in the Siemens microPET Focus 220 preclinical scanner. Mol. Imaging Biol. 2016, 18, 377–385. [Google Scholar] [CrossRef]
- Bhattacharyya, S.; Kurdziel, K.; Wei, L.; Riffle, L.; Kaur, G.; Hill, G.C.; Jacobs, P.M.; Tatum, J.L.; Doroshow, J.H.; Kalen, J.D. Zirconium-89 labeled panitumumab: A potential immuno-PET probe for HER1-expressing carcinomas. Nucl. Med. Biol. 2013, 40, 451–457. [Google Scholar] [CrossRef] [PubMed]
- Beckford Vera, D.R.; Smith, C.C.; Bixby, L.M.; Glatt, D.M.; Dunn, S.S.; Saito, R.; Kim, W.Y.; Serody, J.S.; Vincent, B.G.; Parrott, M.C. Immuno-PET imaging of tumor-infiltrating lymphocytes using zirconium-89 radiolabeled anti-CD3 antibody in immune-competent mice bearing syngeneic tumors. PLoS ONE 2018, 13, e0193832. [Google Scholar] [CrossRef] [PubMed]
- Sato, N.; Wu, H.; Asiedu, K.O.; Szajek, L.P.; Griffiths, G.L.; Choyke, P.L. (89)Zr-Oxine Complex PET Cell Imaging in Monitoring Cell-based Therapies. Radiology 2015, 275, 490–500. [Google Scholar] [CrossRef] [PubMed]
- Perk, L.R.; Vosjan, M.J.; Visser, G.W.; Budde, M.; Jurek, P.; Kiefer, G.E.; van Dongen, G.A. p-Isothiocyanatobenzyl-desferrioxamine: A new bifunctional chelate for facile radiolabeling of monoclonal antibodies with zirconium-89 for immuno-PET imaging. Eur. J. Nucl. Med. Mol. Imaging 2010, 37, 250–259. [Google Scholar] [CrossRef] [PubMed]
- Yoon, J.K.; Park, B.N.; Paik, J.Y.; Jung, K.H.; Ko, B.H.; Lee, K.H. Effects of theophylline on radioiodide uptake in MCF-7 breast cancer and NIS gene-transduced SNU-C5 colon cancer cells. Cancer Biother. Radiopharm. 2009, 24, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Bigott-Hennkens, H.M.; Dannoon, S.; Lewis, M.R.; Jurisson, S.S. In vitro receptor binding assays: General methods and considerations. Q. J. Nucl. Med. Mol. Imaging 2008, 52, 245–253. [Google Scholar] [PubMed]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, B.-N.; Kim, G.-H.; Ko, S.-A.; Shin, G.-H.; Lee, S.-J.; An, Y.-S.; Yoon, J.-K. Zr-89 Immuno-PET Targeting Ectopic ATP Synthase Enables In-Vivo Imaging of Tumor Angiogenesis. Int. J. Mol. Sci. 2019, 20, 3928. https://doi.org/10.3390/ijms20163928
Park B-N, Kim G-H, Ko S-A, Shin G-H, Lee S-J, An Y-S, Yoon J-K. Zr-89 Immuno-PET Targeting Ectopic ATP Synthase Enables In-Vivo Imaging of Tumor Angiogenesis. International Journal of Molecular Sciences. 2019; 20(16):3928. https://doi.org/10.3390/ijms20163928
Chicago/Turabian StylePark, Bok-Nam, Ga-Hee Kim, Seung-A Ko, Ga-Hee Shin, Su-Jin Lee, Young-Sil An, and Joon-Kee Yoon. 2019. "Zr-89 Immuno-PET Targeting Ectopic ATP Synthase Enables In-Vivo Imaging of Tumor Angiogenesis" International Journal of Molecular Sciences 20, no. 16: 3928. https://doi.org/10.3390/ijms20163928
APA StylePark, B.-N., Kim, G.-H., Ko, S.-A., Shin, G.-H., Lee, S.-J., An, Y.-S., & Yoon, J.-K. (2019). Zr-89 Immuno-PET Targeting Ectopic ATP Synthase Enables In-Vivo Imaging of Tumor Angiogenesis. International Journal of Molecular Sciences, 20(16), 3928. https://doi.org/10.3390/ijms20163928




