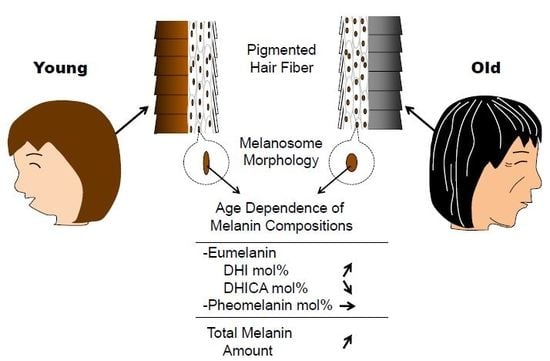
Effects of Aging on Hair Color, Melanosome Morphology, and Melanin Composition in Japanese Females
Abstract
1. Introduction
2. Results
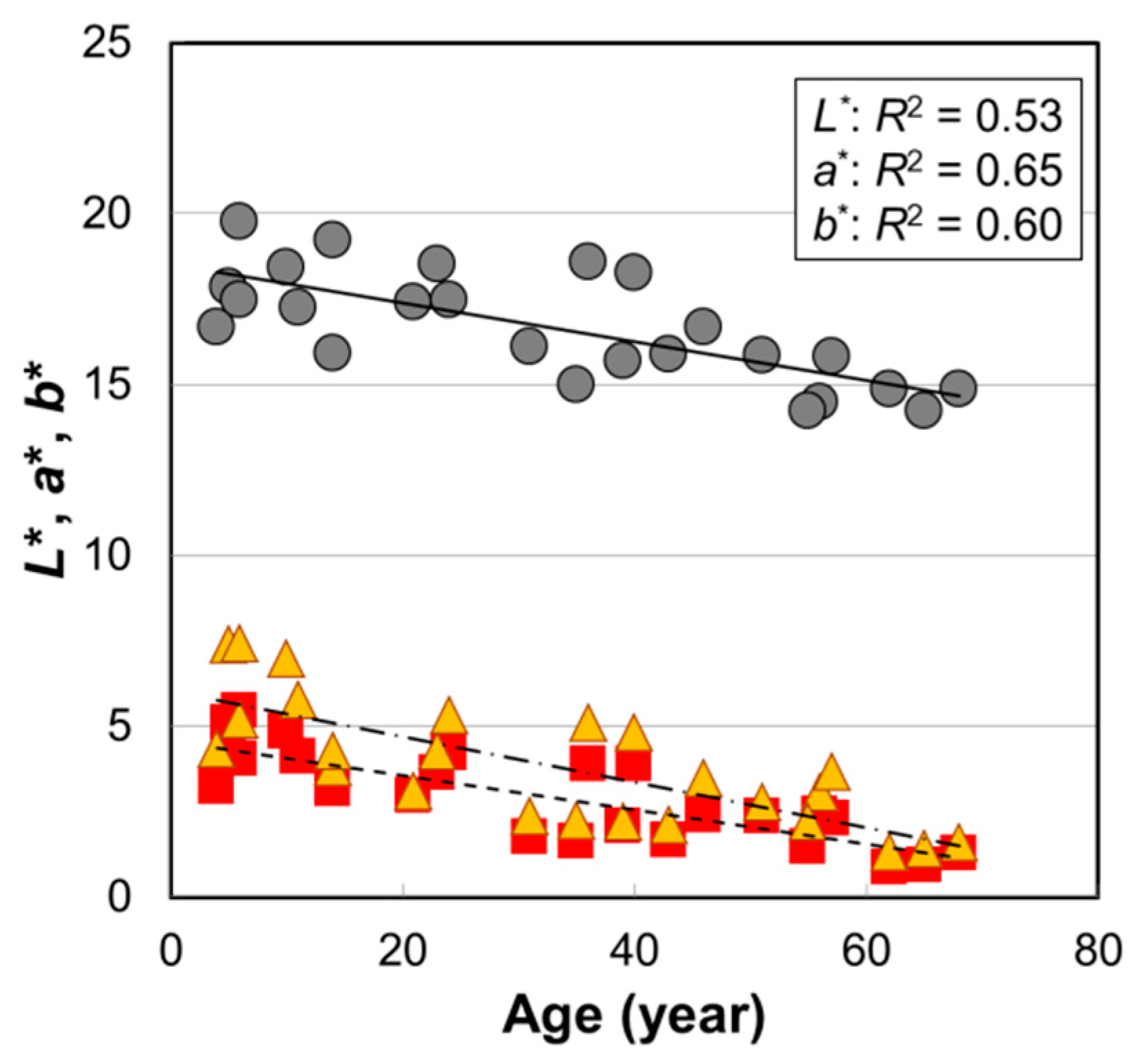
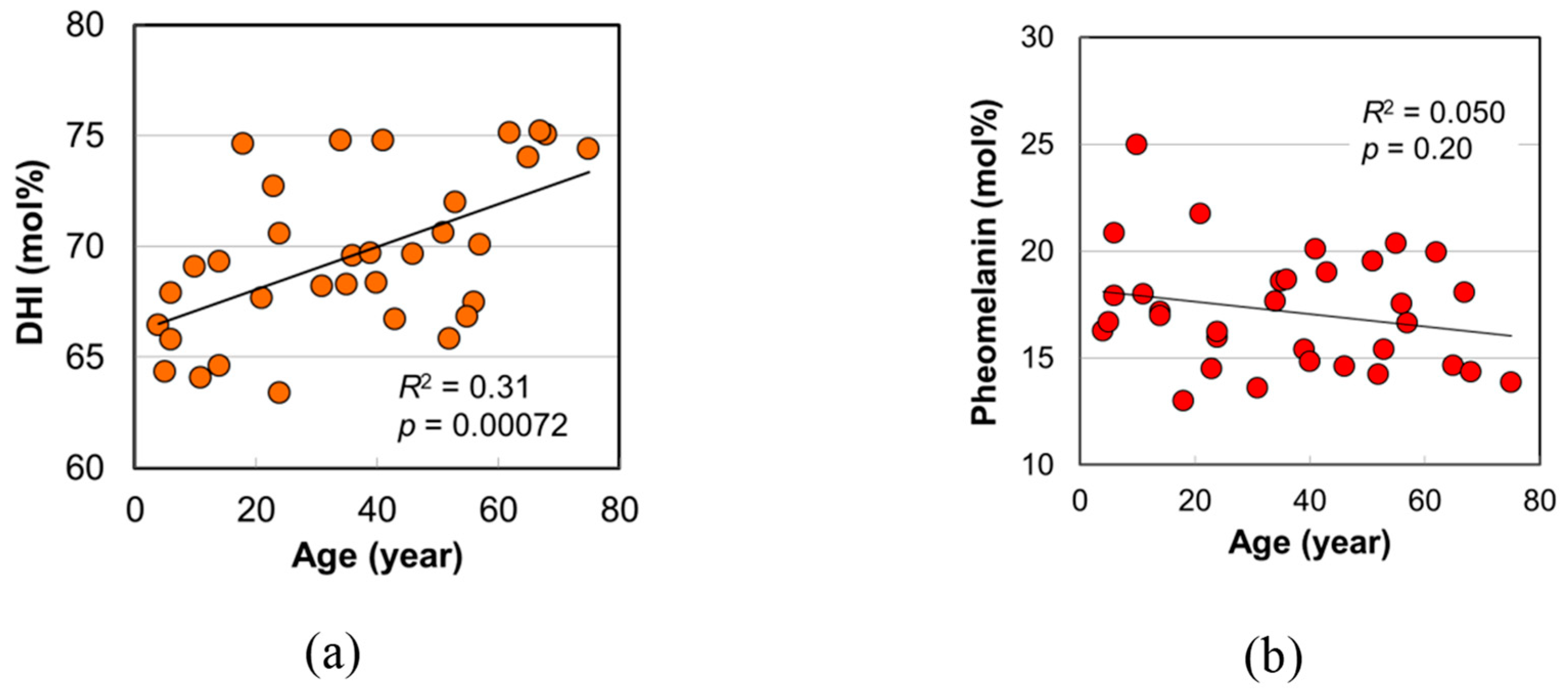
2.1. Age Dependence of Hair Color
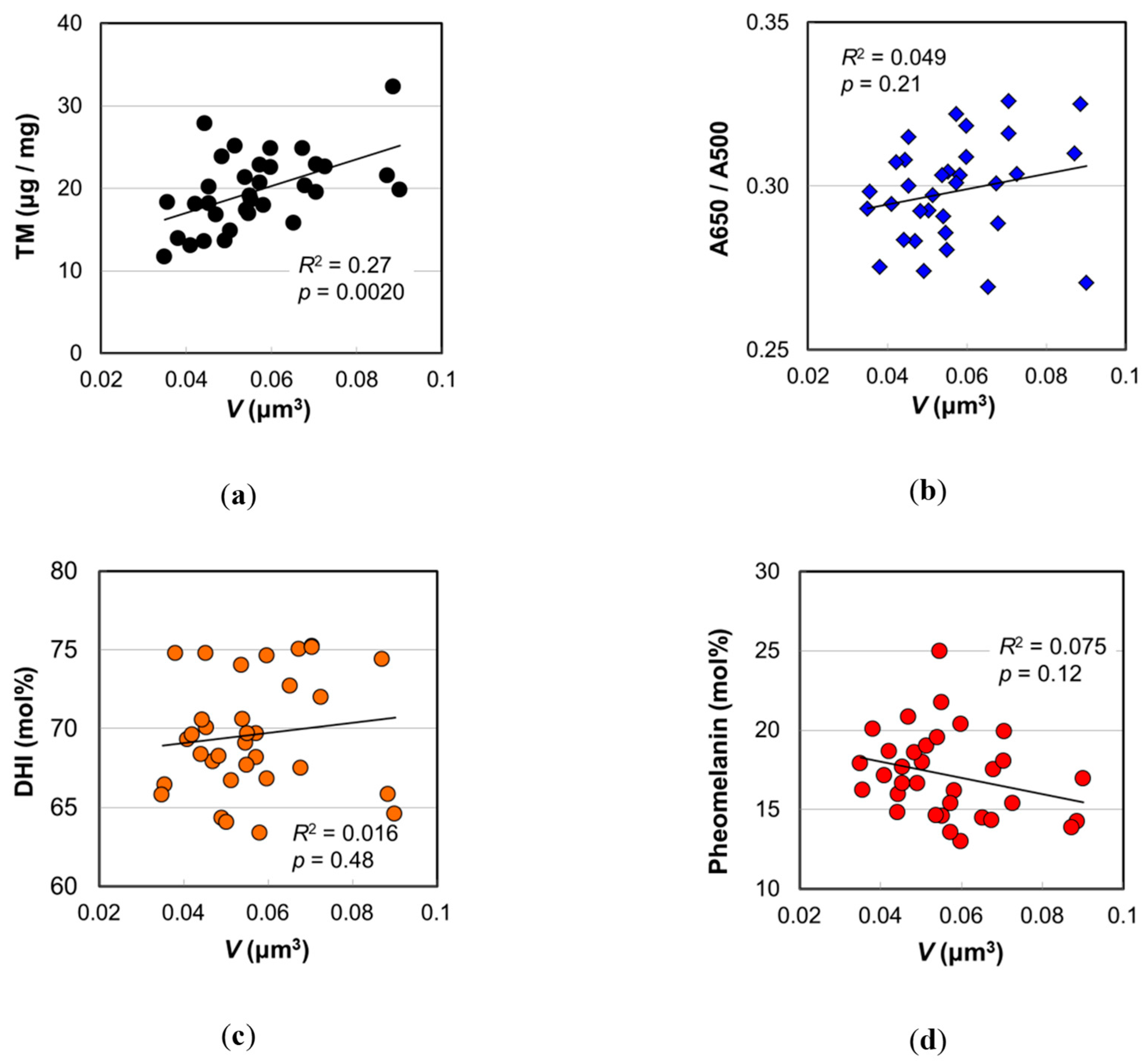
2.2. Melanosome Morphology
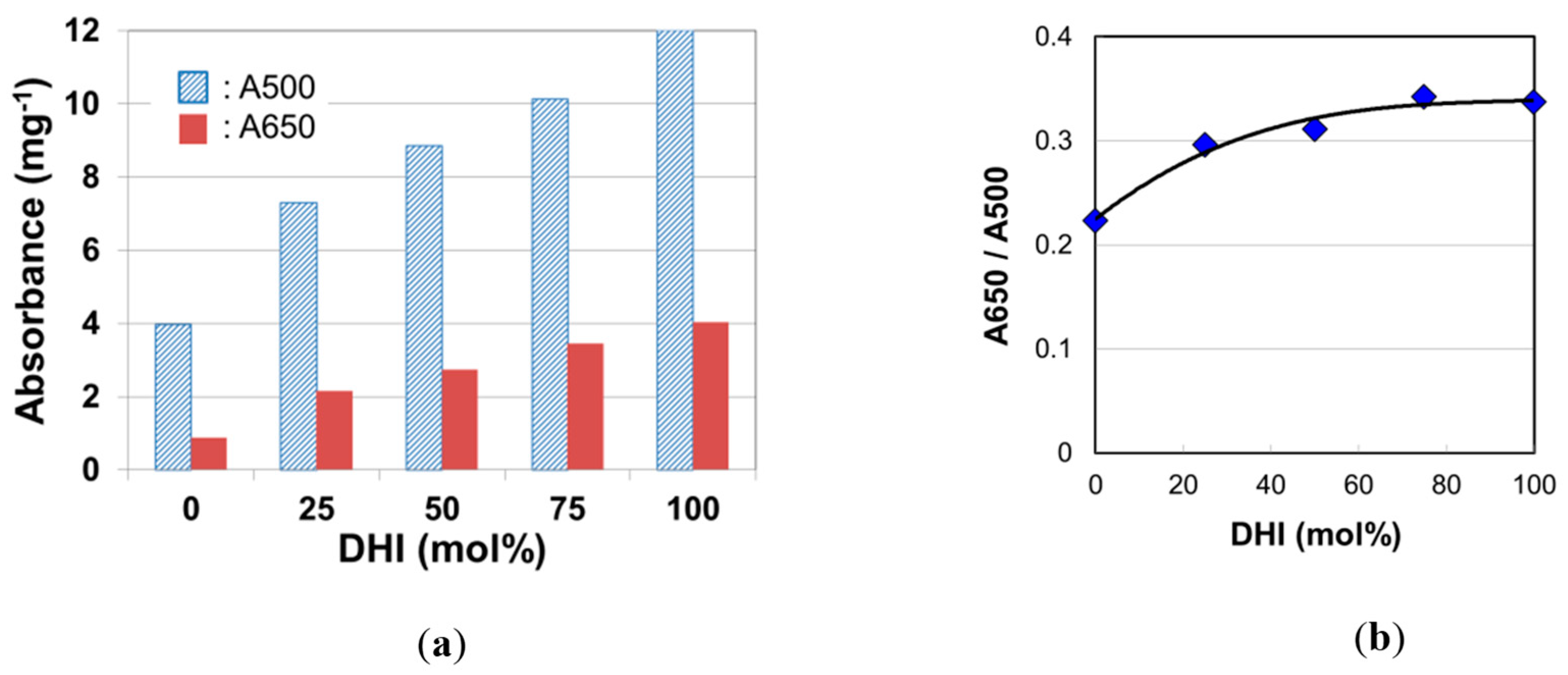
2.3. Absorption Spectra of Eumelanins Synthesized with DHI and DHICA
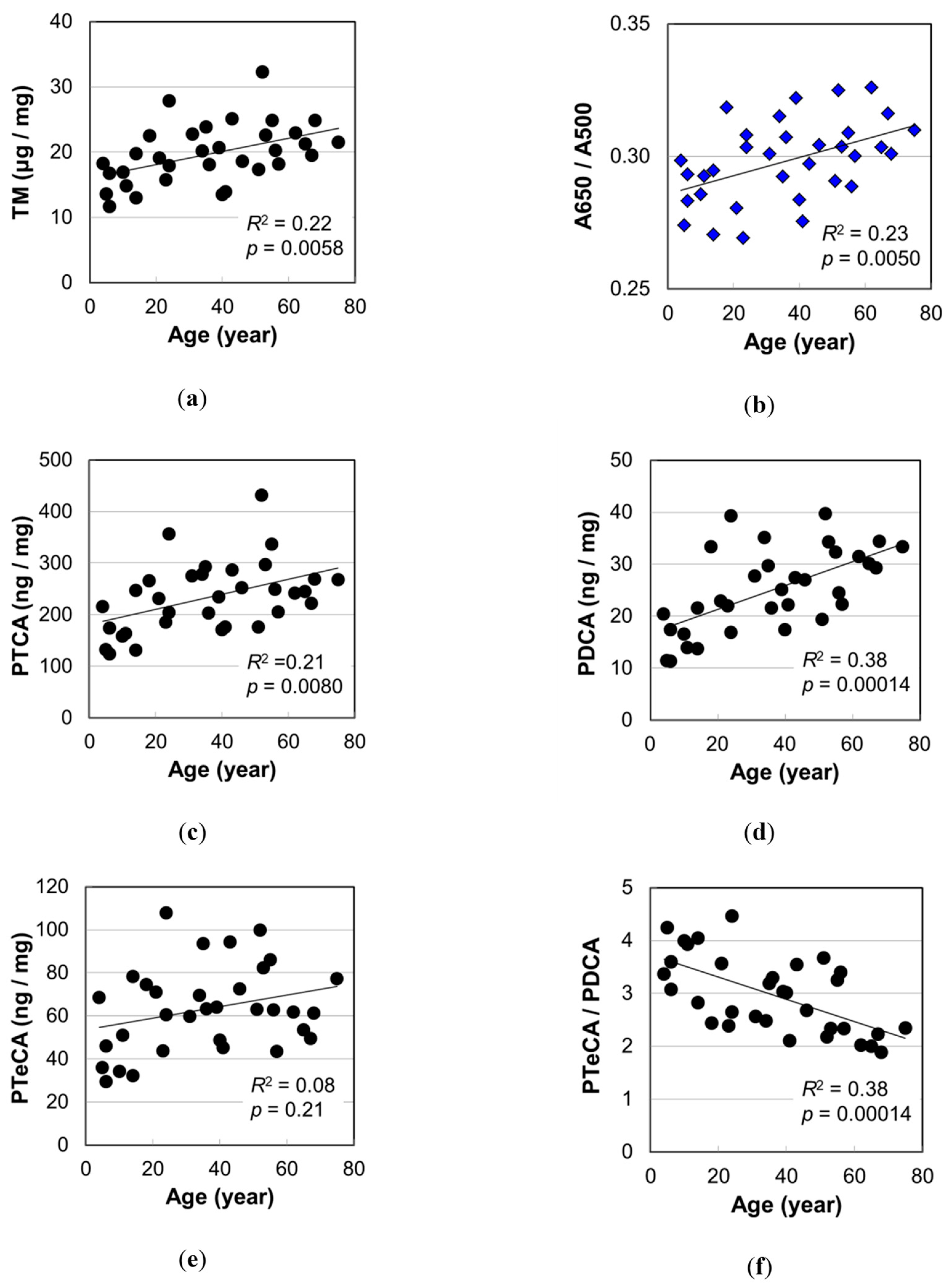
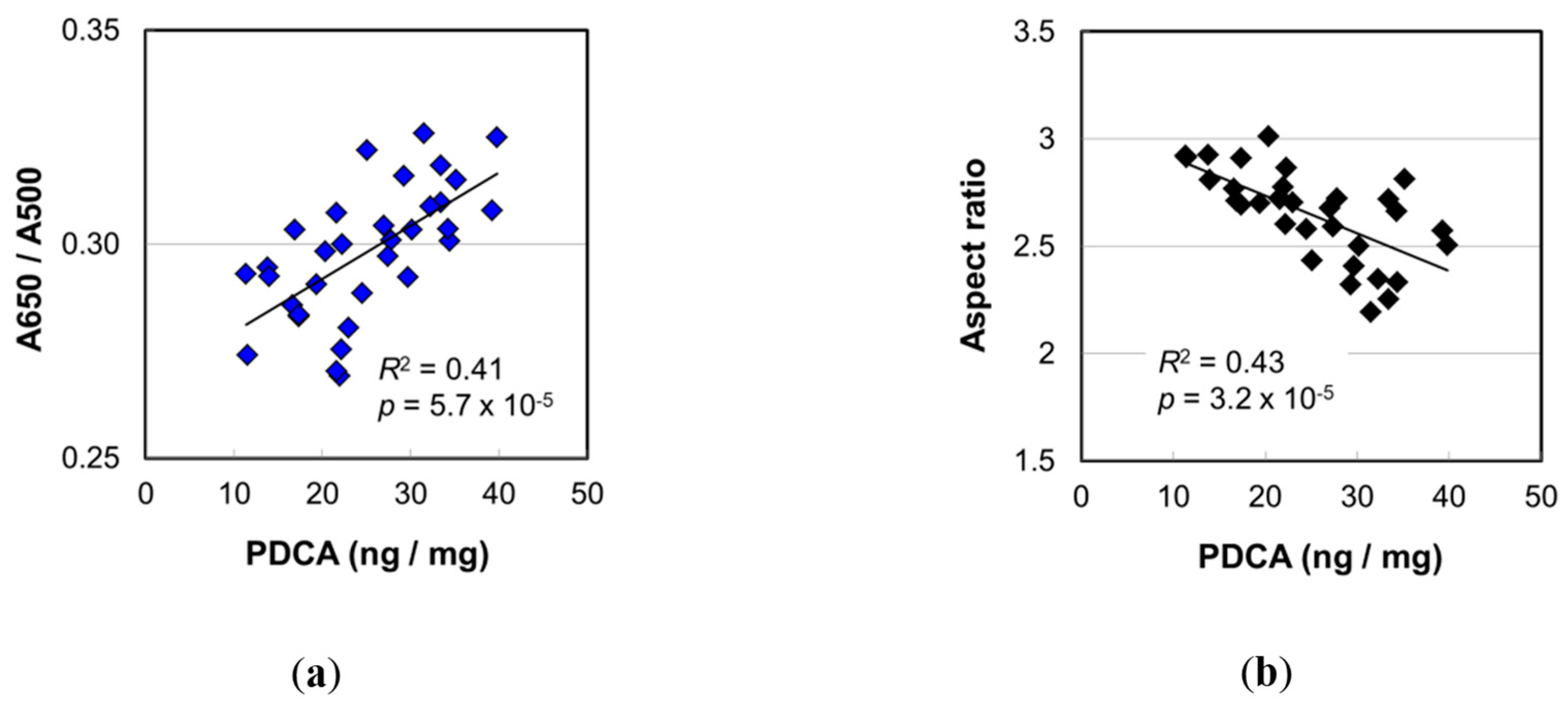
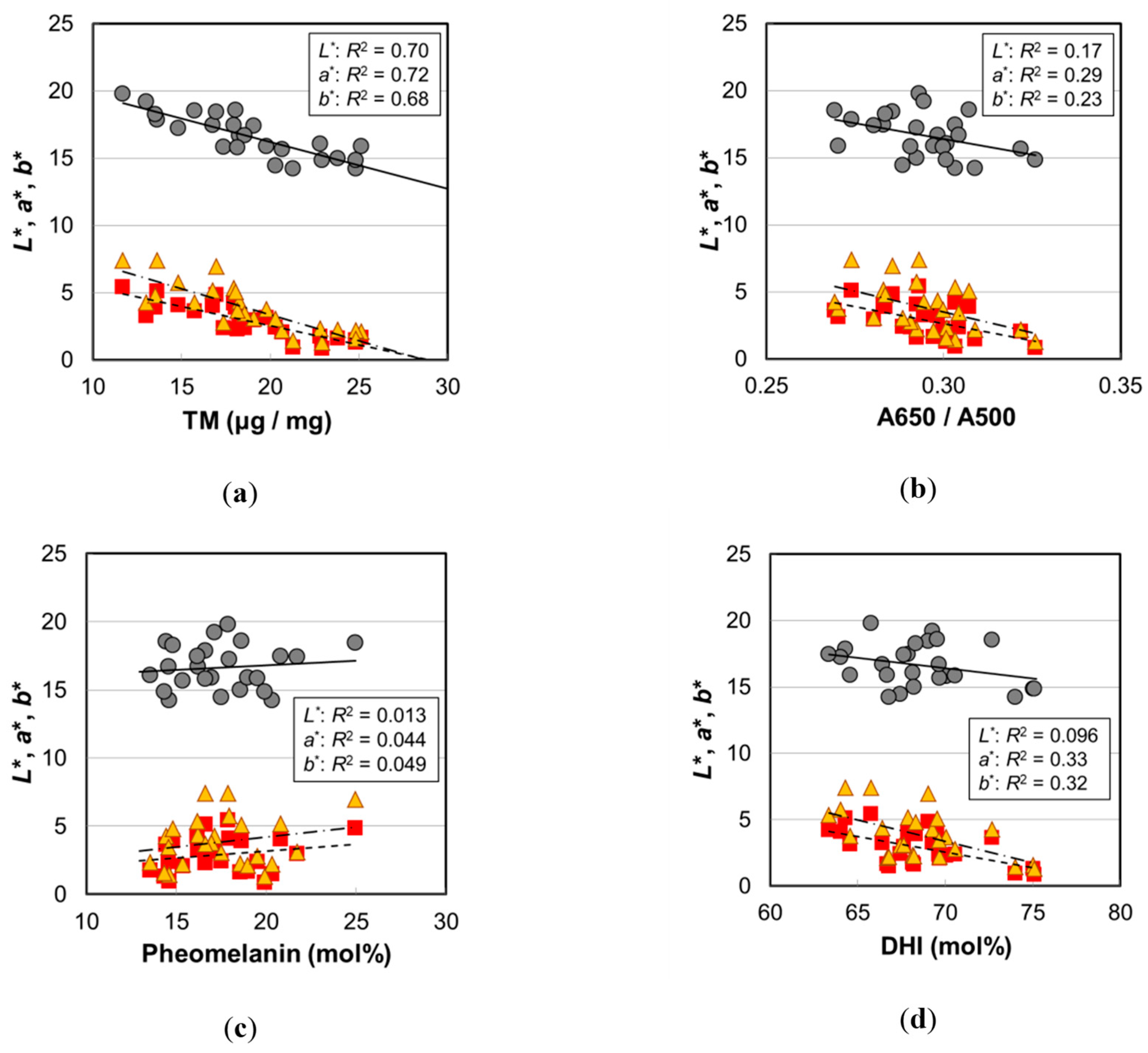
2.4. Melanin Compositions
3. Discussion
3.1. Impacts of the Increase in PDCA by Aging
3.2. Causes of Hair Color Darkening by Aging
4. Materials and Methods
4.1. Hair Samples
4.2. Chemicals
4.3. Hair Diameter
4.4. Colorimetric Measurement
4.5. Isolation of Melanosomes and Morphology Measurement
4.6. Melanin Analyses
4.7. Preparation of Synthetic Melanins and Analyses
4.8. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| A500 | Absorbance at the wave length of 500 nm |
| A650 | Absorbance at the wave length of 650 nm |
| 4-AHP | 4-Amino-3-hydroxyphenylalanine |
| DHI | 5,6-Dihydroxyindole |
| DHICA | 5,6-Dihydroxyindole-2-carboxylic acid |
| H2O2 | Hydrogen peroxide |
| HPLC | High performance liquid chromatography |
| PDCA | Pyrrole-2,3-dicarboxylic acid |
| PTCA | Pyrrole-2,3,5-tricarboxylic acid |
| PTeCA | Pyrrole-2,3,4,5-tetracarboxylic acid |
| TDCA | Thiazole-4,5-dicarboxylic acid |
| TM | Total melanin amount |
| TTCA | Thiazole-2,4,5-tricarboxylic acid |
| UV | Ultraviolet |
References
- Borovanský, J.; Riley, P.A. Physiological and pathological functions of melanosomes. In Melanins and Melanosomes; Borovanský, J., Riley, P.A., Eds.; Wiley-VCH Verlag & Co.: Weinheim, Germany, 2011; pp. 343–381. [Google Scholar]
- Commo, S.; Gaillard, O.; Bernard, B.A. Human hair greying is linked to a specific depletion of hair follicle melanocytes affecting both the bulb and the outer root sheath. Br. J. Derm. 2004, 150, 435–443. [Google Scholar] [CrossRef] [PubMed]
- d’Ischia, M.; Wakamatsu, K.; Cicoira, F.; di Mauro, E.; Garcia-Borron, J.C.; Commo, S.; Galván, I.; Ghanem, G.; Koike, K.; Meredith, P.; et al. Melanins and melanogenesis: From pigment cells to human health and technological applications. Pigment Cell Melanoma Res. 2015, 28, 520–544. [Google Scholar] [CrossRef] [PubMed]
- Hysi, P.G.; Valdes, A.M.; Liu, F.; Furlotte, N.A.; Evans, D.M.; Bataille, V.; Visconti, A.; Hemani, G.; McMahon, G.; Ring, S.M.; et al. Genome-wide association meta-analysis of individuals of European ancestry identifies new loci explaining a substantial fraction of hair color variation and heritability. Nat. Genet. 2018, 50, 652–656. [Google Scholar] [CrossRef] [PubMed]
- Morgan, M.D.; Pairo-Castineira, E.; Rawlik, K.; Canela-Xandri, O.; Rees, J.; Sims, D.; Tenesa, A.; Jackson, I.J. Genome-wide study of hair colour in UK biobank explains most of the SNP heritability. Nat. Commun. 2018, 9, 5271. [Google Scholar] [CrossRef] [PubMed]
- Sulem, P.; Gudbjartsson, D.F.; Stacey, S.N.; Helgason, A.; Rafnar, T.; Magnusson, K.P.; Manolescu, A.; Karason, A.; Palsson, A.; Thorleifsson, G.; et al. Genetic determinants of hair, eye and skin pigmentation in Europeans. Nat. Genet. 2007, 39, 1443–1452. [Google Scholar] [CrossRef] [PubMed]
- Kukla-Bartoszek, M.; Pośpiech, E.; Spólnicka, M.; Karłowska-Pik, J.; Strapagiel, D.; Żądzińska, E.; Rosset, I.; Sobalska-Kwapis, M.; Słomka, M.; Walsh, S.; et al. Investigating the impact of age-depended hair colour darkening during childhood on DNA-based hair colour prediction with the HIrisPlex system. Forensic Sci. Int. Genet. 2018, 36, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Ito, S.; Wakamatsu, K. Quantitative analysis of eumelanin and pheomelanin in humans, mice, and other animals: A comparative review. Pigment Cell Res. 2003, 16, 523–531. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Liu, X.-M.; Dai, X.; Zhou, Q.; Lei, T.; Beermann, F.; Wakamatsu, K.; Xu, S.-Z. Regulation of DHICA-mediated antioxidation by dopachrome tautomerase: Implication for skin photoprotection against UVA radiation. Free Radic. Biol. Med. 2010, 48, 1144–1151. [Google Scholar] [CrossRef] [PubMed]
- Panzella, L.; Gentile, G.; D’Errico, G.; Della Vecchia, N.F.; Errico, M.E.; Napolitano, A.; Carfagna, C.; d’Ischia, M. Atypical structural and π-electron features of a melanin polymer that lead to superior free-radical-scavenging properties. Angew. Chem. Int. Ed. 2013, 52, 12684–12687. [Google Scholar] [CrossRef]
- Commo, S.; Wakamatsu, K.; Lozano, I.; Panhard, S.; Loussouarn, G.; Bernard, B.A.; Ito, S. Age-dependent changes in eumelanin composition in hairs of various ethnic origins. Int. J. Cosmet. Sci. 2012, 34, 102–107. [Google Scholar] [CrossRef]
- Ito, S.; Miyake, S.; Maruyama, S.; Suzuki, I.; Commo, S.; Nakanishi, Y.; Wakamatsu, K. Acid hydrolysis reveals a low but constant level of pheomelanin in human black to brown hair. Pigment Cell Melanoma Res. 2018, 31, 393–403. [Google Scholar] [CrossRef] [PubMed]
- Itou, T. Morphological changes in hair melanosomes by aging. Pigment Cell Melanoma Res. 2018, 31, 630–635. [Google Scholar] [CrossRef] [PubMed]
- Nagase, S.; Kajiura, Y.; Mamada, A.; Abe, H.; Shibuichi, S.; Satoh, N.; Itou, T.; Shinohara, Y.; Amemiya, Y. Changes in structure and geometric properties of human hair by aging. J. Cosmet. Sci. 2009, 60, 637–648. [Google Scholar] [CrossRef] [PubMed]
- Ozeki, H.; Ito, S.; Wakamatsu, K.; Thody, A.J. Spectrophotometric characterization of eumelanin and pheomelanin in hair. Pigment Cell Res. 1996, 9, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Ito, S.; Nakanishi, Y.; Valenzuela, R.K.; Brilliant, M.H.; Kolbe, L.; Wakamatsu, K. Usefulness of alkaline hydrogen peroxide oxidation to analyze eumelanin and pheomelanin in various tissue samples: Application to chemical analysis of human hair melanins. Pigment Cell Melanoma Res. 2011, 24, 605–613. [Google Scholar] [CrossRef] [PubMed]
- d’Ischia, M.; Wakamatsu, K.; Napolitano, A.; Briganti, S.; Garcia-Borron, J.; Kovacs, D.; Meredith, P.; Pezzella, A.; Picardo, M.; Sarna, T.; et al. Melanins and melanogenesis: Methods, standards, protocols. Pigment Cell Melanoma Res. 2013, 26, 616–633. [Google Scholar] [CrossRef] [PubMed]
- Ito, S.; Wakamatsu, K.; Glass, K.; Simon, J.D. High-performance liquid chromatography estimation of cross-linking of dihydroxyindole moiety in eumelanin. Anal. Biochem. 2013, 434, 221–225. [Google Scholar] [CrossRef]
- Gordon, G.F. Sex and age related differences in trace element concentrations in hair. Sci. Total Environ. 1985, 42, 133–147. [Google Scholar] [CrossRef]
- Deeming, S.B.; Weber, C.W. Hair analysis of trace minerals in human subjects as influenced by age, sex, and contraceptive drugs. Am. J. Clin. Nutr. 1978, 31, 1175–1180. [Google Scholar] [CrossRef]
- Ito, S.; Suzuki, N.; Takebayashi, S.; Commo, S.; Wakamatsu, K. Neutral pH and copper ions promote eumelanogenesis after the dopachrome stage. Pigment Cell Melanoma Res. 2013, 26, 817–825. [Google Scholar] [CrossRef]
- Ito, S.; Wakamatsu, K.; Sarna, T. Photodegradation of eumelanin and pheomelanin and its pathophysiological implications. Photochem. Photobiol. 2018, 94, 409–420. [Google Scholar] [CrossRef] [PubMed]
- Ito, S.; Wakamatsu, K. Chemistry of mixed melanogenesis – pivotal roles of dopaquinone. Photochem. Photobiol. 2008, 84, 582–592. [Google Scholar] [CrossRef] [PubMed]
- Ito, S. A chemist’s view of melanogenesis. Pigment Cell Res. 2003, 16, 230–236. [Google Scholar] [CrossRef] [PubMed]
- Palumbo, A.; Solano, F.; Misuraca, G.; Aroca, P.; Garcia-Borron, J.C.; Lozano, J.A.; Prota, G. Comparative action of dopachrome tautomerase and metal ions on the rearrangement of dopachrome. Biochim. Biophys. Acta. 1991, 1115, 1–5. [Google Scholar] [CrossRef]
- Nagase, S.; Shibuichi, S.; Ando, K.; Kariya, E.; Satoh, N. Influence of internal structures of hair fiber on hair appearance. I. Light scattering from the porous structure of the medulla of human hair. J. Cosmet. Sci. 2002, 53, 89–100. [Google Scholar] [PubMed]
- Chardon, A.; Cretois, I.; Hourseau, C. Skin colour typology and suntanning pathways. Int. J. Cosmet. Sci. 1991, 13, 191–208. [Google Scholar] [CrossRef] [PubMed]
- Novellino, L.; Napolitano, A.; Prota, G. Isolation and characterization of mammalian eumelanins from hair and irides. Biochim. Biophys. Acta. 2000, 1475, 295–306. [Google Scholar] [CrossRef]
- Rasband, W.S. ImageJ; U.S. National Institutes of Health: Bethesda, MD, USA. Available online: https://imagej.nih.gov/ij/ (accessed on 15 March 2017).
- Wakamatsu, K.; Ito, S.; Rees, J.L. The usefulness of 4-amino-3-hydroxyphenylalanine as a specific marker of pheomelanin. Pigment Cell Res. 2002, 15, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Ito, S.; Wakamatsu, K. Chemical degradation of melanins: application to identification of dopamine-melanin. Pigment Cell Res. 1998, 11, 120–126. [Google Scholar] [CrossRef]
- Callaghan, T.M.; Wilhelm, K.-P. A review of ageing and an examination of clinical methods in the assessment of ageing skin. Part 2: Clinical perspectives and clinical methods in the evaluation of ageing skin. Int. J. Cosmet. Sci. 2008, 30, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Del Bino, S.; Ito, S.; Sok, J.; Nakanishi, Y.; Bastien, P.; Wakamatsu, K.; Bernerd, F. Chemical analysis of constitutive pigmentation of human epidermis reveals constant eumelanin to pheomelanin ratio. Pigment Cell Melanoma Res. 2015, 28, 707–717. [Google Scholar] [CrossRef] [PubMed]
| Parameter | Hair Color | |||
|---|---|---|---|---|
| L* | a* | b* | ||
| Human | Age (year) | 3.3 × 10−5 | 1.1 × 10−6 | 5.6 × 10−6 |
| Fiber | Cross sectional area (μm2) | 0.63 | 0.28 | 0.12 |
| Melanosome | Major axis (μm) | 0.72 | 0.26 | 0.37 |
| Minor axis (μm) | 0.0063 | 0.0043 | 0.0024 | |
| Aspect ratio | 4.1 × 10−5 | 1.2 × 10−5 | 1.5 × 10−5 | |
| Volume (μm3) | 0.011 | 0.066 | 0.038 | |
| Melanin | Total melanin amount (μg/mg) | 2.2 × 10−7 | 1.0 × 10−7 | 3.9 × 10−7 |
| A650/A500 1 | 0.043 | 0.0054 | 0.015 | |
| DHI 2 (mol%) | 0.13 | 0.0027 | 0.0032 | |
| Pheomelanin (mol%) | 0.59 | 0.31 | 0.29 | |
| PTCA 3 level (ng/mg) | 1.2 × 10−6 | 9.4 × 10−7 | 1.7 × 10−6 | |
| PDCA 4 level (ng/mg) | 8.6 × 10−7 | 1.8 × 10−10 | 1.5 × 10−9 | |
| PTeCA 5 level (ng/mg) | 0.00087 | 0.0094 | 0.0057 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Itou, T.; Ito, S.; Wakamatsu, K. Effects of Aging on Hair Color, Melanosome Morphology, and Melanin Composition in Japanese Females. Int. J. Mol. Sci. 2019, 20, 3739. https://doi.org/10.3390/ijms20153739
Itou T, Ito S, Wakamatsu K. Effects of Aging on Hair Color, Melanosome Morphology, and Melanin Composition in Japanese Females. International Journal of Molecular Sciences. 2019; 20(15):3739. https://doi.org/10.3390/ijms20153739
Chicago/Turabian StyleItou, Takashi, Shosuke Ito, and Kazumasa Wakamatsu. 2019. "Effects of Aging on Hair Color, Melanosome Morphology, and Melanin Composition in Japanese Females" International Journal of Molecular Sciences 20, no. 15: 3739. https://doi.org/10.3390/ijms20153739
APA StyleItou, T., Ito, S., & Wakamatsu, K. (2019). Effects of Aging on Hair Color, Melanosome Morphology, and Melanin Composition in Japanese Females. International Journal of Molecular Sciences, 20(15), 3739. https://doi.org/10.3390/ijms20153739