Proteolytic Rafts for Improving Intraparenchymal Migration of Minimally Invasively Administered Hydrogel-Embedded Stem Cells
Abstract
1. Introduction
2. Results
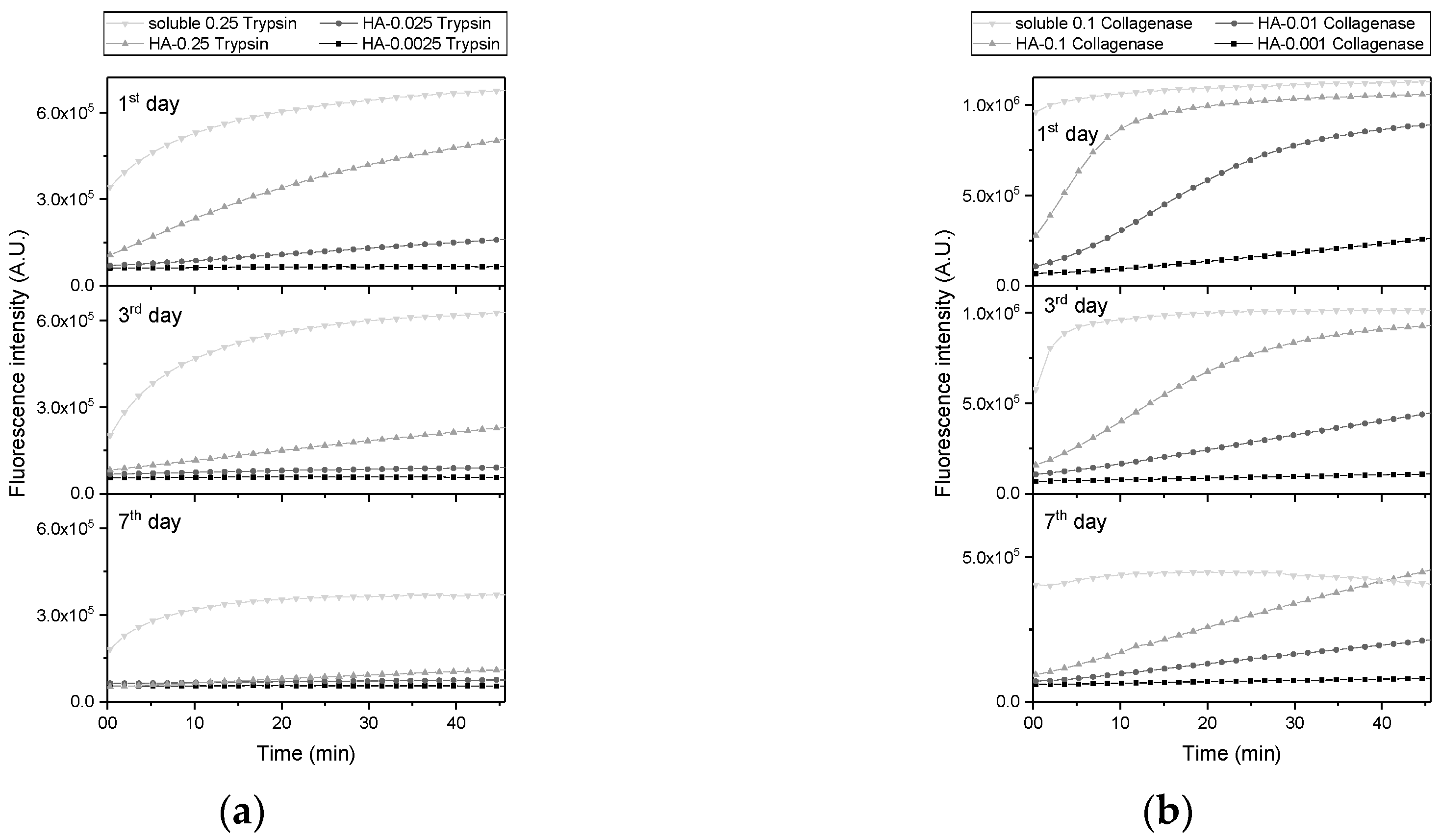
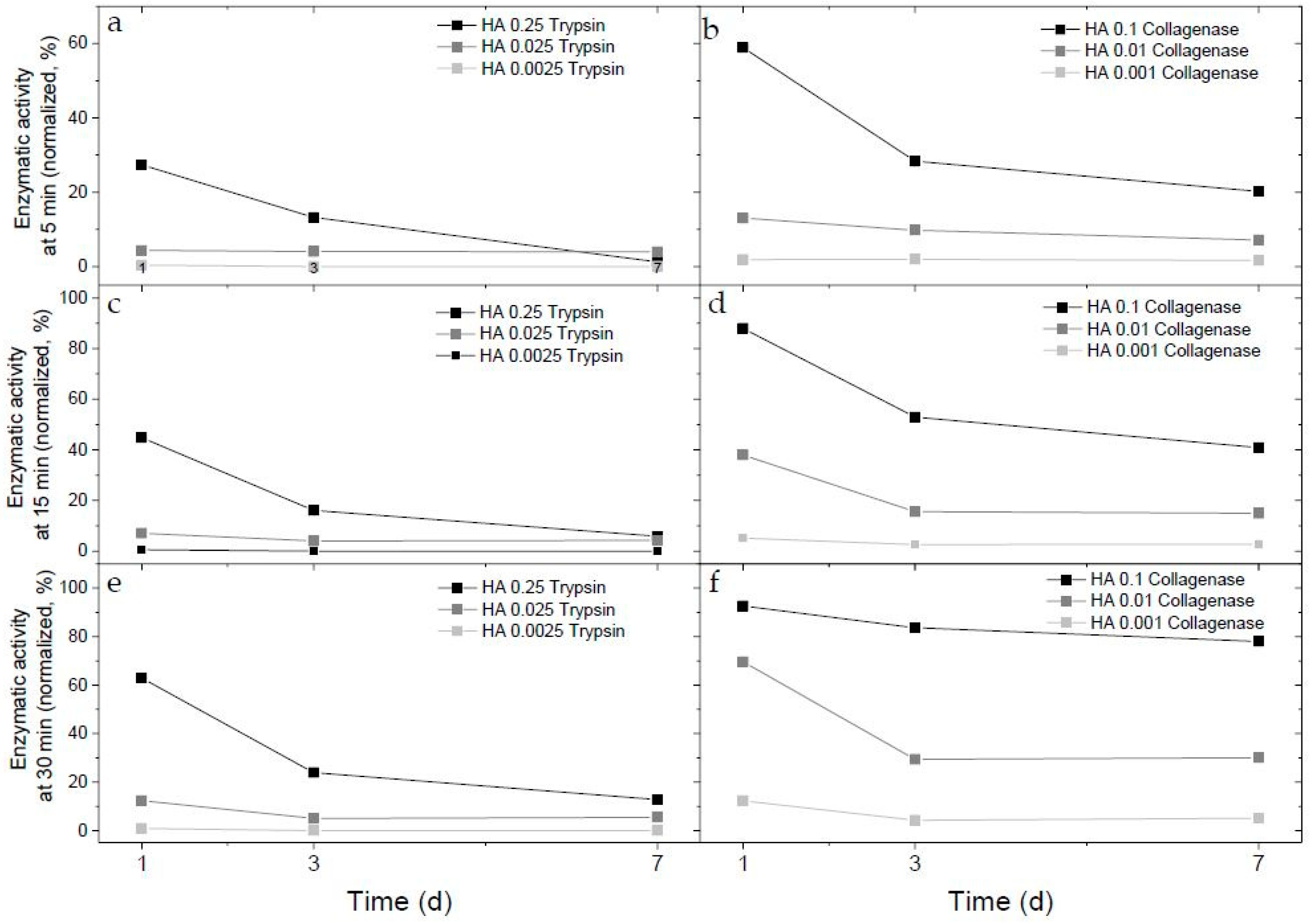
2.1. Enzymatic Activity of Immobilized Trypsin and Collagenase in Hyaluronan-Based Hydrogels
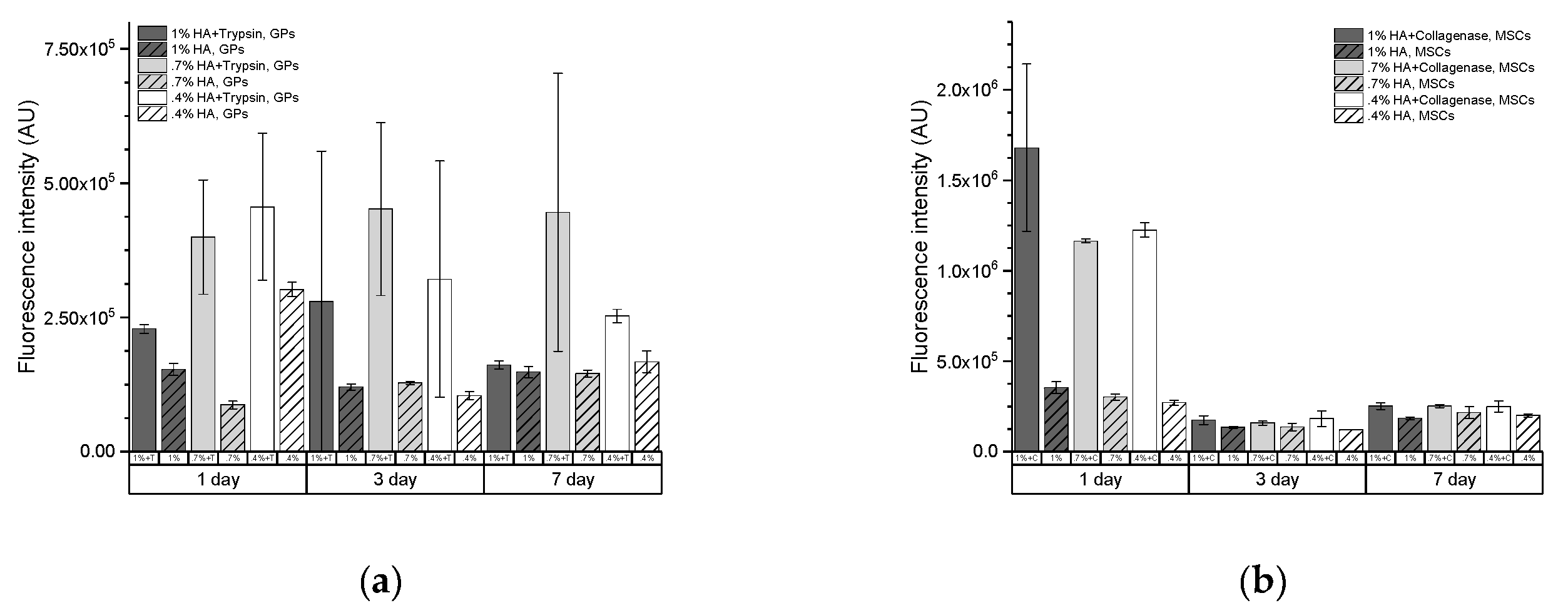
2.2. Enzymatic Activity Measurements for Hydrogel-Embedded Stem Cells
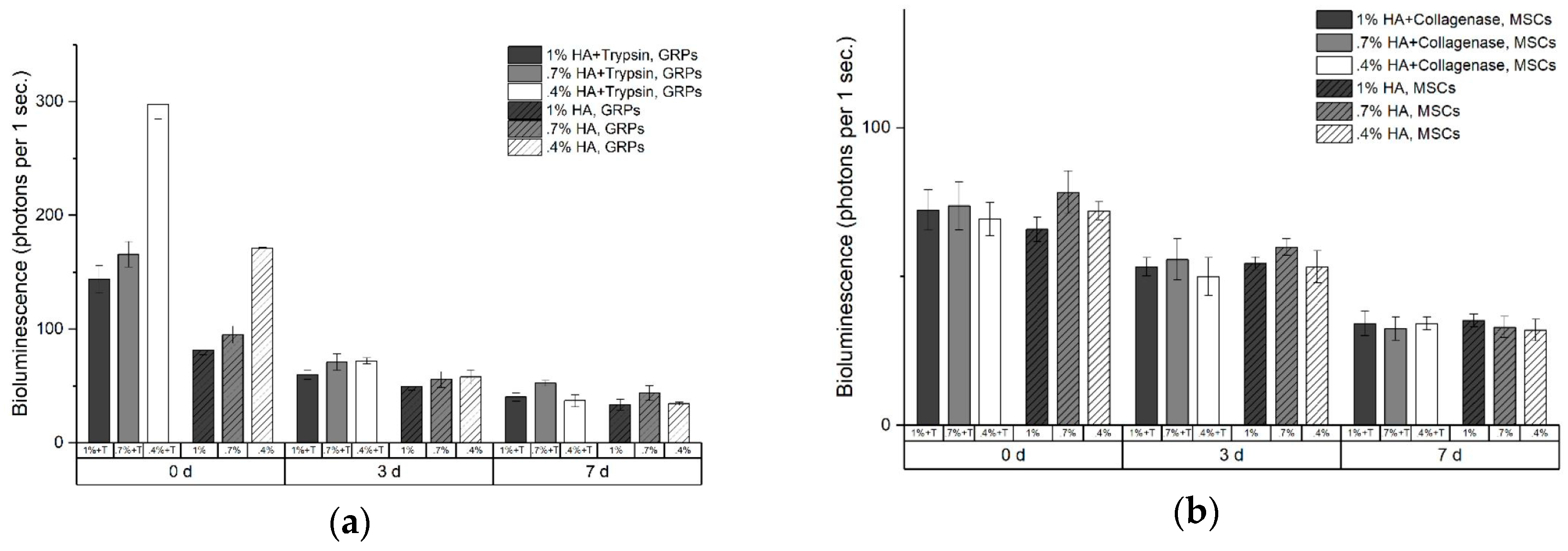
2.3. In Vitro Cell Viability in Enzymatic Hydrogels
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Cell Harvest and Culture
4.3. Trypsin and Collagenase Derivation
4.4. Hydrogel Preparation
4.5. Enzymatic Activity Assay
4.6. In Vitro Viability Assessments
4.7. Statistics
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| HA | activated hyaluronan acid |
| PEGDA | poly(ethylene glycol) diacrylate |
| GRPs | glial-restricted progenitors |
| MSC | mesenchymal stem cells |
| Sulfo-SMCC | sulfosuccinimidyl 4-[N-maleimidomethyl]cyclohexane-1-carboxylate |
Appendix A
| Type 3 Tests of Fixed Effects | ||||
|---|---|---|---|---|
| Num DF | DenDF | F Value | Pr > F | |
| Time | 2 | 34 | 203.48 | <0.0001 |
| Enzyme | 1 | 34 | 157.37 | <0.0001 |
| Hydrogel | 2 | 34 | 3.96 | 0.0284 |
| Time*Enzyme | 2 | 34 | 121.78 | <.0001 |
| Time*Hydrogel | 4 | 34 | 4.26 | 0.0067 |
| Enzyme*Hydrogel | 2 | 34 | 2.81 | 0.0745 |
| Time*Enzyme*Hydrogel | 4 | 34 | 2.28 | 0.0804 |
| Type 3 Tests of Fixed Effects | ||||
|---|---|---|---|---|
| Num DF | DenDF | F Value | Pr > F | |
| Time | 2 | 34 | 0.98 | 0.3868 |
| Enzyme | 1 | 34 | 35.12 | <0.0001 |
| Hydrogel | 2 | 34 | 3.82 | 0.0320 |
| Time*Enzyme | 2 | 34 | 0.90 | 0.4173 |
| Time*Hydrogel | 4 | 34 | 2.04 | 0.1113 |
| Enzyme*Hydrogel | 2 | 34 | 4.87 | 0.0138 |
| Time*Enzyme*Hydrogel | 4 | 34 | 0.13 | 0.9687 |
| Type 3 Tests of Fixed Effects | ||||
|---|---|---|---|---|
| Num DF | DenDF | F Value | Pr > F | |
| Time | 2 | 34 | 306.49 | <0.0001 |
| Enzyme | 1 | 34 | 0.58 | 0.4534 |
| Hydrogel | 2 | 34 | 3.20 | 0.0533 |
| Time*Enzyme | 2 | 34 | 0.55 | 0.5843 |
| Time*Hydrogel | 4 | 34 | 1.74 | 0.1634 |
| Enzyme*Hydrogel | 2 | 34 | 1.07 | 0.3545 |
| Time*Enzyme*Hydrogel | 4 | 34 | 0.85 | 0.5011 |
| Type 3 Tests of Fixed Effects | ||||
|---|---|---|---|---|
| Num DF | DenDF | F Value | Pr > F | |
| Time | 2 | 34 | 2005.19 | <0.0001 |
| Enzyme | 1 | 34 | 466.80 | <0.0001 |
| Hydrogel | 2 | 34 | 251.43 | <0.0001 |
| Time*Enzyme | 2 | 34 | 246.87 | <0.0001 |
| Time*Hydrogel | 4 | 34 | 239.29 | <0.0001 |
| Enzyme*Hydrogel | 2 | 34 | 15.31 | <0.0001 |
| Time*Enzyme*Hydrogel | 4 | 34 | 18.16 | <0.0001 |
| HA | Fluorescence (A.U.) | |||
|---|---|---|---|---|
| 1st day | 3rd day | 7th day | ||
| HA-1 mg/mL collagenase | 1.0% + C | 1.68 × 106 | 1.74 × 105 | 2.52 × 105 |
| 1.0% | 3.55 × 105 | 1.35 × 105 | 1.86 × 105 | |
| 0.7% + C | 1.16 × 106 | 1.58 × 105 | 2.52 × 105 | |
| 0.7% | 3.02 × 105 | 1.37 × 105 | 2.16 × 105 | |
| 0.4% + C | 1.22 × 106 | 1.82 × 105 | 2.49 × 105 | |
| 0.4% | 2.71 × 105 | 1.21 × 105 | 2.01 × 105 | |
| HA-0.25 mg/mL trypsin | 1.0% + T | 2.29 × 105 | 2.80 × 105 | 1.61 × 105 |
| 1.0% | 1.53 × 105 | 1.20 × 105 | 1.48 × 105 | |
| 0.7% +T | 3.99 × 105 | 4.52 × 105 | 4.46 × 105 | |
| 0.7% | 8.73 × 104 | 1.28 × 105 | 1.46 × 105 | |
| 0.4% + T | 4.56 × 105 | 3.21 × 105 | 2.53 × 105 | |
| 0.4% | 3.02 × 105 | 1.04 × 105 | 1.67 × 105 | |
References
- Qin, Z.H.; Xu, J.F.; Qu, J.M.; Zhang, J.; Sai, Y.; Chen, C.M.; Wu, L.; Yu, L. Intrapleural delivery of MSCs attenuates acute lung injury by paracrine/endocrine mechanism. J. Cell Mol. Med. 2012, 16, 2745–2753. [Google Scholar] [CrossRef]
- Zhang, J.; Wu, Z.; Fan, Z.; Qin, Z.; Wang, Y.; Chen, J.; Wu, M.; Chen, Y.; Wu, C.; Wang, J.F. Pericardial application as a new route for implanting stem-cell cardiospheres to treat myocardial infarction. J. Physiol. 2018, 596, 2037–2054. [Google Scholar] [CrossRef]
- Nicholls, F.J.; Ling, W.; Ferrauto, G.; Aime, S.; Modo, M. Simultaneous MR imaging for tissue engineering in a rat model of stroke. Sci. Rep. 2015, 5, 1–15. [Google Scholar] [CrossRef]
- Jablonska, A.; Shea, D.J.; Cao, S.; Bulte, J.W.; Janowski, M.; Konstantopoulos, K.; Walczak, P. Overexpression of VLA-4 in glial-restricted precursors enhances their endothelial docking and induces diapedesis in a mouse stroke model. J. Cereb. Blood Flow Metab. 2018, 38, 835–846. [Google Scholar] [CrossRef] [PubMed]
- Guzman, R.; Janowski, M.; Walczak, P. Intra-Arterial Delivery of Cell Therapies for Stroke. Stroke 2018, 49, 1075–1082. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, E.M.; Lamanna, J.; Boulis, N.M. Stem cell therapy for the spinal cord. Stem Cell Res. Ther. 2012, 3, 24. [Google Scholar] [CrossRef]
- Malysz-Cymborska, I.; Golubczyk, D.; Kalkowski, L.; Burczyk, A.; Janowski, M.; Holak, P.; Olbrych, K.; Sanford, J.; Stachowiak, K.; Milewska, K.; et al. MRI-guided intrathecal transplantation of hydrogel-embedded glial progenitors in large animals. Sci. Rep. 2018, 8, 1–9. [Google Scholar] [CrossRef]
- Janowski, M.; Engels, C.; Gorelik, M.; Lyczek, A.; Bernard, S.; Bulte, J.W.M.; Walczak, P. Survival of neural progenitors allografted into the CNS of immunocompetent recipients is highly dependent on transplantation site. Cell Transplant. 2014, 23, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, J.M.; Carvalho, L.; Silva-Correia, J.; Vieira, S.; Majchrzak, M.; Lukomska, B.; Stanaszek, L.; Strymecka, P.; . Malysz-Cymborska, I.; Golubczyk, D.; et al. Hydrogel-based scaffolds to support intrathecal stem cell transplantation as a gateway to the spinal cord: clinical needs, biomaterials, and imaging technologies. NPJ Regen. Med. 2018, 3, 1–9. [Google Scholar] [CrossRef]
- Lyczek, A.; Arnold, A.; Zhang, J.; Campanelli, J.T.; Janowski, M.; Bulte, J.W.; Walczak, P. Transplanted human glial-restricted progenitors can rescue the survival of dysmyelinated mice independent of the production of mature, compact myelin. Exp. Neurol. 2017, 291, 74–86. [Google Scholar] [CrossRef]
- Srivastava, R.K.; Bulte, J.W.M.; Walczak, P.; Janowski, M. Migratory potential of transplanted glial progenitors as critical factor for successful translation of glia replacement therapy: The gap between mice and men. Glia 2018, 66, 907–919. [Google Scholar] [CrossRef] [PubMed]
- Blázquez, R.; Sánchez-Margallo, F.M.; Crisóstomo, V.; Báez, C.; Maestre, J.; García-Lindo, M.; Usón, A.; Álvarez, V.; Casado, J.G. Intrapericardial administration of mesenchymal stem cells in a large animal model: A bio-distribution analysis. PLoS ONE 2015, 10, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Jia, F.; Narasimhan, B.; Mallapragada, S. Materials-based strategies for multi-enzyme immobilization and co-localization: A review. Biotechnol. Bioeng. 2014, 111, 209–222. [Google Scholar] [CrossRef] [PubMed]
- Schoffelen, S.; van Hest, J.C.M. Multi-enzyme systems: bringing enzymes together in vitro. Soft Matter 2012, 8, 1736–1746. [Google Scholar] [CrossRef]
- Datta, S.; Christena, L.R.; Rajaram, Y.R.S. Enzyme immobilization: an overview on techniques and support materials. 3 Biotech. 2013, 3, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Brady, D.; Jordaan, J. Advances in enzyme immobilisation. Biotechnol. Lett. 2009, 31, 1639–1650. [Google Scholar] [CrossRef] [PubMed]
- Deller, R.C.; Richardson, T.; Richardson, R.; Bevan, L.; Zampetakis, I.; Scarpa, F.; Perriman, A.W. Artificial cell membrane binding thrombin constructs drive in situ fibrin hydrogel formation. Nat. Commun. 2019, 10, 1887. [Google Scholar] [CrossRef]
- Thomas, F.T.; Contreras, J.L.; Bilbao, G.; Ricordi, C.; Curiel, D.; Thomas, J.M. Anoikis, extracellular matrix, and apoptosis factors in isolated cell transplantation. Surgery 1999, 126, 299–304. [Google Scholar] [CrossRef]
- Ballios, B.G.; Cooke, M.J.; Donaldson, L.; Coles, B.L.; Morshead, C.M.; van der Kooy, D.; Shoichet, M.S. A Hyaluronan-Based Injectable Hydrogel Improves the Survival and Integration of Stem Cell Progeny following Transplantation. Stem Cell Rep. 2015, 4, 1031–1045. [Google Scholar] [CrossRef]
- Führmann, T.; Tam, R.Y.; Ballarin, B.; Coles, B.; Donaghue, I.E.; Van Der Kooy, D.; Nagy, A.; Tator, C.H.; Morshead, C.M.; Shoichet, M.S. Injectable hydrogel promotes early survival of induced pluripotent stem cell-derived oligodendrocytes and attenuates longterm teratoma formation in a spinal cord injury model. Biomaterials 2016, 83, 23–36. [Google Scholar] [CrossRef]
- Mohand-Kaci, F.; Assoul, N.; Martelly, I.; Allaire, E.; Zidi, M. Optimized Hyaluronic Acid–Hydrogel Design and Culture Conditions for Preservation of Mesenchymal Stem Cell Properties. Tissue Eng. Part. C Methods 2013, 19, 288–298. [Google Scholar] [CrossRef] [PubMed]
- Mannello, F.; Tonti, G.A.M.; Bagnara, G.P.; Papa, S. Role and Function of Matrix Metalloproteinases in the Differentiation and Biological Characterization of Mesenchymal Stem Cells. Stem Cells 2006, 24, 475–481. [Google Scholar] [CrossRef] [PubMed]
- Uhlen, M.; Fagerberg, L.; Hallström, B.M.; Lindskog, C.; Oksvold, P.; Mardinoglu, A.; Sivertsson, A.; Kampf, C.; Sjöstedt, E.; Asplund, A.; et al. Tissue-based map of the human proteome. Science 2015, 347, 1260419. [Google Scholar] [CrossRef] [PubMed]
- Broda, E.; Suschny, O.; Rücker, W. Effects of trypsin treatment on tissue in culture. Exp. Cell Res. 1959, 18, 169–171. [Google Scholar] [CrossRef]
- Tsuji, K.; Ojima, M.; Otabe, K.; Horie, M.; Koga, H.; Sekiya, I.; Muneta, T. Effects of Different Cell-Detaching Methods on the Viability and Cell Surface Antigen Expression of Synovial Mesenchymal Stem Cells. Cell Transplant. 2017, 26, 1089–1102. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Li, C.; Zhang, C.Y.; Zhao, J. Effect of accutase or trypsin dissociation on the apoptosis of human striatum-derived neural stem cells. Acta Acad. Med. Sci. 2015, 37, 185–194. [Google Scholar]
- Jasensky, J.; Ferguson, K.; Baria, M.; Zou, X.; McGinnis, R.; Kaneshiro, A.; Badieyan, S.; Wei, S.; Marsh, E.N.G.; Chen, Z. Simultaneous Observation of the Orientation and Activity of Surface-Immobilized Enzymes. Langmuir 2018, 34, 9133–9140. [Google Scholar] [CrossRef]
- Rodrigues, R.C.; Ortiz, C.; Berenguer-Murcia, Á.; Torres, R.; Fernández-Lafuente, R. Modifying enzyme activity and selectivity by immobilization. Chem. Soc. Rev. 2013, 42, 6290–6307. [Google Scholar] [CrossRef]
- Phinney, D.G.; Kopen, G.; Isaacson, R.L.; Prockop, D.J. Plastic adherent stromal cells from the bone marrow of commonly used strains of inbred mice: Variations in yield, growth, and differentiation. J. Cell. Biochem. 1999, 72, 570–585. [Google Scholar] [CrossRef]
- Piejko, M.; Walczak, P.; Li, X.; Bulte, J.W.M.; Janowski, M. In Vitro Assessment of Fluorine Nanoemulsion-Labeled Hyaluronan-Based Hydrogels for Precise Intrathecal Transplantation of Glial-Restricted Precursors. Mol. Imaging Biol. 2019, 1–8. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piejko, M.; Jablonska, A.; Walczak, P.; Janowski, M. Proteolytic Rafts for Improving Intraparenchymal Migration of Minimally Invasively Administered Hydrogel-Embedded Stem Cells. Int. J. Mol. Sci. 2019, 20, 3083. https://doi.org/10.3390/ijms20123083
Piejko M, Jablonska A, Walczak P, Janowski M. Proteolytic Rafts for Improving Intraparenchymal Migration of Minimally Invasively Administered Hydrogel-Embedded Stem Cells. International Journal of Molecular Sciences. 2019; 20(12):3083. https://doi.org/10.3390/ijms20123083
Chicago/Turabian StylePiejko, Marcin, Anna Jablonska, Piotr Walczak, and Miroslaw Janowski. 2019. "Proteolytic Rafts for Improving Intraparenchymal Migration of Minimally Invasively Administered Hydrogel-Embedded Stem Cells" International Journal of Molecular Sciences 20, no. 12: 3083. https://doi.org/10.3390/ijms20123083
APA StylePiejko, M., Jablonska, A., Walczak, P., & Janowski, M. (2019). Proteolytic Rafts for Improving Intraparenchymal Migration of Minimally Invasively Administered Hydrogel-Embedded Stem Cells. International Journal of Molecular Sciences, 20(12), 3083. https://doi.org/10.3390/ijms20123083





