Identification of Potential Novel Interacting Partners for Coagulation Factor XIII B (FXIII-B) Subunit, a Protein Associated with a Rare Bleeding Disorder
Abstract
1. Introduction
2. Results
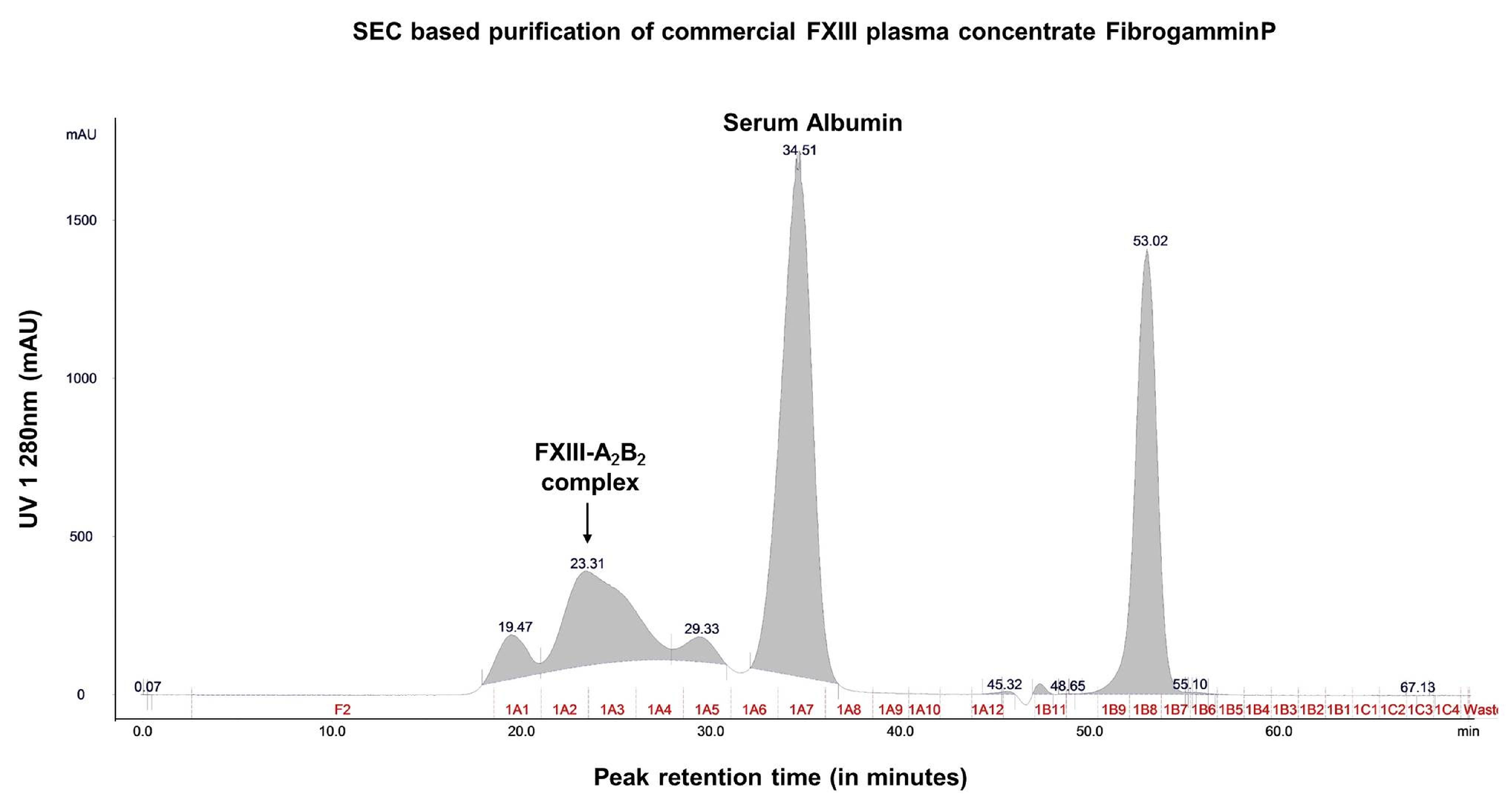
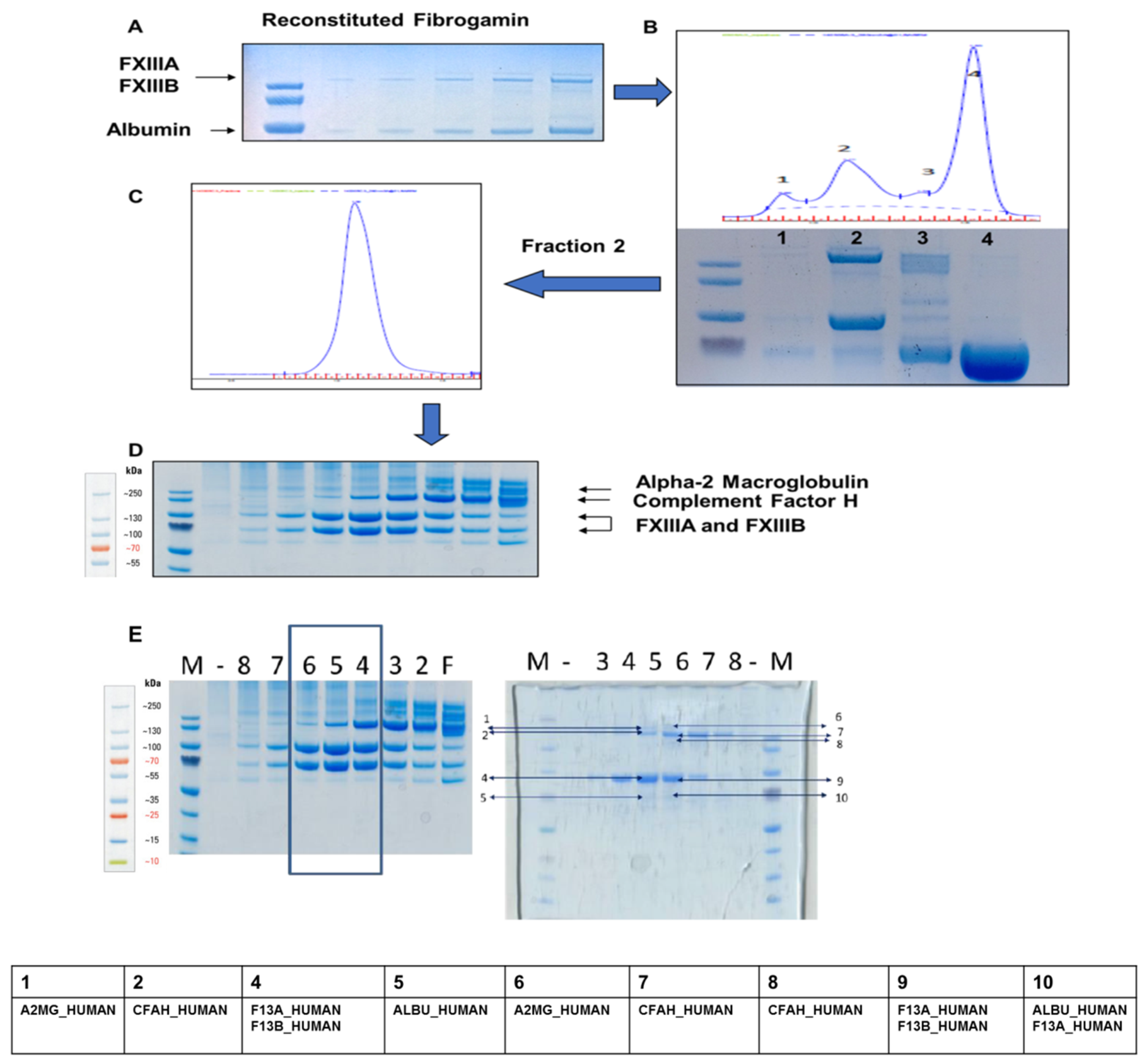
2.1. Content Characterization of Plasma FXIII Concentrate Reveals Co-Presence of Complement Factor H and Alpha-2-Macroglobulin Along with FXIII Complex
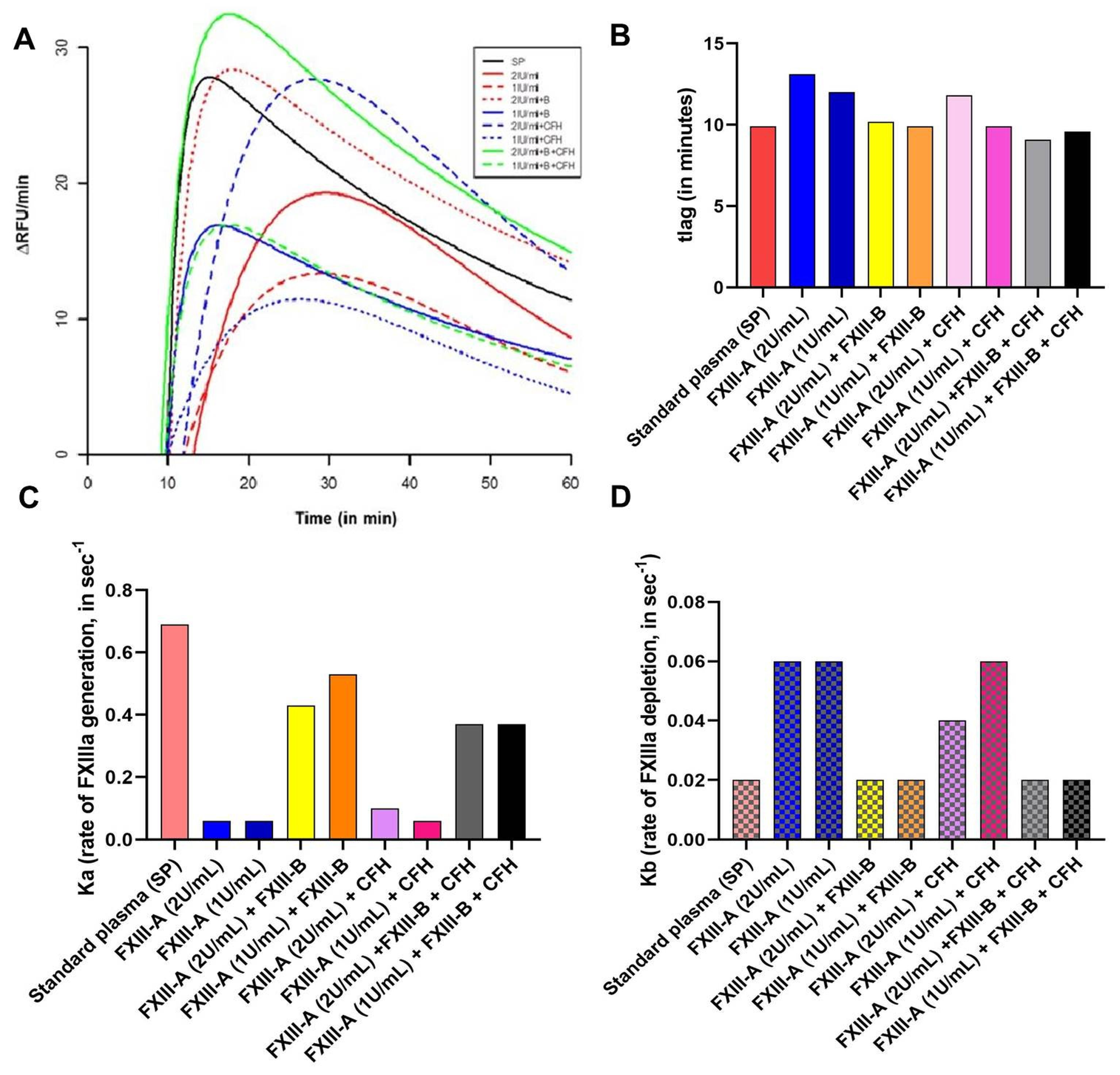
2.2. No Significant Effect of CFH on FXIII-Aa Activation Observed in the FXIIIa Generation Assay
2.3. In a FXIII Deficient Background, FXIII-B Pulls Down Fibrinogen, Few Complement Proteins, and Alpha-2-Macroglobulin
3. Discussion
4. Materials and Methods
4.1. Coagulation Factor XIII Complex and FXIII Subunits
4.2. Separation of the FXIII Plasma Concentrate, FibrogamminP into its Constituents
4.3. Mass Spectrometric Analysis
4.4. FXIIIa Generation Assay
4.5. Isolation and Verification of Plasma Sub-Proteome Interacting with FXIII-B by Immunoaffinity Based Pull-Down Assays
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| FXIII-DP | Factor XIII deficient plasma |
| CFH | Complement Factor H |
| FXIIIa | Activated FXIII-A |
| SP | Standard Plasma |
| MS | Mass spectrometry |
References
- Biswas, A.; Ivaskevicius, V.; Thomas, A.; Oldenburg, J. Coagulation factor XIII deficiency. Diagnosis, prevalence and management of inherited and acquired forms. Hamostaseologie 2014, 34, 160–166. [Google Scholar] [PubMed]
- Muszbek, L.; Katona, É. Diagnosis and Management of Congenital and Acquired FXIII Deficiencies. Semin. Thromb. Hemostasis 2016, 42, 429–439. [Google Scholar]
- Fox, B.A.; Yee, V.C.; Pedersen, L.C.; Le Trong, I.; Bishop, P.D.; Stenkamp, R.E.; Teller, D.C. Identification of the calcium binding site and a novel ytterbium site in blood coagulation factor XIII by x-ray crystallography. J. Biol. Chem. 1999, 274, 4917–4923. [Google Scholar] [CrossRef] [PubMed]
- Stieler, M.; Weber, J.; Hils, M.; Kolb, P.; Heine, A.; Buchold, C.; Pasternack, R.; Klebe, G. Structure of active coagulation factor XIII triggered by calcium binding: basis for the design of next-generation anticoagulants. Angew. Chem. Int. Ed. Engl. 2013, 52, 11930–11934. [Google Scholar] [CrossRef]
- Souri, M.; Kaetsu, H.; Ichinose, A. Sushi domains in the B subunit of factor XIII responsible for oligomer assembly. Biochemistry 2008, 47, 8656–8664. [Google Scholar] [CrossRef] [PubMed]
- Thomas, A.; Biswas, A.; Ivaskevicius, V.; Oldenburg, J. Structural and functional influences of coagulation factor XIII subunit B heterozygous missense mutants. Mol. Genet. Genomic Med. 2015, 3, 258–271. [Google Scholar] [CrossRef]
- Schroeder, V.; Kohler, H.P. Factor XIII: Structure and Function. Semin. Thromb. Hemostasis 2016, 42, 422–428. [Google Scholar]
- Akhter, M.S.; Singh, S.; Yadegari, H.; Ivaskevicius, V.; Oldenburg, J.; Biswas, A. Exploring the structural similarity yet functional distinction between coagulation factor XIII-B and complement factor H sushi domains. J. Thromb. Thrombolysis 2019. [Google Scholar] [CrossRef]
- Lassila, R. Clinical Use of Factor XIII Concentrates. Semin. Thromb. Hemostasis 2016, 42, 440–444. [Google Scholar] [CrossRef]
- Yan, M.T.S.; Rydz, N.; Goodyear, D.; Sholzberg, M. Acquired factor XIII deficiency: A review. Trans. Apheresis Sci. 2018, 57, 724–730. [Google Scholar] [CrossRef]
- Nugent, D. Corifact™/Fibrogammin® P in the prophylactic treatment of hereditary factor XIII deficiency: results of a prospective, multicenter, open-label study. Thromb. Res. 2012, 130, S12–S14. [Google Scholar] [CrossRef]
- Dreyfus, M.; Barrois, D.; Borg, J.-Y.; Claeyssens, S.; Torchet, M.-F.; Arnuti, B.; Pautard, B. Successful long-term replacement therapy with FXIII concentrate (Fibrogammin(®) P) for severe congenital factor XIII deficiency: a prospective multicentre study. J. Thromb. Haemostasis 2011, 9, 1264–1266. [Google Scholar] [CrossRef]
- Dodt, J.; Volkers, P.; Seitz, R. Factor XIIIa generation assay: a tool for studying factor XIII function in plasma. Anal. Biochem. 2013, 439, 145–151. [Google Scholar] [CrossRef]
- Norman, D.G.; Barlow, P.N.; Baron, M.; Day, A.J.; Sim, R.B.; Campbell, I.D. Three-dimensional structure of a complement control protein module in solution. J. Mol. Biol. 1991, 219, 717–725. [Google Scholar] [CrossRef]
- Biswas, A.; Thomas, A.; Bevans, C.G.; Ivaskevicius, V.; Oldenburg, J. In vitro secretion deficits are common among human coagulation factor XIII subunit B missense mutants: correlations with patient phenotypes and molecular models. Hum. Mutat. 2013, 34, 1490–1500. [Google Scholar] [CrossRef]
- Perkins, S.J.; Nan, R.; Li, K.; Khan, S.; Miller, A. Complement factor H-ligand interactions: self-association, multivalency and dissociation constants. Immunobiology 2012, 217, 281–297. [Google Scholar] [CrossRef] [PubMed]
- Krushkal, J.; Bat, O.; Gigli, I. Evolutionary relationships among proteins encoded by the regulator of complement activation gene cluster. Mol. Biol. Evol. 2000, 17, 1718–1730. [Google Scholar] [CrossRef] [PubMed]
- Dodds, A.W.; Law, S.K. The phylogeny and evolution of the thioester bond-containing proteins C3, C4 and alpha 2-macroglobulin. Immunol. Rev. 1998, 166, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, P.B.; Quigley, J.P. Alpha2-macroglobulin: an evolutionarily conserved arm of the innate immune system. Dev. Comp. Immunol. 1999, 23, 375–390. [Google Scholar] [CrossRef]
- Nikolajsen, C.L.; Dyrlund, T.F.; Poulsen, E.T.; Enghild, J.J.; Scavenius, C. Coagulation factor XIIIa substrates in human plasma: identification and incorporation into the clot. J. Biol. Chem. 2014, 289, 6526–6534. [Google Scholar] [CrossRef]
- Gupta, S.; Biswas, A.; Akhter, M.S.; Krettler, C.; Reinhart, C.; Dodt, J.; Reuter, A.; Philippou, H.; Ivaskevicius, V.; Oldenburg, J. Revisiting the mechanism of coagulation factor XIII activation and regulation from a structure/functional perspective. Sci. Rep. 2016, 6, 30105. [Google Scholar] [CrossRef] [PubMed]
- Souri, M.; Osaki, T.; Ichinose, A. The Non-catalytic B Subunit of Coagulation Factor XIII Accelerates Fibrin Cross-linking. J. Biol. Chem. 2015, 290, 12027–12039. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Pang, Y.; Liu, X.; Li, Q.-W. Structure, distribution, classification, and function of C1q protein family: a review. Yi Chuan = Hereditas 2013, 35, 1072–1080. [Google Scholar] [CrossRef]
- Devasahayam, J.; Erode-Singaravelu, G.; Bhat, Z.; Oliver, T.; Chandran, A.; Zeng, X.; Dakshinesh, P.; Pillai, U. C1q Nephropathy: The Unique Underrecognized Pathological Entity. Anal. Cell. Pathol. 2015, 2015, 490413. [Google Scholar] [CrossRef]
- Rossi, O.; Coward, C.; Goh, Y.S.; Claassens, J.W.C.; MacLennan, C.A.; Verbeek, S.J.; Mastroeni, P. The essential role of complement in antibody-mediated resistance to Salmonella. Immunology 2019, 1, 69–73. [Google Scholar] [CrossRef]
- De Sain-van der Velden, M.G.; Kaysen, G.A.; de Meer, K.; Stellaard, F.; Voorbij, H.A.; Reijngoud, D.J.; Rabelink, T.J.; Koomans, H.A. Proportionate increase of fibrinogen and albumin synthesis in nephrotic patients: measurements with stable isotopes. Kidney Int. 1998, 53, 181–188. [Google Scholar] [CrossRef] [PubMed]
- De Sain-van der Velden, M.G.; Rabelink, T.J.; Reijngoud, D.J.; Gadellaa, M.M.; Voorbij, H.A.; Stellaard, F.; Kaysen, G.A. Plasma alpha 2 macroglobulin is increased in nephrotic patients as a result of increased synthesis alone. Kidney Int. 1998, 54, 530–535. [Google Scholar] [CrossRef]
- Ritchie, R.F.; Palomaki, G.E.; Neveux, L.M.; Navolotskaia, O.; Ledue, T.B.; Craig, W.Y. Reference distributions for alpha2-macroglobulin: a practical, simple and clinically relevant approach in a large cohort. J. Clin. Lab. Anal. 2004, 18, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Richardson, V.R.; Schroeder, V.; Grant, P.J.; Standeven, K.F.; Carter, A.M. Complement C3 is a substrate for activated factor XIII that is cross-linked to fibrin during clot formation. Br. J. Haematol. 2013, 160, 116–119. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Akhter, M.S.; Dodt, J.; Sharma, A.; Kaniyappan, S.; Yadegari, H.; Ivaskevicius, V.; Oldenburg, J.; Biswas, A. Disruption of Structural Disulfides of Coagulation FXIII-B Subunit; Functional Implications for a Rare Bleeding Disorder. Int. J. Mol. Sci. 2019, 20, 1956. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Singh, S.; Akhter, M.S.; Dodt, J.; Volkers, P.; Reuter, A.; Reinhart, C.; Krettler, C.; Oldenburg, J.; Biswas, A. Identification of Potential Novel Interacting Partners for Coagulation Factor XIII B (FXIII-B) Subunit, a Protein Associated with a Rare Bleeding Disorder. Int. J. Mol. Sci. 2019, 20, 2682. https://doi.org/10.3390/ijms20112682
Singh S, Akhter MS, Dodt J, Volkers P, Reuter A, Reinhart C, Krettler C, Oldenburg J, Biswas A. Identification of Potential Novel Interacting Partners for Coagulation Factor XIII B (FXIII-B) Subunit, a Protein Associated with a Rare Bleeding Disorder. International Journal of Molecular Sciences. 2019; 20(11):2682. https://doi.org/10.3390/ijms20112682
Chicago/Turabian StyleSingh, Sneha, Mohammad Suhail Akhter, Johannes Dodt, Peter Volkers, Andreas Reuter, Christoph Reinhart, Christoph Krettler, Johannes Oldenburg, and Arijit Biswas. 2019. "Identification of Potential Novel Interacting Partners for Coagulation Factor XIII B (FXIII-B) Subunit, a Protein Associated with a Rare Bleeding Disorder" International Journal of Molecular Sciences 20, no. 11: 2682. https://doi.org/10.3390/ijms20112682
APA StyleSingh, S., Akhter, M. S., Dodt, J., Volkers, P., Reuter, A., Reinhart, C., Krettler, C., Oldenburg, J., & Biswas, A. (2019). Identification of Potential Novel Interacting Partners for Coagulation Factor XIII B (FXIII-B) Subunit, a Protein Associated with a Rare Bleeding Disorder. International Journal of Molecular Sciences, 20(11), 2682. https://doi.org/10.3390/ijms20112682





