The Unique Mechanisms of Cellular Proliferation, Migration and Apoptosis are Regulated through Oocyte Maturational Development—A Complete Transcriptomic and Histochemical Study
Abstract
1. Introduction
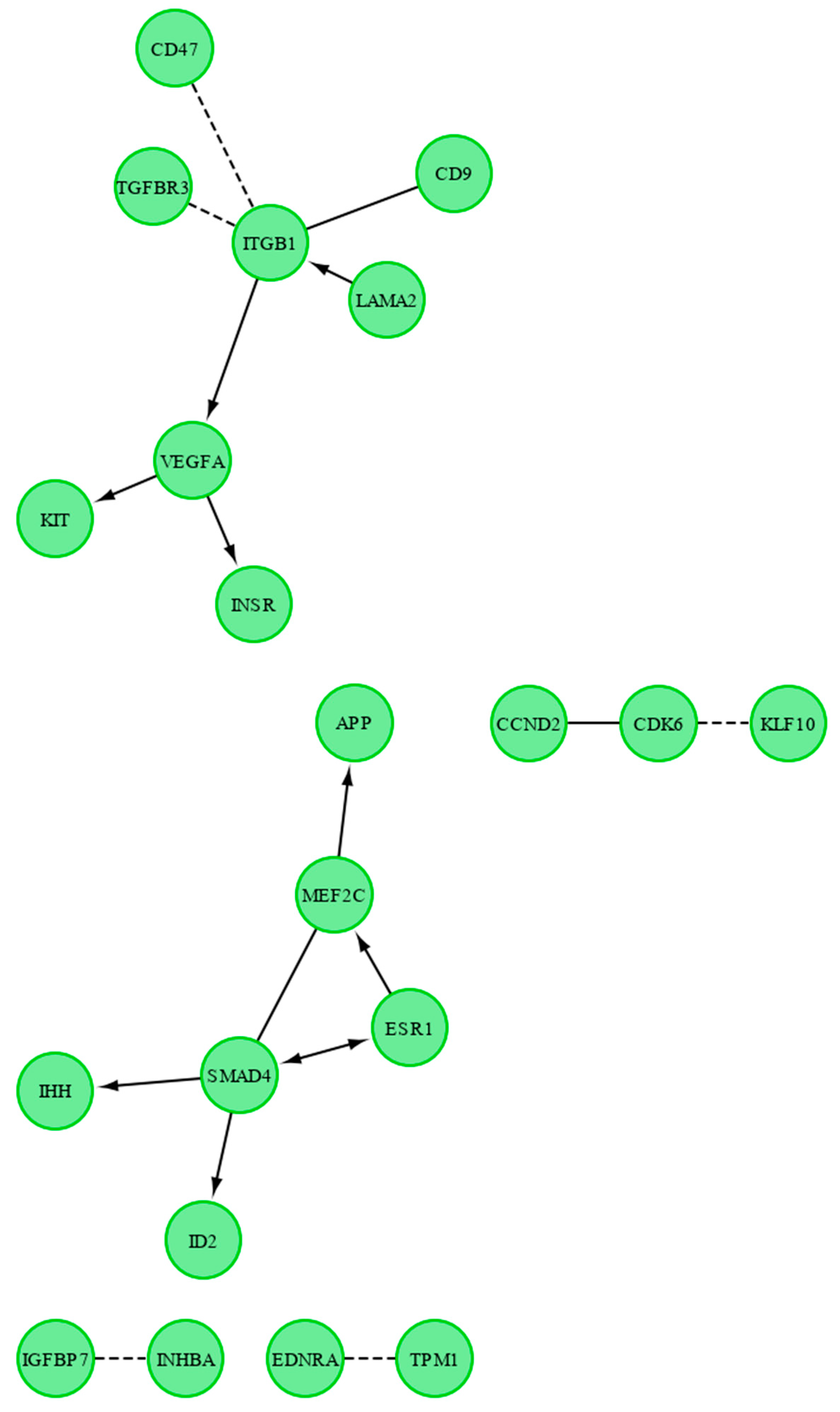
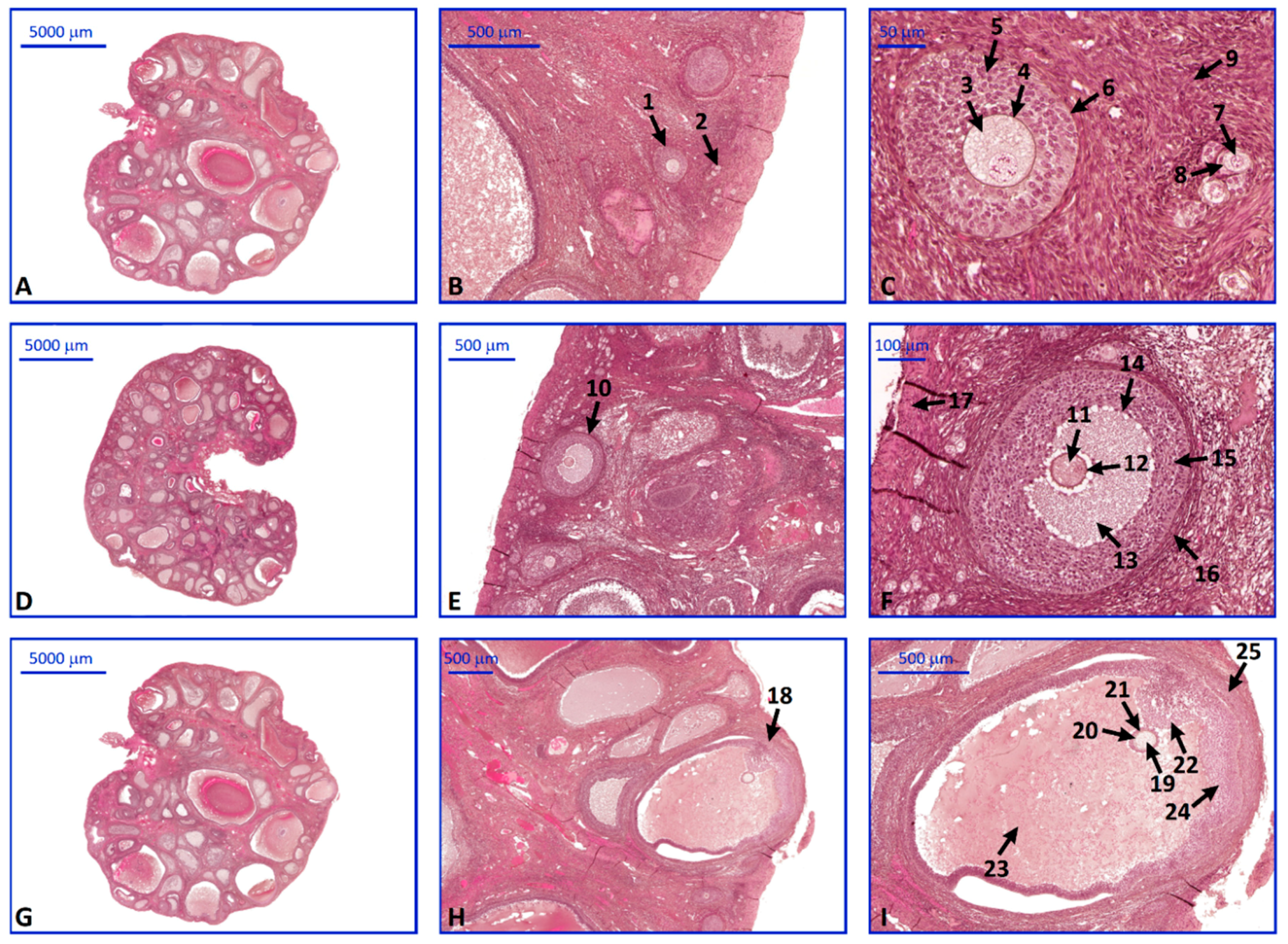
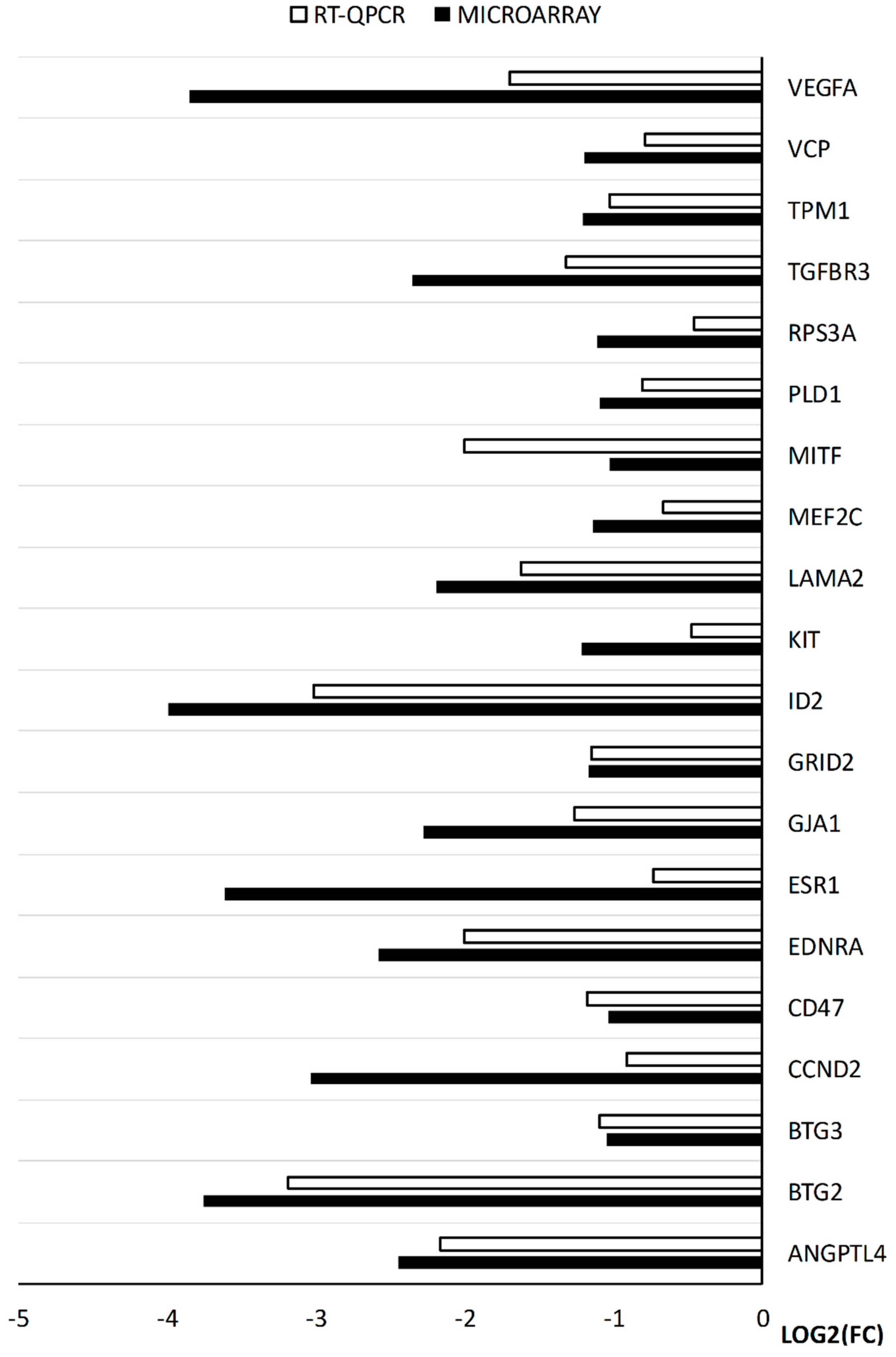
2. Results
3. Discussion
4. Material and Methods
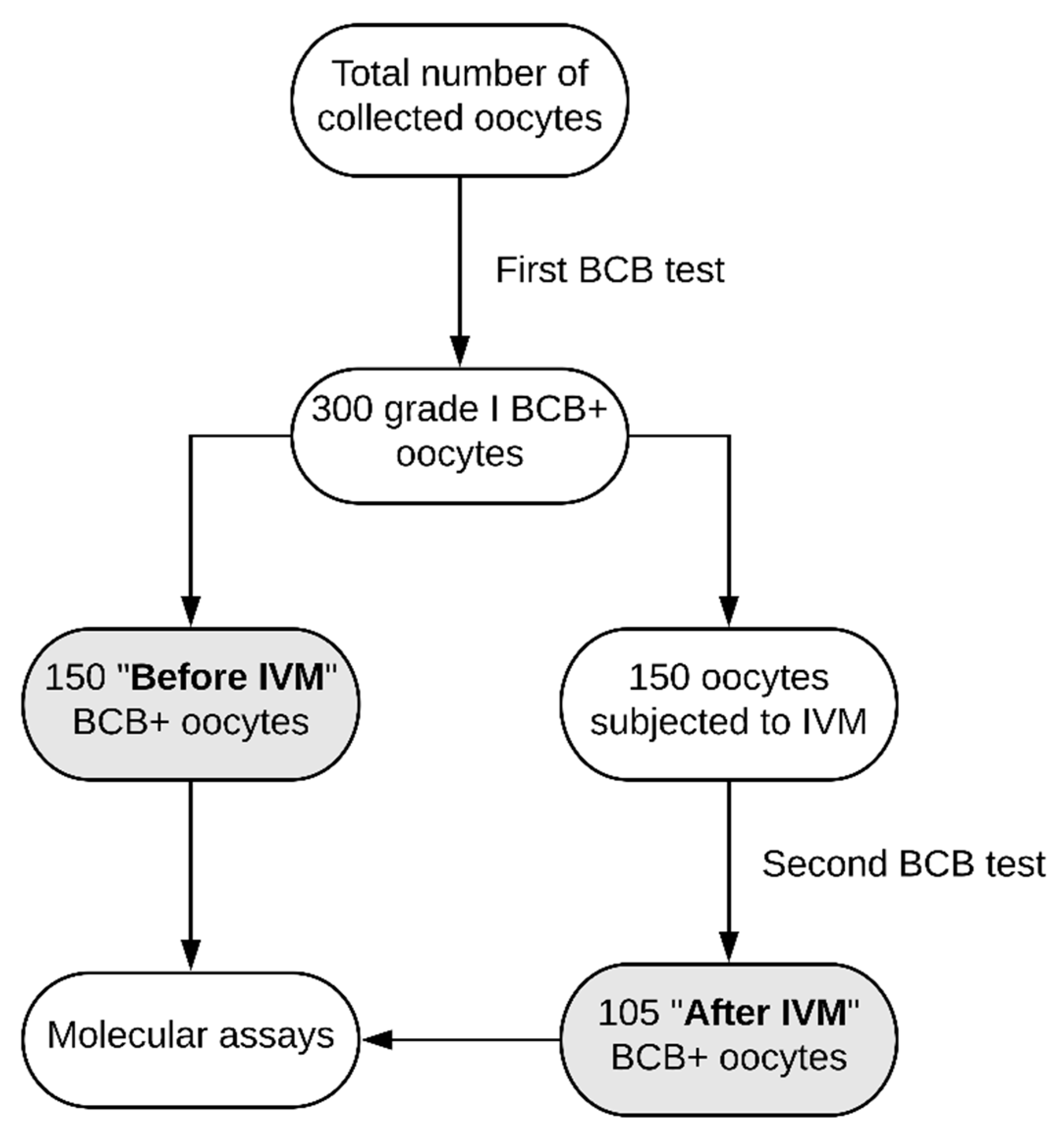
4.1. Experimental Design
4.2. Animals
4.3. Collection of Porcine Ovaries and Cumulus-Oocyte-Complexes (COCs)
4.4. Assessment of Oocyte Developmental Competence by BCB Test
4.5. In Vitro Maturation of Porcine Cumulus-Oocyte-Complexes (COCs)
4.6. Evaluation of Oocytes
4.7. RNA Extraction from Porcine Oocytes
4.8. Microarray Expression Analysis and Statistics
4.9. Real-time Quantitative Polymerase Chain Reaction (RT-qPCR) Analysis
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Hunt, P.A.; Hassold, T.J. Human female meiosis: What makes a good egg go bad? Trends Genet. 2008, 24, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Viswanathan, S.R.; Mermel, C.H.; Lu, J.; Lu, C.-W.; Golub, T.R.; Daley, G.Q. microRNA Expression during Trophectoderm Specification. PLoS ONE 2009, 4, e6143. [Google Scholar] [CrossRef] [PubMed]
- Sinha, P.B.; Tesfaye, D.; Rings, F.; Hossien, M.; Hoelker, M.; Held, E.; Neuhoff, C.; Tholen, E.; Schellander, K.; Salilew-Wondim, D. MicroRNA-130b is involved in bovine granulosa and cumulus cells function, oocyte maturation and blastocyst formation. J. Ovarian Res. 2017, 10, 37. [Google Scholar] [CrossRef] [PubMed]
- Kempisty, B.; Ziółkowska, A.; Ciesiółka, S.; Piotrowska, H.; Antosik, P.; Bukowska, D.; Nowicki, M.; Brüssow, K.P.; Zabel, M. Study on connexin gene and protein expression and cellular distribution in relation to real-time proliferation of porcine granulosa cells. J. Biol. Regul. Homeost. Agents 2014, 28, 625–635. [Google Scholar] [PubMed]
- Diaz, F.J.; Wigglesworth, K.; Eppig, J.J. Oocytes are required for the preantral granulosa cell to cumulus cell transition in mice. Dev. Biol. 2007, 305, 300–311. [Google Scholar] [CrossRef] [PubMed]
- Rybska, M.; Knap, S.; Jankowski, M.; Jeseta, M.; Bukowska, D.; Antosik, P.; Nowicki, M.; Zabel, M.; Kempisty, B.; Jaśkowski, J.M. Cytoplasmic and nuclear maturation of oocytes in mammals–living in the shadow of cells developmental capability. Med. J. Cell Biol. 2018, 6, 13–17. [Google Scholar] [CrossRef]
- Gilchrist, R.; Ritter, L.; Armstrong, D. Oocyte–somatic cell interactions during follicle development in mammals. Anim. Reprod. Sci. 2004, 82–83, 431–446. [Google Scholar] [CrossRef]
- Li, R.; Norman, R.J.; Armstrong, D.T.; Gilchrist, R.B. Oocyte-secreted factor(s) determine functional differences between bovine mural granulosa cells and cumulus cells. Biol. Reprod. 2000, 63, 839–845. [Google Scholar] [CrossRef]
- Macaulay, A.D.; Gilbert, I.; Caballero, J.; Barreto, R.; Fournier, E.; Tossou, P.; Sirard, M.-A.; Clarke, H.J.; Khandjian, É.W.; Richard, F.J.; et al. The Gametic Synapse: RNA Transfer to the Bovine Oocyte1. Biol. Reprod. 2014, 91, 90. [Google Scholar] [CrossRef]
- Biase, F.H.; Kimble, K.M. Functional signaling and gene regulatory networks between the oocyte and the surrounding cumulus cells. BMC Genomics 2018, 19, 351. [Google Scholar] [CrossRef]
- Nawrocki, M.J.; Celichowski, P.; Budna, J.; Bryja, A.; Kranc, W.; Ciesiółka, S.; Borys, S.; Knap, S.; Jeseta, M.; Khozmi, R.; et al. The blood vessels development, morphogenesis and blood circulation are three ontologic groups highly up-regulated in porcine oocytes before in vitro maturation. Adv. Cell Biol. 2017, 5, 135–142. [Google Scholar] [CrossRef]
- Budna, J.; Bryja, A.; Celichowski, P.; Kranc, W.; Ciesiółka, S.; Borys, S.; Rybska, M.; Kolecka-Bednarczyk, A.; Jeseta, M.; Bukowska, D.; et al. “Bone Development” Is an Ontology Group Upregulated in Porcine Oocytes Before In Vitro Maturation: A Microarray Approach. DNA Cell Biol. 2017, 36, 638–646. [Google Scholar] [CrossRef] [PubMed]
- Kordus, R.J.; LaVoie, H.A. Granulosa cell biomarkers to predict pregnancy in ART: Pieces to solve the puzzle. Reproduction 2017, 153, R69–R83. [Google Scholar] [CrossRef]
- Rybska, M.; Knap, S.; Jankowski, M.; Jeseta, M.; Bukowska, D.; Antosik, P.; Nowicki, M.; Zabel, M.; Kempisty, B.; Jaśkowski, J.M. Characteristic of factors influencing the proper course of folliculogenesis in mammals. Med. J. Cell Biol. 2018, 6, 33–38. [Google Scholar] [CrossRef]
- Barrett, S.L.; Albertini, D.F. Cumulus cell contact during oocyte maturation in mice regulates meiotic spindle positioning and enhances developmental competence. J. Assist. Reprod. Genet. 2010, 27, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Komatsu, K.; Masubuchi, S. Mouse oocytes connect with granulosa cells by fusing with cell membranes and form a large complex during follicle development. Biol. Reprod. 2018, 99, 527–535. [Google Scholar] [CrossRef]
- Chachuła, A.; Kranc, W.; Budna, J.; Bryja, A.; Ciesiólka, S.; Wojtanowicz-Markiewicz, K.; Piotrowska, H.; Bukowska, D.; Krajecki, M.; Antosik, P.; et al. The differentiation of mammalian ovarian granulosa cells living in the shadow of cellular developmental capacity. J. Biol. Regul. Homeost. Agents 2016, 30, 627–634. [Google Scholar]
- Budna, J.; Celichowski, P.; Karimi, P.; Kranc, W.; Bryja, A.; Ciesiółka, S.; Rybska, M.; Borys, S.; Jeseta, M.; Bukowska, D.; et al. Does Porcine Oocytes Maturation in Vitro is Regulated by Genes Involved in Transforming Growth Factor B Receptor Signaling Pathway? Adv. Cell Biol. 2017, 5, 1–14. [Google Scholar] [CrossRef]
- Hutt, K.J.; Albertini, D.F. An oocentric view of folliculogenesis and embryogenesis. Reprod. Biomed. Online 2007, 14, 758–764. [Google Scholar] [CrossRef]
- Dias, F.C.F.; Khan, M.I.R.; Sirard, M.A.; Adams, G.P.; Singh, J. Transcriptome analysis of granulosa cells after conventional vs long FSH-induced superstimulation in cattle. BMC Genomics 2018, 19, 258. [Google Scholar] [CrossRef]
- Zheng, W.; Liu, K. Maternal Control of Mouse Preimplantation Development. Results Probl. Cell Differ. 2012, 55, 115–139. [Google Scholar] [PubMed]
- Kranc, W.; Budna, J.; Chachuła, A.; Borys, S.; Bryja, A.; Rybska, M.; Ciesiółka, S.; Sumelka, E.; Jeseta, M.; Brüssow, K.P.; et al. “Cell Migration” Is the Ontology Group Differentially Expressed in Porcine Oocytes Before and After In Vitro Maturation: A Microarray Approach. DNA Cell Biol. 2017, 36, 273–282. [Google Scholar] [CrossRef] [PubMed]
- Kempisty, B.; Ziółkowska, A.; Piotrowska, H.; Zawierucha, P.; Antosik, P.; Bukowska, D.; Ciesiółka, S.; Jaśkowski, J.M.; Brüssow, K.P.; Nowicki, M.; Zabel, M. Real-time proliferation of porcine cumulus cells is related to the protein levels and cellular distribution of Cdk4 and Cx43. Theriogenology 2013, 80, 411–420. [Google Scholar] [CrossRef] [PubMed]
- Borup, R.; Thuesen, L.L.; Andersen, C.Y.; Nyboe-Andersen, A.; Ziebe, S.; Winther, O.; Grøndahl, M.L. Competence Classification of Cumulus and Granulosa Cell Transcriptome in Embryos Matched by Morphology and Female Age. PLoS ONE 2016, 11, e0153562. [Google Scholar] [CrossRef] [PubMed]
- Borys, S.; Khozmi, R.; Kranc, W.; Bryja, A.; Dyszkiewicz-Konwińska, M.; Jeseta, M.; Kempisty, B. Recent findings of the types of programmed cell death. Adv. Cell Biol. 2017, 5, 43–49. [Google Scholar] [CrossRef]
- Artini, P.G.; Tatone, C.; Sperduti, S.; D’Aurora, M.; Franchi, S.; Di Emidio, G.; Ciriminna, R.; Vento, M.; Di Pietro, C.; Stuppia, L.; et al. Cumulus cells surrounding oocytes with high developmental competence exhibit down-regulation of phosphoinositol 1, 3 kinase/protein kinase B (PI3K/AKT) signalling genes involved in proliferation and survival. Hum. Reprod. 2017, 32, 2474–2484. [Google Scholar] [CrossRef] [PubMed]
- Massari, M.E.; Murre, C. Helix-loop-helix proteins: Regulators of transcription in eucaryotic organisms. Mol. Cell. Biol. 2000, 20, 429–440. [Google Scholar] [CrossRef]
- Budna, J.; Chachuła, A.; Kaźmierczak, D.; Rybska, M.; Ciesiółka, S.; Bryja, A.; Kranc, W.; Borys, S.; Żok, A.; Bukowska, D.; et al. Morphogenesis-related gene-expression profile in porcine oocytes before and after in vitro maturation. Zygote 2017, 25, 331–340. [Google Scholar] [CrossRef]
- Guo, L.; Lan, J.; Lin, Y.; Guo, P.; Nie, Q.; Mao, Q.; Ren, L.; Qiu, Y. Hypoxia/ischemia up-regulates Id2 expression in neuronal cells in vivo and in vitro. Neurosci. Lett. 2013, 554, 88–93. [Google Scholar] [CrossRef]
- Hazzard, T.M.; Xu, F.; Stouffer, R.L. Injection of soluble vascular endothelial growth factor receptor 1 into the preovulatory follicle disrupts ovulation and subsequent luteal function in rhesus monkeys. Biol. Reprod. 2002, 67, 1305–1312. [Google Scholar] [CrossRef]
- Trau, H.A.; Brännström, M.; Curry, T.E.; Duffy, D.M. Prostaglandin E2 and vascular endothelial growth factor A mediate angiogenesis of human ovarian follicular endothelial cells. Hum. Reprod. 2016, 31, dev320. [Google Scholar] [CrossRef] [PubMed]
- Kranc, W.; Celichowski, P.; Budna, J.; Khozmi, R.; Bryja, A.; Ciesiółka, S.; Rybska, M.; Borys, S.; Jeseta, M.; Bukowska, D.; et al. Positive Regulation Of Macromolecule Metabolic Process Belongs To The Main Mechanisms Crucial For Porcine Ooocytes Maturation. Adv. Cell Biol. 2017, 5, 15–31. [Google Scholar] [CrossRef]
- Anchordoquy, J.M.; Anchordoquy, J.P.; Testa, J.A.; Sirini, M.Á.; Furnus, C.C. Influence of vascular endothelial growth factor and Cysteamine on in vitro bovine oocyte maturation and subsequent embryo development. Cell Biol. Int. 2015, 39, 1090–1098. [Google Scholar] [CrossRef] [PubMed]
- Celichowski, P.; Nawrocki, M.J.M.J.; Dyszkiewicz-Konwińska, M.; Jankowski, M.; Budna, J.; Bryja, A.; Kranc, W.; Borys, S.; Knap, S.; Ciesiółka, S.; et al. “Positive Regulation of RNA Metabolic Process” Ontology Group Highly Regulated in Porcine Oocytes Matured In Vitro: A Microarray Approach. Biomed Res. Int. 2018, 2018, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Tirone, F. The gene PC3TIS21/BTG2, prototype member of the PC3/BTG/TOB family: Regulator in control of cell growth, differentiation, and DNA repair? J. Cell. Physiol. 2001, 187, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Goldenberg, R.L.; Vaitukaitis, J.L.; Ross, G.T. Estrogen and Follicle Stimulating Hormone Interactions on Follicle Growth in Rats. Endocrinology 1972, 90, 1492–1498. [Google Scholar] [CrossRef]
- Al-Edani, T.; Assou, S.; Ferrières, A.; Bringer Deutsch, S.; Gala, A.; Lecellier, C.-H.; Aït-Ahmed, O.; Hamamah, S. Female aging alters expression of human cumulus cells genes that are essential for oocyte quality. Biomed Res. Int. 2014, 2014, 964614. [Google Scholar] [CrossRef]
- Robker, R.L.; Richards, J.S. Hormonal control of the cell cycle in ovarian cells: Proliferation versus differentiation. Biol. Reprod. 1998, 59, 476–482. [Google Scholar] [CrossRef]
- Van Montfoort, A.P.A.; Geraedts, J.P.M.; Dumoulin, J.C.M.; Stassen, A.P.M.; Evers, J.L.H.; Ayoubi, T.A.Y. Differential gene expression in cumulus cells as a prognostic indicator of embryo viability: A microarray analysis. Mol. Hum. Reprod. 2008, 14, 157–168. [Google Scholar] [CrossRef]
- Bigham, A.W.; Julian, C.G.; Wilson, M.J.; Vargas, E.; Browne, V.A.; Shriver, M.D.; Moore, L.G. Maternal PRKAA1 and EDNRA genotypes are associated with birth weight, and PRKAA1 with uterine artery diameter and metabolic homeostasis at high altitude. Physiol. Genomics 2014, 46, 687–697. [Google Scholar] [CrossRef]
- Kawamura, K.; Ye, Y.; Liang, C.G.; Kawamura, N.; Gelpke, M.S.; Rauch, R.; Tanaka, T.; Hsueh, A.J.W. Paracrine regulation of the resumption of oocyte meiosis by endothelin-1. Dev. Biol. 2009, 327, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.X.; Tong, D.; El-Gehani, F.; Tekpetey, F.R.; Kidder, G.M. Connexin expression and gap junctional coupling in human cumulus cells: Contribution to embryo quality. J. Cell. Mol. Med. 2009, 13, 972–984. [Google Scholar] [CrossRef] [PubMed]
- Li, S.-H.; Lin, M.-H.; Hwu, Y.-M.; Lu, C.-H.; Yeh, L.-Y.; Chen, Y.-J.; Lee, R.K.-K. Correlation of cumulus gene expression of GJA1, PRSS35, PTX3, and SERPINE2 with oocyte maturation, fertilization, and embryo development. Reprod. Biol. Endocrinol. 2015, 13, 93. [Google Scholar] [CrossRef] [PubMed]
- DeLaughter, D.M.; Clark, C.R.; Christodoulou, D.C.; Seidman, C.E.; Baldwin, H.S.; Seidman, J.G.; Barnett, J.V. Transcriptional Profiling of Cultured, Embryonic Epicardial Cells Identifies Novel Genes and Signaling Pathways Regulated by TGFβR3 In Vitro. PLoS ONE 2016, 11, e0159710. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.M.; Sif, S.; Ostrowski, M.C.; Sankar, U. Defective co-activator recruitment in osteoclasts from microphthalmia-oak ridge mutant mice. J. Cell. Physiol. 2009, 220, 230–237. [Google Scholar] [CrossRef] [PubMed]
- Mathias, M.D.; Sockolosky, J.T.; Chang, A.Y.; Tan, K.S.; Liu, C.; Garcia, K.C.; Scheinberg, D.A. CD47 blockade enhances therapeutic activity of TCR mimic antibodies to ultra-low density cancer epitopes. Leukemia 2017, 31, 2254–2257. [Google Scholar] [CrossRef] [PubMed]
- Lv, C.; Wang, H.; Tong, Y.; Yin, H.; Wang, D.; Yan, Z.; Liang, Y.; Wu, D.; Su, Q. The function of BTG3 in colorectal cancer cells and its possible signaling pathway. J. Cancer Res. Clin. Oncol. 2018, 144, 295–308. [Google Scholar] [CrossRef]
- Garrido, J.L.; Wheeler, D.; Vega, L.L.; Friedman, P.A.; Romero, G. Role of Phospholipase D in Parathyroid Hormone Type 1 Receptor Signaling and Trafficking. Mol. Endocrinol. 2009, 23, 2048–2059. [Google Scholar] [CrossRef]
- Tang, Y.; He, Y.; Li, C.; Mu, W.; Zou, Y.; Liu, C.; Qian, S.; Zhang, F.; Pan, J.; Wang, Y.; et al. RPS3A positively regulates the mitochondrial function of human periaortic adipose tissue and is associated with coronary artery diseases. Cell Discov. 2018, 4, 52. [Google Scholar] [CrossRef]
- Ali, Z.; Zulfiqar, S.; Klar, J.; Wikström, J.; Ullah, F.; Khan, A.; Abdullah, U.; Baig, S.; Dahl, N. Homozygous GRID2 missense mutation predicts a shift in the D-serine binding domain of GluD2 in a case with generalized brain atrophy and unusual clinical features. BMC Med. Genet. 2017, 18, 144. [Google Scholar] [CrossRef]
- Bastola, P.; Neums, L.; Schoenen, F.J.; Chien, J. VCP inhibitors induce endoplasmic reticulum stress, cause cell cycle arrest, trigger caspase-mediated cell death and synergistically kill ovarian cancer cells in combination with Salubrinal. Mol. Oncol. 2016, 10, 1559–1574. [Google Scholar] [CrossRef] [PubMed]
- Roca, J.; Martinez, E.; Vazquez, J.M.; Lucas, X. Selection of immature pig oocytes for homologous in vitro penetration assays with the brilliant cresyl blue test. Reprod. Fertil. Dev. 1998, 10, 479–485. [Google Scholar] [CrossRef]
- Nawrocki, M.J.; Budna, J.; Celichowski, P.; Khozmi, R.; Bryja, A.; Kranc, W.; Borys, S.; Ciesiółka, S.; Knap, S.; Jeseta, M.; et al. Analysis of fructose and mannose–regulatory peptides signaling pathway in porcine epithelial oviductal cells (OECs) primary cultured long-term in vitro. Adv. Cell Biol. 2017, 5, 129–135. [Google Scholar] [CrossRef]
- Kranc, W.; Jankowski, M.; Budna, J.; Celichowski, P.; Khozmi, R.; Bryja, A.; Borys, S.; Dyszkiewicz-Konwińska, M.; Jeseta, M.; Magas, M.; et al. Amino acids metabolism and degradation is regulated during porcine oviductal epithelial cells (OECs) primary culture in vitro—A signaling pathways activation approach. Med. J. Cell Biol. 2018, 6, 18–26. [Google Scholar] [CrossRef]
- Walter, W.; Sánchez-Cabo, F.; Ricote, M. GOplot: An R package for visually combining expression data with functional analysis. Bioinformatics 2015, 31, 2912–2914. [Google Scholar] [CrossRef]
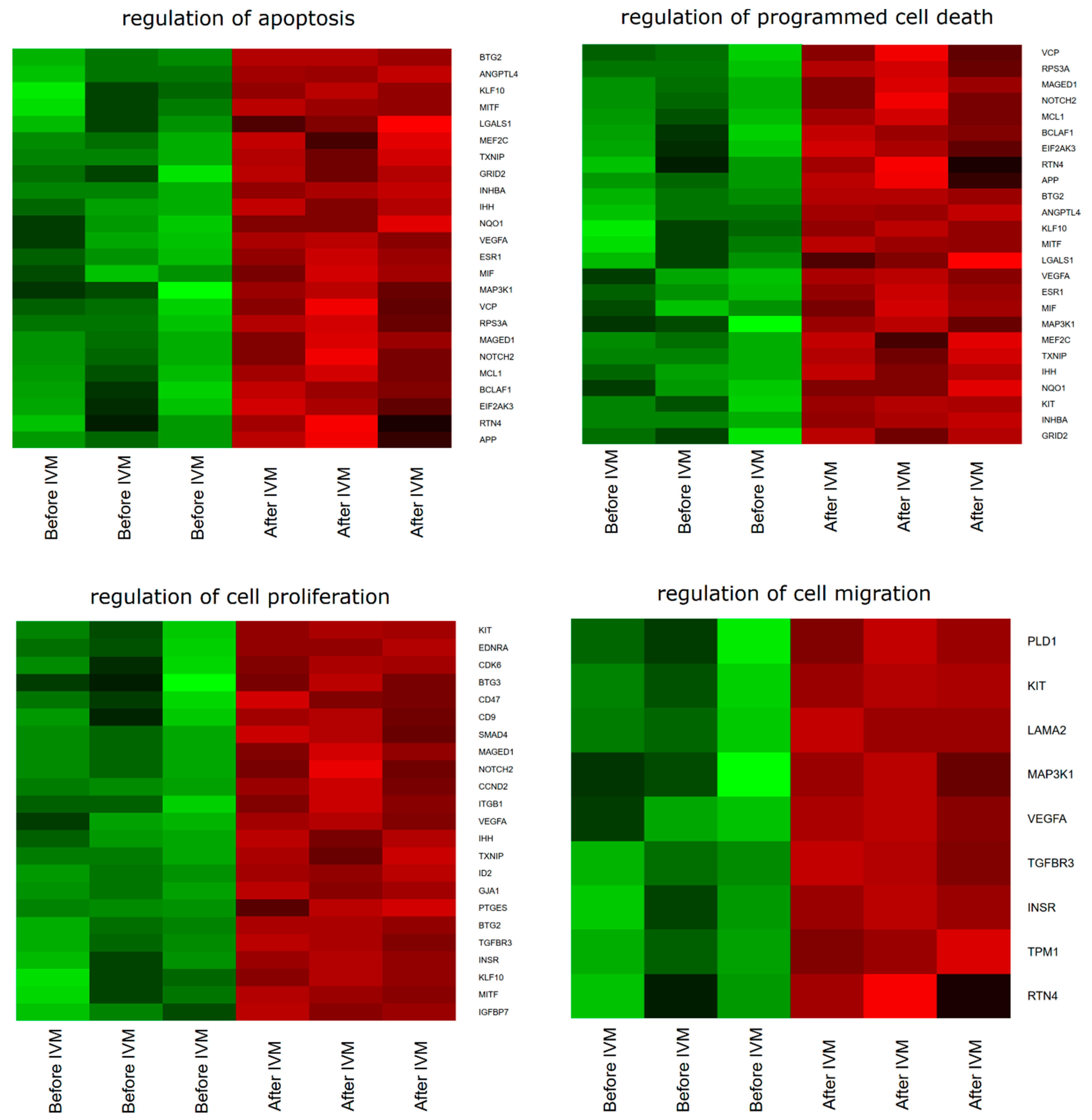

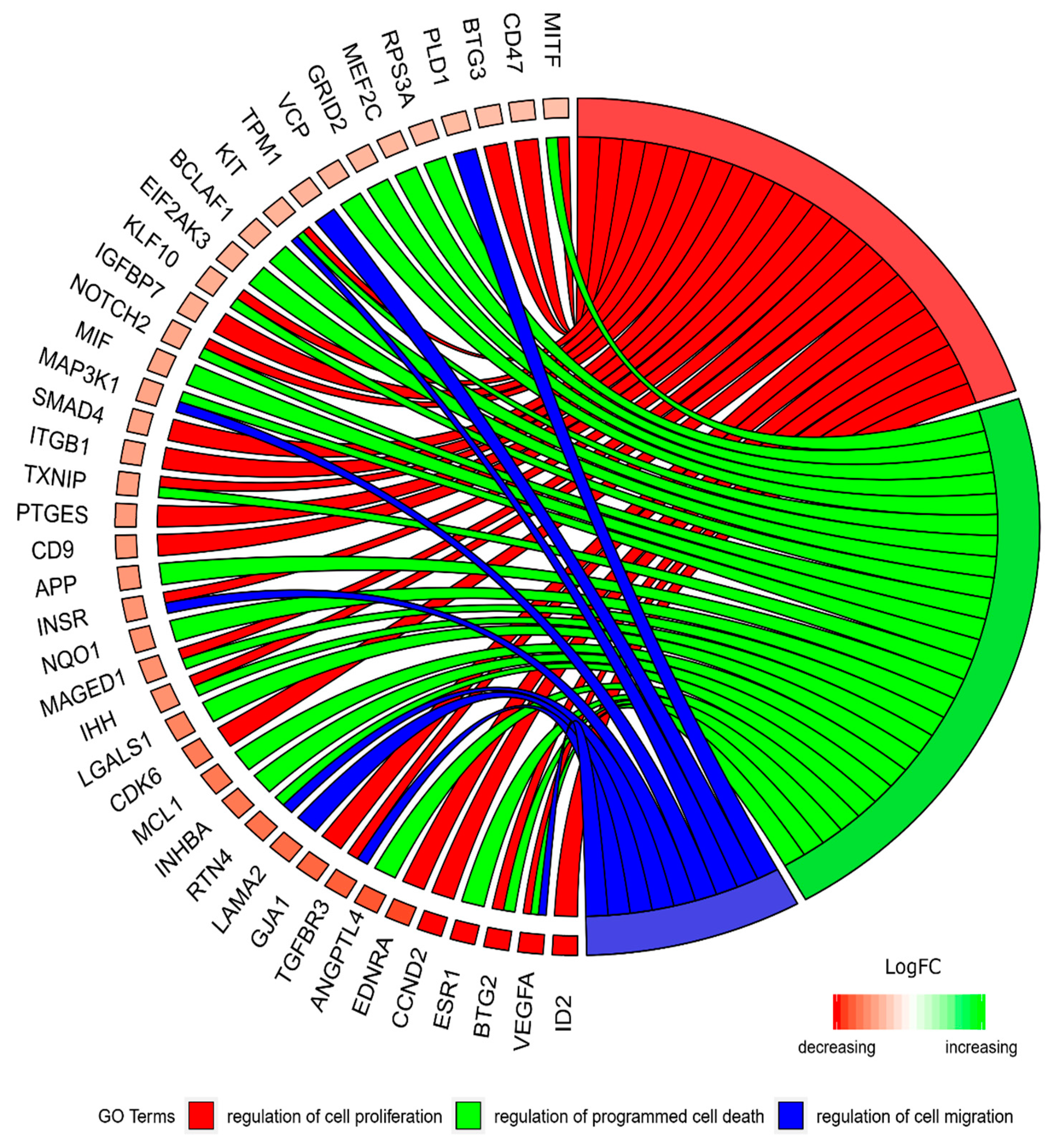
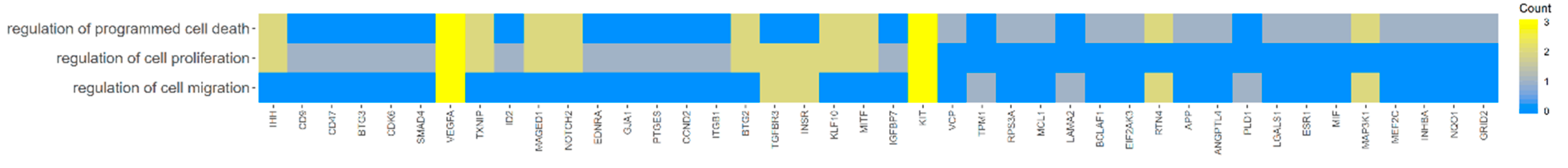
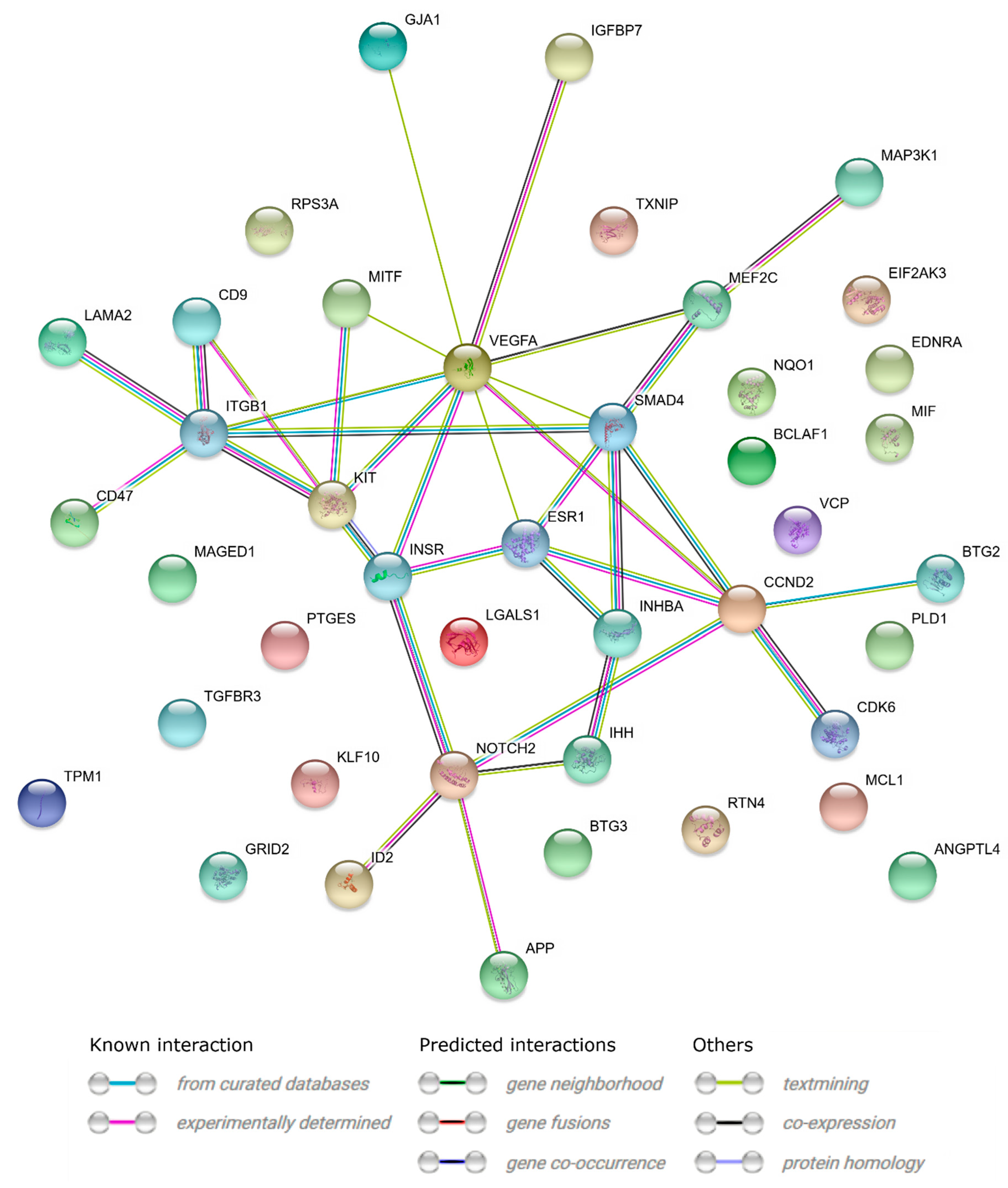
| Gene | Fold Change | p-Value |
|---|---|---|
| ID2 | 0.062979704 | 4.74E-05 |
| VEGFA | 0.069689389 | 0.001912689 |
| BTG2 | 0.074386393 | 9.55E-05 |
| ESR1 | 0.081629841 | 0.000522187 |
| CCND2 | 0.121809064 | 0.000178804 |
| EDNRA | 0.166939028 | 0.00185422 |
| ANGPTL4 | 0.183631311 | 0.000513422 |
| TGFBR3 | 0.196522244 | 0.000405979 |
| GJA1 | 0.206907347 | 0.000107676 |
| LAMA2 | 0.219665855 | 0.000794627 |
| KIT | 0.430444215 | 0.00255635 |
| TPM1 | 0.433963109 | 0.001632742 |
| VCP | 0.435612412 | 0.007402292 |
| GRID2 | 0.444288529 | 0.008594981 |
| MEF2C | 0.453593212 | 0.00396401 |
| RPS3A | 0.462721116 | 0.002620209 |
| PLD1 | 0.468341554 | 0.011044722 |
| BTG3 | 0.485731167 | 0.040345869 |
| CD47 | 0.486714062 | 0.009288999 |
| MITF | 0.491659656 | 0.006329736 |
| Steps | Solution | Duration |
|---|---|---|
| Deparaffinization and rehydration | Xylene I | 10 min |
| Xylene II | 5 min | |
| Absolute alcohol | 5 min | |
| 90% alcohol | 5 min | |
| 70% alcohol | 5 min | |
| Water wash | 10 min | |
| Staining | Hematoxylin | 20 min |
| Water wash | 8 min | |
| Eosin | 20 min | |
| Water wash | 8 min | |
| Dehydration | 70% alcohol | 5 min |
| 90% alcohol | 5 min | |
| Absolute alcohol | 5 min | |
| Xylene I | 5 min | |
| Xylene II | 10 min |
| Gene | Gene Accession Number | Primer Sequence (5′-3′) | Product Size (bp) |
|---|---|---|---|
| ID2 | NM_001037965.1 | CCAGTGAGGTCCGTTAGGAA | 243 |
| GACAATAGTGGGGTGCGAGT | |||
| VEGFA | NM_214084.1 | CTACCTCCACCATGCCAAGT | 232 |
| ACACTCCAGACCTTCGTCGT | |||
| BTG2 | NM_001097505.2 | TGGTTTCCTGAAAAGCCATC | 150 |
| GGACACTTCATAGGGGTCCA | |||
| ESR1 | NM_214220.1 | AGCACCCTGAAGTCTCTGGA | 160 |
| TGTGCCTGAAGTGAGACAGG | |||
| CCND2 | NM_214088.1 | CGTCCAAGCTCAAAGAGACC | 169 |
| CGAAGAATGTGCTCGATGAA | |||
| EDNRA | NM_214229.1 | CACCACATTTCGTGGAACAG | 205 |
| AATGATCCTGAGCAGGGTTG | |||
| ANGPTL4 | NM_214229.1 | TGCCAAGAGCTGTTTGAAGA | 152 |
| TTAAAGTCCACCGAGCCATC | |||
| TGFBR3 | NM_214272.1 | TCATCTCCCCGTACTCGAAC | 219 |
| TCTTGGTACACAGCGTGAGC | |||
| GJA1 | NM_001244212.1 | CACCAGGTGGACTGTTTCCT | 151 |
| TCTTTCCCTTCACACGATCC | |||
| LAMA2 | XM_021084463.1 | CTCCAGGCTATTCTGGCTTG | 153 |
| AGGTTTCAGGGTCACACAGG | |||
| KIT | NM_001044525.1 | TCAGGGAGAACAGCCAGACT | 170 |
| GGTGGTTGTGACATTTGCAG | |||
| TPM1 | NM_001097483.2 | CTCTGAACAGACGCATCCAA | 243 |
| TACGGGCCACCTCTTCATAC | |||
| VCP | NM_214280.1 | ACCCTCCAAGGGAGTGCTAT | 223 |
| GGCAATTGAATCCAGCTCAT | |||
| GRID2 | XM_021100736.1 | GTCCCATCGAAAGAGGATGA | 176 |
| TCACTGATTTGCTCCAGTGC | |||
| MEF2C | NM_001044540.1 | GCCCTGAGTCTGAGGACAAG | 163 |
| AGTGAGCTGACAGGGTTGCT | |||
| RPS3A | NM_001137619.1 | CCGGAAGAAGATGATGGAAA | 179 |
| CAAACTTGGGCTTCTTCAGC | |||
| PLD1 | NM_001244589.1 | TTCTGGACCCAGTGAAGACC | 229 |
| GAACCCACGGATCTTTTTCA | |||
| BTG3 | NM_001097517.1 | TACAGATGCATTCGGGTCAA | 153 |
| CTCTCCATACCGACAGCACA | |||
| CD47 | NM_213982.1 | TGGAGCCATTCTTTTCATCC | 191 |
| GTCCAACCACAGAGAGCACA | |||
| MITF | NM_001038001.1 | GCCAATCGGCATTTGTTACT | 236 |
| CCGAGGTTGTTGTTGAAGGT |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chermuła, B.; Brązert, M.; Jeseta, M.; Ożegowska, K.; Sujka-Kordowska, P.; Konwerska, A.; Bryja, A.; Kranc, W.; Jankowski, M.; Nawrocki, M.J.; et al. The Unique Mechanisms of Cellular Proliferation, Migration and Apoptosis are Regulated through Oocyte Maturational Development—A Complete Transcriptomic and Histochemical Study. Int. J. Mol. Sci. 2019, 20, 84. https://doi.org/10.3390/ijms20010084
Chermuła B, Brązert M, Jeseta M, Ożegowska K, Sujka-Kordowska P, Konwerska A, Bryja A, Kranc W, Jankowski M, Nawrocki MJ, et al. The Unique Mechanisms of Cellular Proliferation, Migration and Apoptosis are Regulated through Oocyte Maturational Development—A Complete Transcriptomic and Histochemical Study. International Journal of Molecular Sciences. 2019; 20(1):84. https://doi.org/10.3390/ijms20010084
Chicago/Turabian StyleChermuła, Błażej, Maciej Brązert, Michal Jeseta, Katarzyna Ożegowska, Patrycja Sujka-Kordowska, Aneta Konwerska, Artur Bryja, Wiesława Kranc, Maurycy Jankowski, Mariusz J. Nawrocki, and et al. 2019. "The Unique Mechanisms of Cellular Proliferation, Migration and Apoptosis are Regulated through Oocyte Maturational Development—A Complete Transcriptomic and Histochemical Study" International Journal of Molecular Sciences 20, no. 1: 84. https://doi.org/10.3390/ijms20010084
APA StyleChermuła, B., Brązert, M., Jeseta, M., Ożegowska, K., Sujka-Kordowska, P., Konwerska, A., Bryja, A., Kranc, W., Jankowski, M., Nawrocki, M. J., Kocherova, I., Celichowski, P., Borowiec, B., Popis, M., Budna-Tukan, J., Antosik, P., Bukowska, D., Brussow, K. P., Pawelczyk, L., ... Kempisty, B. (2019). The Unique Mechanisms of Cellular Proliferation, Migration and Apoptosis are Regulated through Oocyte Maturational Development—A Complete Transcriptomic and Histochemical Study. International Journal of Molecular Sciences, 20(1), 84. https://doi.org/10.3390/ijms20010084








