Electrochemical Detection of Dopamine at a Gold Electrode Modified with a Polypyrrole–Mesoporous Silica Molecular Sieves (MCM-48) Film
Abstract
1. Introduction
2. Results and Discussion
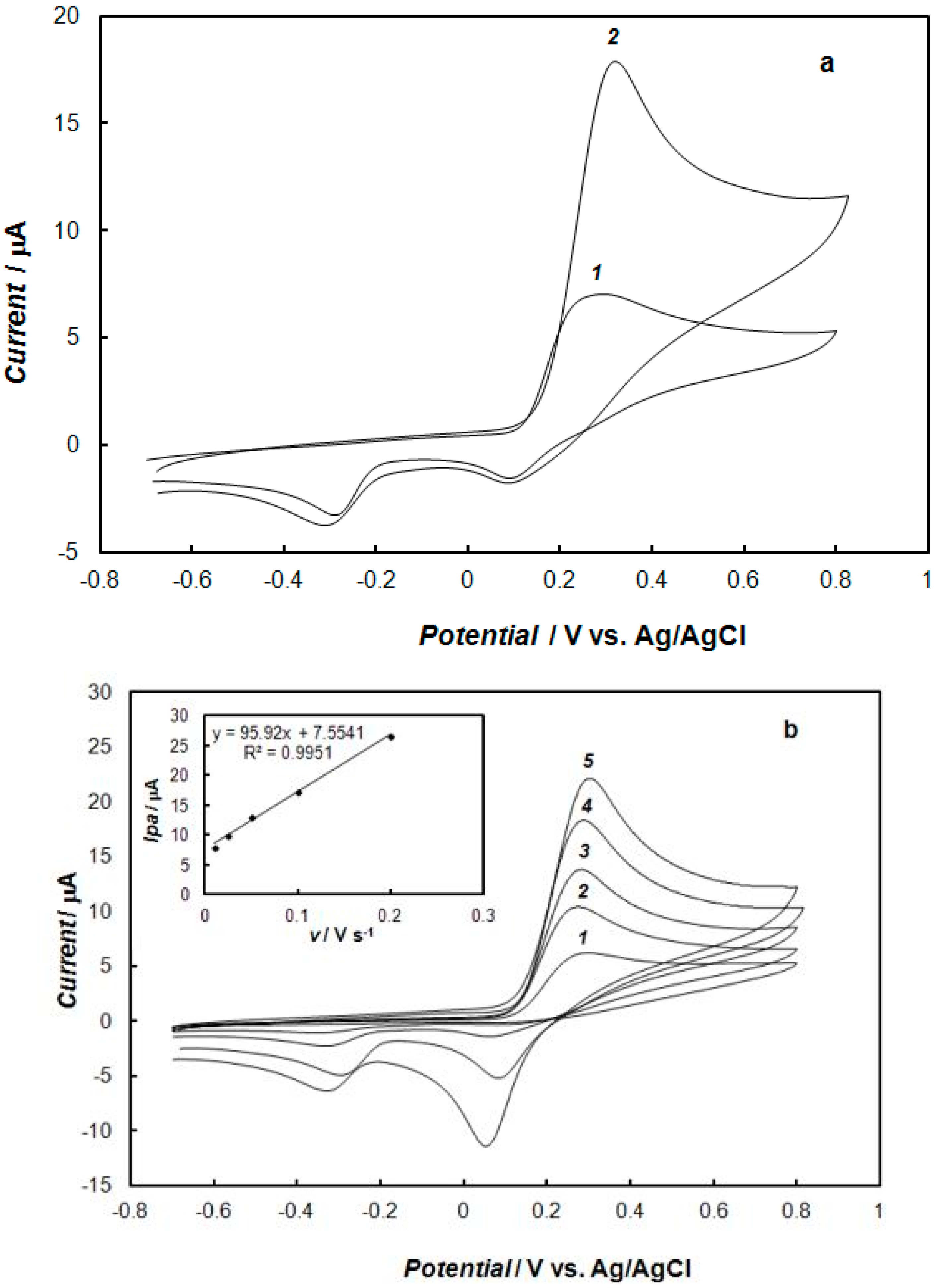
2.1. Formation and Electrochemical Properties of the Modified Electrodes
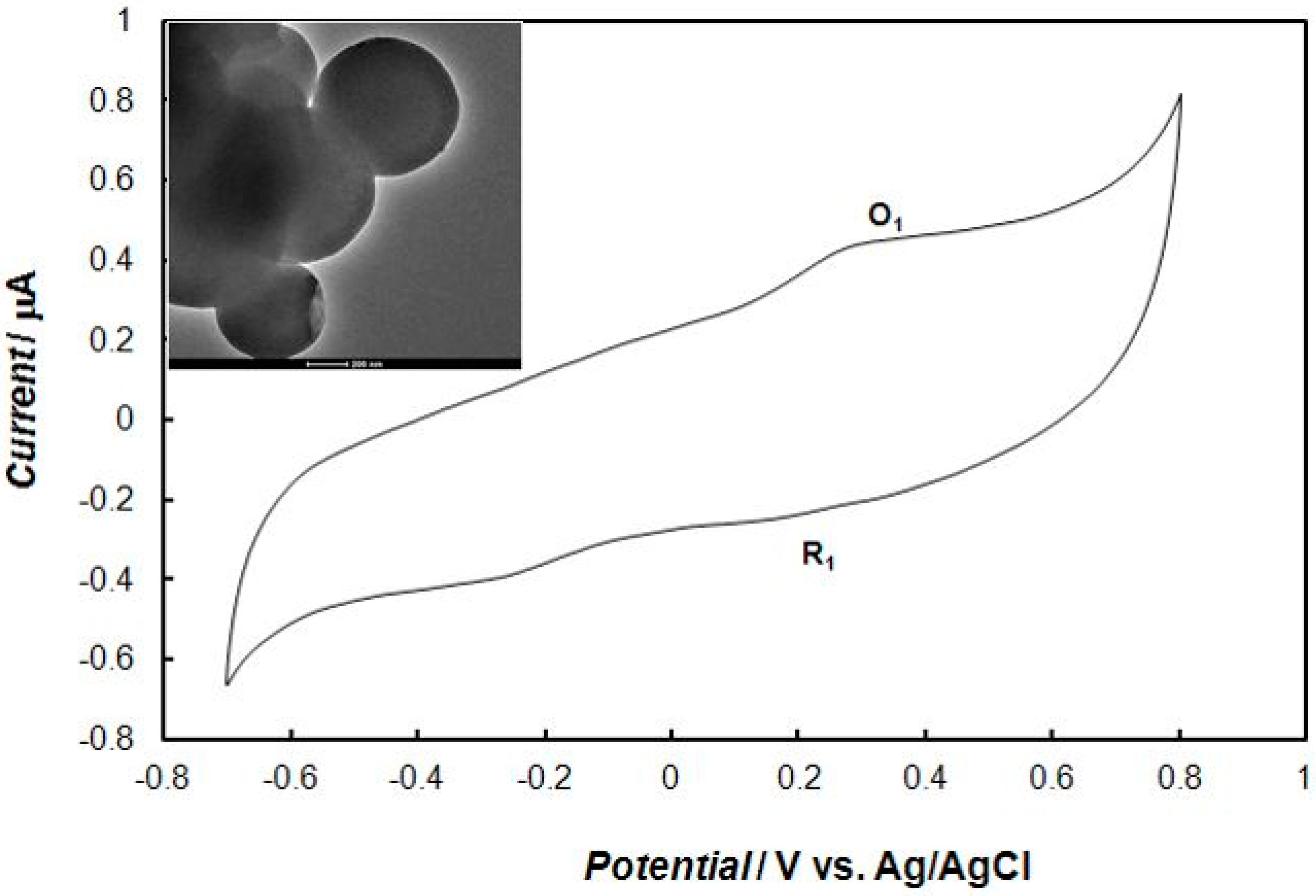
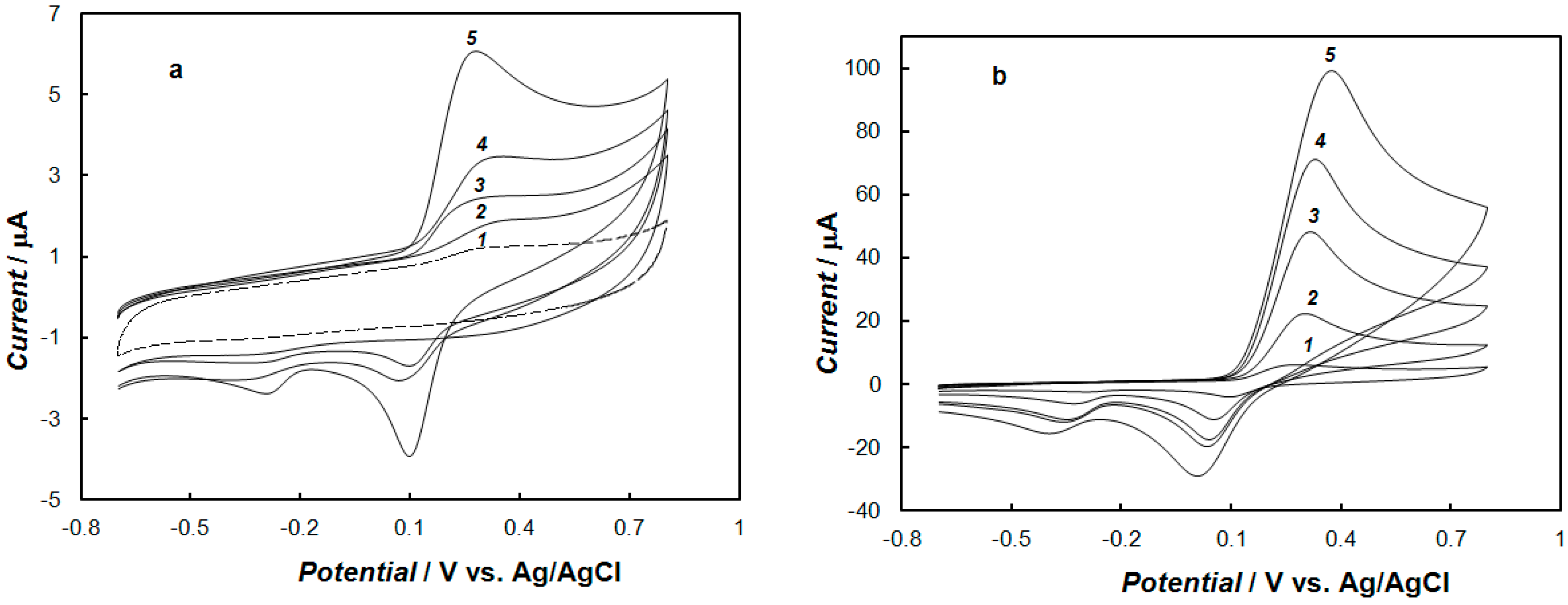
2.2. Voltammetric Behaviors of Dopamine at the Bare Gold Electrode
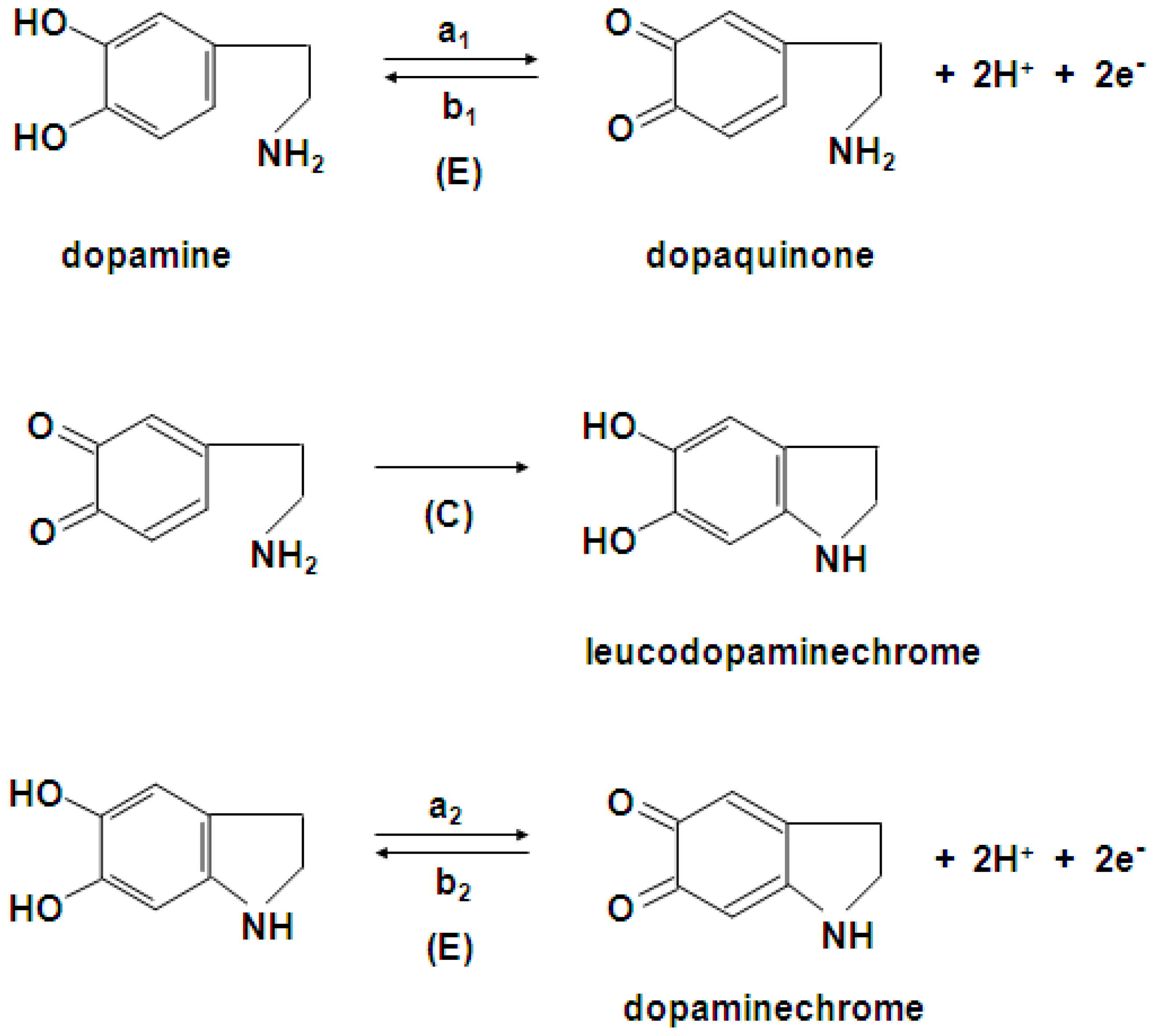
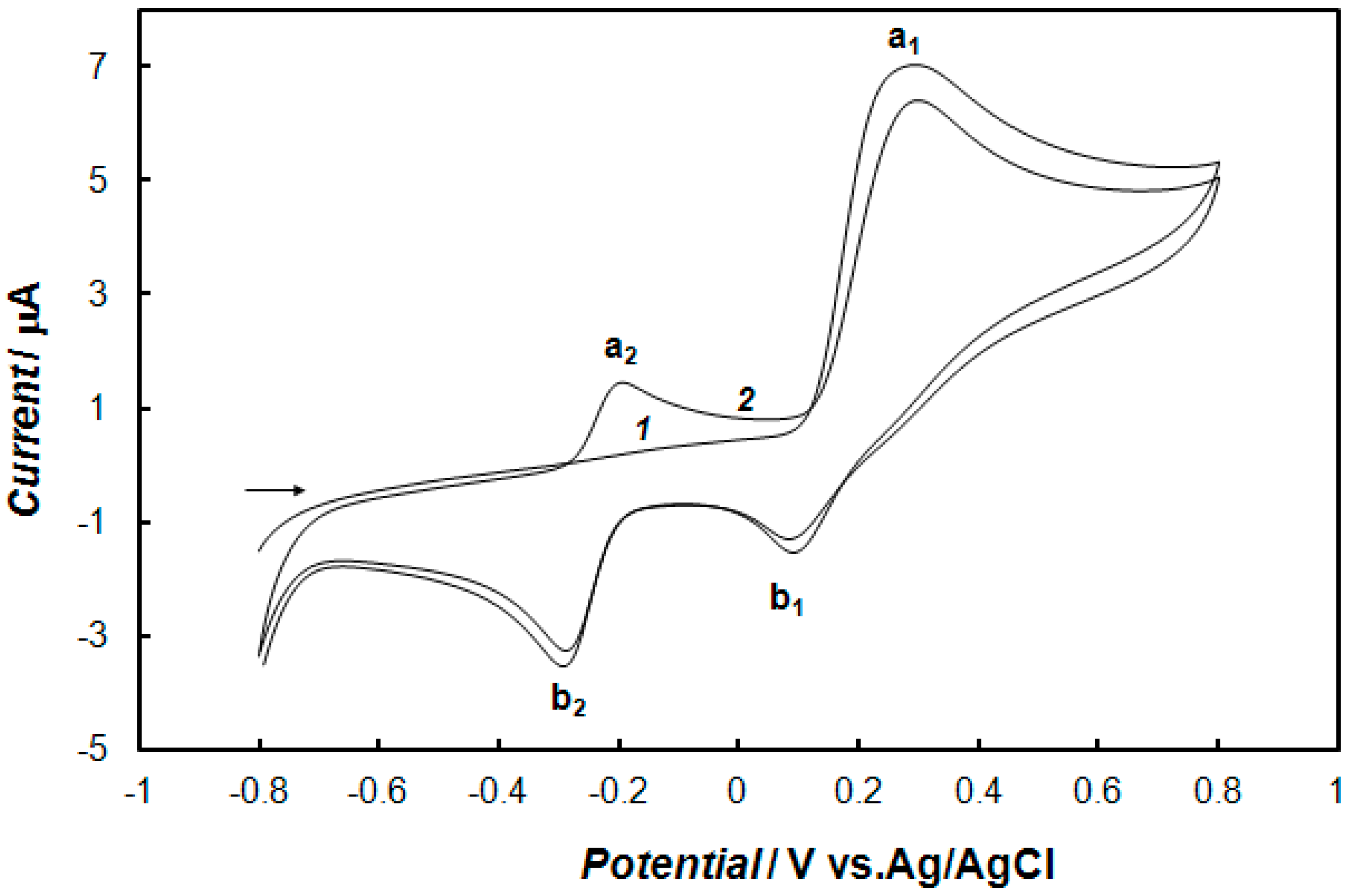
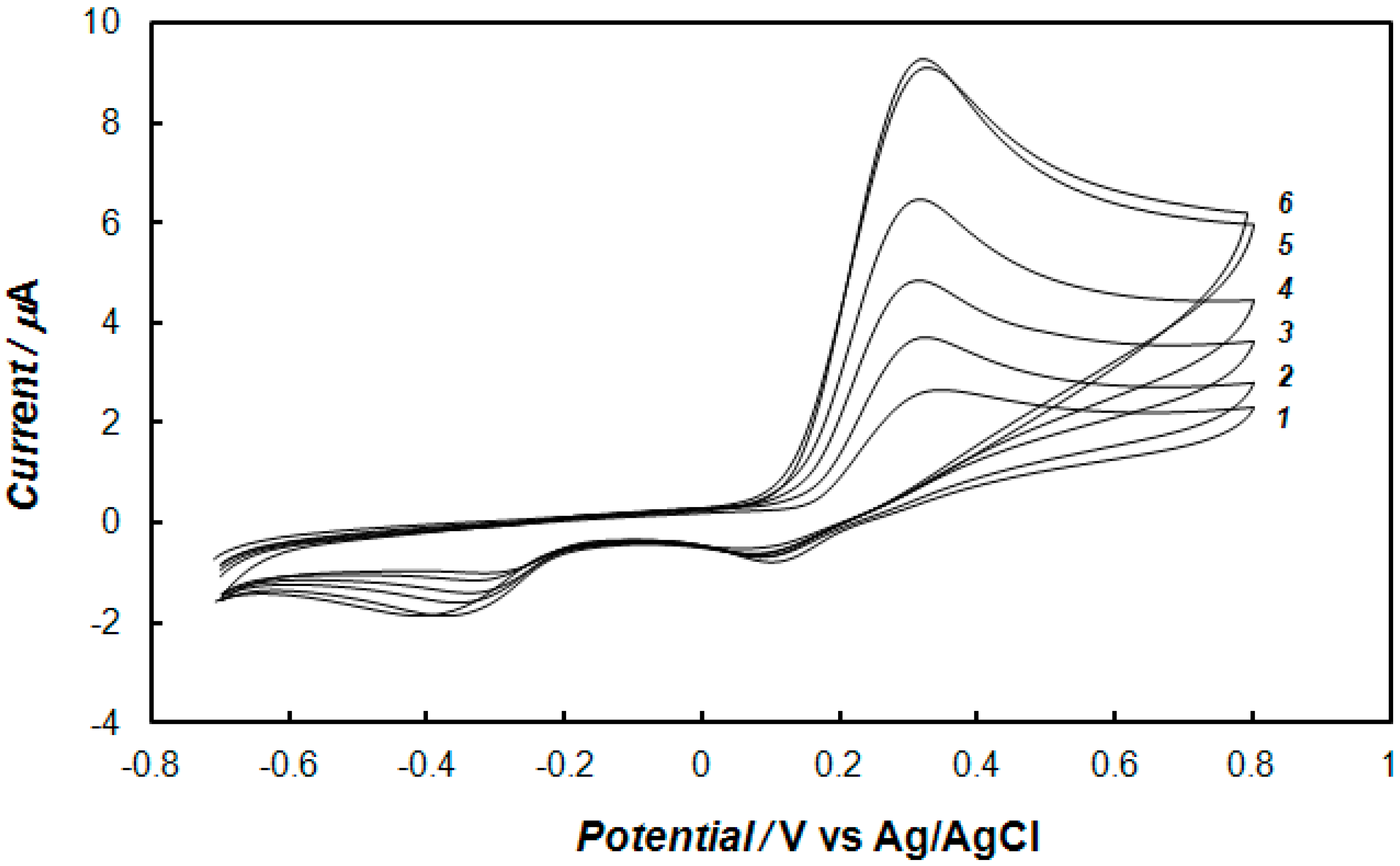
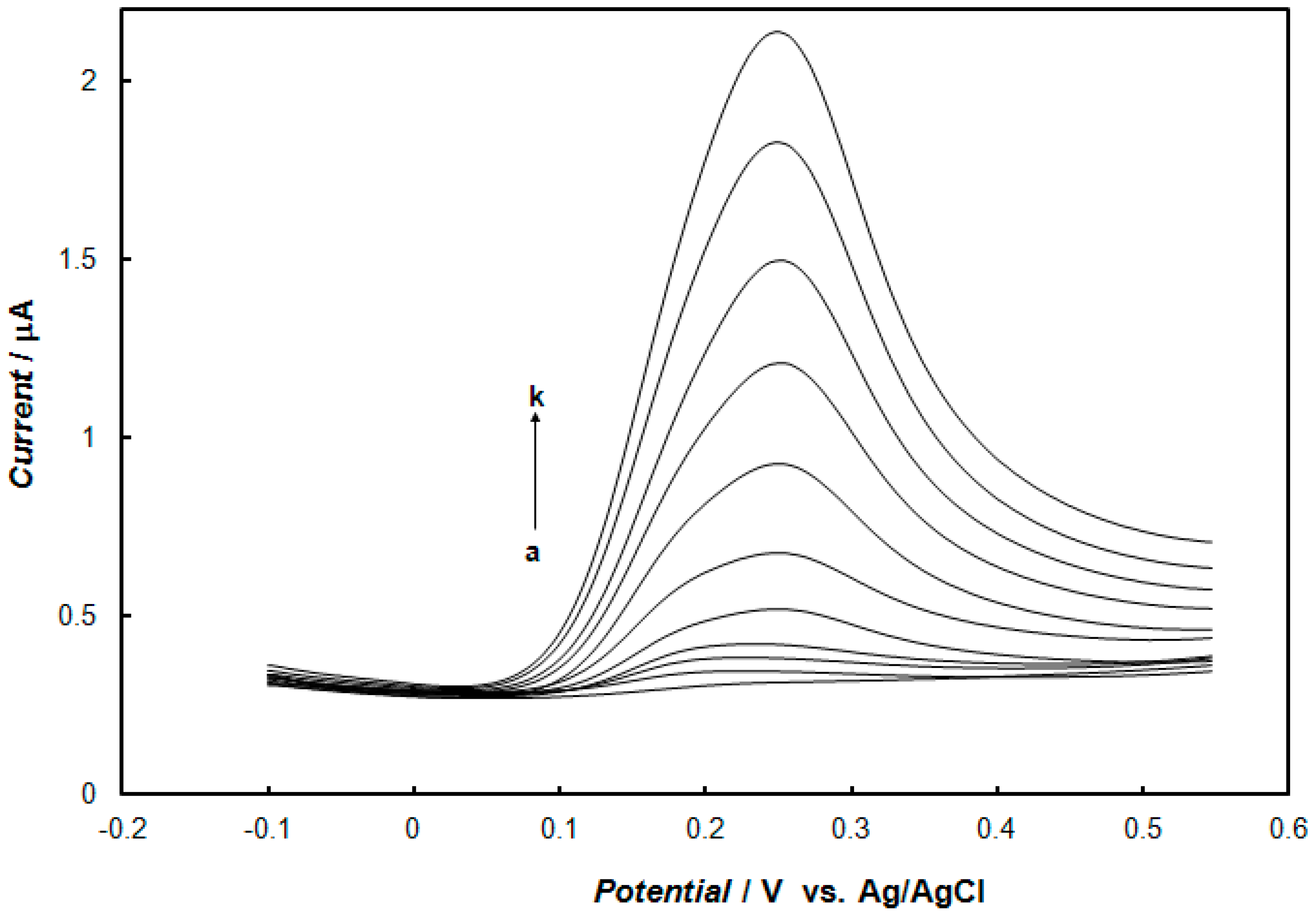
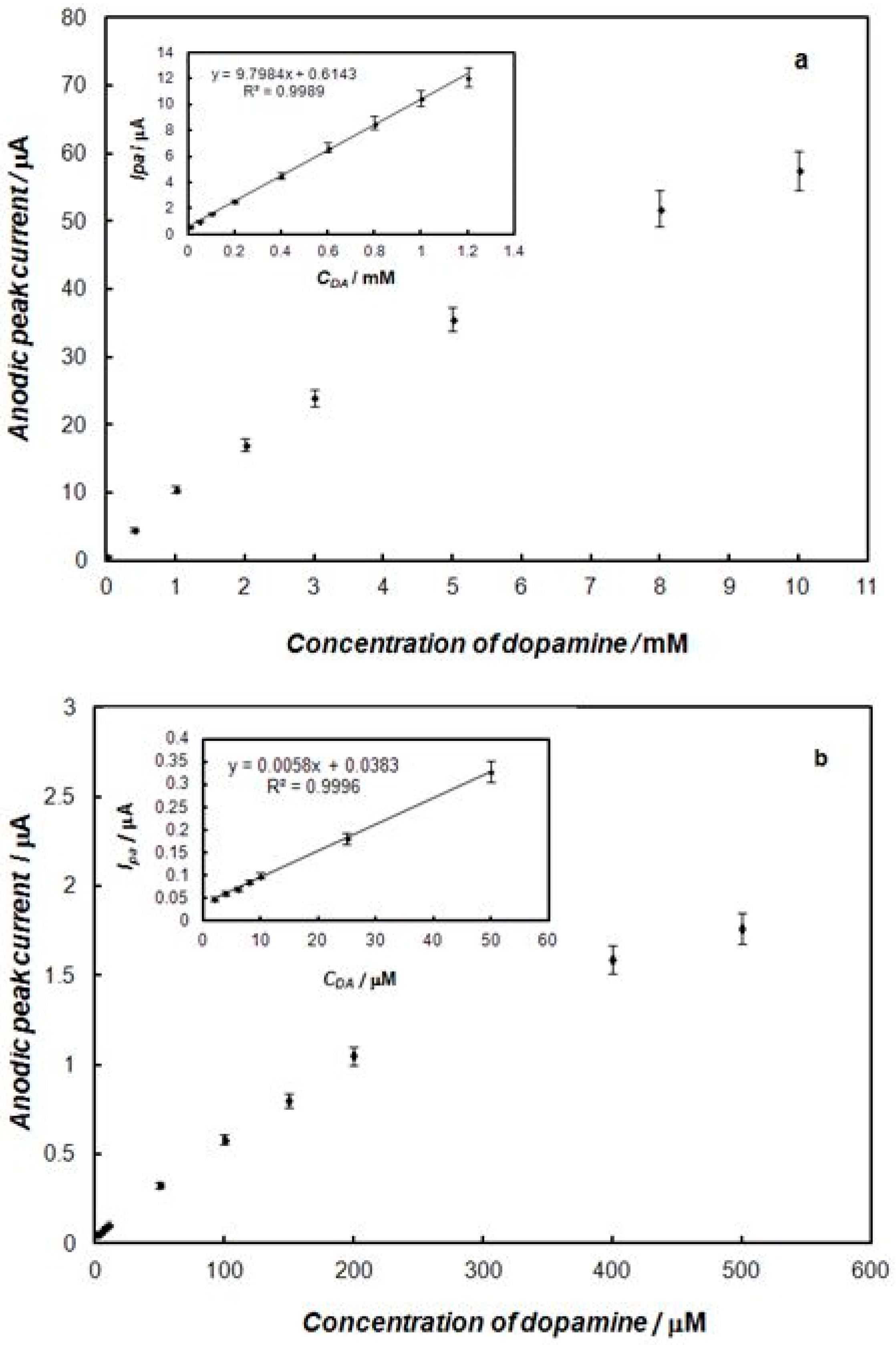
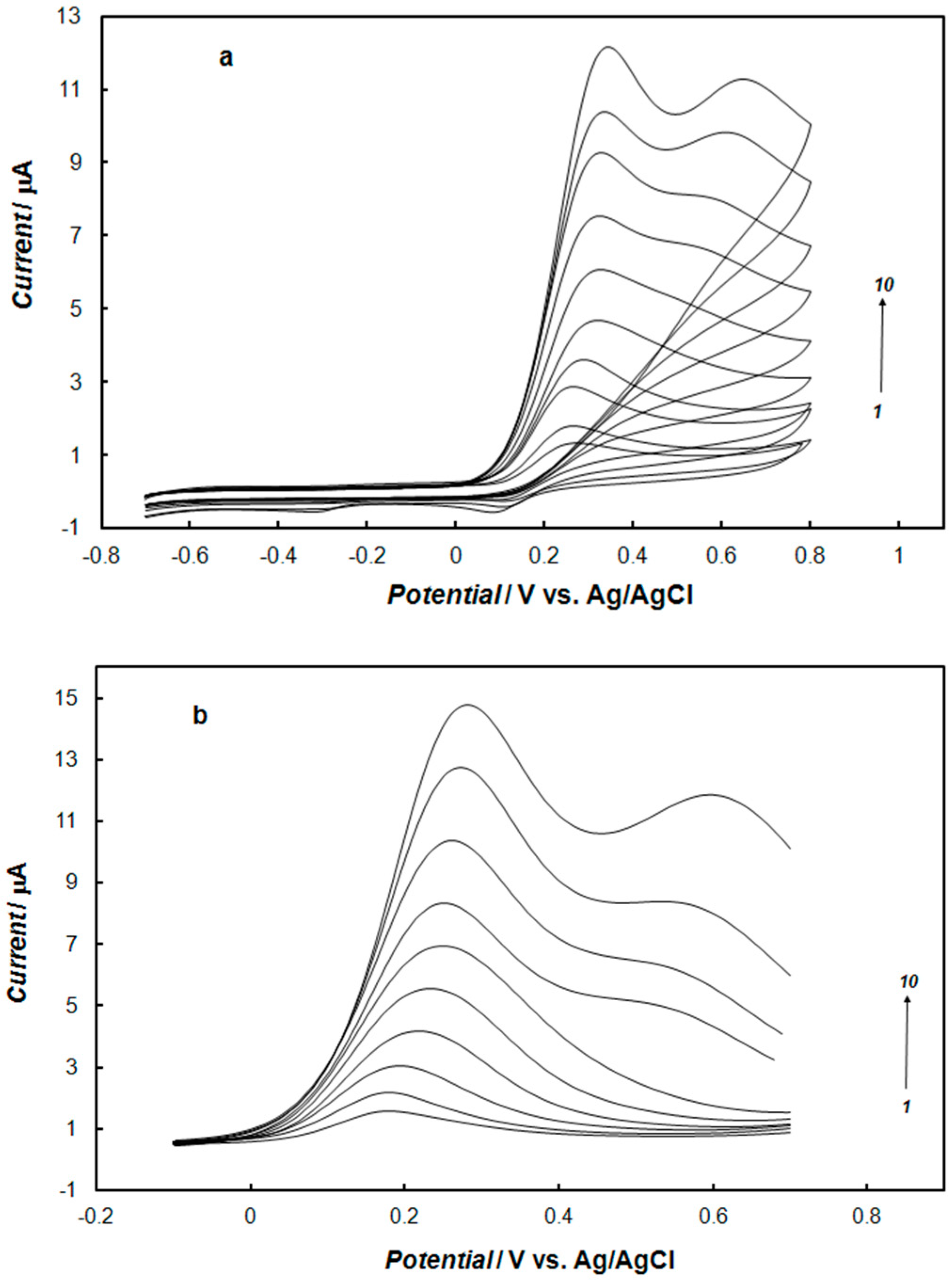
2.3. Electrochemical Detection of Dopamine at the Gold Electrode Modified with the Polypyrrole—MCM-48 Nanocomposite
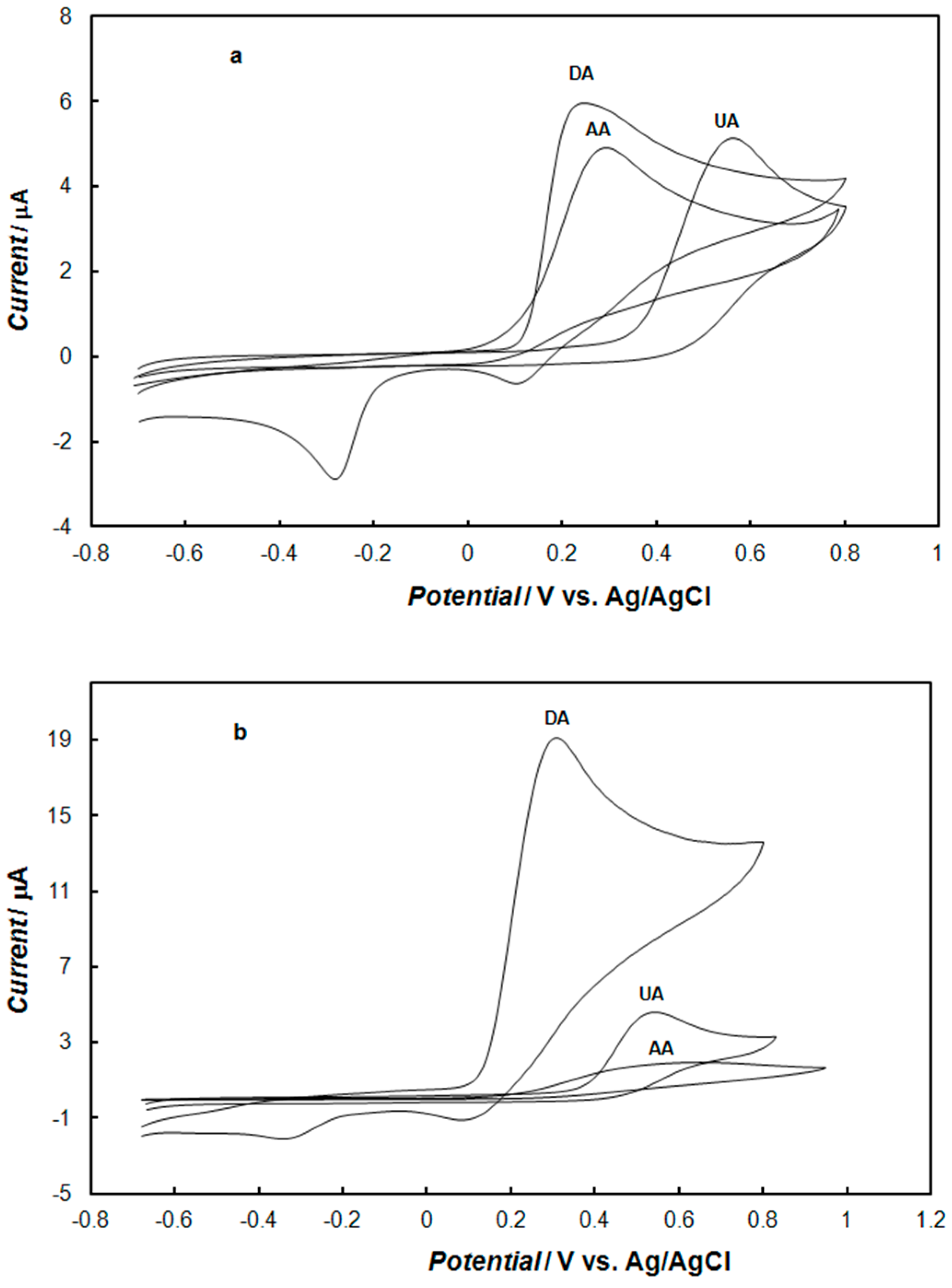
2.4. Interference by Ascorbic Acid and Uric Acid
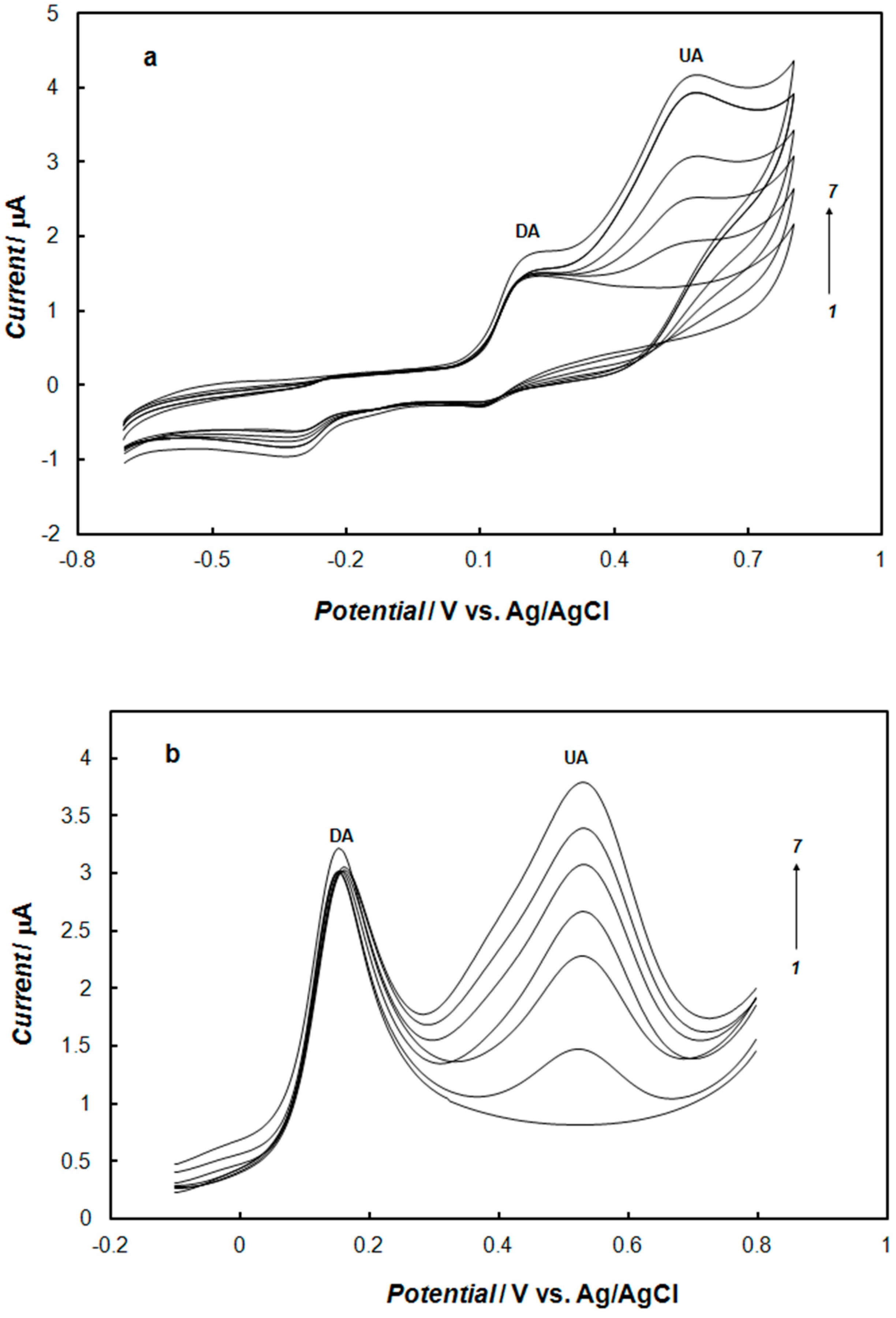
2.5. The Influence of Ascorbic Acid or Uric Acid on the Determination of Dopamine
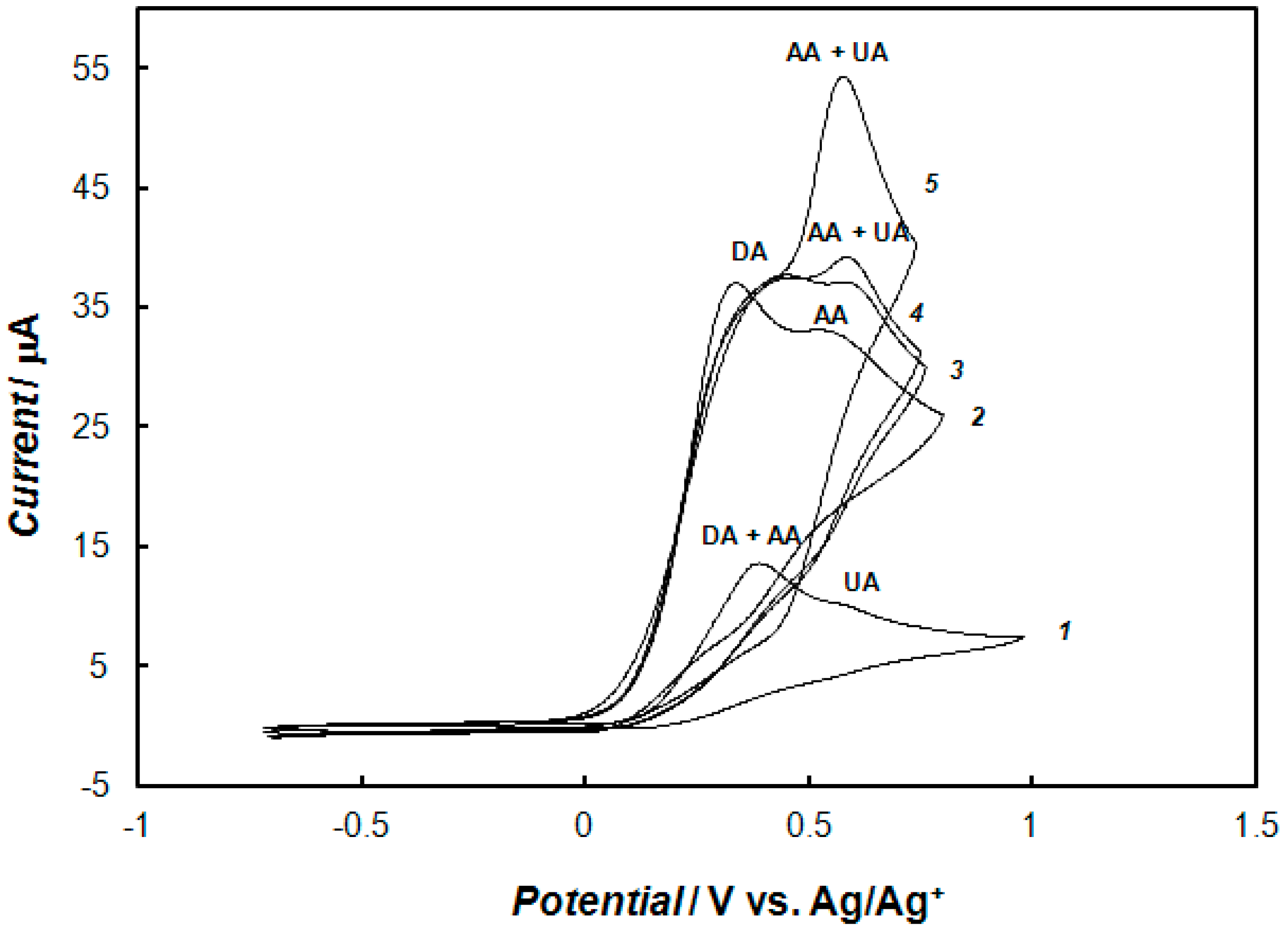
2.6. Simultaneous Detection of Dopamine, Ascorbic Acid, and Uric Acid
3. Conclusions
4. Experimental Section
4.1. Materials
4.2. Instrumentation
4.3. Preparation of the Polypyrrole—MCM-48 Nanocomposite
Author Contributions
Funding
Conflicts of Interest
References
- Sarkar, C.; Basu, B.; Chakroborty, D.; Dasgupta, P.S.; Basu, S. The immunoregulatory role of dopamine: An update. Brain Behav. Immun. 2010, 24, 525–528. [Google Scholar] [CrossRef]
- Stocchi, F. The therapeutic concept of continuous dopaminergic stimulation (CDS) in the treatment of Parkinson’s disease. Parkinsonism Relat. Disord. 2009, 15, 68–71. [Google Scholar] [CrossRef]
- Adams, R.N. Probing Brain chemistry with Electrosnslytical. Anal. Chem. 1976, 48, 1130A–1138A. [Google Scholar] [CrossRef]
- Raj, C.R.; Tokuda, K.; Ohsaka, T. Electroanalytical applications of cationic self-assembled monolayers: Square-wave voltammetric determination of dopamine and ascorbate. Bioelectrochemistry 2001, 53, 183–191. [Google Scholar] [CrossRef]
- Fabregat, G.; Cordova-Mattteo, E.; Armelin, E.; Bertran, O.; Aleman, C. Ultrathin films of polypyrrole derivatives for dopamine detection. J. Phys. Chem. 2011, 115, 14933–14941. [Google Scholar] [CrossRef]
- Jiang, L.; Xie, Q.; Li, Z.; Li, Y.; Yao, S. A study on tannic acid-doped polypyrrole films on gold electrodes for selective electrochemical detection of dopamine. Sensor 2005, 5, 199–208. [Google Scholar] [CrossRef]
- Roy, P.R.; Okajima, T.; Ohsaka, T. Simultaneous electroanalysis of dopamine and ascorbic acid using poly (N,N-dimethylaniline)-modified electrodes. Bioelectrochemistry 2003, 59, 11–19. [Google Scholar] [CrossRef]
- Jin, G.; Zhang, Y.; Cheng, W. Poly(p-aminobenzene sulfonic acid)-modified glassy carbon electrode for simultaneous detection of dopamine and ascorbic acid. Sens. Actuators B 2005, 107, 528–534. [Google Scholar] [CrossRef]
- Ates, M.; Castillo, J.; Sarac, A.S.; Schuhmann, W. Carbon fiber microelectrodes electrocoated with polycarbazole and poly(carbazole-co-p-tolylsulfonyl pyrrole) films for the detection of dopamine in presence of ascorbic acid. Microchim. Acta 2008, 160, 247–251. [Google Scholar] [CrossRef]
- Fabre, B.; Taillebois, L. Poly(aniline boronic acid)-based conductimetric sensor of dopamine. Chem. Commun. 2003, 2982–2983. [Google Scholar] [CrossRef]
- Lin, X.; Zhang, Y.; Chen, W.; Wu, P. Electrocatalytic oxidation and determination of dopamine in the presence of ascorbic acid and uric acid at a poly(p-nitrobenzenazoresorcinol) modified glassy carbon electrode. Sens. Actuators B 2007, 122, 309–314. [Google Scholar] [CrossRef]
- Kalimuthu, P.; John, S.A. Electropolymerized film of functionalized thiadiazole on glassy carbon electrode for the simultaneous determination of ascorbic acid, dopamine and uric acid. Bioelectrochemistry 2009, 77, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Oztekin, Y.; Tok, M.; Bilici, E.; Mikoliunaite, L.; Yazicigil, Z.; Ramanaviciena, A.; Ramanavicius, A. Copper nanoparticle modified carbon electrode for determination of dopamine. Electrochim. Acta 2012, 76, 201–207. [Google Scholar] [CrossRef]
- Tashkhourian, J.; Nezhad, M.R.H.; Khodavesi, J.; Javadi, S. Silver nanoparticles modified carbon nanotube paste electrode for simultaneous determination of dopamine and ascorbic acid. J. Electroanal. Chem. 2009, 633, 85–91. [Google Scholar] [CrossRef]
- Li, J.; Yang, J.; Yang, Z.; Li, Y.; Yu, S.; Xu, Q.; Hu, X. Graphene–Au nanoparticles nanocomposite film for selective electrochemical determination of dopamine. Anal. Methods 2012, 4, 1725–1728. [Google Scholar] [CrossRef]
- Huang, J.; Liu, Y.; Hou, H.; You, T. Simultaneous electrochemical determination of dopamine, uric acid and ascorbic acid using palladium nanoparticle-loaded carbon nanofibers modified electrode. Biosens. Bioelectron. 2008, 24, 632–637. [Google Scholar] [CrossRef] [PubMed]
- Yau, J.; Liu, S.; Zhang, Z.; He, G.; Zhou, P.; Liang, H.; Tian, L.; Zhou, X.; Jiang, H. Simultaneous electrochemical detection of ascorbic acid, dopamine and uric acid based on graphene anchored with Pd–Pt nanoparticles. Colloids Surf. B Biointerfaces 2013, 111, 392–397. [Google Scholar]
- Li, L.; Liu, H.; Shen, Y.; Zhang, J.; Zhu, J.J. Electrogenerated chemiluminescence of Au nanoclusters for the detection of dopamine. Anal. Chem. 2011, 83, 661–665. [Google Scholar] [CrossRef]
- Goyal, R.N.; Gupta, V.K.; Bachhetin, N.; Sharma, R.A. Electrochemical sensor for the determination of dopamine in presence of high concentration of ascorbic acid using a fullerene-C60 coated gold electrode. Electroanalysis 2008, 20, 757–764. [Google Scholar] [CrossRef]
- Wang, Z.; Liu, J.; Liang, Q.; Wang, Y.; Luo, G. Carbon nanotube-modified electrodes for the simultaneous determination of dopamine and ascorbic acid. Analyst 2002, 127, 653–658. [Google Scholar] [CrossRef]
- Wang, Y.; Li, Y.; Tang, L.; Lu, J.; Li, J. Application of graphene-modified electrode for selective detection of dopamine. Electrochem. Commun. 2009, 11, 889–892. [Google Scholar] [CrossRef]
- Kim, Y.R.; Bong, S.; Kang, Y.J.; Yang, Y.; Mahajan, R.K.; Kim, J.S.; Kim, H. Electrochemical detection of dopamine in the presence of ascorbic acid using graphene modified electrodes. Biosens. Bioelectron. 2010, 25, 2366–2369. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Gao, Y.; Zhan, D.; Liu, H.; Zhao, Q.; Kou, Y.; Shao, Y.; Li, M.; Zhuang, Q.; Zhu, Z. Selective detection of dopamine in the presence of ascorbic acid and uric acid by a carbon nanotubes-ionic liquid gel modified electrode. Talanta 2005, 66, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Liang, Q.; Wang, Y.; Luo, G. Carbon nanotube-intercalated graphite electrodes for simultaneous determination of dopamine and serotonin in the presence of ascorbic acid. J. Electroanal. Chem. 2003, 540, 129–134. [Google Scholar] [CrossRef]
- Reddy, S.; Swamy, B.E.K.; Jayadevappa, H. CuO nanoparticle sensor for the electrochemical determination of dopamine. Electrochim. Acta 2012, 61, 78–86. [Google Scholar] [CrossRef]
- Reddy, S.; Swamy, B.E.K.; Chandra, U.; Sherigara, B.S.; Jayadevappa, H. Synthesis of CdO nanoparticles and their modified carbon paste electrode for determination of dopamine and ascorbic acid by using cyclic voltammetry. Int. J. Electrochem. Sci. 2010, 5, 10–17. [Google Scholar]
- Li, Q.; Wang, Y.; Luo, G. Voltammetric separation of dopamine and ascorbic acid with graphite electrodes modified with ultrafine TiO2. Mater. Sci. Eng C 2000, 11, 71–74. [Google Scholar] [CrossRef]
- Elhag, S.; Ibupoto, Z.H.; Liu, X.; Nur, O.; Willander, M. Dopamine wide range detection sensor based on modified Co3O4 nanowires electrode. Sens. Actuators B 2014, 203, 543–549. [Google Scholar] [CrossRef]
- Yang, L.; Liu, S.; Zhang, Q.; Li, F. Simultaneous electrochemical determination of dopamine and ascorbic acid using AuNPs@ polyaniline core–shell nanocomposites modified electrode. Talanta 2012, 89, 136–141. [Google Scholar] [CrossRef]
- Manivel, P.; Dhakshnamoorthy, M.; Balamurugan, A.; Ponpandian, N.; Mangalaraj, D.; Viswanathan, C. Conducting polyaniline-graphene oxide fibrous nanocomposites: Preparation, characterization and simultaneous electrochemical detection of ascorbic acid, dopamine and uric acid. RSC Adv. 2013, 3, 14428–14437. [Google Scholar] [CrossRef]
- Zhuang, Z.; Li, J.; Xu, R.; Xiao, D. Electrochemical detection of dopamine in the presence of ascorbic acid using overoxidized polypyrrole/graphene modified electrodes. Int. J. Electrochem. Sci. 2011, 6, 2149–2161. [Google Scholar]
- Qian, T.; Wu, S.; Shen, J. Facilely prepared polypyrrole-reduced graphite oxide core–shell microspheres with high dispersibility for electrochemical detection of dopamine. Chem. Commun. 2013, 49, 4610–4612. [Google Scholar] [CrossRef] [PubMed]
- Min, K.; Yoo, Y.J. Amperometric detection of dopamine based on tyrosinase–SWNTs–Ppy composite electrode. Talanta 2009, 80, 1007–1011. [Google Scholar] [CrossRef] [PubMed]
- Ulubay, S.; Dursun, Z. Cu nanoparticles incorporated polypyrrole modified GCE for sensitive simultaneous determination of dopamine and uric acid. Talanta 2010, 80, 1461–1466. [Google Scholar] [CrossRef] [PubMed]
- Saha, S.; Sarkar, P.; Turner, A.P.F. Interference-free electrochemical detection of nanomolar dopamine using doped polypyrrole and silver nanoparticles. Electroanalysis 2014, 26, 2197–2206. [Google Scholar] [CrossRef]
- Li, J.; Lin, X. Simultaneous determination of dopamine and serotonin on gold nanocluster/overoxidized-polypyrrole composite modified glassy carbon electrode. Sens. Actuators B 2007, 124, 486–493. [Google Scholar] [CrossRef]
- Qian, T.; Yu, C.; Wu, S.; Shen, J. In situ polymerization of highly dispersed polypyrrole on reduced graphite oxide for dopamine detection. Biosens. Bioelectron. 2013, 50, 157–160. [Google Scholar] [CrossRef]
- Ramanavičius, A.; Ramanavičienė, A.; Malinauskas, A. Electrochemical sensors based on conducting polymer-polypyrrole. Electrochim. Acta 2006, 51, 6025–6037. [Google Scholar] [CrossRef]
- Duchet, J.; Legras, R.; Demoustier-Champagne, S. Chemical synthesis of polypyrrole: Structure–properties relationship. Synth. Met. 1998, 98, 113–122. [Google Scholar] [CrossRef]
- Liu, J.; Wan, M. Synthesis, characterization and electrical properties of microtubules of polypyrrole synthesized by a template-free method. J. Mater. Chem. 2001, 11, 404–407. [Google Scholar] [CrossRef]
- Pringle, J.M.; Efthimiadis, J.; Howlett, P.C.; Efthimiadis, J.; MacFarlane, D.R.; Chaplin, A.B.; Hall, S.B.; Officer, D.L.; Wallace, G.G.; Forsyth, M. Electrochemical synthesis of polypyrrole in ionic liquids. Polymer 2004, 45, 1447–1453. [Google Scholar] [CrossRef]
- Gong, J.; Wang, L.; Zhang, L. Electrochemical biosensing of methyl parathion pesticide based on acetylcholinesterase immobilized onto Au–polypyrrole interlaced network-like nanocomposite. Biosens. Bioelectron. 2009, 24, 2285–2288. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Kong, L.B.; Cai, J.J.; Luo, Y.C.; Kang, L. Nano-composite of polypyrrole/modified mesoporous carbon for electrochemical capacitor application. Electrochim. Acta 2010, 55, 8067–8073. [Google Scholar] [CrossRef]
- Qu, L.; Shi, G.; Chen, F.; Zhang, J. Electrochemical growth of polypyrrole microcontainers. Macromolecules 2003, 36, 1063–1067. [Google Scholar] [CrossRef]
- Qu, L.; Shi, G. Hollow microstructures of polypyrrole doped by poly(styrene sulfonic acid). J. Poly. Sci. A Polym. Chem. 2004, 42, 3170–3177. [Google Scholar] [CrossRef]
- Schumacher, K.; Grun, M.; Unger, K.K. Novel synthesis of spherical MCM-48. Microporous Mesoporou Mater. 1999, 27, 201–206. [Google Scholar] [CrossRef]
- Wysocka-Zolopa, M.; Zablocka, I.; Basa, A.; Winkler, K. Formation and characterization of mesoporous silica MCM-48 and polypyrrole composite. Chem. Heterocycl. Compd. 2017, 53, 78–86. [Google Scholar] [CrossRef]
- Kim, J.M.; Kim, S.K.; Ryoo, R. Synthesis of MCM-48 single crystals. Chem. Commun. 1998, 2, 259–260. [Google Scholar] [CrossRef]
- He, H.; Xie, Q.; Yao, S. An electrochemical quartz crystal impedance study on anti-human immunoglobulin G immobilization in the polymer grown during dopamine oxidation at an Au electrode. J. Colloid Interface Sci. 2005, 289, 446–454. [Google Scholar] [CrossRef] [PubMed]
- Majewska, U.E.; Chmurski, K.; Biesiada, K.; Olszyna, A.R.; Bilewicz, R. Dopamine oxidation at per (6-deoxy-6-thio)-α-cyclodextrin monolayer modified gold electrodes. Electroanalysis 2006, 18, 1463–1470. [Google Scholar] [CrossRef]
- Hegde, R.N.; Swamy, B.E.S.; Shetti, N.P.; Nandibewoor, S.T. Electro-oxidation and determination of gabapentin at gold electrode. J. Electroanal. Chem. 2009, 635, 51–57. [Google Scholar] [CrossRef]
- Raoof, J.B.; Ojani, R.; Rashid-Nadimi, S. Voltammetric determination of ascorbic acid and dopamine in the same sample at the surface of a carbon paste electrode modified with polypyrrole/ferrocyanide films. Electrochim. Acta 2005, 50, 4694–4698. [Google Scholar] [CrossRef]
- Feng, X.; Huang, H.; Ye, Q.; Zhu, J.J.; Hou, W. Ag/polypyrrole core–shell nanostructures: Interface polymerization, characterization, and modification by gold nanoparticles. J. Phys. Chem. C 2007, 111, 8463–8468. [Google Scholar] [CrossRef]
- Izaoumen, N.; Bouchta, D.; Zejli, H.; El Kaoutit, M.; Temsamani, K.R. The Electrochemical behavior of neurotransmitters at a poly(pyrrole-β-cyclodextrin) modified glassy carbon electrode. Anal. Lett. 2005, 38, 1869–1885. [Google Scholar] [CrossRef]
- Ghanbari, K.; Hajheidari, N. ZnO–CuxO/polypyrrole nanocomposite modified electrode for simultaneous determination of ascorbic acid, dopamine, and uric acid. Anal. Biochem. 2015, 473, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Harley, C.C.; Rooney, A.D.; Breslin, C.B. The selective detection of dopamine at a polypyrrole film doped with sulfonated β-cyclodextrins. Sens. Actuators B Chem. 2010, 150, 498–504. [Google Scholar] [CrossRef]
- Carelli, I.; Chiarotto, I.; Curulli, A.; Palleschi, G. Electropolymerization of hydroxybenzene and aminobenzene isomers on platinum electrodes to assemble interference-free electrochemical biosensors. Electrochim. Acta 1996, 41, 1793–1800. [Google Scholar] [CrossRef]
- Zen, J.M.; Chen, P.J. A selective voltammetric method for uric acid and dopamine detection using clay-modified electrode. Anal. Chem. 1997, 69, 5087–5093. [Google Scholar] [CrossRef]
- Malem, F.; Mandler, D. Self-assembled monolayers in electroanalytlcal chemistry: Application of -mercapto carboxylic acid monolayers for the electrochemical detection of dopamine in the presence of a high concentration of ascorbic acid. Anal. Chem. 1993, 65, 37–41. [Google Scholar] [CrossRef]
- Rubianes, M.D.; Rivas, G.A. Highly selective dopamine quantification using a glassy carbon electrode modified with a melanin-type polymer. Anal. Chim. Acta 2001, 440, 99–108. [Google Scholar] [CrossRef]

| Modified Electrode | Method | Linear Relationship | Correlation Coefficient | Limit Detection | Reference |
|---|---|---|---|---|---|
| PPyox/graphene/GCE | CV | 0.5–10 μM 25 μM–1.0 mM | 0.997 0.996 | 0.1 μM | [31] |
| PPy-RGO core-shell microspheres/GCE | DPV | 10 μM–10 mM | 0.9996 | 1 nM | [32] |
| PPy/ferrocyanide/carbon paste electrode | LSV DPV | 0.1–1.2 mM 0.2–0.95 mM | 0.9984 0.9998 | 38.6 μM 15.1 μM | [52] |
| Au/PPy/Ag/GCE | Amperometric response | 0.1–5 mM | - | 50 μM | [53] |
| PPy/-cyclodextrin/GCE | CV | 2–10 μM | - | 6 μM | [54] |
| ZnO-CuxO-PPy/GCE | DPV | 0.1–130 μM | 0.9922 | 0.04 μM | [55] |
| PPy doped with sulfonated-cyclodextrin/GCE | Constant potential amperometry | 7.5–40 μM | 0.999 | 3.2 μM | [56] |
| PPy—MCM-48/Au electrode | CV SWV | 10 μM–1.2 mM 2–250 μM | 0.9989 0.9996 | 2.5 μM 0.7 μM | This work |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zablocka, I.; Wysocka-Zolopa, M.; Winkler, K. Electrochemical Detection of Dopamine at a Gold Electrode Modified with a Polypyrrole–Mesoporous Silica Molecular Sieves (MCM-48) Film. Int. J. Mol. Sci. 2019, 20, 111. https://doi.org/10.3390/ijms20010111
Zablocka I, Wysocka-Zolopa M, Winkler K. Electrochemical Detection of Dopamine at a Gold Electrode Modified with a Polypyrrole–Mesoporous Silica Molecular Sieves (MCM-48) Film. International Journal of Molecular Sciences. 2019; 20(1):111. https://doi.org/10.3390/ijms20010111
Chicago/Turabian StyleZablocka, Izabela, Monika Wysocka-Zolopa, and Krzysztof Winkler. 2019. "Electrochemical Detection of Dopamine at a Gold Electrode Modified with a Polypyrrole–Mesoporous Silica Molecular Sieves (MCM-48) Film" International Journal of Molecular Sciences 20, no. 1: 111. https://doi.org/10.3390/ijms20010111
APA StyleZablocka, I., Wysocka-Zolopa, M., & Winkler, K. (2019). Electrochemical Detection of Dopamine at a Gold Electrode Modified with a Polypyrrole–Mesoporous Silica Molecular Sieves (MCM-48) Film. International Journal of Molecular Sciences, 20(1), 111. https://doi.org/10.3390/ijms20010111





