Mesenchymal Stem Cells as New Therapeutic Approach for Diabetes and Pancreatic Disorders
Abstract
1. Introduction
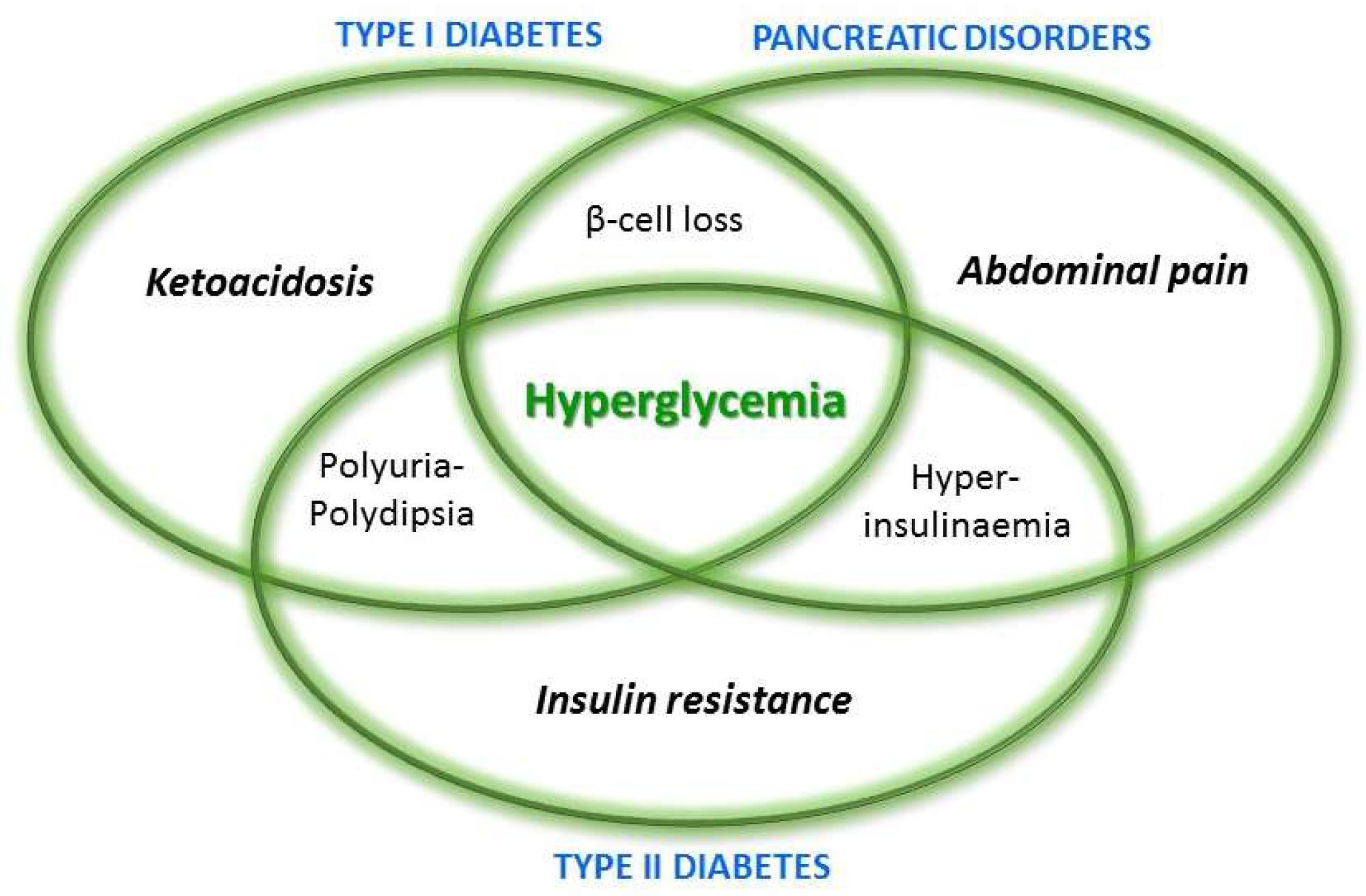
2. Pancreatic Disorders
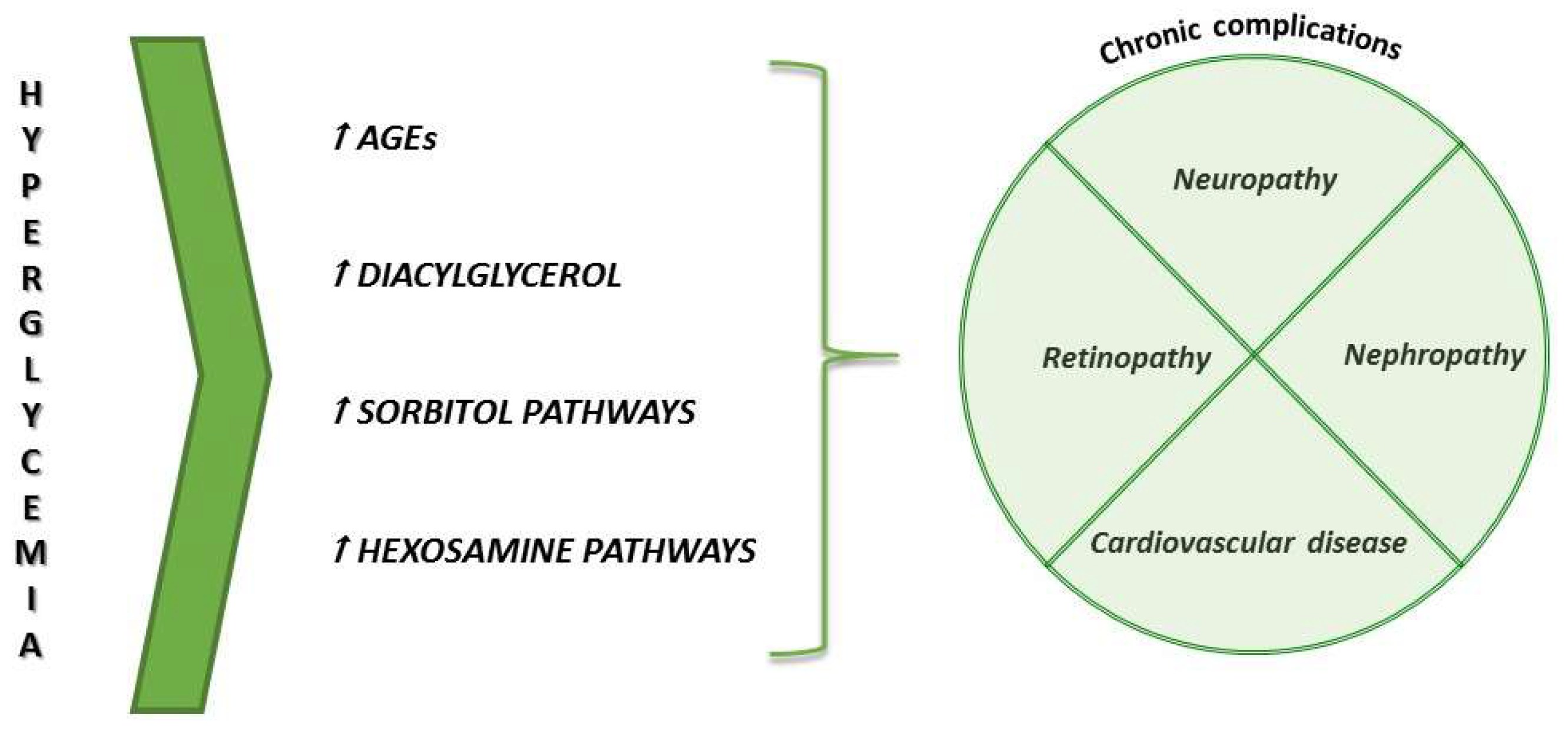
2.1. Diabetes
2.2. Pancreatitis
2.2.1. Acute Pancreatitis
2.2.2. Chronic Pancreatitis
2.3. Pancreatic Cancer
3. Mesenchymal Stem Cells (MSCs)
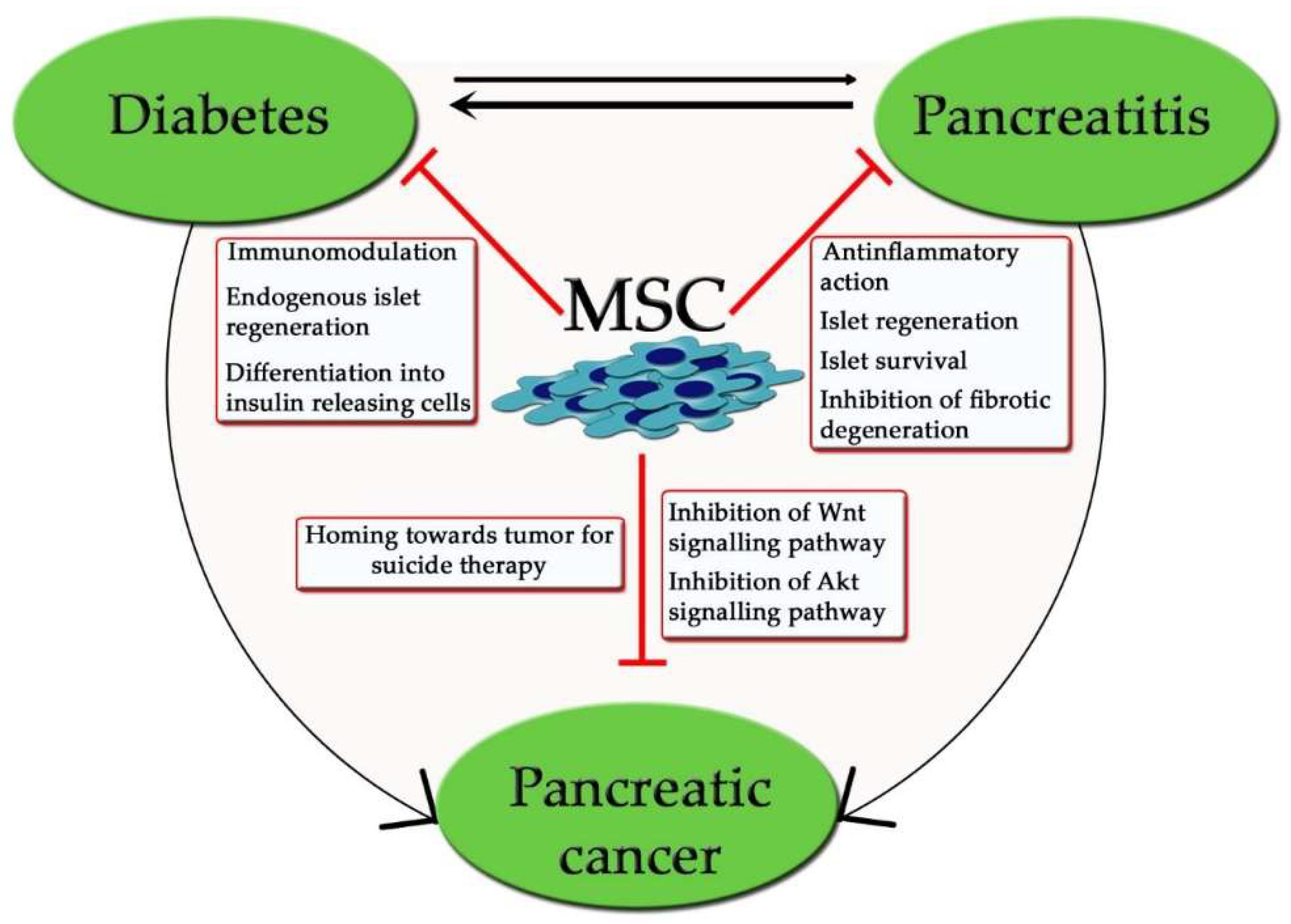
4. MSCs and Diabetes
5. MSCs and Acute and Chronic Pancreatitis
6. MSCs and Pancreatic Cancer
7. Potential Drawbacks
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| MSCs | Mesenchymal Stem Cells |
| ADA | American Diabetes Association |
| AGEs | Advanced Glycosylation End products |
| PKC | Protein Kinase C |
| Cox2 | Cyclooxigenase 2 |
| PDK-1 | Pancreatic Duodenal Homeobox-1 |
| GLUT-4 | Glucose Transporter 4 |
| TNF-α | Tumor Necrosis Factor-α |
| HDL | High Density Lipoprotein |
| VLDL | Very Low Density Lipoprotein |
| FFA | Free Fatty Acids |
| IL | Interleukyn |
| IGF-1 | Insulin-like Growth Factor-1 |
| VEGF | Vascular Endothelial Growth Factor |
| TCA | Taurocholic Acid |
| LPS | Lipopolysaccharides |
| TGF-β | Tumor Growth Factor-β |
| INF-γ | Interferon-γ |
| Trail-1 | Tumor Necrosis Factor (TNF)-related apoptosis-inducing ligand |
| ISCT | International Society of Cellular Therapy |
References
- Gonzalez-Perez, A.; Schlienger, R.G.; Rodríguez, L.A. Acute pancreatitis in association with type 2 diabetes and antidiabetic drugs: A population-based cohort study. Diabetes Care 2010, 33, 2580–2585. [Google Scholar] [CrossRef] [PubMed]
- Moniri, M.R.; Dai, L.J.; Warnock, G.L. The challenge of pancreatic cancer therapy and novel treatment strategy using engineered mesenchymal stem cells. Cancer Gene Ther. 2014, 21, 12–23. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care 2014, 37, S81–S90. [Google Scholar] [CrossRef] [PubMed]
- Fauci, A.S.; Braunwald, E.; Kasper, D.; Hauser, S.L.; Longo, D.L.; Jameson, J.L.; Loscalzo, J. Harrison’s Principles of Internal Medicine, 17th ed.; Mc Graw Hill: New York, NY, USA, 2008; ISBN 978-0-07-147693-5. [Google Scholar]
- Volpe, C.M.O.; Villar-Delfino, P.H.; Dos Anjos, P.M.; Nogueira-Machado, J.A. Cellular death, reactive oxygen species (ROS) and diabetic complications. Cell Death Dis. 2018, 9, 119. [Google Scholar] [CrossRef] [PubMed]
- Gurel, Z.; Sheibani, N. O-Linked β-N-acetylglucosamine (O-GlcNAc) modification: A new pathway to decode pathogenesis of diabetic retinopathy. Clin. Sci. (London) 2018, 132, 185–198. [Google Scholar] [CrossRef] [PubMed]
- Albai, O.; Roman, D. and Frandes M. Hypertriglyceridemia, an important and independent risk factor for acute pancreatitis in patients with type 2 diabetes mellitus. Ther. Clin. Risk Manag. 2017, 13, 515–522. [Google Scholar] [CrossRef] [PubMed]
- Majumder, S.; Chari, S.T. Chronic pancreatitis. Lancet 2016, 387, 1957–1966. [Google Scholar] [CrossRef]
- Pinho, A.V.; Chantrill, L.; Rooman, I. Chronic pancreatitis: A path to pancreatic cancer. Cancer Lett. 2014, 345, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Ewald, N.; Hardt, P.D. Diagnosis and treatment of diabetes mellitus in chronic pancreatitis. World J. Gastroenterol. 2013, 19, 7276–7281. [Google Scholar] [CrossRef] [PubMed]
- Ewald, N.; Kaufmann, C.; Raspe, A.; Kloer, H.; Bretzel, R.G.; Hardt, P.D. Prevalence of diabetes mellitus secondary to pancreatic diseases (type 3c). Diabetes Metab. Res. Rev. 2012, 28, 338–342. [Google Scholar] [CrossRef] [PubMed]
- Andersen, D.K.; Korc, M.; Petersen, G.M.; Eibl, G.; Li, D.; Rickels, M.R.; Chari, S.T.; Abbruzzese, J.L. Diabetes, Pancreatogenic Diabetes, and Pancreatic Cancer. Diabetes 2017, 66, 1103–1110. [Google Scholar] [CrossRef] [PubMed]
- Salvatore, T.; Marfella, R.; Rizzo, M.; Sasso, F. Pancreatic cancer and diabetes. A two-way relationship in the perspective of diabetologist. Int. J. Surg. 2015, 21, S72–S77. [Google Scholar] [CrossRef] [PubMed]
- De Ferranti, S.D.; De Boer, I.H.; Fonseca, V.; Fox, C.S.; Hill Golden, S.; Lavie, C.J.; Magge, S.N.; Marx, N.; McGuire, D.K.; Orchard, T.J.; et al. Type 1 Diabetes Mellitus and Cardiovascular Disease: A Scientific Statement From the American Heart Association and American Diabetes Association. Diabetes Care 2014, 37, 2843–2863. [Google Scholar] [CrossRef] [PubMed]
- Boden, G.; Laakso, M. Lipids and glucose in type 2 diabetes: What is the cause and effect? Diabetes Care 2004, 27, 2253–2259. [Google Scholar] [CrossRef] [PubMed]
- Gillies, N.A.; Pendharkar, S.A.; Singh, R.G.; Asrani, V.M.; Petrov, M.S. Lipid metabolism in patients with chronic hyperglycemia after an episode of acute pancreatitis. Diabetes Metab. Syndr. 2017, 11, S233–S241. [Google Scholar] [CrossRef] [PubMed]
- Ni, Q.; Yun, L.; Xu, R.; Shang, D. Correlation between blood lipid levels and chronic pancreatitis: A retrospective case-control study of 48 cases. Medicine (Baltimore) 2014, 93. [Google Scholar] [CrossRef] [PubMed]
- Li, D. Diabetes and pancreatic cancer. Mol. Carcinog. 2012, 51, 64–74. [Google Scholar] [CrossRef] [PubMed]
- Galdiero, M.R.; Marone, G.; Mantovani, A. Cancer Inflammation and Cytokines. Cold Spring Harb. Perspect. Biol. 2018, 10, a028662. [Google Scholar] [CrossRef] [PubMed]
- Munigala, S.; Kanwal, F.; Xian, H.; Scherrer, J.F.; Agarwal, B. Increased risk of pancreatic adenocarcinoma after acute pancreatitis. Clin. Gastroenterol. Hepatol. 2014, 12, 1143–1150. [Google Scholar] [CrossRef] [PubMed]
- Kong, X.; Sun, T.; Kong, F.; Du, Y.; Li, Z. Chronic Pancreatitis and Pancreatic Cancer. Gastrointest. Tumors 2014, 1, 123–134. [Google Scholar] [CrossRef] [PubMed]
- Dhar, P.; Kalghatgi, S.; Saraf, V. Pancreatic cancer in chronic pancreatitis. Indian. J. Surg. Oncol. 2015, 6, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Yi, T.; Song, S.U. Immunomodulatory properties of mesenchymal stem cells and their therapeutic applications. Arch. Pharm. Res. 2012, 35, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Scuteri, A.; Miloso, M.; Foudah, D.; Orciani, M.; Cavaletti, G.; Tredici, G. Mesenchymal stem cells neuronal differentiation ability: A real perspective for nervous system repair? Curr. Stem Cell Res. Ther. 2011, 6, 82–92. [Google Scholar] [CrossRef] [PubMed]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.; Krause, D.; Deans, R.; Keating, A.; Prockop, D.; Horwitz, E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy Position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef] [PubMed]
- Satija, N.K.; Singh, V.K.; Verma, Y.K.; Gupta, P.; Sharma, S.; Afrin, F.; Sharma, M.; Sharma, P.; Tripathi, R.P.; Gurudutta, G.U. Mesenchymal stem cell-based therapy: A new paradigm in regenerative medicine. J. Cell. Mol. Med. 2009, 13, 4385–4402. [Google Scholar] [CrossRef] [PubMed]
- Kaundal, U.; Bagai, U.; Rakha, A. Immunomodulatory plasticity of mesenchymal stem cells: A potential key to successful solid organ transplantation. J. Transl. Med. 2018, 16. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Guo, X.; Chen, S.Y. Function and Therapeutic Potential of Mesenchymal Stem Cells in Atherosclerosis. Front. Cardiovasc. Med. 2017, 4. [Google Scholar] [CrossRef] [PubMed]
- Monfrini, M.; Donzelli, E.; Rodriguez-Menendez, V.; Ballarini, E.; Carozzi, V.A.; Chiorazzi, A.; Meregalli, C.; Canta, A.; Oggioni, N.; Crippa, L.; et al. Therapeutic potential of Mesenchymal Stem Cells for the treatment of diabetic peripheral neuropathy. Exp. Neurol. 2017, 288, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.M.; Morsi, M.; Ghoneim, N.I.; Abdel-Daim, M.M.; El-Badri, N. Mesenchymal stromal cell therapy for Pancreatitis: A Systematic Review. Oxid. Med. Cell. Longev. 2018, 2018, 3250864. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, M.; Kumar, A.; Bhonde, R.R. Reversal of experimental diabetes by multiple bone marrow transplantation. Biochem. Biophys. Res. Commun. 2005, 328, 318–325. [Google Scholar] [CrossRef] [PubMed]
- Bueno Pde, G.; Yochite, J.N.; Derigge-Pisani, G.F.; Malmegrim de Farias, K.C.; de Avó, L.R.; Voltarelli, J.C.; Leal, Â.M. Metabolic and pancreatic effects of bone marrow mesenchymal stem cells transplantation in mice fed high-fat diet. PLoS ONE 2015, 10, e0124369. [Google Scholar] [CrossRef] [PubMed]
- Ezquer, F.; Ezquer, M.; Contador, D.; Ricca, M.; Simon, V.; Conget, P. The antidiabetic effect of mesenchymal stem cells is unrelated to their transdifferentiation potential but to their capability to restore Th1/Th2 balance and to modify the pancreatic microenvironment. Stem Cells 2012, 30, 1664–1674. [Google Scholar] [CrossRef] [PubMed]
- Maccario, R.; Podestà, M.; Moretta, A.; Cometa, A.; Comoli, P.; Montagna, D.; Daudt, L.; Ibatici, A.; Piaggio, G.; Pozzi, S.; et al. Interaction of human mesenchymal stem cells with cells involved in alloantigen-specific immune response favors the differentiation of CD4+ T-cell subsets expressing a regulatory/suppressive phenotype. Haematologica 2005, 90, 516–525. [Google Scholar] [PubMed]
- Chen, J.; Liu, Z.; Hong, M.M.; Zhang, H.; Chen, C.; Xiao, M.; Wang, J.; Yao, F.; Ba, M.; Liu, J.; et al. Proangiogenic compositions of microvesicles derived from human umbilical cord mesenchymal stem cells. PLoS ONE 2014, 9, e115316. [Google Scholar] [CrossRef] [PubMed]
- Huang, P.; Gebhart, N.; Richelson, E.; Brott, T.G.; Meschia, J.F.; Zubair, A.C. Mechanism of mesenchymal stem cell-induced neuron recovery and anti-inflammation. Cytotherapy 2014, 16, 1336–1344. [Google Scholar] [CrossRef] [PubMed]
- Gao, D.; Xie, J.; Zhang, J.; Feng, C.; Yao, B.; Ma, K.; Li, J.; Wu, X.; Huang, S.; Fu, X. MSC attenuate diabetes-induced functional impairment in adipocytes via secretion of insulin-like growth factor-1. Biochem. Biophys. Res. Commun. 2014, 452, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Hashemian, S.J.; Kouhnavard, M.; Nasli-Esfahani, E. Mesenchymal stem cells: Rising concerns over their application in treatment of Type One Diabetes Mellitus. J. Diabetes Res. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Katuchova, J.; Harvanova, D.; Spakova, T.; Kalanin, R.; Farkas, D.; Durny, P.; Rosocha, J.; Radonak, J.; Petrovic, D.; Siniscalco, D.; et al. Mesenchymal stem cells in the treatment of type 1 diabetes mellitus. Endocr. Pathol. 2015, 26, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Taneera, J.; Rosengren, A.; Renstrom, E.; Nygren, J.M.; Serup, P.; Rorsman, P.; Jacobsen, S.E. Failure of transplanted bone marrow cells to adopt a pancreatic beta-cell fate. Diabetes 2006, 55, 290–296. [Google Scholar] [CrossRef] [PubMed]
- Tsai, P.J.; Wang, H.S.; Lin, G.J.; Chou, S.C.; Chu, T.H.; Chuan, W.T.; Lu, Y.J.; Weng, Z.C.; Su, C.H.; Hsieh, P.S.; et al. Undifferentiated Wharton’s Jelly Mesenchymal Stem Cell Transplantation Induces Insulin-Producing Cell Differentiation and Suppression of T-Cell-Mediated Autoimmunity in Nonobese Diabetic Mice. Cell Transplant. 2015, 24, 1555–1570. [Google Scholar] [CrossRef] [PubMed]
- Dave, S.D.; Vanikar, A.V.; Trivedi, H.L. Extrinsic factors promoting in vitro differentiation of insulin-secreting cells from human adipose tissue-derived mesenchymal stem cells. Appl. Biochem. Biotechnol. 2013, 170, 962–971. [Google Scholar] [CrossRef] [PubMed]
- Bal, T.; Nazli, C.; Okcu, A.; Duruksu, G.; Karaöz, E.; Kizilel, S. Mesenchymal stem cells and ligand incorporation in biomimetic poly(ethylene glycol) hydrogels significantly improve insulin secretion from pancreatic islets. J. Tissue Eng. Regen. Med. 2017, 11, 694–703. [Google Scholar] [CrossRef] [PubMed]
- Van Pham, P.; Thi-My Nguyen, P.; Thai-Quynh Nguyen, A.; Minh Pham, V.; Nguyen-Tu Bui, A.; Thi-Tung Dang, L.; Gia Nguyen, K.; Kim Phan, N. Improved differentiation of umbilical cord blood-derived mesenchymal stem cells into insulin-producing cells by PDX-1 mRNA transfection. Differentiation 2014, 87, 200–208. [Google Scholar] [CrossRef] [PubMed]
- Scuteri, A.; Donzelli, E.; Rodriguez-Menendez, V.; Ravasi, M.; Monfrini, M.; Bonandrini, B.; Figliuzzi, M.; Remuzzi, A.; Tredici, G. A double mechanism for the mesenchymal stem cells’ positive effect on pancreatic islets. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Ezquer, F.; Ezquer, M.; Arango-Rodriguez, M.; Conget, P. Could donor multipotent mesenchymal stromal cells prevent or delay the onset of diabetic retinopathy? Acta Ophthalmol. 2014, 92, e86–e95. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Zhang, D.; Zeng, Y.; Ma, J.; Wang, J.; Guo, H.; Zhang, J.; Wang, M.; Zhang, W.; Gong, N. Bone Marrow-Derived Mesenchymal Stem Cells Protect Islet Grafts Against Endoplasmic Reticulum Stress-Induced Apoptosis During the Early Stage After Transplantation. Stem Cells 2018, 36, 1045–1061. [Google Scholar] [CrossRef] [PubMed]
- Carlsson, P.O.; Korsgren, O.; Le Blanc, K. Mesenchymal stromal cells to halt the progression of type 1 diabetes? Curr. Diab. Rep. 2015, 15. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.H.; Song, S.U.; Yi, T.; Jeon, M.S.; Hong, S.W.; Zheng, H.M.; Lee, H.S.; Choi, M.J.; Lee, D.H.; Hong, S.S. Human bone marrow-derived clonal mesenchymal stem cells inhibit inflammation and reduce acute pancreatitis in rats. Gastroenterology 2011, 140, 998–1008. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.H.; Yi, T.; Son, M.K.; Song, S.U.; Hong, S.S. Therapeutic effect of human clonal bone marrow-derived mesenchymal stem cells in severe acute pancreatitis. Arch. Pharm. Res. 2015, 38, 742–751. [Google Scholar] [CrossRef] [PubMed]
- Yin, G.; Hu, G.; Wan, R.; Yu, G.; Cang, X.; Xiong, J.; Ni, J.; Hu, Y.; Xing, M.; Fan, Y.; et al. Role of Microvesicles From Bone Marrow Mesenchymal Stem Cells in Acute Pancreatitis. Pancreas 2016, 45, 1282–1293. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; He, Z.; Huang, D.; Gao, J.; Gong, Y.; Wu, H.; Xu, A.; Meng, X.; Li, Z. Infusion of Bone Marrow Mesenchymal Stem Cells Attenuates Experimental Severe Acute Pancreatitis in Rats. Stem Cells Int. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Yin, G.; Hu, G.; Wan, R.; Yu, G.; Cang, X.; Ni, J.; Xiong, J.; Hu, Y.; Xing, M.; Fan, Y.; et al. Role of bone marrow mesenchymal stem cells in L-arg-induced acute pancreatitis: Effects and possible mechanisms. Int. J. Clin. Exp. Pathol. 2015, 8, 4457–4468. [Google Scholar] [PubMed]
- Hua, J.; He, Z.G.; Qian, D.H.; Lin, S.P.; Gong, J.; Meng, H.B.; Yang, T.S.; Sun, W.; Xu, B.; Zhou, B.; et al. Angiopoietin-1 gene-modified human mesenchymal stem cells promote angiogenesis and reduce acute pancreatitis in rats. Int. J. Clin. Exp. Pathol. 2014, 7, 3580–3595. [Google Scholar] [PubMed]
- Qin, T.; Liu, C.J.; Zhang, H.W.; Pan, Y.F.; Tang, Q.; Liu, J.K.; Wang, Y.Z.; Hu, M.X.; Xue, F. Effect of the IκBα mutant gene delivery to mesenchymal stem cells on rat chronic pancreatitis. Genet. Mol. Res. 2014, 13, 371–385. [Google Scholar] [CrossRef] [PubMed]
- Moniri, M.R.; Sun, X.Y.; Rayat, J.; Dai, D.; Ao, Z.; He, Z.; Verchere, C.B.; Dai, L.J.; Warnock, G.L. TRAIL-engineered pancreas-derived mesenchymal stem cells: Characterization and cytotoxic effects on pancreatic cancer cells. Cancer Gene Ther. 2012, 19, 652–658. [Google Scholar] [CrossRef] [PubMed]
- Qiao, L.; Xu, Z.; Zhao, T.; Zhao, Z.; Shi, M.; Zhao, R.C.; Ye, L.; Zhang, X. Suppression of tumorigenesis by human mesenchymal stem cells in a hepatoma model. Cell Res. 2008, 18, 500–507. [Google Scholar] [CrossRef] [PubMed]
- Khakoo, A.Y.; Pati, S.; Anderson, S.A.; Reid, W.; Elshal, M.F.; Rovira, I.I.; Nguyen, A.T.; Malide, D.; Combs, C.A.; Hall, G.; et al. Human mesenchymal stem cells exert potent antitumorigenic effects in a model of Kaposi’s sarcoma. J. Exp. Med. 2006, 203, 1235–1247. [Google Scholar] [CrossRef] [PubMed]
- Amara, I.; Touati, W.; Beaune, P.; de Waziers, I. Mesenchymal stem cells as cellular vehicles for prodrug gene therapy against tumors. Biochimie 2014, 105, 4–11. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Mahato, R.I. Mesenchymal stem cell-based therapy for type 1 diabetes. Discov. Med. 2014, 17, 139–143. [Google Scholar] [PubMed]
- Yang, B.; Bai, B.; Liu, C.X.; Wang, S.Q.; Jiang, X.; Zhu, C.L.; Zhao, Q.C. Effect of umbilical cord mesenchymal stem cells on treatment of severe acute pancreatitis in rats. Cytotherapy 2013, 15, 154–162. [Google Scholar] [CrossRef] [PubMed]
- Kornicka, K.; Houston, J.; Marycz, K. Dysfunction of Mesenchymal Stem Cells Isolated from Metabolic Syndrome and Type 2 Diabetic Patients as Result of Oxidative Stress and Autophagy may Limit Their Potential Therapeutic Use. Stem Cell Rev. 2018, 14, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Yaochite, J.N.; de Lima, K.W.; Caliari-Oliveira, C.; Palma, P.V.; Couri, C.E.; Simões, B.P.; Covas, D.T.; Voltarelli, J.C.; Oliveira, M.C.; Donadi, E.A.; et al. Multipotent mesenchymal stromal cells from patients with newly diagnosed type 1 diabetes mellitus exhibit preserved in vitro and in vivo immunomodulatory properties. Stem Cell Res. Ther. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
| Disorder | Underlying mechanisms |
|---|---|
| Type I Diabetes | Immune-mediated β-cell destruction |
| Type II Diabetes | Insulin resistance and insulin deficiency |
| Type III Diabetes | a. Genetic defects β-cell function |
| b. Genetic defects of insulin action | |
| c. Diseases of the exocrine pancreas | |
| d. Endocrinopathies | |
| e. Drug- or chemical-induced | |
| f. Infections | |
| g. Uncommon forms of immune-mediated diabetes | |
| h. Genetic Syndrome | |
| Acute Pancreatitis | Inflammation due to pancreatic enzyme activation inside the pancreas |
| Chronic Pancreatitis | Inflammation, fibrosis and progressive destruction of exocrine and endocrine tissue |
| Pancreatic Cancer | Genetic mutations |
| Positive Markers | Negative Markers | |
|---|---|---|
| Marker Expression | CD73/5’-Nucleotidase CD90/Thy1 CD105 | CD34 CD45 CD11b/CD14 CD79α HLA ClassII |
| Culture Type | Adhesion to plastic | |
| Differentiation Ability | Adipogenic differentiation, Osteogenic differentiation, Chondrogenic differentiation after specific stimulation | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scuteri, A.; Monfrini, M. Mesenchymal Stem Cells as New Therapeutic Approach for Diabetes and Pancreatic Disorders. Int. J. Mol. Sci. 2018, 19, 2783. https://doi.org/10.3390/ijms19092783
Scuteri A, Monfrini M. Mesenchymal Stem Cells as New Therapeutic Approach for Diabetes and Pancreatic Disorders. International Journal of Molecular Sciences. 2018; 19(9):2783. https://doi.org/10.3390/ijms19092783
Chicago/Turabian StyleScuteri, Arianna, and Marianna Monfrini. 2018. "Mesenchymal Stem Cells as New Therapeutic Approach for Diabetes and Pancreatic Disorders" International Journal of Molecular Sciences 19, no. 9: 2783. https://doi.org/10.3390/ijms19092783
APA StyleScuteri, A., & Monfrini, M. (2018). Mesenchymal Stem Cells as New Therapeutic Approach for Diabetes and Pancreatic Disorders. International Journal of Molecular Sciences, 19(9), 2783. https://doi.org/10.3390/ijms19092783






