Molecular Mechanism and Health Role of Functional Ingredients in Blueberry for Chronic Disease in Human Beings
Abstract
1. Introduction
2. Functional Ingredients in Blueberry
3. Preventive Chronic Disease of Blueberry
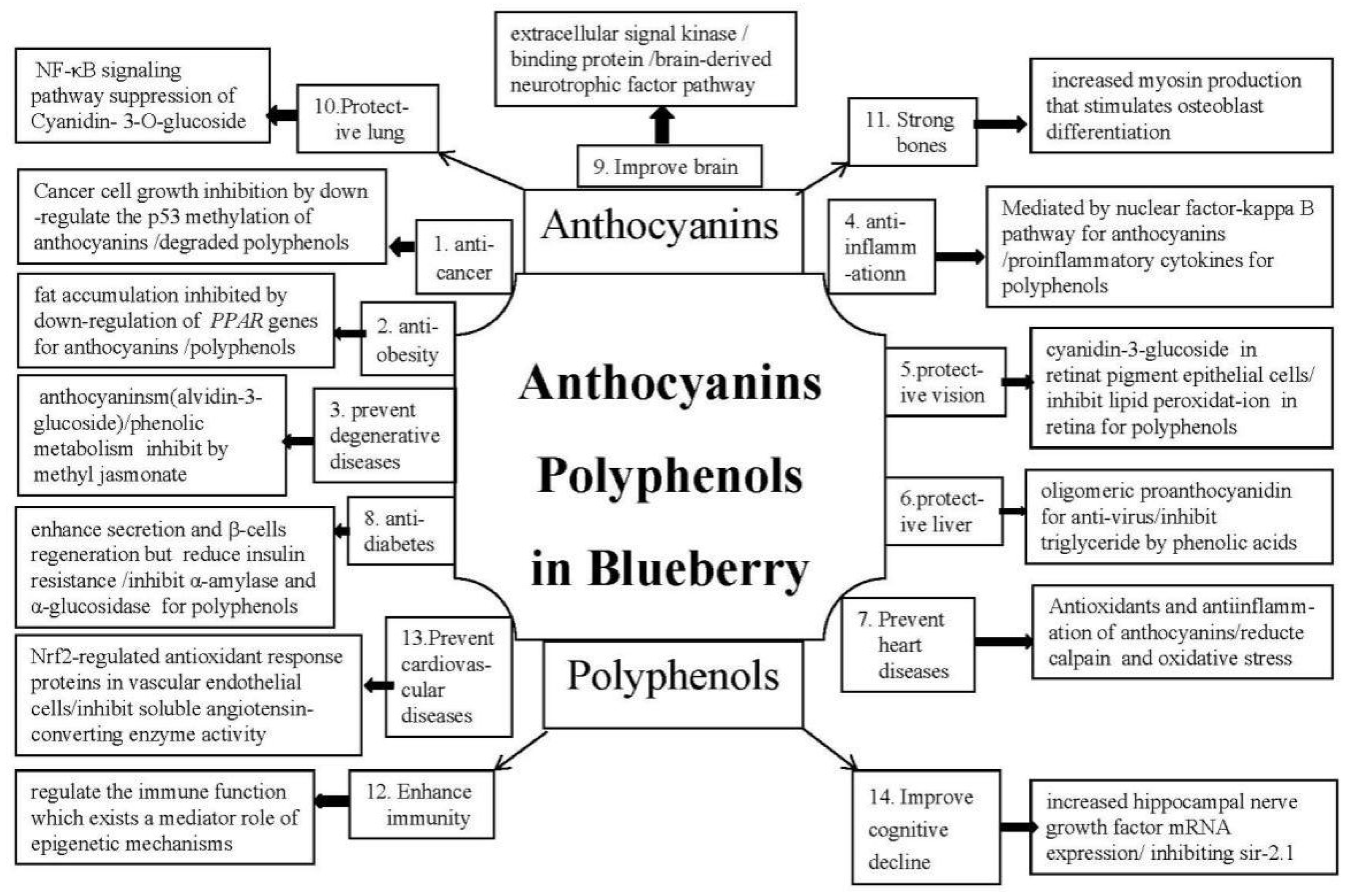
3.1. Anticancer and Functional Ingredients of Blueberry
3.2. Anti-Obesity and Functional Ingredients of Blueberry
3.3. Prevent Degenerative Diseases and Functional Ingredients of Blueberry
3.4. Anti-Inflammatory and Functional Ingredients of Blueberry
3.5. Protective Vision and Functional Ingredients of Blueberry
3.6. Protective Liver and Functional Ingredients of Blueberry
3.7. Heart Disease Prevention and Functional Ingredients of Blueberry
3.8. Antidiabetic Properties and Functional Ingredients of Blueberry
3.9. Improving Brain Health and Functional Ingredients of Blueberry
3.10. Lung-Protective Properties and Functional Ingredients of Blueberry
3.11. Strong Bones and Functional Ingredients of Blueberry
3.12. Enhance Immunological Effects and Functional Ingredients of Blueberry
3.13. Prevent Cardiovascular Diseases and Functional Ingredients of Blueberry
3.14. Blueberries can Improve Cognitive Decline, Functional Ingredients of Blueberry
4. Major Mechanisms and Structural Activity of Blueberry Compounds for Preventive Chronic Disease
4.1. Anthocyanins Mechanism and its Structural Activity
4.2. Polyphenols Mechanism and its Structural Activity
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Skrovankova, S.; Sumczynski, D.; Mlcek, J.; Jurikova, T.; Sochor, J. Bioactive compounds and antioxidant activity in different types of berries. Int. J. Mol. Sci. 2015, 16, 24673–24706. [Google Scholar] [CrossRef] [PubMed]
- Kausar, H.; Jeyabalan, J.; Aqil, F.; Chabba, D.; Sidana, J.; Singh, I.P.; Gupta, R.C. Berry anthocyanidins synergistically suppress growth and invasive potential of human non-small-cell lung cancer cells. Cancer Lett. 2012, 325, 54–62. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.W.; Du, J.; Pu, X.Y.; Yang, J.Z.; Yang, T.; Yang, S.; Yang, X. Coevolution between cancer activities and food structure of human being from southwest China. BioMed Res. Int. 2015, 2015, 497934. [Google Scholar] [CrossRef] [PubMed]
- Ayoub, M.; de Camargo, A.C.; Shahidi, F. Antioxidants and bioactivities of free, esterified and insoluble-bound phenolics from berry seed meals. Food Chem. 2016, 197, 221–232. [Google Scholar] [CrossRef] [PubMed]
- Marhuenda, J.; Alemán, M.D.; Gironés-Vilaplana, A.; Pérez, A.; Caravaca, G.; Figueroa, F.; Mulero, J.; Zafrilla, P. Phenolic composition, antioxidant activity, and in vitro availability of four different berries. J. Chem. 2016, 2016, 5194901. [Google Scholar] [CrossRef]
- Ciric, A.; Jelikic-Stankov, M.; Cvijovic, M.; Djurdjevic, P. Statistical optimization of an RP-HPLC method for the determination of selected flavonoids in berry juices and evaluation of their antioxidant activities. Biomed. Chromatogr. 2018, 32, e4150. [Google Scholar] [CrossRef] [PubMed]
- Cásedas, G.; Les, F.; Gómez-Serranillos, M.P.; Smith, C.; López, V. Anthocyanin profile, antioxidant activity and enzyme inhibiting properties of blueberry and cranberry juices: A comparative study. Food Funct. 2017, 8, 4187–4193. [Google Scholar] [CrossRef] [PubMed]
- Cortez, R.; Luna-Vital, D.A.; Margulis, D.; Gonzalez de Mejia, E. Natural pigments: Stabilization methods of anthocyanins for food applications. Compr. Rev. Food Sci. Food Saf. 2017, 16, 180–198. [Google Scholar] [CrossRef]
- Chen, C.F.; Li, Y.D.; Xu, Z. Chemical principles and bioactivities of blueberry. Yao Xue Xue Bao 2010, 45, 422–429. (In Chinese) [Google Scholar] [PubMed]
- Rowland, L.J.; Alkharouf, N.; Darwish, O.; Ogden, E.L.; Polashock, J.J.; Bassil, N.V.; Main, D. Generation and analysis of blueberry transcriptome sequences from leaves, developing fruit, and flower buds from cold acclimation through deacclimation. BMC Plant Biol. 2012, 12, 46. [Google Scholar] [CrossRef] [PubMed]
- Ravoori, S.; Vadhanam, M.V.; Aqil, F.; Gupta, R.C. Inhibition of estrogen-mediated mammary tumorigenesis by blueberry and black raspberry. J. Agric. Food Chem. 2012, 60, 5547–5555. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Tang, Q.; Gao, Z.; Yu, Z.; Song, H.; Zheng, X.; Chen, W. Blueberry and mulberry juice prevent obesity development in C57BL/6 mice. PLoS ONE 2013, 8, e77585. [Google Scholar] [CrossRef] [PubMed]
- Ren, T.; Huang, C.; Cheng, M. Dietary Blueberry and bifidobacteria attenuate nonalcoholic fatty liver disease in rats by affecting SIRT1-mediated signaling pathway. Oxid. Med. Cell. Longev. 2014, 2014, 469059. [Google Scholar] [CrossRef] [PubMed]
- Lin, Z.; Fischer, J. Wicker, L. Intermolecular binding of blueberry pectin-rich fractions and anthocyanin. Food Chem. 2016, 194, 986–993. [Google Scholar] [CrossRef] [PubMed]
- Rahal, O.M.; Pabona, J.M.; Kelly, T.; Huang, Y.; Hennings, L.J.; Prior, R.L.; Al-Dwairi, A.; Simmen, F.A.; Simmen, R.C. Suppression of Wnt1-induced mammary tumor growth and lower serum insulin in offspring exposed to maternal blueberry diet suggest early dietary influence on developmental programming. Carcinogenesis 2013, 34, 464–474. [Google Scholar] [CrossRef] [PubMed]
- Kraujalytė, V.; Venskutonis, P.R.; Pukalskas, A.; Česonienė, L.; Daubaras, R. Antioxidant properties, phenolic composition and potentiometric sensor array evaluation of commercial and new blueberry (Vaccinium corymbosum) and bog blueberry (Vaccinium uliginosum) genotypes. Food Chem. 2015, 188, 583–590. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Yongku, L.; Wang, X.; Wang, F.; Wang, X.; Wang, Y.; Meng, X. Simultaneous separation and determination of organic acids in blueberry juices by capillary electrophoresis-electrospray ionization mass spectrometry. J. Food Sci. Technol. 2015, 52, 5228–5235. [Google Scholar] [CrossRef] [PubMed]
- Correa-Betanzo, J.; Allen-Vercoe, E.; McDonald, J.; Schroeter, K.; Corredig, M.; Paliyath, G. Stability and biological activity of wild blueberry (Vaccinium angustifolium) polyphenols during simulated in vitro gastrointestinal digestion. Food Chem. 2014, 165, 522–531. [Google Scholar] [CrossRef] [PubMed]
- Zu, X.Y.; Zhang, Z.Y.; Zhang, X.W.; Yoshioka, M.; Yang, Y.N.; Li, J. Anthocyanins extracted from Chinese blueberry (Vaccinium uliginosum L.) and its anticancer effects on DLD-1 and COLO205 cells. Chin. Med. J. 2010, 123, 2714–2719. [Google Scholar] [PubMed]
- Pertuzatti, P.B.; Barcia, M.T.; Rodrigues, D.; da Cruz, P.N.; Hermosín-Gutiérrez, I.; Smith, R.; Godoy, H.T. Antioxidant activity of hydrophilic and lipophilic extracts of Brazilian blueberries. Food Chem. 2014, 164, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.G.; Vance, T.M.; Nam, T.G.; Kim, D.O.; Koo, S.I.; Chun, O.K. Contribution of anthocyanin composition to total antioxidant capacity of berries. Plant Foods Hum. Nutr. 2015, 70, 427–432. [Google Scholar] [CrossRef] [PubMed]
- Bobinaitė, R.; Pataro, G.; Lamanauskas, N.; Šatkauskas, S.; Viškelis, P.; Ferrari, G. Application of pulsed electric field in the production of juice and extraction of bioactive compounds from blueberry fruits and their by-products. J. Food Sci. Technol. 2015, 52, 5898–58905. [Google Scholar] [CrossRef] [PubMed]
- Davidson, K.T.; Zhu, Z.; Balabanov, D.; Zhao, L.; Wakefield, M.R.; Bai, Q.; Fang, Y. Beyond conventional medicine—A look at blueberry, a cancer-fighting superfruit. Pathol. Oncol. Res. 2017, 24, 733–738. [Google Scholar] [CrossRef] [PubMed]
- Davidson, K.T.; Zhu, Z.; Bai, Q.; Xiao, H.; Wakefield, M.R.; Fang, Y. Blueberry as a potential radiosensitizer for treating cervical cancer. Pathol. Oncol. Res. 2017. [Google Scholar] [CrossRef] [PubMed]
- Faria, A.; Pestana, D.; Teixeira, D.; de Freitas, V.; Mateus, N.; Calhau, C. Blueberry anthocyanins and pyruvic acid adducts: Anticancer properties in breast cancer cell lines. Phytother. Res. 2010, 24, 1862–1869. [Google Scholar] [CrossRef] [PubMed]
- Bunea, A.; Rugină, D.; Sconţa, Z.; Pop, R.M.; Pintea, A.; Socaciu, C.; Tăbăran, F.; Grootaert, C.; Struijs, K.; VanCamp, J. Anthocyanin determination in blueberry extracts from various cultivars and their antiproliferative and apoptotic properties in B16-F10 metastatic murine melanoma cells. Phytochemistry 2013, 95, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Wang, T.; Zhang, B.; Zhao, H. Addition of sucrose during the blueberry heating process is good or bad? Evaluating the changes of anthocyanins/anthocyanidins and the anticancer ability in HepG-2 cells. Food Res. Int. 2018, 107, 509–517. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Wang, P.; Zhu, Y.; Liu, X.; Hu, X.; Chen, F. The chemoprotection of a blueberry anthocyanin extract against the acrylamide-induced oxidative stress in mitochondria: Unequivocal evidence in mice liver. Food Funct. 2015, 6, 3006–3012. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Xu, Z.; Chang, G.; Hou, J.; Hu, L.; Zhang, Y.; Yu, D.; Li, B.; Chang, S.; Xie, Y.; et al. The blueberry component pterostilbene has potent anti-myeloma activity in bortezomib-resistant cells. Oncol. Rep. 2017, 38, 488–496. [Google Scholar] [CrossRef] [PubMed]
- Minker, C.; Duban, L.; Karas, D.; Järvinen, P.; Lobstein, A.; Muller, C.D. Impact of procyanidins from different berries on caspase 8 activation in colon cancer. Oxid. Med. Cell. Longev. 2015, 2015, 154164. [Google Scholar] [CrossRef] [PubMed]
- Håkansson, A.; Bränning, C.; Molin, G.; Adawi, D.; Hagslätt, M.-L.; Jeppsson, B.; Nyman, M.; Ahrné, S. Blueberry husks and probiotics attenuate colorectal inflammation and oncogenesis, and liver injuries in rats exposed to cycling DSS-treatment. PLoS ONE 2012, 7, e33510. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Na, H.; Kasai, H.; Kawai, K.; Li, Y.S.; Yang, M. Comparison of blueberry (Vaccinium spp.) and vitamin C via antioxidative and epigenetic effects in human. J. Cancer Prev. 2017, 22, 174–181. [Google Scholar] [CrossRef] [PubMed]
- Wang, E.; Liu, Y.; Xu, C.; Liu, J. Antiproliferative and proapoptotic activities of anthocyanin and anthocyanidin extracts from blueberry fruits on B16-F10 melanoma cells. Food Nutr. Res. 2017, 61, 1325308. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.; Li, Z. Blueberries inhibit cyclooxygenase-1 and cyclooxygenase-2 activity in human epithelial ovarian cancer. Oncol. Lett. 2017, 13, 4897–4904. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Diaz, D.F.; Johnson, M.H.; de Mejia, E.G. Anthocyanins from fermented berry beverages inhibit inflammation-related adiposity response in vitro. J. Med. Food 2015, 18, 489–496. [Google Scholar] [CrossRef] [PubMed]
- Moghe, S.S.; Juma, S.; Imrhan, V.; Vijayagopal, P. Effect of blueberry polyphenols on 3T3-F442A preadipocyte differentiation. J. Med. Food 2012, 15, 448–452. [Google Scholar] [CrossRef] [PubMed]
- Diaconeasa, Z.; Leopold, L.; Rugină, D.; Ayvaz, H.; Socaciu, C. Antiproliferative and antioxidant properties of anthocyanin rich extracts from blueberry and blackcurrant juice. Int. J. Mol. Sci. 2015, 16, 2352–2365. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.Y.; Zhu, Y.M.; Li, C.Y.; Sui, Z.Q.; Min, W.H. Effect of blueberry anthocyanins malvidin and glycosides on the antioxidant properties in endothelial cells. Oxid. Med. Cell. Longev. 2016, 2016, 1591803. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.G.; Kim, H.L.; Kim, S.J.; Park, K.S. Fruit quality, anthocyanin and total phenolic contents, and antioxidant activities of 45 blueberry cultivars grown in Suwon, Korea. J. Zhejiang Univ. Sci. B 2013, 14, 793–799. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.; Thakali, K.M.; Jensen, G.S.; Wu, X. Phenolic acids of the two major blueberry species in the US Market and their antioxidant and anti-inflammatory activities. Plant Foods Hum. Nutr. 2015, 70, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Feng, J.; Huang, W.Y.; An, X.T. Composition of polyphenols and antioxidant activity of rabbiteye blueberry (Vaccinium ashei) in Nanjing. J. Agric. Food Chem. 2013, 61, 523–531. [Google Scholar] [CrossRef] [PubMed]
- Ben Lagha, A.; Dudonné, S.; Desjardins, Y.; Grenier, D. Wild blueberry (Vaccinium angustifolium Ait.) polyphenols target Fusobacterium nucleatum and the host inflammatory response: Potential innovative molecules for treating periodontal diseases. J. Agric. Food Chem. 2015, 63, 6999–7008. [Google Scholar] [CrossRef] [PubMed]
- McAnulty, L.S.; Nieman, D.C.; Dumke, C.L.; Shooter, L.A.; Henson, D.A.; Utter, A.C.; Milne, G.; McAnulty, S.R. Effect of blueberry ingestion on natural killer cell counts, oxidative stress, and inflammation prior to and after 2.5 h of running. Appl. Physiol. Nutr. Metabl. 2011, 36, 976–984. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.Y.; Liu, Y.M.; Wang, J.; Wang, X.N.; Li, C.Y. Anti-inflammatory effect of the blueberry anthocyanins malvidin-3-glucoside and malvidin-3-galactoside in endothelial cells. Molecules 2014, 19, 12827–12841. [Google Scholar] [CrossRef] [PubMed]
- Ben Lagha, A.; LeBel, G.; Grenier, D. Dual action of highbush blueberry proanthocyanidins on Aggregatibacter actinomycetemcomitans and the host inflammatory response. BMC Complem. Altern. Med. 2018, 18, 10. [Google Scholar] [CrossRef] [PubMed]
- Kalt, W.; McDonald, J.E.; Fillmore, S.A.; Tremblay, F. Blueberry effects on dark vision and recovery after photobleaching: Placebo-controlled crossover studies. J. Agric. Food Chem. 2014, 62, 11180–11189. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, D.; Liu, Y.; Wang, D.; Liu, J.; Ji, B. The protective effects of berry-derived anthocyanins against visible light-induced damage in human retinal pigment epithelial cells. J. Sci. Food Agri. 2015, 95, 936–944. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, D.; Hu, J.; Liu, G.; Chen, J.; Sun, L.; Jiang, Z.; Zhang, X.; Chen, Q.; Ji, B. Visible light-induced lipid peroxidation of unsaturated fatty acids in the retina and the inhibitory effects of blueberry polyphenols. J. Agric. Food Chem. 2015, 63, 9295–9305. [Google Scholar] [CrossRef] [PubMed]
- Srovnalova, A.; Svecarova, M.; Zapletalova, M.K.; Anzenbacher, P.; Bachleda, P.; Anzenbacherova, E.; Dvorak, Z. Effects of anthocyanidins and anthocyanins on the expression and catalytic activities of CYP2A6, CYP2B6, CYP2C9, and CYP3A4 in primary human hepatocytes and human liver microsomes. J. Agric. Food Chem. 2014, 62, 789–797. [Google Scholar] [CrossRef] [PubMed]
- Ferlemi, A.-V.; Mermigki, P.G.; Makri, O.E.; Anagnostopoulos, D.; Koulakiotis, N.S.; Margarity, M.; Tsarbopoulos, A.; Georgakopoulos, C.D.; Lamari, F.N. Cerebral area differential redox response of neonatal rats to selenite-induced oxidative stress and to concurrent administration of highbush blueberry leaf polyphenols. Neurochem. Res. 2015, 40, 2280–2292. [Google Scholar] [CrossRef] [PubMed]
- Zhan, W.; Liao, X.; Xie, R.J.; Tian, T.; Yu, L.; Liu, X.; Liu, J.; Li, P.; Han, B.; Yang, T.; et al. The effects of blueberry anthocyanins on histone acetylation in rat liver fibrosis. Oncotarget 2017, 8, 96761–96773. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Tan, D.; Shi, L.; Liu, X.; Zhang, Y.; Tong, C.; Song, D.; Hou, M. Blueberry anthocyanins-enriched extracts attenuate cyclophosphamide-induced cardiac injury. PLoS ONE 2015, 10, e0127813. [Google Scholar] [CrossRef] [PubMed]
- Khanal, R.C.; Howard, L.R.; Brownmiller, C.R.; Prior, R.L. Influence of extrusion processing on procyanidin composition and total anthocyanin contents of blueberry pomace. J. Food Sci. 2009, 74, H52–H58. [Google Scholar] [CrossRef] [PubMed]
- Louis, X.L.; Thandapilly, S.J.; Kalt, W.; Vinqvist-Tymchuk, M.; Aloud, B.M.; Raj, P.; Yu, L.; Le, H.; Netticadan, T. Blueberry polyphenols prevent cardiomyocyte death by preventing calpain activation and oxidative stress. Food Funct. 2014, 5, 1785–1794. [Google Scholar] [CrossRef] [PubMed]
- Bharat, D.; Cavalcanti, R.R.M.; Petersen, C.; Begaye, N.; Cutler, B.R.; Costa, M.M.A.; Ramos, R.K.L.G.; Ferreira, M.R.; Li, Y.; Bharath, L.P.; et al. Blueberry metabolites attenuate lipotoxicity-induced endothelial dysfunction. Mol. Nutr. Food Res. 2017, 62. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Gao, F.; Ji, B.; Wang, R.; Yang, J.; Liu, H.; Zhou, F. Anthocyanins-rich extract of wild Chinese blueberry protects glucolipotoxicity -induced INS832/13 β-cell against dysfunction and death. J. Food Sci. Technol. 2015, 52, 3022–3029. [Google Scholar] [CrossRef] [PubMed]
- Bell, L.; Lamport, D.J.; Butler, L.T.; Williams, C.M. A study of glycaemic effects following acute anthocyanin-rich blueberry supplementation in healthy young adults. Food Funct. 2017, 8, 3104–3110. [Google Scholar] [CrossRef] [PubMed]
- Bowtell, J.L.; Aboo-Bakkar, Z.; Conway, M.; Adlam, A.R.; Fulford, J. Enhanced task related brain activation and resting perfusion in healthy older adults after chronic blueberry supplementation. Appl. Physiol. Nutr. Metab. 2017, 42, 773–779. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.G.; Hamilton, D.A.; Joseph, J.A.; Shukitt-Hale, B. Dietary blueberry improves cognition among older adults in a randomized, double-blind, placebo-controlled trial. Eur. J. Nutr. 2017, 57, 1169–1180. [Google Scholar] [CrossRef] [PubMed]
- Krikorian, R.; Shidler, M.D.; Nash, T.A.; Kalt, W.; Vinqvist-Tymchuk, M.R.; Shukitt-Hale, B.; Joseph, J.A. Blueberry supplementation improves memory in older adults. J. Agric. Food Chem. 2010, 58, 3996–4000. [Google Scholar] [CrossRef] [PubMed]
- Xu, N.; Meng, H.; Liu, T.; Feng, Y.; Qi, Y.; Zhang, D.; Wang, H. Blueberry phenolics reduce gastrointestinal infection of patients with cerebral venous thrombosis by improving depressant-induced autoimmune disorder via miR-155-mediated brain-derived neurotrophic factor. Front. Pharmacol. 2017, 8, 853. [Google Scholar] [CrossRef] [PubMed]
- McNamara, R.K.; Kalt, W.; Shidler, M.D.; McDonald, J.; Summer, S.S.; Stein, A.L.; Stover, A.N.; Krikorian, R. Cognitive response to fish oil, blueberry, and combined supplementation in older adults with subjective cognitive impairment. Neurobiol. Aging 2018, 64, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.G.; Kim, B.; Soung do, Y.; Vance, T.; Lee, J.S.; Lee, J.Y.; Koo, S.I.; Kim, D.O.; Drissi, H.; Chun, O.K. Relationship between oxidative stress and bone mass in obesity and effects of berry supplementation on bone remodeling in obese male mice: An exploratory study. J. Med. Food 2015, 18, 476–482. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Liu, N.; Wu, Z.; Feng, Y.; Meng, X. Anti-tumor activity of a polysaccharide from blueberry. Molecules 2015, 20, 3841–3853. [Google Scholar] [CrossRef] [PubMed]
- Shi, D.; Xu, M.; Ren, M.; Pan, E.; Luo, C.; Zhang, W.; Tang, Q. Immunomodulatory effect of flavonoids of blueberry (Vaccinium corymbosum L.) leaves via the NF-κB signal pathway in LPS-stimulated RAW 264.7 cells. J. Immunol. Res. 2017, 2017, 5476903. [Google Scholar] [CrossRef] [PubMed]
- Del Bo’, C.; Deon, V.; Campolo, J.; Lanti, C.; Parolini, M.; Porrini, M.; Klimis-Zacas, D.; Riso, P. A serving of blueberry (V. corymbosum) acutely improves peripheral arterial dysfunction in young smokers and nonsmokers: Two randomized, controlled, crossover pilot studies. Food Funct. 2017, 8, 4108–4117. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.S.; Vissers, M.C.M.; Anderson, R.F.; Sreebhavan, S.; Bozonet, S.M.; Scheepens, A.; Melton, L.D. Bioavailable blueberry-derived phenolic acids at physiological concentrations enhance Nrf2-regulated antioxidant pesponses in human vascular endothelial cells. Mol. Nutr. Food Res. 2017, 62. [Google Scholar] [CrossRef]
- Wiseman, W.; Egan, J.M.; Slemmer, J.E.; Shaughnessy, K.S.; Ballem, K.; Gottschall-Pass, K.T.; Sweeney, M.I. Feeding blueberry diets inhibits angiotensin II-converting enzyme (ACE) activity in spontaneously hypertensive stroke-prone rats. Can. J. Physiol. Pharmacol. 2011, 89, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Bensalem, J.; Dudonné, S.; Etchamendy, N.; Pellay, H.; Amadieu, C.; Gaudout, D.; Dubreuil, S.; Paradis, M.E.; Pomerleau, S.; Capuron, L.; et al. Polyphenols from grape and blueberry improve episodic memory in healthy elderly with lower level of memory performance: A bicentric double-blind, randomized, placebo-controlled clinical study. J. Gerontol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.C.; Kim, D.Y.; Choi, B.Y. Antioxidative activity of blueberry leaf extract prevents high-fat diet-induced obesity in C57BL/6 mice. J. Cancer Prev. 2014, 19, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Farias, M.; Vasquez, K.; Ovalle-Marin, A.; Fuentes, F.; Parra, C.; Quitral, V.; Jimenez, P.; Garcia-Diaz, D.F. Chilean native fruit extracts inhibit inflammation linked to the pathogenic interaction between adipocytes and macrophages. J. Med. Food 2015, 18, 601–608. [Google Scholar] [CrossRef] [PubMed]
- Vendrame, S.; Daugherty, A.; Kristo, A.S.; Klimis-Zacas, D. Wild blueberry (Vaccinium angustifolium)-enriched diet improves dyslipidaemia and modulates the expression of genes related to lipid metabolism in obese Zucker rats. Br. J. Nutr. 2014, 111, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Aranaz, P.; Romo-Hualde, A.; Zabala, M.; Navarro-Herrera, D.; Ruiz de Galarreta, M.; Gil, A.G.; Martinez, J.A.; Milagro, F.I.; González-Navarro, C.J. Freeze-dried strawberry and blueberry attenuates diet-induced obesity and insulin resistance in rats by inhibiting adipogenesis and lipogenesis. Food Funct. 2017, 8, 3999–4013. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Zhou, Q.; Chen, X.Y.; Li, X.; Wang, Y.; Zhang, J.L. Comparison and screening of bioactive phenolic compounds in different blueberry cultivars: Evaluation of anti-oxidation and α-glucosidase inhibition effect. Food Res. Int. 2017, 100, 312–324. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.M.; Yoon, Y.; Yoon, H.; Park, H.M.; Song, S.; Yeum, K.J. Dietary anthocyanins against obesity and inflammation. Nutrients 2017, 9, 1089. [Google Scholar] [CrossRef] [PubMed]
- Vendrame, S.; Daugherty, A.; Kristo, A.S.; Riso, P.; Klimis-Zacas, D. Wild blueberry (Vaccinium angustifolium) consumption improves inflammatory status in the obese Zucker rat model of the metabolic syndrome. J. Nutr. Biochem. 2013, 24, 1508–1512. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Song, X.; Han, Y.; Zhou, F.; Zhang, D.; Ji, B.; Hu, J.; Lv, Y.; Cai, S.; Wei, Y.; et al. Identification of anthocyanin components of wild Chinese blueberries and amelioration of light-induced retinal damage in pigmented rabbit using whole berries. J. Agric. Food Chem. 2011, 59, 356–363. [Google Scholar] [CrossRef] [PubMed]
- Ishida, Y.; Takeshita, M.; Kataoka, H. Functional foods effective for hepatitis C: Identification of oligomeric proanthocyanidin and its action mechanism. World J. Hepatol. 2014, 6, 870–879. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Liu, X.; Luo, Y.; Guo, H.; Hu, X.; Chen, F. Evaluation of protective effect of freeze-dried strawberry, grape, and blueberry powder on acrylamide toxicity in mice. J. Food Sci. 2015, 80, H869–H874. [Google Scholar] [CrossRef] [PubMed]
- Gong, P.; Chen, F.X.; Wang, L.; Wang, J.; Jin, S.; Ma, Y.M. Protective effects of blueberries (Vaccinium corymbosum L.) extract against cadmium-induced hepatotoxicity in mice. Environ. Toxicol. Pharmacol. 2014, 37, 1015–1027. [Google Scholar] [CrossRef] [PubMed]
- Ozcelik, E.; Uslu, S.; Burukoglu, D.; Musmul, A. Chitosan and blueberry treatment induces arginase activity and inhibits nitric oxide production during acetaminophen-induced hepatotoxicity. Pharmacogn. Mag. 2014, 10, S217–S224. [Google Scholar] [CrossRef] [PubMed]
- Vendrame, S.; Zhao, A.; Merrow, T.; Klimis-Zacas, D. The effects of wild blueberry consumption on plasma markers and gene expression related to glucose metabolism in the obese Zucker rat. J. Med. Food 2015, 18, 619–624. [Google Scholar] [CrossRef] [PubMed]
- Cutler, B.R.; Gholami, S.; Chua, J.S.; Kuberan, B.; Anandh Babu, P.V. Blueberry metabolites restore cell surface glycosaminoglycans and attenuate endothelial inflammation in diabetic human aortic endothelial cells. Int. J. Cardiol. 2018, 261, 155–158. [Google Scholar] [CrossRef] [PubMed]
- Nemes-Nagy, E.; Szocs-Molnár, T.; Dunca, I.; Balogh-Sămărghiţan, V.; Hobai, S.; Morar, R.; Pusta, D.L.; Crăciun, E.C. Effect of a dietary supplement containing blueberry and sea buckthorn concentrate on antioxidant capacity in type 1 diabetic children. Acta Physiol. Hung. 2008, 95, 383–393. [Google Scholar] [CrossRef] [PubMed]
- Elks, C.M.; Terrebonne, J.D.; Ingram, D.K.; Stephens, J.M. Blueberries improve glucose tolerance without altering body composition in obese postmenopausal mice. Obesity 2015, 23, 573–580. [Google Scholar] [CrossRef] [PubMed]
- DeFuria, J.; Bennett, G.; Strissel, K.J.; Perfield, J.W.; Milbury, P.E.; Greenberg, A.S.; Obin, M.S. Dietary blueberry attenuates whole-body insulin resistance in high fat-fed mice by reducing adipocyte death and its inflammatory sequelae. J. Nutr. 2009, 139, 1510–1516. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Keirsey, K.I.; Kirkland, R.; Grunewald, Z.I.; Fischer, J.G.; de La Serre, C.B. Blueberry supplementation influences the gut microbiota, inflammation, and insulin resistance in high-fat-diet-fed rats. J. Nutr. 2018, 148, 209–219. [Google Scholar] [CrossRef] [PubMed]
- Çoban, J.; Doğan-Ekici, I.; Aydın, A.F.; Betül-Kalaz, E.; Doğru-Abbasoğlu, S.; Uysal, M. Blueberry treatment decreased d-galactose-induced oxidative stress and brain damage in rats. Metab. Brain Dis. 2015, 30, 793–802. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Wang, L.; Gu, L.; Zhao, W.; Su, H.; Cheng, X. Higher transcription levels in ascorbic acid biosynthetic and recycling genes were associated with higher ascorbic acid accumulation in blueberry. Food Chem. 2015, 188, 399–405. [Google Scholar] [CrossRef] [PubMed]
- Poulose, S.M.; Bielinski, D.F.; Carrihill-Knoll, K.L.; Rabin, B.M.; Shukitt-Hale, B. Protective effects of blueberry-and strawberry diets on neuronal stress following exposure to (56)Fe particles. Brain Res. 2014, 1593, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Oh, D.R.; Kim, Y.; Choi, E.J.; Jung, M.A.; Oh, K.N.; Hong, J.A.; Bae, D.; Kim, K.; Kang, H.; Kim, J.; et al. Antidepressant-like effects of Vaccinium bracteatum in chronic restraint stress mice: Functional actions and mechanism explorations. Am. J. Chin. Med. 2018, 46, 357–387. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.; Yang, H.P.; Pang, W.; Lu, H.; Hu, Y.D.; Li, J.; Lu, S.J.; Zhang, W.Q.; Jiang, Y.G. Cyanidin-3-O-galactoside and blueberry extracts supplementation improves spatial memory and regulates hippocampal ERK expression in senescence-accelerated mice. Biomed. Environ. Sci. 2014, 27, 186–196. [Google Scholar] [PubMed]
- Jeong, H.R.; Jo, Y.N.; Jeong, J.H.; Kim, H.J.; Kim, M.J.; Heo, H.J. Blueberry (Vaccinium virgatum) leaf extracts protect against Aβ-induced cytotoxicity and cognitive impairment. J. Med. Food 2013, 16, 968–976. [Google Scholar] [CrossRef] [PubMed]
- Fragua, V.; Lepoudère, A.; Leray, V.; Baron, C.; Araujo, J.A.; Nguyen, P.; Milgram, N.W. Effects of dietary supplementation with a mixed blueberry and grape extract on working memory in aged beagle dogs. J. Nutr. Sci. 2017, 6, e35. [Google Scholar] [CrossRef] [PubMed]
- Boespflug, E.L.; Eliassen, J.C.; Dudley, J.A.; Shidler, M.D.; Kalt, W.; Summer, S.S.; Stein, A.L.; Stover, A.N.; Krikorian, R. Enhanced neural activation with blueberry supplementation in mild cognitive impairment. Nutr. Neurosci. 2017, 21, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Johnson, S.L.; Liu, W.; DaSilva, N.A.; Meschwitz, S.; Dain, J.A.; Seeram, N.P. Evaluation of polyphenol anthocyanin-enriched extracts of blackberry, black raspberry, blueberry, cranberry, red raspberry, and strawberry for free radical scavenging, reactive carbonyl species trapping, anti-glycation, anti-β-amyloid aggregation, and microglial neuroprotective effects. Int. J. Mol. Sci. 2018, 19, E461. [Google Scholar] [CrossRef] [PubMed]
- Senevirathne, M.; Kim, S.H.; Jeon, Y.J. Protective effect of enzymatic hydrolysates from highbush blueberry (Vaccinium corymbosum L.) against hydrogen peroxide-induced oxidative damage in Chinese hamster lung fibroblast cell line. Nutr. Res. Pract. 2010, 4, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Lazarenko, O.P.; Blackburn, M.L.; Shankar, K.; Badger, T.M.; Ronis, M.J.J.; Chen, J.R. Feeding blueberry diets in early life prevent senescence of osteoblasts and bone loss in ovariectomized adult female rats. PLoS ONE 2011, 6, e24486. [Google Scholar] [CrossRef] [PubMed]
- Devareddy, L.; Hooshmand, S.; Collins, J.K.; Lucas, E.A.; Chai, S.C.; Arjmandi, B.H. Blueberry prevents bone loss in ovariectomized rat model of postmenopausal osteoporosis. J. Nutr. Biochem. 2008, 19, 694–699. [Google Scholar] [CrossRef] [PubMed]
- Nair, A.R.; Mariappan, N.; Stull, A.J.; Francis, J. Blueberry supplementation attenuates oxidative stress within monocytes and modulates immune cell levels in adults with metabolic syndrome: A randomized, double-blind, placebo-controlled trial. Food Funct. 2017, 8, 4118–4128. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.P.; Cheng, M.L.; Zhang, B.F.; Mu, M.; Zhou, M.Y.; Wu, J.; Li, C.X. Effect of blueberry on hepatic and immunological functions in mice. Hepatobil. Pancreat. Dis. Int. 2010, 9, 164–168. [Google Scholar]
- Lu, S.; Cheng, M.L.; Li, H.; Wu, J. Study of potential protective effects of blueberry on hepatic cytochrome P450 2E1 expression in rats with immune hepatic fibrosis. Zhonghua Gan Zang Bing Za Zhi 2012, 20, 497–502. (In Chinese) [Google Scholar] [PubMed]
- Lewis, E.D.; Ren, Z.; DeFuria, J.; Obin, M.S.; Meydani, S.N.; Wu, D. Dietary supplementation with blueberry partially restores T-cell-mediated function in high-fat-diet-induced obese mice. Br. J. Nutr. 2018, 119, 1393–1399. [Google Scholar] [CrossRef] [PubMed]
- Ahrén, I.L.; Xu, J.; Önning, G.; Olsson, C.; Ahrné, S.; Molin, G. Antihypertensive activity of blueberries fermented by Lactobacillus plantarum DSM 15313 and effects on the gut microbiota in healthy rats. Clin. Nutr. 2015, 34, 719–726. [Google Scholar] [CrossRef] [PubMed]
- McAnulty, L.S.; Collier, S.R.; Landram, M.J.; Whittaker, D.S.; Isaacs, S.E.; Klemka, J.M.; Cheek, S.L.; Arms, J.C.; McAnulty, S.R. Six weeks daily ingestion of whole blueberry powder increases natural killer cell counts and reduces arterial stiffness in sedentary males and females. Nutr. Res. 2014, 34, 577–584. [Google Scholar] [CrossRef] [PubMed]
- Johnson, S.A.; Figueroa, A.; Navaei, N.; Wong, A.; Kalfon, R.; Ormsbee, L.T.; Feresin, R.G.; Elam, M.L.; Hooshmand, S.; Payton, M.E.; et al. Daily blueberry consumption improves blood pressure and arterial stiffness in postmenopausal women with pre- and stage 1-hypertension: A randomized, double-blind, placebo-controlled clinical trial. J. Acad. Nutr. Diet. 2015, 115, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Lasekan, O. Exotic berries as a functional food. Curr. Opin. Clin. Nutr. Metab. Care 2014, 17, 589–595. [Google Scholar] [CrossRef] [PubMed]
- Whyte, A.R.; Cheng, N.; Fromentin, E.; Williams, C.M. A randomized, double-blinded, placebo-controlled study to compare the safety and efficacy of low dose enhanced wild blueberry powder and wild blueberry extract (ThinkBlue™) in maintenance of episodic and working memory in older adults. Nutrients 2018, 10, 660. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Tan, L.; Yang, H.P.; Pang, W.; Xu, T.; Jiang, Y.G. Changes of hippocampus proteomic profiles after blueberry extracts supplementation in APP/PS1 transgenic mice. Nutr. Neurosci. 2018, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.Y.; Guo, Q.-G.; Liu, Y.J.; Liu, H.C.; Wang, F.G.; Jia, C.G. Molecular cloning and functional analysis of a flavanone 3-hydroxylase gene from blueberry. J. Hortic. Sci. Biotechnol. 2017, 92, 57–64. [Google Scholar] [CrossRef]
- Baba, A.B.; Nivetha, R.; Chattopadhyay, I.; Nagini, S. Blueberry and malvidin inhibit cell cycle progression and induce mitochondrial-mediated apoptosis by abrogating the JAK/STAT-3 signalling pathway. Food Chem. Toxicol. 2017, 109, 534–543. [Google Scholar] [CrossRef] [PubMed]
- McGill, C.M.; Brown, T.J.; Cheng, Y.Y.; Fisher, L.N.; Shanmugavelandy, S.S.; Gustafson, S.J.; Dunlap, K.L.; Lila, M.A.; Kester, M.; Toran, P.T.; et al. Therapeutic effect of blueberry extracts for acute myeloid leukemia. Int. J. Biopharm. Sci. 2018, 1, 102. [Google Scholar] [PubMed]
- Adams, L.S.; Phung, S.; Yee, N.; Seeram, N.P.; Li, L.; Chen, S. Blueberry phytochemicals inhibit growth and metastatic potential of MDA-MB-231 breast cancer cells through modulation of the phosphatidylinositol 3-kinase pathway. Cancer Res. 2010, 70, 3594–3605. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Li, S.; Jia, Y.; Wang, L. Blueberry anthocyanins induce G2/M cell cycle arrest and apoptosis of oral cancer KB cells through down-regulation methylation of p53. Yi Chuan 2014, 36, 566–573. (In Chinese) [Google Scholar]
- Wu, T.; Gao, Y.; Guo, X.; Zhang, M.; Gong, L. Blackberry and blueberry anthocyanin supplementation counteract high-fat-diet-induced obesity by alleviating oxidative stress and inflammation and accelerating energy expenditure. Oxid. Med. Cell Longev. 2018, 2018, 4051232. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Park, H.J.; Kang, S.N.; Jang, S.H.; Lee, S.J.; Ko, Y.G.; Kim, G.S.; Cho, J.H. Blueberry peel extracts inhibit adipogenesis in 3T3-L1 cells and reduce high-fat diet-induced obesity. PLoS ONE 2013, 8, e69925. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Sun, H.; Pei, J.; Dong, Y.; Wang, F.; Chen, H.; Sun, Y.; Wang, N.; Li, H.; Li, Y. De novo sequencing and comparative analysis of the blueberry transcriptome to discover putative genes related to antioxidants. Gene 2012, 511, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.Y.; Wu, H.; Li, D.J.; Song, J.F.; Xiao, Y.D.; Liu, C.Q.; Zhou, J.Z.; Sui, Z.Q. Protective effects of blueberry anthocyanins against H2O2-induced oxidative injuries in human retinal pigment epithelial cells. J. Agric. Food Chem. 2018, 66, 1638–1648. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Yan, Z.; Li, D.; Ma, Y.; Zhou, J.; Sui, Z. Antioxidant and anti-inflammatory effects of blueberry anthocyanins on high glucose-induced human retinal capillary endothelial cells. Oxid. Med. Cell Longev. 2018, 201, 1862462. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Wu, Y.; Long, C.; He, P.; Gu, J.; Yang, L.; Liang, Y.; Wang, Y. Anthocyanins isolated from blueberry ameliorates CCl4 induced liver fibrosis by modulation of oxidative stress, inflammation and stellate cell activation in mice. Food Chem. Toxicol. 2018, 120, 491–499. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.J.; Cheng, M.L.; Zhou, M.Y.; Zhao, X.K. The effect of p-JAK1/p-STAT3 signaling way in intervening NAFLD by blueberry probiotic serum. J. Sichuan Univ. (Med. Sci. Ed.) 2017, 48, 203–209. (In Chinese) [Google Scholar]
- Nair, A.R.; Elks, C.M.; Vila, J.; Del Piero, F.; Paulsen, D.B.; Francis, J. A blueberry-enriched diet improves renal function and reduces oxidative stress in metabolic syndrome animals: Potential mechanism of TLR4-MAPK signaling pathway. PLoS ONE 2014, 9, e111976. [Google Scholar] [CrossRef] [PubMed]
- Koupý, D.; Kotolová, H.; Kučerová, J. Effectiveness of phytotherapy in supportive treatment of type 2 diabetes mellitus billberry (Vaccinium myrtillus). Ceska. Slov. Farm. 2015, 64, 3–6. [Google Scholar] [PubMed]
- Milbury, P.E.; Kalt, W. Xenobiotic metabolism and berry flavonoid transport across the blood–brain barrier. J. Agric. Food Chem. 2010, 58, 3950–3956. [Google Scholar] [CrossRef] [PubMed]
- Williams, C.M.; El Mohsen, M.A.; Vauzour, D.; Rendeiro, C.; Butler, L.T.; Ellis, J.A.; Whiteman, M.; Spencer, J.P. Blueberry-induced changes in spatial working memory correlate with changes in hippocampal CREB phosphorylation and brain-derived neurotrophic factor (BDNF) levels. Free Rad. Biol. Med. 2008, 45, 295–305. [Google Scholar] [CrossRef] [PubMed]
- Cásedas, G.; González-Burgos, E.; Smith, C.; López, V.; Gómez-Serranillos, M.P. Regulation of redox status in neuronal SH-SY5Y cells by blueberry (Vaccinium myrtillus L.) juice, cranberry (Vaccinium macrocarpon A.) juice and cyanidin. Food Chem. Toxicol. 2018, 118, 572–580. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Wu, L.; Li, B.; Meng, X.; Dai, H.; Zheng, Y.; Fu, J. Cyanidin-3-O-glucoside attenuates acute lung injury in sepsis rats. J. Surg. Res. 2015, 199, 592–600. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.; Li, Y.; Yang, J.; Pu, X.; Du, J.; Yang, X.; Yang, T.; Yang, S. Therapeutic role of functional components in alliums for preventive chronic disease in human being. Evid.-Based Complement. Altern. Med. 2017, 2017, 9402849. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.W.; Pu, X.Y.; Yang, J.Z.; Du, J.; Yang, X.; Li, X.; Li, L.; Zhou, Y.; Yang, T. Preventive and therapeutic role of functional ingredients of barley grass for chronic diseases in human beings. Oxid. Med. Cell. Longev. 2018, 2018, 3232080. [Google Scholar] [CrossRef] [PubMed]
- Cocetta, G.; Rossoni, M.; Gardana, C.; Mignani, I.; Ferrante, A.; Spinardi, A. Methyl jasmonate affects phenolic metabolism and gene expression in blueberry (Vaccinium corymbosum). Physiol. Plant. 2015, 153, 269–283. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wang, D.; Zhang, D.; Lv, Y.; Wei, Y.; Wu, W.; Zhou, F.; Tang, M.; Mao, T.; Li, M.; et al. Inhibitory effect of blueberry polyphenolic compounds on oleic acid-induced hepatic steatosis in vitro. J. Agric. Food Chem. 2011, 59, 12254–12263. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Yao, L.; He, X.; Wang, L.; Li, M.; Yang, Y.; Wan, C. Hypoglycemic activity and constituents analysis of blueberry (Vaccinium corymbosum) fruit extracts. Diabetes Metab. Syndr. Obes. 2018, 11, 357–366. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.H.; Lucius, A.; Meyer, T.; de Mejia, E.G. Cultivar evaluation and effect of fermentation on antioxidant capacity and in vitro inhibition of α-amylase and α-glucosidase by highbush blueberry (Vaccinium corombosum). J. Agric. Food Chem. 2011, 59, 8923–8930. [Google Scholar] [CrossRef] [PubMed]
- Cuevas, A.; Saavedra, N.; Salazar, L.A.; Abdalla, D.S. Modulation of immune function by polyphenols: Possible contribution of epigenetic factors. Nutrients 2013, 5, 2314–2332. [Google Scholar] [CrossRef] [PubMed]
- Uysal, U.; Seremet, S.; Lamping, J.W.; Adams, J.M.; Liu, D.Y.; Swerdlow, R.H.; Aires, D.J. Consumption of polyphenol plants may slow aging and associated diseases. Curr. Pharm. Des. 2013, 19, 6094–6111. [Google Scholar] [CrossRef] [PubMed]
- Nair, A.R.; Masson, G.S.; Ebenezer, P.J.; Del Piero, F.; Francis, J. Role of TLR4 in lipopolysaccharide -induced acute kidney injury: Protection by blueberry. Free Rad. Biol. Med. 2014, 71, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Bensalem, J.; Dudonné, S.; Gaudout, D.; Servant, L.; Calon, F.; Desjardins, Y.; Layé, S.; Lafenetre, P.; Pallet, V. Polyphenol-rich extract from grape and blueberry attenuates cognitive decline and improves neuronal function in aged mice. J. Nutr. Sci. 2018, 7, e19. [Google Scholar] [CrossRef] [PubMed]
- Maulik, M.; Mitra, S.; Hunter, S.; Hunstiger, M.; Oliver, S.R.; Bult-Ito, A.; Taylor, B.E. Sir-2.1 mediated attenuation of α-synuclein expression by Alaskan bog blueberry polyphenols in a transgenic model of Caenorhabditis elegans. Sci. Rep. 2018, 8, 10216. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.M.; Soe, K.H.; Lee, T.H.; Kim, I.S.; Lee, Y.M.; Lim, B.O. Cognitive improving effects by highbush blueberry (Vaccinium crymbosum L.) vinegar on scopolamine-induced amnesia mice model. J. Agric. Food Chem. 2017, 66, 99–107. [Google Scholar] [CrossRef] [PubMed]
| Chronic Disease | Functional Components | References |
|---|---|---|
| Anticancer | Anthocyanins, phenolic acids, pyruvic acid, pterostilbene | [15,25,26,27,28,29,30,33,34] |
| Anti-obesity | Anthocyanins, polyphenols | [35,36] |
| Prevent degenerative diseases | Anthocyanins, chlorogenic acid, polyphenols | [37,38,39,40,41] |
| Anti-inflammation | Anthocyanins, phenolic acids, flavonoids | [42,43,44,45] |
| Protective vision | Anthocyanins, polyphenols | [46,47,48] |
| Protective liver | Anthocyanins, polyphenols | [28,49,50,51] |
| Prevent heart diseases | Anthocyanins, procyanidin, polyphenols | [52,53,54] |
| Antidiabetes | Anthocyanins, polyphenols | [35,55,56,57] |
| Improve brain | Anthocyanins, phenolics | [58,59,60,61,62] |
| Protective lung | Anthocyanins | [2] |
| Strong bones | Anthocyanins | [63] |
| Enhance immunity | Polysaccharide, polyphenols | [64,65] |
| Prevent cardiovascular diseases | Phenolic acids, flavonoid | [66,67,68] |
| Improve cognitive decline | Polyphenols | [69] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, L.; Sun, Z.; Zeng, Y.; Luo, M.; Yang, J. Molecular Mechanism and Health Role of Functional Ingredients in Blueberry for Chronic Disease in Human Beings. Int. J. Mol. Sci. 2018, 19, 2785. https://doi.org/10.3390/ijms19092785
Ma L, Sun Z, Zeng Y, Luo M, Yang J. Molecular Mechanism and Health Role of Functional Ingredients in Blueberry for Chronic Disease in Human Beings. International Journal of Molecular Sciences. 2018; 19(9):2785. https://doi.org/10.3390/ijms19092785
Chicago/Turabian StyleMa, Luyao, Zhenghai Sun, Yawen Zeng, Mingcan Luo, and Jiazhen Yang. 2018. "Molecular Mechanism and Health Role of Functional Ingredients in Blueberry for Chronic Disease in Human Beings" International Journal of Molecular Sciences 19, no. 9: 2785. https://doi.org/10.3390/ijms19092785
APA StyleMa, L., Sun, Z., Zeng, Y., Luo, M., & Yang, J. (2018). Molecular Mechanism and Health Role of Functional Ingredients in Blueberry for Chronic Disease in Human Beings. International Journal of Molecular Sciences, 19(9), 2785. https://doi.org/10.3390/ijms19092785







