The Reprimo Gene Family: A Novel Gene Lineage in Gastric Cancer with Tumor Suppressive Properties
Abstract
1. Introduction
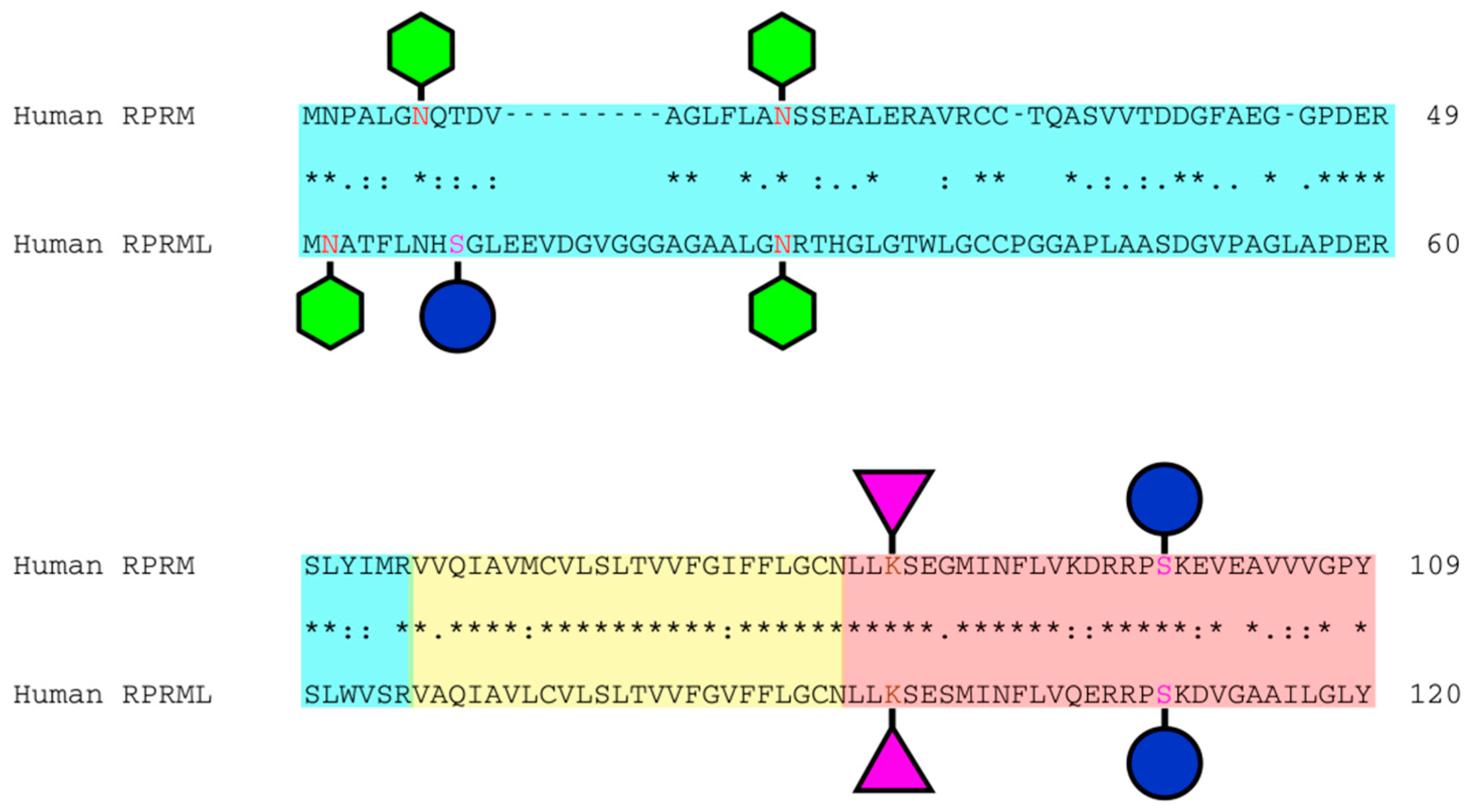
2. Structure and Genomic Location of the RPRM Gene Family
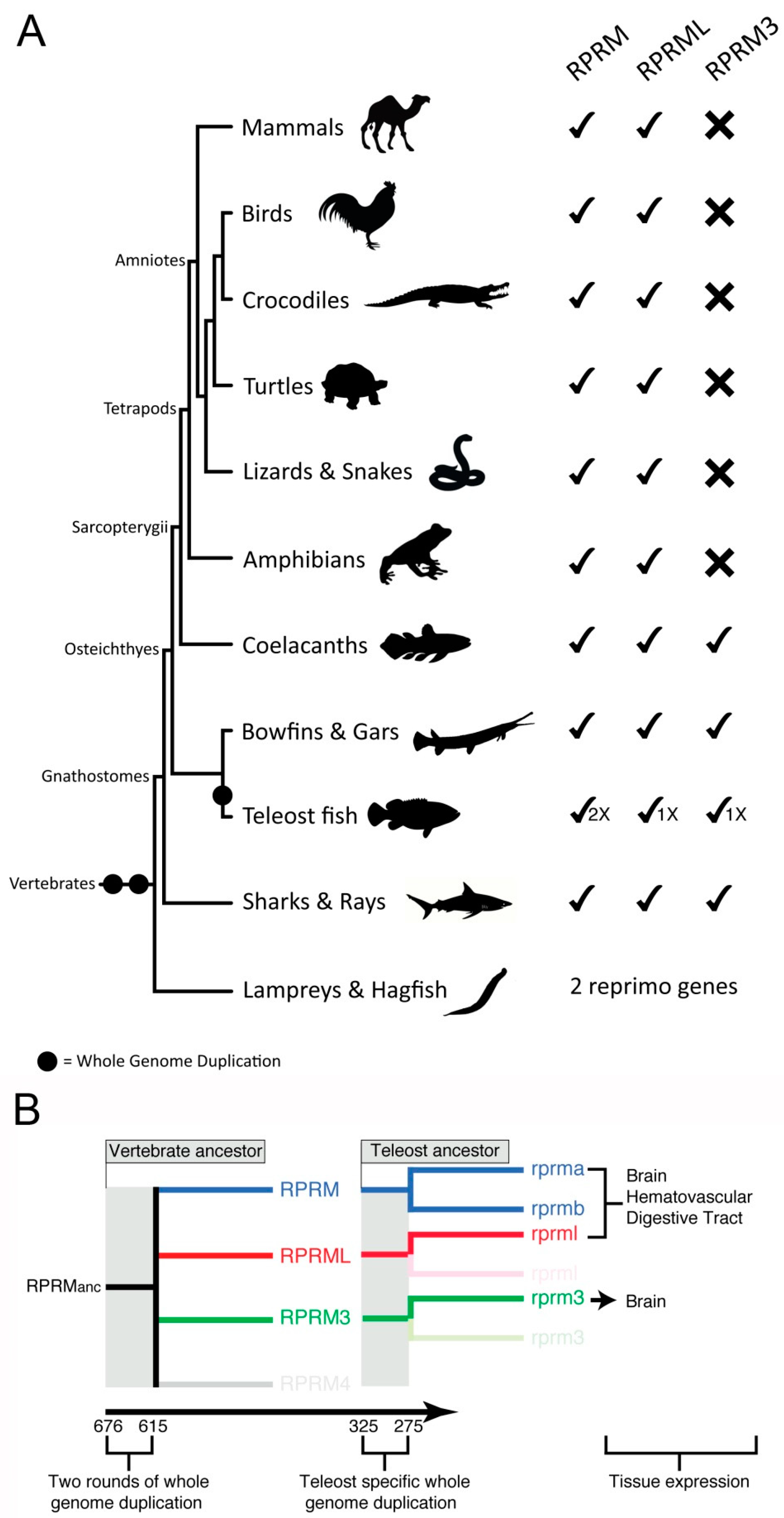
3. Evolution of the RPRM Gene Family
4. Developmental Expression Patterns of RPRM Gene Family
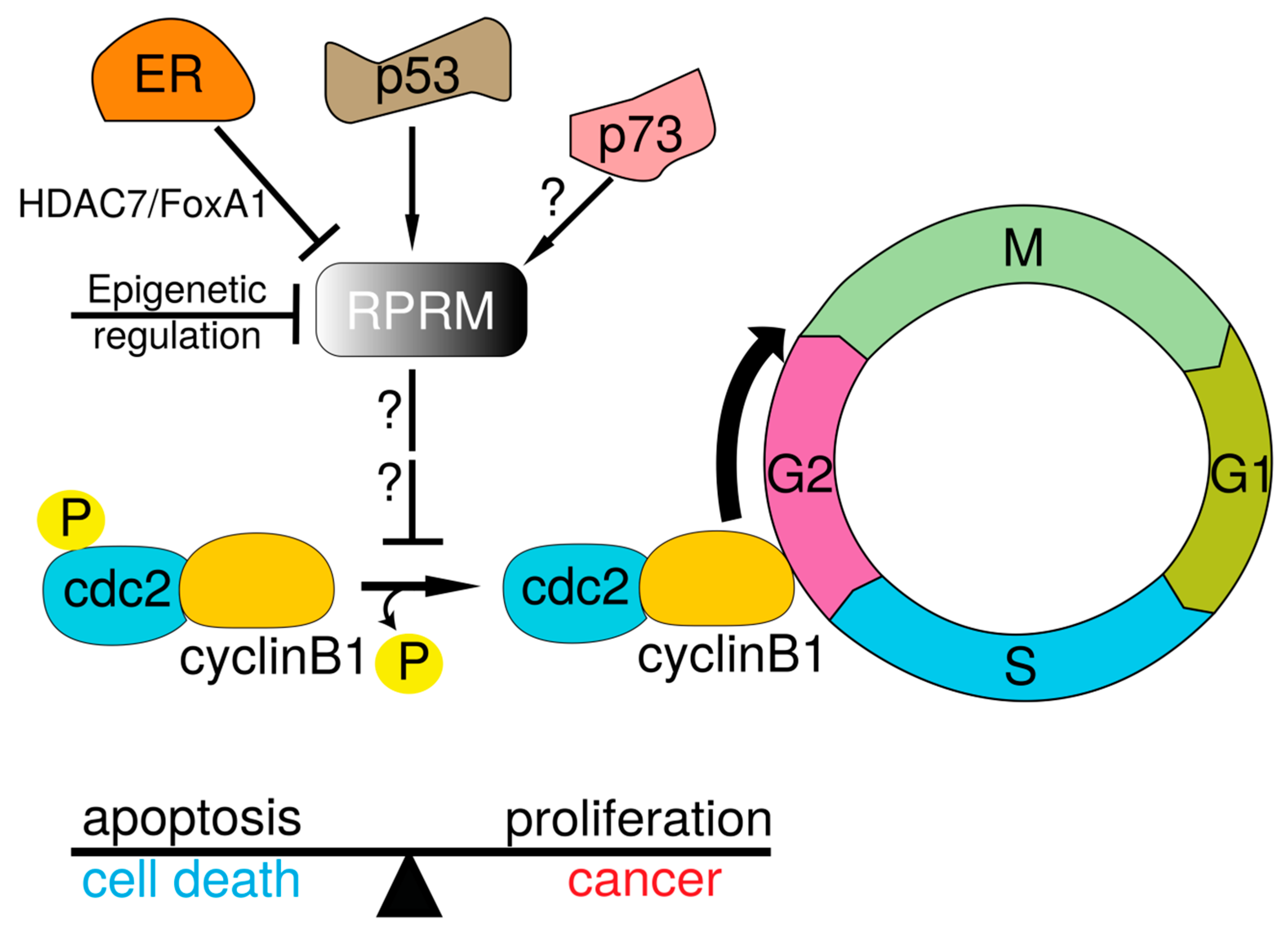
5. Functional Diversification of the RPRM Gene Family
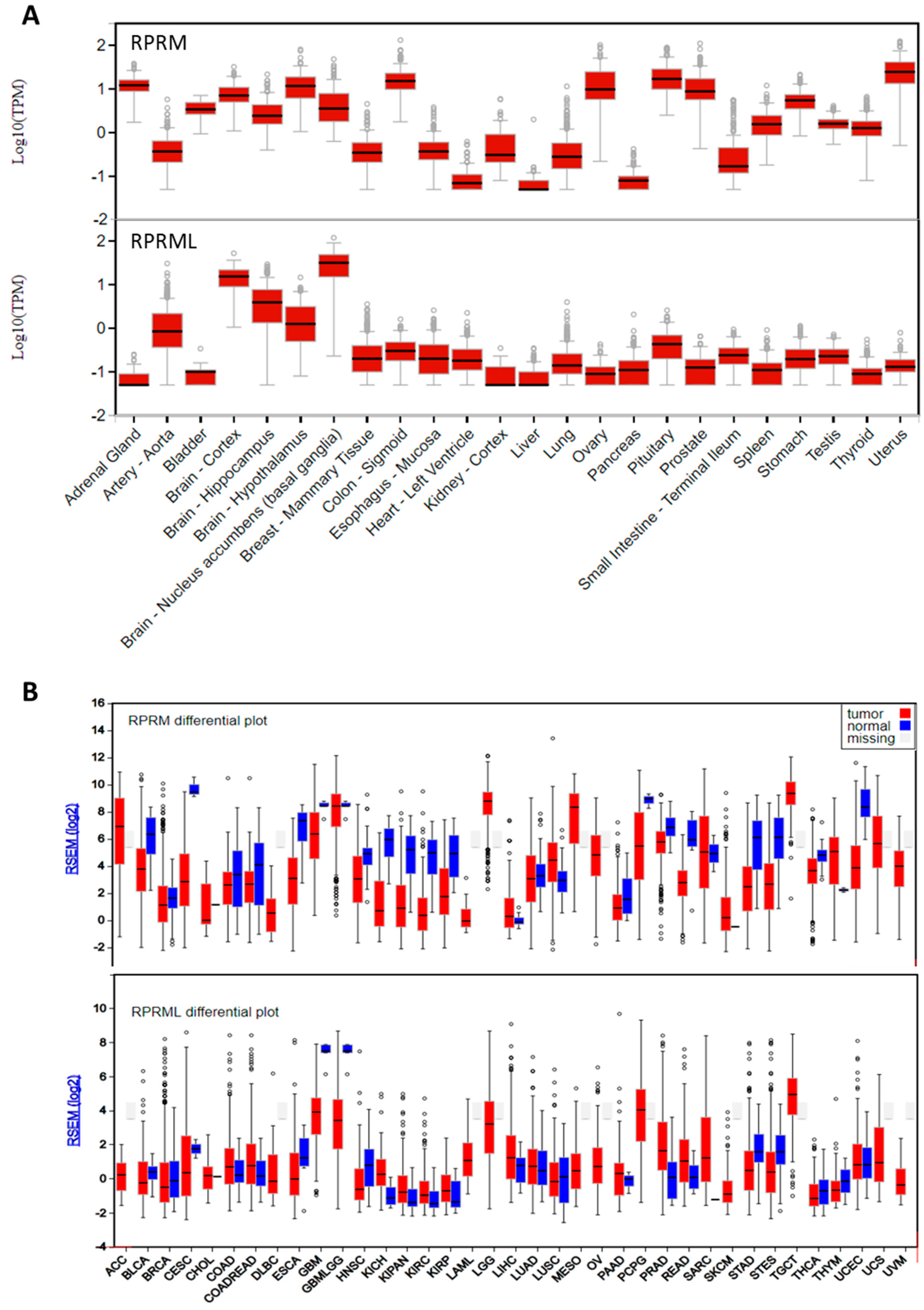
6. Role of the RPRM Gene Family in Human Carcinogenesis
7. Methylated RPRM Cell-Free DNA as a Potential Non-Invasive Biomarker in Gastric Cancer
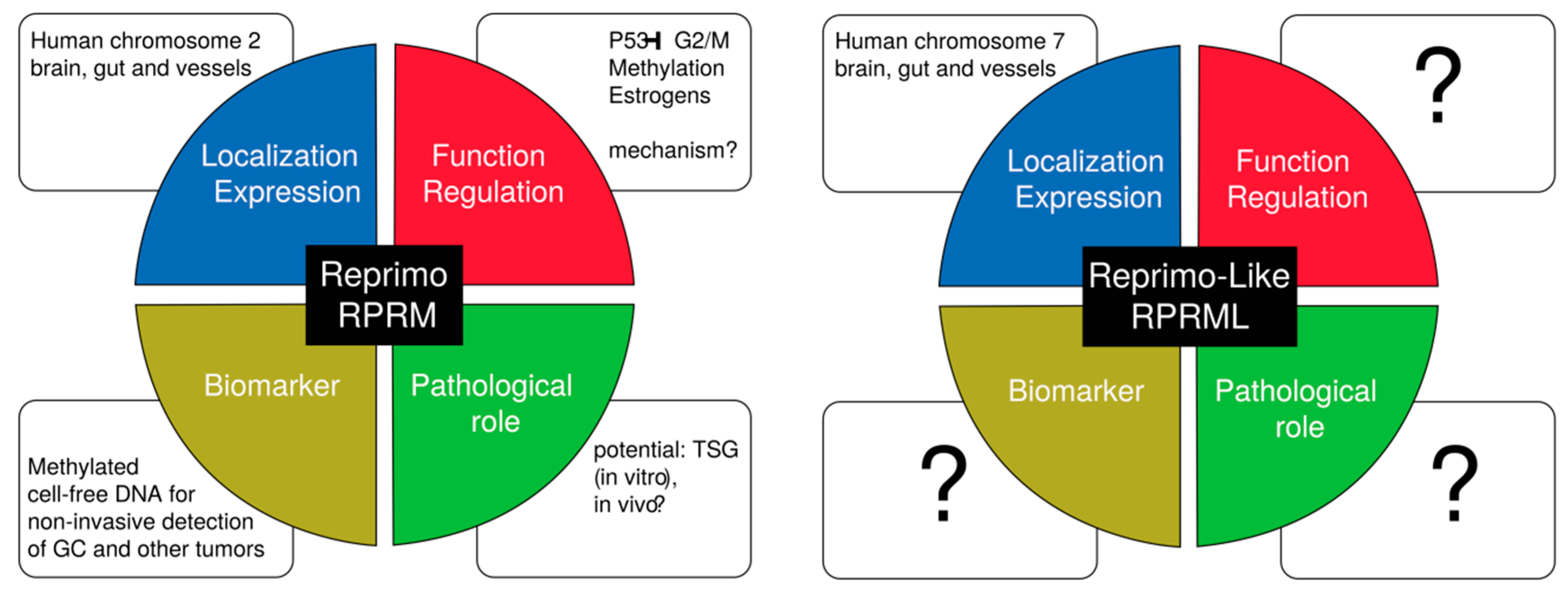
8. Unanswered Questions and the RPRM Gene Family
9. Concluding Remarks
Acknowledgments
Conflicts of Interest
References
- Wichmann, I.A.; Zavala, K.; Hoffmann, F.G.; Vandewege, M.W.; Corvalan, A.H.; Amigo, J.D.; Owen, G.I.; Opazo, J.C. Evolutionary history of the reprimo tumor suppressor gene family in vertebrates with a description of a new reprimo gene lineage. Gene 2016, 591, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Figueroa, R.J.; Carrasco-Avino, G.; Wichmann, I.A.; Lange, M.; Owen, G.I.; Siekmann, A.F.; Corvalan, A.H.; Opazo, J.C.; Amigo, J.D. Reprimo tissue-specific expression pattern is conserved between zebrafish and human. PLoS ONE 2017, 12, e0178274. [Google Scholar] [CrossRef] [PubMed]
- Saavedra, K.; Valbuena, J.; Olivares, W.; Marchant, M.J.; Rodriguez, A.; Torres-Estay, V.; Carrasco-Avino, G.; Guzman, L.; Aguayo, F.; Roa, J.C.; et al. Loss of expression of reprimo, a p53-induced cell cycle arrest gene, correlates with invasive stage of tumor progression and p73 expression in gastric cancer. PLoS ONE 2015, 10, e0125834. [Google Scholar] [CrossRef] [PubMed]
- Ohki, R.; Nemoto, J.; Murasawa, H.; Oda, E.; Inazawa, J.; Tanaka, N.; Taniguchi, T. Reprimo, a new candidate mediator of the p53-mediated cell cycle arrest at the G2 phase. J. Biol. Chem. 2000, 275, 22627–22630. [Google Scholar] [CrossRef] [PubMed]
- Louhichi, A.; Fourati, A.; Rebai, A. Igd: A resource for intronless genes in the human genome. Gene 2011, 488, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Huret, J.L.; Ahmad, M.; Arsaban, M.; Bernheim, A.; Cigna, J.; Desangles, F.; Guignard, J.C.; Jacquemot-Perbal, M.C.; Labarussias, M.; Leberre, V.; et al. Atlas of genetics and cytogenetics in oncology and haematology in 2013. Nucleic Acids Res. 2013, 41, D920–D924. [Google Scholar] [CrossRef] [PubMed]
- Grzybowska, E.A. Human intronless genes: Functional groups, associated diseases, evolution, and mRNA processing in absence of splicing. Biochem. Biophys. Res. Commun. 2012, 424, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Holland, P.W.; Garcia-Fernandez, J.; Williams, N.A.; Sidow, A. Gene duplications and the origins of vertebrate development. Development 1994, 1994, 125–133. [Google Scholar]
- Dehal, P.; Boore, J.L. Two rounds of whole genome duplication in the ancestral vertebrate. PLoS Boil. 2005, 3, e314. [Google Scholar] [CrossRef] [PubMed]
- Putnam, N.H.; Butts, T.; Ferrier, D.E.; Furlong, R.F.; Hellsten, U.; Kawashima, T.; Robinson-Rechavi, M.; Shoguchi, E.; Terry, A.; Yu, J.K.; et al. The amphioxus genome and the evolution of the chordate karyotype. Nature 2008, 453, 1064–1071. [Google Scholar] [CrossRef] [PubMed]
- Qiu, H.; Hildebrand, F.; Kuraku, S.; Meyer, A. Unresolved orthology and peculiar coding sequence properties of lamprey genes: The KCNA gene family as test case. BMC Genom. 2011, 12, 325. [Google Scholar] [CrossRef] [PubMed]
- Kuraku, S. Impact of asymmetric gene repertoire between cyclostomes and gnathostomes. Semin. Cell Dev. Boil. 2013, 24, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.J.; Kuraku, S.; Holt, C.; Sauka-Spengler, T.; Jiang, N.; Campbell, M.S.; Yandell, M.D.; Manousaki, T.; Meyer, A.; Bloom, O.E.; et al. Sequencing of the sea lamprey (Petromyzon marinus) genome provides insights into vertebrate evolution. Nat. Genet. 2013, 45, 415–421. [Google Scholar] [CrossRef] [PubMed]
- Meyer, A.; Van de Peer, Y. From 2R to 3R: Evidence for a fish-specific genome duplication (FSGD). BioEssays 2005, 27, 937–945. [Google Scholar] [CrossRef] [PubMed]
- Kasahara, M. The 2R hypothesis: An update. Curr. Opin. Immunol. 2007, 19, 547–552. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez-Lovera, C.; Vazquez-Rios, A.J.; Guerra-Varela, J.; Sánchez, L.; de la Fuente, M. The potential of zebrafish as a model organism for improving the translation of genetic anticancer nanomedicines. Gene 2017, 8, 349. [Google Scholar] [CrossRef] [PubMed]
- Vogel, G. Genomics: Sanger will sequence zebrafish genome. Science 2000, 290, 1671. [Google Scholar] [CrossRef] [PubMed]
- Staton, C.A.; Reed, M.W.; Brown, N.J. A critical analysis of current in vitro and in vivo angiogenesis assays. Int. J. Exp. Pathol. 2009, 90, 195–221. [Google Scholar] [CrossRef] [PubMed]
- MacRae, C.A.; Peterson, R.T. Zebrafish as tools for drug discovery. Nat. Rev. Drug Discov. 2015, 14, 721–731. [Google Scholar] [CrossRef] [PubMed]
- Wallace, K.N.; Akhter, S.; Smith, E.M.; Lorent, K.; Pack, M. Intestinal growth and differentiation in zebrafish. Mech. Dev. 2005, 122, 157–173. [Google Scholar] [CrossRef] [PubMed]
- Stanic, K.; Quiroz, A.; Wichmann, I.; Corvalan, A.H.; Owen, G.I.; Opazo, J.C.; Lemus, C.; Concha, M.; Amigo, J.D. Expression of RPRM/rprm in the olfactory system of embryonic zebrafish (Danio rerio). Front. Neuroanat. 2018. [Google Scholar] [CrossRef] [PubMed]
- Ming, G.L.; Song, H. Adult neurogenesis in the mammalian brain: Significant answers and significant questions. Neuron 2011, 70, 687–702. [Google Scholar] [CrossRef] [PubMed]
- Van de Peer, Y.; Maere, S.; Meyer, A. The evolutionary significance of ancient genome duplications. Nat. Rev. Genet. 2009, 10, 725–732. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, F.G.; Opazo, J.C.; Storz, J.F. Whole-genome duplications spurred the functional diversification of the globin gene superfamily in vertebrates. Mol. Biol. Evol. 2012, 29, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, F.G.; Opazo, J.C.; Storz, J.F. Differential loss and retention of cytoglobin, myoglobin, and globin-e during the radiation of vertebrates. Genome Biol. Evol. 2011, 3, 588–600. [Google Scholar] [CrossRef] [PubMed]
- Storz, J.F.; Opazo, J.C.; Hoffmann, F.G. Gene duplication, genome duplication, and the functional diversification of vertebrate globins. Mol. Phylogenet. Evol. 2013, 66, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Braasch, I.; Gehrke, A.R.; Smith, J.J. The spotted gar genome illuminates vertebrate evolution and facilitates human-teleost comparisons. Nat. Genet. 2016, 48, 427–437. [Google Scholar] [CrossRef] [PubMed]
- GTEx Consortium, T. The genotype-tissue expression (GTEx) project. Nat. Genet. 2013, 45, 580–585. [Google Scholar]
- Shabalina, S.A.; Ogurtsov, A.Y.; Spiridonov, A.N.; Novichkov, P.S.; Spiridonov, N.A.; Koonin, E.V. Distinct patterns of expression and evolution of intronless and intron-containing mammalian genes. Mol. Biol. Evol. 2010, 27, 1745–1749. [Google Scholar] [CrossRef] [PubMed]
- Broad Institute TCGA Genome Data Analysis Center. Analysis-Ready Standardized TCGA Data from Broad GDAC Firehose 2016_01_28 Run; Broad Institute of MIT and Harvard: Cambridge, MA, USA, 2016. [Google Scholar]
- Ooki, A.; Yamashita, K.; Yamaguchi, K.; Mondal, A.; Nishimiya, H.; Watanabe, M. DNA damage-inducible gene, reprimo functions as a tumor-suppressor and is suppressed by promoter methylation in gastric cancer. Mol. Cancer Res. 2013, 11, 1362–1374. [Google Scholar] [CrossRef] [PubMed]
- Lai, J.; Wang, H.; Luo, Q.; Huang, S.; Lin, S.; Zheng, Y.; Chen, Q. The relationship between DNA methylation and reprimo gene expression in gastric cancer cells. Oncotarget 2017, 8, 108610–108623. [Google Scholar] [CrossRef] [PubMed]
- Morris, M.R.; Ricketts, C.; Gentle, D.; Abdulrahman, M.; Clarke, N.; Brown, M.; Kishida, T.; Yao, M.; Latif, F.; Maher, E.R. Identification of candidate tumour suppressor genes frequently methylated in renal cell carcinoma. Oncogene 2010, 29, 2104–2117. [Google Scholar] [PubMed]
- Xu, M.; Knox, A.J.; Michaelis, K.A.; Kiseljak-Vassiliades, K.; Kleinschmidt-DeMasters, B.K.; Lillehei, K.O.; Wierman, M.E. Reprimo (RPRM) is a novel tumor suppressor in pituitary tumors and regulates survival, proliferation, and tumorigenicity. Endocrinology 2012, 153, 2963–2973. [Google Scholar] [CrossRef] [PubMed]
- Buchegger, K.; Ili, C.; Riquelme, I.; Letelier, P.; Corvalan, A.H.; Brebi, P.; Huang, T.H.; Roa, J.C. Reprimo as a modulator of cell migration and invasion in the mda-mb-231 breast cancer cell line. Biol. Res. 2016, 49, 5. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Zhu, Y.; Yang, G.; Gong, L.; Wang, B.; Liu, H. Loss of reprimo and s100a2 expression in human gastric adenocarcinoma. Diagn. Cytopathol. 2011, 39, 752–757. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, J.P.; Sato, F.; Jin, Z.; Greenwald, B.D.; Ito, T.; Mori, Y.; Paun, B.C.; Kan, T.; Cheng, Y.; Wang, S.; et al. Reprimo methylation is a potential biomarker of barrett’s-associated esophageal neoplastic progression. Clin. Cancer Res. 2006, 12, 6637–6642. [Google Scholar] [PubMed]
- Nakazato, T.; Suzuki, Y.; Tanaka, R.; Abe, N.; Masaki, T.; Mori, T.; Ohkura, Y.; Sugiyama, M. Effect of reprimo down-regulation on malignant transformation of intraductal papillary mucinous neoplasm. Pancreas 2018, 47, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Sato, N.; Fukushima, N.; Hruban, R.H.; Goggins, M. CPG island methylation profile of pancreatic intraepithelial neoplasia. Mod. Pathol. 2008, 21, 238–244. [Google Scholar] [PubMed]
- Chang, W.L.; Jackson, C.; Riel, S.; Cooper, H.S.; Devarajan, K.; Hensley, H.H.; Zhou, Y.; Vanderveer, L.A.; Nguyen, M.T.; Clapper, M.L. Differential preventive activity of sulindac and atorvastatin in Apc+/Min-FCCCmice with or without colorectal adenomas. Gut 2017. [Google Scholar] [CrossRef] [PubMed]
- Beasley, W.D.; Beynon, J.; Jenkins, G.J.; Parry, J.M. Reprimo 824 G>C and p53R2 4696 C>G single nucleotide polymorphisms and colorectal cancer: A case-control disease association study. Int. J. Colorectal Dis. 2008, 23, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Buchegger, K.; Riquelme, I.; Viscarra, T.; Ili, C.; Brebi, P.; Huang, T.H.; Roa, J.C. Reprimo, a potential p53-dependent tumor suppressor gene, is frequently hypermethylated in estrogen receptor alpha-positive breast cancer. Int. J. Mol. Sci. 2017, 18, 1525. [Google Scholar] [CrossRef] [PubMed]
- Soon, P.S.; Gill, A.J.; Benn, D.E.; Clarkson, A.; Robinson, B.G.; McDonald, K.L.; Sidhu, S.B. Microarray gene expression and immunohistochemistry analyses of adrenocortical tumors identify IGF2 and Ki-67 as useful in differentiating carcinomas from adenomas. Endocr. Relat. Cancer 2009, 16, 573–583. [Google Scholar] [CrossRef] [PubMed]
- Zohrabian, V.M.; Nandu, H.; Gulati, N.; Khitrov, G.; Zhao, C.; Mohan, A.; Demattia, J.; Braun, A.; Das, K.; Murali, R.; et al. Gene expression profiling of metastatic brain cancer. Oncol. Rep. 2007, 18, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, B.E.; Meissner, A.; Lander, E.S. The mammalian epigenome. Cell 2007, 128, 669–681. [Google Scholar] [CrossRef] [PubMed]
- Berdasco, M.; Esteller, M. Aberrant epigenetic landscape in cancer: How cellular identity goes awry. Dev. Cell 2010, 19, 698–711. [Google Scholar] [CrossRef] [PubMed]
- Esteller, M.; Hamilton, S.R.; Burger, P.C.; Baylin, S.B.; Herman, J.G. Inactivation of the DNA repair gene o6-methylguanine-DNA methyltransferase by promoter hypermethylation is a common event in primary human neoplasia. Cancer Res. 1999, 59, 793–797. [Google Scholar] [PubMed]
- Bernal, C.; Aguayo, F.R.; Villarroel, C.; Vargas, M.; Diaz, I.; Ossandón, F.J.; Santibáñez, E.; Palma, M.; Aravena, E.; Barrientos, C.; et al. Reprimo as a potential biomarker for early detection in gastric cancer. Clin. Cancer Res. 2008, 14, 6264–6269. [Google Scholar] [CrossRef] [PubMed]
- Malik, S.; Jiang, S.; Garee, J.P.; Verdin, E.; Lee, A.V.; O’Malley, B.W.; Zhang, M.; Belaguli, N.S.; Oesterreich, S. Histone deacetylase 7 and foxa1 in estrogen-mediated repression of rprm. Mol. Cell. Biol. 2010, 30, 399–412. [Google Scholar] [CrossRef] [PubMed]
- Cerda-Opazo, P.; Valenzuela-Valderrama, M.; Wichmann, I.; Rodriguez, A.; Contreras-Reyes, D.; Fernandez, E.; Carrasco-Aviño, G.; Corvalan, A.H.; Quest, A. Inverse expression of survivin and reprimo correlates with poor patient prognosis in gastric cancer. Oncotarget 2018, 9, 12853–12867. [Google Scholar] [CrossRef] [PubMed]
- The Japanese Research Society for Gastric Cancer. Japanese classification of gastric carcinoma: 3rd English edition. Gastric Cancer 2011, 14, 101–112. [Google Scholar]
- Torre, L.A.; Bray, F.; Siegel, R.L.; Ferlay, J.; Lortet-Tieulent, J.; Jemal, A. Global cancer statistics, 2012. CA Cancer J. Clin. 2015, 65, 87–108. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.L.; Gao, W.; Kang, Q.M.; Zhang, X.J.; Yang, S.G. Prognostic value of survivin in patients with gastric cancer: A systematic review with meta-analysis. PLoS ONE 2013, 8, e71930. [Google Scholar] [CrossRef] [PubMed]
- Perri, F.; Longo, F.; Giuliano, M.; Sabbatino, F.; Favia, G.; Ionna, F.; Addeo, R.; Della Vittoria Scarpati, G.; Di Lorenzo, G.; Pisconti, S. Epigenetic control of gene expression: Potential implications for cancer treatment. Crit. Rev. Oncol. Hematol. 2017, 111, 166–172. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Bloj, B.; Moses, C.; Sgro, A.; Plani-Lam, J.; Arooj, M.; Duffy, C.; Thiruvengadam, S.; Sorolla, A.; Rashwan, R.; Mancera, R.L.; et al. Waking up dormant tumor suppressor genes with zinc fingers, tales and the crispr/dcas9 system. Oncotarget 2016, 7, 60535–60554. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Wei, L.; Law, C.T.; Tsang, F.H.; Shen, J.; Cheng, C.L.; Tsang, L.H.; Ho, D.W.; Chiu, D.K.; Lee, J.M.; et al. RNA N6-methyladenosine methyltransferase-like 3 promotes liver cancer progression through YTHDF2 dependent post-transcriptional silencing of SOCS2. Hepatology 2017. [Google Scholar] [CrossRef]
- Liu, L.; Yang, X. Implication of reprimo and hMLH1 gene methylation in early diagnosis of gastric carcinoma. Int. J. Clin. Exp. Pathol. 2015, 8, 14977–14982. [Google Scholar] [PubMed]
- Schneider, B.G.; Peng, D.F.; Camargo, M.C.; Piazuelo, M.B.; Sicinschi, L.A.; Mera, R.; Romero-Gallo, J.; Delgado, A.G.; Bravo, L.E.; Wilson, K.T.; et al. Promoter DNA hypermethylation in gastric biopsies from subjects at high and low risk for gastric cancer. Int. J. Cancer 2010, 127, 2588–2597. [Google Scholar] [CrossRef] [PubMed]
- Maeda, M.; Yamashita, S.; Shimazu, T.; Iida, N.; Takeshima, H.; Nakajima, T.; Oda, I.; Nanjo, S.; Kusano, C.; Mori, A.; et al. Novel epigenetic markers for gastric cancer risk stratification in individuals after helicobacter pylori eradication. Gastric Cancer 2018. [Google Scholar] [CrossRef] [PubMed]
- Schneider, B.G.; Mera, R.; Piazuelo, M.B.; Bravo, J.C.; Zabaleta, J.; Delgado, A.G.; Bravo, L.E.; Wilson, K.T.; El-Rifai, W.; Peek, R.M., Jr.; et al. DNA methylation predicts progression of human gastric lesions. Cancer Epidemiol. Biomark. Prev. 2015, 24, 1607–1613. [Google Scholar] [CrossRef] [PubMed]
- Duffy, M.J.; Napieralski, R.; Martens, J.W.; Span, P.N.; Spyratos, F.; Sweep, F.C.; Brunner, N.; Foekens, J.A.; Schmitt, M. Methylated genes as new cancer biomarkers. Eur. J. Cancer 2009, 45, 335–346. [Google Scholar] [CrossRef] [PubMed]
- Padmanabhan, N.; Ushijima, T.; Tan, P. How to stomach an epigenetic insult: The gastric cancer epigenome. Nature reviews. Gastroenterol. Hepatol. 2017, 14, 467–478. [Google Scholar]
- Leygo, C.; Williams, M.; Jin, H.C.; Chan, M.W.Y.; Chu, W.K.; Grusch, M.; Cheng, Y.Y. DNA methylation as a noninvasive epigenetic biomarker for the detection of cancer. Dis. Markers 2017, 2017, 3726595. [Google Scholar] [CrossRef] [PubMed]
- Sapari, N.S.; Loh, M.; Vaithilingam, A.; Soong, R. Clinical potential of DNA methylation in gastric cancer: A meta-analysis. PLoS ONE 2012, 7, e36275. [Google Scholar] [CrossRef] [PubMed]
- Wen, J.; Zheng, T.; Hu, K.; Zhu, C.; Guo, L.; Ye, G. Promoter methylation of tumor-related genes as a potential biomarker using blood samples for gastric cancer detection. Oncotarget 2017, 8, 77783–77793. [Google Scholar] [CrossRef] [PubMed]
- Jorquera, R.; Ortiz, R.; Ossandon, F.; Cardenas, J.P.; Sepulveda, R.; Gonzalez, C.; Holmes, D.S. SinEx DB: A database for single exon coding sequences in mammalian genomes. Database 2016. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amigo, J.D.; Opazo, J.C.; Jorquera, R.; Wichmann, I.A.; Garcia-Bloj, B.A.; Alarcon, M.A.; Owen, G.I.; Corvalán, A.H. The Reprimo Gene Family: A Novel Gene Lineage in Gastric Cancer with Tumor Suppressive Properties. Int. J. Mol. Sci. 2018, 19, 1862. https://doi.org/10.3390/ijms19071862
Amigo JD, Opazo JC, Jorquera R, Wichmann IA, Garcia-Bloj BA, Alarcon MA, Owen GI, Corvalán AH. The Reprimo Gene Family: A Novel Gene Lineage in Gastric Cancer with Tumor Suppressive Properties. International Journal of Molecular Sciences. 2018; 19(7):1862. https://doi.org/10.3390/ijms19071862
Chicago/Turabian StyleAmigo, Julio D., Juan C. Opazo, Roddy Jorquera, Ignacio A. Wichmann, Benjamin A. Garcia-Bloj, Maria Alejandra Alarcon, Gareth I. Owen, and Alejandro H. Corvalán. 2018. "The Reprimo Gene Family: A Novel Gene Lineage in Gastric Cancer with Tumor Suppressive Properties" International Journal of Molecular Sciences 19, no. 7: 1862. https://doi.org/10.3390/ijms19071862
APA StyleAmigo, J. D., Opazo, J. C., Jorquera, R., Wichmann, I. A., Garcia-Bloj, B. A., Alarcon, M. A., Owen, G. I., & Corvalán, A. H. (2018). The Reprimo Gene Family: A Novel Gene Lineage in Gastric Cancer with Tumor Suppressive Properties. International Journal of Molecular Sciences, 19(7), 1862. https://doi.org/10.3390/ijms19071862




