Macrophage Populations in Visceral Adipose Tissue from Pregnant Women: Potential Role of Obesity in Maternal Inflammation
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients and Donor Selection
2.2. Collection of Visceral Adipose Tissue
2.3. Isolation of the Stromal Vascular Fraction from Visceral Adipose Tissue
2.4. Immunostaining for Macrophages from Stromal Vascular Fraction
2.5. Hematoxylin and eosin stain of Adipose Tissue Sections
2.6. Statistical Analysis
3. Results
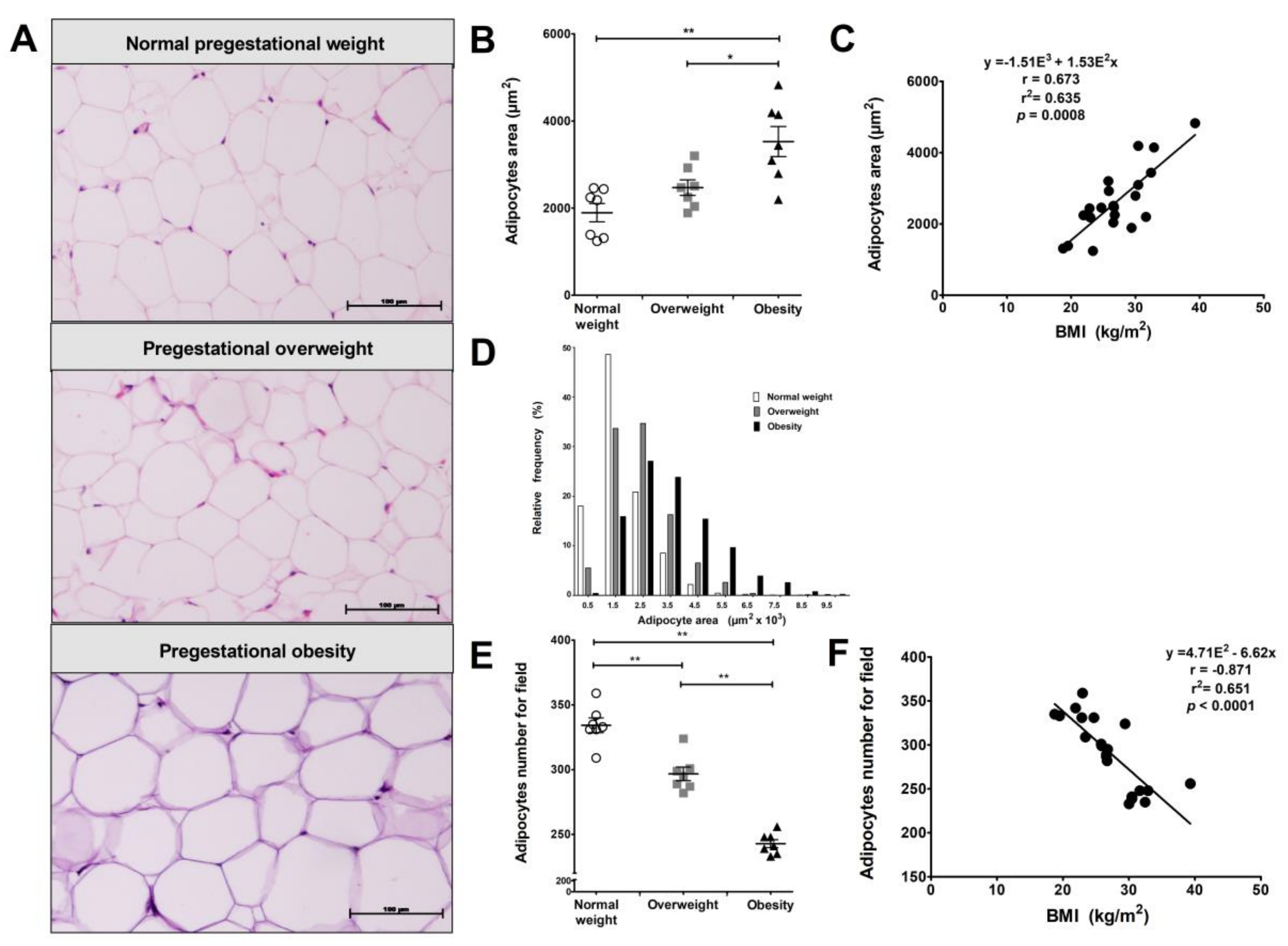
3.1. Pregestational Obesity Is Associated with Visceral Adipocyte Hypertrophy
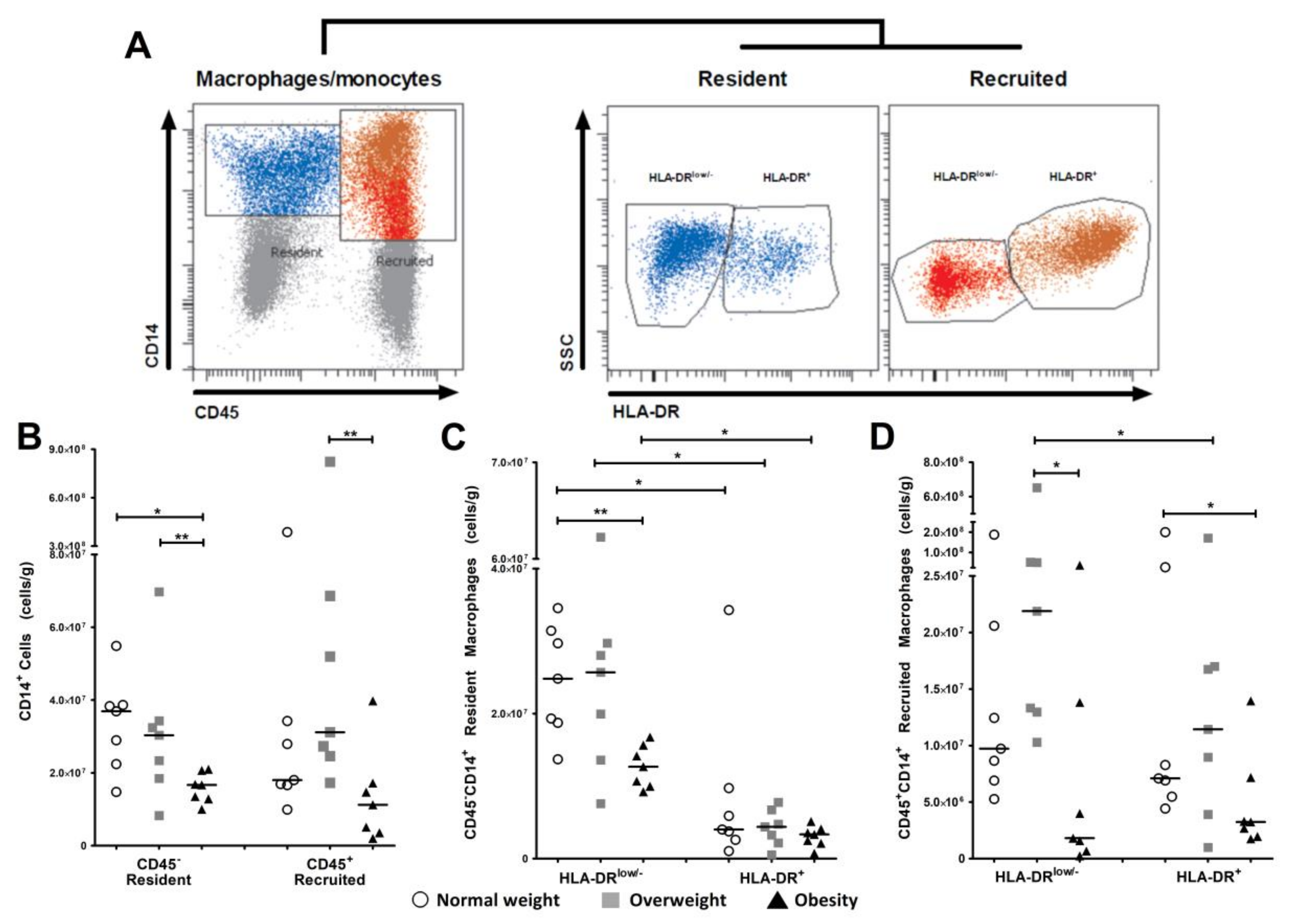
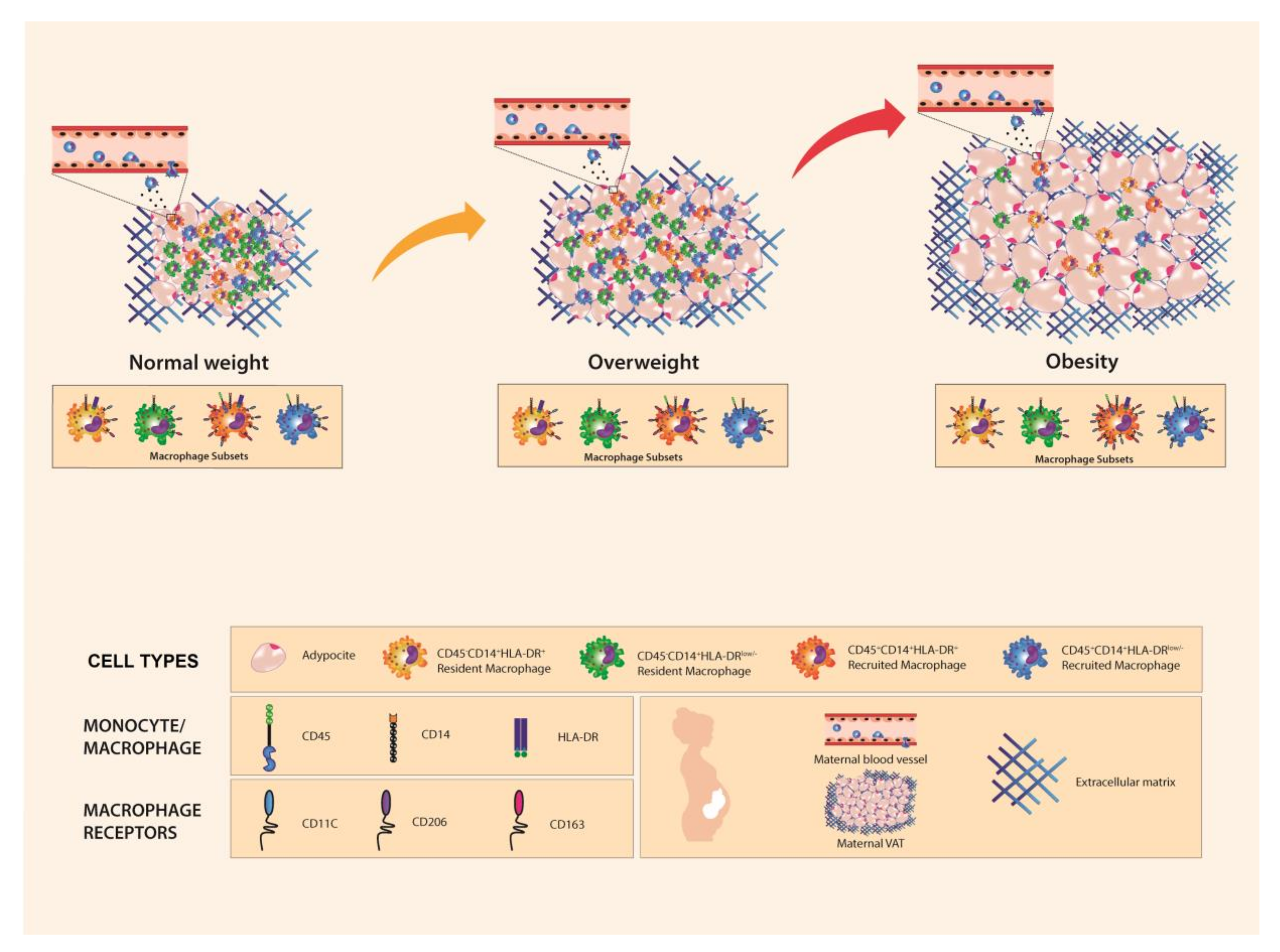
3.2. Resident and Recruited Macrophage Subsets Are Present in Visceral Adipose Tissue from Pregnant Women
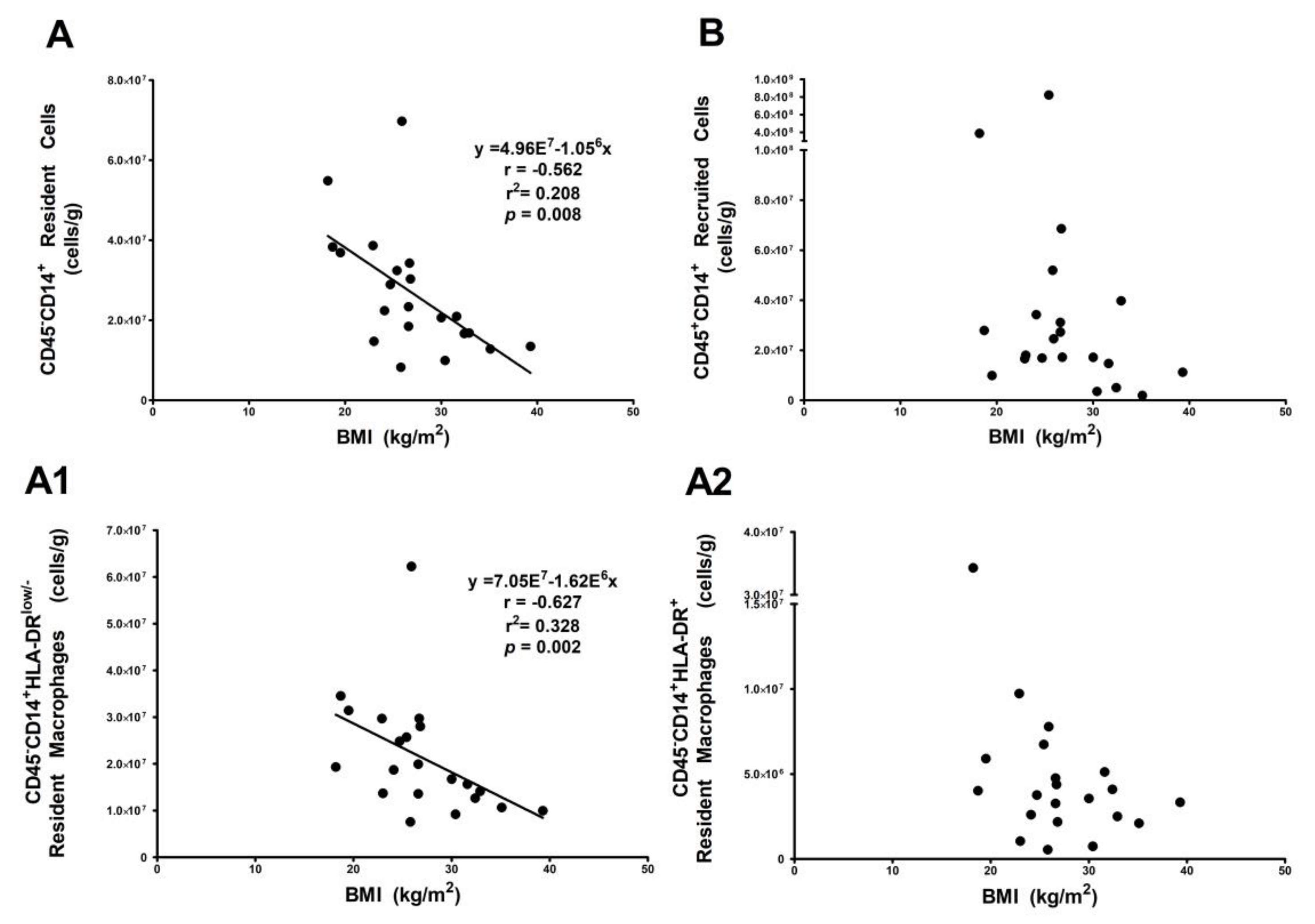
3.3. Resident HLA-DRlow/− Macrophage Subset Is Influenced by Body Mass Index
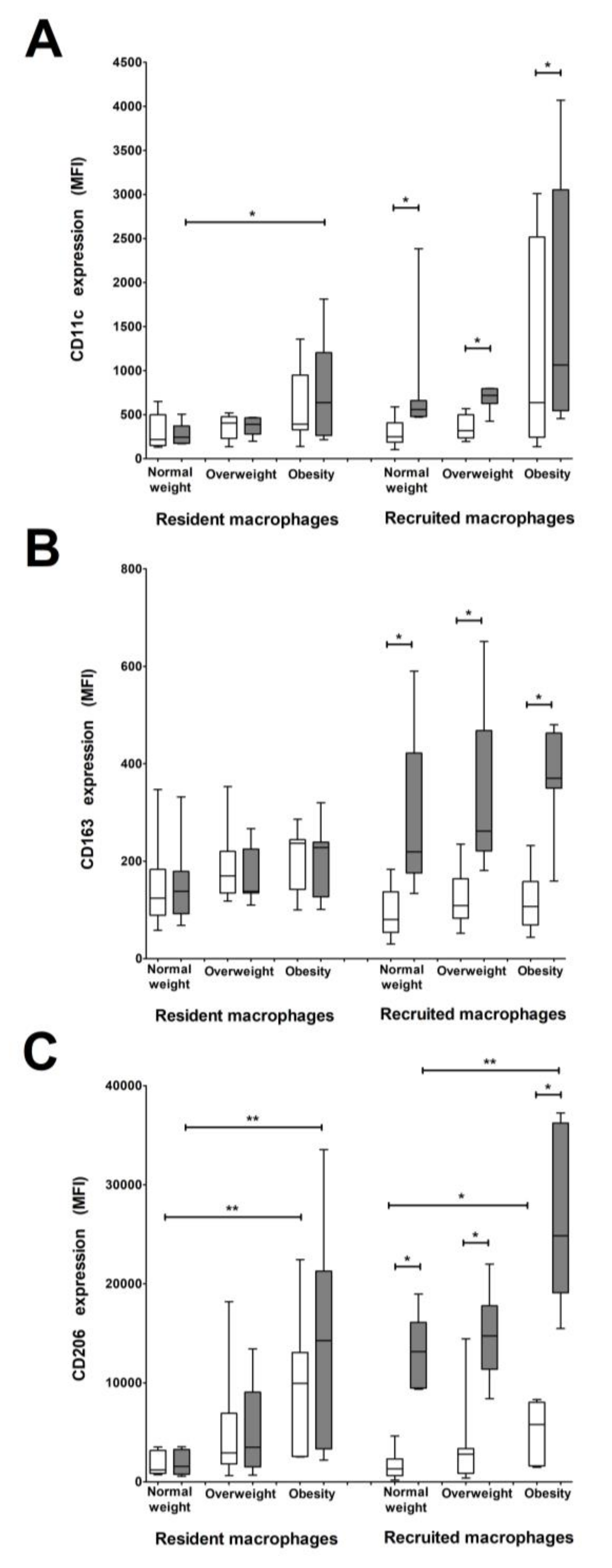
3.4. CD206 and CD11c Surface Expression Is Higher in Recruited and Resident Macrophages from Women with Pregestational Obesity
4. Discussion and Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cousin, B.; Casteilla, L.; Laharrague, P.; Luche, E.; Lorsignol, A.; Cuminetti, V.; Paupert, J. Immuno-metabolism and adipose tissue: The key role of hematopoietic stem cells. Biochimie 2016, 124, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Mraz, M.; Haluzik, M. The role of adipose tissue immune cells in obesity and low-grade inflammation. J. Endocrinol. 2014, 222, R113–R127. [Google Scholar] [CrossRef] [PubMed]
- Vieira-Potter, V.J. Inflammation and macrophage modulation in adipose tissues. Cell. Microbiol. 2014, 16, 1484–1492. [Google Scholar] [CrossRef] [PubMed]
- Boutens, L.; Stienstra, R. Adipose tissue macrophages: Going off track during obesity. Diabetologia 2016, 59, 879–894. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, A.S.; Obin, M.S. Obesity and the role of adipose tissue in inflammation and metabolism. Am. J. Clin. Nutr. 2006, 83, S461–S465. [Google Scholar] [CrossRef]
- Grant, R.W.; Dixit, V.D. Adipose tissue as an immunological organ. Obesity 2015, 23, 512–518. [Google Scholar] [CrossRef] [PubMed]
- Masoodi, M.; Kuda, O.; Rossmeisl, M.; Flachs, P.; Kopecky, J. Lipid signaling in adipose tissue: Connecting inflammation and metabolism. Biochim. Biophys. Acta 2015, 1851, 503–518. [Google Scholar] [CrossRef] [PubMed]
- Ouchi, N.; Parker, J.L.; Lugus, J.J.; Walsh, K. Adipokines in inflammation and metabolic disease. Nat. Rev. Immunol. 2011, 11, 85–97. [Google Scholar] [CrossRef] [PubMed]
- Segovia, S.A.; Vickers, M.H.; Reynolds, C.M. The impact of maternal obesity on inflammatory processes and consequences for later offspring health outcomes. J. Dev. Orig. Health Dis. 2017, 8, 529–540. [Google Scholar] [CrossRef] [PubMed]
- Nathanielsz, P.W.; Ford, S.P.; Long, N.M.; Vega, C.C.; Reyes-Castro, L.A.; Zambrano, E. Interventions to prevent adverse fetal programming due to maternal obesity during pregnancy. Nutr. Rev. 2013, 71, S78–S87. [Google Scholar] [CrossRef] [PubMed]
- Ong, T.P.; Guest, P.C. Nutritional programming effects on development of metabolic disorders in later life. Methods Mol. Biol. 2018, 1735, 3–17. [Google Scholar] [PubMed]
- Ginhoux, F.; Greter, M.; Leboeuf, M.; Nandi, S.; See, P.; Gokhan, S.; Mehler, M.F.; Conway, S.J.; Ng, L.G.; Stanley, E.R.; et al. Fate mapping analysis reveals that adult microglia derive from primitive macrophages. Science 2010, 330, 841–845. [Google Scholar] [CrossRef] [PubMed]
- Hoeffel, G.; Wang, Y.; Greter, M.; See, P.; Teo, P.; Malleret, B.; Leboeuf, M.; Low, D.; Oller, G.; Almeida, F.; et al. Adult langerhans cells derive predominantly from embryonic fetal liver monocytes with a minor contribution of yolk sac-derived macrophages. J. Exp. Med. 2012, 209, 1167–1181. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, D.; Chow, A.; Noizat, C.; Teo, P.; Beasley, M.B.; Leboeuf, M.; Becker, C.D.; See, P.; Price, J.; Lucas, D.; et al. Tissue-resident macrophages self-maintain locally throughout adult life with minimal contribution from circulating monocytes. Immunity 2013, 38, 792–804. [Google Scholar] [CrossRef] [PubMed]
- Landsman, L.; Varol, C.; Jung, S. Distinct differentiation potential of blood monocyte subsets in the lung. J. Immunol. 2007, 178, 2000–2007. [Google Scholar] [CrossRef] [PubMed]
- Ajuebor, M.N.; Das, A.M.; Virag, L.; Flower, R.J.; Szabo, C.; Perretti, M. Role of resident peritoneal macrophages and mast cells in chemokine production and neutrophil migration in acute inflammation: Evidence for an inhibitory loop involving endogenous il-10. J. Immunol. 1999, 162, 1685–1691. [Google Scholar] [PubMed]
- Italiani, P.; Boraschi, D. From monocytes to m1/m2 macrophages: Phenotypical vs. Functional differentiation. Front. Immunol. 2014, 5, 514. [Google Scholar] [CrossRef] [PubMed]
- Dey, A.; Allen, J.; Hankey-Giblin, P.A. Ontogeny and polarization of macrophages in inflammation: Blood monocytes versus tissue macrophages. Front. Immunol. 2014, 5, 683. [Google Scholar] [CrossRef] [PubMed]
- Perdiguero, E.G.; Geissmann, F. The development and maintenance of resident macrophages. Nat. Immunol. 2016, 17, 2–8. [Google Scholar] [CrossRef] [PubMed]
- Hoeffel, G.; Ginhoux, F. Ontogeny of tissue-resident macrophages. Front. Immunol. 2015, 6, 486. [Google Scholar] [CrossRef] [PubMed]
- Cousin, B.; Munoz, O.; Andre, M.; Fontanilles, A.M.; Dani, C.; Cousin, J.L.; Laharrague, P.; Casteilla, L.; Penicaud, L. A role for preadipocytes as macrophage-like cells. FASEB J. 1999, 13, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Charriere, G.; Cousin, B.; Arnaud, E.; Andre, M.; Bacou, F.; Penicaud, L.; Casteilla, L. Preadipocyte conversion to macrophage. Evidence of plasticity. J. Biol. Chem. 2003, 278, 9850–9855. [Google Scholar] [CrossRef] [PubMed]
- Schlitzer, A.; Schultze, J.L. Tissue-resident macrophages—How to humanize our knowledge. Immunol. Cell Biol. 2017, 95, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, Y.; Maeda, N.; Izumiya, C.; Kusume, T.; Oguri, H.; Kawashima, M.; Hayashi, K.; Nomura, A.; Yamashita, C.; Fukaya, T. Decreased human leukocyte antigen-dr expression in the lipid raft by peritoneal macrophages from women with endometriosis. Fertil. Steril. 2008, 89, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Wang, X.; Li, X.; Fan, Y.; Li, G.; Guo, C.; Zhu, F.; Zhang, L.; Shi, Y. Cd68(+)hla-dr(+) m1-like macrophages promote motility of hcc cells via nf-kappab/fak pathway. Cancer Lett. 2014, 345, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Amano, S.U.; Cohen, J.L.; Vangala, P.; Tencerova, M.; Nicoloro, S.M.; Yawe, J.C.; Shen, Y.; Czech, M.P.; Aouadi, M. Local proliferation of macrophages contributes to obesity-associated adipose tissue inflammation. Cell Metab. 2014, 19, 162–171. [Google Scholar] [CrossRef] [PubMed]
- Olefsky, J.M.; Glass, C.K. Macrophages, inflammation, and insulin resistance. Annu. Rev. Physiol. 2010, 72, 219–246. [Google Scholar] [CrossRef] [PubMed]
- Weisberg, S.P.; McCann, D.; Desai, M.; Rosenbaum, M.; Leibel, R.L.; Ferrante, A.W., Jr. Obesity is associated with macrophage accumulation in adipose tissue. J. Clin. Investig. 2003, 112, 1796–1808. [Google Scholar] [CrossRef] [PubMed]
- Braune, J.; Weyer, U.; Hobusch, C.; Mauer, J.; Bruning, J.C.; Bechmann, I.; Gericke, M. Il-6 regulates m2 polarization and local proliferation of adipose tissue macrophages in obesity. J. Immunol. 2017, 198, 2927–2934. [Google Scholar] [CrossRef] [PubMed]
- Prieur, X.; Mok, C.Y.; Velagapudi, V.R.; Nunez, V.; Fuentes, L.; Montaner, D.; Ishikawa, K.; Camacho, A.; Barbarroja, N.; O’Rahilly, S.; et al. Differential lipid partitioning between adipocytes and tissue macrophages modulates macrophage lipotoxicity and M2/M1 polarization in obese mice. Diabetes 2011, 60, 797–809. [Google Scholar] [CrossRef] [PubMed]
- Wentworth, J.M.; Naselli, G.; Brown, W.A.; Doyle, L.; Phipson, B.; Smyth, G.K.; Wabitsch, M.; O’Brien, P.E.; Harrison, L.C. Pro-inflammatory cd11c+cd206+ adipose tissue macrophages are associated with insulin resistance in human obesity. Diabetes 2010, 59, 1648–1656. [Google Scholar] [CrossRef] [PubMed]
- Patsouris, D.; Li, P.P.; Thapar, D.; Chapman, J.; Olefsky, J.M.; Neels, J.G. Ablation of cd11c-positive cells normalizes insulin sensitivity in obese insulin resistant animals. Cell Metab. 2008, 8, 301–309. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Navarrete, J.M.; Ortega, F.; Gomez-Serrano, M.; Garcia-Santos, E.; Ricart, W.; Tinahones, F.; Mingrone, G.; Peral, B.; Fernandez-Real, J.M. The mrc1/cd68 ratio is positively associated with adipose tissue lipogenesis and with muscle mitochondrial gene expression in humans. PLoS ONE 2013, 8, e70810. [Google Scholar] [CrossRef] [PubMed]
- Stout, R.D.; Watkins, S.K.; Suttles, J. Functional plasticity of macrophages: In situ reprogramming of tumor-associated macrophages. J. Leukoc. Biol. 2009, 86, 1105–1109. [Google Scholar] [CrossRef] [PubMed]
- Bourlier, V.; Bouloumie, A. Role of macrophage tissue infiltration in obesity and insulin resistance. Diabetes Metab. 2009, 35, 251–260. [Google Scholar] [CrossRef] [PubMed]
- Davey Smith, G.; Hypponen, E.; Power, C.; Lawlor, D.A. Offspring birth weight and parental mortality: Prospective observational study and meta-analysis. Am. J. Epidemiol. 2007, 166, 160–169. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhang, F.; Didelot, X.; Bruce, K.D.; Cagampang, F.R.; Vatish, M.; Hanson, M.; Lehnert, H.; Ceriello, A.; Byrne, C.D. Maternal high fat diet during pregnancy and lactation alters hepatic expression of insulin like growth factor-2 and key micrornas in the adult offspring. BMC Genom. 2009, 10, 478. [Google Scholar] [CrossRef] [PubMed]
- Boney, C.M.; Verma, A.; Tucker, R.; Vohr, B.R. Metabolic syndrome in childhood: Association with birth weight, maternal obesity, and gestational diabetes mellitus. Pediatrics 2005, 115, e290–e296. [Google Scholar] [CrossRef] [PubMed]
- Armitage, J.A.; Khan, I.Y.; Taylor, P.D.; Nathanielsz, P.W.; Poston, L. Developmental programming of the metabolic syndrome by maternal nutritional imbalance: How strong is the evidence from experimental models in mammals? J. Physiol. 2004, 561, 355–377. [Google Scholar] [CrossRef] [PubMed]
- Samuelsson, A.M.; Matthews, P.A.; Argenton, M.; Christie, M.R.; McConnell, J.M.; Jansen, E.H.; Piersma, A.H.; Ozanne, S.E.; Twinn, D.F.; Remacle, C.; et al. Diet-induced obesity in female mice leads to offspring hyperphagia, adiposity, hypertension, and insulin resistance: A novel murine model of developmental programming. Hypertension 2008, 51, 383–392. [Google Scholar] [CrossRef] [PubMed]
- Kirk, S.L.; Samuelsson, A.M.; Argenton, M.; Dhonye, H.; Kalamatianos, T.; Poston, L.; Taylor, P.D.; Coen, C.W. Maternal obesity induced by diet in rats permanently influences central processes regulating food intake in offspring. PLoS ONE 2009, 4, e5870. [Google Scholar] [CrossRef] [PubMed]
- Hemmeryckx, B.; van Bree, R.; Van Hoef, B.; Vercruysse, L.; Lijnen, H.R.; Verhaeghe, J. Adverse adipose phenotype and hyperinsulinemia in gravid mice deficient in placental growth factor. Endocrinology 2008, 149, 2176–2183. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Petry, C.J.; Evans, M.L.; Wingate, D.L.; Ong, K.K.; Reik, W.; Constancia, M.; Dunger, D.B. Raised late pregnancy glucose concentrations in mice carrying pups with targeted disruption of h19delta13. Diabetes 2010, 59, 282–286. [Google Scholar] [CrossRef] [PubMed]
- Haghiac, M.; Vora, N.L.; Basu, S.; Johnson, K.L.; Presley, L.; Bianchi, D.W.; Hauguel-de Mouzon, S. Increased death of adipose cells, a path to release cell-free DNA into systemic circulation of obese women. Obesity 2012, 20, 2213–2219. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Sugiyama, T.; Murabayashi, N.; Umekawa, T.; Ma, N.; Kamimoto, Y.; Ogawa, Y.; Sagawa, N. The inflammatory changes of adipose tissue in late pregnant mice. J. Mol. Endocrinol. 2011, 47, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Shoelson, S.E.; Herrero, L.; Naaz, A. Obesity, inflammation, and insulin resistance. Gastroenterology 2007, 132, 2169–2180. [Google Scholar] [CrossRef] [PubMed]
- Huh, J.Y.; Park, Y.J.; Ham, M.; Kim, J.B. Crosstalk between adipocytes and immune cells in adipose tissue inflammation and metabolic dysregulation in obesity. Mol. Cells 2014, 37, 365–371. [Google Scholar] [CrossRef] [PubMed]
- Cao, H. Adipocytokines in obesity and metabolic disease. J. Endocrinol. 2014, 220, T47–T59. [Google Scholar] [CrossRef] [PubMed]
- Ghigliotti, G.; Barisione, C.; Garibaldi, S.; Fabbi, P.; Brunelli, C.; Spallarossa, P.; Altieri, P.; Rosa, G.; Spinella, G.; Palombo, D.; et al. Adipose tissue immune response: Novel triggers and consequences for chronic inflammatory conditions. Inflammation 2014, 37, 1337–1353. [Google Scholar] [CrossRef] [PubMed]
- Klimcakova, E.; Roussel, B.; Kovacova, Z.; Kovacikova, M.; Siklova-Vitkova, M.; Combes, M.; Hejnova, J.; Decaunes, P.; Maoret, J.J.; Vedral, T.; et al. Macrophage gene expression is related to obesity and the metabolic syndrome in human subcutaneous fat as well as in visceral fat. Diabetologia 2011, 54, 876–887. [Google Scholar] [CrossRef] [PubMed]
- Itoh, M.; Suganami, T.; Hachiya, R.; Ogawa, Y. Adipose tissue remodeling as homeostatic inflammation. Int. J. Inflamm. 2011, 2011, 720926. [Google Scholar] [CrossRef] [PubMed]
- Surmi, B.K.; Hasty, A.H. Macrophage infiltration into adipose tissue: Initiation, propagation and remodeling. Future Lipidol. 2008, 3, 545–556. [Google Scholar] [CrossRef] [PubMed]
- Kratz, M.; Coats, B.R.; Hisert, K.B.; Hagman, D.; Mutskov, V.; Peris, E.; Schoenfelt, K.Q.; Kuzma, J.N.; Larson, I.; Billing, P.S.; et al. Metabolic dysfunction drives a mechanistically distinct proinflammatory phenotype in adipose tissue macrophages. Cell Metab. 2014, 20, 614–625. [Google Scholar] [CrossRef] [PubMed]
| Normal Weight n = 7 | Overweight n = 7 | Obesity n = 7 | p | |
|---|---|---|---|---|
| Mother characteristics | ||||
| Maternal age (years) | 27.6 ± 6.5 | 32.1 ± 8.3 | 32.8 ± 4.3 | 0.286 |
| Pregestational BMI (kg m−2) | 21.6 ± 2.7 | 26.7 ± 1.2 | 33.3 ± 3.4 | <0.0001 |
| Parity | 3.1 ± 1.4 | 3.1 ± 1.4 | 3.5 ± 1.9 | 0.859 |
| Fasting glucose (mg/dL) | 79.4 ± 6.3 | 78.1 ± 11.0 | 83.3 ± 8.5 | 0.498 |
| Newborn characteristics | ||||
| Gender | 2F/5M | 4F/3M | 7F/0M | |
| Birth weight (g) | 2868.6 ± 248.9 | 3408.4 ± 647.3 | 3363.1 ± 468.1 | 0.092 |
| Birth height (cm) | 47.0 ± 1.6 | 50.2 ± 2.5 | 49.1 ± 1.4 | 0.014 |
| Gestational age (weeks) | 37.9 ± 0.8 | 38.6 ± 1.0 | 38.5 ± 0.5 | 0.168 |
| Capurro method (weeks) | 38.8 ± 0.7 | 39.1 ± 1.1 | 38.2 ± 1.4 | 0.250 |
| Apgar 1st min (score) | 8.0 ± 0.0 | 8.0 ± 0.5 | 7.9 ± 0.4 | 0.741 |
| Apgar 5th min (score) | 9.0 ± 0.0 | 9.0 ± 0.0 | 9.0 ± 0.0 | 1.0 |
| Silverman–Andersen | 1.6 ± 0.5 | 1.3 ± 0.5 | 1.5 ± 1.1 | 0.357 |
| BMI | |||||
|---|---|---|---|---|---|
| Resident | Recruited | ||||
| HLA-DRlow/− | HLA-DR+ | HLA-DRlow/− | HLA-DR+ | ||
| MFI CD11c | r | 0.4014 | 0.5528 | 0.3800 | 0.4001 |
| p | (ns) | (0.009) | (ns) | (ns) | |
| MFI CD163 | r | 0.2605 | 0.2726 | 0.0474 | 0.2449 |
| p | (ns) | (ns) | (ns) | (ns) | |
| MFI CD206 | r | 0.6255 | 0.6665 | 0.4651 | 0.7197 |
| p | (0.002) | (0.001) | (0.034) | (0.0002) | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bravo-Flores, E.; Mancilla-Herrera, I.; Espino y Sosa, S.; Ortiz-Ramirez, M.; Flores-Rueda, V.; Ibargüengoitia-Ochoa, F.; Ibañez, C.A.; Zambrano, E.; Solis-Paredes, M.; Perichart-Perera, O.; et al. Macrophage Populations in Visceral Adipose Tissue from Pregnant Women: Potential Role of Obesity in Maternal Inflammation. Int. J. Mol. Sci. 2018, 19, 1074. https://doi.org/10.3390/ijms19041074
Bravo-Flores E, Mancilla-Herrera I, Espino y Sosa S, Ortiz-Ramirez M, Flores-Rueda V, Ibargüengoitia-Ochoa F, Ibañez CA, Zambrano E, Solis-Paredes M, Perichart-Perera O, et al. Macrophage Populations in Visceral Adipose Tissue from Pregnant Women: Potential Role of Obesity in Maternal Inflammation. International Journal of Molecular Sciences. 2018; 19(4):1074. https://doi.org/10.3390/ijms19041074
Chicago/Turabian StyleBravo-Flores, Eyerahi, Ismael Mancilla-Herrera, Salvador Espino y Sosa, Marco Ortiz-Ramirez, Verónica Flores-Rueda, Francisco Ibargüengoitia-Ochoa, Carlos A. Ibañez, Elena Zambrano, Mario Solis-Paredes, Otilia Perichart-Perera, and et al. 2018. "Macrophage Populations in Visceral Adipose Tissue from Pregnant Women: Potential Role of Obesity in Maternal Inflammation" International Journal of Molecular Sciences 19, no. 4: 1074. https://doi.org/10.3390/ijms19041074
APA StyleBravo-Flores, E., Mancilla-Herrera, I., Espino y Sosa, S., Ortiz-Ramirez, M., Flores-Rueda, V., Ibargüengoitia-Ochoa, F., Ibañez, C. A., Zambrano, E., Solis-Paredes, M., Perichart-Perera, O., Sanchez-Martinez, M., Medina-Bastidas, D., Reyes-Muñoz, E., & Estrada-Gutierrez, G. (2018). Macrophage Populations in Visceral Adipose Tissue from Pregnant Women: Potential Role of Obesity in Maternal Inflammation. International Journal of Molecular Sciences, 19(4), 1074. https://doi.org/10.3390/ijms19041074







