Amino Acid Transporters and Glutamine Metabolism in Breast Cancer
Abstract
1. Introduction
2. Amino Acid Transporters
3. Amino Acid Transporters in Breast Cancer
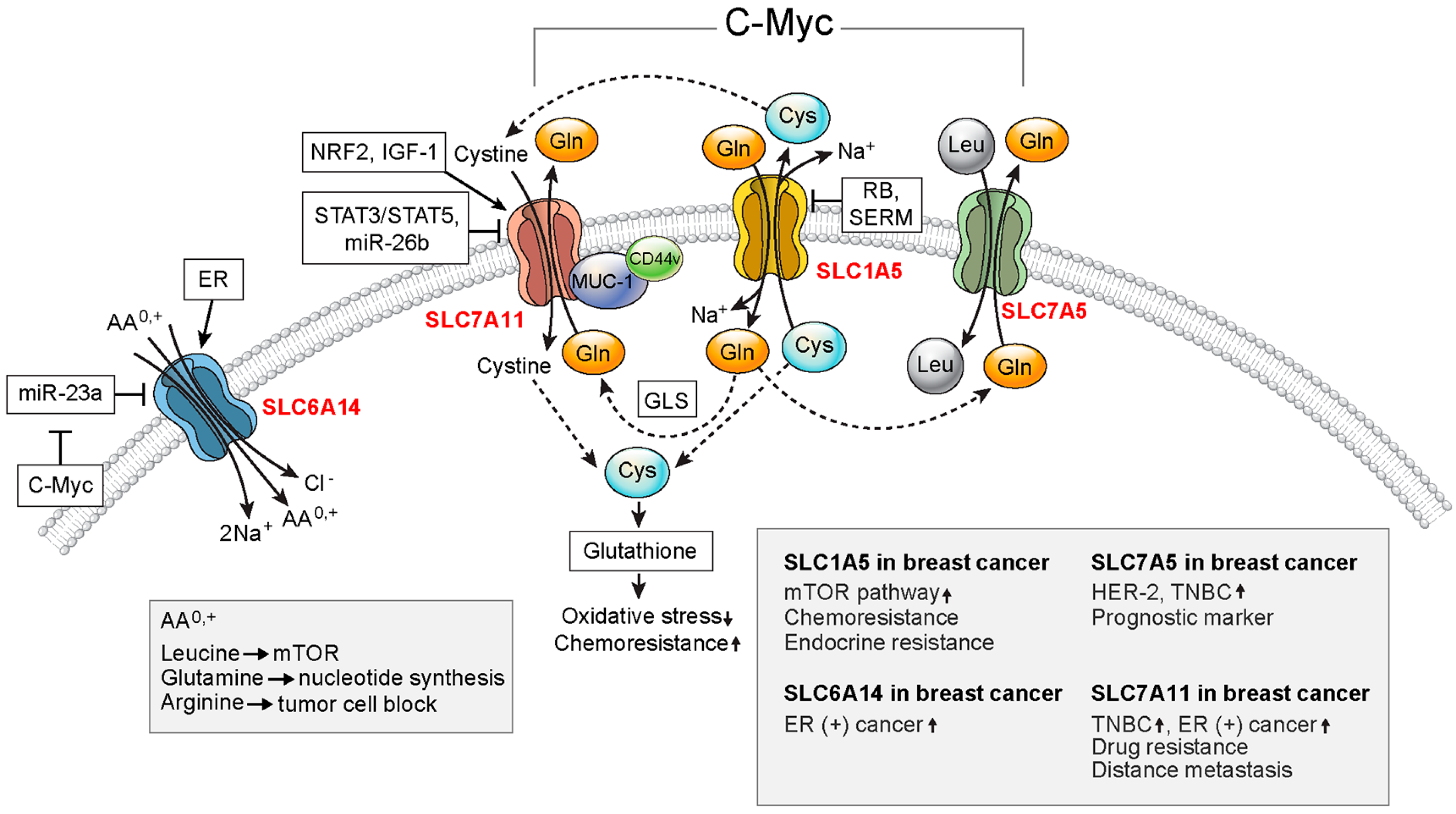
3.1. SLC1A5
3.2. SLC6A14
3.3. SLC7A5
3.4. SLC7A11
3.5. Other SLCs
4. Clinical Applications of Amino Acid Transporters in Breast Cancer
4.1. Amino Acid Transporters as Therapeutic Targets
4.2. Cancer Imaging
4.3. Drug Delivery
5. Glutamine Metabolism in Breast Cancer
5.1. Role of Glutamine and Glutamine Metabolism in Breast Cancer Cells
5.2. Regulation of Glutamine Metabolism in Breast Cancer
5.3. Alteration of Glutamine Metabolism in Breast Cancer According to Molecular Subtypes
5.4. Therapeutic Implication of Glutamine Metabolism in Breast Cancer
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Benson, J.R.; Jatoi, I. The global breast cancer burden. Future Oncol. 2012, 8, 697–702. [Google Scholar] [CrossRef] [PubMed]
- Ismail-Khan, R.; Bui, M.M. A review of triple-negative breast cancer. Cancer Control 2010, 17, 173–176. [Google Scholar] [CrossRef] [PubMed]
- Bhutia, Y.D.; Babu, E.; Ramachandran, S.; Ganapathy, V. Amino acid transporters in cancer and their relevance to “glutamine addiction”: Novel targets for the design of a new class of anticancer drugs. Cancer Res. 2015, 75, 1782–1788. [Google Scholar] [CrossRef] [PubMed]
- Wise, D.R.; Thompson, C.B. Glutamine addiction: A new therapeutic target in cancer. Trends Biochem. Sci. 2010, 35, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Locasale, J.W. Serine, glycine and one-carbon units: Cancer metabolism in full circle. Nat. Rev. Cancer 2013, 13, 572–583. [Google Scholar] [CrossRef] [PubMed]
- El Ansari, R.; McIntyre, A.; Craze, M.L.; Ellis, I.O.; Rakha, E.A.; Green, A.R. Altered glutamine metabolism in breast cancer; subtype dependencies and alternative adaptations. Histopathology 2018, 72, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Kim, D.H.; Jung, W.H.; Koo, J.S. Expression of glutamine metabolism-related proteins according to molecular subtype of breast cancer. Endocr. Relat. Cancer 2013, 20, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.K.; Jung, W.H.; Koo, J.S. Differential expression of enzymes associated with serine/glycine metabolism in different breast cancer subtypes. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Noh, S.; Kim, D.H.; Jung, W.H.; Koo, J.S. Expression levels of serine/glycine metabolism-related proteins in triple negative breast cancer tissues. Tumour Biol. 2014, 35, 4457–4468. [Google Scholar] [CrossRef] [PubMed]
- Craze, M.L.; Cheung, H.; Jewa, N.; Coimbra, N.D.M.; Soria, D.; El-Ansari, R.; Aleskandarany, M.A.; Wai Cheng, K.; Diez-Rodriguez, M.; Nolan, C.C.; et al. Myc regulation of glutamine-proline regulatory axis is key in luminal b breast cancer. Br. J. Cancer 2017, 118, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Smith, B.; Schafer, X.L.; Ambeskovic, A.; Spencer, C.M.; Land, H.; Munger, J. Addiction to coupling of the warburg effect with glutamine catabolism in cancer cells. Cell Rep. 2016, 17, 821–836. [Google Scholar] [CrossRef] [PubMed]
- DeBerardinis, R.J.; Mancuso, A.; Daikhin, E.; Nissim, I.; Yudkoff, M.; Wehrli, S.; Thompson, C.B. Beyond aerobic glycolysis: Transformed cells can engage in glutamine metabolism that exceeds the requirement for protein and nucleotide synthesis. Proc. Natl. Acad. Sci. USA 2007, 104, 19345–19350. [Google Scholar] [CrossRef] [PubMed]
- Van Geldermalsen, M.; Wang, Q.; Nagarajah, R.; Marshall, A.D.; Thoeng, A.; Gao, D.; Ritchie, W.; Feng, Y.; Bailey, C.G.; Deng, N.; et al. Asct2/slc1a5 controls glutamine uptake and tumour growth in triple-negative basal-like breast cancer. Oncogene 2016, 35, 3201–3208. [Google Scholar] [CrossRef] [PubMed]
- Karunakaran, S.; Ramachandran, S.; Coothankandaswamy, V.; Elangovan, S.; Babu, E.; Periyasamy-Thandavan, S.; Gurav, A.; Gnanaprakasam, J.P.; Singh, N.; Schoenlein, P.V.; et al. Slc6a14 (atb0,+) protein, a highly concentrative and broad specific amino acid transporter, is a novel and effective drug target for treatment of estrogen receptor-positive breast cancer. J. Biol. Chem. 2011, 286, 31830–31838. [Google Scholar] [CrossRef] [PubMed]
- Perland, E.; Fredriksson, R. Classification systems of secondary active transporters. Trends Pharmacol. Sci. 2017, 38, 305–315. [Google Scholar] [CrossRef] [PubMed]
- Gray, K.A.; Seal, R.L.; Tweedie, S.; Wright, M.W.; Bruford, E.A. A review of the new hgnc gene family resource. Hum. Genom. 2016, 10. [Google Scholar] [CrossRef] [PubMed]
- Finn, R.D.; Coggill, P.; Eberhardt, R.Y.; Eddy, S.R.; Mistry, J.; Mitchell, A.L.; Potter, S.C.; Punta, M.; Qureshi, M.; Sangrador-Vegas, A.; et al. The pfam protein families database: Towards a more sustainable future. Nucleic Acids Res. 2016, 44, D279–D285. [Google Scholar] [CrossRef] [PubMed]
- Fredriksson, R.; Nordstrom, K.J.; Stephansson, O.; Hagglund, M.G.; Schioth, H.B. The solute carrier (slc) complement of the human genome: Phylogenetic classification reveals four major families. FEBS Lett. 2008, 582, 3811–3816. [Google Scholar] [CrossRef] [PubMed]
- Hoglund, P.J.; Nordstrom, K.J.; Schioth, H.B.; Fredriksson, R. The solute carrier families have a remarkably long evolutionary history with the majority of the human families present before divergence of bilaterian species. Mol. Biol. Evol. 2011, 28, 1531–1541. [Google Scholar] [CrossRef] [PubMed]
- Schweikhard, E.S.; Ziegler, C.M. Amino acid secondary transporters: Toward a common transport mechanism. Curr. Top. Membr. 2012, 70, 1–28. [Google Scholar] [PubMed]
- Broer, S.; Palacin, M. The role of amino acid transporters in inherited and acquired diseases. Biochem. J. 2011, 436, 193–211. [Google Scholar] [CrossRef] [PubMed]
- Hediger, M.A.; Romero, M.F.; Peng, J.B.; Rolfs, A.; Takanaga, H.; Bruford, E.A. The abcs of solute carriers: Physiological, pathological and therapeutic implications of human membrane transport proteinsintroduction. Pflug. Arch. 2004, 447, 465–468. [Google Scholar] [CrossRef] [PubMed]
- Broer, S. Adaptation of plasma membrane amino acid transport mechanisms to physiological demands. Pflugers Arch. 2002, 444, 457–466. [Google Scholar] [CrossRef] [PubMed]
- Rasko, J.E.; Battini, J.L.; Gottschalk, R.J.; Mazo, I.; Miller, A.D. The rd114/simian type d retrovirus receptor is a neutral amino acid transporter. Proc. Natl. Acad. Sci. USA 1999, 96, 2129–2134. [Google Scholar] [CrossRef] [PubMed]
- Oppedisano, F.; Pochini, L.; Galluccio, M.; Indiveri, C. The glutamine/amino acid transporter (asct2) reconstituted in liposomes: Transport mechanism, regulation by atp and characterization of the glutamine/glutamate antiport. Biochim. Biophys. Acta 2007, 1768, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Gao, P.; Tchernyshyov, I.; Chang, T.C.; Lee, Y.S.; Kita, K.; Ochi, T.; Zeller, K.I.; De Marzo, A.M.; Van Eyk, J.E.; Mendell, J.T.; et al. C-myc suppression of mir-23a/b enhances mitochondrial glutaminase expression and glutamine metabolism. Nature 2009, 458, 762–765. [Google Scholar] [CrossRef] [PubMed]
- Wise, D.R.; DeBerardinis, R.J.; Mancuso, A.; Sayed, N.; Zhang, X.Y.; Pfeiffer, H.K.; Nissim, I.; Daikhin, E.; Yudkoff, M.; McMahon, S.B.; et al. Myc regulates a transcriptional program that stimulates mitochondrial glutaminolysis and leads to glutamine addiction. Proc. Natl. Acad. Sci. USA 2008, 105, 18782–18787. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, M.R.; Lane, A.N.; Robertson, B.; Kemp, S.; Liu, Y.; Hill, B.G.; Dean, D.C.; Clem, B.F. Control of glutamine metabolism by the tumor suppressor rb. Oncogene 2014, 33, 556–566. [Google Scholar] [CrossRef] [PubMed]
- Nicklin, P.; Bergman, P.; Zhang, B.; Triantafellow, E.; Wang, H.; Nyfeler, B.; Yang, H.; Hild, M.; Kung, C.; Wilson, C.; et al. Bidirectional transport of amino acids regulates mtor and autophagy. Cell 2009, 136, 521–534. [Google Scholar] [CrossRef] [PubMed]
- Cormerais, Y.; Massard, P.A.; Vucetic, M.; Giuliano, S.; Tambutte, E.; Durivault, J.; Vial, V.; Endou, H.; Wempe, M.F.; Parks, S.K.; et al. The glutamine transporter asct2 (slc1a5) promotes tumor growth independently of the amino acid transporter lat1 (slc7a5). J. Biol. Chem. 2018, 293, 2877–2887. [Google Scholar] [CrossRef] [PubMed]
- Broer, A.; Rahimi, F.; Broer, S. Deletion of amino acid transporter asct2 (slc1a5) reveals an essential role for transporters snat1 (slc38a1) and snat2 (slc38a2) to sustain glutaminolysis in cancer cells. J. Biol. Chem. 2016, 291, 13194–13205. [Google Scholar] [CrossRef] [PubMed]
- Todorova, V.K.; Kaufmann, Y.; Luo, S.; Klimberg, V.S. Tamoxifen and raloxifene suppress the proliferation of estrogen receptor-negative cells through inhibition of glutamine uptake. Cancer Chemother. Pharmacol. 2011, 67, 285–291. [Google Scholar] [CrossRef] [PubMed]
- Jeon, Y.J.; Khelifa, S.; Ratnikov, B.; Scott, D.A.; Feng, Y.; Parisi, F.; Ruller, C.; Lau, E.; Kim, H.; Brill, L.M.; et al. Regulation of glutamine carrier proteins by rnf5 determines breast cancer response to er stress-inducing chemotherapies. Cancer Cell 2015, 27, 354–369. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Wang, Y.; Warden, C.; Chen, S. Cross-talk between er and her2 regulates c-myc-mediated glutamine metabolism in aromatase inhibitor resistant breast cancer cells. J. Steroid Biochem. Mol. Biol. 2015, 149, 118–127. [Google Scholar] [CrossRef] [PubMed]
- Bernhardt, S.; Bayerlova, M.; Vetter, M.; Wachter, A.; Mitra, D.; Hanf, V.; Lantzsch, T.; Uleer, C.; Peschel, S.; John, J.; et al. Proteomic profiling of breast cancer metabolism identifies shmt2 and asct2 as prognostic factors. Breast Cancer Res. 2017, 19, 112. [Google Scholar] [CrossRef] [PubMed]
- Sloan, J.L.; Mager, S. Cloning and functional expression of a human na(+) and cl(-)-dependent neutral and cationic amino acid transporter b(0+). J. Biol. Chem. 1999, 274, 23740–23745. [Google Scholar] [CrossRef] [PubMed]
- Babu, E.; Bhutia, Y.D.; Ramachandran, S.; Gnanaprakasam, J.P.; Prasad, P.D.; Thangaraju, M.; Ganapathy, V. Deletion of the amino acid transporter slc6a14 suppresses tumour growth in spontaneous mouse models of breast cancer. Biochem. J. 2015, 469, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.; Wang, N.; Tsao, S.W.; Yuen, M.F.; Feng, Y.; Wan, T.S.; Man, K.; Feng, Y. Up-regulation of micrornas, mir21 and mir23a in human liver cancer cells treated with coptidis rhizoma aqueous extract. Exp. Ther. Med. 2011, 2, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Fotiadis, D.; Kanai, Y.; Palacin, M. The slc3 and slc7 families of amino acid transporters. Mol. Asp. Med. 2013, 34, 139–158. [Google Scholar] [CrossRef] [PubMed]
- Kaira, K.; Oriuchi, N.; Imai, H.; Shimizu, K.; Yanagitani, N.; Sunaga, N.; Hisada, T.; Tanaka, S.; Ishizuka, T.; Kanai, Y.; et al. l-Type amino acid transporter 1 and cd98 expression in primary and metastatic sites of human neoplasms. Cancer Sci. 2008, 99, 2380–2386. [Google Scholar] [CrossRef] [PubMed]
- Prasad, P.D.; Wang, H.; Huang, W.; Kekuda, R.; Rajan, D.P.; Leibach, F.H.; Ganapathy, V. Human lat1, a subunit of system l amino acid transporter: Molecular cloning and transport function. Biochem. Biophys. Res. Commun. 1999, 255, 283–288. [Google Scholar] [CrossRef] [PubMed]
- Shennan, D.B.; Thomson, J.; Barber, M.C.; Travers, M.T. Functional and molecular characteristics of system l in human breast cancer cells. Biochim. Biophys. Acta 2003, 1611, 81–90. [Google Scholar] [CrossRef]
- Shennan, D.B.; Thomson, J.; Gow, I.F.; Travers, M.T.; Barber, M.C. L-leucine transport in human breast cancer cells (mcf-7 and mda-mb-231): Kinetics, regulation by estrogen and molecular identity of the transporter. Biochim. Biophys. Acta 2004, 1664, 206–216. [Google Scholar] [CrossRef] [PubMed]
- Furuya, M.; Horiguchi, J.; Nakajima, H.; Kanai, Y.; Oyama, T. Correlation of l-type amino acid transporter 1 and cd98 expression with triple negative breast cancer prognosis. Cancer Sci. 2012, 103, 382–389. [Google Scholar] [CrossRef] [PubMed]
- Mihaly, Z.; Kormos, M.; Lanczky, A.; Dank, M.; Budczies, J.; Szasz, M.A.; Gyorffy, B. A meta-analysis of gene expression-based biomarkers predicting outcome after tamoxifen treatment in breast cancer. Breast Cancer Res. Treat. 2013, 140, 219–232. [Google Scholar] [CrossRef] [PubMed]
- Bartlett, J.M.; Bloom, K.J.; Piper, T.; Lawton, T.J.; van de Velde, C.J.; Ross, D.T.; Ring, B.Z.; Seitz, R.S.; Beck, R.A.; Hasenburg, A.; et al. Mammostrat as an immunohistochemical multigene assay for prediction of early relapse risk in the tamoxifen versus exemestane adjuvant multicenter trial pathology study. J. Clin. Oncol. 2012, 30, 4477–4484. [Google Scholar] [CrossRef] [PubMed]
- Ring, B.Z.; Seitz, R.S.; Beck, R.; Shasteen, W.J.; Tarr, S.M.; Cheang, M.C.; Yoder, B.J.; Budd, G.T.; Nielsen, T.O.; Hicks, D.G.; et al. Novel prognostic immunohistochemical biomarker panel for estrogen receptor-positive breast cancer. J. Clin. Oncol. 2006, 24, 3039–3047. [Google Scholar] [CrossRef] [PubMed]
- Bartlett, J.M.; Thomas, J.; Ross, D.T.; Seitz, R.S.; Ring, B.Z.; Beck, R.A.; Pedersen, H.C.; Munro, A.; Kunkler, I.H.; Campbell, F.M.; et al. Mammostrat as a tool to stratify breast cancer patients at risk of recurrence during endocrine therapy. Breast Cancer Res. 2010, 12. [Google Scholar] [CrossRef] [PubMed]
- Ross, D.T.; Kim, C.Y.; Tang, G.; Bohn, O.L.; Beck, R.A.; Ring, B.Z.; Seitz, R.S.; Paik, S.; Costantino, J.P.; Wolmark, N. Chemosensitivity and stratification by a five monoclonal antibody immunohistochemistry test in the nsabp b14 and b20 trials. Clin. Cancer Res. 2008, 14, 6602–6609. [Google Scholar] [CrossRef] [PubMed]
- Bansal, A.; Garg, M.; Chintamani, C.; Saxena, S. Immunohistochemical expression of carcinoembryonic antigen-related cell adhesion molecules 5, ceacam6, and slc7a5: Do they aid in predicting the response to neo-adjuvant chemotherapy in locally advanced breast cancer? Clin. Cancer Investig. J. 2014, 3, 521–525. [Google Scholar] [CrossRef]
- Lyons, S.A.; Chung, W.J.; Weaver, A.K.; Ogunrinu, T.; Sontheimer, H. Autocrine glutamate signaling promotes glioma cell invasion. Cancer Res. 2007, 67, 9463–9471. [Google Scholar] [CrossRef] [PubMed]
- Lewerenz, J.; Hewett, S.J.; Huang, Y.; Lambros, M.; Gout, P.W.; Kalivas, P.W.; Massie, A.; Smolders, I.; Methner, A.; Pergande, M.; et al. The cystine/glutamate antiporter system x(c)(-) in health and disease: From molecular mechanisms to novel therapeutic opportunities. Antioxid. Redox Signal. 2013, 18, 522–555. [Google Scholar] [CrossRef] [PubMed]
- Stepulak, A.; Rola, R.; Polberg, K.; Ikonomidou, C. Glutamate and its receptors in cancer. J. Neural Transm. (Vienna) 2014, 121, 933–944. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Takano, N.; Xiang, L.; Gilkes, D.M.; Luo, W.; Semenza, G.L. Hypoxia-inducible factors enhance glutamate signaling in cancer cells. Oncotarget 2014, 5, 8853–8868. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa, M.; Takahashi, H.; Rajabi, H.; Alam, M.; Suzuki, Y.; Yin, L.; Tagde, A.; Maeda, T.; Hiraki, M.; Sukhatme, V.P.; et al. Functional interactions of the cystine/glutamate antiporter, cd44v and muc1-c oncoprotein in triple-negative breast cancer cells. Oncotarget 2016, 7, 11756–11769. [Google Scholar] [CrossRef] [PubMed]
- Timmerman, L.A.; Holton, T.; Yuneva, M.; Louie, R.J.; Padro, M.; Daemen, A.; Hu, M.; Chan, D.A.; Ethier, S.P.; van’t Veer, L.J.; et al. Glutamine sensitivity analysis identifies the xct antiporter as a common triple-negative breast tumor therapeutic target. Cancer Cell 2013, 24, 450–465. [Google Scholar] [CrossRef] [PubMed]
- Habib, E.; Linher-Melville, K.; Lin, H.X.; Singh, G. Expression of xct and activity of system xc(-) are regulated by nrf2 in human breast cancer cells in response to oxidative stress. Redox Biol. 2015, 5, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Yee, D. Igf-i regulates redox status in breast cancer cells by activating the amino acid transport molecule xc. Cancer Res. 2014, 74, 2295–2305. [Google Scholar] [CrossRef] [PubMed]
- Linher-Melville, K.; Haftchenary, S.; Gunning, P.; Singh, G. Signal transducer and activator of transcription 3 and 5 regulate system xc- and redox balance in human breast cancer cells. Mol. Cell. Biochem. 2015, 405, 205–221. [Google Scholar] [CrossRef] [PubMed]
- Linher-Melville, K.; Nashed, M.G.; Ungard, R.G.; Haftchenary, S.; Rosa, D.A.; Gunning, P.T.; Singh, G. Chronic inhibition of stat3/stat5 in treatment-resistant human breast cancer cell subtypes: Convergence on the ros/sumo pathway and its effects on xct expression and system xc- activity. PLoS ONE 2016, 11. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.X.; Li, X.J.; Zhang, B.; Liang, Y.J.; Zhou, C.X.; Cao, D.X.; He, M.; Chen, G.Q.; He, J.R.; Zhao, Q. Microrna-26b is underexpressed in human breast cancer and induces cell apoptosis by targeting slc7a11. FEBS Lett. 2011, 585, 1363–1367. [Google Scholar] [CrossRef] [PubMed]
- Ge, C.; Cao, B.; Feng, D.; Zhou, F.; Zhang, J.; Yang, N.; Feng, S.; Wang, G.; Aa, J. The down-regulation of slc7a11 enhances ros induced p-gp over-expression and drug resistance in mcf-7 breast cancer cells. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Sato, R.; Nakano, T.; Hosonaga, M.; Sampetrean, O.; Harigai, R.; Sasaki, T.; Koya, I.; Okano, H.; Kudoh, J.; Saya, H.; et al. Rna sequencing analysis reveals interactions between breast cancer or melanoma cells and the tissue microenvironment during brain metastasis. Biomed. Res. Int. 2017, 2017. [Google Scholar] [CrossRef] [PubMed]
- Lanzardo, S.; Conti, L.; Rooke, R.; Ruiu, R.; Accart, N.; Bolli, E.; Arigoni, M.; Macagno, M.; Barrera, G.; Pizzimenti, S.; et al. Immunotargeting of antigen xct attenuates stem-like cell behavior and metastatic progression in breast cancer. Cancer Res. 2016, 76, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Cao, Y.; Wang, Y.; Li, W.; Liu, X.; Lv, Y.; Li, X.; Mi, J. Cysteine transporter slc3a1 promotes breast cancer tumorigenesis. Theranostics 2017, 7, 1036–1046. [Google Scholar] [CrossRef] [PubMed]
- Leo, J.C.; Wang, S.M.; Guo, C.H.; Aw, S.E.; Zhao, Y.; Li, J.M.; Hui, K.M.; Lin, V.C. Gene regulation profile reveals consistent anticancer properties of progesterone in hormone-independent breast cancer cells transfected with progesterone receptor. Int. J. Cancer 2005, 117, 561–568. [Google Scholar] [CrossRef] [PubMed]
- Sweet, R.; Paul, A.; Zastre, J. Hypoxia induced upregulation and function of the thiamine transporter, slc19a3 in a breast cancer cell line. Cancer Biol. Ther. 2010, 10, 1101–1111. [Google Scholar] [CrossRef] [PubMed]
- Thakkar, A.; Raj, H.; Ravishankar; Muthuvelan, B.; Balakrishnan, A.; Padigaru, M. High expression of three-gene signature improves prediction of relapse-free survival in estrogen receptor-positive and node-positive breast tumors. Biomark. Insights 2015, 10, 103–112. [Google Scholar] [CrossRef] [PubMed]
- Thakkar, A.D.; Raj, H.; Chakrabarti, D.; Ravishankar; Saravanan, N.; Muthuvelan, B.; Balakrishnan, A.; Padigaru, M. Identification of gene expression signature in estrogen receptor positive breast carcinoma. Biomark. Cancer 2010, 2, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Cao, F.; Fang, W.; Hu, Y.; Chen, Y.; Ding, H.; Yu, G. Activation of snat1/slc38a1 in human breast cancer: Correlation with p-akt overexpression. BMC Cancer 2013, 13. [Google Scholar] [CrossRef] [PubMed]
- Ganapathy, V.; Thangaraju, M.; Prasad, P.D. Nutrient transporters in cancer: Relevance to warburg hypothesis and beyond. Pharmacol. Ther. 2009, 121, 29–40. [Google Scholar] [CrossRef] [PubMed]
- McCracken, A.N.; Edinger, A.L. Nutrient transporters: The achilles’ heel of anabolism. Trends Endocrinol. Metab. 2013, 24, 200–208. [Google Scholar] [CrossRef] [PubMed]
- Sato, H.; Shiiya, A.; Kimata, M.; Maebara, K.; Tamba, M.; Sakakura, Y.; Makino, N.; Sugiyama, F.; Yagami, K.; Moriguchi, T.; et al. Redox imbalance in cystine/glutamate transporter-deficient mice. J. Biol. Chem. 2005, 280, 37423–37429. [Google Scholar] [CrossRef] [PubMed]
- Nakaya, M.; Xiao, Y.; Zhou, X.; Chang, J.H.; Chang, M.; Cheng, X.; Blonska, M.; Lin, X.; Sun, S.C. Inflammatory t cell responses rely on amino acid transporter asct2 facilitation of glutamine uptake and mtorc1 kinase activation. Immunity 2014, 40, 692–705. [Google Scholar] [CrossRef] [PubMed]
- Poncet, N.; Mitchell, F.E.; Ibrahim, A.F.; McGuire, V.A.; English, G.; Arthur, J.S.; Shi, Y.B.; Taylor, P.M. The catalytic subunit of the system l1 amino acid transporter (slc7a5) facilitates nutrient signalling in mouse skeletal muscle. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Oda, K.; Hosoda, N.; Endo, H.; Saito, K.; Tsujihara, K.; Yamamura, M.; Sakata, T.; Anzai, N.; Wempe, M.F.; Kanai, Y.; et al. L-type amino acid transporter 1 inhibitors inhibit tumor cell growth. Cancer Sci. 2010, 101, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Rosilio, C.; Nebout, M.; Imbert, V.; Griessinger, E.; Neffati, Z.; Benadiba, J.; Hagenbeek, T.; Spits, H.; Reverso, J.; Ambrosetti, D.; et al. L-type amino-acid transporter 1 (lat1): A therapeutic target supporting growth and survival of t-cell lymphoblastic lymphoma/t-cell acute lymphoblastic leukemia. Leukemia 2015, 29, 1253–1266. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Zheng, W.; Nagana Gowda, G.A.; Raftery, D.; Donkin, S.S.; Bequette, B.; Teegarden, D. 1,25-dihydroxyvitamin d inhibits glutamine metabolism in harvey-ras transformed mcf10a human breast epithelial cell. J. Steroid Biochem. Mol. Biol. 2016, 163, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Shennan, D.B.; Thomson, J. Inhibition of system L (lat1/cd98hc) reduces the growth of cultured human breast cancer cells. Oncol. Rep. 2008, 20, 885–889. [Google Scholar] [CrossRef] [PubMed]
- Narang, V.S.; Pauletti, G.M.; Gout, P.W.; Buckley, D.J.; Buckley, A.R. Sulfasalazine-induced reduction of glutathione levels in breast cancer cells: Enhancement of growth-inhibitory activity of doxorubicin. Chemotherapy 2007, 53, 210–217. [Google Scholar] [CrossRef] [PubMed]
- Warburg, O. On the origin of cancer cells. Science 1956, 123, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Way, J.D.; Wang, M.; Hamann, I.; Wuest, M.; Wuest, F. Synthesis and evaluation of 2-amino-5-(4-[(18)f]fluorophenyl)pent-4-ynoic acid ([(18)f]fphpa): A novel (18)f-labeled amino acid for oncologic pet imaging. Nucl. Med. Biol. 2014, 41, 660–669. [Google Scholar] [CrossRef] [PubMed]
- Okudaira, H.; Shikano, N.; Nishii, R.; Miyagi, T.; Yoshimoto, M.; Kobayashi, M.; Ohe, K.; Nakanishi, T.; Tamai, I.; Namiki, M.; et al. Putative transport mechanism and intracellular fate of trans-1-amino-3-18f-fluorocyclobutanecarboxylic acid in human prostate cancer. J. Nucl. Med. 2011, 52, 822–829. [Google Scholar] [CrossRef] [PubMed]
- Okudaira, H.; Nakanishi, T.; Oka, S.; Kobayashi, M.; Tamagami, H.; Schuster, D.M.; Goodman, M.M.; Shirakami, Y.; Tamai, I.; Kawai, K. Kinetic analyses of trans-1-amino-3-[18f]fluorocyclobutanecarboxylic acid transport in xenopus laevis oocytes expressing human asct2 and snat2. Nucl. Med. Biol. 2013, 40, 670–675. [Google Scholar] [CrossRef] [PubMed]
- Oka, S.; Okudaira, H.; Yoshida, Y.; Schuster, D.M.; Goodman, M.M.; Shirakami, Y. Transport mechanisms of trans-1-amino-3-fluoro[1-(14)c]cyclobutanecarboxylic acid in prostate cancer cells. Nucl. Med. Biol. 2012, 39, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Tade, F.I.; Cohen, M.A.; Styblo, T.M.; Odewole, O.A.; Holbrook, A.I.; Newell, M.S.; Savir-Baruch, B.; Li, X.; Goodman, M.M.; Nye, J.A.; et al. Anti-3–18f-facbc (18f-fluciclovine) pet/ct of breast cancer: An exploratory study. J. Nucl. Med. 2016, 57, 1357–1363. [Google Scholar] [CrossRef] [PubMed]
- Ulaner, G.A.; Goldman, D.A.; Gonen, M.; Pham, H.; Castillo, R.; Lyashchenko, S.K.; Lewis, J.S.; Dang, C. Initial results of a prospective clinical trial of 18f-fluciclovine pet/ct in newly diagnosed invasive ductal and invasive lobular breast cancers. J. Nucl. Med. 2016, 57, 1350–1356. [Google Scholar] [CrossRef] [PubMed]
- Kaim, A.H.; Weber, B.; Kurrer, M.O.; Westera, G.; Schweitzer, A.; Gottschalk, J.; von Schulthess, G.K.; Buck, A. (18)f-fdg and (18)f-fet uptake in experimental soft tissue infection. Eur. J. Nucl. Med. Mol. Imaging 2002, 29, 648–654. [Google Scholar] [CrossRef] [PubMed]
- Rau, F.C.; Weber, W.A.; Wester, H.J.; Herz, M.; Becker, I.; Kruger, A.; Schwaiger, M.; Senekowitsch-Schmidtke, R. O-(2-[(18)f]fluoroethyl)- l-tyrosine (fet): A tracer for differentiation of tumour from inflammation in murine lymph nodes. Eur. J. Nucl. Med. Mol. Imaging 2002, 29, 1039–1046. [Google Scholar] [CrossRef] [PubMed]
- Ren, W.; Liu, G.; Yin, J.; Tan, B.; Wu, G.; Bazer, F.W.; Peng, Y.; Yin, Y. Amino-acid transporters in t-cell activation and differentiation. Cell Death Dis. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Baek, S.; Choi, C.M.; Ahn, S.H.; Lee, J.W.; Gong, G.; Ryu, J.S.; Oh, S.J.; Bacher-Stier, C.; Fels, L.; Koglin, N.; et al. Exploratory clinical trial of (4s)-4-(3-[18f]fluoropropyl)-l-glutamate for imaging xc- transporter using positron emission tomography in patients with non-small cell lung or breast cancer. Clin. Cancer Res. 2012, 18, 5427–5437. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Jenni, S.; Colovic, M.; Merkens, H.; Poleschuk, C.; Rodrigo, I.; Miao, Q.; Johnson, B.F.; Rishel, M.J.; Sossi, V.; et al. (18)f-5-fluoroaminosuberic acid as a potential tracer to gauge oxidative stress in breast cancer models. J. Nucl. Med. 2017, 58, 367–373. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kopka, K.; Riemann, B.; Friedrich, M.; Winters, S.; Halfter, H.; Weckesser, M.; Stögbauer, F.; Bernd Ringelstein, E.; Schober, O. Characterization of 3-[123i]iodo-l-α-methyl tyrosine transport in astrocytes of neonatal rats. J. Neurochem. 2001, 76, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Jager, P.L.; Franssen, E.J.; Kool, W.; Szabo, B.G.; Hoekstra, H.J.; Groen, H.J.; de Vries, E.G.; van Imhoff, G.W.; Vaalburg, W.; Piers, D.A. Feasibility of tumor imaging using l-3-[iodine-123]-iodo-alpha-methyl-tyrosine in extracranial tumors. J. Nucl. Med. 1998, 39, 1736–1743. [Google Scholar] [PubMed]
- Sharma, S.; Singh, B.; Mishra, A.K.; Rathod, D.; Hazari, P.P.; Chuttani, K.; Chopra, S.; Singh, P.M.; Abrar, M.L.; Mittal, B.R.; et al. Lat-1 based primary breast cancer detection by [99m]tc-labeled dtpa-bis-methionine scintimammography: First results using indigenously developed single vial kit preparation. Cancer Biother. Radiopharm. 2014, 29, 283–288. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Di, X.; Wu, M.; Sun, Z.; Zhong, L.; Wang, Y.; Fu, Q.; Kan, Q.; Sun, J.; He, Z. Targeting tumor highly-expressed lat1 transporter with amino acid-modified nanoparticles: Toward a novel active targeting strategy in breast cancer therapy. Nanomedicine 2017, 13, 987–998. [Google Scholar] [CrossRef] [PubMed]
- Ong, Z.Y.; Chen, S.; Nabavi, E.; Regoutz, A.; Payne, D.J.; Elson, D.S.; Dexter, D.T.; Dunlop, I.E.; Porter, A.E. Multibranched gold nanoparticles with intrinsic lat-1 targeting capabilities for selective photothermal therapy of breast cancer. ACS Appl. Mater. Interfaces 2017, 9, 39259–39270. [Google Scholar] [CrossRef] [PubMed]
- Cory, J.G.; Cory, A.H. Critical roles of glutamine as nitrogen donors in purine and pyrimidine nucleotide synthesis: Asparaginase treatment in childhood acute lymphoblastic leukemia. In Vivo 2006, 20, 587–589. [Google Scholar] [PubMed]
- Metallo, C.M.; Gameiro, P.A.; Bell, E.L.; Mattaini, K.R.; Yang, J.; Hiller, K.; Jewell, C.M.; Johnson, Z.R.; Irvine, D.J.; Guarente, L.; et al. Reductive glutamine metabolism by idh1 mediates lipogenesis under hypoxia. Nature 2011, 481, 380–384. [Google Scholar] [CrossRef] [PubMed]
- Yudkoff, M.; Pleasure, D.; Cregar, L.; Lin, Z.P.; Nissim, I.; Stern, J.; Nissim, I. Glutathione turnover in cultured astrocytes: Studies with [15n]glutamate. J. Neurochem. 1990, 55, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Yanagida, O.; Kanai, Y.; Chairoungdua, A.; Kim, D.K.; Segawa, H.; Nii, T.; Cha, S.H.; Matsuo, H.; Fukushima, J.; Fukasawa, Y.; et al. Human l-type amino acid transporter 1 (lat1): Characterization of function and expression in tumor cell lines. Biochim. Biophys. Acta 2001, 1514, 291–302. [Google Scholar] [CrossRef]
- Duran, R.V.; Oppliger, W.; Robitaille, A.M.; Heiserich, L.; Skendaj, R.; Gottlieb, E.; Hall, M.N. Glutaminolysis activates rag-mtorc1 signaling. Mol. Cell 2012, 47, 349–358. [Google Scholar] [CrossRef] [PubMed]
- DeBerardinis, R.J.; Cheng, T. Q’s next: The diverse functions of glutamine in metabolism, cell biology and cancer. Oncogene 2010, 29, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Terunuma, A.; Putluri, N.; Mishra, P.; Mathe, E.A.; Dorsey, T.H.; Yi, M.; Wallace, T.A.; Issaq, H.J.; Zhou, M.; Killian, J.K.; et al. Myc-driven accumulation of 2-hydroxyglutarate is associated with breast cancer prognosis. J. Clin. Investig. 2014, 124, 398–412. [Google Scholar] [CrossRef] [PubMed]
- Korangath, P.; Teo, W.W.; Sadik, H.; Han, L.; Mori, N.; Huijts, C.M.; Wildes, F.; Bharti, S.; Zhang, Z.; Santa-Maria, C.A.; et al. Targeting glutamine metabolism in breast cancer with aminooxyacetate. Clin. Cancer Res. 2015, 21, 3263–3273. [Google Scholar] [CrossRef] [PubMed]
- Haider, S.; McIntyre, A.; van Stiphout, R.G.; Winchester, L.M.; Wigfield, S.; Harris, A.L.; Buffa, F.M. Genomic alterations underlie a pan-cancer metabolic shift associated with tumour hypoxia. Genome Biol. 2016, 17. [Google Scholar] [CrossRef] [PubMed]
- Cao, M.D.; Lamichhane, S.; Lundgren, S.; Bofin, A.; Fjosne, H.; Giskeodegard, G.F.; Bathen, T.F. Metabolic characterization of triple negative breast cancer. BMC Cancer 2014, 14. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Chen, Y.; Olopade, O.I. Myc and breast cancer. Genes Cancer 2010, 1, 629–640. [Google Scholar] [CrossRef] [PubMed]
- Perou, C.M.; Sorlie, T.; Eisen, M.B.; van de Rijn, M.; Jeffrey, S.S.; Rees, C.A.; Pollack, J.R.; Ross, D.T.; Johnsen, H.; Akslen, L.A.; et al. Molecular portraits of human breast tumours. Nature 2000, 406, 747–752. [Google Scholar] [CrossRef] [PubMed]
- Sorlie, T.; Perou, C.M.; Tibshirani, R.; Aas, T.; Geisler, S.; Johnsen, H.; Hastie, T.; Eisen, M.B.; van de Rijn, M.; Jeffrey, S.S.; et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc. Natl. Acad. Sci. USA 2001, 98, 10869–10874. [Google Scholar] [CrossRef] [PubMed]
- Hilvo, M.; Denkert, C.; Lehtinen, L.; Muller, B.; Brockmoller, S.; Seppanen-Laakso, T.; Budczies, J.; Bucher, E.; Yetukuri, L.; Castillo, S.; et al. Novel theranostic opportunities offered by characterization of altered membrane lipid metabolism in breast cancer progression. Cancer Res. 2011, 71, 3236–3245. [Google Scholar] [CrossRef] [PubMed]
- Budczies, J.; Denkert, C.; Muller, B.M.; Brockmoller, S.F.; Klauschen, F.; Gyorffy, B.; Dietel, M.; Richter-Ehrenstein, C.; Marten, U.; Salek, R.M.; et al. Remodeling of central metabolism in invasive breast cancer compared to normal breast tissue—A gc-tofms based metabolomics study. BMC Genom. 2012, 13. [Google Scholar] [CrossRef] [PubMed]
- Kanaan, Y.M.; Sampey, B.P.; Beyene, D.; Esnakula, A.K.; Naab, T.J.; Ricks-Santi, L.J.; Dasi, S.; Day, A.; Blackman, K.W.; Frederick, W.; et al. Metabolic profile of triple-negative breast cancer in african-american women reveals potential biomarkers of aggressive disease. Cancer Genom. Proteom. 2014, 11, 279–294. [Google Scholar]
- Gross, M.I.; Demo, S.D.; Dennison, J.B.; Chen, L.; Chernov-Rogan, T.; Goyal, B.; Janes, J.R.; Laidig, G.J.; Lewis, E.R.; Li, J.; et al. Antitumor activity of the glutaminase inhibitor cb-839 in triple-negative breast cancer. Mol. Cancer Ther. 2014, 13, 890–901. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Cui, H.; Fang, J.; Deng, H.; Kuang, P.; Guo, H.; Wang, X.; Zhao, L. Glutamine deprivation plus bptes alters etoposide- and cisplatin-induced apoptosis in triple negative breast cancer cells. Oncotarget 2016, 7, 54691–54701. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Xu, H.; Di Antonio, M.; McKinney, S.; Mathew, V.; Ho, B.; O’Neil, N.J.; Santos, N.D.; Silvester, J.; Wei, V.; Garcia, J.; et al. Cx-5461 is a DNA g-quadruplex stabilizer with selective lethality in brca1/2 deficient tumours. Nat. Commun. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Seong, M.W.; Kim, K.H.; Chung, I.Y.; Kang, E.; Lee, J.W.; Park, S.K.; Lee, M.H.; Lee, J.E.; Noh, D.Y.; Son, B.H.; et al. A multi-institutional study on the association between brca1/brca2 mutational status and triple-negative breast cancer in familial breast cancer patients. Breast Cancer Res. Treat. 2014, 146, 63–69. [Google Scholar] [CrossRef] [PubMed]
| Gene | Synonym | Transport Mechanism | Substrates | Inhibitor | Imaging Tracer | Drug Delivery Candidate |
|---|---|---|---|---|---|---|
| SLC1A5 | ASCT2 | Obligatory exchange; Na+/AA exchanged for Na+/AA | Ala, Ser, Cys, Thr, Gln | 1,25-dihydroxyvitamin D | [(18)F]FPhPA; (18)F-FACBC | |
| SLC6A14 | ATB0,+ | Unidirectional; Na+/Cl−/AA0,+ symport | All neutral amino acids; All cationic amino acids | α-methyl-dl-tryptophan | ||
| SLC7A5 | LAT1 | Obligatory exchange; AA exchanged for AA | Large neutral amino acids | BCH (2-amino-2-norbornane-carboxylic acid) JHP203 | [(18)F]FPhPA; (18)F-FACBC; [99m]Tc-labeled diethylenetriaminepentaaceticacid (DTPA-bis)-methionine | Polylactic-co-glycolic acid (PLGA) nanoparticles; AuNPs |
| SLC7A11 | xCT | Obligatory exchange; AA exchanged for AA | Cystine, glutamate | Sulfasalazine; Erastin | [(18)F]FSPG 18F-5-FASu | |
| SLC3A1 | rBAT | Obligatory exchange; AA exchanged for AA | cystine and neutral and dibasic amino acids | |||
| SLC7A8 | LAT2 | Obligatory exchange; AA exchanged for AA | Large neutral amino acids | |||
| SLC38A1 | ATA1 | Obligatory exchange; Na+/AA exchanged for Na+/AA | All neutral amino acids |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cha, Y.J.; Kim, E.-S.; Koo, J.S. Amino Acid Transporters and Glutamine Metabolism in Breast Cancer. Int. J. Mol. Sci. 2018, 19, 907. https://doi.org/10.3390/ijms19030907
Cha YJ, Kim E-S, Koo JS. Amino Acid Transporters and Glutamine Metabolism in Breast Cancer. International Journal of Molecular Sciences. 2018; 19(3):907. https://doi.org/10.3390/ijms19030907
Chicago/Turabian StyleCha, Yoon Jin, Eun-Sol Kim, and Ja Seung Koo. 2018. "Amino Acid Transporters and Glutamine Metabolism in Breast Cancer" International Journal of Molecular Sciences 19, no. 3: 907. https://doi.org/10.3390/ijms19030907
APA StyleCha, Y. J., Kim, E.-S., & Koo, J. S. (2018). Amino Acid Transporters and Glutamine Metabolism in Breast Cancer. International Journal of Molecular Sciences, 19(3), 907. https://doi.org/10.3390/ijms19030907








