Dietary Plants for the Prevention and Management of Kidney Stones: Preclinical and Clinical Evidence and Molecular Mechanisms
Abstract
1. Introduction
2. Literature Search Methodology
3. Role of Natural Diet in the Prevention of Kidney Stones
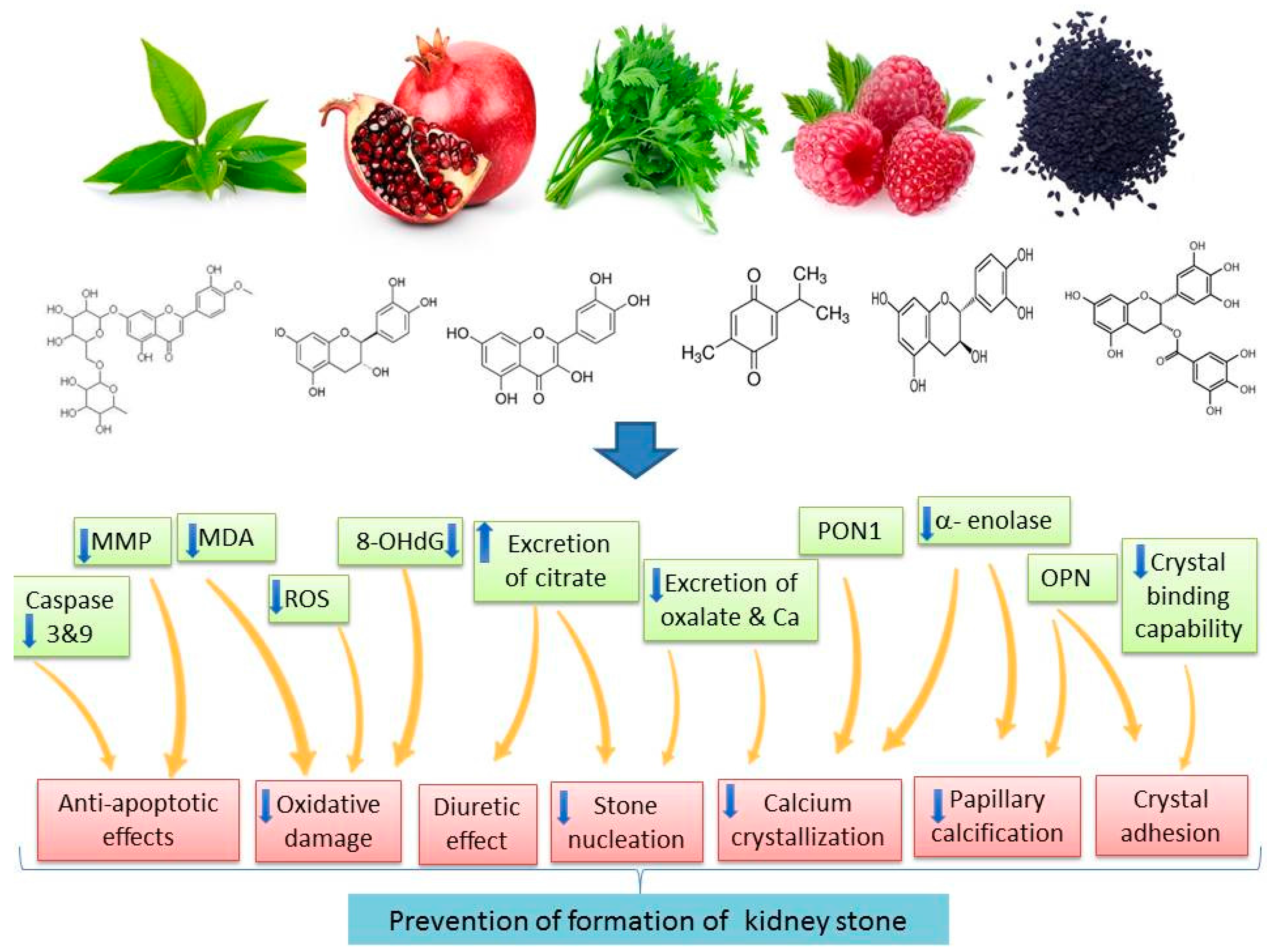
4. Dietary Plants for the Prevention of Kidney Stones
4.1. Green Tea
4.2. Raspberry
4.3. Rubia cordifolia
4.4. Parsley
4.5. Pomegranate (Punica granatum)
4.6. Pistacia lentiscus
4.7. Solanum xanthocarpum
4.8. Urtica dioica
4.9. Dolichos biflorus
4.10. Ammi visnaga
4.11. Nigella sativa
4.12. Hibiscus sabdariffa
4.13. Origanum vulgare
5. Medicinal Plants and Phytoconstituents as Dietary Supplements for the Prevention of Kidney Stones
6. Effect of Pharmacologically Active Phytochemicals on the Inhibition of Urolithiasis
7. Major Pharmacological Mechanisms of Plants and Natural Products in the Prevention of Kidney Stones
8. Concluding Remarks
Acknowledgments
Author Contributions
Conflicts of Interest
References
- López, M.; Hoppe, B. History, epidemiology and regional diversities of urolithiasis. Pediatr. Nephrol. 2010, 25, 49–59. [Google Scholar] [CrossRef] [PubMed]
- Tiselius, H.G. Epidemiology and medical management of stone disease. BJU Int. 2003, 91, 758–767. [Google Scholar] [CrossRef] [PubMed]
- Moe, O.W. Kidney stones: Pathophysiology and medical management. Lancet 2006, 367, 333–344. [Google Scholar] [CrossRef]
- Gindi, S.; Methra, T.; Chandu, B.R.; Boyina, R.; Dasari, V. Antiurolithiatic and invitro anti-oxidant activity of leaves of Ageratum conyzoides in rat. World J. Pharm. Pharm. Sci. 2013, 2, 636–649. [Google Scholar]
- Heron, S.; Yarnell, E. Recurrent kidney stones: A naturopathic approach. Altern. Complement. Ther. 1998, 4, 60–67. [Google Scholar] [CrossRef]
- Aggarwal, A.; Tandon, S.; Singla, S.; Tandon, C. Diminution of oxalate induced renal tubular epithelial cell injury and inhibition of calcium oxalate crystallization in vitro by aqueous extract of Tribulus terrestris. Int. Braz. J. Urol. 2010, 36, 480–489. [Google Scholar] [CrossRef] [PubMed]
- Baumann, J.M. Stone preventionhy so little progress? Urol. Res. 1998, 26, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Baynes, R.; Riviere, J. Risks associated with melamine and related triazine contamination of food. Emerg. Health Threats J. 2010, 3, e5. [Google Scholar] [CrossRef] [PubMed]
- Mandavia, D.R.; Patel, M.K.; Patel, J.C.; Anovadiya, A.P.; Baxi, S.N.; Tripathi, C.R. Anti-urolithiatic effect of ethanolic extract of Pedalium murex linn. fruits on ethylene glycol-induced renal calculi. Urol. J. 2013, 10, 946–952. [Google Scholar] [PubMed]
- Miller, N.L.; Lingeman, J.E. Management of kidney stones. Br. Med. J. 2007, 334, 468. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.; Bashir, S.; Khan, S.R.; Gilani, A.H. Antiurolithic activity of Origanum vulgare is mediated through multiple pathways. BMC Complement. Altern. Med. 2011, 11, 96. [Google Scholar] [CrossRef] [PubMed]
- Rathod, N.; Biswas, D.; Chitme, H.; Ratna, S.; Muchandi, I.; Chandra, R. Anti-urolithiatic effects of Punica granatum in male rats. J. Ethnopharmacol. 2012, 140, 234–238. [Google Scholar] [CrossRef] [PubMed]
- Coe, F.L.; Parks, J.H.; Asplin, J.R. The pathogenesis and treatment of kidney stones. N. Engl. J. Med. 1992, 327, 1141–1152. [Google Scholar] [CrossRef] [PubMed]
- Curhan, G.C.; Willett, W.C.; Knight, E.L.; Stampfer, M.J. Dietary factors and the risk of incident kidney stones in younger women: Nurses’ Health Study II. Arch. Intern. Med. 2004, 164, 885–891. [Google Scholar] [CrossRef] [PubMed]
- Taylor, E.N.; Stampfer, M.J.; Curhan, G.C. Dietary factors and the risk of incident kidney stones in men: New insights after 14 years of follow-up. J. Am. Soc. Nephrol. 2004, 15, 3225–3232. [Google Scholar] [CrossRef] [PubMed]
- Meschi, T.; Nouvenne, A.; Ticinesi, A.; Prati, B.; Guerra, A.; Allegri, F.; Pigna, F.; Soldati, L.; Vezzoli, G.; Gambaro, G. Dietary habits in women with recurrent idiopathic calcium nephrolithiasis. J. Transl. Med. 2012, 10, 63. [Google Scholar] [CrossRef] [PubMed]
- Goraya, N.; Wesson, D.E. Dietary interventions to improve outcomes in chronic kidney disease. Curr. Opin. Nephrol. Hypertens. 2015, 24, 505–510. [Google Scholar] [CrossRef] [PubMed]
- Meschi, T.; Maggiore, U.; Fiaccadori, E.; Schianchi, T.; Bosi, S.; Adorni, G.; Ridolo, E.; Guerra, A.; Allegri, F.; Novarini, A. The effect of fruits and vegetables on urinary stone risk factors. Kidney Int. 2004, 66, 2402–2410. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Sinha, A.K.; Makkar, H.P.; Becker, K. Dietary roles of phytate and phytase in human nutrition: A review. Food Chem. 2010, 120, 945–959. [Google Scholar] [CrossRef]
- Seltzer, M.A.; Low, R.K.; McDonald, M.; Shami, G.S.; Stoller, M.L. Dietary manipulation with lemonade to treat hypocitraturic calcium nephrolithiasis. J. Urol. 1996, 156, 907–909. [Google Scholar] [CrossRef]
- Sorensen, M.D.; Hsi, R.S.; Chi, T.; Shara, N.; Wactawski-Wende, J.; Kahn, A.J.; Wang, H.; Hou, L.; Stoller, M.L. Dietary intake of fiber, fruit and vegetables decreases the risk of incident kidney stones in women Women’s Health Initiative report. J. Urol. 2014, 192, 1694–1699. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.W.; Choi, J.S.; Yoon, G.S.; Yang, E.K.; Kim, D.Y. Effect of green tea on calcium oxalate stone formation and excretion in ethylene glycol-treated rats. Korean J. Urol. 2005, 46, 299–305. [Google Scholar]
- Itoh, Y.; Yasui, T.; Okada, A.; Tozawa, K.; Hayashi, Y.; Kohri, K. Preventive effects of green tea on renal stone formation and the role of oxidative stress in nephrolithiasis. J. Urol. 2005, 173, 271–275. [Google Scholar] [CrossRef] [PubMed]
- Ghalayini, I.F.; Al-Ghazo, M.A.; Harfeil, M.N. Prophylaxis and therapeutic effects of raspberry (Rubus idaeus) on renal stone formation in Balb/c mice. Int. Braz. J. Urol. 2011, 37, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, Z.; Yang, Y.; Zu, X.; Guan, D.; Guan, Y. Diuretic activity of Rubus idaeus L (Rosaceae) in rats. Trop. J. Pharm. Res. 2011, 10. [Google Scholar] [CrossRef]
- Divakar, K.; Pawar, A.; Chandrasekhar, S.; Dighe, S.; Divakar, G. Protective effect of the hydro-alcoholic extract of Rubia cordifolia roots against ethylene glycol induced urolithiasis in rats. Food Chem. Toxicol. 2010, 48, 1013–1018. [Google Scholar] [CrossRef] [PubMed]
- Al-Yousofy, F.; Gumaih, H.; Ibrahim, H.; Alasbahy, A. Parsley! Mechanism as antiurolithiasis remedy. Am. J. Clin. Exp. Urol. 2017, 5, 55. [Google Scholar] [PubMed]
- Jafar, S.; Mehri, L.; Hadi, B. The antiurolithiasic activity of aqueous extract of Petroselinum sativum on ethylene glycolinduced kidney calculi in rats. In Proceedings of the International Conference on Chemical, Environmental and Biological Sciences, Pattaya, Thailand, 7–8 October 2011; pp. 1577–1583. [Google Scholar]
- Saeidi, J.; Bozorgi, H.; Zendehdel, A.; Mehrzad, J. Therapeutic effects of aqueous extracts of Petroselinum sativum on ethylene glycol-induced kidney calculi in rats. Urol. J. 2012, 9, 361–366. [Google Scholar] [PubMed]
- Patel, P.; Patel, M.; Saralai, M.; Gandhi, T. Antiurolithiatic effects of Solanum xanthocarpum fruit extract on ethylene-glycol-induced nephrolithiasis in rats. J. Young Pharm. 2012, 4, 164–170. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Li, N.; Li, K.; Li, P. Protective effect of Urtica dioica methanol extract against experimentally induced urinary calculi in rats. Mol. Med. Rep. 2014, 10, 3157–3162. [Google Scholar] [CrossRef] [PubMed]
- Vanachayangkul, P.; Chow, N.; Khan, S.; Butterweck, V. Prevention of renal crystal deposition by an extract of Ammi visnaga L. and its constituents khellin and visnagin in hyperoxaluric rats. Urol. Res. 2011, 39, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Khoei, A.; Hadjzadeh, Z.; Parizady, M. Ethanolic extract of nigella sativa L seeds on ethylene glycol-induced kidney calculi in rats. Urol. J. 2009, 4, 86–90. [Google Scholar]
- Hajzadeh, M.; Mohammadian, N.; Rahmani, Z.; Rassouli, F.B. Effect of thymoquinone on ethylene glycol-induced kidney calculi in rats. Urol. J. 2008, 5, 149–155. [Google Scholar]
- Li, X.; Liang, Q.; Sun, Y.; Diao, L.; Qin, Z.; Wang, W.; Lu, J.; Fu, S.; Ma, B.; Yue, Z. Potential mechanisms responsible for the antinephrolithic effects of an aqueous extract of Fructus aurantii. Evid.-Based Complement. Altern. Med. 2015. [Google Scholar] [CrossRef] [PubMed]
- Laikangbam, R.; Devi, M.D. Inhibition of calcium oxalate crystal deposition on kidneys of urolithiatic rats by Hibiscus sabdariffa L. extract. Urol. Res. 2012, 40, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Meiouet, F.; El Kabbaj, S.; Daudon, M. In vitro study of the litholytic effects of herbal extracts on cystine urinary calculi. Prog. Urol. 2011, 21, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Cheraft-Bahloul, N.; Husson, C.; Ourtioualous, M.; Sinaeve, S.; Atmani, D.; Stévigny, C.; Nortier, J.L.; Antoine, M.-H. Protective Effects of Pistacia lentiscus L. fruit extract against calcium oxalate monohydrate induced proximal tubular injury. J. Ethnopharmacol. 2017, 209, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Saha, S.; Verma, R.J. Evaluation of hydro-alcoholic extract of Dolichos biflorus seeds on inhibition of calcium oxalate crystallization. J. Herb. Med. 2015, 5, 41–47. [Google Scholar] [CrossRef]
- Atodariya, U.; Barad, R.; Upadhyay, S.; Upadhyay, U. Anti-urolithiatic activity of Dolichos biflorus seeds. J. Pharmacogn. Phytochem. 2013, 2, 45051. [Google Scholar]
- Jeong, B.C.; Kim, B.S.; Kim, J.I.; Kim, H.H. Effects of green tea on urinary stone formationn in vivo and in vitro study. J. Endourol. 2006, 20, 356–361. [Google Scholar] [CrossRef] [PubMed]
- Tracy, C.R.; Henning, J.R.; Newton, M.R.; Aviram, M.; Zimmerman, M.B. Oxidative stress and nephrolithiasis: A comparative pilot study evaluating the effect of pomegranate extract on stone risk factors and elevated oxidative stress levels of recurrent stone formers and controls. Urolithiasis 2014, 42, 401–408. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.G.; Behura, S.K.; Kumar, R. Litholytic property of Kulattha (Dolichous biflorus) vs. potassium citrate in renal calculus disease comparative study. JAPI 2010, 58, 286–289. [Google Scholar] [PubMed]
- Prasongwatana, V.; Woottisin, S.; Sriboonlue, P.; Kukongviriyapan, V. Uricosuric effect of Roselle (Hibiscus sabdariffa) in normal and renal-stone former subjects. J. Ethnopharmacol. 2008, 117, 491–495. [Google Scholar] [CrossRef] [PubMed]
- Rodgers, A.; Mokoena, M.; Durbach, I.; Lazarus, J.; de Jager, S.; Ackermann, H.; Breytenbach, I.; Okada, A.; Usami, M.; Hirose, Y. Do teas rich in antioxidants reduce the physicochemical and peroxidative risk factors for calcium oxalate nephrolithiasis in humans? Pilot studies with Rooibos herbal tea and Japanese green tea. Urolithiasis 2016, 44, 299–310. [Google Scholar] [CrossRef] [PubMed]
- Anttonen, M.J.; Karjalainen, R.O. Environmental and genetic variation of phenolic compounds in red raspberry. J. Food Compos. Anal. 2005, 18, 759–769. [Google Scholar] [CrossRef]
- Bhatt, P.; Kushwah, A. Rubia cordifolia overview: A new approach to treat cardiac disorders. Int. J. Drug Dev. Res. 2013, 5, 47–54. [Google Scholar]
- Deshkar, N.; Tilloo, S.; Pande, V. A comprehensive review of Rubia cordifolia Linn. Pharmacogn. Rev. 2008, 2, 124–134. [Google Scholar]
- Dev, S. Selection of Prime Ayurvedic Plant Drugs; Anamaya Publishers: New Delhi, India, 2006. [Google Scholar]
- Son, J.K.; Jung, S.J.; Jung, J.H.; Fang, Z.; Lee, C.S.; Seo, C.S.; Moon, D.C.; Min, B.S.; Kim, M.R.; Woo, M.H. Anticancer Constituents from the Roots of Rubia cordifolia L. Chem. Pharm. Bull. 2008, 56, 213–216. [Google Scholar] [CrossRef] [PubMed]
- Lodi, S.; Sharma, V.; Kansal, L. The protective effect of Rubia cordifolia against lead nitrate-induced immune response impairment and kidney oxidative damage. Indian J. Pharm. 2011, 43, 441–444. [Google Scholar] [CrossRef] [PubMed]
- Farzaei, M.H.; Abbasabadi, Z.; Ardekani, M.R.S.; Rahimi, R.; Farzaei, F. Parsley: A review of ethnopharmacology, phytochemistry and biological activities. J. Tradit. Chin. Med. 2013, 33, 815–826. [Google Scholar] [CrossRef]
- Zhang, H.; Chen, F.; Wang, X.; Yao, H.-Y. Evaluation of antioxidant activity of parsley (Petroselinum crispum) essential oil and identification of its antioxidant constituents. Food Res. Int. 2006, 39, 833–839. [Google Scholar] [CrossRef]
- Moazedi, A.; Mirzaie, D.; Seyyednejad, S.; Zadkarami, M.; Amirzargar, A. Spasmolytic effect of Petroselinum crispum (Parsley) on rat’s ileum at different calcium chloride concentrations. Pak. J. Biol. Sci. 2007, 10, 4036–4042. [Google Scholar] [PubMed]
- Rezazad, M.; Farokhi, F. Protective effect of Petroselinum crispum extract in abortion using prostadin-induced renal dysfunction in female rats. Avicenna J. Phytomed. 2014, 4, 312–319. [Google Scholar] [PubMed]
- Kreydiyyeh, S.I.; Usta, J. Diuretic effect and mechanism of action of parsley. J. Ethnopharmacol. 2002, 79, 353–357. [Google Scholar] [CrossRef]
- Vamenta-Morris, H.; Dreisbach, A.; Shoemaker-Moyle, M.; Abdel-Rahman, E.M. Internet claims on dietary and herbal supplements in advanced nephropathyruth or myth. Am. J. Nephrol. 2014, 40, 393–398. [Google Scholar] [CrossRef] [PubMed]
- Kuźma, P.; Drużyńska, B.; Obiedziński, M. Optimization of extraction conditions of some polyphenolic compounds from parsley leaves (Petroselinum crispum). Acta Sci. Pol. Technol. Aliment. 2014, 13, 145–154. [Google Scholar] [CrossRef] [PubMed]
- Jurenka, J. Therapeutic applications of pomegranate (Punica granatum L.): A review. Altern. Med. Rev. 2008, 13, 128–144. [Google Scholar] [PubMed]
- Bhandari, P.R. Pomegranate (Punica granatum L). Ancient seeds for modern cure? Review of potential therapeutic applications. Int. J. Nutr. Pharmacol. Neurol. Dis. 2012, 2, 171–184. [Google Scholar] [CrossRef]
- Tugcu, V.; Kemahli, E.; Ozbek, E.; Arinci, Y.V.; Uhri, M.; Erturkuner, P.; Metin, G.; Seckin, I.; Karaca, C.; Ipekoglu, N. Protective effect of a potent antioxidant, pomegranate juice, in the kidney of rats with nephrolithiasis induced by ethylene glycol. J. Endourol. 2008, 22, 2723–2732. [Google Scholar] [CrossRef] [PubMed]
- Elwej, A.; Ghorbel, I.; Marrekchi, R.; Boudawara, O.; Jamoussi, K.; Boudawara, T.; Zeghal, N.; Sefi, M. Improvement of kidney redox states contributes to the beneficial effects of dietary pomegranate peel against barium chloride-induced nephrotoxicity in adult rats. Arch. Physiol. Biochem. 2016, 122, 130–140. [Google Scholar] [CrossRef] [PubMed]
- Boroushaki, M.T.; Asadpour, E.; Sadeghnia, H.R.; Dolati, K. Effect of pomegranate seed oil against gentamicin-induced nephrotoxicity in rat. J. Food Sci. Technol. 2014, 51, 3510–3514. [Google Scholar] [CrossRef] [PubMed]
- Alimoradian, A.; Changizi-Ashtiyani, S.; Ghiasabadi Farahani, A.; Kheder, L.; Rajabi, R.; Sharifi, A. Protective Effects of Pomegranate Juice on Nephrotoxicity Induced by Captopril and Gentamicin in Rats. Iran. J. Kidney Dis. 2017, 11, 422–429. [Google Scholar] [PubMed]
- Sadeghi, F.; Nematbakhsh, M.; Noori-Diziche, A.; Eshraghi-Jazi, F.; Talebi, A.; Nasri, H.; Mansouri, A.; Dehghani, A.; Saberi, S.; Shirdavani, S.; et al. Protective effect of pomegranate flower extract against gentamicin-induced renal toxicity in male rats. J. Ren. Inj. Prev. 2015, 4, 45–50. [Google Scholar] [PubMed]
- Primarizky, H.; Yuniarti, W.M.; Lukiswanto, B.S. Benefits of pomegranate (Punica granatum Linn) fruit extracts to weight changes, total protein, and uric acid in white rats (Rattus norvegicus) as an animal model of acute renal failure. Vet. World 2016, 9, 1269–1274. [Google Scholar] [CrossRef] [PubMed]
- Sharifiyan, F.; Movahedian-Attar, A.; Nili, N.; Asgary, S. Study of pomegranate (Punica granatum L.) peel extract containing anthocyanins on fatty streak formation in the renal arteries in hypercholesterolemic rabbits. Adv. Biomed. Res. 2016, 5, 8. [Google Scholar] [PubMed]
- Bagri, P.; Ali, M.; Aeri, V.; Bhowmik, M.; Sultana, S. Antidiabetic effect of Punica granatum flowers: Effect on hyperlipidemia, pancreatic cells lipid peroxidation and antioxidant enzymes in experimental diabetes. Food Chem. Toxicol. 2009, 47, 50–54. [Google Scholar] [CrossRef] [PubMed]
- Ilbey, Y.O.; Ozbek, E.; Simsek, A.; Cekmen, M.; Somay, A.; Tasci, A.I. Effects of pomegranate juice on hyperoxaluria-induced oxidative stress in the rat kidneys. Ren. Fail. 2009, 31, 522–531. [Google Scholar] [CrossRef] [PubMed]
- Ljubuncic, P.; Song, H.; Cogan, U.; Azaizeh, H.; Bomzon, A. The effects of aqueous extracts prepared from the leaves of Pistacia lentiscus in experimental liver disease. J. Ethnopharmacol. 2005, 100, 198–204. [Google Scholar] [CrossRef] [PubMed]
- Benhammou, N.; Bekkara, F.A.; Panovska, T.K. Antioxidant and antimicrobial activities of the Pistacia lentiscus and Pistacia atlantica extracts. Afr. J. Pharm. Pharmacol. 2008, 2, 22–28. [Google Scholar]
- Kumar, S.; Pandey, A.K. Medicinal attributes of Solanum xanthocarpum fruit consumed by several tribal communities as foodn in vitro antioxidant, anticancer and anti HIV perspective. BMC Complement. Altern. Med. 2014, 14, 112. [Google Scholar] [CrossRef] [PubMed]
- Saha, S.; Goswami, G.; Pandrangi, A. Isolation and prevention of calcium oxalate-induced apoptotic death and oxidative stress in MDCK cells by diosgenin. Chem. Biol. Interact. 2014, 224, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Ranka, D.; Aswar, M.; Aswar, U.; Bodhankar, S. Diuretic potential of aqueous extract of roots of Solanum xanthocarpum Schrad & Wendl, a preliminary study. Indian J. Exp. Biol. 2013, 51, 833–839. [Google Scholar] [PubMed]
- Hussain, T.; Gupta, R.K.; Sweety, K.; Eswaran, B.; Vijayakumar, M.; Rao, C.V. Nephroprotective activity of Solanum xanthocarpum fruit extract against gentamicin–induced nephrotoxicity and renal dysfunction in experimental rodents. Asian Pac. J. Trop. Med. 2012, 5, 686–691. [Google Scholar] [CrossRef]
- Durak, I.; Biri, H.; Devrim, E.; Sözen, S.; Avcı, A. Aqueous extract of Urtica dioica makes significant inhibition on adenosine deaminase activity in prostate tissue from patients with prostate cancer. Cancer Biol. Ther. 2004, 3, 855–857. [Google Scholar] [CrossRef] [PubMed]
- Mathew, L.E.; Sindhu, G.; Helen, A. Dolichos biflorus exhibits anti-inflammatory and antioxidant properties in an acute inflammatory model. J. Food Drug Anal. 2014, 22, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Hayatdavoudi, P.; Rad, A.K.; Rajaei, Z.; Hadjzadeh, M.A.-R. Renal injury, nephrolithiasis and Nigella sativa: A mini review. Avicenna J. Phytomed. 2016, 6, 1–8. [Google Scholar] [PubMed]
- Amin, G.R. Popular Medicinal Plants of Iran; Iranian Research Institute of Medicinal Plants Tehran: Tehran, Iran, 1991; Volume 1. [Google Scholar]
- Woottisin, S.; Hossain, R.Z.; Yachantha, C.; Sriboonlue, P.; Ogawa, Y.; Saito, S. Effects of Orthosiphon grandiflorus, Hibiscus sabdariffa and Phyllanthus amarus extracts on risk factors for urinary calcium oxalate stones in rats. J. Urol. 2011, 185, 323–328. [Google Scholar] [CrossRef] [PubMed]
- Farzaei, M.H.; Abdollahi, M.; Rahimi, R. Role of dietary polyphenols in the management of peptic ulcer. World J. Gastroenterol. 2015, 21, 6499. [Google Scholar] [CrossRef] [PubMed]
- Gürocak, S.; Küpeli, B. Consumption of historical and current phytotherapeutic agents for urolithiasis: A critical review. J. Urol. 2006, 176, 450–455. [Google Scholar] [CrossRef] [PubMed]
- Alok, S.; Jain, S.K.; Verma, A.; Kumar, M.; Sabharwal, M. Pathophysiology of kidney, gallbladder and urinary stones treatment with herbal and allopathic medicine: A review. Asian Pac. J. Trop. Dis. 2013, 3, 496–504. [Google Scholar] [CrossRef]
- Lien, E.J.-C.; Lien, L.L.-M.; Wang, R.; Wang, J. Phytochemical analysis of medicinal plants with kidney protective activities. Chin. J. Integr. Med. 2012, 18, 790–800. [Google Scholar] [CrossRef] [PubMed]
- Saha, S.; Verma, R.J. Inhibition of calcium oxalate crystallisation in vitro by an extract of Bergenia ciliata. Arab J. Urol. 2013, 11, 187–192. [Google Scholar] [CrossRef] [PubMed]
- Bashir, S.; Gilani, A.H. Antiurolithic effect of Bergenia ligulata rhizome: An explanation of the underlying mechanisms. J. Ethnopharmacol. 2009, 122, 106–116. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, D.; Kaushal, R.; Kaur, T.; Bijarnia, R.K.; Puri, S.; Singla, S.K. The most potent antilithiatic agent ameliorating renal dysfunction and oxidative stress from Bergenia ligulata rhizome. J. Ethnopharmacol. 2014, 158, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, C.; Joshi, M.; Vaidya, A. Growth inhibition of struvite crystals in the presence of herbal extract Commiphora wightii. J. Mater. Sci. Mater. Med. 2009, 20, 85. [Google Scholar] [CrossRef] [PubMed]
- De Cógáin, M.R.; Linnes, M.P.; Lee, H.J.; Krambeck, A.E.; de Mendonça Uchôa, J.C.; Kim, S.-H.; Lieske, J.C. Aqueous extract of Costus arabicus inhibits calcium oxalate crystal growth and adhesion to renal epithelial cells. Urolithiasis 2015, 43, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Atmani, F.; Slimani, Y.; Mbark, A.; Bnouham, M.; Ramdani, A. In vitro and in vivo antilithiasic effect of saponin rich fraction isolated from Herniaria hirsute. J. Bras. Nefrol. 2006, 28, 199–203. [Google Scholar]
- Tayal, S.; Duggal, S.; Bandyopadhyay, P.; Aggarwal, A.; Tandon, S.; Tandon, C. Cytoprotective role of the aqueous extract of Terminalia chebula on renal epithelial cells. Int. Braz. J. Urol. 2012, 38, 204–214. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Aggarwal, A.; Tandon, S.; Kumar Singla, S.; Tandon, C. A novel antilithiatic protein from Tribulus terrestris having cytoprotective potency. Protein Pept. Lett. 2012, 19, 812–819. [Google Scholar] [CrossRef] [PubMed]
- Sathya, M.; Kokilavani, R.; Teepa, K.A.; Balakrishnan, A. Biopotency of Acalypha indica Linn on Membrane Bound ATPases and Marker Enzymes urolithic Rats. Anc. Sci. Life 2011, 31, 3–9. [Google Scholar] [PubMed]
- Soundararajan, P.; Mahesh, R.; Ramesh, T.; Begum, V.H. Effect of Aerva lanata on calcium oxalate urolithiasis in rats. Indian J. Exp. Biol. 2006, 44, 981–986. [Google Scholar] [PubMed]
- Ahmadi, M.; Rad, A.K.; Rajaei, Z.; Hadjzadeh, M.-A.-R.; Mohammadian, N.; Tabasi, N.S. Alcea rosea root extract as a preventive and curative agent in ethylene glycol-induced urolithiasis in rats. Indian J. Pharmacol. 2012, 44, 304–307. [Google Scholar] [PubMed]
- Christina, A.; Ashok, K.; Packialakshmi, M.; Tobin, G.; Preethi, J.; Murugesh, N. Antilithiatic effect of Asparagus racemosus Willd on ethylene glycol-induced lithiasis in male albino Wistar rats. Methods Find. Exp. Clin. Pharmacol. 2005, 27, 633–638. [Google Scholar] [CrossRef] [PubMed]
- Gadge, N.; Jalalpure, S. Curative treatment with extracts of Bombax ceiba fruit reduces risk of calcium oxalate urolithiasis in rats. Pharm. Biol. 2012, 50, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.-C.; Lai, M.-T.; Chen, H.-Y.; Ho, C.-Y.; Man, K.-M.; Shen, J.-L.; Lee, Y.-J.; Tsai, F.-J.; Chen, Y.-H.; Chen, W.-C. Protective effect of Flos carthami extract against ethylene glycol-induced urolithiasis in rats. Urol. Res. 2012, 40, 655–661. [Google Scholar] [CrossRef] [PubMed]
- Rad, A.K.; Rajaei, Z.; Mohammadian, N.; Valiollahi, S.; Sonei, M. The Beneficial effect of Cynodon Dactylon fractions on ethylene glycol-induced kidney calculi in rats. Urol. J. 2011, 8, 179–184. [Google Scholar]
- Orhan, N.; Onaran, M.; Şen, İ.; Gönül, İ.I.; Aslan, M. Preventive treatment of calcium oxalate crystal deposition with immortal flowers. J. Ethnopharmacol. 2015, 163, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Shah, J.G.; Patel, B.G.; Patel, S.B.; Patel, R.K. Antiurolithiatic and antioxidant activity of Hordeum vulgare seeds on ethylene glycol-induced urolithiasis in rats. Indian J. Pharmacol. 2012, 44, 672–677. [Google Scholar] [CrossRef] [PubMed]
- Ingale, K.G.; Thakurdesai, P.A.; Vyawahare, N.S. Effect of Hygrophila spinosa in ethylene glycol induced nephrolithiasis in rats. Indian J. Pharmacol. 2012, 44, 639–642. [Google Scholar] [CrossRef] [PubMed]
- Khalili, M.; Jalali, M.R.; Mirzaei-Azandaryani, M. Effect of hydroalcoholic extract of Hypericum perforatum L. leaves on ethylene glycol-induced kidney calculi in rats. Urol. J. 2012, 9, 472–479. [Google Scholar] [PubMed]
- Makasana, A.; Ranpariya, V.; Desai, D.; Mendpara, J.; Parekh, V. Evaluation for the anti-urolithiatic activity of Launaea procumbens against ethylene glycol-induced renal calculi in rats. Toxicol. Rep. 2014, 1, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.J.; Bae, W.J.; Kim, S.J.; Hong, S.H.; Lee, J.Y.; Hwang, T.-K.; Choi, Y.J.; Hwang, S.Y.; Kim, S.W. The inhibitory effect of an ethanol extract of the spores of Lygodium japonicum on ethylene glycol-induced kidney calculi in rats. Urolithiasis 2014, 42, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Akanae, W.; Tsujihata, M.; Yoshioka, I.; Nonomura, N.; Okuyama, A. Orthosiphon grandiflorum has a protective effect in a calcium oxalate stone forming rat model. Urol. Res. 2010, 38, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Bouanani, S.; Henchiri, C.; Migianu-Griffoni, E.; Aouf, N.; Lecouvey, M. Pharmacological and toxicological effects of Paronychia argentea in experimental calcium oxalate nephrolithiasis in rats. J. Ethnopharmacol. 2010, 129, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Vyas, B.; Vyas, R.; Joshi, S.; Santani, D. Antiurolithiatic activity of whole-plant hydroalcoholic extract of Pergularia daemia in rats. J. Young Pharm. 2011, 3, 36–40. [Google Scholar] [CrossRef] [PubMed]
- Moriyama, M.T.; Suga, K.; Miyazawa, K.; Tanaka, T.; Higashioka, M.; Noda, K.; Oka, M.; Tanaka, M.; Suzuki, K. Inhibitions of urinary oxidative stress and renal calcium level by an extract of Quercus salicina Blume/Quercus stenophylla Makino in a rat calcium oxalate urolithiasis model. Int. J. Urol. 2009, 16, 397–401. [Google Scholar] [CrossRef] [PubMed]
- Geetha, K.; Manavalan, R.; Venkappayya, D. Control of urinary risk factors of stone formation by Salvadora persica in experimental hyperoxaluria. Methods Find. Exp. Clin. Pharmacol. 2010, 32, 623–629. [Google Scholar] [CrossRef] [PubMed]
- Mirian, E.-C.M.; Juanita, N.-M.; Christophe, B.O.; Estela, M.-C.M. Molecular mechanisms involved in the protective effect of the chloroform extract of Selaginella lepidophylla (Hook. et Grev.) Spring in a lithiasic rat model. Urolithiasis 2013, 41, 205–215. [Google Scholar] [CrossRef] [PubMed]
- Brardi, S.; Imperiali, P.; Cevenini, G.; Verdacchi, T.; Ponchietti, R. Effects of the association of potassium citrate and agropyrum repens in renal stone treatment: Results of a prospective randomized comparison with potassium citrate. Arch. Ital. Urol. Androl. 2012, 84, 61–67. [Google Scholar] [PubMed]
- Micali, S.; Sighinolfi, M.; Celia, A.; de Stefani, S.; Grande, M.; Cicero, A.; Bianchi, G. Can Phyllanthus niruri affect the efficacy of extracorporeal shock wave lithotripsy for renal stones? A randomized, prospective, long-term study. J. Urol. 2006, 176, 1020–1022. [Google Scholar] [CrossRef] [PubMed]
- Joshi, V.S.; Parekh, B.B.; Joshi, M.J.; Vaidya, A.D. Inhibition of the growth of urinary calcium hydrogen phosphate dihydrate crystals with aqueous extracts of Tribulus terrestris and Bergenia ligulata. Urol. Res. 2005, 33, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Byahatti, V.V.; Pai, K.V.; D’Souza, M.G. Effect of phenolic compounds from Bergenia ciliata (Haw.) Sternb. leaves on experimental kidney stones. Anc. Sci. Life 2010, 30, 14–17. [Google Scholar] [PubMed]
- Han, X.; Shen, T.; Lou, H. Dietary polyphenols and their biological significance. Int. J. Mol. Sci. 2007, 8, 950–988. [Google Scholar] [CrossRef]
- Mendoza-Wilson, A.M.; Glossman-Mitnik, D. Theoretical study of the molecular properties and chemical reactivity of (+)-catechin and (−)-epicatechin related to their antioxidant ability. J. Mol. Struct. THEOCHEM 2006, 761, 97–106. [Google Scholar] [CrossRef]
- Higdon, J.V.; Frei, B. Tea catechins and polyphenols: Health effects, metabolism, and antioxidant functions. Crit. Rev. Food Sci. Nutr. 2003, 43, 89–143. [Google Scholar] [CrossRef] [PubMed]
- Amengual-Cladera, E.; Nadal-Casellas, A.; Gómez-Pérez, Y.; Gomila, I.; Prieto, R.M.; Proenza, A.M.; Lladó, I. Phytotherapy in a rat model of hyperoxaluriahe antioxidant effects of quercetin involve serum paraoxonase 1 activation. Exp. Biol. Med. 2011, 236, 1133–1138. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.A.; Priyamvada, S.; Farooq, N.; Khan, S.; Khan, M.W.; Yusufi, A.N. Protective effect of green tea extract on gentamicin-induced nephrotoxicity and oxidative damage in rat kidney. Pharmacol. Res. 2009, 59, 254–262. [Google Scholar] [CrossRef] [PubMed]
- Zhai, W.; Zheng, J.; Yao, X.; Peng, B.; Liu, M.; Huang, J.; Wang, G.; Xu, Y. Catechin prevents the calcium oxalate monohydrate induced renal calcium crystallization in NRK-52E cells and the ethylene glycol induced renal stone formation in rat. BMC Complement. Altern. Med. 2013, 13, 228. [Google Scholar] [CrossRef] [PubMed]
- Grases, F.; Prieto, R.M.; Fernandez-Cabot, R.A.; Costa-Bauzá, A.; Tur, F.; Torres, J.J. Effects of polyphenols from grape seeds on renal lithiasis. Oxid. Med. Cell. Longev. 2015, 2015, 813737. [Google Scholar] [CrossRef] [PubMed]
- Kanlaya, R.; Singhto, N.; Thongboonkerd, V. EGCG decreases binding of calcium oxalate monohydrate crystals onto renal tubular cells via decreased surface expression of alpha-enolase. J. Biol. Inorg. Chem. 2016, 21, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Crespo, M.; Galvez, J.; Cruz, T.; Ocete, M.; Zarzuelo, A. Anti-inflammatory activity of diosmin and hesperidin in rat colitis induced by TNBS. Planta Med. 1999, 65, 651–653. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Daim, M.M.; Khalifa, H.A.; Abushouk, A.I.; Dkhil, M.A.; Al-Quraishy, S.A. Diosmin Attenuates Methotrexate-Induced Hepatic, Renal, and Cardiac Injury: A Biochemical and Histopathological Study in Mice. Oxid. Med. Cell. Longev. 2017, 2017, 3281670. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Mundhe, N.; Borgohain, M.; Chowdhury, L.; Kwatra, M.; Bolshette, N.; Ahmed, A.; Lahkar, M. Diosmin Modulates the NF-κB Signal Transduction Pathways and Downregulation of Various Oxidative Stress Markers in Alloxan-Induced Diabetic Nephropathy. Inflammation 2016, 39, 1783–1797. [Google Scholar] [CrossRef] [PubMed]
- Rehman, M.U.; Tahir, M.; Quaiyoom Khan, A.; Khan, R.; Lateef, A.; Hamiza, O.O.; Ali, F.; Sultana, S. Diosmin protects against trichloroethylene-induced renal injury in Wistar ratslausible role of p53, Bax and caspases. Br. J. Nutr. 2013, 110, 699–710. [Google Scholar] [CrossRef] [PubMed]
- Schlottfeldt Fdos, S.; Fernandes, S.M.; Martins, D.M.; Cordeiro, P.; Fonseca, C.D.; Watanabe, M.; Vattimo Mde, F. Prevention of amphotericin B nephrotoxicity through use of phytotherapeutic medication. Rev. Esc. Enferm. USP 2015, 49, 74–79. [Google Scholar] [PubMed]
- Noorafshan, A.; Karbalay-Doust, S.; Karimi, F. Diosmin reduces calcium oxalate deposition and tissue degeneration in nephrolithiasis in rats: A stereological study. Korean J. Urol. 2013, 54, 252–257. [Google Scholar] [CrossRef] [PubMed]
- Prabhu, V.V.; Sathyamurthy, D.; Ramasamy, A.; Das, S.; Anuradha, M.; Pachiappan, S. Evaluation of protective effects of diosmin (a citrus flavonoid) in chemical-induced urolithiasis in experimental rats. Pharm. Biol. 2016, 54, 1513–1521. [Google Scholar] [CrossRef] [PubMed]
- Pastukhov, A.V.; Levchenko, L.A.; Sadkov, A.P. Spectroscopic study on binding of rutin to human serum albumin. J. Mol. Struct. 2007, 842, 60–66. [Google Scholar] [CrossRef]
- Colombo, R.; de L Batista, A.N.; Teles, H.L.; Silva, G.H.; Bomfim, G.C.; Burgos, R.C.; Cavalheiro, A.J.; da Silva Bolzani, V.; Silva, D.H.; Pelicia, C.R.; et al. Validated HPLC method for the standardization of Phyllanthus niruri (herb and commercial extracts) using corilagin as a phytochemical marker. Biomed. Chromatogr. BMC 2009, 23, 573–580. [Google Scholar] [CrossRef] [PubMed]
- Ghodasara, J.; Pawar, A.; Deshmukh, C.; Kuchekar, B. Inhibitory effect of rutin and curcumin on experimentally-induced calcium oxalate urolithiasis in rats. Pharmacogn. Res. 2010, 2, 388–392. [Google Scholar]
- Zhu, W.; Xu, Y.F.; Feng, Y.; Peng, B.; Che, J.P.; Liu, M.; Zheng, J.H. Prophylactic effects of quercetin and hyperoside in a calcium oxalate stone forming rat model. Urolithiasis 2014, 42, 519–526. [Google Scholar] [CrossRef] [PubMed]
- Park, H.K.; Jeong, B.C.; Sung, M.-K.; Park, M.-Y.; Choi, E.Y.; Kim, B.S.; Kim, H.H.; Kim, J.I. Reduction of oxidative stress in cultured renal tubular cells and preventive effects on renal stone formation by the bioflavonoid quercetin. J. Urol. 2008, 179, 1620–1626. [Google Scholar] [CrossRef] [PubMed]
- Mutalib, L.Y. Comparison between phytotherapy and conventional drug therapy used in urolithiasis management in Hawler city, Kurdistan Region\Iraq. J. Pharmacogn. Phytochem. 2015, 4, 83–86. [Google Scholar]
| Plant | Part of Plant | Study Type | Study Design | Results | Reference |
|---|---|---|---|---|---|
| Green tea (Camellia sinensis) | Leaves of kidney stones | In vivo | Ethylene glycol (EG)-induced nephrolithiasis in rat | ↓ Calcium crystal depositions in the kidneys ↓ The osteopontin mRNA level | [22] |
| Leaves | In vivo | EG-induced nephrolithiasis in rat | ↓ Urinary oxalate excretion, calcium oxalate deposit formation ↑ Sodium Oxide Dismutase (SOD) activity | [23] | |
| Rasberry (Rubus idaeus) | Aqueous extract | In vivo | Glyoxylate-induced calcium oxalate (CaOx) nephrolithiasis in mice | ↓ Generation of malondialdehyde (MDA) and protein carbonyls ↓ Urinary calcium and phosphorus levels ↓ The growth rate of calculus | [24] |
| Methanolic extract | In vivo | Bicarbonate saline solution (containing 110 mM NaCl and 30 mM NaHCO3) induced nephrolithiasis in rats | ↓ Activity of aldosterone or epithelial sodium channels ↑ Urine volume | [25] | |
| Common madder (Rubia cordifolia) | Hydro-alcoholic extract | In vivo | EG-induced urolithiasis | ↓ The growth of calcium oxalate crystals ↓ The formation of urinary oxalate ↑ Tubular citrate | [26] |
| Parsley (Petroselinum sativum Hoffm.) | Ethanolic extract | In vivo | EG+ ammonium chloride (AlCl3)-induced urolithiasis in rat | ↓ Urinary calcium and protein excretion ↑ Urinary pH | [27] |
| Aqueous Extract | In vivo | EG-induced urolithiasis in rats | ↓ Serum urea and uric acid concentrations ↑ Serum magnesium concentration | [28] | |
| Parsley (Petroselinum sativum Hoffm.) | Aerial parts and roots aqueous extract | In vivo | EG-feeding rats | ↓ The number of calcium oxalate deposits | [29] |
| Pomegranate (Punica granatum) | Fruits chloroform and methanol extract | In vivo | EG-induced urolithiasis | ↓ Urine oxalate, calcium and phosphate, renal tissue oxalates ↓ Serum creatinine, urea and uric acid | [24] |
| Yellow-fruit nightshade (Solanum xanthocarpum) | The methanolic extract | In vivo | EG-induced urolithiasis in rats | ↓ Renal hyperoxaluria and crystalluria, ↓ Supersaturation of calcium oxalate | [30] |
| Stinging nettle (Urtica dioica) | Methanolic extract | In vivo | EG-induced urolithiasis in rats | ↓ Urinary creatinine level and the supersaturation of lithogenic enhancing agents | [31] |
| Khella (Ammi visnaga L.) | Aqueous extract of fruits | In vivo | EG+ aluminum chloride-induced urolithiasis in rats | ↓ Calcium oxalate crystal deposition ↑ Urinary excretion of citrate ↓ Oxalate excretion | [32] |
| Black-cumin (Nigella Sativa L.) | Ethanolicextract of seeds | In vivo | Ethylene glycol for induction of calcium oxalate calculus formation in rats | ↓ Number of calcium oxalate deposits ↓ Urine concentration of calcium oxalate | [33] |
| Thymoquinone (major component of seeds) | In vivo | Ethylene glycol-induced kidney calculi in rats | ↓ Number and size of calcium oxalate deposits in the renal tubules | [34] | |
| Citrus aurantium L. | Aqueous extract of unripe fruit | In vivo | EG -induced calcium oxalate crystallization | Preventing the formation of calcium oxalate nephrolithiasis and pathological alterations in rats | [35] |
| Oregano (Origanum vulgare L.) | Aerial part aqueous-methanolic extract | In vivo | EG-induced urolithiasis in rats | Preventing loss of body weight, polyurea, crystalluria, oxaluria ↑ Serum urea and creatinine levels | [34] |
| Roselle (Hibiscus sabdariffa L.) | Plant aqueous extracts | In vivo | EG-induced hyperoxaluria | ↓ Deposition of stone-forming constituents in the kidneys and serum | [36] |
| Khella (Ammi visnaga L.) | aqueous extract | In vitro | A flask containing a cystine stone | ↑ Dissolution rate of cystine stones | [37] |
| Mastic (Pistacia lentiscus) | ethanolic fruit extract | In vitro | Calcium oxalate monohydrate-induced in Human Kidney (HK)-2 cells | ↓ Cell death induced by COM, ↓ The level of E-cadherin and H2O2 | [38] |
| Roselle (Dolichos biflorus L.) | Hydro-alcoholic extract of seeds | In vitro | Calcium oxalate crystallization using a synthetic urine system | ↓ Nucleation and aggregation of calcium oxalate monohydrate crystals | [39] |
| Aqueous, chloroform, and benzene extracts of seed | In vitro | Experimental preparation of kidney stones; calcium oxalate and calcium phosphate | Dissolving calcium oxalate stones | [40] | |
| Oregano (Origanum vulgare L.) | Crude aqueous-methanolic extract | In vitro | Supersaturated solution of calcium oxalate, kidney epithelial cell lines (MDCK) and urinary bladder of rabbits | ↓ Calcium oxalate crystallization Exerting antioxidant, renal epithelial cell protective and antispasmodic activities | [41] |
| Solanum xanthocarpum | Saponin rich fraction prepared from fruits | In vitro | calcium oxalate crystal nucleation. artificial urine solution | ↓ Calcium oxalate crystal formation ↑ Glycosaminoglycan level | [42] |
| Pomegranate (Punica granatum) | Extract capsule | Clinical | 23 recurrent stone formers (RSFs) and 7 non-stone formers (NSFs) (1000 mg daily) for 90 days | ↓ Serum paraoxonasearylesterase activity ↓ Supersaturation of calcium oxalate | [43] |
| Horse gram (Dolichos biflorus L.) | Seed | Clinical | 24 patients received Dolichosbiflorus and 23 patients were given potassium citrate | ↓ Recurrence of calcium oxalate stone | [44] |
| Roselle (Hibiscus sabdariffa L.) | A tea bag of dried plant | Clinical | 9 patients with renal stones and 9 with non-renal stone received tea (A cup of tea made from 1.5 g of dry herb two times daily | ↑ Uric acid excretion and clearance | [45] |
| Plant | Part or Chemical Constituents | Study Type | Study Design | Results | Reference |
|---|---|---|---|---|---|
| Bergenia ciliata (Haw.) Sternb | Hydro-alcoholic extract of rhizomes | In vitro | Calcium oxalate induced in a synthetic urine system | ↑ Nucleation and aggregation of COM crystals ↓ The number and size of COM crystals | [85] |
| Bergenia ligulata Engl. | Aqueous-methanolic extract of rhizome | In vitro | Calcium oxalate induced crystal in a synthetic urine system | Inhibition of crystal aggregation and formation ↑ Radical scavenging ability and lipid peroxidation | [86] |
| Commiphora wightii (Arn.) Bhandari | Extract | In vitro | Struvite crystals induced using gel growth technique | ↓ Growth and the size of the struvite crystals | [88] |
| Costus arabicus L. | Aqueous dried plant extract | In vitro | Calcium oxalate monohydrate (COM) crystals induced in MDCK cells | ↓ Crystal growth and calculogenesis | [89] |
| Herniaria hirsuta L. | Ether and methanol extracts of aerial parts | In vitro | Calcium oxalate-induced stone in urine | ↓ The size and supersaturation rate of crystals | [90] |
| Terminalia chebula Retz. | Aqueous fruits extract | In vitro | Calcium oxalate induced cell injury in NRK-52E and MDCK renal epithelial cells | ↓ Lactate dehydrogenase release ↑ Cell viability | [91] |
| Tribulus terrestris L. | Protein biomolecules | In vitro | Oxalate induced injury on NRK-52E cells | ↓ Lactate dehydrogenase release ↑ Cell viability | [92] |
| Plant | Part or Chemical Constituents | Study Type | Study Design | Results | Reference |
|---|---|---|---|---|---|
| Acalypha indica L. | Ethanolic extract | In vivo | Ethylene glycol (EG)-induced urolithiasis in Wistar albino rats | ↑ Ca2+ ATPase, Mg2+ ATPase, Na+K+ ATPase ↑ Aspartate Transaminase (AST), Alanine Transaminase (ALT), Acid phosphatase (ACP) and Alkaline Phosphatase (ALP) | [93] |
| Aerva lanata (L.) Juss. | Aqueous suspension of aerial parts | In vivo | EG-induced urolithiasis in rats | ↓ Glycolic acid oxidase (GAO), and lactate dehydrogenase (LDH) | [94] |
| Ageratum conyzoides (L.) L. | Hydroalcolohlic extract of whole plant | In vivo | EG-induced urolithiasis in rats | ↓ Stone forming constituents, Blood urea nitrogen (BUN), uric acid and creatinine | [11] |
| Alcea rosea L. | Hydroalcoholic extract of roots | In vivo | EG-induced lithiasis in rats | ↓ The number of calcium oxalate deposits ↓ Urinary oxalate level | [95] |
| Asparagus racemosus Willd. | Ethanolic extract of tuberous roots | In vivo | EG-induced urolithiasis in rats | ↓ The level of calcium, oxalate, phosphate, and serum creatinine; ↑ Urinary concentration of magnesium | [96] |
| Bergenia ciliata (Haw.) Sternb. | The hydro-methanolic extract of rhizomes | In vivo | EG-induced urolithiasis in rats | ↓ Nucleation and aggregation of crystals ↓ The number and size of COM crystals | [85] |
| Bergenia ligulata Engl. | Aqueous-methanolic extract of rhizome | In vivo | EG-induced urolithiasis in rats | ↓ Calcium oxalate crystal deposition, and lithogenic signs ↑ Urinary magnesium | [96] |
| Ethanolic extract of rhizome; bergenin | In vivo | EG+ aluminium chloride-induced urolithiasis in rats | ↓ MDA level, ↑ H2O2 scavenging ability ↑ SOD, Catalase (CAT) and GP levels | [87] | |
| Bombax ceiba L. | Fruit aqueous and ethanol extract | In vivo | EG-induced urolithiasis in rats | ↓ Urinary oxalate ↓ Stone forming constituents | [97] |
| Carthamus tinctorius L. | Commercial herbal powder- gastric gavage | In vivo | EG-induced stones in rats | ↓ Deposition of calcium oxalate crystal | [98] |
| Cynodon dactylon (L.) Pers. | N-butanol and ethyl acetate extract of root | In vivo | EG-induced calculus in rats | Preventing calcium oxalate deposition ↓ The size of crystals | [99] |
| Helichrysum graveolens (M.Bieb.) Sweet and Helichrysum stoechas ssp. barellieri (Ten.) Nyman | Capitulum aqueous extract | In vivo | Sodium oxalate- induced urolithiasis in rats | ↓ Formation and growth of crystals ↓ Urine oxalate and uric acid levels, ↑ Citrate level | [100] |
| Hordeum vulgare L. | Seeds ethanolic extract | In vivo | EG-induced urolithiasis in rats | ↓ Stone forming constituents ↓ Lipid peroxidation ↑ SOD and CAT | [101] |
| Hygrophila spinosa T.Anderson | Methanolic extract of aerial parts | In vivo | EG-induced nephrolithiasis in rats | ↓ Urinary oxalate ↓ Calcium and oxalate in kidney; ↑ Urinary magnesium | [102] |
| Hypericum perforatum L. | Hydroalcoholic extract of leaves | In vivo | EG+ ammonium chloride- induced stone in rats | ↓ The size and number of calcium oxalate deposits | [103] |
| Launaea procumbens L. | Methanolic extract of leaves | In vivo | EG-induced urolithiasis in rats | ↓ Urinary calcium, oxalate and phosphate excretion ↓ Creatinine and uric acid | [104] |
| Lygodium japonicum (Thunb.) Sw. | Ethanolic extract of spore | In vivo | EG-induced kidney calculi in rats | ↓ Urinary calcium, oxalate and uric acid ↓ Kidney peroxides, and the number of oxalate deposits ↑ Urinary citrate levels | [105] |
| Orthosiphon grandiflorus Bold. | Aqueous extract of leaves | In vivo | EG-induced stones in rats | ↓ Crystal deposits ↑ SOD and CAT | [106] |
| Paronychia argentea Lam. | Butanolic extract of aerial parts | In vivo | Sodium oxalate-induced lithiasis in rats | ↓ Renal necrosis ↓ Serum creatinine and blood urea levels | [107] |
| Pergularia daemia (Forssk.) Chiov. | Whole-plant hydroalcoholic extract | In vivo | EG- induced kidney stone in rats | ↓ Serum urea nitrogen, creatinine and uric acid levels | [108] |
| Quercus salicina Blume | Leaves aqueous extract | In vivo | EG and the vitamin D3 analog(α-calcidol)-induced urolithiasis in rats | ↓ MDA and serum creatinine level ↓ Oxidative stress ↓ Calcium level in kidney | [109] |
| Salvadora persica L. | Aqueous and alcoholic extract of the leaves | In vivo | EG- induced urolithiasis in rats | ↓ Urinary oxalate levels and deposition | [110] |
| Selaginella lepidophylla (Hook. et Grev) Spring | Chloroform extract of the plant | In vivo | EG and ammonium chloride- induced urolithiasis in rats | ↑ Urinary flow rate, glomerular filtration rate (GFR) ↓ ROS and lipid-peroxidation ↓ Renal cortical organic anion transporter (OAT3) expression | [111] |
| Agropyron repens (L.) P.Beauv. | Extract | Clinical | Unblinded treatment to the patients (treatment group received potassium citrate + Agropyrum repens and control group recieved potassium citrate alone (100 mg/day for 5 month)) | ↓ Number and size of urinary stones ↓ Uric acid urinary secretion | [112] |
| Phyllanthus niruri L. | Extract | Clinical | 150 patients received 1 to 3 extracorporeal shock wave lithotripsy sessions. After treatment 78 patients received extract and 72 were served as a control group (2 g/day for 3 month) | ↑ Stone-free rate (stone-free defined as the absence of any stone or residual fragments less than 3 mm) | [113] |
| Photochemical | In Vitro/In Vivo | Model | Result | Reference |
|---|---|---|---|---|
| Catechin | In vitro | Calcium oxalate monohydrate(COM)-induced NRK-52E cells | ↑ SOD activity ↓ Mitochondrial membrane potential (MMP), Caspase-3 activity, and renal calcium crystallization | [121] |
| In vivo | Ethylene glycol (EG) induced nephrolithiasis in rat | ↑ OPN, ↓ MDA, 8-OHdG ↓ Renal calcium crystallization | [121] | |
| In vivo | EG-induced nephrolithiasis in rat | ↓ Calcium oxalate monohydrate and Papillary calculus formation ↓ Renal papillary calcification | [122] | |
| Epigallocatechin-3-gallate | In vitro | COM-induced Madin–Darby canine kidney (MDCK) cells | ↓ α-enolase protein expression ↓ crystal-binding capability | [123] |
| In vitro | Oxalate-induced NRK-52E cells | ↓ Free-radical production | [41] | |
| In vivo | Oxalate-induced renal stone in rats | ↓ Excretion of urinary oxalate ↓ Activities of urinary gammaglutamyl transpeptidase and N-acetylglucosaminidase | [41] | |
| Diosmin | In vivo | EG-induced nephrolithiasis in rat | ↓ Capillary hyper-permeability ↓ Degeneration of glomeruli and tubules, Restoring the diameter of the capillaries and vessels in the cortex | [129] |
| Rutin | In vivo | EG-induced nephrolithiasis in rat | Prevention of stone formation Inhibition of calcium oxalate urolithiasis | [133] |
| Quercetin | In vivo | EG induced calcium oxalate (CaOx) formation | Hypo-Uricemic, and anti-inflammatory activities Inhibitory effect on the deposition of CaOx crystal | [134] |
| In vitro | Sodium oxalate | ↓ Cell viability ↓ Lipid peroxidation | [135] | |
| In vivo | Hyperoxaluria-induced rats | ↓ Urinary crystal deposit formation | [135] | |
| In vivo | EG-induced nephrolithiasis in rat | ↓ Oxidative damage ↑ Serum paraoxonase 1 (PON1) | [119] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nirumand, M.C.; Hajialyani, M.; Rahimi, R.; Farzaei, M.H.; Zingue, S.; Nabavi, S.M.; Bishayee, A. Dietary Plants for the Prevention and Management of Kidney Stones: Preclinical and Clinical Evidence and Molecular Mechanisms. Int. J. Mol. Sci. 2018, 19, 765. https://doi.org/10.3390/ijms19030765
Nirumand MC, Hajialyani M, Rahimi R, Farzaei MH, Zingue S, Nabavi SM, Bishayee A. Dietary Plants for the Prevention and Management of Kidney Stones: Preclinical and Clinical Evidence and Molecular Mechanisms. International Journal of Molecular Sciences. 2018; 19(3):765. https://doi.org/10.3390/ijms19030765
Chicago/Turabian StyleNirumand, Mina Cheraghi, Marziyeh Hajialyani, Roja Rahimi, Mohammad Hosein Farzaei, Stéphane Zingue, Seyed Mohammad Nabavi, and Anupam Bishayee. 2018. "Dietary Plants for the Prevention and Management of Kidney Stones: Preclinical and Clinical Evidence and Molecular Mechanisms" International Journal of Molecular Sciences 19, no. 3: 765. https://doi.org/10.3390/ijms19030765
APA StyleNirumand, M. C., Hajialyani, M., Rahimi, R., Farzaei, M. H., Zingue, S., Nabavi, S. M., & Bishayee, A. (2018). Dietary Plants for the Prevention and Management of Kidney Stones: Preclinical and Clinical Evidence and Molecular Mechanisms. International Journal of Molecular Sciences, 19(3), 765. https://doi.org/10.3390/ijms19030765






