Interventional Left Atrial Appendage Closure Affects the Metabolism of Acylcarnitines
Abstract
1. Introduction
2. Results
2.1. Baseline Characteristics and Procedures’ Indications
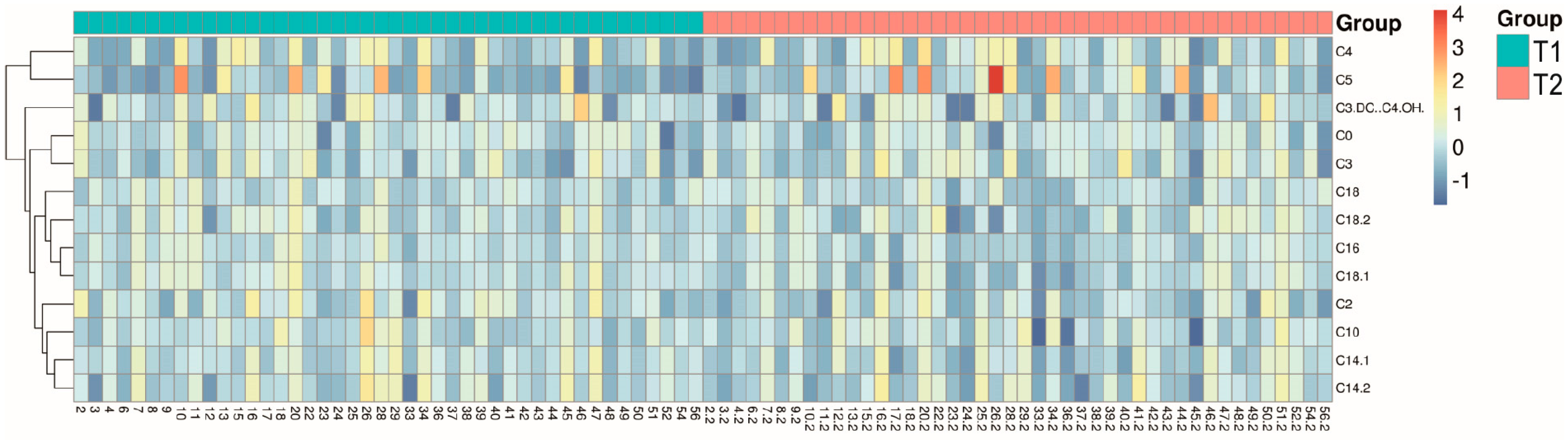
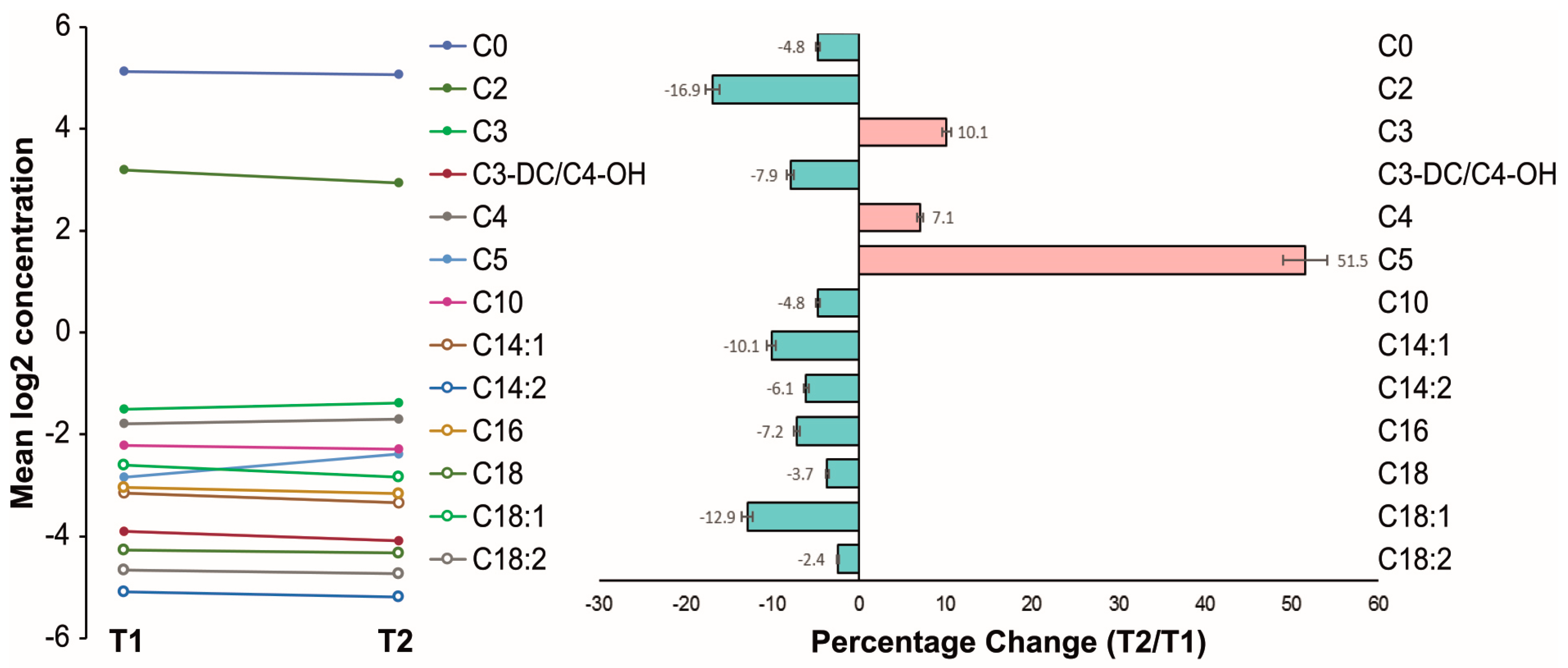
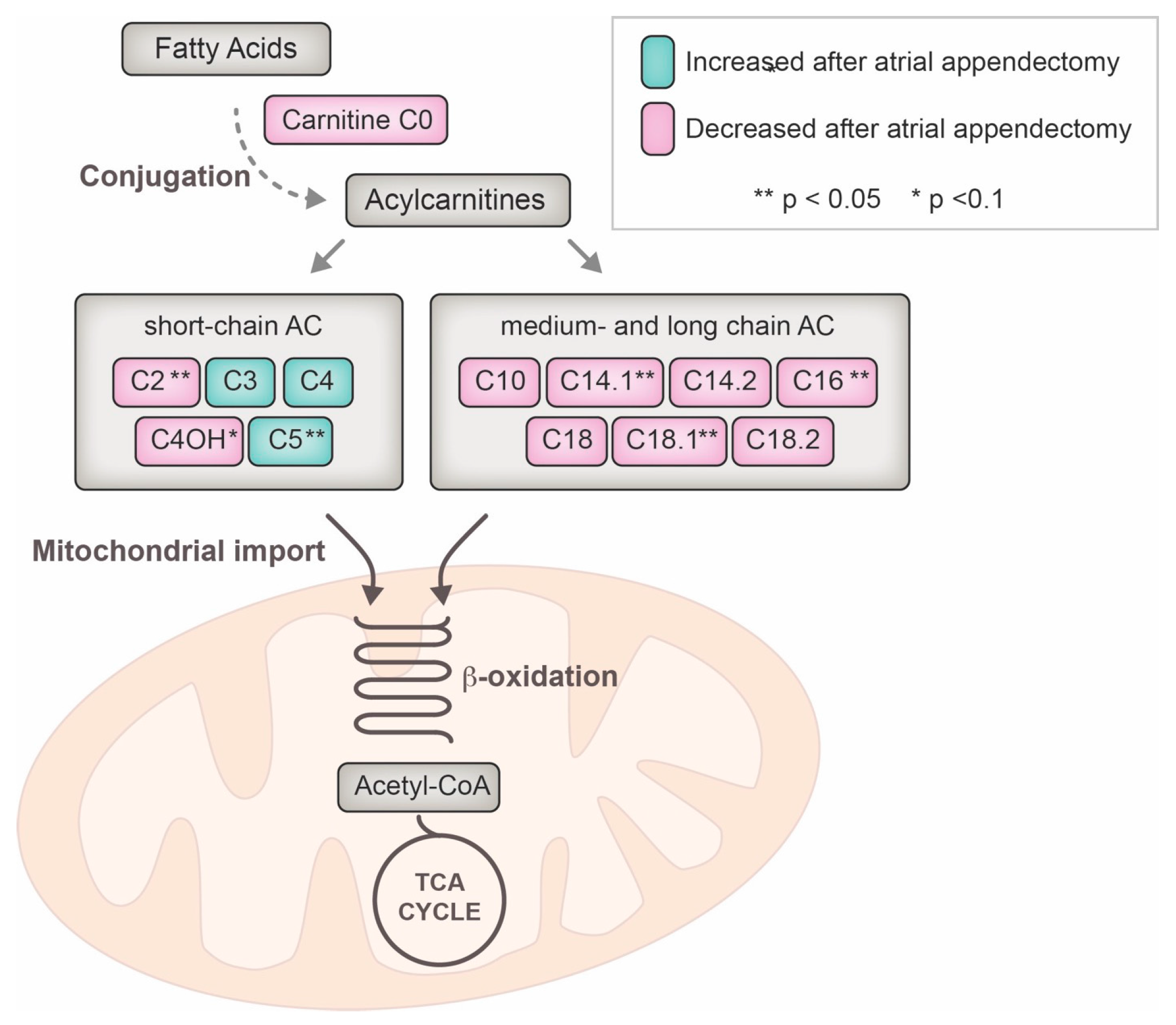
2.2. Numerical Non-Adjusted Changes of Acylcarnitine Levels
2.3. Subgroup Analyses
2.4. Adjusted Multivariable Regression Model
3. Discussion
Study Limitations
4. Materials and Methods
4.1. Sample Collection
4.2. Characterization of the Study Population and Changes over the Mid-Term Follow-Up
4.3. Metabolite Analyses
4.4. Outcome Measures
4.5. Statistics
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| AF | atrial fibrillation |
| ANOVA | analysis of variance |
| BMI | body mass index |
| CAC | carnitine/acylcarnitine carrier protein |
| CHA2DS2-VASc | congestive heart failure, hypertension, age ≥ 75 years, diabetes mellitus, prior stroke or TIA or thromboembolism, vascular disease, age 65–74 years, sex category (i.e., female sex) |
| CPT | carnitine palmitoytransferase |
| CRP | C reactive protein |
| CT | computed tomography |
| EDTA | ethylenediaminetetra-acetic acid |
| ESI–LC–MS/MS | electrospray ionization–liquid chromatography– mass spectrometry |
| FDR | false discovery rate |
| HAS-BLED | hypertension, abnormal renal or liver function, prior stroke, prior major bleeding or predisposition to bleeding, labile INR, elderly, i.e., age > 65 years, prior alcohol, drug, or medication usage predisposing to bleeding |
| HCA | hierarchical cluster analysis |
| INR | International Normalized Ratio |
| LAA | left atrial appendage |
| LAAC | left atrial appendage |
| LABEL | Left Atrial Appendage Occlusion and Biomarker Evaluation |
| LC | long chain |
| LDH | lactate dehydrogenase |
| LOD | limit of detection |
| LV | left ventricular |
| LVEF | left ventricular ejection fraction |
| MC | medium chain |
| MC | medium chain |
| MDRD | Modification of Diet in Renal Disease |
| NT-proBNP | amino-terminal pro-brain natriuretic peptide |
| OAC | oral anticoagulation |
| PCA | principal component analysis |
| PLS-DA | partial least squares discrimination analysis |
| SC | short chain |
| TCA | tricarboxylic acid |
References
- Kirchhof, P.; Benussi, S.; Kotecha, D.; Ahlsson, A.; Atar, D.; Casadei, B.; Hindricks, G. 2016 ESC Guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Eur. Heart J. 2016, 37, 2893–2962. [Google Scholar] [CrossRef] [PubMed]
- January, C.T.; Wann, L.S.; Alpert, J.S.; Calkins, H.; Cigarroa, J.E.; Cleveland, J.C., Jr.; Sacco, R.L. 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society. J. Am. Coll. Cardiol. 2014, 64, e1–e76. [Google Scholar] [CrossRef] [PubMed]
- Reddy, V.Y.; Sievert, H.; Halperin, J.; Doshi, S.K.; Buchbinder, M.; Neuzil, P.; Gordon, N. Percutaneous left atrial appendage closure vs warfarin for atrial fibrillation: A randomized clinical trial. JAMA 2014, 312, 1988–1998. [Google Scholar] [CrossRef] [PubMed]
- Patti, G.; Pengo, V.; Marcucci, R.; Cirillo, P.; Renda, G.; Santilli, F.; Parato, V.M. The left atrial appendage: From embryology to prevention of thromboembolism. Eur. Heart J. 2017, 38, 877–887. [Google Scholar] [CrossRef] [PubMed]
- Majunke, N.; Sandri, M.; Adams, V.; Daehnert, I.; Mangner, N.; Schuler, G.; Moebius-Winkler, S. Atrial and Brain Natriuretic Peptide Secretion After Percutaneous Closure of the Left Atrial Appendage With the Watchman Device. J. Invasive Cardiol. 2015, 27, 448–452. [Google Scholar] [PubMed]
- Fastner, C.; Behnes, M.; Sartorius, B.; Yildiz, M.; Mashayekhi, K.; El-Battrawy, I.; Akin, I. Left atrial appendage morphology, echocardiographic characterization, procedural data and in-hospital outcome of patients receiving left atrial appendage occlusion device implantation: A prospective observational study. BMC Cardiovasc. Disord. 2016, 16, 25. [Google Scholar] [CrossRef] [PubMed]
- Behnes, M.; Sartorius, B.; Wenke, A.; Lang, S.; Hoffmann, U.; Fastner, C.; Borggrefe, M.; Roth, T.; Triebel, J.; Bertsch, T.; Akin, I. Percutaneous closure of left atrial appendage affects mid-term release of MR-proANP. Sci. Rep. 2017, 7, 9028. [Google Scholar] [CrossRef] [PubMed]
- Lewis, G.D.; Asnani, A.; Gerszten, R.E. Application of metabolomics to cardiovascular biomarker and pathway discovery. J. Am. Coll. Cardiol. 2008, 52, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Ussher, J.R.; Elmariah, S.; Gerszten, R.E.; Dyck, J.R. The Emerging Role of Metabolomics in the Diagnosis and Prognosis of Cardiovascular Disease. J. Am. Coll. Cardiol. 2016, 68, 2850–2870. [Google Scholar] [CrossRef] [PubMed]
- Lewis, G.D.; Wei, R.; Liu, E.; Yang, E.; Shi, X.; Martinovic, M.; Shaham, O. Metabolite profiling of blood from individuals undergoing planned myocardial infarction reveals early markers of myocardial injury. J. Clin. Investig. 2008, 118, 3503–3512. [Google Scholar] [CrossRef] [PubMed]
- Nemutlu, E.; Zhang, S.; Xu, Y.Z.; Terzic, A.; Zhong, L.; Dzeja, P.D.; Cha, Y.M. Cardiac resynchronization therapy induces adaptive metabolic transitions in the metabolomic profile of heart failure. J. Card. Fail. 2015, 21, 460–469. [Google Scholar] [CrossRef] [PubMed]
- Sattler, K.; Behnes, M.; Barth, C.; Wenke, A.; Sartorius, B.; El-Battrawy, I.; Mashayekhi, K.; Kuschyk, J.; Hoffmann, U.; Papavasiliu, T.; et al. Occlusion of left atrial appendage affects metabolomic profile: Focus on glycolysis, tricarboxylic acid and urea metabolism. Metabolomics 2017, 13, 127. [Google Scholar] [CrossRef] [PubMed]
- McCoin, C.S.; Knotts, T.A.; Adams, S.H. Acylcarnitines—Old actors auditioning for new roles in metabolic physiology. Nat. Rev. Endocrinol. 2015, 11, 617–625. [Google Scholar] [CrossRef] [PubMed]
- Rizza, S.; Copetti, M.; Rossi, C.; Cianfarani, M.A.; Zucchelli, M.; Luzi, A.; Pellegrini, F. Metabolomics signature improves the prediction of cardiovascular events in elderly subjects. Atherosclerosis 2014, 232, 260–264. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, T.; Kelly, J.P.; McGarrah, R.W.; Hellkamp, A.S.; Fiuzat, M.; Testani, J.M.; Ilkayeva, O.R. Prognostic Implications of Long-Chain Acylcarnitines in Heart Failure and Reversibility With Mechanical Circulatory Support. J. Am. Coll. Cardiol. 2016, 67, 291–299. [Google Scholar] [CrossRef] [PubMed]
- Hunter, W.G.; Kelly, J.P.; McGarrah, R.W., 3rd; Khouri, M.G.; Craig, D.; Haynes, C.; Newgard, C.B. Metabolomic Profiling Identifies Novel Circulating Biomarkers of Mitochondrial Dysfunction Differentially Elevated in Heart Failure With Preserved Versus Reduced Ejection Fraction: Evidence for Shared Metabolic Impairments in Clinical Heart Failure. J. Am. Heart Assoc. 2016, 5, e003190. [Google Scholar] [PubMed]
- Reuter, S.E.; Evans, A.M. Carnitine and acylcarnitines: Pharmacokinetic, pharmacological and clinical aspects. Clin. Pharmacokinet. 2012, 51, 553–572. [Google Scholar] [CrossRef] [PubMed]
- Meincke, F.; Schmidt-Salzmann, M.; Kreidel, F.; Kuck, K.H.; Bergmann, M.W. New technical and anticoagulation aspects for left atrial appendage closure using the WATCHMAN(R) device in patients not taking warfarin. EuroIntervention 2013, 9, 463–468. [Google Scholar] [CrossRef] [PubMed]
- Sick, P.B.; Schuler, G.; Hauptmann, K.E.; Grube, E.; Yakubov, S.; Turi, Z.G.; Holmes, D.R. Initial worldwide experience with the WATCHMAN left atrial appendage system for stroke prevention in atrial fibrillation. J. Am. Coll. Cardiol. 2007, 49, 1490–1495. [Google Scholar] [CrossRef] [PubMed]
- Saw, J.; Fahmy, P.; DeJong, P.; Lempereur, M.; Spencer, R.; Tsang, M.; Nicolaou, S. Cardiac CT angiography for device surveillance after endovascular left atrial appendage closure. Eur. Heart J. Cardiovasc. Imaging 2015, 16, 1198–1206. [Google Scholar] [CrossRef] [PubMed]
- Behnes, M.; Akin, I.; Sartorius, B.; Fastner, C.; El-Battrawy, I.; Borggrefe, M.; Henzler, T. LAA Occluder View for post-implantation Evaluation (LOVE)—Standardized imaging proposal evaluating implanted left atrial appendage occlusion devices by cardiac computed tomography. BMC Med. Imaging 2016, 16, 25. [Google Scholar] [CrossRef] [PubMed]
- Meier, B.; Blaauw, Y.; Khattab, A.A.; Lewalter, T.; Sievert, H.; Tondo, C.; Roffi, M. EHRA/EAPCI expert consensus statement on catheter-based left atrial appendage occlusion. Europace 2014, 16, 1397–1416. [Google Scholar] [CrossRef] [PubMed]
- Violante, S.; Ijlst, L.; Te Brinke, H.; de Almeida, I.T.; Wanders, R.J.; Ventura, F.V.; Houten, S.M. Carnitine palmitoyltransferase 2 and carnitine/acylcarnitine translocase are involved in the mitochondrial synthesis and export of acylcarnitines. FASEB J. 2013, 27, 2039–2044. [Google Scholar] [CrossRef] [PubMed]
- Kalim, S.; Clish, C.B.; Wenger, J.; Elmariah, S.; Yeh, R.W.; Deferio, J.J.; Rhee, E.P. A plasma long-chain acylcarnitine predicts cardiovascular mortality in incident dialysis patients. J. Am. Heart Assoc. 2013, 2, e000542. [Google Scholar] [CrossRef] [PubMed]
- Aguer, C.; McCoin, C.S.; Knotts, T.A.; Thrush, A.B.; Ono-Moore, K.; McPherson, R.; Harper, M.E. Acylcarnitines: Potential implications for skeletal muscle insulin resistance. FASEB J. 2015, 29, 336–345. [Google Scholar] [CrossRef] [PubMed]
- Schooneman, M.G.; Vaz, F.M.; Houten, S.M.; Soeters, M.R. Acylcarnitines: Reflecting or inflicting insulin resistance? Diabetes 2013, 62, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Mihalik, S.J.; Goodpaster, B.H.; Kelley, D.E.; Chace, D.H.; Vockley, J.; Toledo, F.G.; DeLany, J.P. Increased levels of plasma acylcarnitines in obesity and type 2 diabetes and identification of a marker of glucolipotoxicity. Obesity 2010, 18, 1695–1700. [Google Scholar] [CrossRef] [PubMed]
- Boersma, L.V.; Schmidt, B.; Betts, T.R.; Sievert, H.; Tamburino, C.; Teiger, E.; Bergmann, M.W. Implant success and safety of left atrial appendage closure with the WATCHMAN device: Peri-procedural outcomes from the EWOLUTION registry. Eur. Heart J. 2016, 37, 2465–2474. [Google Scholar] [CrossRef] [PubMed]
- Di Guida, R.; Engel, J.; Allwood, J.W.; Weber, R.J.; Jones, M.R.; Sommer, U.; Dunn, W.B. Non-targeted UHPLC-MS metabolomic data processing methods: A comparative investigation of normalisation, missing value imputation, transformation and scaling. Metabolomics 2016, 12, 93. [Google Scholar] [CrossRef] [PubMed]
- Kooperberg, C.; Stone, C.J. Logspine Density Estimation for Censored Data. J. Comput. Graph. Stat. 1992, 1, 301–328. [Google Scholar]
- Worley, B.; Powers, R. Multivariate Analysis in Metabolomics. Curr. Metab. 2013, 1, 92–107. [Google Scholar]
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate: A Practical and Powerful Approach to Multiple Testing. J. R. Stat. Soc. Ser. B (Methodol.) 1995, 57, 289–300. [Google Scholar]
| Characteristic | Value |
|---|---|
| Demographics | |
| sex, male n (%) | 30 (68.2) |
| age, y (IQR) | 78 (75.8–83) |
| BMI, kg/m2 (IQR) | 28.1 (24.7–32.7) |
| Cardiovascular risk factors | |
| arterial hypertension, n (%) | 42 (95.4) |
| diabetes mellitus, n (%) | 16 (36.7) |
| HbA1c, % (IQR) | 7.0 (6.2–7.0) |
| hypercholesterinemia, n (%) | 22 (50) |
| cholesterol, mg/dL (IQR) | 143 (124–186) |
| low-density lipoprotein, mg/dL (IQR) | 89 (68–111) |
| high-density lipoprotein, mg/dL (IQR) | 55 (41–62) |
| triglycerides, mg/dL (IQR) | 117 (95–162) |
| impaired left ventricular function, each n (%) | |
| LVEF 45–54% | 4 (9.1) |
| LVEF 30–44% | 4 (9.1) |
| LVEF < 30% | 2 (4.5) |
| diastolic dysfunction, n (%) | 11 (25.0%) |
| combined with impaired left ventricular function, n (%) | 1 (2.3%) |
| Medical history | |
| atrial fibrillation, each n (%) | |
| Paroxysmal | 24 (54.5) |
| Persistent | 6 (13.6) |
| Permanent | 14 (31.8) |
| pulmonary vein isolation, n (%) | 4 (9.1) |
| transitory ischemic attack, n (%) | 3 (6.8) |
| stroke, n (%) | 7 (15.9) |
| coronary artery disease, n (%) | 25 (56.8) |
| myocardial infarction, n (%) | 10 (22.7) |
| heart failure, n (%) | 10 (22.7) |
| peripheral vascular disease, n (%) | 4 (9.1) |
| chronic kidney disease, n (%) | 18 (40.9) |
| chronic liver disease, n (%) | 3 (6.8) |
| sleep apnea, n (%) | 4 (9.1) |
| prior bleeding, n (%) | 34 (77.3) |
| CHA2DS2-VASc score (IQR) | 4 (3–5) |
| HAS-BLED score (IQR) | 4 (3–5) |
| Postinterventional antithrombotic regimen | |
| dual antiplatelet therapy for 6 months, n (%) | 44 (100.0) |
| Reasons for rehospitalization/severe clinical events during mid-term follow-up | |
| myocardial infarction, n (%) | 1 (2.3) |
| heart failure, n (%) | 6 (13.6) |
| arrhythmia, n (%) | 2 (4.5) |
| vascular problem, n (%) | 2 (4.5) |
| gastrointestinal bleeding, n (%) | 8 (18.2) |
| renal failure, n (%) | 1 (2.3) |
| infectious disease, n (%) | 2 (4.5) |
| orthopedic, n (%) | 2 (4.5) |
| Bleeding during mid-term follow-up, BARC-Score | |
| 1, n (%) | 1 (2.3) |
| 2, n (%) | 5 (11.4) |
| 3a, n (%) | 2 (4.5) |
| ≥3b, n (%) | 0 (0) |
| Characteristics | T1 | T2 | p Value |
|---|---|---|---|
| Smoking status | |||
| never, n (%) | 37 (84.1) | 37 (84.1) | 1.000 |
| >1 year former, n (%) | 1 (2.3) | 1 (2.3) | 1.000 |
| current, n (%) | 6 (13.6) | 6 (13.6) | 1.000 |
| Pharmacotherapy | |||
| beta blockers, n (%) | 17 (38.6) | 15 (34.1) | 0.825 |
| calcium channel blockers, n (%) | 9 (20.5) | 7 (15.9) | 0.783 |
| combined therapy, n (%) | 14 (31.8) | 15 (34.1) | 1.000 |
| statines, n (%) | 27 (61.4) | 28 (63.6) | 1.000 |
| statin and ezetimibe, n (%) | 1 (2.3) | 1 (2.3) | 1.000 |
| other lipid-lowering drugs, n (%) | 1 (2.3) | 1 (2.3) | 1.000 |
| Anticoagulation | |||
| None | 15 (34.1) | 43 (97.7) | <0.001 |
| Any | 29 (65.9) | 1 (2.3) | <0.001 |
| phenprocoumon, n (%) | 10 (22.7) | 0 (0.0) | 0.001 |
| dabigatran, n (%) | 3 (6.8) | 0 (0.0) | 0.241 |
| rivaroxaban, n (%) | 3 (6.8) | 0 (0.0) | 0.241 |
| apixaban, n (%) | 3 (6.8) | 1 (2.3) | 0.616 |
| LMWH, n (%) | 10 (22.7) | 0 (0.0) | 0.001 |
| Echocardiographic data | |||
| LA diameter, mm (IQR) | 48.0 (43.7–55.0) | 49.0 (44.0–53.0) | 0.657 |
| LA surface, cm2 (IQR) | 24.0 (19.7–28.0) | 22.0 (19.0–25.0) | 0.010 |
| LA volume, cm3 (IQR) | 88.5 (70.2–105.3) | 83.0 (66.2–100.5) | 0.739 |
| Laboratory values | |||
| cholesterol, mg/dL (IQR) | 143 (124–186) | 155 (128–161) | 0.370 |
| NT-proBNP, pg/mL (IQR) | 975 (455–1429) | 981 (488–1852) | 0.323 |
| creatinine, mg/dL (IQR) | 1.10 (0.96–1.42) | 1.24 (1.01–1.71) | 0.430 |
| MDRD-GFR, mL/min/1.73 m2 (IQR) | 65 (43–65) | 56 (37–65) | 0.140 |
| Hb, g/dl (IQR) | 12.4 (10.7–14.5) | 10.8 (9.6–12.7) | 0.810 |
| anemia < 10 g/dL, n (%) | 8 (18.2) | 8 (18.2) | 1.000 |
| average blood glucose, g/dL (IQR) | 113 (94–133) | 108 (90–112) | 0.900 |
| CRP, mg/L (IQR) | 5.1 (2.9–11.1) | 4.0 (2.9–14.3) | 0.560 |
| LDH, U/L (IQR) | 198 (176–240) | 240 (199–253) | 0.930 |
| T1 | T2 | ||||||
|---|---|---|---|---|---|---|---|
| Rank | Metabolite | Mean Conc. (µM) | SD | Mean Conc. (µM) | SD | Percentage Change | p Value |
| 1 | C18:1 | 0.171 | 0.051 | 0.149 | 0.054 | −12.9 | 0.005 |
| 2 | C2 | 10.12 | 5.02 | 8.41 | 3.77 | −16.9 | 0.005 |
| 3 | C5 | 0.206 | 0.252 | 0.312 | 0.481 | 51.5 | 0.013 |
| 4 | C14:1 | 0.119 | 0.042 | 0.107 | 0.043 | −10.1 | 0.035 |
| 5 | C16 | 0.125 | 0.034 | 0.116 | 0.035 | −7.2 | 0.050 |
| 6 | C3-DC/C4-OH | 0.076 | 0.043 | 0.070 | 0.073 | −7.9 | 0.071 |
| 7 | C3 | 0.385 | 0.161 | 0.424 | 0.232 | 10.1 | 0.155 |
| 8 | C4 | 0.336 | 0.221 | 0.360 | 0.212 | 7.1 | 0.337 |
| 9 | C14:2 | 0.033 | 0.021 | 0.031 | 0.025 | −6.1 | 0.380 |
| 10 | C18 | 0.054 | 0.024 | 0.052 | 0.012 | −3.7 | 0.422 |
| 11 | C0 | 36.93 | 11.42 | 35.15 | 10.15 | −4.8 | 0.430 |
| 12 | C18:2 | 0.042 | 0.014 | 0.041 | 0.023 | −2.4 | 0.430 |
| 13 | C10 | 0.231 | 0.114 | 0.220 | 0.091 | −4.8 | 0.493 |
| Rank | Metabolite | FDR | Beta | Standard Error | t Value | p Value |
|---|---|---|---|---|---|---|
| 1 | C18:1 | 0.0351 | −0.236 | 0.080 | −2.961 | 0.0050 |
| 2 | C2 | 0.0351 | −0.254 | 0.087 | 2.930 | 0.0054 |
| 3 | C5 | 0.0549 | 0.456 | 0.175 | 2.602 | 0.0127 |
| 4 | C14:1 | 0.1130 | −0.189 | 0.087 | −2.180 | 0.0348 |
| 5 | C16 | 0.1288 | −0.121 | 0.060 | −2.021 | 0.0495 |
| 6 | C3-DC/C4-OH | 0.1534 | −0.186 | 0.100 | −1.853 | 0.0708 |
| 7 | C3 | 0.2886 | 0.123 | 0.085 | 1.446 | 0.1554 |
| 8 | C4 | 0.4663 | 0.092 | 0.095 | 0.971 | 0.3368 |
| 9 | C14:2 | 0.4663 | −0.102 | 0.115 | −0.887 | 0.3801 |
| 10 | C18 | 0.4663 | −0.056 | 0.069 | −0.810 | 0.4222 |
| 11 | C0 | 0.4663 | −0.061 | 0.077 | −0.797 | 0.4297 |
| 12 | C18:2 | 0.4663 | −0.071 | 0.089 | −0.796 | 0.4304 |
| 13 | C10 | 0.4932 | −0.074 | 0.107 | −0.691 | 0.4932 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fastner, C.; Behnes, M.; Sartorius, B.; Wenke, A.; Lang, S.; Yücel, G.; Sattler, K.; Rusnak, J.; Saleh, A.; Barth, C.; et al. Interventional Left Atrial Appendage Closure Affects the Metabolism of Acylcarnitines. Int. J. Mol. Sci. 2018, 19, 500. https://doi.org/10.3390/ijms19020500
Fastner C, Behnes M, Sartorius B, Wenke A, Lang S, Yücel G, Sattler K, Rusnak J, Saleh A, Barth C, et al. Interventional Left Atrial Appendage Closure Affects the Metabolism of Acylcarnitines. International Journal of Molecular Sciences. 2018; 19(2):500. https://doi.org/10.3390/ijms19020500
Chicago/Turabian StyleFastner, Christian, Michael Behnes, Benjamin Sartorius, Annika Wenke, Siegfried Lang, Gökhan Yücel, Katherine Sattler, Jonas Rusnak, Ahmad Saleh, Christian Barth, and et al. 2018. "Interventional Left Atrial Appendage Closure Affects the Metabolism of Acylcarnitines" International Journal of Molecular Sciences 19, no. 2: 500. https://doi.org/10.3390/ijms19020500
APA StyleFastner, C., Behnes, M., Sartorius, B., Wenke, A., Lang, S., Yücel, G., Sattler, K., Rusnak, J., Saleh, A., Barth, C., Mashayekhi, K., Hoffmann, U., Borggrefe, M., & Akin, I. (2018). Interventional Left Atrial Appendage Closure Affects the Metabolism of Acylcarnitines. International Journal of Molecular Sciences, 19(2), 500. https://doi.org/10.3390/ijms19020500




