Naturally Occurring Canine Melanoma as a Predictive Comparative Oncology Model for Human Mucosal and Other Triple Wild-Type Melanomas
Abstract
1. Introduction
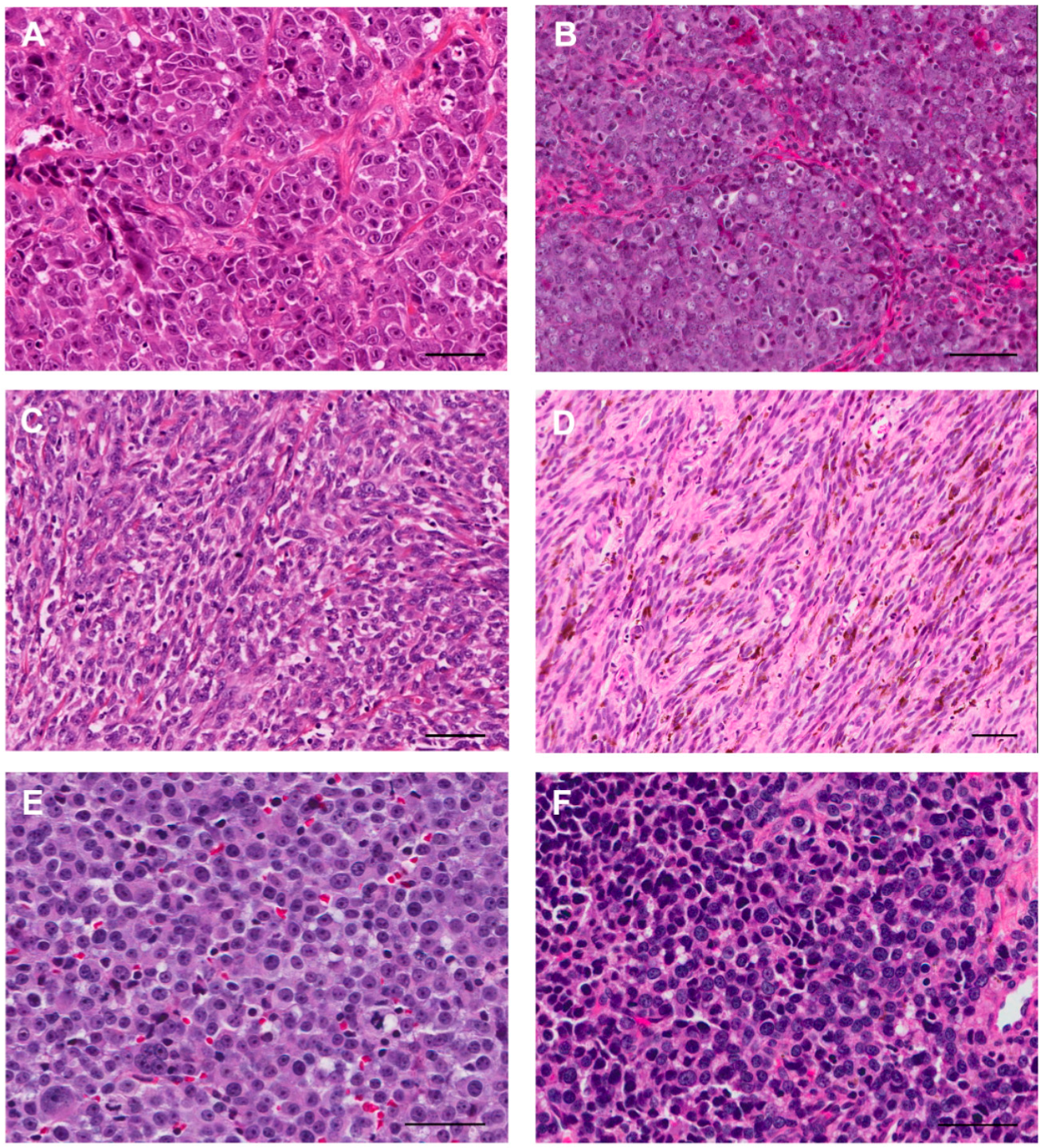
2. Clinical Manifestations of Canine Melanomas
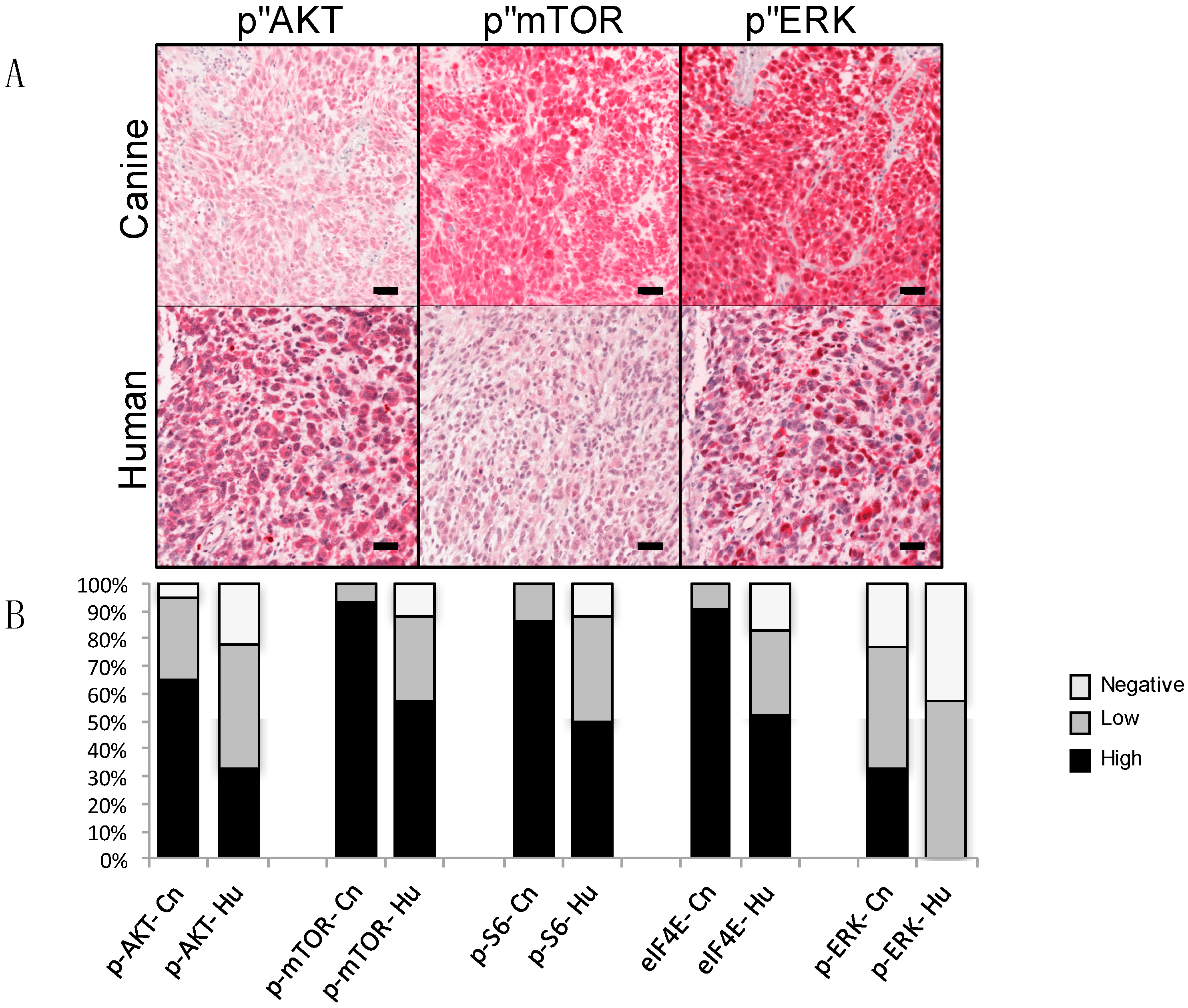
3. Comparative Genetics and Molecular Signaling Pathways
4. Canine Mucosal Melanoma as a Preclinical Model
4.1. Small Molecule Signaling Inhibitors
4.1.1. BRAF, KIT, and MAPK Pathway Inhibitors
4.1.2. PI3K Pathway Inhibition and Combined Targeted Therapy
4.2. Immunomodulators
4.2.1. Immune Checkpoint Blockade
4.2.2. Adoptively transferred Cell Therapies
4.2.3. Tumor Vaccines
4.2.4. Cytokine Therapy
5. Conclusions and Perspectives
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer Statistics, 2017. CA Cancer J. Clin. 2017, 67, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Chang, A.E.; Karnell, L.H.; Menck, H.R. The National Cancer Data Base report on cutaneous and noncutaneous melanoma: A summary of 84,836 cases from the past decade. The American College of Surgeons Commission on Cancer and the American Cancer Society. Cancer 1998, 83, 1664–1678. [Google Scholar] [CrossRef]
- Kong, Y.; Si, L.; Zhu, Y.; Xu, X.; Corless, C.L.; Flaherty, K.T.; Li, L.; Li, H.; Sheng, X.; Cui, C.; et al. Large-scale analysis of KIT aberrations in Chinese patients with melanoma. Clin. Cancer Res. 2011, 17, 1684–1691. [Google Scholar] [CrossRef] [PubMed]
- Bastian, B.C. The molecular pathology of melanoma: An integrated taxonomy of melanocytic neoplasia. Annu. Rev. Pathol. 2014, 9, 239–271. [Google Scholar] [CrossRef] [PubMed]
- Hayward, N.K.; Wilmott, J.S.; Waddell, N.; Johansson, P.A.; Field, M.A.; Nones, K.; Patch, A.M.; Kakavand, H.; Alexandrov, L.B.; Burke, H.; et al. Whole-genome landscapes of major melanoma subtypes. Nature 2017, 545, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Van der Weyden, L.; Patton, E.E.; Wood, G.A.; Foote, A.K.; Brenn, T.; Arends, M.J.; Adams, D.J. Cross-species models of human melanoma. J. Pathol. 2016, 238, 152–165. [Google Scholar] [CrossRef] [PubMed]
- Hodis, E.; Watson, I.R.; Kryukov, G.V.; Arold, S.T.; Imielinski, M.; Theurillat, J.-P.; Nickerson, E.; Auclair, D.; Li, L.; Place, C.; et al. A Landscape of Driver Mutations in Melanoma. Cell 2012, 150, 251–263. [Google Scholar] [CrossRef] [PubMed]
- Davies, M.A.; Kopetz, S. Overcoming resistance to MAPK pathway inhibitors. J. Natl. Cancer Inst. 2013, 105, 9–10. [Google Scholar] [CrossRef] [PubMed]
- Van ‘t Veer, L.J.; Burgering, B.M.; Versteeg, R.; Boot, A.J.; Ruiter, D.J.; Osanto, S.; Schrier, P.I.; Bos, J.L. N-ras mutations in human cutaneous melanoma from sun-exposed body sites. Mol. Cell. Biol. 1989, 9, 3114–3116. [Google Scholar] [CrossRef] [PubMed]
- Ascierto, P.A.; Kirkwood, J.M.; Grob, J.J.; Simeone, E.; Grimaldi, A.M.; Maio, M.; Palmieri, G.; Testori, A.; Marincola, F.M.; Mozzillo, N. The role of BRAF V600 mutation in melanoma. J. Transl. Med. 2012, 10, 85. [Google Scholar] [CrossRef] [PubMed]
- Curtin, J.A.; Busam, K.; Pinkel, D.; Bastian, B.C. Somatic activation of KIT in distinct subtypes of melanoma. J. Clin. Oncol. 2006, 24, 4340–4346. [Google Scholar] [CrossRef] [PubMed]
- Johnson, D.B.; Puzanov, I. Treatment of NRAS-mutant melanoma. Curr. Treat. Options Oncol. 2015, 16, 15. [Google Scholar] [CrossRef] [PubMed]
- Network, C.G.A. Genomic Classification of Cutaneous Melanoma. Cell 2015, 161, 1681–1696. [Google Scholar] [CrossRef]
- Wilkins, D.K.; Nathan, P.D. Therapeutic opportunities in noncutaneous melanoma. Ther. Adv. Med. Oncol. 2009, 1, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Del Vecchio, M.; Di Guardo, L.; Ascierto, P.A.; Grimaldi, A.M.; Sileni, V.C.; Pigozzo, J.; Ferraresi, V.; Nuzzo, C.; Rinaldi, G.; Testori, A.; et al. Efficacy and safety of ipilimumab 3mg/kg in patients with pretreated, metastatic, mucosal melanoma. Eur. J. Cancer 2014, 50, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Day, C.P.; Merlino, G.; Van Dyke, T. Preclinical mouse cancer models: A maze of opportunities and challenges. Cell 2015, 163, 39–53. [Google Scholar] [CrossRef] [PubMed]
- Perez-Guijarro, E.; Day, C.P.; Merlino, G.; Zaidi, M.R. Genetically engineered mouse models of melanoma. Cancer 2017, 123, 2089–2103. [Google Scholar] [CrossRef] [PubMed]
- Kaufman, C.K.; Mosimann, C.; Fan, Z.P.; Yang, S.; Thomas, A.J.; Ablain, J.; Tan, J.L.; Fogley, R.D.; van Rooijen, E.; Hagedorn, E.J.; et al. A zebrafish melanoma model reveals emergence of neural crest identity during melanoma initiation. Science 2016, 351, aad2197. [Google Scholar] [CrossRef] [PubMed]
- Noonan, F.P.; Recio, J.A.; Takayama, H.; Duray, P.; Anver, M.R.; Rush, W.L.; De Fabo, E.C.; Merlino, G. Neonatal sunburn and melanoma in mice. Nature 2001, 413, 271–272. [Google Scholar] [CrossRef] [PubMed]
- Jarrett, S.G.; Novak, M.; Harris, N.; Merlino, G.; Slominski, A.; Kaetzel, D.M. NM23 deficiency promotes metastasis in a UV radiation-induced mouse model of human melanoma. Clin. Exp. Metastasis 2013, 30, 25–36. [Google Scholar] [CrossRef] [PubMed]
- Noonan, F.P.; Zaidi, M.R.; Wolnicka-Glubisz, A.; Anver, M.R.; Bahn, J.; Wielgus, A.; Cadet, J.; Douki, T.; Mouret, S.; Tucker, M.A.; et al. Melanoma induction by ultraviolet A but not ultraviolet B radiation requires melanin pigment. Nat. Commun. 2012, 3, 884. [Google Scholar] [CrossRef] [PubMed]
- Khanna, C.; Lindblad-Toh, K.; Vail, D.; London, C.; Bergman, P.; Barber, L.; Breen, M.; Kitchell, B.; McNeil, E.; Modiano, J.F.; et al. The dog as a cancer model. Nat. Biotechnol. 2006, 24, 1065–1066. [Google Scholar] [CrossRef] [PubMed]
- Nishiya, A.T.; Massoco, C.O.; Felizzola, C.R.; Perlmann, E.; Batschinski, K.; Tedardi, M.V.; Garcia, J.S.; Mendonça, P.P.; Teixeira, T.F.; Zaidan Dagli, M.L. Comparative Aspects of Canine Melanoma. Vet. Sci. 2016, 3, 7. [Google Scholar] [CrossRef] [PubMed]
- Paoloni, M.; Khanna, C. Translation of new cancer treatments from pet dogs to humans. Nat. Rev. Cancer 2008, 8, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Smedley, R.C.; Spangler, W.L.; Esplin, D.G.; Kitchell, B.E.; Bergman, P.J.; Ho, H.Y.; Bergin, I.L.; Kiupel, M. Prognostic markers for canine melanocytic neoplasms: A comparative review of the literature and goals for future investigation. Vet. Pathol. 2011, 48, 54–72. [Google Scholar] [CrossRef] [PubMed]
- Bosenberg, M.; Arnheiter, H.; Kelsh, R. Melanoma in mankind’s best friend. Pigment Cell Melanoma Res. 2014, 27, 1. [Google Scholar] [CrossRef] [PubMed]
- Poorman, K.; Borst, L.; Moroff, S.; Roy, S.; Labelle, P.; Motsinger-Reif, A.; Breen, M. Comparative cytogenetic characterization of primary canine melanocytic lesions using array CGH and fluorescence in situ hybridization. Chromosome Res. 2015, 23, 171–186. [Google Scholar] [CrossRef] [PubMed]
- Bolon, B.; Calderwood Mays, M.B.; Hall, B.J. Characteristics of canine melanomas and comparison of histology and DNA ploidy to their biologic behavior. Vet. Pathol. 1990, 27, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Goldschmidt, M.; Hendrick, M. Tumors of the Skin and Soft Tissues; Iowa State Press: Ames, IA, USA, 2002; pp. 78–84. [Google Scholar]
- MacEwen, E.G.; Patnaik, A.K.; Harvey, H.J.; Hayes, A.A.; Matus, R. Canine oral melanoma: Comparison of surgery versus surgery plus Corynebacterium parvum. Cancer Investig. 1986, 4, 397–402. [Google Scholar] [CrossRef]
- Gillard, M.; Cadieu, E.; De Brito, C.; Abadie, J.; Vergier, B.; Devauchelle, P.; Degorce, F.; Dreano, S.; Primot, A.; Dorso, L.; et al. Naturally occurring melanomas in dogs as models for non-UV pathways of human melanomas. Pigment Cell Melanoma Res. 2014, 27, 90–102. [Google Scholar] [CrossRef] [PubMed]
- Bergman, P.J.; Kent, M.S.; Farese, J.P. Melanoma. In Withrow and MacEwen’s Small Animal Clinical Oncology; Page, R.L., Withrow, S.J., Vail, D.M., Eds.; Elsevier/Saunders: St. Louis, MO, USA, 2013. [Google Scholar]
- Bergman, P.J. Canine oral melanoma. Clin. Tech. Small Anim. Pract. 2007, 22, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Carvajal, R.D.; Spencer, S.A.; Lydiatt, W. Mucosal melanoma: A clinically and biologically unique disease entity. J. Natl. Compr. Cancer Netw. 2012, 10, 345–356. [Google Scholar] [CrossRef]
- Simpson, R.M.; Bastian, B.C.; Michael, H.T.; Webster, J.D.; Prasad, M.L.; Conway, C.M.; Prieto, V.M.; Gary, J.M.; Goldschmidt, M.H.; Esplin, D.G.; et al. Sporadic naturally occurring melanoma in dogs as a preclinical model for human melanoma. Pigment Cell Melanoma Res. 2014, 27, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Bostock, D.E. Prognosis after surgical excision of canine melanomas. Vet. Pathol. 1979, 16, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Blackwood, L.; Dobson, J.M. Radiotherapy of oral malignant melanomas in dogs. J. Am. Vet. Med. Assoc. 1996, 209, 98–102. [Google Scholar] [PubMed]
- Theon, A.P.; Rodriguez, C.; Madewell, B.R. Analysis of prognostic factors and patterns of failure in dogs with malignant oral tumors treated with megavoltage irradiation. J. Am. Vet. Med. Assoc. 1997, 210, 778–784. [Google Scholar] [PubMed]
- Rassnick, K.M.; Ruslander, D.M.; Cotter, S.M.; Al-Sarraf, R.; Bruyette, D.S.; Gamblin, R.M.; Meleo, K.A.; Moore, A.S. Use of carboplatin for treatment of dogs with malignant melanoma: 27 cases (1989–2000). J. Am. Vet. Med. Assoc. 2001, 218, 1444–1448. [Google Scholar] [CrossRef] [PubMed]
- Meier, F.; Busch, S.; Lasithiotakis, K.; Kulms, D.; Garbe, C.; Maczey, E.; Herlyn, M.; Schittek, B. Combined targeting of MAPK and AKT signalling pathways is a promising strategy for melanoma treatment. Br. J. Dermatol. 2007, 156, 1204–1213. [Google Scholar] [CrossRef] [PubMed]
- Garman, B.; Anastopoulos, I.N.; Krepler, C.; Brafford, P.; Sproesser, K.; Jiang, Y.; Wubbenhorst, B.; Amaravadi, R.; Bennett, J.; Beqiri, M.; et al. Genetic and Genomic Characterization of 462 Melanoma Patient-Derived Xenografts, Tumor Biopsies, and Cell Lines. Cell Rep. 2017, 21, 1936–1952. [Google Scholar] [CrossRef] [PubMed]
- Krepler, C.; Sproesser, K.; Brafford, P.; Beqiri, M.; Garman, B.; Xiao, M.; Shannan, B.; Watters, A.; Perego, M.; Zhang, G.; et al. A Comprehensive Patient-Derived Xenograft Collection Representing the Heterogeneity of Melanoma. Cell Rep. 2017, 21, 1953–1967. [Google Scholar] [CrossRef] [PubMed]
- Furney, S.J.; Turajlic, S.; Stamp, G.; Nohadani, M.; Carlisle, A.; Thomas, J.M.; Hayes, A.; Strauss, D.; Gore, M.; van den Oord, J.; et al. Genome sequencing of mucosal melanomas reveals that they are driven by distinct mechanisms from cutaneous melanoma. J. Pathol. 2013, 230, 261–269. [Google Scholar] [CrossRef] [PubMed]
- Fowles, J.S.; Denton, C.L.; Gustafson, D.L. Comparative analysis of MAPK and PI3K/AKT pathway activation and inhibition in human and canine melanoma. Vet. Comp. Oncol. 2015, 13, 288–304. [Google Scholar] [CrossRef] [PubMed]
- Wei, B.R.; Michael, H.T.; Halsey, C.H.C.; Peer, C.J.; Adhikari, A.; Dwyer, J.E.; Hoover, S.B.; El Meskini, R.; Kozlov, S.; Weaver Ohler, Z.; et al. Synergistic targeted inhibition of MEK and dual PI3K/mTOR diminishes viability and inhibits tumor growth of canine melanoma underscoring its utility as a preclinical model for human mucosal melanoma. Pigment Cell Melanoma Res. 2016, 29, 643–655. [Google Scholar] [CrossRef] [PubMed]
- Cosgarea, I.; Ugurel, S.; Sucker, A.; Livingstone, E.; Zimmer, L.; Ziemer, M.; Utikal, J.; Mohr, P.; Pfeiffer, C.; Pfohler, C.; et al. Targeted next generation sequencing of mucosal melanomas identifies frequent NF1 and RAS mutations. Oncotarget 2017, 8, 40683–40692. [Google Scholar] [CrossRef] [PubMed]
- Lyu, J.; Song, Z.; Chen, J.; Shepard, M.J.; Song, H.; Ren, G.; Li, Z.; Guo, W.; Zhuang, Z.; Shi, Y. Whole-exome sequencing of oral mucosal melanoma reveals mutational profile and therapeutic targets. J. Pathol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Shelly, S.; Chien, M.B.; Yip, B.; Kent, M.S.; Theon, A.P.; McCallan, J.L.; London, C.A. Exon 15 BRAF mutations are uncommon in canine oral malignant melanomas. Mamm. Genome 2005, 16, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Mochizuki, H.; Kennedy, K.; Shapiro, S.G.; Breen, M. BRAF Mutations in Canine Cancers. PLoS ONE 2015, 10, e0129534. [Google Scholar] [CrossRef] [PubMed]
- Koenig, A.; Bianco, S.R.; Fosmire, S.; Wojcieszyn, J.; Modiano, J.F. Expression and significance of p53, rb, p21/waf-1, p16/ink-4a, and PTEN tumor suppressors in canine melanoma. Vet. Pathol. 2002, 39, 458–472. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Li, Y.; Long, Y.; Tang, E.; Wang, R.; Huang, K.; Xie, C.; Chen, G. Increased p16 and p53 protein expression predicts poor prognosis in mucosal melanoma. Oncotarget 2017, 8, 53226–53233. [Google Scholar] [CrossRef] [PubMed]
- Sheng, X.; Kong, Y.; Li, Y.; Zhang, Q.; Si, L.; Cui, C.; Chi, Z.; Tang, B.; Mao, L.; Lian, B.; et al. GNAQ and GNA11 mutations occur in 9.5% of mucosal melanoma and are associated with poor prognosis. Eur. J. Cancer 2016, 65, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Newman, S.J.; Jankovsky, J.M.; Rohrbach, B.W.; LeBlanc, A.K. C-kit expression in canine mucosal melanomas. Vet. Pathol. 2012, 49, 760–765. [Google Scholar] [CrossRef] [PubMed]
- Murakami, A.; Mori, T.; Sakai, H.; Murakami, M.; Yanai, T.; Hoshino, Y.; Maruo, K. Analysis of KIT expression and KIT exon 11 mutations in canine oral malignant melanomas. Vet. Comp. Oncol. 2011, 9, 219–224. [Google Scholar] [CrossRef] [PubMed]
- Chu, P.Y.; Pan, S.L.; Liu, C.H.; Lee, J.; Yeh, L.S.; Liao, A.T. KIT gene exon 11 mutations in canine malignant melanoma. Vet. J. 2013, 196, 226–230. [Google Scholar] [CrossRef] [PubMed]
- Iussich, S.; Maniscalco, L.; Di Sciuva, A.; Iotti, B.; Morello, E.; Martano, M.; Gattino, F.; Buracco, P.; De Maria, R. PDGFRs expression in dogs affected by malignant oral melanomas: Correlation with prognosis. Vet. Comp. Oncol. 2017, 15, 462–469. [Google Scholar] [CrossRef] [PubMed]
- Mayr, B.; Schaffner, G.; Reifinger, M.; Zwetkoff, S.; Prodinger, B. N-ras mutations in canine malignant melanomas. Vet. J. 2003, 165, 169–171. [Google Scholar] [CrossRef]
- Kim, H.S.; Jung, M.; Kang, H.N.; Kim, H.; Park, C.W.; Kim, S.M.; Shin, S.J.; Kim, S.H.; Kim, S.G.; Kim, E.K.; et al. Oncogenic BRAF fusions in mucosal melanomas activate the MAPK pathway and are sensitive to MEK/PI3K inhibition or MEK/CDK4/6 inhibition. Oncogene 2017, 36, 3334–3345. [Google Scholar] [CrossRef] [PubMed]
- Tanami, H.; Imoto, I.; Hirasawa, A.; Yuki, Y.; Sonoda, I.; Inoue, J.; Yasui, K.; Misawa-Furihata, A.; Kawakami, Y.; Inazawa, J. Involvement of overexpressed wild-type BRAF in the growth of malignant melanoma cell lines. Oncogene 2004, 23, 8796–8804. [Google Scholar] [CrossRef] [PubMed]
- Guldberg, P.; thor Straten, P.; Birck, A.; Ahrenkiel, V.; Kirkin, A.F.; Zeuthen, J. Disruption of the MMAC1/PTEN gene by deletion or mutation is a frequent event in malignant melanoma. Cancer Res. 1997, 57, 3660–3663. [Google Scholar] [PubMed]
- Manca, A.; Lissia, A.; Capone, M.; Ascierto, P.A.; Botti, G.; Caraco, C.; Stanganelli, I.; Colombino, M.; Sini, M.; Cossu, A.; et al. Activating PIK3CA mutations coexist with BRAF or NRAS mutations in a limited fraction of melanomas. J. Transl. Med. 2015, 13, 37. [Google Scholar] [CrossRef] [PubMed]
- Hendricks, W.; Zismann, V. Somatic inactivating PTPRJ mutations and dysregulated pathways identified in canine melanoma by integrated comparative genomic analysis. BioRxiv 2017. [Google Scholar] [CrossRef]
- Marech, I.; Patruno, R.; Zizzo, N.; Gadaleta, C.; Introna, M.; Zito, A.F.; Gadaleta, C.D.; Ranieri, G. Masitinib (AB1010), from canine tumor model to human clinical development: Where we are? Crit. Rev. Oncol. Hematol. 2014, 91, 98–111. [Google Scholar] [CrossRef] [PubMed]
- Paoloni, M.; Davis, S.; Lana, S.; Withrow, S.; Sangiorgi, L.; Picci, P.; Hewitt, S.; Triche, T.; Meltzer, P.; Khanna, C. Canine tumor cross-species genomics uncovers targets linked to osteosarcoma progression. BMC Genom. 2009, 10, 625. [Google Scholar] [CrossRef] [PubMed]
- Rusk, A.; Cozzi, E.; Stebbins, M.; Vail, D.; Graham, J.; Valli, V.; Henkin, J.; Sharpee, R.; Khanna, C. Cooperative activity of cytotoxic chemotherapy with antiangiogenic thrombospondin-I peptides, ABT-526 in pet dogs with relapsed lymphoma. Clin. Cancer Res. 2006, 12, 7456–7464. [Google Scholar] [CrossRef] [PubMed]
- Eggermont, A.M.; Robert, C. New drugs in melanoma: It’s a whole new world. Eur. J. Cancer 2011, 47, 2150–2157. [Google Scholar] [CrossRef] [PubMed]
- McArthur, G.A.; Ribas, A. Targeting oncogenic drivers and the immune system in melanoma. J. Clin. Oncol. 2013, 31, 499–506. [Google Scholar] [CrossRef] [PubMed]
- Chapman, P.B.; Hauschild, A.; Robert, C.; Haanen, J.B.; Ascierto, P.; Larkin, J.; Dummer, R.; Garbe, C.; Testori, A.; Maio, M.; et al. Improved survival with vemurafenib in melanoma with BRAF V600E mutation. N. Engl. J. Med. 2011, 364, 2507–2516. [Google Scholar] [CrossRef] [PubMed]
- Dossett, L.A.; Kudchadkar, R.R.; Zager, J.S. BRAF and MEK inhibition in melanoma. Expert Opin. Drug Saf. 2015, 14, 559–570. [Google Scholar] [CrossRef] [PubMed]
- Decker, B.; Parker, H.G.; Dhawan, D.; Kwon, E.M.; Karlins, E.; Davis, B.W.; Ramos-Vara, J.A.; Bonney, P.L.; McNiel, E.A.; Knapp, D.W.; et al. Homologous Mutation to Human BRAF V600E Is Common in Naturally Occurring Canine Bladder Cancer–Evidence for a Relevant Model System and Urine-Based Diagnostic Test. Mol. Cancer Res. 2015, 13, 993–1002. [Google Scholar] [CrossRef] [PubMed]
- Handolias, D.; Salemi, R.; Murray, W.; Tan, A.; Liu, W.; Viros, A.; Dobrovic, A.; Kelly, J.; McArthur, G.A. Mutations in KIT occur at low frequency in melanomas arising from anatomical sites associated with chronic and intermittent sun exposure. Pigment Cell Melanoma Res. 2010, 23, 210–215. [Google Scholar] [CrossRef] [PubMed]
- Hodi, F.S.; Friedlander, P.; Corless, C.L.; Heinrich, M.C.; Mac Rae, S.; Kruse, A.; Jagannathan, J.; Van den Abbeele, A.D.; Velazquez, E.F.; Demetri, G.D.; et al. Major response to imatinib mesylate in KIT-mutated melanoma. J. Clin. Oncol. 2008, 26, 2046–2051. [Google Scholar] [CrossRef] [PubMed]
- Hodi, F.S.; Corless, C.L.; Giobbie-Hurder, A.; Fletcher, J.A.; Zhu, M.; Marino-Enriquez, A.; Friedlander, P.; Gonzalez, R.; Weber, J.S.; Gajewski, T.F.; et al. Imatinib for melanomas harboring mutationally activated or amplified KIT arising on mucosal, acral, and chronically sun-damaged skin. J. Clin. Oncol. 2013, 31, 3182–3190. [Google Scholar] [CrossRef] [PubMed]
- Bonkobara, M. Dysregulation of tyrosine kinases and use of imatinib in small animal practice. Vet. J. 2015, 205, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Hahn, K.A.; Ogilvie, G.; Rusk, T.; Devauchelle, P.; Leblanc, A.; Legendre, A.; Powers, B.; Leventhal, P.S.; Kinet, J.P.; Palmerini, F.; et al. Masitinib is safe and effective for the treatment of canine mast cell tumors. J. Vet. Intern. Med. 2008, 22, 1301–1309. [Google Scholar] [CrossRef] [PubMed]
- Ito, K.; Kobayashi, M.; Kuroki, S.; Sasaki, Y.; Iwata, T.; Mori, K.; Kuroki, T.; Ozawa, Y.; Tetsuka, M.; Nakagawa, T.; et al. The proteasome inhibitor bortezomib inhibits the growth of canine malignant melanoma cells in vitro and in vivo. Vet. J. 2013, 198, 577–582. [Google Scholar] [CrossRef] [PubMed]
- Cleary, J.M.; Shapiro, G.I. Development of phosphoinositide-3 kinase pathway inhibitors for advanced cancer. Curr. Oncol. Rep. 2010, 12, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Kwong, L.N.; Boland, G.M.; Frederick, D.T.; Helms, T.L.; Akid, A.T.; Miller, J.P.; Jiang, S.; Cooper, Z.A.; Song, X.; Seth, S.; et al. Co-clinical assessment identifies patterns of BRAF inhibitor resistance in melanoma. J. Clin. Investig. 2015, 125, 1459–1470. [Google Scholar] [CrossRef] [PubMed]
- Kent, M.S.; Collins, C.J.; Ye, F. Activation of the AKT and mammalian target of rapamycin pathways and the inhibitory effects of rapamycin on those pathways in canine malignant melanoma cell lines. Am. J. Vet. Res. 2009, 70, 263–269. [Google Scholar] [CrossRef] [PubMed]
- She, Q.B.; Halilovic, E.; Ye, Q.; Zhen, W.; Shirasawa, S.; Sasazuki, T.; Solit, D.B.; Rosen, N. 4E-BP1 is a key effector of the oncogenic activation of the AKT and ERK signaling pathways that integrates their function in tumors. Cancer Cell 2010, 18, 39–51. [Google Scholar] [CrossRef] [PubMed]
- Bailey, S.T.; Zhou, B.; Damrauer, J.S.; Krishnan, B.; Wilson, H.L.; Smith, A.M.; Li, M.; Yeh, J.J.; Kim, W.Y. mTOR inhibition induces compensatory, therapeutically targetable MEK activation in renal cell carcinoma. PLoS ONE 2014, 9, e104413. [Google Scholar] [CrossRef] [PubMed]
- Carracedo, A.; Ma, L.; Teruya-Feldstein, J.; Rojo, F.; Salmena, L.; Alimonti, A.; Egia, A.; Sasaki, A.T.; Thomas, G.; Kozma, S.C.; et al. Inhibition of mTORC1 leads to MAPK pathway activation through a PI3K-dependent feedback loop in human cancer. J. Clin. Investig. 2008, 118, 3065–3074. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.R.; Min, H.; Fang, J.F.; Zhou, F.; Deng, X.W.; Zhang, Y.Q. Activity of the novel dual phosphatidylinositol 3-kinase/mammalian target of rapamycin inhibitor NVP-BEZ235 against osteosarcoma. Cancer Biol. Ther. 2015, 16, 602–609. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of Cancer: The Next Generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Hamid, O.; Robert, C.; Daud, A.; Hodi, F.S.; Hwu, W.J.; Kefford, R.; Wolchok, J.D.; Hersey, P.; Joseph, R.W.; Weber, J.S.; et al. Safety and tumor responses with lambrolizumab (anti-PD-1) in melanoma. N. Engl. J. Med. 2013, 369, 134–144. [Google Scholar] [CrossRef] [PubMed]
- Snyder, A.; Makarov, V.; Merghoub, T.; Yuan, J.; Zaretsky, J.M.; Desrichard, A.; Walsh, L.A.; Postow, M.A.; Wong, P.; Ho, T.S.; et al. Genetic basis for clinical response to CTLA-4 blockade in melanoma. N. Engl. J. Med. 2014, 371, 2189–2199. [Google Scholar] [CrossRef] [PubMed]
- Maekawa, N.; Konnai, S.; Ikebuchi, R.; Okagawa, T.; Adachi, M.; Takagi, S.; Kagawa, Y.; Nakajima, C.; Suzuki, Y.; Murata, S.; et al. Expression of PD-L1 on canine tumor cells and enhancement of IFN-gamma production from tumor-infiltrating cells by PD-L1 blockade. PLoS ONE 2014, 9, e98415. [Google Scholar] [CrossRef] [PubMed]
- Hartley, G.; Faulhaber, E.; Caldwell, A.; Coy, J.; Kurihara, J.; Guth, A.; Regan, D.; Dow, S. Immune regulation of canine tumour and macrophage PD-L1 expression. Vet. Comp. Oncol. 2017, 15, 534–549. [Google Scholar] [CrossRef] [PubMed]
- Shosu, K.; Sakurai, M.; Inoue, K.; Nakagawa, T.; Sakai, H.; Morimoto, M.; Okuda, M.; Noguchi, S.; Mizuno, T. Programmed Cell Death Ligand 1 Expression in Canine Cancer. In Vivo 2016, 30, 195–204. [Google Scholar] [PubMed]
- Maekawa, N.; Konnai, S.; Okagawa, T.; Nishimori, A.; Ikebuchi, R.; Izumi, Y.; Takagi, S.; Kagawa, Y.; Nakajima, C.; Suzuki, Y.; et al. Immunohistochemical Analysis of PD-L1 Expression in Canine Malignant Cancers and PD-1 Expression on Lymphocytes in Canine Oral Melanoma. PLoS ONE 2016, 11, e0157176. [Google Scholar] [CrossRef] [PubMed]
- Muller, D.; Kontermann, R.E. Bispecific antibodies for cancer immunotherapy: Current perspectives. BioDrugs 2010, 24, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Jain, M.D.; Davila, M.L. Concise Review: Emerging Principles from the Clinical Application of Chimeric Antigen Receptor T Cell Therapies for B Cell Malignancies. Stem Cells 2017. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, S.A.; Dudley, M.E. Adoptive cell therapy for the treatment of patients with metastatic melanoma. Curr. Opin. Immunol. 2009, 21, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Wang, L.; Cui, H.; Wang, X.; Zhang, G.; Ma, J.; Han, H.; He, W.; Wang, W.; Zhao, Y.; et al. Anti-melanoma activity of T cells redirected with a TCR-like chimeric antigen receptor. Sci. Rep. 2014, 4, 3571. [Google Scholar] [CrossRef] [PubMed]
- Gyorffy, S.; Rodriguez-Lecompte, J.C.; Woods, J.P.; Foley, R.; Kruth, S.; Liaw, P.C.; Gauldie, J. Bone marrow-derived dendritic cell vaccination of dogs with naturally occurring melanoma by using human gp100 antigen. J. Vet. Intern. Med. 2005, 19, 56–63. [Google Scholar] [PubMed]
- Alexander, A.N.; Huelsmeyer, M.K.; Mitzey, A.; Dubielzig, R.R.; Kurzman, I.D.; Macewen, E.G.; Vail, D.M. Development of an allogeneic whole-cell tumor vaccine expressing xenogeneic gp100 and its implementation in a phase II clinical trial in canine patients with malignant melanoma. Cancer Immunol. Archol. Immunother. 2006, 55, 433–442. [Google Scholar] [CrossRef] [PubMed]
- Atherton, M.J.; Morris, J.S.; McDermott, M.R.; Lichty, B.D. Cancer immunology and canine malignant melanoma: A comparative review. Vet. Immunol. Immunopathol. 2016, 169, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Mata, M.; Vera, J.F.; Gerken, C.; Rooney, C.M.; Miller, T.; Pfent, C.; Wang, L.L.; Wilson-Robles, H.M.; Gottschalk, S. Toward immunotherapy with redirected T cells in a large animal model: Ex vivo activation, expansion, and genetic modification of canine T cells. J. Immunother. 2014, 37, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Bergman, P.J. Immunotherapy in veterinary oncology. Vet. Clin. N. Am. Small Anim. Pract. 2014, 44, 925–939. [Google Scholar] [CrossRef] [PubMed]
- Bergman, P.J.; McKnight, J.; Novosad, A.; Charney, S.; Farrelly, J.; Craft, D.; Wulderk, M.; Jeffers, Y.; Sadelain, M.; Hohenhaus, A.E.; et al. Long-term survival of dogs with advanced malignant melanoma after DNA vaccination with xenogeneic human tyrosinase: A phase I trial. Clin. Cancer Res. 2003, 9, 1284–1290. [Google Scholar] [PubMed]
- Grosenbaugh, D.A.; Leard, A.T.; Bergman, P.J.; Klein, M.K.; Meleo, K.; Susaneck, S.; Hess, P.R.; Jankowski, M.K.; Jones, P.D.; Leibman, N.F.; et al. Safety and efficacy of a xenogeneic DNA vaccine encoding for human tyrosinase as adjunctive treatment for oral malignant melanoma in dogs following surgical excision of the primary tumor. Am. J. Vet. Res. 2011, 72, 1631–1638. [Google Scholar] [CrossRef] [PubMed]
- Liao, J.C.; Gregor, P.; Wolchok, J.D.; Orlandi, F.; Craft, D.; Leung, C.; Houghton, A.N.; Bergman, P.J. Vaccination with human tyrosinase DNA induces antibody responses in dogs with advanced melanoma. Cancer Immunol. Arch. 2006, 6, 8. [Google Scholar]
- Wolchok, J.D.; Srinivasan, R.; Perales, M.A.; Houghton, A.N.; Bowne, W.B.; Blachere, N.E. Alternative roles for interferon-gamma in the immune response to DNA vaccines encoding related melanosomal antigens. Cancer Immunol. Arch. 2001, 1, 9. [Google Scholar]
- Ottnod, J.M.; Smedley, R.C.; Walshaw, R.; Hauptman, J.G.; Kiupel, M.; Obradovich, J.E. A retrospective analysis of the efficacy of Oncept vaccine for the adjunct treatment of canine oral malignant melanoma. Vet. Comp. Oncol. 2013, 11, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, S.A.; Yang, J.C.; Topalian, S.L.; Schwartzentruber, D.J.; Weber, J.S.; Parkinson, D.R.; Seipp, C.A.; Einhorn, J.H.; White, D.E. Treatment of 283 consecutive patients with metastatic melanoma or renal cell cancer using high-dose bolus interleukin 2. JAMA 1994, 271, 907–913. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, S.A.; Yang, J.C.; White, D.E.; Steinberg, S.M. Durability of complete responses in patients with metastatic cancer treated with high-dose interleukin-2: Identification of the antigens mediating response. Ann. Surg. 1998, 228, 307–319. [Google Scholar] [CrossRef] [PubMed]
- Atkins, M.B. Interleukin-2: Clinical applications. Semin. Oncol. 2002, 29, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Finocchiaro, L.M.; Fiszman, G.L.; Karara, A.L.; Glikin, G.C. Suicide gene and cytokines combined nonviral gene therapy for spontaneous canine melanoma. Cancer Gene Ther. 2008, 15, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Briggs, J.; Paoloni, M.; Chen, Q.-R.; Wen, X.; Khan, J.; Khanna, C. A Compendium of Canine Normal Tissue Gene Expression. PLoS ONE 2011, 6, e17107. [Google Scholar] [CrossRef] [PubMed]
- Gordon, I.; Paoloni, M.; Mazcko, C.; Khanna, C. The Comparative Oncology Trials Consortium: Using Spontaneously Occurring Cancers in Dogs to Inform the Cancer Drug Development Pathway. PLoS Med. 2009, 6, e1000161. [Google Scholar] [CrossRef] [PubMed]
- Paoloni, M.; Vail, D. Clinical Trials and Developmental Therapeutic; Elsevier/Saunders: St. Louis, MO, USA, 2013. [Google Scholar]

| Molecular/Genetic Evaluation | Canine | Human | ||||
|---|---|---|---|---|---|---|
| Specimen | # Affected/# Examined | Finding | Specimen | # Affected/# Examined | Finding | |
| NRAS | Tumor | 1/12 | Silent mutation at codon 52 [44] | PDX | 0/17 | All lack mutations [42] |
| Cell lines | 1/5 | Q61R mutation [44] | PDX | 2/10 | 1 G12A mutation; 1 G13D mutation [41] | |
| Tumor | 2/28 | Q61 mutation [45] | Tumor | 8/71 | G12, G13, or Q61 mutation [46] | |
| Cell lines | 2/5 | Q61 mutation [45] | Tumor; LN | 3/8 | 1 G12C mutation and copy number gain; 1 Q61R mutation; 1 copy number loss [5] | |
| BRAF | Tumor | 0/12 | All lack mutations [44] | PDX | 0/10 | All lack mutations [41] |
| Cell lines | 0/5 | All lack mutations [44] | PDX | 0/17 | All lack mutations [42] | |
| Tumor | 0/28 | All lack mutations [45] | Tumor | 0/19 | All lack mutations [47] | |
| Cell lines | 0/5 | All lack mutations [45] | Tumor | 2/8 | 1 K486E mutation; 1 copy number gain [5] | |
| Tumor | 0/11 | All lack mutations [48] | Tumor | 6/74 | 4 V600E mutations; 1 V600K mutation; | |
| Cell lines | 0/6 | All lack mutations [48] | 1 N188S mutation [46] | |||
| Tumor | 2/47 | V600E mutation [49] | ||||
| CDKN2A/p16 | Tumor | 14/20 | Copy number loss [27] | Tumor | 3/8 | Copy number loss [5] |
| Tumor | 10/12 | Decreased expression by IHC [50] | Tumor | 59/59 | 12 High CDKN2A expression; | |
| Cell lines | 4/6 | Decreased expression by IHC [50] | 47 Low CDKN2A expression by IHC [51] | |||
| p-ERK | TMA | 33/43 | IHC immunopositive [45] | TMA | 21/37 | IHC immunopositive [45] |
| Tumor | 19/28 | ERK activation by WB [45] | ||||
| Cell lines | 6/6 | Basal p-ERK increased by WB [48] | ||||
| Cell lines | 4/4 | Basal p-ERK increased by WB; cell lines sensitive to MEK inhibitor [44] | ||||
| GNAQ | Tumor | 13/284 | Mutation at codon 209 [52] | |||
| GNA11 | Tumor | 14/284 | Mutation at codon 209 [52] | |||
| KIT | Tumor | 13/20 | Copy number gain [27] | PDX | 0/17 | All lack mutations [42] |
| Tumor | 30/61 | IHC immunopositive [53] | PDX | 2/10 | Non-synonymous mutations [41] | |
| Tumor | 20/39 | All lack mutations; 20 IHC immunopositive [54] | Tumor | 2/8 | Non-synonymous mutations [5] | |
| Tumor | 33/34 | 1 missense mutation; | Tumor | 4/19 | 2 Non-synonymous mutations; 2 Deletions; | |
| 5 synonymous mutations at nt1743; | 3 of 4 in hotspot domains [47] | |||||
| 33 IHC immunopositive [55] | Tumor | 5/75 | Non-synonymous mutations; 1 of 5 mutations were activating [46] | |||
| MYC | Tumor | 16/20 | Copy number gain [27] | PDX | 1/10 | Single mutation [41] |
| NF1 | PDX | 0/17 | All lack mutations [42] | |||
| PDX | 1/10 | Frameshift mutation [41] | ||||
| Tumor | 0/19 | All lack mutations [47] | ||||
| Tumor | 1/8 | Copy number loss [5] | ||||
| Tumor | 13/75 | Non-synonymous mutations; 9 of 13 mutations were inactivating [46] | ||||
| p53 | Tumor | 8/12 | Decreased by IHC [50] | Tumor | 59/59 | 12 High expression; |
| Cell lines | 3/6 | Decreased by IHC [50] | 47 Low expression by IHC [51] | |||
| Tumor | 7/20 | 6 Copy number loss; 1 Copy number gain [27] | Tumor | 2/8 | Copy number loss [5] | |
| PDGFR | Tumor | 18/48 | IHC immunopositive [56] | |||
| PTEN | Tumor | 10/12 | Decreased by IHC [50] | Tumor | 1/8 | Copy number loss [5] |
| Cell lines | 3/6 | Decreased by IHC and WB [50] | PDX | 1/10 | Frameshift mutation [41] | |
| p-AKT | TMA | 41/43 | IHC immunopositive [45] | TMA | 31/40 | IHC immunopositive [45] |
| Tumor | 12/28 | AKT activation by WB [45] | ||||
| Cell lines | 3/5 | Basal p-AKT increased by WB; cell lines sensitive to rapamycin [44] |
| Elements of Strategy | Fundamental Action/Procedure | Constructive Consideration |
|---|---|---|
| Clinical documentation | ||
| • Patient data | Presentation/history, duration, previous work up, management | Breed and other background information useful to generate data on incidence |
| • Gross lesion documentation | Extent of disease. Description of specific anatomic location (not just indication of oral cavity); dimensions in mm, two axes; ulceration, evidence of dissemination | Photograph lesion with a ruler if possible |
| • Biopsy | Inclusion for diagnostic intent/therapeutic intent (excisional, incisional); preservation for correlative molecular analysis | Consideration of lateral extent as well as vertical depth of invasion; Attention paid to quality of sampling, preservation, QA, and utilization |
| • Pathology review | Development of features of malignancy for initial assessment for trial enrollments: Proliferation, growth pattern, invasion, and dissemination, etc. Continue refining prognostic summation; Inclusion of IHC panel if needed to establish diagnosis | Capture classical features outlined—Adapt how used initially vs. what becomes useful from adjunct molecular data and outcomes [ 25]; |
| Clinical staging/prognosis and monitoring | ||
| • Imaging for dissemination | Ultrasound of lymph nodes to detect metastasis (includes submandibular) | +/− consideration of removal for staging; alternative consideration ultrasound-guided fine needle aspirate cytology for staging |
| • CT (MRI) imaging evaluation • Biopsy | Lung particularly; lymph node; abdomen Monitoring response to therapy, as appropriate | Consideration of monitoring for brain involvement; inclusion of cranial imaging Lymph nodes or other palpable disease is recommended |
| • Endpoint assessment | Necropsy examination, with collection of tissue for research, and documentation of extent of disease/host response | |
| Quality of life measures | Assessments of fatigue, cardiac function, mucositis, altered mentation, serial assessments of metabolic and hemotologic toxicity, threshold of toxicity vs. response | Harmonized approach for multicenter trials similar to [ 111] |
| Client education | Informed consent; Should also include education on how the initiative is intended to explore benefits for both dogs and humans; Necropsy education | Necropsy education; emphasis on historical shortcomings impediment to progress. Education design beyond pro forma consent for necropsy |
| Follow up | Directly with owner/clients and indirectly with primary care clinician | |
| Genomics | Global discovery genomics, proteomics and informatic methods: develop and apply. Database and clinical monitoring integration. | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hernandez, B.; Adissu, H.A.; Wei, B.-R.; Michael, H.T.; Merlino, G.; Simpson, R.M. Naturally Occurring Canine Melanoma as a Predictive Comparative Oncology Model for Human Mucosal and Other Triple Wild-Type Melanomas. Int. J. Mol. Sci. 2018, 19, 394. https://doi.org/10.3390/ijms19020394
Hernandez B, Adissu HA, Wei B-R, Michael HT, Merlino G, Simpson RM. Naturally Occurring Canine Melanoma as a Predictive Comparative Oncology Model for Human Mucosal and Other Triple Wild-Type Melanomas. International Journal of Molecular Sciences. 2018; 19(2):394. https://doi.org/10.3390/ijms19020394
Chicago/Turabian StyleHernandez, Belen, Hibret A. Adissu, Bih-Rong Wei, Helen T. Michael, Glenn Merlino, and R. Mark Simpson. 2018. "Naturally Occurring Canine Melanoma as a Predictive Comparative Oncology Model for Human Mucosal and Other Triple Wild-Type Melanomas" International Journal of Molecular Sciences 19, no. 2: 394. https://doi.org/10.3390/ijms19020394
APA StyleHernandez, B., Adissu, H. A., Wei, B.-R., Michael, H. T., Merlino, G., & Simpson, R. M. (2018). Naturally Occurring Canine Melanoma as a Predictive Comparative Oncology Model for Human Mucosal and Other Triple Wild-Type Melanomas. International Journal of Molecular Sciences, 19(2), 394. https://doi.org/10.3390/ijms19020394






