Dissecting Pathogenetic Mechanisms and Therapeutic Strategies in Drosophila Models of Myotonic Dystrophy Type 1
Abstract
1. Introduction
2. DM1 Pathogenesis
3. Drosophila Could Serve as a Model Organism for DM1
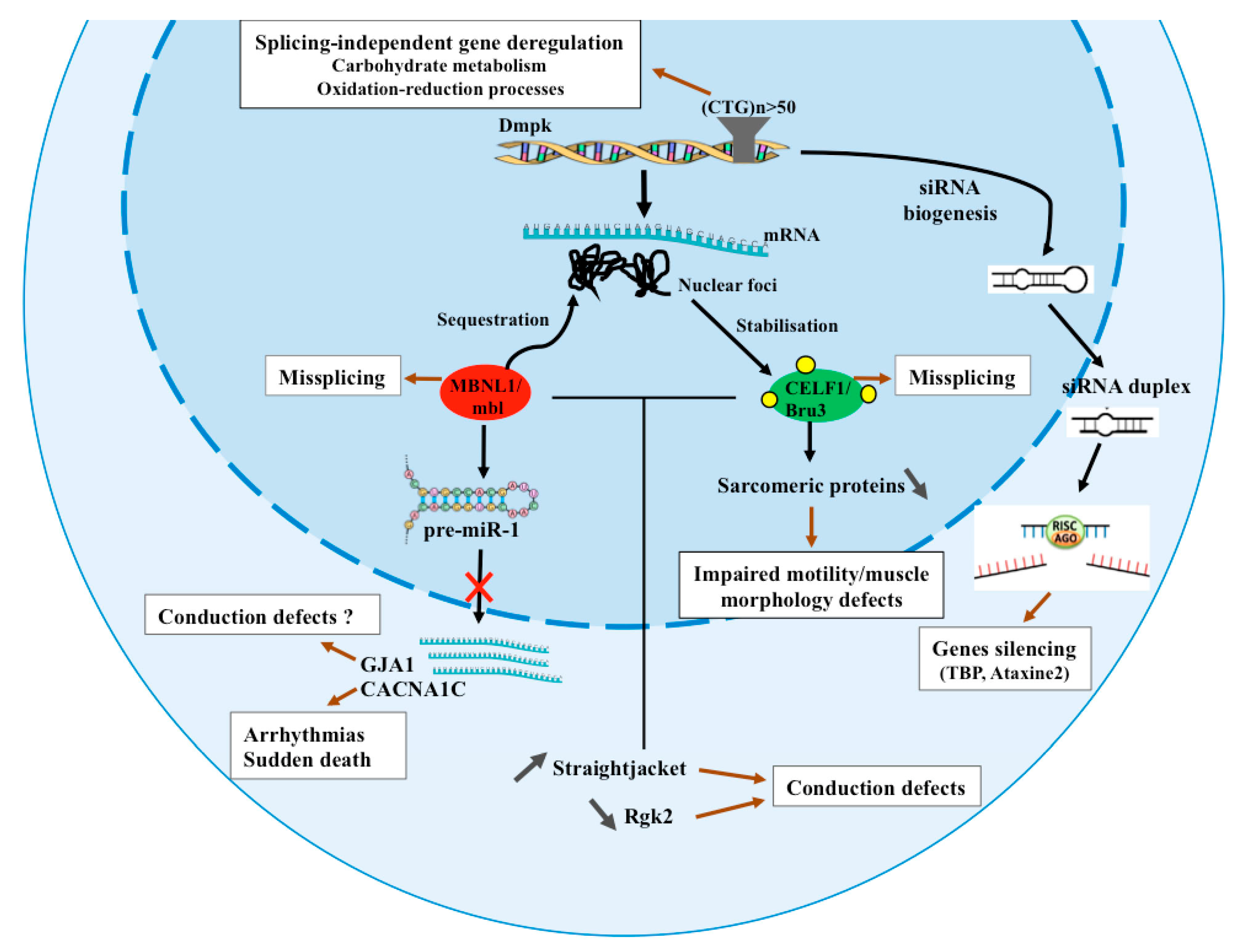
4. Identification of New Mechanisms Underlying DM1 Using Drosophila Models
5. Discovering Potential Gene and Drug-Based Therapeutic Strategies Using Fly DM1 Models
6. Conclusion and Remarks
Author Contributions
Funding
Conflicts of Interest
References
- Meola, G.; Cardani, R. Myotonic dystrophies: An update on clinical aspects, genetic, pathology, and molecular pathomechanisms. Biochim. Biophys. Acta BBA—Mol. Basis Dis. 2015, 1852, 594–606. [Google Scholar] [CrossRef] [PubMed]
- Buxton, J.; Shelbourne, P.; Davies, J.; Jones, C.; Tongeren, T.V.; Aslanidis, C.; de Jong, P.; Jansen, G.; Anvret, M.; Riley, B.; et al. Detection of an unstable fragment of DNA specific to individuals with myotonic dystrophy. Nature 1992, 355, 547–548. [Google Scholar] [CrossRef] [PubMed]
- Kroksmark, A.-K.; Ekström, A.-B.; Björck, E.; Tulinius, M. Myotonic dystrophy: muscle involvement in relation to disease type and size of expanded CTG-repeat sequence. Dev. Med. Child Neurol. 2005, 47, 478–485. [Google Scholar] [CrossRef] [PubMed]
- Harper, P.S. Congenital myotonic dystrophy in Britain. I. Clinical aspects. Arch. Dis. Child. 1975, 50, 505–513. [Google Scholar] [CrossRef] [PubMed]
- Fried, K.; Pajewski, M.; Mundel, G.; Caspi, E.; Spira, R. Thin ribs in neonatal myotonic dystrophy. Clin. Genet. 1975, 7, 417–420. [Google Scholar] [CrossRef] [PubMed]
- Tokgozoglu, L.S.; Ashizawa, T.; Pacifico, A.; Armstrong, R.M.; Epstein, H.F.; Zoghbi, W.A. Cardiac involvement in a large kindred with myotonic dystrophy. Quantitative assessment and relation to size of CTG repeat expansion. JAMA 1995, 274, 813–819. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, Y.; Ikeda, U.; Kojo, T.; Nishinaga, M.; Miyashita, H.; Kuroda, T.; Inoue, K.; Nishizawa, M.; Shimada, K. Cardiac abnormalities and cytosine-thymine-guanine trinucleotide repeats in myotonic dystrophy. Am. Heart J. 1997, 134, 292–297. [Google Scholar] [CrossRef]
- Cudia, P.; Bernasconi, P.; Chiodelli, R.; Mangiola, F.; Bellocci, F.; Dello Russo, A.; Angelini, C.; Romeo, V.; Melacini, P.; Politano, L.; et al. Risk of arrhythmia in type I myotonic dystrophy: the role of clinical and genetic variables. J. Neurol. Neurosurg. Psychiatry 2009, 80, 790–793. [Google Scholar] [CrossRef] [PubMed]
- Groh, W.J.; Lowe, M.R.; Zipes, D.P. Severity of cardiac conduction involvement and arrhythmias in myotonic dystrophy type 1 correlates with age and CTG repeat length. J. Cardiovasc. Electrophysiol. 2002, 13, 444–448. [Google Scholar] [CrossRef] [PubMed]
- Renna, L.V.; Bosè, F.; Iachettini, S.; Fossati, B.; Saraceno, L.; Milani, V.; Colombo, R.; Meola, G.; Cardani, R. Receptor and post-receptor abnormalities contribute to insulin resistance in myotonic dystrophy type 1 and type 2 skeletal muscle. PLoS ONE 2017, 12, e0184987. [Google Scholar] [CrossRef] [PubMed]
- Thornton, C.A.; Johnson, K.; Moxley, R.T. Myotonic dystrophy patients have larger CTG expansions in skeletal muscle than in leukocytes. Ann. Neurol. 1994, 35, 104–107. [Google Scholar] [CrossRef] [PubMed]
- Morales, F.; Couto, J.M.; Higham, C.F.; Hogg, G.; Cuenca, P.; Braida, C.; Wilson, R.H.; Adam, B.; del Valle, G.; Brian, R.; et al. Somatic instability of the expanded CTG triplet repeat in myotonic dystrophy type 1 is a heritable quantitative trait and modifier of disease severity. Hum. Mol. Genet. 2012, 21, 3558–3567. [Google Scholar] [CrossRef] [PubMed]
- Ashizawa, T.; Anvret, M.; Baiget, M.; Barceló, J.M.; Brunner, H.; Cobo, A.M.; Dallapiccola, B.; Fenwick, R.G.; Grandell, U.; Harley, H. Characteristics of intergenerational contractions of the CTG repeat in myotonic dystrophy. Am. J. Hum. Genet. 1994, 54, 414–423. [Google Scholar] [PubMed]
- Fu, Y.H.; Friedman, D.L.; Richards, S.; Pearlman, J.A.; Gibbs, R.A.; Pizzuti, A.; Ashizawa, T.; Perryman, M.B.; Scarlato, G.; Fenwick, R.G. Decreased expression of myotonin-protein kinase messenger RNA and protein in adult form of myotonic dystrophy. Science 1993, 260, 235–238. [Google Scholar] [CrossRef] [PubMed]
- Jansen, G.; Groenen, P.J.; Bächner, D.; Jap, P.H.; Coerwinkel, M.; Oerlemans, F.; van den Broek, W.; Gohlsch, B.; Pette, D.; Plomp, J.J.; et al. Abnormal myotonic dystrophy protein kinase levels produce only mild myopathy in mice. Nat. Genet. 1996, 13, 316–324. [Google Scholar] [CrossRef] [PubMed]
- Inukai, A.; Doyu, M.; Kato, T.; Liang, Y.; Kuru, S.; Yamamoto, M.; Kobayashi, Y.; Sobue, G. Reduced expression of DMAHP/SIX5 gene in myotonic dystrophy muscle. Muscle Nerve 2000, 23, 1421–1426. [Google Scholar] [CrossRef]
- Harris, S.E.; Winchester, C.L.; Johnson, K.J. Functional analysis of the homeodomain protein SIX5. Nucleic Acids Res. 2000, 28, 1871–1878. [Google Scholar] [CrossRef] [PubMed]
- Klesert, T.R.; Cho, D.H.; Clark, J.I.; Maylie, J.; Adelman, J.; Snider, L.; Yuen, E.C.; Soriano, P.; Tapscott, S.J. Mice deficient in Six5 develop cataracts: implications for myotonic dystrophy. Nat. Genet. 2000, 25, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Mankodi, A.; Logigian, E.; Callahan, L.; McClain, C.; White, R.; Henderson, D.; Krym, M.; Thornton, C.A. Myotonic dystrophy in transgenic mice expressing an expanded CUG repeat. Science 2000, 289, 1769–1773. [Google Scholar] [CrossRef] [PubMed]
- Davis, B.M.; McCurrach, M.E.; Taneja, K.L.; Singer, R.H.; Housman, D.E. Expansion of a CUG trinucleotide repeat in the 3’ untranslated region of myotonic dystrophy protein kinase transcripts results in nuclear retention of transcripts. Proc. Natl. Acad. Sci. USA 1997, 94, 7388–7393. [Google Scholar] [CrossRef] [PubMed]
- Taneja, K.L.; McCurrach, M.; Schalling, M.; Housman, D.; Singer, R.H. Foci of trinucleotide repeat transcripts in nuclei of myotonic dystrophy cells and tissues. J. Cell Biol. 1995, 128, 995–1002. [Google Scholar] [CrossRef] [PubMed]
- Fardaei, M.; Larkin, K.; Brook, J.D.; Hamshere, M.G. In vivo co-localisation of MBNL protein with DMPK expanded-repeat transcripts. Nucleic Acids Res. 2001, 29, 2766–2771. [Google Scholar] [CrossRef] [PubMed]
- Mankodi, A.; Urbinati, C.R.; Yuan, Q.P.; Moxley, R.T.; Sansone, V.; Krym, M.; Henderson, D.; Schalling, M.; Swanson, M.S.; Thornton, C.A. Muscleblind localizes to nuclear foci of aberrant RNA in myotonic dystrophy types 1 and 2. Hum. Mol. Genet. 2001, 10, 2165–2170. [Google Scholar] [CrossRef] [PubMed]
- Tian, B.; White, R.J.; Xia, T.; Welle, S.; Turner, D.H.; Mathews, M.B.; Thornton, C.A. Expanded CUG repeat RNAs form hairpins that activate the double-stranded RNA-dependent protein kinase PKR. RNA 2000, 6, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Kino, Y.; Washizu, C.; Oma, Y.; Onishi, H.; Nezu, Y.; Sasagawa, N.; Nukina, N.; Ishiura, S. MBNL and CELF proteins regulate alternative splicing of the skeletal muscle chloride channel CLCN1. Nucleic Acids Res. 2009, 37, 6477–6490. [Google Scholar] [CrossRef] [PubMed]
- Kalsotra, A.; Xiao, X.; Ward, A.J.; Castle, J.C.; Johnson, J.M.; Burge, C.B.; Cooper, T.A. A postnatal switch of CELF and MBNL proteins reprograms alternative splicing in the developing heart. Proc. Natl. Acad. Sci. USA 2008, 105, 20333–20338. [Google Scholar] [CrossRef] [PubMed]
- Kanadia, R.N.; Johnstone, K.A.; Mankodi, A.; Lungu, C.; Thornton, C.A.; Esson, D.; Timmers, A.M.; Hauswirth, W.W.; Swanson, M.S. A Muscleblind Knockout Model for Myotonic Dystrophy. Science 2003, 302, 1978–1980. [Google Scholar] [CrossRef] [PubMed]
- Kanadia, R.N.; Shin, J.; Yuan, Y.; Beattie, S.G.; Wheeler, T.M.; Thornton, C.A.; Swanson, M.S. Reversal of RNA missplicing and myotonia after muscleblind overexpression in a mouse poly(CUG) model for myotonic dystrophy. Proc. Natl. Acad. Sci. USA 2006, 103, 11748–11753. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, M.; Sellier, C.; Ney, M.; Villa, P.; Charlet-Berguerand, N.; Artero, R.; Llamusi, B. Daunorubicin reduces MBNL1 titration by expanded CUG repeat RNA and rescues cardiac dysfunctions in a Drosophila model of myotonic dystrophy. Dis. Model. Mech. 2018, dmm032557. [Google Scholar] [CrossRef] [PubMed]
- Vlasova, I.A.; Tahoe, N.M.; Fan, D.; Larsson, O.; Rattenbacher, B.; Sternjohn, J.R.; Vasdewani, J.; Karypis, G.; Reilly, C.S.; Bitterman, P.B.; et al. Conserved GU-rich elements mediate mRNA decay by binding to CUG-binding protein 1. Mol. Cell 2008, 29, 263–270. [Google Scholar] [CrossRef] [PubMed]
- Paillard, L.; Legagneux, V.; Beverley Osborne, H. A functional deadenylation assay identifies human CUG-BP as a deadenylation factor. Biol. Cell 2003, 95, 107–113. [Google Scholar] [CrossRef]
- Kim, Y.K.; Mandal, M.; Yadava, R.S.; Paillard, L.; Mahadevan, M.S. Evaluating the effects of CELF1 deficiency in a mouse model of RNA toxicity. Hum. Mol. Genet. 2014, 23, 293–302. [Google Scholar] [CrossRef] [PubMed]
- Reiter, L.T.; Potocki, L.; Chien, S.; Gribskov, M.; Bier, E. A systematic analysis of human disease-associated gene sequences in Drosophila melanogaster. Genome Res. 2001, 11, 1114–1125. [Google Scholar] [CrossRef] [PubMed]
- Bier, E. Drosophila, the golden bug, emerges as a tool for human genetics. Nat. Rev. Genet. 2005, 6, 9–23. [Google Scholar] [CrossRef] [PubMed]
- Houseley, J.M.; Wang, Z.; Brock, G.J.R.; Soloway, J.; Artero, R.; Perez-Alonso, M.; O’Dell, K.M.C.; Monckton, D.G. Myotonic dystrophy associated expanded CUG repeat muscleblind positive ribonuclear foci are not toxic to Drosophila. Hum. Mol. Genet. 2005, 14, 873–883. [Google Scholar] [CrossRef] [PubMed]
- de Haro, M.; Al-Ramahi, I.; De Gouyon, B.; Ukani, L.; Rosa, A.; Faustino, N.A.; Ashizawa, T.; Cooper, T.A.; Botas, J. MBNL1 and CUGBP1 modify expanded CUG-induced toxicity in a Drosophila model of myotonic dystrophy type 1. Hum. Mol. Genet. 2006, 15, 2138–2145. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Lopez, A.; Monferrer, L.; Garcia-Alcover, I.; Vicente-Crespo, M.; Alvarez-Abril, M.C.; Artero, R.D. Genetic and Chemical Modifiers of a CUG Toxicity Model in Drosophila. PLoS ONE 2008, 3, e1595. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Teng, X.; Bonini, N.M. Triplet Repeat–Derived siRNAs Enhance RNA–Mediated Toxicity in a Drosophila Model for Myotonic Dystrophy. PLoS Genet. 2011, 7, e1001340. [Google Scholar] [CrossRef] [PubMed]
- Cammarato, A.; Ahrens, C.H.; Alayari, N.N.; Qeli, E.; Rucker, J.; Reedy, M.C.; Zmasek, C.M.; Gucek, M.; Cole, R.N.; Eyk, J.E.V.; et al. A Mighty Small Heart: The Cardiac Proteome of Adult Drosophila melanogaster. PLoS ONE 2011, 6, e18497. [Google Scholar] [CrossRef] [PubMed]
- Bodmer, R. The gene tinman is required for specification of the heart and visceral muscles in Drosophila. Dev. Camb. Engl. 1993, 118, 719–729. [Google Scholar]
- Olson, E.N. Gene Regulatory Networks in the Evolution and Development of the Heart. Science 2006, 313, 1922–1927. [Google Scholar] [CrossRef] [PubMed]
- Bodmer, R. Heart development in Drosophila and its relationship to vertebrates. Trends Cardiovasc. Med. 1995, 5, 21–28. [Google Scholar] [CrossRef]
- Nguyen, H.H.; Wolfe, J.T.; Holmes, D.R.; Edwards, W.D. Pathology of the cardiac conduction system in myotonic dystrophy: A study of 12 cases. J. Am. Coll. Cardiol. 1988, 11, 662–671. [Google Scholar] [CrossRef]
- Benhayon, D.; Lugo, R.; Patel, R.; Carballeira, L.; Elman, L.; Cooper, J.M. Long-term arrhythmia follow-up of patients with myotonic dystrophy. J. Cardiovasc. Electrophysiol. 2015, 26, 305–310. [Google Scholar] [CrossRef] [PubMed]
- Groh, W.J.; Groh, M.R.; Saha, C.; Kincaid, J.C.; Simmons, Z.; Ciafaloni, E.; Pourmand, R.; Otten, R.F.; Bhakta, D.; Nair, G.V.; et al. Electrocardiographic abnormalities and sudden death in myotonic dystrophy type 1. N. Engl. J. Med. 2008, 358, 2688–2697. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, M.; Selma-Soriano, E.; Magny, E.; Couso, J.P.; Pérez-Alonso, M.; Charlet-Berguerand, N.; Artero, R.; Llamusi, B. Pentamidine rescues contractility and rhythmicity in a Drosophila model of myotonic dystrophy heart dysfunction. Dis. Model. Mech. 2015, 8, 1569–1578. [Google Scholar] [CrossRef] [PubMed]
- Plantie, E.; Nakamori, M.; Renaud, Y.; Huguet, A.; Choquet, C.; Dondi, C.; Miquerol, L.; Takahashi, M.; Gourdon, G.; Junion, G.; et al. Straightjacket/α 2δ 3 deregulation is associated with cardiac conduction defects in Myotonic Dystrophy type 1. bioRxiv 2018, 431569. [Google Scholar]
- Yenigun, V.B.; Sirito, M.; Amcheslavky, A.; Czernuszewicz, T.; Colonques-Bellmunt, J.; García-Alcover, I.; Wojciechowska, M.; Bolduc, C.; Chen, Z.; López Castel, A.; et al. (CCUG)n RNA toxicity in a Drosophila model of myotonic dystrophy type 2 (DM2) activates apoptosis. Dis. Model. Mech. 2017, 10, 993–1003. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Jackson, P.G.; Makan, J. Cardiac troponins. J. Clin. Pathol. 2004, 57, 1025–1026. [Google Scholar] [CrossRef] [PubMed]
- Philips, A.V.; Timchenko, L.T.; Cooper, T.A. Disruption of splicing regulated by a CUG-binding protein in myotonic dystrophy. Science 1998, 280, 737–741. [Google Scholar] [CrossRef] [PubMed]
- Artero, R.; Prokop, A.; Paricio, N.; Begemann, G.; Pueyo, I.; Mlodzik, M.; Perez-Alonso, M.; Baylies, M.K. The muscleblind gene participates in the organization of Z-bands and epidermal attachments of Drosophila muscles and is regulated by Dmef2. Dev. Biol. 1998, 195, 131–143. [Google Scholar] [CrossRef] [PubMed]
- Picchio, L.; Plantie, E.; Renaud, Y.; Poovthumkadavil, P.; Jagla, K. Novel Drosophila model of myotonic dystrophy type 1: phenotypic characterization and genome-wide view of altered gene expression. Hum. Mol. Genet. 2013, 22, 2795–2810. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Costa, J.M.; Garcia-Lopez, A.; Zuñiga, S.; Fernandez-Pedrosa, V.; Felipo-Benavent, A.; Mata, M.; Jaka, O.; Aiastui, A.; Hernandez-Torres, F.; Aguado, B.; et al. Expanded CTG repeats trigger miRNA alterations in Drosophila that are conserved in myotonic dystrophy type 1 patients. Hum. Mol. Genet. 2013, 22, 704–716. [Google Scholar] [CrossRef] [PubMed]
- Rau, F.; Freyermuth, F.; Fugier, C.; Villemin, J.-P.; Fischer, M.-C.; Jost, B.; Dembele, D.; Gourdon, G.; Nicole, A.; Duboc, D.; et al. Misregulation of miR-1 processing is associated with heart defects in myotonic dystrophy. Nat. Struct. Mol. Biol. 2011, 18, 840–845. [Google Scholar] [CrossRef] [PubMed]
- Splawski, I.; Timothy, K.W.; Sharpe, L.M.; Decher, N.; Kumar, P.; Bloise, R.; Napolitano, C.; Schwartz, P.J.; Joseph, R.M.; Condouris, K.; et al. Ca(V)1.2 calcium channel dysfunction causes a multisystem disorder including arrhythmia and autism. Cell 2004, 119, 19–31. [Google Scholar] [CrossRef] [PubMed]
- Naga Prasad, S.V.; Duan, Z.-H.; Gupta, M.K.; Surampudi, V.S.K.; Volinia, S.; Calin, G.A.; Liu, C.-G.; Kotwal, A.; Moravec, C.S.; Starling, R.C.; et al. Unique MicroRNA Profile in End-stage Heart Failure Indicates Alterations in Specific Cardiovascular Signaling Networks. J. Biol. Chem. 2009, 284, 27487–27499. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Li, R.; Zhang, C.; Bian, H.; Wang, F.; Xiao, J.; Liu, S.; Yi, W.; Zhang, M.; Wang, S.; et al. MicroRNA-7a/b Protects against Cardiac Myocyte Injury in Ischemia/Reperfusion by Targeting Poly(ADP-Ribose) Polymerase. PLoS ONE 2014, 9, e90096. [Google Scholar] [CrossRef] [PubMed]
- Geng, H.-H.; Li, R.; Su, Y.-M.; Xiao, J.; Pan, M.; Cai, X.-X.; Ji, X.-P. The Circular RNA Cdr1as Promotes Myocardial Infarction by Mediating the Regulation of miR-7a on Its Target Genes Expression. PLoS ONE 2016, 11, e0151753. [Google Scholar] [CrossRef] [PubMed]
- Picchio, L.; Legagneux, V.; Deschamps, S.; Renaud, Y.; Chauveau, S.; Paillard, L.; Jagla, K. Bruno-3 regulates sarcomere component expression and contributes to muscle phenotypes of myotonic dystrophy type 1. Dis. Model. Mech. 2018, 11. [Google Scholar] [CrossRef] [PubMed]
- de Haro, M.; Al-Ramahi, I.; Jones, K.R.; Holth, J.K.; Timchenko, L.T.; Botas, J. Smaug/SAMD4A Restores Translational Activity of CUGBP1 and Suppresses CUG-Induced Myopathy. PLoS Genet. 2013, 9, e1003445. [Google Scholar]
- Bargiela, A.; Cerro-Herreros, E.; Fernandez-Costa, J.M.; Vilchez, J.J.; Llamusi, B.; Artero, R. Increased autophagy and apoptosis contribute to muscle atrophy in a myotonic dystrophy type 1 Drosophila model. Dis. Model. Mech. 2015, 8, 679–690. [Google Scholar] [CrossRef] [PubMed]
- Cerro-Herreros, E.; Chakraborty, M.; Pérez-Alonso, M.; Artero, R.; Llamusí, B. Expanded CCUG repeat RNA expression in Drosophila heart and muscle trigger Myotonic Dystrophy type 1-like phenotypes and activate autophagocytosis genes. Sci. Rep. 2017, 7, 2843. [Google Scholar] [CrossRef] [PubMed]
- Cerro-Herreros, E.; Fernandez-Costa, J.M.; Sabater-Arcis, M.; Llamusi, B.; Artero, R. Derepressing muscleblind expression by miRNA sponges ameliorates myotonic dystrophy-like phenotypes in Drosophila. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Angelbello, A.J.; González, À.L.; Rzuczek, S.G.; Disney, M.D. Development of pharmacophore models for small molecules targeting RNA: Application to the RNA repeat expansion in myotonic dystrophy type 1. Bioorg. Med. Chem. Lett. 2016, 26, 5792–5796. [Google Scholar] [CrossRef] [PubMed]
- López-Morató, M.; Brook, J.D.; Wojciechowska, M. Small Molecules Which Improve Pathogenesis of Myotonic Dystrophy Type 1. Front. Neurol. 2018, 9, 349. [Google Scholar] [CrossRef] [PubMed]
- García-López, A.; Llamusí, B.; Orzáez, M.; Pérez-Payá, E.; Artero, R.D. In vivo discovery of a peptide that prevents CUG-RNA hairpin formation and reverses RNA toxicity in myotonic dystrophy models. Proc. Natl. Acad. Sci. USA 2011, 108, 11866–11871. [Google Scholar]
- Wong, C.-H.; Nguyen, L.; Peh, J.; Luu, L.M.; Sanchez, J.S.; Richardson, S.L.; Tuccinardi, T.; Tsoi, H.; Chan, W.Y.; Chan, H.Y.E.; et al. Targeting toxic RNAs that cause myotonic dystrophy type 1 (DM1) with a bisamidinium inhibitor. J. Am. Chem. Soc. 2014, 136, 6355–6361. [Google Scholar] [CrossRef] [PubMed]
- Luu, L.M.; Nguyen, L.; Peng, S.; Lee, J.; Lee, H.Y.; Wong, C.-H.; Hergenrother, P.J.; Chan, H.Y.E.; Zimmerman, S.C. A Potent Inhibitor of Protein Sequestration by Expanded Triplet (CUG) Repeats that Shows Phenotypic Improvements in a Drosophila Model of Myotonic Dystrophy. ChemMedChem 2016, 11, 1428–1435. [Google Scholar] [CrossRef] [PubMed]
| Screen | Identified Drug | Drosophila Model | Phenotype/Mode of Action | Ref. |
|---|---|---|---|---|
| Genetic Modifier Screening | Suppressors: cnc, Nurf-38, foi, coro, csk, spinster; Enhancers: seven up, viking, cg4589 | 480 interrupted CTG | CUG-induced rough-eye phenotype | [37] |
| Smaug | 480 interrupted CTG | Restoration of translational activity of CELF1/Bru-3, rescue of CUG-induced myopathy (prevents muscle wasting/restore muscle function) | [60] | |
| apoptosis/autophagy inhibitors (overexpression of DIAP1, mTOR or muscleblind) | 480 interrupted CTG | Rescue of the muscle-loss phenotype (atrophy) | [61] | |
| Drug Screening | Non-steroidal anti-inflammatory agents, dopamine receptors and monoamine uptake inhibitors, Na+ and Ca2+ metabolism, Muscarinic, cholinergic and histamine receptors inhibitors | 480 interrupted CTG) | Suppression of CUG-induced lethality | [37] |
| d-amino acid hexapeptide (ABP1) | 480 interrupted CTG | Biding to CUG repeats, elimination of CUG-RNA hairpin formation, suppression of CUG-induced lethality and muscle degeneration | [66] | |
| Ligand 3 | 480 interrupted CTG | Inhibition of MBNL1-CUG interaction, reduction of ribonuclear foci, partial rescue of misregulated splicing and degenerative phenotypes | [67] | |
| Ligand 2a | 480 interrupted CTG | Inhibition of MBNL1-CUG interaction, reduction of ribonuclear foci, amelioration of adult external eye degeneration and larval crawling defect | [68] | |
| Pentamidine | 250 pure CTG repeats | Inhibition of MBNL1-CUG interaction, reduction of ribonuclear foci, rescue of heart arrhythmicity and contractility, fly survival | [46] | |
| Daunorubicin hydrochloride | 250 pure CTG repeats | Inhibition of MBNL1-CUG interaction, rescue of Mbl-dependent missplicing/cardiac function recovery (systolic interval and diastolic interval), fly survival | [29] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Souidi, A.; Zmojdzian, M.; Jagla, K. Dissecting Pathogenetic Mechanisms and Therapeutic Strategies in Drosophila Models of Myotonic Dystrophy Type 1. Int. J. Mol. Sci. 2018, 19, 4104. https://doi.org/10.3390/ijms19124104
Souidi A, Zmojdzian M, Jagla K. Dissecting Pathogenetic Mechanisms and Therapeutic Strategies in Drosophila Models of Myotonic Dystrophy Type 1. International Journal of Molecular Sciences. 2018; 19(12):4104. https://doi.org/10.3390/ijms19124104
Chicago/Turabian StyleSouidi, Anissa, Monika Zmojdzian, and Krzysztof Jagla. 2018. "Dissecting Pathogenetic Mechanisms and Therapeutic Strategies in Drosophila Models of Myotonic Dystrophy Type 1" International Journal of Molecular Sciences 19, no. 12: 4104. https://doi.org/10.3390/ijms19124104
APA StyleSouidi, A., Zmojdzian, M., & Jagla, K. (2018). Dissecting Pathogenetic Mechanisms and Therapeutic Strategies in Drosophila Models of Myotonic Dystrophy Type 1. International Journal of Molecular Sciences, 19(12), 4104. https://doi.org/10.3390/ijms19124104




