Methodology of Drought Stress Research: Experimental Setup and Physiological Characterization
Abstract
1. Introduction
2. Experimental Models of Drought Stress
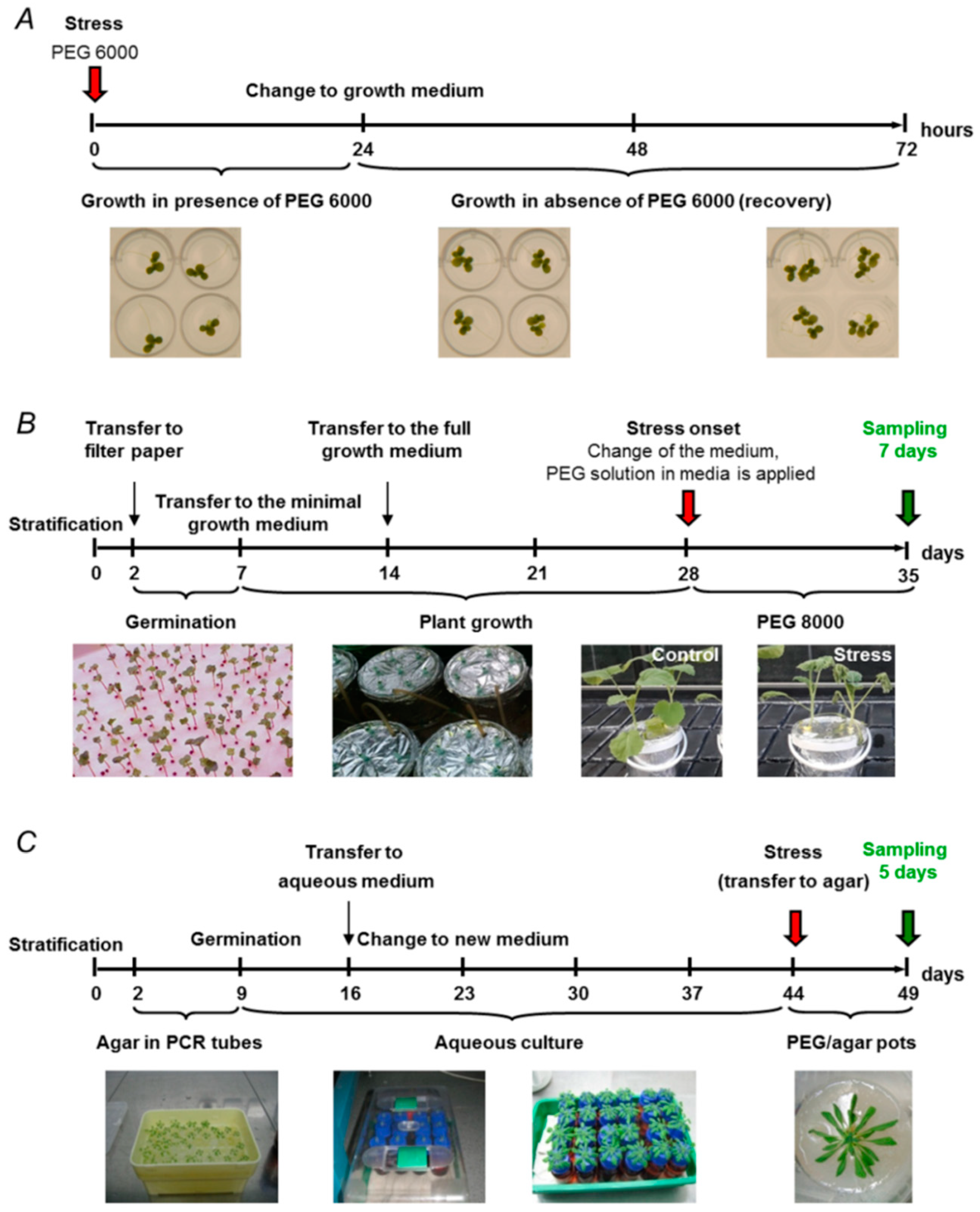
2.1. Soil-Based Drought Models
2.2. Drought Models Based on Hydroponic Aqueous Culture
2.3. Agar-Based Drought Models
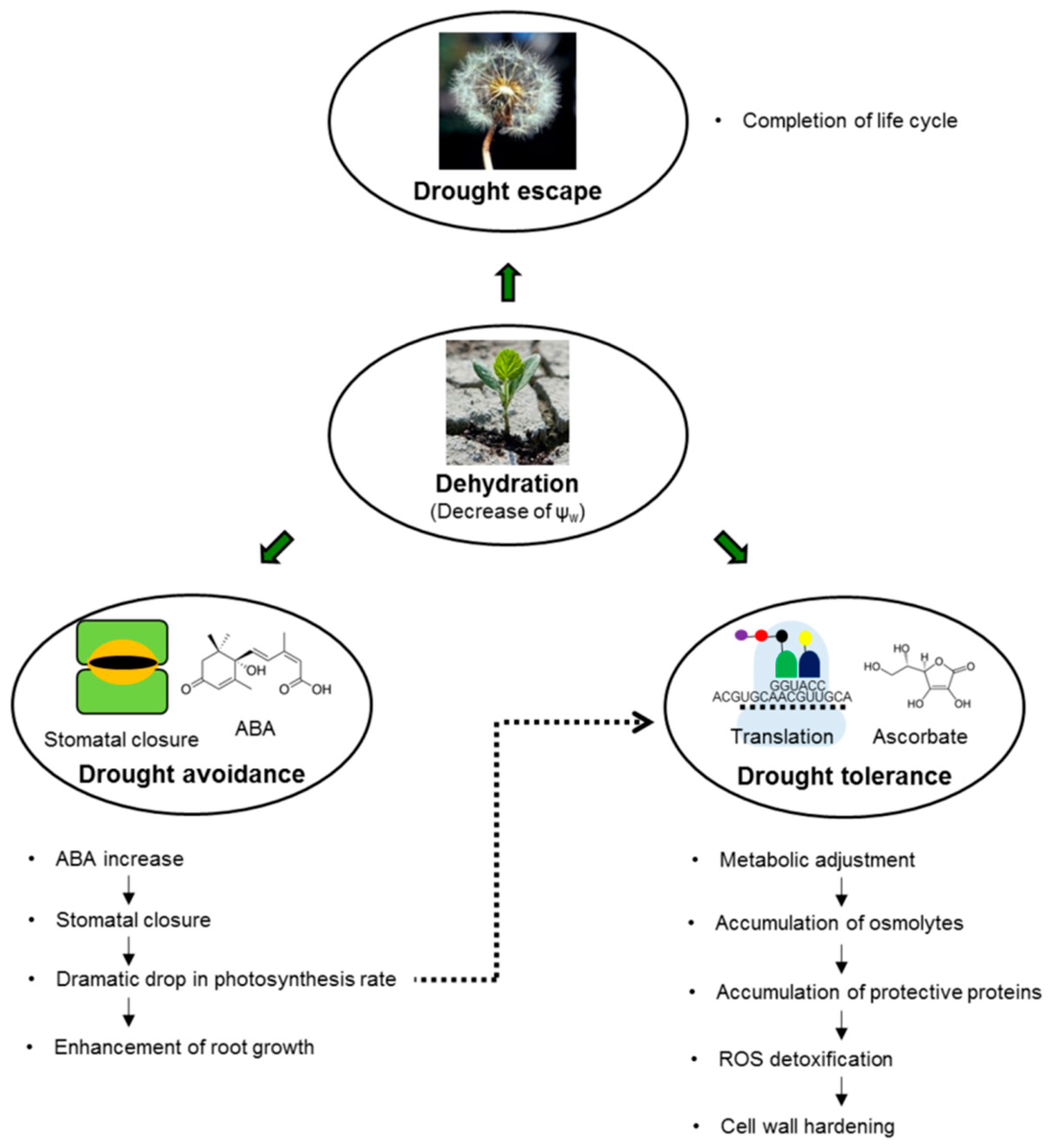
3. Physiological and Biochemical Characterization of Drought Stress
3.1. Water Status and Photosynthetic Parameters as Markers of Drought Stress
3.2. Changes in Phytohormone Patterns as Markers of Drought Stress
3.3. Metabolites as Markers of Drought Stress
3.4. Protective Proteins as Markers of Drought Stress
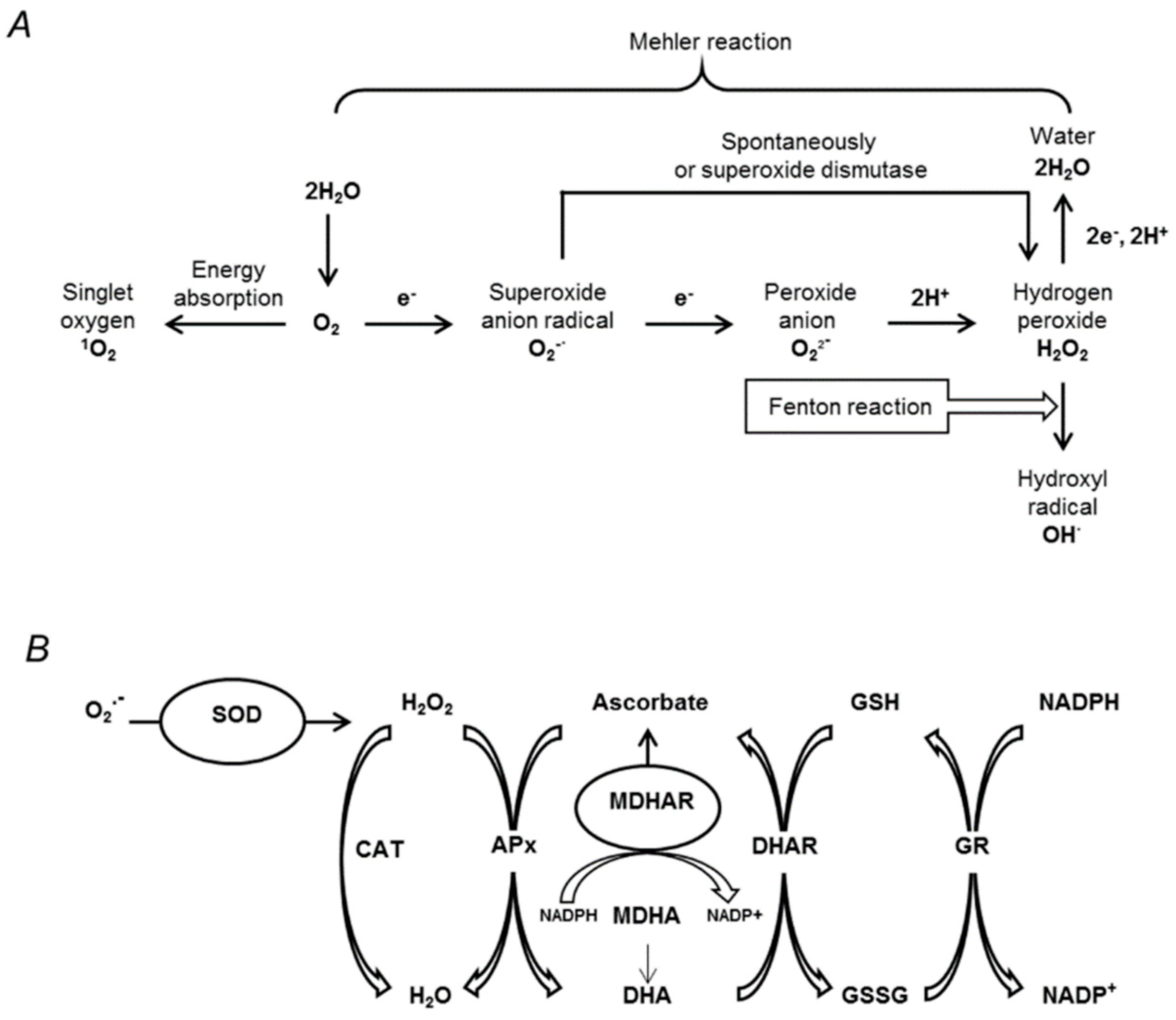
3.5. Oxidative Stress Associated with Drought
4. Conclusions
| Species | Drought Stress Model | Osmotically Active Agent | Age of Plant | Duration of Stress | Reference |
|---|---|---|---|---|---|
| Arabidopsis thaliana L. | Agar system | 50, 300 mmol/L mannitol | 7 days | 2 weeks | [164] |
| Arabidopsis thaliana L. | Agar system | 100, 200, 300 mmol/L mannitol | 8 days | 1 day | [165] |
| Arabidopsis thaliana L. | Agar system | 17% PEG8000 | 2 weeks | 3 days | [100] |
| Lemna minor L. | Hydroponic system (Microtiter plate formate possible) | PEG6000 or 8000 Variable conc. | Adult | 24 h | [68] |
| Hordeum vulgare L. | Soil system | No | Adult | Every 15 days until physiological maturity | [166] |
| Zea mays L. | Soil system | No | Adult | Every 15 days until physiological maturity | [166] |
| Zea mays L. | Hydroponic system | 15% PEG6000 | 5 weeks | 24 h | [167] |
| Populuseuphratica | Soil system | No | 2 months | 0, 4, 8 24, 48, 96 h | [168] |
| Solanum tuberosum L. | Agar system | Sorbitol (0.1, 0.2, 0.3, and 0.4 m) and PEG8000 (0%, 4.8%, and 9.6%) | 2 weeks | 3 weeks | [169] |
| Lolium perenne L. | Hydroponic system | 10, 20% PEG6000 | 1 week | 4 weeks | [170] |
| Solanum lycopersicum L. | Hydroponic system | 15% PEG8000 | 25 days | 0, 3, 6, 24, 48 h | [171] |
| Medicago sativa | Hydroponic system | 15% PEG6000 | 28 days | 24 h | [172] |
| Pistacia lentiscus | Soil system | 5, 10, 15, 20, 25% PEG6000 | 1,5 months | 20, 23 days | [173] |
| Brachypodiumdistachyon | Soil system | No | Vegetativestage | 4, 8, 12 days | [174] |
| Transgenicplum “Claudia verde” | Soil system | No | 8 weeks | 7, 15 days | [175] |
| Stipapurpurea | Soil system | No | Trefoilstage (about 3 weeks’ growth) | 7, 15 days | [176] |
| Saccharum spp. | Soil system | No | 2 months | 17 days | [177] |
| Hordeumvulgare L. | Hydroponic system | 20% PEG6000 | 31 days | 9 days | [178] |
| Brassica campestris ssp. | Hydroponic system | 60, 120%PEG6000 | 34 days | 7 days | [179] |
| Oryzasativa L. | Soil system | No | Reproductive stage | – | [180] |
| Cucumissativus L. | Hydroponic system | 2% PEG6000 | 2 weeks | 7 days | [181] |
| Parameter | Growth Model | Plant Object | Method | Reference |
|---|---|---|---|---|
| Physiological Markers | ||||
| Leaf water potential (MPa) | Soil | Cotton (Gossypium hirsutum L.) | Pressure chamber technique | [182] |
| Relative water content (RWC; %) | Soil | Potato (Solanum tuberosum L.) | RWC (%) = [(FW − DW)/(SW − DW)] × 100, where FW, DW, and SW are fresh, dry, and saturated (turgid)weights of leaf tissues, respectively | [183] |
| Stomatal conductance | Soil | Tomato (Lycopersicon esculentum Mill.) | Abaxial stomatal conductance measurement with diffusion porometer (AP4, Delta-T, Cambridge, UK) | [90] |
| Photosynthetic parameters (chlorophyll content and PSII activity) | Soil | Barley (Hordeum vulgare L.) | Determination of leaf chlorophyll using chlorophyll meter (SPAD-502, Minolta, Japan); measurement of chlorophyll fluorescence with portable fluorescence spectrometer (Handy PEA, Hansatech Instruments, Norfolk, UK) Fluorescence value Fv/Fm represents maximum quantum yield of PSII: Fv = Fm−Fo | [96] |
| Biochemical Markers | ||||
| Phytohormones | Soil | Clover (Trifolium subterraneum L.) | ABA analysis in xylem sap by ELISA | [184] |
| Soil | Wheat (Triticum aestivum L.) | ABA analysis by HPLC | [185] | |
| Metabolites | Soil | Triticum spp. | LMW drought stress–responsive metabolites in root and leaf samples of 7 wild and domesticated wheatrelatives revealed by GC-MS based comparative metabolomicsapproach | [186] |
| Protective proteins | Soil | Rice (Oryza sativa L.) | Expression pattern analysis of OsHSP50.2, an HSP90 family gene | [139] |
| Soil | Cotton (Gossypium tomentosum, Gossypium hirsutum) | LEA gene expression analysis and profiling | [144] | |
| ROS and antioxidant enzymes | Water culture + PEG6000 | Wheat genotypes | – | [187] |
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| 2D | two-dimensional |
| ABA | abscisic acid |
| APx | ascorbate peroxidase |
| CAT | catalase |
| DHA | dehydroascorbate |
| DHAR | dehydroascorbate reductase |
| GC-MS | gas chromatography–mass spectrometry |
| GR | glutathione reductase |
| GSH | reduced glutathione |
| GSSG | oxidized glutathione |
| LEA | late embryogenesis abundant |
| LRWC | leaf relative water content |
| MDHA | monodehydroascorbate |
| MDHAR | monodehydroascorbate reductase |
| NADPH | nicotinamide adenine dinucleotide phosphate |
| PAM | pulse amplitude modulation |
| PARP | poly (ADP-ribose) polymerase |
| PEG | polyethylene glycol |
| PS II | photosystem II |
| RCC | reactive carbonyl compound |
| ROS | reactive oxygen species |
| SOD | superoxide dismutase |
| St | Solanum tuberosum |
References
- Drought Assessment, Management, and Planning: Theory and Case Studies; Wilhite, D.A., Ed.; Springer: Boston, MA, USA, 1993; ISBN 978-1-4613-6416-0. [Google Scholar]
- Zhang, X.; Lu, G.; Long, W.; Zou, X.; Li, F.; Nishio, T. Recent progress in drought and salt tolerance studies in Brassica crops. Breed. Sci. 2014, 64, 60–73. [Google Scholar] [CrossRef] [PubMed]
- Shao, H.B.; Chu, L.-Y.; Jaleel, C.A.; Manivannan, P.; Panneerselvam, R.; Shao, M.-A. Understanding water deficit stress-induced changes in the basic metabolism of higher plants—Biotechnologically and sustainably improving agriculture and the ecoenvironment in arid regions of the globe. Crit. Rev. Biotechnol. 2009, 29, 131–151. [Google Scholar] [CrossRef] [PubMed]
- Basu, S.; Ramegowda, V.; Kumar, A.; Pereira, A. Plant adaptation to drought stress. F1000Research 2016, 5. [Google Scholar] [CrossRef] [PubMed]
- Farooq, M.; Wahid, A.; Kobayashi, N.; Fujita, D.; Basra, S.M.A. Plant drought stress: Effects, mechanisms and management. Agron. Sustain. Dev. 2009, 29, 185–212. [Google Scholar] [CrossRef]
- Jaleel, C.A.; Manivannan, P.; Wahid, A.; Farooq, M.; Al-Juburi, J.; Somasundaram, R.; Panneerselvam, R. Drought Stress in Plants: A Review on Morphological Characteristics and Pigments Composition. Int. J. Agric. Biol. 2009, 11, 7. [Google Scholar]
- Anjum, S.A.; Xie, X.; Wang, L.; Saleem, M.F.; Man, C.; Lei, W. Morphological, physiological and biochemical responses of plants to drought stress. Acta Physiol. Plant. 2015, 37. [Google Scholar] [CrossRef]
- Trenberth, K.E.; Dai, A.; van der Schrier, G.; Jones, P.D.; Barichivich, J.; Briffa, K.R.; Sheffield, J. Global warming and changes in drought. Nat. Clim. Chang. 2014, 4, 17–22. [Google Scholar] [CrossRef]
- Zhao, T.; Dai, A. The Magnitude and Causes of Global Drought Changes in the Twenty-First Century under a Low–Moderate Emissions Scenario. J. Clim. 2015, 28, 4490–4512. [Google Scholar] [CrossRef]
- Verslues, P.E.; Agarwal, M.; Katiyar-Agarwal, S.; Zhu, J.; Zhu, J.-K. Methods and concepts in quantifying resistance to drought, salt and freezing, abiotic stresses that affect plant water status. Plant J. 2006, 45, 523–539. [Google Scholar] [CrossRef] [PubMed]
- Deikman, J.; Petracek, M.; Heard, J.E. Drought tolerance through biotechnology: Improving translation from the laboratory to farmers’ fields. Curr. Opin. Biotechnol. 2012, 23, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Harb, A.; Krishnan, A.; Ambavaram, M.M.R.; Pereira, A. Molecular and Physiological Analysis of Drought Stress in Arabidopsis Reveals Early Responses Leading to Acclimation in Plant Growth. Plant Physiol. 2010, 154, 1254–1271. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Zhao, X.; Ling, Q.; Li, H.; Gao, X. Exotic shrub species (Caragana korshinskii) is more resistant to extreme natural drought than native species (Artemisia gmelinii) in a semiarid revegetated ecosystem. Agric. For. Meteorol. 2018, 263, 207–216. [Google Scholar] [CrossRef]
- Chirkova, T.V. Physiologicheskiie Osnovy Ustojchivosti Rastenii; Izdatelstvo Sankt-Peterburgskogo Universiteta: Saint Petersburg, Russia, 2002. [Google Scholar]
- Wang, X.; Cai, X.; Xu, C.; Wang, Q.; Dai, S. Drought-Responsive Mechanisms in Plant Leaves Revealed by Proteomics. Int. J. Mol. Sci. 2016, 17, 1706. [Google Scholar] [CrossRef] [PubMed]
- Kar, R.K. Plant responses to water stress: Role of reactive oxygen species. Plant Signal. Behav. 2011, 6, 1741–1745. [Google Scholar] [CrossRef] [PubMed]
- Gill, S.S.; Tuteja, N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol. Biochem. 2010, 48, 909–930. [Google Scholar] [CrossRef] [PubMed]
- Boyer, J.S.; Kramer, P.J. Water Relations of Plants and Soils; Academic Press Inc.: Waltham, MA, USA, 1995; ISBN 978-0-12-425060-4. [Google Scholar]
- Haswell, E.S.; Verslues, P.E. The ongoing search for the molecular basis of plant osmosensing. J. General Physiol. 2015, 145, 389–394. [Google Scholar] [CrossRef] [PubMed]
- Paudel, G.; Bilova, T.; Schmidt, R.; Greifenhagen, U.; Berger, R.; Tarakhovskaya, E.; Stöckhardt, S.; Balcke, G.U.; Humbeck, K.; Brandt, W.; et al. Changes in Arabidopsis thaliana advanced glycated proteome induced by the polyethylene glycol-related osmotic stress. J. Exp. Bot. 2016, 67, 6283–6295. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, C.L.; de Borja Reis, A.F.; Mazzafera, P.; Favarin, J.L. Determination of the Water Potential Threshold at Which Rice Growth Is Impacted. Plants 2018, 7, 48. [Google Scholar] [CrossRef] [PubMed]
- Btittelli, M. Measuring Soil Water Potential for Water Management in Agriculture: A Review. Sustainability 2010, 2, 1226–1251. [Google Scholar] [CrossRef]
- Kikuta, S.B.; Richter, H. Leaf discs or press saps? A comparison of techniques for the determination of osmotic potentials in freeze-thawed leaf material. J. Exp. Bot. 1992, 43, 1039–1044. [Google Scholar] [CrossRef]
- Voronin, P.Y.; Rakhmankulova, Z.F.; Shuyskaya, E.V.; Maevskaya, S.N.; Nikolaeva, M.K.; Maksimov, A.P.; Maximov, T.C.; Myasoedov, N.A.; Balnokin, Y.V.; Rymar, V.P.; et al. New method for quantitative determination of water potential of mesophyll cells’ apoplast in substomatal cavity of the leaf. Russ. J. Plant Physiol. 2017, 64, 452–456. [Google Scholar] [CrossRef]
- Razmkhah, H. Comparing Threshold Level Methods in Development of Stream Flow Drought Severity-Duration-Frequency Curves. Water Resour. Manag. 2017, 31, 4045–4061. [Google Scholar] [CrossRef]
- Grover, A.; Kapoor, A.; Lakshmi, O.S.; Agarwal, S.; Sahi, C.; Katiyar-Agarwal, S.; Agarwal, M.; Dubey, H. Understanding molecular alphabets of the plant abiotic stress responses. Plant Mol. Biol. 2001, 80, 206–216. [Google Scholar]
- Duque, A.S.; de Almeida, A.M.; da Silva, A.B.; da Silva, J.M.; Farinha, A.P.; Santos, D.; Fevereiro, P.; Araujo, S.S. Abiotic Stress Responses in Plants: Unraveling the Complexity of Genes and Networks to Survive. In Abiotic Stress—Plant Responses and Applications in Agriculture; InTech: London, UK, 2013; pp. 49–101. [Google Scholar] [CrossRef]
- Zhang, Q. Strategies for developing Green Super Rice. Proc. Natl. Acad. Sci. USA 2007, 104, 16402–16409. [Google Scholar] [CrossRef] [PubMed]
- Carboni, M.; Zeleny, D.; Acosta, A.T.R. Measuring ecological specialization along a natural stress gradient using a set of complementary niche breadth indices. J. Veg. Sci. 2016, 27, 892–903. [Google Scholar] [CrossRef]
- Levitt, J. Responses of Plants to Environmental Stresses; Academic Press: New York, NY, USA, 1972. [Google Scholar] [CrossRef]
- Joao, Z.; Ziany, B.; Silva, I.; Josiane, R.; Valdinei, S. Cotton response to water deficits at different growth stages. Rev. Caatinga 2017, 30, 980–990. [Google Scholar] [CrossRef]
- Antunes, C.; Pereira, A.J.; Fernandes, P.; Ramos, M.; Ascensão, L.; Correia, O.; Maguas, C. Understanding plant drought resistance in a Mediterranean coastal sand dune ecosystem: Differences between native and exotic invasive species. J. Plant Ecol. 2018, 11, 26–38. [Google Scholar] [CrossRef]
- Muller, B.; Pantin, F.; Génard, M.; Turc, O.; Freixes, S.; Piques, M.; Gibon, Y. Water deficits uncouple growth from photosynthesis, increase C content, and modify the relationships between C and growth in sink organs. J. Exp. Bot. 2011, 62, 1715–1729. [Google Scholar] [CrossRef] [PubMed]
- Li, L.J.; Gu, W.R.; Meng, Y.; Wang, Y.L.; Mu, J.Y.; Li, J.; Wei, S. Physiological and biochemical mechanism of spermidine improving drought resistance in maize seedlings under drought stress. Ying Yong Sheng Tai Xue Bao 2018, 29, 554–564. [Google Scholar] [CrossRef] [PubMed]
- Frolov, A.; Bilova, T.; Paudel, G.; Berger, R.; Balcke, G.U.; Birkemeyer, C.; Wessjohann, L.A. Early responses of mature Arabidopsis thaliana plants to reduced water potential in the agar-based polyethylene glycol infusion drought model. J. Plant Physiol. 2017, 208, 70–83. [Google Scholar] [CrossRef] [PubMed]
- Lipiec, J.; Doussan, C.; Nosalewicz, A.; Kondracka, K. Effect of drought and heat stresses on plant growth and yield: A review. Int. Agrophys. 2013, 27, 463–477. [Google Scholar] [CrossRef]
- Bhargava, S.; Sawant, K. Drought stress adaptation: Metabolic adjustment and regulation of gene expression. Plant Breed. 2013, 132, 21–32. [Google Scholar] [CrossRef]
- Tatrai, Z.A.; Sanoubar, R.; Pluhar, Z.; Mancarella, S.; Orsini, F.; Gianquinto, G. Morphological and Physiological Plant Responses to Drought Stress in Thymus citriodorus. Int. J. Agron. 2016, 10, 1–8. [Google Scholar] [CrossRef]
- Lamaoui, M.; Jemo, M.; Datla, R.; Bekkaoui, F. Heat and drought stresses in crops and approaches for their mitigation. Front. Chem. 2018, 6, 26. [Google Scholar] [CrossRef] [PubMed]
- Todaka, D.; Zhao, Y.; Yoshida, T.; Kudo, M.; Kidokoro, S.; Mizoi, J.; Kodaira, K.-S.; Takebayashi, Y.; Kojima, M.; Sakakibara, H. Temporal and spatial changes in gene expression, metabolite accumulation and phytohormone content in rice seedlings grown under drought stress conditions. Plant J. 2017, 90, 61–78. [Google Scholar] [CrossRef] [PubMed]
- Vinocur, B.; Altman, A. Recent advances in engineering plant tolerance to abiotic stress: Achievements and limitations. Curr. Opin. Biotechnol. 2005, 16, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Ford, K.L.; Cassin, A.; Bacic, A. Quantitative proteomic analysis of wheat cultivars with differing drought stress tolerance. Front. Plant Sci. 2011, 2, 44. [Google Scholar] [CrossRef] [PubMed]
- Gernot, B.; Alireza, A.; Kaul, H.-P. Management of crop water under drought: A review. Agron. Sustain. Dev. 2015, 35. [Google Scholar] [CrossRef]
- Thompson, A.J.; Thorne, E.T.; Burbidge, A.; Jackson, A.C.; Sharp, R.E.; Taylor, I.B. Complementation of notabilis, an abscisic acid-deficient mutant of tomato: Importance of sequence context and utility of partial complementation. Plant Cell Environ. 2004, 27, 459–471. [Google Scholar] [CrossRef]
- Vrablova, M.; Vrabl, D.; Hronkova, M.; Kubasek, J.; Santrucek, J. Stomatal function, density and pattern, and CO2 assimilation in Arabidopsis thaliana tmm1 and sdd1-1 mutants. Plant Biol. 2017, 19, 689–701. [Google Scholar] [CrossRef] [PubMed]
- Seminario, A.; Song, L.; Zulet, A.; Nguyen, H.T.; González, E.M.; Larrainzar, E. Drought stress causes a reduction in the biosynthesis of ascorbic acid in soybean plants. Front. Plant Sci. 2017, 8, 1042. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, N.; Malagoli, M.; Wirtz, M.; Hell, R. Drought stress in maize causes differential acclimation responses of glutathione and sulfur metabolism in leaves and roots. BMC Plant Biol. 2016, 16, 247. [Google Scholar] [CrossRef] [PubMed]
- Staudinger, C.; Mehmeti-Tershani, V.; Gil-Quintana, E.; Gonzalez, E.M.; Hofhansl, F.; Bachmann, G.; Wienkoop, S. Evidence for a rhizobia-induced drought stress response strategy in Medicago truncatula. J. Proteom. 2016, 136, 202–213. [Google Scholar] [CrossRef] [PubMed]
- Koyama, R.; Itoh, H.; Kimura, S.; Morioka, A.; Uno, Y. Augmentation of Antioxidant Constituents by Drought Stress to Roots in Leafy Vegetables. HortTechnology 2012, 22, 121–125. [Google Scholar] [CrossRef]
- Ito, Y.; Katsura, K.; Maruyama, K.; Taji, T.; Kobayashi, M.; Seki, M.; Shinozaki, K.; Yamaguchi-Shinozaki, K. Functional analysis of rice DREB1/CBF-type transcription factors involved in cold-responsive gene expression in transgenic rice. Plant Cell Physiol. 2006, 47, 141–153. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Duursma, R.A.; Medlyn, B.E.; Kelly, J.W.G.; Prentice, I.C. How should we model plant responses to drought? An analysis of stomatal and non-stomatal responses to water stress. Agric. Forest Meteorol. 2013, 182, 204–214. [Google Scholar] [CrossRef]
- Ji, H.; Liu, L.; Li, K.; Xie, Q.; Wang, Z.; Zhao, X.; Li, X. PEG-mediated osmotic stress induces premature differentiation of the root apical meristem and outgrowth of lateral roots in wheat. J. Exp. Bot. 2014, 65, 4863–4872. [Google Scholar] [CrossRef] [PubMed]
- Hellal, F.A.; El-Shabrawi, H.M.; Abd El-Hady, M.; Khatab, I.A.; El-Sayed, S.A.A.; Abdelly, C. Influence of PEG induced drought stress on molecular and biochemical constituents and seedling growth of Egyptian barley cultivars. J. Genet. Eng. Biotechnol. 2018, 16, 203–212. [Google Scholar] [CrossRef]
- Amist, N.; Singh, N.B. PEG imposed water deficit and physiological alterations in hydroponic cabbage. Iran. J. Plant Physiol. 2016, 8, 1653–1658. [Google Scholar]
- Hohl, M.; Schopfer, P. Water Relations of Growing Maize Coleoptiles: Comparison between Mannitol and Polyethylene Glycol 6000 as External Osmotica for Adjusting Turgor Pressure. Plant Physiol. 1991, 95, 716–722. [Google Scholar] [CrossRef] [PubMed]
- Munns, R. Comparative physiology of salt and water stress. Plant Cell Environ. 2002, 25, 239–250. [Google Scholar] [CrossRef] [PubMed]
- Verslues, P.E.; Ober, E.S.; Sharp, R.E. Root growth and oxygen relations at low water potentials. Impact of oxygen availability in polyethylene glycol solutions. Plant Physiol. 1998, 116, 1403–1412. [Google Scholar] [CrossRef] [PubMed]
- Hassan, N.S.; Shaaban, L.D.; Hashem, E.S.A.; Seleem, E.E. In vitro selection for water stress tolerant callus line of Helianthus annus L. cv. Myak. Int. J. Agric. Biol. 2004, 6, 13–18. [Google Scholar]
- Zhong, Y.-P.; Li, Z.; Bai, D.-F.; Qi, X.-J.; Chen, J.-Y.; Wei, C.-G.; Lin, M.-M.; Fang, J.-B. In Vitro Variation of Drought Tolerance in Five Actinidia Species. J. Am. Soc. Hortic. Sci. 2018, 143, 226–234. [Google Scholar] [CrossRef]
- Chutia, J.; Borah, S.P. Water Stress Effects on Leaf Growth and Chlorophyll Content but Not the Grain Yield in Traditional Rice (Oryza sativa Linn.) Genotypes of Assam, India II. Protein and Proline Status in Seedlings under PEG Induced Water Stress. Am. J. Plant Sci. 2012, 3, 971. [Google Scholar] [CrossRef]
- Meher; Shivakrishna, P.; Reddy, K.A.; Rao, D.M. Effect of PEG-6000 imposed drought sress on RNA content, relative water content (RWC), and chlorophyll content in peanut leaves and roots. Saudi J. Biol. Sci. 2018, 25, 285–289. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Yang, H.; Gosling, S.N.; Kummu, M.; Flörke, M.; Pfister, S.; Hanasaki, N.; Wada, Y.; Zhang, X.; Zheng, C.; et al. Water scarcity assessments in the past, present, and future: REVIEW ON WATER SCARCITY ASSESSMENT. Earth’s Future 2017, 5, 545–559. [Google Scholar] [CrossRef] [PubMed]
- Bressan, R.A.; Hasegawa, P.M.; Handa, A.K. Resistance of cultured higher plant cells to polyethylene glycol-induced water stress. Plant Sci. Lett. 1981, 21, 23–30. [Google Scholar] [CrossRef]
- Rao, S.; Ftz, J. In vitro selection and characterization of polyethylene glycol (PEG) tolerant callus lines and regeneration of plantlets from the selected callus lines in sugarcane (Saccharum officinarum L.). Physiol. Mol. Biol. Plants 2013, 19, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Senaratna, T.; Touchell, D.; Bunn, E.; Dixon, K. Acetyl salicylic acid (Aspirin) and salicylic acid induce multiple stress tolerance in bean and tomato plants. Plant Growth Regul. 2000, 30, 157–161. [Google Scholar] [CrossRef]
- Vanden Bossche, H.; Marichal, P.; Gorrens, J.; Geerts, H.; Janssen, P.A.J. Mode of action studies: basis for the search of new antifungal drugs. Ann. N Y Acad. Sci. 1988, 544, 191–207. [Google Scholar] [CrossRef] [PubMed]
- Thielert, W. A unique product: The story of the imidacloprid stress shield. Pflanzenschutz Nachrichten-Bayer 2007, 59, 73. [Google Scholar]
- Geissler, T.; Wessjohann, L.A. Whole-Plant Microtiter Plate Assay for Drought Stress Tolerance-Inducing Effects. J. Plant Growth Regul. 2011, 30, 504–511. [Google Scholar] [CrossRef]
- Rissel, D.; Heym, P.P.; Thor, K.; Brandt, W.; Wessjohann, L.A.; Peiter, E. No silver bullet-Canonical Poly (ADP-Ribose) Polymerases (PARPs) are no universal factors of abiotic and biotic stress resistance of Arabidopsis thaliana. Front. Plant Sci. 2017, 8, 59. [Google Scholar] [CrossRef] [PubMed]
- Funar-Timofei, S.; Borota, A.; Crisan, L. Combined molecular docking and QSAR study of fused heterocyclic herbicide inhibitors of D1 protein in photosystem II of plants. Mol. Divers. 2018, 21, 437–454. [Google Scholar] [CrossRef] [PubMed]
- Sardare, M.D.; Admane, S.V. A review on plant without soil. Int. J. Res. Eng. Technol. 2013, 2, 299–304. [Google Scholar] [CrossRef]
- Bilova, T.; Lukasheva, E.; Brauch, D.; Greifenhagen, U.; Paudel, G.; Tarakhovskaya, E.; Frolova, N.; Mittasch, J.; Balcke, G.U.; Tissier, A.; et al. A Snapshot of the Plant Glycated Proteome: Structural, Functional, and Mechanistic Aspects. J. Biol. Chem. 2016, 291, 7621–7636. [Google Scholar] [CrossRef] [PubMed]
- Simon, C.; Brad, H.; Maclin, D.; Bo, X.; Asmini, A.; Sam, H.; Lucy, A.; Vanessa, C.; Monique, S.; Sigfredo, F.; et al. Protocol: Optimising hydroponic growth systems for nutritional and physiological analysis of Arabidopsis thaliana and other plants. Plant Methods 2013, 9, 4. [Google Scholar] [CrossRef]
- Jacomini, E.; Bertani, A.; Mapelli, S. Accumulation of polyethylene glycol 6000 and its effects on water content and carbohydrate level in water-stressed tomato plants. Can. J. Bot. 1988, 66, 970–973. [Google Scholar] [CrossRef]
- Blum, A. Osmotic adjustment is a prime drought stress adaptive engine in support of plant production: Osmotic adjustment and plant production. Plant Cell Environ. 2017, 40, 4–10. [Google Scholar] [CrossRef] [PubMed]
- Van der Weele, C.M.; Spollen, W.G.; Sharp, R.E.; Baskin, T.I. Growth of Arabidopsis thaliana seedlings under water deficit studied by control of water potential in nutrient-agar media. J. Exp. Bot. 2000, 51, 1555–1562. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.H.; Bhaskaran, S.; Miller, F.R. Screening for drought tolerance in Sorghum using cell culture. In Vitro Cell Dev. Biol. 1985, 21, 541. [Google Scholar] [CrossRef]
- Chen, T.; Fluhr, R. Singlet Oxygen Plays an Essential Role in the Root’s Response to Osmotic Stress. Plant Physiol. 2018, 177, 1717–1727. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Wu, J.; Lv, B.; Li, J.; Gao, Z.; Xu, W.; Baluška, F.; Shi, W.; Shaw, P.C.; Zhang, J. Involvement of 14-3-3 protein GRF9 in root growth and response under polyethylene glycol-induced water stress. J. Exp. Bot. 2015, 66, 2271–2281. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.J.; Zhang, X.K.; Zou, C.S.; Cheng, Y.; Zhen, P.Y.; Li, G.Y. Effects of drought simulated by PEG-6000 on germination and seedling growth of rapeseed (Brassica napus L.). Chin.J.Oil Crop Sci. 1998, 29, 425–430. [Google Scholar] [CrossRef]
- Channaoui, S.; Kahkahi, R.E.; Charafi, J.; Mazouz, H.; Fechtali, M.E.; Nabloussi, A. Germination and Seedling Growth of a Set of Rapeseed (Brassica napus) Varieties under Drought Stress Conditions. Int. J. Environ. Agric. Biotechnol. 2017, 2. [Google Scholar] [CrossRef]
- Hamayun, M.; Khan, S.A.; Shinwari, Z.K.; Khan, A.L.; Ahmad, N.; Lee, I.-J. Effect of polyethylene glycol induced drought stress on physio-hormonal attributes of soybean. Abstractsofpapers 2010, 42, 977–986. [Google Scholar]
- Tardieu, F. Drought perception by plants do cells of droughted plants experience water stress? Plant Growth Regul. 1996, 20, 93–104. [Google Scholar] [CrossRef]
- Raghavendra, A.S.; Gonugunta, V.K.; Christmann, A.; Grill, E. ABA perception and signalling. Trends Plant Sci. 2010, 15, 395–401. [Google Scholar] [CrossRef] [PubMed]
- Govender, M.; Govender, P.; Weiersbye, I.; Witkowski, E.; Ahmed, F. Review of commonly used remote sensing and ground-based technologies to measure plant water stress. Water SA 2009, 35. [Google Scholar] [CrossRef]
- Donovan, L.; Linton, M.; Richards, J. Predawn plant water potential does not necessarily equilibrate with soil water potential under well-watered conditions. Oecologia 2001, 129, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Soltys-Kalina, D.; Plich, J.; Strzelczyk-Zyta, D.; Sliwka, J.; Marczewski, W. The effect of drought stress on the leaf relative water content and tuber yield of a half-sib family of ‘Katahdin’-derived potato cultivars. Breed. Sci. 2016, 66, 328–331. [Google Scholar] [CrossRef] [PubMed]
- De Silva, M.A.; Jifon, J.L.; da Silva, J.A.G.; Sharma, V. Use of physiological parameters as fast tools to screen for drought tolerance in sugarcane. Braz. J. Plant Physiol. 2007, 19, 193–201. [Google Scholar] [CrossRef]
- Novak, V.A.; Osmolovskaya, N. Phytomonitoring in plant physiology: Organization, arrangement, and possibilities. J. Plant Physiol. 1997, 44, 121–128. [Google Scholar]
- Sobeih, W.Y. Long-distance signals regulating stomatal conductance and leaf growth in tomato (Lycopersicon esculentum) plants subjected to partial root-zone drying. J. Exp. Bot. 2004, 55, 2353–2363. [Google Scholar] [CrossRef] [PubMed]
- Damour, G.; Simonneau, T.; Cochard, H.; Urban, L. An overview of models of stomatal conductance at the leaf level: Models of stomatal conductance. Plant Cell Environ. 2010, 33, 1419–1438. [Google Scholar] [CrossRef] [PubMed]
- Burkhardt, J.; Kaiser, H.; Goldbach, H.; Kappen, L. Measurements of electrical leaf surface conductance reveal re-condensation of transpired water vapour on leaf surfaces. Plant Cell Environ. 1999, 22, 189–196. [Google Scholar] [CrossRef]
- Monteith, J.; Campbell, G.; Potter, E. Theory and performance of a dynamic diffusion porometer. Agric. Forest Meteorol. 1988, 44, 27–38. [Google Scholar] [CrossRef]
- Dbira, S.; Al Hassan, M.; Gramazio, P.; Ferchichi, A.; Vicente, O.; Prohens, J.; Boscaiu, M. Variable Levels of Tolerance to Water Stress (Drought) and Associated Biochemical Markers in Tunisian Barley Landraces. Molecules 2018, 23, 613. [Google Scholar] [CrossRef] [PubMed]
- Fathi, A.; Tari, D.B. Effect of Drought Stress and its Mechanism in Plants. Int. J. Life Sci. 2016, 10, 1–6. [Google Scholar] [CrossRef]
- Guo, P.; Baum, M.; Grando, S.; Ceccarelli, S.; Bai, G.; Li, R.; von Korff, M.; Varshney, R.K.; Graner, A.; Valkoun, J. Differentially expressed genes between drought-tolerant and drought-sensitive barley genotypes in response to drought stress during the reproductive stage. J. Exp. Bot. 2009, 60, 3531–3544. [Google Scholar] [CrossRef] [PubMed]
- López-Jurado, J.; Balao, F.; Mateos-Naranjo, E. Deciphering the ecophysiological traits involved during water stress acclimation and recovery of the threatened wild carnation, Dianthus inoxianus. Plant Physiol. Biochem. 2016, 109, 397–405. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-E.; Liu, W.-J.; Su, Y.-Q.; Cui, J.-M.; Zhang, Z.-W.; Yuan, M.; Zhang, H.-Y.; Yuan, S. Different response of photosystem II to short and long-term drought stress in Arabidopsis thaliana. Physiol. Plant. 2016, 158, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Klughammer, C.; Schreiber, U. Complementary PS II quantum yields calculated from simple fluorescence parameters measured by PAM fluorometry and the Saturation Pulse method. PAM Appl. Notes 2008, 1, 27–35. [Google Scholar]
- Ruhle, T.; Reiter, B.; Leister, D. Chlorophyll Fluorescence Video Imaging: A Versatile Tool for Identifying Factors Related to Photosynthesis. Front. Plant Sci. 2018, 9, 55. [Google Scholar] [CrossRef] [PubMed]
- Krause, G.H. Photoinhibition of photosynthesis. An evaluation of damaging and protective mechanisms. Physiol. Plant. 1988, 74, 566–574. [Google Scholar] [CrossRef]
- La Rocca, N.; Pupillo, P.; Puppi, G.; Rascio, N. Erythronium dens-canis L. (Liliaceae): An unusual case of change of leaf mottling. Plant Physiol. Biochem. 2014, 74, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Jefferies, R.A. Drought and chlorophyll fluorescence in field-grown potato (Solanum tuberosum). Physiol. Plant. 1994, 90, 93–97. [Google Scholar] [CrossRef]
- Wingler, A.; Marès, M.; Pourtau, N. Spatial Patterns and Metabolic Regulation of Photosynthetic Parameters during Leaf Senescence. New Phytol. 2004, 161, 781–789. [Google Scholar] [CrossRef]
- Kauser, R.; Athar, H.U.R.; Ashraf, M.; Roubina, K.; Habib, A. Chlorophyll fluorescence: A potential indicator for rapid assessment of water stress tolerance in Canola (Brassica napus L.). Pak. J. Bot. 2006, 38, 1501–1509. [Google Scholar]
- Shinozaki, K.; Yamaguchi-Shinozaki, K. Gene networks involved in drought stress response and tolerance. J. Exp. Bot. 2006, 58, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Daszkowska-Golec, A.; Szarejko, I. Open or Close the Gate—Stomata Action under the Control of Phytohormones in Drought Stress Conditions. Front. Plant Sci. 2013, 4. [Google Scholar] [CrossRef] [PubMed]
- Seki, M.; Umezawa, T.; Urano, K.; Shinozaki, K. Regulatory metabolic networks in drought stress responses. Curr. Opin. Plant Biol. 2007, 10, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, S.; Davies, W.J. Drought, ozone, ABA and ethylene: New insights from cell to plant to community. Plant Cell Environ. 2010, 33, 510–525. [Google Scholar] [CrossRef] [PubMed]
- Munemasa, S.; Oda, K.; Watanabe-Sugimoto, M.; Nakamura, Y.; Shimoishi, Y.; Murata, Y. The coronatine-insensitive 1 Mutation Reveals the Hormonal Signaling Interaction between Abscisic Acid and Methyl Jasmonate in Arabidopsis Guard Cells. Specific Impairment of Ion Channel Activation and Second Messenger Production. Plant Physiol. 2007, 143, 1398–1407. [Google Scholar] [CrossRef] [PubMed]
- Dalal, M.; Tayal, D.; Chinnusamy, V.; Bansal, K.C. Abiotic stress and ABA-inducible Group 4 LEA from Brassica napus plays a key role in salt and drought tolerance. J. Biotechnol. 2009, 139, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Desikan, R.; Last, K.; Harrett-Williams, R.; Tagliavia, C.; Harter, K.; Hooley, R.; Hancock, J.T.; Neill, S.J. Ethylene-induced stomatal closure in Arabidopsis occurs via AtrbohF-mediated hydrogen peroxide synthesis. Plant J. 2006, 47, 907–916. [Google Scholar] [CrossRef] [PubMed]
- Fraire-Velazquez, S.; Emmanuel, V. Abiotic Stress in Plants and Metabolic Responses. In Abiotic Stress—Plant Responses and Applications in Agriculture; InTech: London, UK, 2013; ISBN 978-953-51-1024-8. [Google Scholar]
- Krasensky, J.; Jonak, C. Drought, salt, and temperature stress-induced metabolic rearrangements and regulatory networks. J. Exp. Bot. 2012, 63, 1593–1608. [Google Scholar] [CrossRef] [PubMed]
- Valadez-Bustos, M.G.; Aguado-Santacruz, GA.; Tiessen-Favier, A.; Robledo-Paz, A.; Munoz-Orozco, A.; Rascon-Cruz, Q.; Santacruz-Varela, A. A reliable method for spectrophotometric determination of glycine betaine in cell suspension and other systems. Anal. Biochem. 2016, 498, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Alimohammadi, M.; Lahiani, M.H.; Khodakovskaya, M.V. Genetic reduction of inositol triphosphate increases tolerance of tomato plants to oxidative stress. Planta 2015, 242, 123–135. [Google Scholar] [CrossRef] [PubMed]
- Birkemeyer, C.; Osmolovskaya, N.; Kuchaeva, L.; Tarakhovskaya, E. Distribution of natural ingredients suggests a complex network of metabolic transport between source and sink tissues in the brown alga Fucus vesiculosus. Planta 2018. [Google Scholar] [CrossRef] [PubMed]
- Tarakhovskaya, E.; Lemesheva, V.; Bilova, T.; Birkemeyer, C. Early Embryogenesis of Brown Alga Fucus vesiculosus L. is Characterized by Significant Changes in Carbon and Energy Metabolism. Molecules 2017, 22, 1509. [Google Scholar] [CrossRef] [PubMed]
- Milkovska-Stamenova, S.; Schmidt, R.; Frolov, A.; Birkemeyer, C. GC-MS Method for the Quantitation of Carbohydrate Intermediates in Glycation Systems. J. Agric. Food Chem. 2015, 63, 5911–5919. [Google Scholar] [CrossRef] [PubMed]
- Ghasemzadeh, A.; Jaafar, H.Z.; Rahmat, A. Synthesis of phenolics and flavonoids in ginger (Zingiber officinale Roscoe) and their effects on photosynthesis rate. Int. J. Mol. Sci. 2010, 11, 4539–4555. [Google Scholar] [CrossRef] [PubMed]
- Ma, D.; Sun, D.; Wang, C.; Li, Y.; Guo, T. Expression of flavonoid biosynthesis genes and accumulation of flavonoid in wheat leaves in response to drought stress. Plant Physiol. Biochem. 2014, 80, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Bechtold, U.; Rabbani, N.; Mullineaux, P.M.; Thornalley, P.J. Quantitative measurement of specific biomarkers for protein oxidation, nitration and glycation in Arabidopsis leaves. Plant J. 2009, 59, 661–671. [Google Scholar] [CrossRef] [PubMed]
- Bilova, T.; Paudel, G.; Shilyaev, N.; Schmidt, R.; Brauch, D.; Tarakhovskaya, E.; Milrud, S.; Smolikova, G.; Tissier, A.; Vogt, T.; et al. Global proteomic analysis of advanced glycation end products in the Arabidopsis proteome provides evidence for age-related glycation hot spots. J. Biol. Chem. 2017, 292, 15758–15776. [Google Scholar] [CrossRef] [PubMed]
- Frolov, A.; Schmidt, R.; Spiller, S.; Greifenhagen, U.; Hoffmann, R. Arginine-derived advanced glycation end products generated in peptide-glucose mixtures during boiling. J. Agric. Food Chem. 2014, 62, 3626–3635. [Google Scholar] [CrossRef] [PubMed]
- Greifenhagen, U.; Nguyen, V.D.; Moschner, J.; Giannis, A.; Frolov, A.; Hoffmann, R. Sensitive and site-specific identification of carboxymethylated and carboxyethylated peptides in tryptic digests of proteins and human plasma. J. Proteome Res. 2015, 14, 768–777. [Google Scholar] [CrossRef] [PubMed]
- Greifenhagen, U.; Frolov, A.; Hoffmann, R. Oxidative degradation of N(ε)-fructosylamine-substituted peptides in heated aqueous systems. Amino Acids 2015, 47, 1065–1076. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, R.; Böhme, D.; Singer, D.; Frolov, A. Specific tandem mass spectrometric detection of AGE-modified arginine residues in peptides. J. Mass Spectrom. 2015, 50, 613–624. [Google Scholar] [CrossRef] [PubMed]
- Soboleva, A.; Vikhnina, M.; Grishina, T.; Frolov, A. Probing Protein Glycation by Chromatography and Mass Spectrometry: Analysis of Glycation Adducts. Int. J. Mol. Sci. 2017, 18, 2557. [Google Scholar] [CrossRef] [PubMed]
- Fedorova, M.; Frolov, A.; Hoffmann, R. Fragmentation behavior of Amadori-peptides obtained by non-enzymatic glycosylation of lysine residues with ADP-ribose in tandem mass spectrometry. J. Mass Spectrom. 2010, 45, 664–669. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, E.L.; Cervantes-Laurean, D.; Jacobson, M.K. Glycation of proteins by ADP-ribose. Mol. Cell. Biochem. 1994, 138, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, M.; Foolad, M.R. Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ. Exp. Bot. 2007, 59, 206–216. [Google Scholar] [CrossRef]
- Giri, J. Glycinebetaine and abiotic stress tolerance in plants. Plant Signal Behav 2011, 6, 1746–1751. [Google Scholar] [CrossRef] [PubMed]
- Templer, S.E.; Ammon, A.; Pscheidt, D.; Ciobotea, O.; Schuy, C.; McCollum, C.; Sonnewald, U.; Hanemann, A.; Förster, J.; Ordon, F.; et al. Metabolite profiling of barley flag leaves under drought and combined heat and drought stress reveals metabolic QTLs for metabolites associated with antioxidant defense. J. Exp. Bot. 2017, 68, 1697–1713. [Google Scholar] [CrossRef] [PubMed]
- Doczi, R.; Csanaki, C.Z.; Banfalvi, Z. Expression and promoter activity of the desiccation-specific Solanum tuberosum gene, StDS2. Plant Cell Environ. 2002, 25, 1197–1203. [Google Scholar] [CrossRef]
- Muvunyi, B.P.; Yan, Q.; Wu, F.; Min, X.; Yan, Z.Z.; Kanzana, G.; Wang, Y.; Zhang, J. Mining Late Embryogenesis Abundant (LEA) Family Genes in Cleistogenes songorica, a Xerophyte Perennial Desert Plant. Int. J. Mol. Sci. 2018, 19, 3430. [Google Scholar] [CrossRef] [PubMed]
- Park, C.J.; Seo, Y.S. Heat Shock Proteins: A Review of the Molecular Chaperones for Plant Immunity. Plant Pathol. J. 2015, 31, 323–333. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Vinocur, B.; Shoseyov, O.; Altman, A. Role of plant heat-shock proteins and molecular chaperones in the abiotic stress response. Trends Plant Sci. 2004, 9, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Dong, C.; Yang, S.; Li, X.; Sun, X.; Yang, Y. Physiological and Proteomic Adaptation of the Alpine Grass Stipa purpurea to a Drought Gradient. PLoS ONE 2015, 10, e0117475. [Google Scholar] [CrossRef] [PubMed]
- Xiang, J.; Chen, X.; Hu, W.; Xiang, Y.; Yan, M.; Wang, J. Overexpressing heat-shock protein OsHSP50.2 improves drought tolerance in rice. Plant Cell Rep. 2018, 37, 1585–1595. [Google Scholar] [CrossRef] [PubMed]
- Kovacs, D.; Kalmar, E.; Torok, Z.; Tompa, P. Chaperone Activity of ERD10 and ERD14, Two Disordered Stress-Related Plant Proteins. Plant Physiol. 2008, 147, 381–390. [Google Scholar] [CrossRef] [PubMed]
- Dure, L., III; Greenway, S.C.; Galau, G.A. Developmental biochemistry of cottonseed embryogenesis and germination: Changing messenger ribonucleic acid populations as shown by in vitro and in vivo protein synthesis. Biochemistry 1981, 20, 4162–4168. [Google Scholar] [CrossRef] [PubMed]
- Hatanaka, R.; Hagiwara-Komoda, Y.; Furuki, T.; Kanamori, Y.; Fujita, M.; Cornette, R.; Sakurai, M.; Okuda, T.; Kikawada, T. An abundant LEA protein in the anhydrobiotic midge, PvLEA4, acts as a molecular shield by limiting growth of aggregating protein particles. Insect Biochem. Mol. Biol. 2013, 43, 1055–1067. [Google Scholar] [CrossRef] [PubMed]
- Kovacs, D.; Agoston, B.; Tompa, P. Disordered plant LEA proteins as molecular chaperones. Plant Signal. Behav. 2008, 3, 710–713. [Google Scholar] [CrossRef] [PubMed]
- Magwanga, R.O.; Lu, P.; Kirungu, J.N.; Lu, H.; Wang, X.; Cai, X.; Zhou, Z.; Zhang, Z.; Salih, H.; Wang, K.; et al. Characterization of the late embryogenesis abundant (LEA) proteins family and their role in drought stress tolerance in upland cotton. BMC Genet. 2018, 19, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Cruz de Carvalho, M.H. Drought stress and reactive oxygen species: Production, scavenging and signaling. Plant Signal. Behav. 2008, 3, 156–165. [Google Scholar] [CrossRef] [PubMed]
- Møller, I.M.; Jensen, P.E.; Hansson, A. Oxidative modifications to cellular components in plants. Annu. Rev. Plant Biol. 2007, 58, 459–481. [Google Scholar] [CrossRef] [PubMed]
- Ma, F.; Chen, X.-B.; Sang, M.; Wang, P.; Zhang, J.-P.; Li, L.-B.; Kuang, T.-Y. Singlet oxygen formation and chlorophyll. Photosynth. Res. 2009, 100, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Møller, I.M. Plant mitochondria and oxidative stress: Electron transport, NADPH turnover, and metabolism of reactive oxygen species. Annu. Rev. Plant Biol. 2001, 52, 561–591. [Google Scholar] [CrossRef] [PubMed]
- Foyer, C.H. Reactive oxygen species, oxidative signaling and the regulation of photosynthesis. Environ. Exp. Bot. 2018, 154, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Pospisil, P. Molecular mechanisms of production and scavenging of reactive oxygen species by photosystem II. Biochim. Biophys. Acta 2012, 1817, 218–231. [Google Scholar] [CrossRef] [PubMed]
- Zulfugarov, I.S.; Tovuu, A.; Eu, Y.-J.; Dogsom, B.; Poudyal, R.S.; Nath, K.; Hall, M.; Banerjee, M.; Yoon, U.C.; Moon, Y.-H.; et al. Production of superoxide from Photosystem II in a rice (Oryza sativa L.) mutant lacking PsbS. BMC Plant Biol. 2014, 14, 242. [Google Scholar] [CrossRef] [PubMed]
- Velasquez, M.; Santander, I.P.; Contreras, D.R.; Yanez, J.; Zaror, C.; Salazar, R.A.; Pérez-Moya, M.; Mansilla, H.D. Oxidative degradation of sulfathiazole by Fenton and photo-Fenton reactions. J. Environ. Sci. Health Part A 2014, 49, 661–670. [Google Scholar] [CrossRef] [PubMed]
- Noctor, G.; Veljovic-Jovanovic, S.; Driscoll, S.; Novitskaya, L.; Foyer, C.H. Drought and oxidative load in the leaves of C3 plants: A predominant role for photorespiration? Ann Bot. 2002, 89, 841–850. [Google Scholar] [CrossRef] [PubMed]
- Atkin, O.K.; Macherel, D. The crucial role of plant mitochondria in orchestrating drought tolerance. Ann. Bot. 2009, 103, 581–597. [Google Scholar] [CrossRef] [PubMed]
- Moore, K.; Roberts, L.J. Measurement of Lipid Peroxidation. Free Radic. Res. 1998, 28, 659–671. [Google Scholar] [CrossRef] [PubMed]
- Del Rio, D.; Stewart, A.J.; Pellegrini, N. A review of recent studies on malondialdehyde as toxic molecule and biological marker of oxidative stress. Nutr. Metab. Cardiovasc. Dis. 2005, 15, 316–328. [Google Scholar] [CrossRef] [PubMed]
- Davey, M.W.; Stals, E.; Panis, B.; Keulemans, J.; Swennen, R.L. High-throughput determination of malondialdehyde in plant tissues. Anal. Biochem. 2005, 347, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Rhee, S.G.; Chang, T.-S.; Jeong, W.; Kang, D. Methods for detection and measurement of hydrogen peroxide inside and outside of cells. Mol. Cells 2010, 29, 539–549. [Google Scholar] [CrossRef] [PubMed]
- Foyer, C.H.; Noctor, G. Ascorbate and glutathione: The heart of the redox hub. Plant Physiol. 2011, 155, 2–18. [Google Scholar] [CrossRef] [PubMed]
- Smirnoff, N. BOTANICAL BRIEFING: The Function and Metabolism of Ascorbic Acid in Plants. Ann. Bot. 1996, 78, 661–669. [Google Scholar] [CrossRef]
- Noctor, G.; Foyer, C.H. Ascorbate and Glutathione: Keeping Active Oxygen under Control. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1998, 49, 249–279. [Google Scholar] [CrossRef] [PubMed]
- Meyer, A.J. The integration of glutathione homeostasis and redox signaling. J. Plant Physiol. 2008, 165, 1390–1403. [Google Scholar] [CrossRef] [PubMed]
- Kang, G.Z.; Li, G.Z.; Liu, G.Q.; Xu, W.; Peng, X.Q.; Wang, C.Y.; Zhu, Y.J.; Guo, T.C. Exogenous salicylic acid enhances wheat drought tolerance by influence on the expression of genes related to ascorbate-glutathione cycle. Biol. Plant. 2013, 57, 718–724. [Google Scholar] [CrossRef]
- Moon, H.-D.; Lee, M.-S.; Kim, S.-H.; Jeong, W.-J.; Choi, D.-W. Identification of a drought responsive gene encoding a nuclear protein involved in drought and freezing stress tolerance in Arabidopsis. Biol. Plant 2016, 60, 105–112. [Google Scholar] [CrossRef]
- Huang, T.; Jander, G. Abscisic acid-regulated protein degradation causes osmotic stress-induced accumulation of branched-chain amino acids in Arabidopsis thaliana. Planta 2017, 246, 737–747. [Google Scholar] [CrossRef] [PubMed]
- Soleymani, A. Light response of barley (Hordeum vulgare L.) and corn (Zea mays L.) as affected by drought stress, plant genotype and N fertilization. Biocatal. Agric. Biotechnol. 2017, 11, 1–8. [Google Scholar] [CrossRef]
- Niu, G.-L.; Gou, W.; Han, X.-L.; Qin, C.; Zhang, L.-X.; Abomohra, A.E.-F.; Ashraf, M. Cloning and Functional Analysis of Phosphoethanolamine Methyltransferase Promoter from Maize (Zea mays L.). Int. J. Mol. Sci. 2018, 19, 191. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Zhang, X.; Duan, H.; Lian, C.; Liu, C.; Yin, W.; Xia, X. Three stress-responsive NAC transcription factors from Populus euphratica differentially regulate salt and drought tolerance in transgenic plants. Physiol. Plant. 2017, 162, 73–97. [Google Scholar] [CrossRef] [PubMed]
- Bundig, C.; Vu, T.H.; Meise, P.; Seddig, S.; Schum, A.; Winkelmann, T. Variability in Osmotic Stress Tolerance of Starch Potato Genotypes (Solanum tuberosum L.) as Revealed by an In Vitro Screening: Role of Proline, Osmotic Adjustment and Drought Response in Pot Trials. J. Agron. Crop Sci. 2016, 203, 206–218. [Google Scholar] [CrossRef]
- Bothe, A.; Westermeier, P.; Wosnitza, A.; Willner, E.; Schum, A.; Dehmer, K.J.; Hartmann, S. Drought tolerance in perennial ryegrass (Lolium perenne L.) as assessed by two contrasting phenotyping systems. J. Agron. Crop Sci. 2018, 204, 375–389. [Google Scholar] [CrossRef]
- Landi, S.; Nurcato, R.; De Lillo, A.; Lentini, M.; Grillo, S.; Esposito, S. Glucose-6-phosphate dehydrogenase plays a central role in the response of tomato (Solanum lycopersicum) plants to short and long-term drought. Plant Physiol. Biochem. 2016, 105, 79–89. [Google Scholar] [CrossRef] [PubMed]
- Defez, R.; Andreozzi, A.; Dickinson, M.; Charlton, A.; Tadini, L.; Pesaresi, P.; Bianco, C. Improved Drought Stress Response in Alfalfa Plants Nodulated by an IAA Over-producing Rhizobium Strain. Front. Microbiol. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Vasques, A.R.; Pinto, G.; Dias, M.C.; Correia, C.M.; Moutinho-Pereira, J.M.; Vallejo, V.R.; Santos, C.; Keizer, J.J. Physiological response to drought in seedlings of Pistacia lentiscus (mastic tree). New For. 2016, 47, 119–130. [Google Scholar] [CrossRef]
- Tatli, O.; Ozdemir, B.S.; Doganay, G.D. Time-dependent leaf proteome alterations of Brachypodium distachyon in response to drought stress. Plant Mol. Biol. 2017, 94, 609–623. [Google Scholar] [CrossRef] [PubMed]
- Diaz-Vivancos, P.; Faize, L.; Nicolás, E.; Clemente-Moreno, M.J.; Bru-Martinez, R.; Burgos, L.; Hernández, J.A. Transformation of plum plants with a cytosolic ascorbate peroxidase transgene leads to enhanced water stress tolerance. Ann. Bot. 2016, 117, 1121–1131. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Yang, Y.; Yang, S.; Sun, X.; Yin, X.; Zhao, Y.; Yang, Y. Comparative proteomics analyses of intraspecific differences in the response of Stipa purpurea to drought. Plant Divers. 2016, 38, 101–117. [Google Scholar] [CrossRef] [PubMed]
- Rampazzo, P.; Marcos, F.; Cipriano, M.; Marchiori, P.; Freitas, S.; Machado, E.; Nascimento, L.; Brocchi, M.; Ribeiro, R. Rhizobacteria improve sugarcane growth and photosynthesis under well-watered conditions. Ann. Appl. Biol. 2018, 172, 309–320. [Google Scholar] [CrossRef]
- Tavakol, E.; Jakli, B.; Cakmak, I.; Dittert, K.; Karlovsky, P.; Pfohl, K.; Senbayram, M. Optimized potassium nutrition improves plant-water-relations of barley under PEG-induced osmotic stress. Plant Soil 2018, 430, 23–35. [Google Scholar] [CrossRef]
- Xiong, X.; Chang, L.; Khalid, M.; Zhang, J.; Huang, D. Alleviation of Drought Stress by Nitrogen Application in Brassica campestris ssp. Chinensis L. Agronomy 2018, 8, 66. [Google Scholar] [CrossRef]
- Pang, Y.; Chen, K.; Wang, X.; Xu, J.; Ali, J.; Li, Z. Recurrent selection breeding by dominant male sterility for multiple abiotic stresses tolerant rice cultivars. Euphytica 2017, 213, 268. [Google Scholar] [CrossRef]
- Redox imbalance contributed differently to membrane damage of cucumber leaves under water stress and Fusarium infection. Plant Sci. 2018, 274, 171–180. [CrossRef] [PubMed]
- Argyrokastritisa, I.G.; Papastylianoub, P.T.; Alexandrisa, S. Leaf Water Potential and Crop Water Stress Index variation for full and deficit irrigated cotton in Mediterranean conditions. Agric. Agric. Sci. Procedia 2015, 4, 463–470. [Google Scholar] [CrossRef]
- Pieczynski, M.; Marczewski, W.; Hennig, J.; Dolata, J.; Bielewicz, D.; Piontek, P.; Wyrzykowska, A.; Krusiewicz, D.; Strzelczyk-Zyta, D.; Konopka-Postupolska, D.; et al. Down-regulation of CBP80 gene expression as a strategy to engineer a drought-tolerant potato. Plant Biotechnol. J. 2013, 11, 459–469. [Google Scholar] [CrossRef] [PubMed]
- Socias, X.; Correia, M.J.; Chaves, M.; Medrano, H. The role of abscisic acid and water relations in drought responses of subterranean clover. J. Exp. Bot. 1997, 48, 1281–1288. [Google Scholar] [CrossRef]
- Bano, A.; Ullah, F.; Nosheen, A. Role of abscisic acid and drought stress on the activitiesof antioxidant enzymes in wheat. Plant Soil Environ. 2012, 58, 181–185. [Google Scholar] [CrossRef]
- Ullah, N.; Yüce, M.; Gökçe, Z.N.Ö.; Budak, H. Comparative metabolite profiling of drought stress in roots and leaves of seven Triticeae species. BMC Genom. 2017, 18, 969. [Google Scholar] [CrossRef] [PubMed]
- Weng, M.; Cui, L.; Liu, F.; Zhang, M.; Shan, L.; Yang, S.; Deng, X. Effects of drought stress on antioxidant enzymes in seedlings of different wheat genotypes. Pak. J. Bot. 2015, 47, 49–56. [Google Scholar]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Osmolovskaya, N.; Shumilina, J.; Kim, A.; Didio, A.; Grishina, T.; Bilova, T.; Keltsieva, O.A.; Zhukov, V.; Tikhonovich, I.; Tarakhovskaya, E.; et al. Methodology of Drought Stress Research: Experimental Setup and Physiological Characterization. Int. J. Mol. Sci. 2018, 19, 4089. https://doi.org/10.3390/ijms19124089
Osmolovskaya N, Shumilina J, Kim A, Didio A, Grishina T, Bilova T, Keltsieva OA, Zhukov V, Tikhonovich I, Tarakhovskaya E, et al. Methodology of Drought Stress Research: Experimental Setup and Physiological Characterization. International Journal of Molecular Sciences. 2018; 19(12):4089. https://doi.org/10.3390/ijms19124089
Chicago/Turabian StyleOsmolovskaya, Natalia, Julia Shumilina, Ahyoung Kim, Anna Didio, Tatiana Grishina, Tatiana Bilova, Olga A. Keltsieva, Vladimir Zhukov, Igor Tikhonovich, Elena Tarakhovskaya, and et al. 2018. "Methodology of Drought Stress Research: Experimental Setup and Physiological Characterization" International Journal of Molecular Sciences 19, no. 12: 4089. https://doi.org/10.3390/ijms19124089
APA StyleOsmolovskaya, N., Shumilina, J., Kim, A., Didio, A., Grishina, T., Bilova, T., Keltsieva, O. A., Zhukov, V., Tikhonovich, I., Tarakhovskaya, E., Frolov, A., & Wessjohann, L. A. (2018). Methodology of Drought Stress Research: Experimental Setup and Physiological Characterization. International Journal of Molecular Sciences, 19(12), 4089. https://doi.org/10.3390/ijms19124089







