Pharmacological Utilization of Bergamottin, Derived from Grapefruits, in Cancer Prevention and Therapy
Abstract
1. Introduction
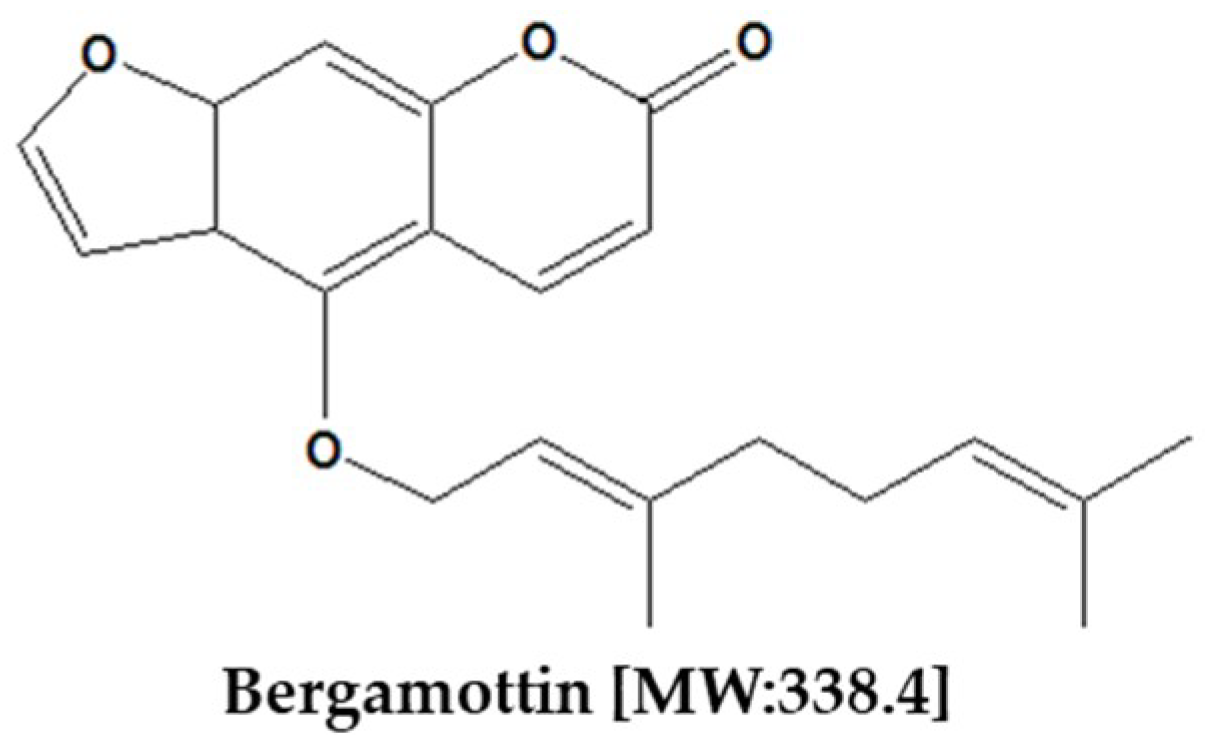
2. Chemical Properties of Bergamottin
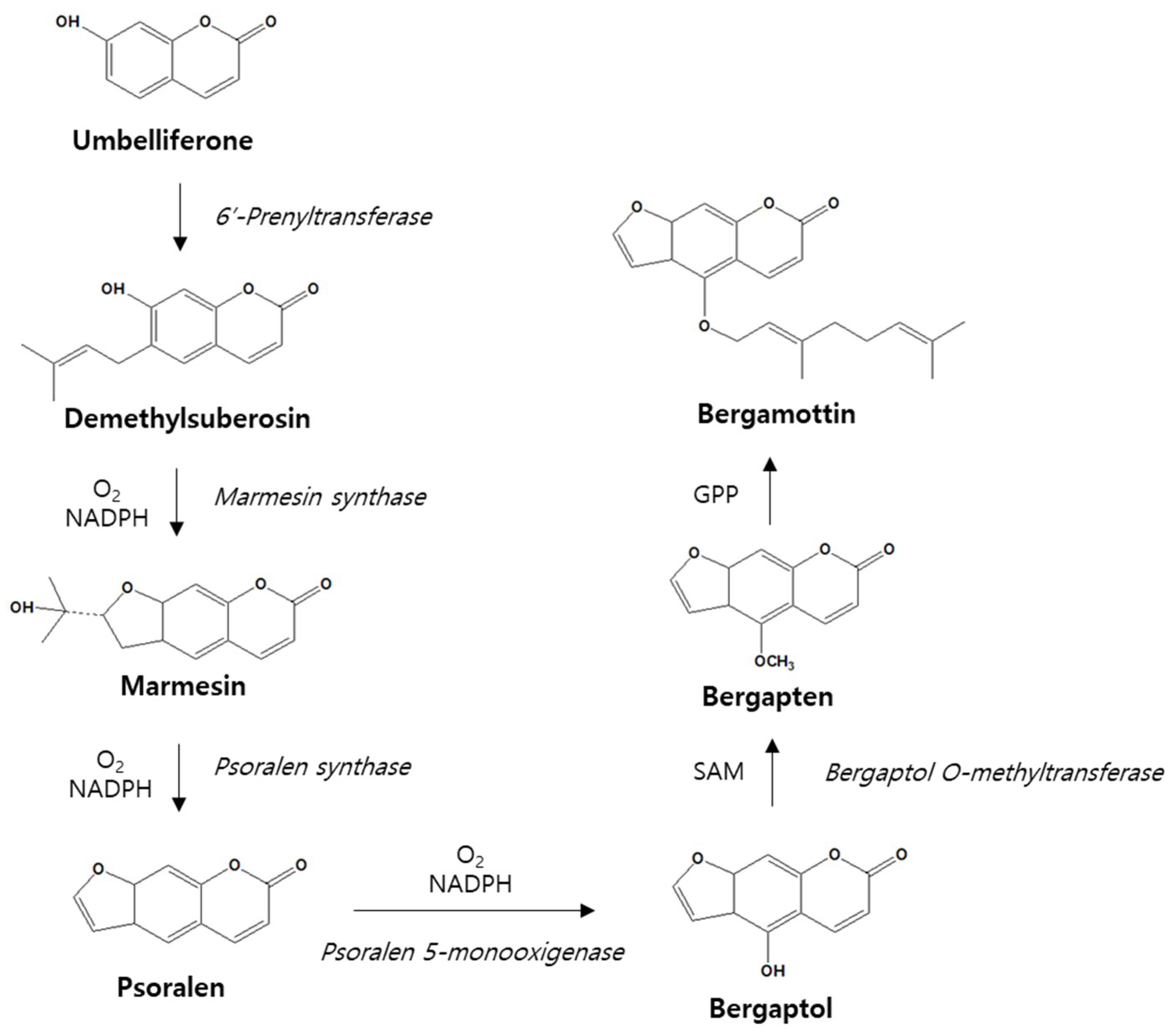
3. Metabolism of Bergamottin
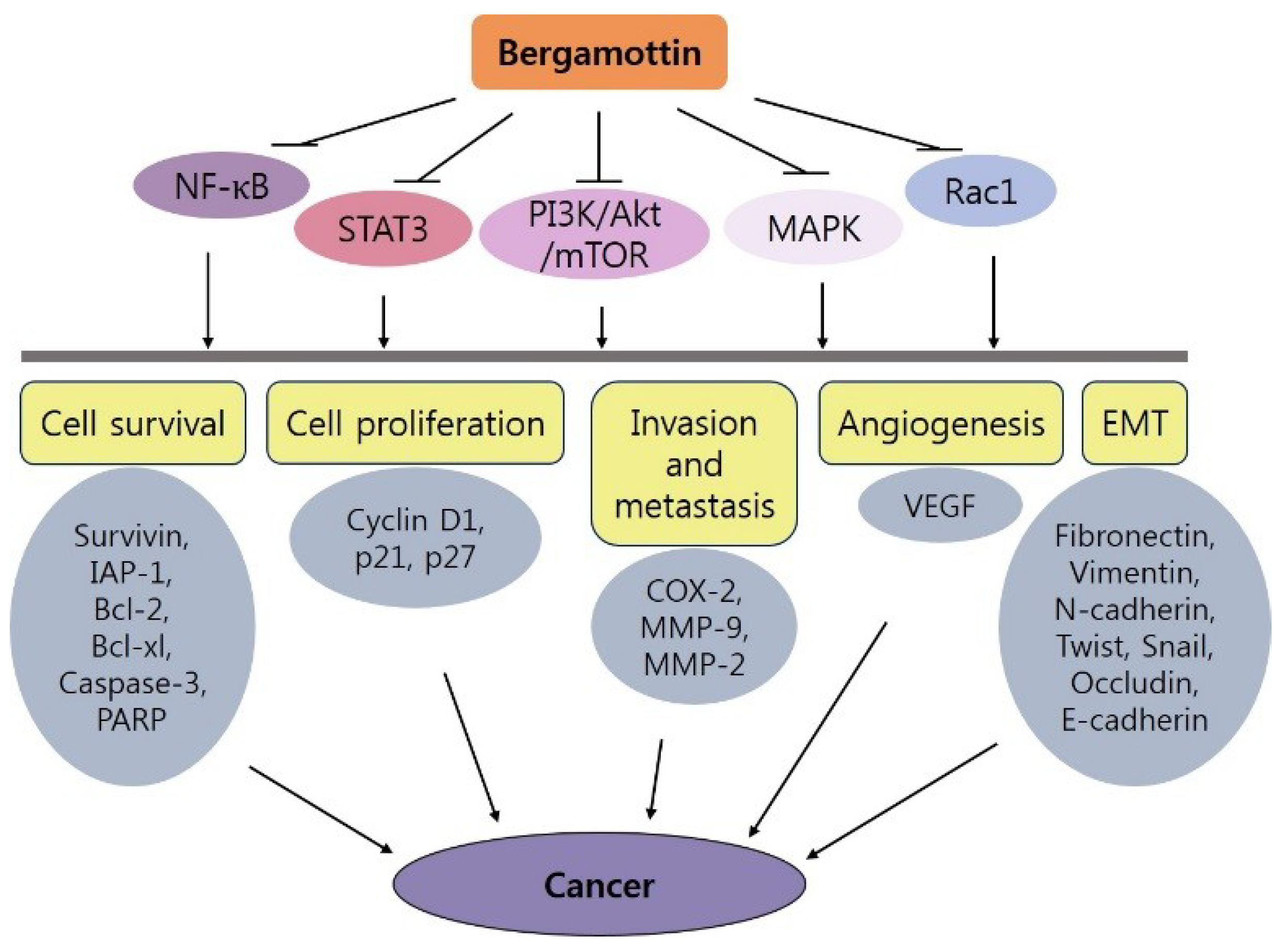
4. Bergamottin and Cancer
4.1. Multiple Myeloma
4.2. Leukemia
4.3. Skin Cancer
4.4. Lung Cancer
4.5. Fibrosarcoma
4.6. Other Cancers
5. Conclusions
Funding
Conflicts of Interest
References
- Deorukhkar, A.; Krishnan, S.; Sethi, G.; Aggarwal, B.B. Back to basics: How natural products can provide the basis for new therapeutics. Expert Opin. Investig. Drugs 2007, 16, 1753–1773. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.F.; Weng, C.J.; Sethi, G.; Hu, D.N. Natural bioactives and phytochemicals serve in cancer treatment and prevention. Euvid. Based Complement. Altern. Med. 2013, 2013, 698190. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.H.; Sethi, G.; Kuo, P.L. Novel medicines and strategies in cancer treatment and prevention. Biomed Res. Int. 2014, 2014, 474078. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, Y.S.; Yang, S.F.; Sethi, G.; Hu, D.N. Natural bioactives in cancer treatment and prevention. Biomed Res. Int. 2015, 2015, 182835. [Google Scholar] [CrossRef] [PubMed]
- Yarla, N.S.; Bishayee, A.; Sethi, G.; Reddanna, P.; Kalle, A.M.; Dhananjaya, B.L.; Dowluru, K.S.; Chintala, R.; Duddukuri, G.R. Targeting arachidonic acid pathway by natural products for cancer prevention and therapy. Semin. Cancer Boil. 2016, 40–41, 48–81. [Google Scholar] [CrossRef] [PubMed]
- Hasanpourghadi, M.; Looi, C.Y.; Pandurangan, A.K.; Sethi, G.; Wong, W.F.; Mustafa, M.R. Phytometabolites Targeting the Warburg Effect in Cancer Cells: A Mechanistic Review. Curr. Drug Targets 2017, 18, 1086–1094. [Google Scholar] [CrossRef] [PubMed]
- Shanmugam, M.K.; Warrier, S.; Kumar, A.P.; Sethi, G.; Arfuso, F. Potential Role of Natural Compounds as Anti-Angiogenic Agents in Cancer. Curr. Vasc. Pharmacol. 2017, 15, 503–519. [Google Scholar] [CrossRef] [PubMed]
- Shanmugam, M.K.; Kannaiyan, R.; Sethi, G. Targeting cell signaling and apoptotic pathways by dietary agents: Role in the prevention and treatment of cancer. Nutr. Cancer 2011, 63, 161–173. [Google Scholar] [CrossRef]
- Aggarwal, B.B.; Sethi, G.; Baladandayuthapani, V.; Krishnan, S.; Shishodia, S. Targeting cell signaling pathways for drug discovery: An old lock needs a new key. J. Cell. Biochem. 2007, 102, 580–592. [Google Scholar] [CrossRef]
- Jung, Y.Y.; Hwang, S.T.; Sethi, G.; Fan, L.; Arfuso, F.; Ahn, K.S. Potential Anti-Inflammatory and Anti-Cancer Properties of Farnesol. Molecules 2018, 23, 2827. [Google Scholar] [CrossRef]
- Merarchi, M.; Sethi, G.; Fan, L.; Mishra, S.; Arfuso, F.; Ahn, K.S. Molecular Targets Modulated by Fangchinoline in Tumor Cells and Preclinical Models. Molecules 2018, 23, 2538. [Google Scholar] [CrossRef] [PubMed]
- Sethi, G.; Shanmugam, M.K.; Warrier, S.; Merarchi, M.; Arfuso, F.; Kumar, A.P.; Bishayee, A. Pro-Apoptotic and Anti-Cancer Properties of Diosgenin: A Comprehensive and Critical Review. Nutrients 2018, 10, 645. [Google Scholar] [CrossRef] [PubMed]
- Ko, J.H.; Sethi, G.; Um, J.Y.; Shanmugam, M.K.; Arfuso, F.; Kumar, A.P.; Bishayee, A.; Ahn, K.S. The Role of Resveratrol in Cancer Therapy. Int. J. Mol. Sci. 2017, 18, 2589. [Google Scholar] [CrossRef] [PubMed]
- Tewari, D.; Nabavi, S.F.; Nabavi, S.M.; Sureda, A.; Farooqi, A.A.; Atanasov, A.G.; Vacca, R.A.; Sethi, G.; Bishayee, A. Targeting activator protein 1 signaling pathway by bioactive natural agents: Possible therapeutic strategy for cancer prevention and intervention. Pharmacol. Res. 2018, 128, 366–375. [Google Scholar] [CrossRef] [PubMed]
- Bishayee, A.; Sethi, G. Bioactive natural products in cancer prevention and therapy: Progress and promise. Semin. Cancer Boil. 2016, 40–41, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Shanmugam, M.K.; Lee, J.H.; Chai, E.Z.; Kanchi, M.M.; Kar, S.; Arfuso, F.; Dharmarajan, A.; Kumar, A.P.; Ramar, P.S.; Looi, C.Y.; et al. Cancer prevention and therapy through the modulation of transcription factors by bioactive natural compounds. Semin. Cancer Boil. 2016, 40–41, 35–47. [Google Scholar] [CrossRef]
- Wagstaff, D.J. Dietary exposure to furocoumarins. Regul. Toxicol. Pharmacol. RTP 1991, 14, 261–272. [Google Scholar] [CrossRef]
- Dolan, L.C.; Matulka, R.A.; Burdock, G.A. Naturally occurring food toxins. Toxins 2010, 2, 2289–2332. [Google Scholar] [CrossRef]
- Hung, W.L.; Suh, J.H.; Wang, Y. Chemistry and health effects of furanocoumarins in grapefruit. J. Food Drug Anal. 2017, 25, 71–83. [Google Scholar] [CrossRef]
- Bailey, D.G.; Malcolm, J.; Arnold, O.; Spence, J.D. Grapefruit juice-drug interactions. Br. J. Clin. Pharmacol. 1998, 46, 101–110. [Google Scholar] [CrossRef]
- He, K.; Iyer, K.R.; Hayes, R.N.; Sinz, M.W.; Woolf, T.F.; Hollenberg, P.F. Inactivation of cytochrome P450 3A4 by bergamottin, a component of grapefruit juice. Chem. Res. Toxicol. 1998, 11, 252–259. [Google Scholar] [CrossRef]
- Girennavar, B.; Poulose, S.M.; Jayaprakasha, G.K.; Bhat, N.G.; Patil, B.S. Furocoumarins from grapefruit juice and their effect on human CYP 3A4 and CYP 1B1 isoenzymes. Bioorg. Med. Chem. 2006, 14, 2606–2612. [Google Scholar] [CrossRef]
- Monroe, K.R.; Stanczyk, F.Z.; Besinque, K.H.; Pike, M.C. The effect of grapefruit intake on endogenous serum estrogen levels in postmenopausal women. Nutr. Cancer 2013, 65, 644–652. [Google Scholar] [CrossRef]
- Kantola, T.; Kivisto, K.T.; Neuvonen, P.J. Grapefruit juice greatly increases serum concentrations of lovastatin and lovastatin acid. Clin. Pharmacol. Ther. 1998, 63, 397–402. [Google Scholar] [CrossRef]
- Lilja, J.J.; Kivisto, K.T.; Neuvonen, P.J. Grapefruit juice-simvastatin interaction: Effect on serum concentrations of simvastatin, simvastatin acid, and HMG-CoA reductase inhibitors. Clin. Pharmacol. Ther. 1998, 64, 477–483. [Google Scholar] [CrossRef]
- Benton, R.E.; Honig, P.K.; Zamani, K.; Cantilena, L.R.; Woosley, R.L. Grapefruit juice alters terfenadine pharmacokinetics, resulting in prolongation of repolarization on the electrocardiogram. Clin. Pharmacol. Ther. 1996, 59, 383–388. [Google Scholar] [CrossRef]
- Lundahl, J.U.; Regardh, C.G.; Edgar, B.; Johnsson, G. The interaction effect of grapefruit juice is maximal after the first glass. Eur. J. Clin. Pharmacol. 1998, 54, 75–81. [Google Scholar] [CrossRef]
- Row, E.C.; Brown, S.A.; Stachulski, A.V.; Lennard, M.S. Design, synthesis and evaluation of furanocoumarin monomers as inhibitors of CYP3A4. Org. Biomol. Chem. 2006, 4, 1604–1610. [Google Scholar] [CrossRef]
- Christensen, H.; Asberg, A.; Holmboe, A.B.; Berg, K.J. Coadministration of grapefruit juice increases systemic exposure of diltiazem in healthy volunteers. Eur. J. Clin. Pharmacol. 2002, 58, 515–520. [Google Scholar] [CrossRef]
- Kane, G.C.; Lipsky, J.J. Drug-grapefruit juice interactions. Mayo Clin. Proc. 2000, 75, 933–942. [Google Scholar] [CrossRef]
- Cheng, K.L.; Nafziger, A.N.; Peloquin, C.A.; Amsden, G.W. Effect of grapefruit juice on clarithromycin pharmacokinetics. Antimicrob. Agents Chemother. 1998, 42, 927–929. [Google Scholar] [CrossRef]
- Ho, P.C.; Chalcroft, S.C.; Coville, P.F.; Wanwimolruk, S. Grapefruit juice has no effect on quinine pharmacokinetics. Eur. J. Clin. Pharmacol. 1999, 55, 393–398. [Google Scholar] [CrossRef]
- Hall, S.D.; Thummel, K.E.; Watkins, P.B.; Lown, K.S.; Benet, L.Z.; Paine, M.F.; Mayo, R.R.; Turgeon, D.K.; Bailey, D.G.; Fontana, R.J.; et al. Molecular and physical mechanisms of first-pass extraction. Drug Metab. Dispos. Boil. Fate Chem. 1999, 27, 161–166. [Google Scholar]
- Takanaga, H.; Ohnishi, A.; Matsuo, H.; Sawada, Y. Inhibition of vinblastine efflux mediated by P-glycoprotein by grapefruit juice components in caco-2 cells. Boil. Pharm. Bull. 1998, 21, 1062–1066. [Google Scholar] [CrossRef]
- Soldner, A.; Christians, U.; Susanto, M.; Wacher, V.J.; Silverman, J.A.; Benet, L.Z. Grapefruit juice activates P-glycoprotein-mediated drug transport. Pharm. Res. 1999, 16, 478–485. [Google Scholar] [CrossRef]
- Kobayashi, K.R.M.J.; Fleming, G.F.; Vogelzang, N.J.; Cooper, N.; Sun, B.L. A Phase I study of CYP3A4 modulation of oral (po) etoposide with ketoconazole (KCZ) in patients (pts) with advanced cancer (CA). Proc. Am. J. Clin. Oncol. 1996, 15, 471. [Google Scholar]
- Hanley, M.J.; Cancalon, P.; Widmer, W.W.; Greenblatt, D.J. The effect of grapefruit juice on drug disposition. Expert Opin. Drug Metab. Toxicol. 2011, 7, 267–286. [Google Scholar] [CrossRef]
- Collado-Borrell, R.; Escudero-Vilaplana, V.; Romero-Jimenez, R.; Iglesias-Peinado, I.; Herranz-Alonso, A.; Sanjurjo-Saez, M. Oral antineoplastic agent interactions with medicinal plants and food: An issue to take into account. J. Cancer Res. Clin. Oncol. 2016, 142, 2319–2330. [Google Scholar] [CrossRef]
- Reif, S.; Nicolson, M.C.; Bisset, D.; Reid, M.; Kloft, C.; Jaehde, U.; McLeod, H.L. Effect of grapefruit juice intake on etoposide bioavailability. Eur. J. Clin. Pharmacol. 2002, 58, 491–494. [Google Scholar] [CrossRef]
- Yin, O.Q.; Gallagher, N.; Li, A.; Zhou, W.; Harrell, R.; Schran, H. Effect of grapefruit juice on the pharmacokinetics of nilotinib in healthy participants. J. Clin. Pharmacol. 2010, 50, 188–194. [Google Scholar] [CrossRef]
- Van Erp, N.P.; Baker, S.D.; Zandvliet, A.S.; Ploeger, B.A.; den Hollander, M.; Chen, Z.; den Hartigh, J.; Konig-Quartel, J.M.; Guchelaar, H.J.; Gelderblom, H. Marginal increase of sunitinib exposure by grapefruit juice. Cancer Chemother. Pharmacol. 2011, 67, 695–703. [Google Scholar] [CrossRef]
- Cohen, E.E.; Wu, K.; Hartford, C.; Kocherginsky, M.; Eaton, K.N.; Zha, Y.; Nallari, A.; Maitland, M.L.; Fox-Kay, K.; Moshier, K.; et al. Phase I studies of sirolimus alone or in combination with pharmacokinetic modulators in advanced cancer patients. Clin. Cancer Res. 2012, 18, 4785–4793. [Google Scholar] [CrossRef]
- Schubert, W.; Cullberg, G.; Edgar, B.; Hedner, T. Inhibition of 17 beta-estradiol metabolism by grapefruit juice in ovariectomized women. Maturitas 1994, 20, 155–163. [Google Scholar] [CrossRef]
- Weber, A.; Jager, R.; Borner, A.; Klinger, G.; Vollanth, R.; Matthey, K.; Balogh, A. Can grapefruit juice influence ethinylestradiol bioavailability? Contraception 1996, 53, 41–47. [Google Scholar] [CrossRef]
- Monroe, K.R.; Murphy, S.P.; Kolonel, L.N.; Pike, M.C. Prospective study of grapefruit intake and risk of breast cancer in postmenopausal women: The Multiethnic Cohort Study. Br. J. Cancer 2007, 97, 440–445. [Google Scholar] [CrossRef]
- Bisagni, E. Synthesis of psoralens and analogues. J. Photochem. Photobiol. Bbiol. 1992, 14, 23–46. [Google Scholar] [CrossRef]
- Voznesensky, A.I.; Schenkman, J.B. The cytochrome P450 2B4-NADPH cytochrome P450 reductase electron transfer complex is not formed by charge-pairing. J. Boil. Chem. 1992, 267, 14669–14676. [Google Scholar]
- Kent, U.M.; Lin, H.L.; Noon, K.R.; Harris, D.L.; Hollenberg, P.F. Metabolism of bergamottin by cytochromes P450 2B6 and 3A5. J. Pharmacol. Exp. Ther. 2006, 318, 992–1005. [Google Scholar] [CrossRef]
- Melough, M.M.; Chun, O.K. Dietary furocoumarins and skin cancer: A review of current biological evidence. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Boil. Res. Assoc. 2018, 122, 163–171. [Google Scholar] [CrossRef]
- Goosen, T.C.; Cillie, D.; Bailey, D.G.; Yu, C.; He, K.; Hollenberg, P.F.; Woster, P.M.; Cohen, L.; Williams, J.A.; Rheeders, M.; et al. Bergamottin contribution to the grapefruit juice-felodipine interaction and disposition in humans. Clin. Pharmacol. Ther. 2004, 76, 607–617. [Google Scholar] [CrossRef]
- Lee, S.G.; Kim, K.; Vance, T.M.; Perkins, C.; Provatas, A.; Wu, S.; Qureshi, A.; Cho, E.; Chun, O.K. Development of a comprehensive analytical method for furanocoumarins in grapefruit and their metabolites in plasma and urine using UPLC-MS/MS: A preliminary study. Int. J. Food Sci. Nutr. 2016, 67, 881–887. [Google Scholar] [CrossRef]
- Messer, A.; Nieborowski, A.; Strasser, C.; Lohr, C.; Schrenk, D. Major furocoumarins in grapefruit juice I: Levels and urinary metabolite(s). Food Chem. Toxicol. Int. J. Publ. Br. Ind. Boil. Res. Assoc. 2011, 49, 3224–3231. [Google Scholar] [CrossRef]
- Lin, H.L.; Kent, U.M.; Hollenberg, P.F. The grapefruit juice effect is not limited to cytochrome P450 (P450) 3A4: Evidence for bergamottin-dependent inactivation, heme destruction, and covalent binding to protein in P450s 2B6 and 3A5. J. Pharmacol. Exp. Ther. 2005, 313, 154–164. [Google Scholar] [CrossRef]
- Abel, G. Chromosome damage induced in human lymphocytes by 5-methoxypsoralen and 8-methoxypsoralen plus UV-A. Mutat. Res. 1987, 190, 63–68. [Google Scholar] [CrossRef]
- Papadopoulo, D.; Moustacchi, E. Mutagenic effects photoinduced in normal human lymphoblasts by a monofunctional pyridopsoralen in comparison to 8-methoxypsoralen. Mutat. Res. 1990, 245, 259–266. [Google Scholar] [CrossRef]
- Lohr, C.; Raquet, N.; Schrenk, D. Application of the concept of relative photomutagenic potencies to selected furocoumarins in V79 cells. Toxicol. Vitr. Int. J. Publ. Assoc. Bibra 2010, 24, 558–566. [Google Scholar] [CrossRef]
- Kim, S.M.; Lee, J.H.; Sethi, G.; Kim, C.; Baek, S.H.; Nam, D.; Chung, W.S.; Kim, S.H.; Shim, B.S.; Ahn, K.S. Bergamottin, a natural furanocoumarin obtained from grapefruit juice induces chemosensitization and apoptosis through the inhibition of STAT3 signaling pathway in tumor cells. Cancer Lett. 2014, 354, 153–163. [Google Scholar] [CrossRef]
- Kawaii, S.; Tomono, Y.; Katase, E.; Ogawa, K.; Yano, M. Isolation of furocoumarins from bergamot fruits as HL-60 differentiation-inducing compounds. J. Agric. Food Chem. 1999, 47, 4073–4078. [Google Scholar] [CrossRef]
- Liu, Y.; Ren, C.; Cao, Y.; Wang, Y.; Duan, W.; Xie, L.; Sun, C.; Li, X. Characterization and Purification of Bergamottin from Citrus grandis (L.) Osbeck cv. Yongjiazaoxiangyou and Its Antiproliferative Activity and Effect on Glucose Consumption in HepG2 cells. Molecules 2017, 22, 1227. [Google Scholar] [CrossRef]
- Kim, S.M.; Lee, E.J.; Lee, J.H.; Yang, W.M.; Nam, D.; Lee, J.H.; Lee, S.G.; Um, J.Y.; Shim, B.S.; Ahn, K.S. Simvastatin in combination with bergamottin potentiates TNF-induced apoptosis through modulation of NF-kappaB signalling pathway in human chronic myelogenous leukaemia. Pharm. Boil. 2016, 54, 2050–2060. [Google Scholar] [CrossRef]
- Cai, Y.; Baer-Dubowska, W.; Ashwood-Smith, M.; DiGiovanni, J. Inhibitory effects of naturally occurring coumarins on the metabolic activation of benzo[a]pyrene and 7,12-dimethylbenz[a]anthracene in cultured mouse keratinocytes. Carcinogenesis 1997, 18, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.J.; Wu, H.B.; Zhao, Y.Q.; Chen, L.J.; Zou, H.Z. Bergamottin isolated from Citrus bergamia exerts in vitro and in vivo antitumor activity in lung adenocarcinoma through the induction of apoptosis, cell cycle arrest, mitochondrial membrane potential loss and inhibition of cell migration and invasion. Oncol. Rep. 2016, 36, 324–332. [Google Scholar] [CrossRef] [PubMed]
- Ko, J.H.; Nam, D.; Um, J.Y.; Jung, S.H.; Sethi, G.; Ahn, K.S. Bergamottin Suppresses Metastasis of Lung Cancer Cells through Abrogation of Diverse Oncogenic Signaling Cascades and Epithelial-to-Mesenchymal Transition. Molecules 2018, 23, 1601. [Google Scholar] [CrossRef] [PubMed]
- Hwang, Y.P.; Yun, H.J.; Choi, J.H.; Kang, K.W.; Jeong, H.G. Suppression of phorbol-12-myristate-13-acetate-induced tumor cell invasion by bergamottin via the inhibition of protein kinase Cdelta/p38 mitogen-activated protein kinase and JNK/nuclear factor-kappaB-dependent matrix metalloproteinase-9 expression. Mol. Nutr. Food Res. 2010, 54, 977–990. [Google Scholar] [CrossRef] [PubMed]
- Sekiguchi, H.; Washida, K.; Murakami, A. Suppressive Effects of Selected Food Phytochemicals on CD74 Expression in NCI-N87 Gastric Carcinoma Cells. J. Clin. Biochem. Nutr. 2008, 43, 109–117. [Google Scholar] [CrossRef]
- Kleiner, H.E.; Reed, M.J.; DiGiovanni, J. Naturally occurring coumarins inhibit human cytochromes P450 and block benzo[a]pyrene and 7,12-dimethylbenz[a]anthracene DNA adduct formation in MCF-7 cells. Chem. Res. Toxicol. 2003, 16, 415–422. [Google Scholar] [CrossRef]
- Navarra, M.; Ferlazzo, N.; Cirmi, S.; Trapasso, E.; Bramanti, P.; Lombardo, G.E.; Minciullo, P.L.; Calapai, G.; Gangemi, S. Effects of bergamot essential oil and its extractive fractions on SH-SY5Y human neuroblastoma cell growth. J. Pharm. Pharmacol. 2015, 67, 1042–1053. [Google Scholar] [CrossRef]
- Luo, W.; Song, Z.; Sun, H.; Liang, J.; Zhao, S. Bergamottin, a natural furanocoumarin abundantly present in grapefruit juice, suppresses the invasiveness of human glioma cells via inactivation of Rac1 signaling. Oncol. Lett. 2018, 15, 3259–3266. [Google Scholar] [CrossRef]
- Cai, Y.; Kleiner, H.; Johnston, D.; Dubowski, A.; Bostic, S.; Ivie, W.; DiGiovanni, J. Effect of naturally occurring coumarins on the formation of epidermal DNA adducts and skin tumors induced by benzo[a]pyrene and 7,12-dimethylbenz[a]anthracene in SENCAR mice. Carcinogenesis 1997, 18, 1521–1527. [Google Scholar] [CrossRef]
- Arora, L.; Kumar, A.P.; Arfuso, F.; Chng, W.J.; Sethi, G. The Role of Signal Transducer and Activator of Transcription 3 (STAT3) and Its Targeted Inhibition in Hematological Malignancies. Cancers 2018, 10, 327. [Google Scholar] [CrossRef]
- Lee, J.H.; Kim, C.; Lee, S.G.; Sethi, G.; Ahn, K.S. Ophiopogonin D, a Steroidal Glycoside Abrogates STAT3 Signaling Cascade and Exhibits Anti-Cancer Activity by Causing GSH/GSSG Imbalance in Lung Carcinoma. Cancers 2018, 10, 427. [Google Scholar] [CrossRef] [PubMed]
- Wong, A.L.A.; Hirpara, J.L.; Pervaiz, S.; Eu, J.Q.; Sethi, G.; Goh, B.C. Do STAT3 inhibitors have potential in the future for cancer therapy? Expert Opin. Investig. Drugs 2017, 26, 883–887. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Kim, C.; Baek, S.H.; Ko, J.H.; Lee, S.G.; Yang, W.M.; Um, J.Y.; Sethi, G.; Ahn, K.S. Capsazepine inhibits JAK/STAT3 signaling, tumor growth, and cell survival in prostate cancer. Oncotarget 2017, 8, 17700–17711. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Ahn, K.S.; Kim, C.; Shanmugam, M.K.; Siveen, K.S.; Arfuso, F.; Samym, R.P.; Deivasigamanim, A.; Lim, L.H.; Wang, L.; et al. Nimbolide-Induced Oxidative Stress Abrogates STAT3 Signaling Cascade and Inhibits Tumor Growth in Transgenic Adenocarcinoma of Mouse Prostate Model. Antioxid. Redox Signal. 2016, 24, 575–589. [Google Scholar] [CrossRef] [PubMed]
- Chai, E.Z.; Shanmugam, M.K.; Arfuso, F.; Dharmarajan, A.; Wang, C.; Kumar, A.P.; Samy, R.P.; Lim, L.H.; Wang, L.; Goh, B.C.; et al. Targeting transcription factor STAT3 for cancer prevention and therapy. Pharmacol. Ther. 2016, 162, 86–97. [Google Scholar] [CrossRef] [PubMed]
- Siveen, K.S.; Sikka, S.; Surana, R.; Dai, X.; Zhang, J.; Kumar, A.P.; Tan, B.K.; Sethi, G.; Bishayee, A. Targeting the STAT3 signaling pathway in cancer: Role of synthetic and natural inhibitors. Biochim. Biophys. Acta 2014, 1845, 136–154. [Google Scholar] [CrossRef] [PubMed]
- Subramaniam, A.; Shanmugam, M.K.; Ong, T.H.; Li, F.; Perumal, E.; Chen, L.; Vali, S.; Abbasi, T.; Kapoor, S.; Ahn, K.S.; et al. Emodin inhibits growth and induces apoptosis in an orthotopic hepatocellular carcinoma model by blocking activation of STAT3. Br. J. Pharmacol. 2013, 170, 807–821. [Google Scholar] [CrossRef]
- Subramaniam, A.; Shanmugam, M.K.; Perumal, E.; Li, F.; Nachiyappan, A.; Dai, X.; Swamy, S.N.; Ahn, K.S.; Kumar, A.P.; Tan, B.K.; et al. Potential role of signal transducer and activator of transcription (STAT)3 signaling pathway in inflammation, survival, proliferation and invasion of hepatocellular carcinoma. Biochim. Biophys. Acta 2013, 1835, 46–60. [Google Scholar] [CrossRef]
- Jung, Y.Y.; Lee, J.H.; Nam, D.; Narula, A.S.; Namjoshi, O.A.; Blough, B.E.; Um, J.Y.; Sethi, G.; Ahn, K.S. Anti-myeloma Effects of Icariin Are Mediated Through the Attenuation of JAK/STAT3-Dependent Signaling Cascade. Front. Pharmacol. 2018, 9, 531. [Google Scholar] [CrossRef]
- Kim, C.; Lee, S.G.; Yang, W.M.; Arfuso, F.; Um, J.Y.; Kumar, A.P.; Bian, J.; Sethi, G.; Ahn, K.S. Formononetin-induced oxidative stress abrogates the activation of STAT3/5 signaling axis and suppresses the tumor growth in multiple myeloma preclinical model. Cancer Lett. 2018, 431, 123–141. [Google Scholar] [CrossRef]
- Li, F.; Zhang, J.; Arfuso, F.; Chinnathambi, A.; Zayed, M.E.; Alharbi, S.A.; Kumar, A.P.; Ahn, K.S.; Sethi, G. NF-kappaB in cancer therapy. Arch. Toxicol. 2015, 89, 711–731. [Google Scholar] [CrossRef]
- Manu, K.A.; Shanmugam, M.K.; Ramachandran, L.; Li, F.; Fong, C.W.; Kumar, A.P.; Tan, P.; Sethi, G. First evidence that gamma-tocotrienol inhibits the growth of human gastric cancer and chemosensitizes it to capecitabine in a xenograft mouse model through the modulation of NF-kappaB pathway. Clin. Cancer Res. 2012, 18, 2220–2229. [Google Scholar] [CrossRef] [PubMed]
- Sethi, G.; Shanmugam, M.K.; Ramachandran, L.; Kumar, A.P.; Tergaonkar, V. Multifaceted link between cancer and inflammation. Biosci. Rep. 2012, 32, 1–15. [Google Scholar] [CrossRef]
- Li, F.; Sethi, G. Targeting transcription factor NF-kappaB to overcome chemoresistance and radioresistance in cancer therapy. Biochim. Biophys. Acta 2010, 1805, 167–180. [Google Scholar]
- Sethi, G.; Tergaonkar, V. Potential pharmacological control of the NF-kappaB pathway. Trends Pharmacol. Sci. 2009, 30, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Ahn, K.S.; Sethi, G.; Aggarwal, B.B. Nuclear factor-kappa B: From clone to clinic. Curr. Mol. Med. 2007, 7, 619–637. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Ahn, K.S.; Shanmugam, M.K.; Wang, H.; Shen, H.; Arfuso, F.; Chinnathambi, A.; Alharbi, S.A.; Chang, Y.; Sethi, G.; et al. Oleuropein induces apoptosis via abrogating NF-kappaB activation cascade in estrogen receptor-negative breast cancer cells. J. Cell. Biochem. 2018. [Google Scholar] [CrossRef]
- Puar, Y.R.; Shanmugam, M.K.; Fan, L.; Arfuso, F.; Sethi, G.; Tergaonkar, V. Evidence for the Involvement of the Master Transcription Factor NF-kappaB in Cancer Initiation and Progression. Biomedicines 2018, 6, 82. [Google Scholar] [CrossRef]
- Chai, E.Z.; Siveen, K.S.; Shanmugam, M.K.; Arfuso, F.; Sethi, G. Analysis of the intricate relationship between chronic inflammation and cancer. Biochem. J. 2015, 468, 1–15. [Google Scholar] [CrossRef]
- Shanmugam, M.K.; Ahn, K.S.; Lee, J.H.; Kannaiyan, R.; Mustafa, N.; Manu, K.A.; Siveen, K.S.; Sethi, G.; Chng, W.J.; Kumar, A.P. Celastrol Attenuates the Invasion and Migration and Augments the Anticancer Effects of Bortezomib in a Xenograft Mouse Model of Multiple Myeloma. Front. Pharmacol. 2018, 9, 365. [Google Scholar] [CrossRef]
- Manu, K.A.; Shanmugam, M.K.; Ramachandran, L.; Li, F.; Siveen, K.S.; Chinnathambi, A.; Zayed, M.E.; Alharbi, S.A.; Arfuso, F.; Kumar, A.P.; et al. Isorhamnetin augments the anti-tumor effect of capeciatbine through the negative regulation of NF-kappaB signaling cascade in gastric cancer. Cancer Lett. 2015, 363, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Shanmugam, M.K.; Siveen, K.S.; Wang, F.; Ong, T.H.; Loo, S.Y.; Swamy, M.M.; Mandal, S.; Kumar, A.P.; Goh, B.C.; et al. Garcinol sensitizes human head and neck carcinoma to cisplatin in a xenograft mouse model despite downregulation of proliferative biomarkers. Oncotarget 2015, 6, 5147–5163. [Google Scholar] [CrossRef] [PubMed]
- DiGiovanni, J. Multistage carcinogenesis in mouse skin. Pharmacol. Ther. 1992, 54, 63–128. [Google Scholar] [CrossRef]
- Kleiner, H.E.; Vulimiri, S.V.; Reed, M.J.; Uberecken, A.; DiGiovanni, J. Role of cytochrome P450 1a1 and 1b1 in the metabolic activation of 7,12-dimethylbenz[a]anthracene and the effects of naturally occurring furanocoumarins on skin tumor initiation. Chem. Res. Toxicol. 2002, 15, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Thiery, J.P. Epithelial-mesenchymal transitions in tumour progression. Nat. Rev. Cancer 2002, 2, 442–454. [Google Scholar] [CrossRef] [PubMed]
- Lo, H.C.; Zhang, X.H. EMT in Metastasis: Finding the Right Balance. Dev. Cell 2018, 45, 663–665. [Google Scholar] [CrossRef] [PubMed]
- Buiatti, E.; Palli, D.; Decarli, A.; Amadori, D.; Avellini, C.; Bianchi, S.; Biserni, R.; Cipriani, F.; Cocco, P.; Giacosa, A.; et al. A case-control study of gastric cancer and diet in Italy. Int. J. Cancer 1989, 44, 611–616. [Google Scholar] [CrossRef] [PubMed]
- Ramon, J.M.; Serra, L.; Cerdo, C.; Oromi, J. Dietary factors and gastric cancer risk. A case-control study in Spain. Cancer 1993, 71, 1731–1735. [Google Scholar] [CrossRef]
- Gonzalez, C.A.; Pera, G.; Agudo, A.; Bueno-de-Mesquita, H.B.; Ceroti, M.; Boeing, H.; Schulz, M.; Del Giudice, G.; Plebani, M.; Carneiro, F.; et al. Fruit and vegetable intake and the risk of stomach and oesophagus adenocarcinoma in the European Prospective Investigation into Cancer and Nutrition (EPIC-EURGAST). Int. J. Cancer 2006, 118, 2559–2566. [Google Scholar] [CrossRef] [PubMed]
| Antineoplastic Drug | Metabolism | Interaction | Ref. |
|---|---|---|---|
| Etoposide | Metabolized by CYP3A4 | Decrease etoposide exposure (area under the concentration time curve (AUC) 26.2% ↓) | [39] |
| Nilotinib | Metabolized by CYP3A4 | Increase nilotinib exposure (Cmax 60% ↑, AUC 29% ↑) No increase in adverse events | [40] |
| Sunitinib | Metabolized by CYP3A4 | Increase sunitinib exposure (Cmax 10.9% ↑, AUC 11% ↑) No increase in toxicity | [41] |
| Type of Cancers | Cell Lines | Dose | Biological Effect | Ref. |
| In Vitro | ||||
| Multiple myeloma | U266 | 50 and 100 µM for 24 h | Inhibits cell proliferation, induces apoptosis, and inhibits JAK/STAT3 activation | [57] |
| Leukemia | HL-60 | 40 µM for 4 days 6.25, 12.5, 25, and 50 µg/mL for 3 days | Inhibits cell proliferation | [58] [59] |
| KBM-5 | 50 µM for 12 h in combination with 10 µM simvastatin | Combination with simvastatin exhibits synergistic effects of TNF-induced cytotoxicity and apoptosis | [60] | |
| Skin cancer | Mouse epidermal keratinocytes | 2 nM | Inhibits DNA adduct formation induced by B[α]P) and DMBA | [61] |
| Lung cancer | A549 | 10, 25, and 50 µM for 48 h | Induces apoptosis, cell cycle arrest, and loss of mitochondrial membrane potential Inhibits cell migration and invasion | [62] |
| A549 | 100 µM for 24 h | Suppresses EMT, TGF-β-induced EMT, and cell invasive potential | [63] | |
| Fibrosarcoma | HT-1080 | 5, 25, and 50 µM for 24 h | Reduces PMA-induced MMP-9 and MMP-2 activation Inhibits cell invasion and migration | [64] |
| Liver cancer | HepG2 | 6.25, 12.5, 25, and 50 µg/mL for 3 days | Abrogates cell proliferation | [59] |
| Gastric cancer | BGC-823 | 6.25, 12.5, 25, and 50 µg/mL for 3 days | Inhibits cell proliferation | [59] |
| NCI-N87 | 4, 20, and 100 µM for 48 h | Attenuates cell proliferation | [65] | |
| Breast cancer | MDA-MB-231 | 100 µM for 6 h 100 µM for 75 h 100 µM for 24 h | Inhibits STAT3 activation Suppresses cell proliferation Attenuates cell invasion | [57] |
| MCF-7 | 40 µM for 24 h | Inhibits DNA adduct formation induced by B[α]P and DMBA | [66] | |
| Prostate cancer | DU145 | 100 µM for 6 h 100 µM for 75 h 100 µM for 24 h | Suppresses STAT3 activation Inhibits cell proliferation Inhibits cell invasion | [57] |
| Neuroblastoma | SH-SY5Y | BEO (0.01, 0.02, and 0.03%) for 24 h | Suppresses cell proliferation | [67] |
| Glioma | U87, U251 | 2 and 10 µM for 48 h | Exhibits anti-invasive activity through the inactivation of Rac1 and the downregulation of MMP-9 | [68] |
| In Vivo | ||||
| Type of Cancers | Animal Models | Dose | Biological Effect | Ref. |
| Skin cancer | SENCAR mice (B[α]P) | 400 nmol; 5 min pretreatment | Suppresses B[α]P-induced tumor initiation | [69] |
| Lung cancer | BALB/c nude mice xenograft model (A549) | 25, 50, and 100 mg/kg; daily; 18 days | Inhibits lung cancer growth | [62] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ko, J.-H.; Arfuso, F.; Sethi, G.; Ahn, K.S. Pharmacological Utilization of Bergamottin, Derived from Grapefruits, in Cancer Prevention and Therapy. Int. J. Mol. Sci. 2018, 19, 4048. https://doi.org/10.3390/ijms19124048
Ko J-H, Arfuso F, Sethi G, Ahn KS. Pharmacological Utilization of Bergamottin, Derived from Grapefruits, in Cancer Prevention and Therapy. International Journal of Molecular Sciences. 2018; 19(12):4048. https://doi.org/10.3390/ijms19124048
Chicago/Turabian StyleKo, Jeong-Hyeon, Frank Arfuso, Gautam Sethi, and Kwang Seok Ahn. 2018. "Pharmacological Utilization of Bergamottin, Derived from Grapefruits, in Cancer Prevention and Therapy" International Journal of Molecular Sciences 19, no. 12: 4048. https://doi.org/10.3390/ijms19124048
APA StyleKo, J.-H., Arfuso, F., Sethi, G., & Ahn, K. S. (2018). Pharmacological Utilization of Bergamottin, Derived from Grapefruits, in Cancer Prevention and Therapy. International Journal of Molecular Sciences, 19(12), 4048. https://doi.org/10.3390/ijms19124048






