NAD Metabolome Analysis in Human Cells Using 1H NMR Spectroscopy
Abstract
1. Introduction
2. Results and Discussion
2.1. Optimization of NMR-Based Quantification of the NAD Metabolome
2.2. Application of 1H NMR Spectroscopy to NAD Metabolome Analysis in Human Cell Extracts
3. Materials and Methods
3.1. Materials
3.2. Large Scale Synthesis of NAR
3.3. Cell Culture
3.4. Platelets and Erythrocytes Isolation from the Human Whole Blood
3.5. NMR Sample Preparation
3.6. NMR Analysis and Quantification
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gupte, R.; Liu, Z.; Kraus, W.L. PARPs and ADP-ribosylation: Recent advances linking molecular functions to biological outcomes. Genes Dev. 2017, 31, 101–126. [Google Scholar] [CrossRef] [PubMed]
- Cohen, M.S.; Chang, P. Insights into the biogenesis, function, and regulation of ADP-ribosylation. Nat. Chem. Biol. 2018, 14, 236–243. [Google Scholar] [CrossRef]
- Houtkooper, R.H.; Pirinen, E.; Auwerx, J. Sirtuins as regulators of metabolism and healthspan. Nat. Rev. Mol. Cell Biol. 2012, 13, 225–238. [Google Scholar] [CrossRef] [PubMed]
- Nikiforov, A.; Kulikova, V.; Ziegler, M. The human NAD metabolome: Functions, metabolism and compartmentalization. Crit. Rev. Biochem. Mol. Biol. 2015, 50, 284–297. [Google Scholar] [CrossRef]
- Malavasi, F.; Deaglio, S.; Funaro, A.; Ferrero, E.; Horenstein, A.L.; Ortolan, E.; Vaisitti, T.; Aydin, S. Evolution and function of the ADP ribosyl cyclase/CD38 gene family in physiology and pathology. Physiol. Rev. 2008, 88, 841–886. [Google Scholar] [CrossRef] [PubMed]
- Fliegert, R.; Gasser, A.; Guse, A.H. Regulation of calcium signalling by adenine-based second messengers. Biochem. Soc. Trans. 2007, 35, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Dölle, C.; Skoge, R.H.; Vanlinden, M.R.; Ziegler, M. NAD biosynthesis in humans--enzymes, metabolites and therapeutic aspects. Curr. Top. Med. Chem. 2013, 13, 2907–2917. [Google Scholar] [CrossRef] [PubMed]
- Yoshino, J.; Baur, J.A.; Imai, S.I. NAD(+) Intermediates: The Biology and Therapeutic Potential of NMN and NR. Cell Metab. 2018, 27, 513–528. [Google Scholar] [CrossRef]
- Rajman, L.; Chwalek, K.; Sinclair, D.A. Therapeutic Potential of NAD-Boosting Molecules: The In Vivo Evidence. Cell Metab. 2018, 27, 529–547. [Google Scholar] [CrossRef] [PubMed]
- Evans, C.; Bogan, K.L.; Song, P.; Burant, C.F.; Kennedy, R.T.; Brenner, C. NAD+ metabolite levels as a function of vitamins and calorie restriction: Evidence for different mechanisms of longevity. BMC Chem. Boil. 2010, 10, 2. [Google Scholar] [CrossRef]
- Trammell, S.A.; Brenner, C. Targeted, LCMS-based Metabolomics for Quantitative Measurement of NAD(+) Metabolites. Comput. Struct. Biotechnol. J. 2013, 4, e201301012. [Google Scholar] [CrossRef] [PubMed]
- Bustamante, S.E.; Jayasena, T.; Richani, D.; Gilchrist, R.; Wu, L.E.; Sinclair, D.A.; Sachdev, P.S.; Braidy, N. Quantifying the cellular NAD+ metabolome using a tandem liquid chromatography mass spectrometry approach. Metabolomics 2018, 14. [Google Scholar] [CrossRef]
- Carpi, F.M.; Cortese, M.; Orsomando, G.; Polzonetti, V.; Vincenzetti, S.; Moreschini, B.; Coleman, M.; Magni, G.; Pucciarelli, S. Simultaneous quantification of nicotinamide mononucleotide and related pyridine compounds in mouse tissues by UHPLC–MS/MS. Sep. Sci. Plus 2018, 1, 22–30. [Google Scholar] [CrossRef]
- Kulikova, V.; Shabalin, K.; Nerinovski, K.; Dolle, C.; Niere, M.; Yakimov, A.; Redpath, P.; Khodorkovskiy, M.; Migaud, M.E.; Ziegler, M.; et al. Generation, Release, and Uptake of the NAD Precursor Nicotinic Acid Riboside by Human Cells. J. Biol. Chem. 2015, 290, 27124–27137. [Google Scholar] [CrossRef] [PubMed]
- Nagana Gowda, G.A.; Abell, L.; Lee, C.F.; Tian, R.; Raftery, D. Simultaneous Analysis of Major Coenzymes of Cellular Redox Reactions and Energy Using ex Vivo (1)H NMR Spectroscopy. Anal. Chem. 2016, 88, 4817–4824. [Google Scholar] [CrossRef] [PubMed]
- Nagana Gowda, G.A.; Raftery, D. Whole Blood Metabolomics by (1)H NMR Spectroscopy Provides a New Opportunity To Evaluate Coenzymes and Antioxidants. Anal. Chem. 2017, 89, 4620–4627. [Google Scholar] [CrossRef] [PubMed]
- Johnson, G.S. Metabolism of NAD and N1-methylnicotinamide in growing and growth-arrested cells. Eur. J. Biochem. 1980, 112, 635–641. [Google Scholar] [CrossRef] [PubMed]
- Formentini, L.; Moroni, F.; Chiarugi, A. Detection and pharmacological modulation of nicotinamide mononucleotide (NMN) in vitro and in vivo. Biochem. Pharmacol. 2009, 77, 1612–1620. [Google Scholar] [CrossRef]
- Niere, M.; Kernstock, S.; Koch-Nolte, F.; Ziegler, M. Functional localization of two poly(ADP-ribose)-degrading enzymes to the mitochondrial matrix. Mol. Cell. Biol. 2008, 28, 814–824. [Google Scholar] [CrossRef]
- Grozio, A.; Sociali, G.; Sturla, L.; Caffa, I.; Soncini, D.; Salis, A.; Raffaelli, N.; De Flora, A.; Nencioni, A.; Bruzzone, S. CD73 protein as a source of extracellular precursors for sustained NAD+ biosynthesis in FK866-treated tumor cells. J. Biol. Chem. 2013, 288, 25938–25949. [Google Scholar] [CrossRef]
- Belenky, P.; Christensen, K.C.; Gazzaniga, F.; Pletnev, A.A.; Brenner, C. Nicotinamide riboside and nicotinic acid riboside salvage in fungi and mammals. Quantitative basis for Urh1 and purine nucleoside phosphorylase function in NAD+ metabolism. J. Biol. Chem. 2009, 284, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Wielgus-Kutrowska, B.; Kulikowska, E.; Wierzchowski, J.; Bzowska, A.; Shugar, D. Nicotinamide riboside, an unusual, non-typical, substrate of purified purine-nucleoside phosphorylases. Eur. J. Biochem. 1997, 243, 408–414. [Google Scholar] [CrossRef] [PubMed]
- Ulanovskaya, O.A.; Zuhl, A.M.; Cravatt, B.F. NNMT promotes epigenetic remodeling in cancer by creating a metabolic methylation sink. Nat. Chem. Biol. 2013, 9, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Kraus, D.; Yang, Q.; Kong, D.; Banks, A.S.; Zhang, L.; Rodgers, J.T.; Pirinen, E.; Pulinilkunnil, T.C.; Gong, F.; Wang, Y.C.; et al. Nicotinamide N-methyltransferase knockdown protects against diet-induced obesity. Nature 2014, 508, 258–262. [Google Scholar] [CrossRef] [PubMed]
- Tomida, M.; Ohtake, H.; Yokota, T.; Kobayashi, Y.; Kurosumi, M. Stat3 up-regulates expression of nicotinamide N-methyltransferase in human cancer cells. J. Cancer Res. Clin. Oncol. 2008, 134, 551–559. [Google Scholar] [CrossRef]
- Uhlen, M.; Fagerberg, L.; Hallstrom, B.M.; Lindskog, C.; Oksvold, P.; Mardinoglu, A.; Sivertsson, A.; Kampf, C.; Sjostedt, E.; Asplund, A.; et al. Proteomics. Tissue-based map of the human proteome. Science 2015, 347, 1260419. [Google Scholar] [CrossRef] [PubMed]
- Fouquerel, E.; Goellner, E.M.; Yu, Z.; Gagne, J.P.; Barbi de Moura, M.; Feinstein, T.; Wheeler, D.; Redpath, P.; Li, J.; Romero, G.; et al. ARTD1/PARP1 negatively regulates glycolysis by inhibiting hexokinase 1 independent of NAD+ depletion. Cell. Rep. 2014, 8, 1819–1831. [Google Scholar] [CrossRef] [PubMed]
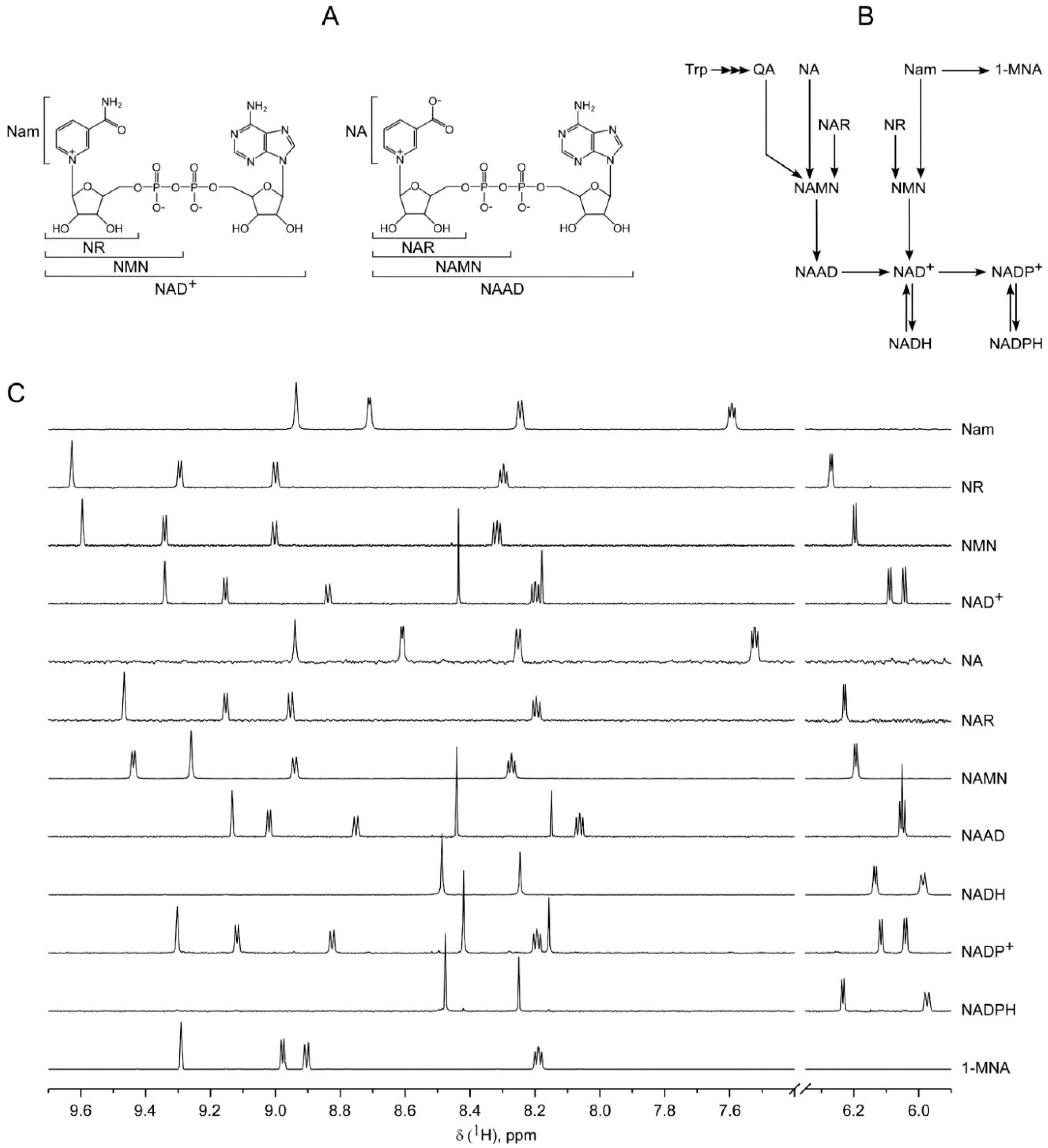
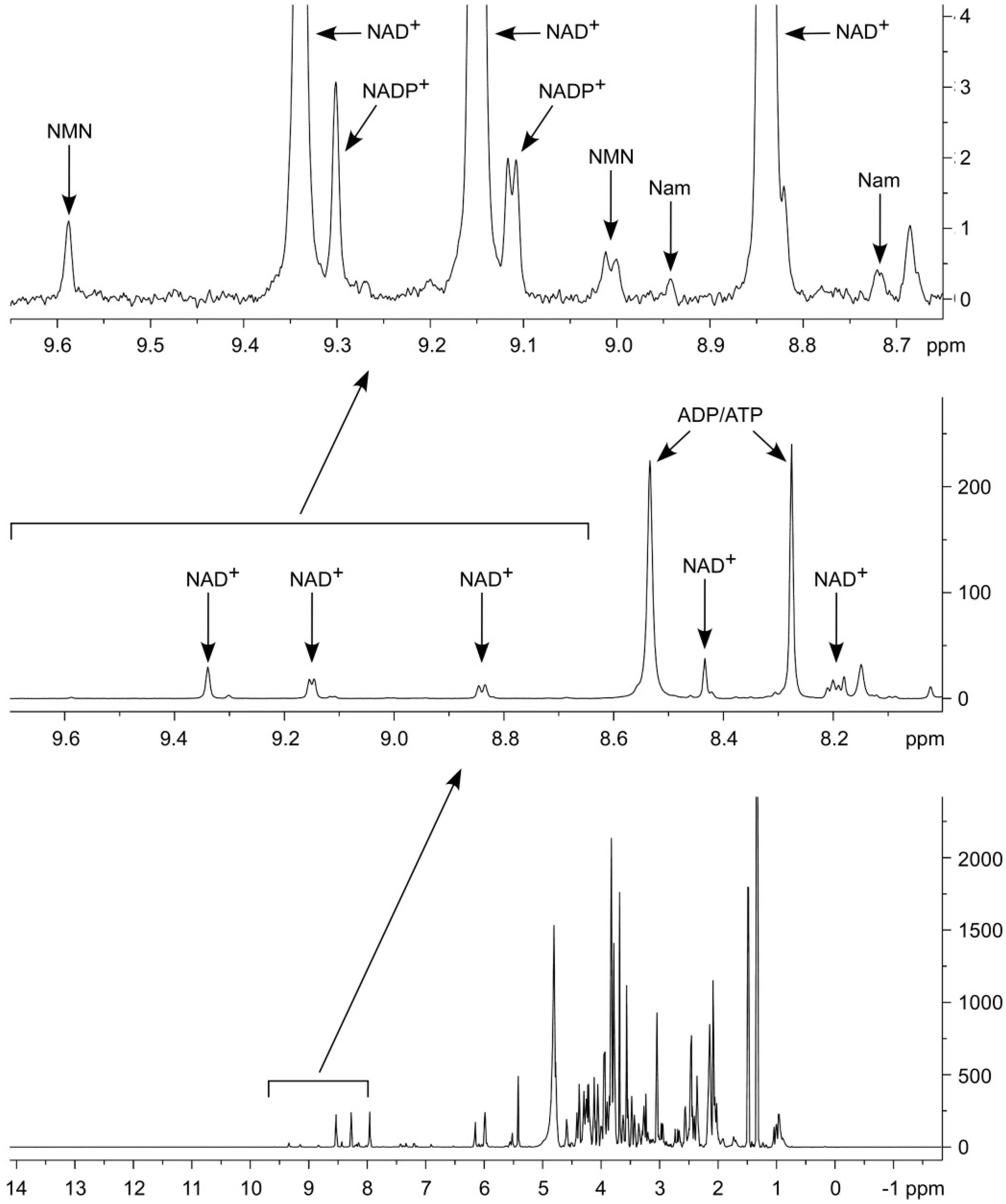

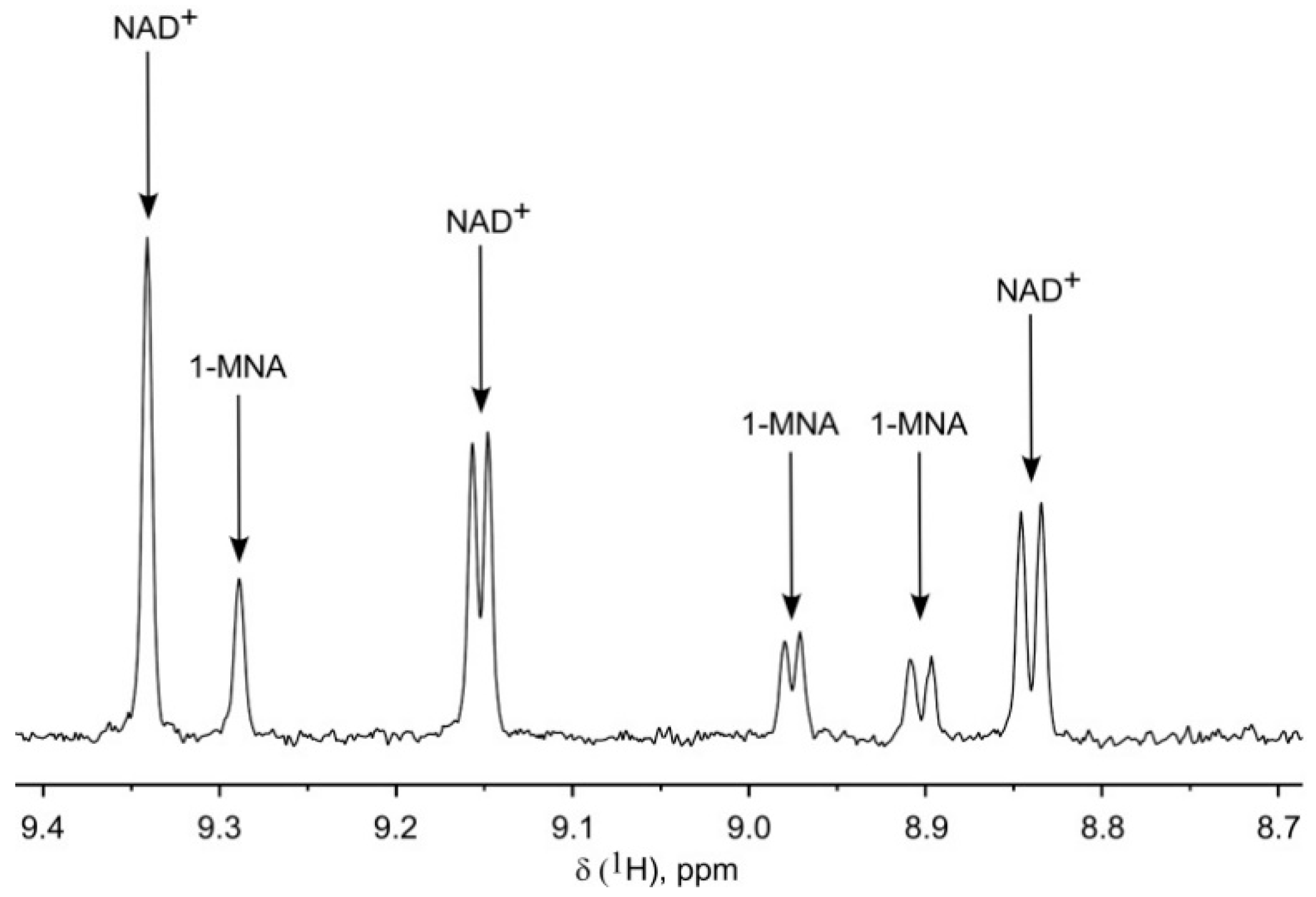
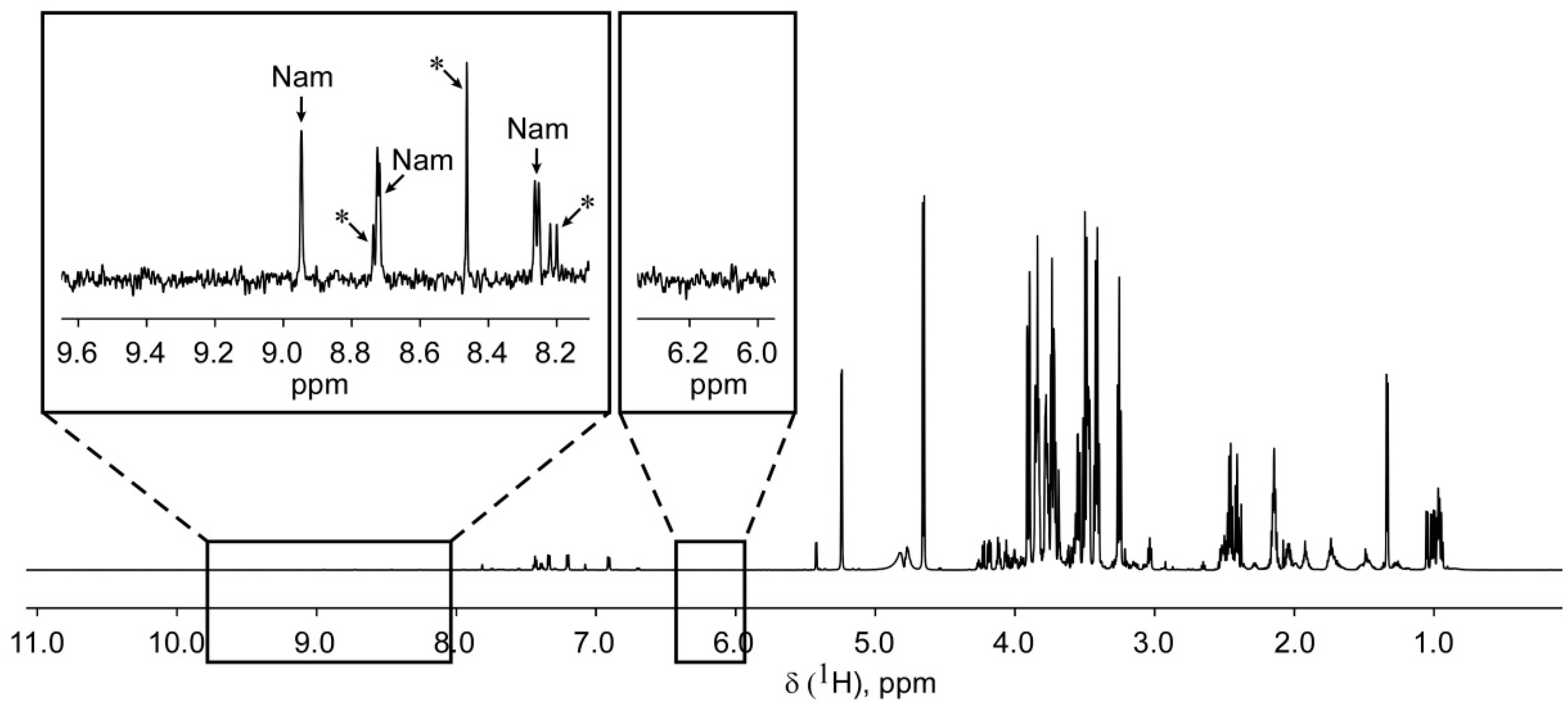
| Metabolite | δ, ppm | M, Class | J’s, Hz | T1, s |
|---|---|---|---|---|
| Nam | 8.94 | s | / | 13.73 |
| 8.71 | d | 5.01 | 7.97 | |
| 8.25 | d | 7.97 | 8.52 | |
| 7.60 | dd | 4.93, 7.99 | 6.29 | |
| NR | 9.64 | s | / | 3.02 |
| 9.31 | d | 6.18 | 2.21 | |
| 9.02 | d | 8.03 | 5.29 | |
| 8.31 | t | 7.11, 7.11 | 2.22 | |
| 6.29 | d | 4.39 | 1.89 | |
| NMN | 9.60 | s | / | 3.16 |
| 9.34 | d | 6.26 | 2.56 | |
| 9.00 | d | 8.03 | 4.68 | |
| 8.32 | t | 7.11, 7.11 | 2.97 | |
| 6.20 | d | 5.83 | 2.27 | |
| NAD+ | 9.34 | s | / | 2.32 |
| 9.15 | d | 6.22 | 1.76 | |
| 8.84 | d | 8.04 | 3.99 | |
| 8.43 | s | / | 2.84 | |
| 8.20 | t | 7.19, 7.19 | 2.29 | |
| 8.18 | s | / | 9.73 | |
| 6.09 | d | 5.44 | 1.8 | |
| 6.04 | d | 5.94 | 4.09 | |
| NA | 8.94 | d | 2.14 | 12.23 |
| 8.61 | d | 4.99 | 9.41 | |
| 8.25 | d | 7.9 | 10 | |
| 7.52 | dd | 4.90, 7.91 | 6.44 | |
| NAR | 9.47 | s | / | 2.93 |
| 9.16 | d | 6.2 | 2.06 | |
| 8.95 | d | 7.91 | 4.92 | |
| 8.20 | t | 7.03, 7.03 | 2.38 | |
| 6.23 | d | 4.68 | 1.89 | |
| NAMN | 9.44 | d | 6.23 | 1.92 |
| 9.26 | s | / | 2.09 | |
| 8.94 | d | 7.88 | 4.11 | |
| 8.27 | t | 7.07, 7.07 | 2.25 | |
| 6.19 | d | 5.37 | 1.75 | |
| NAAD | 9.13 | s | / | 2.1 |
| 9.02 | d | 6.22 | 1.97 | |
| 8.75 | d | 7.87 | 3.96 | |
| 8.44 | s | / | 2.67 | |
| 8.15 | s | / | 8.99 | |
| 8.06 | t | 7.04, 7.04 | 2.18 | |
| 6.05 | t | 5.36, 5.36 | 2.92 | |
| NADH | 8.49 | s | / | 2.61 |
| 8.25 | s | / | 7.79 | |
| 6.95 | s | / | 1.79 | |
| 6.13 | d | 5.51 | 3.78 | |
| 5.99 | d | 8.04 | 1.50 | |
| NADP+ | 9.30 | s | / | 2.4 |
| 9.12 | d | 6.29 | 1.82 | |
| 8.83 | d | 8.09 | 4.09 | |
| 8.42 | s | / | 2.92 | |
| 8.19 | m | / | 2.33 | |
| 8.16 | s | / | 9.6 | |
| 6.12 | d | 5.1 | 3.95 | |
| 6.04 | d | 5.62 | 1.75 | |
| NADPH | 8.48 | s | / | 2.84 |
| 8.25 | s | / | 8.05 | |
| 6.94 | d | 1.53 | 1.92 | |
| 6.23 | d | 4.81 | 3.8 | |
| 5.97 | d | 8.53 | 1.75 | |
| 1-MNA | 9.29 | s | / | 6.98 |
| 8.98 | d | 6.08 | 5.8 | |
| 8.90 | d | 8.20 | 7.17 | |
| 8.19 | dd | 6.16, 8.15 | 5.0 |
| Metabolite | Untreated | Treated with NR and NAR |
|---|---|---|
| NAD+ | 4.19 ± 0.15 | 8.69 ± 0.69 |
| NMN | 0.24 ± 0.04 | 0.27 ± 0.06 |
| NR | nd * | nd |
| Nam | 0.15 ± 0.03 | 0.54 ± 0.12 |
| NADP+ | 0.42 ± 0.05 | 0.55 ± 0.03 |
| NAAD | nd | 0.62 ± 0.04 |
| NAMN | nd | 0.42 ± 0.05 |
| NAR | nd | 0.09 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shabalin, K.; Nerinovski, K.; Yakimov, A.; Kulikova, V.; Svetlova, M.; Solovjeva, L.; Khodorkovskiy, M.; Gambaryan, S.; Cunningham, R.; Migaud, M.E.; et al. NAD Metabolome Analysis in Human Cells Using 1H NMR Spectroscopy. Int. J. Mol. Sci. 2018, 19, 3906. https://doi.org/10.3390/ijms19123906
Shabalin K, Nerinovski K, Yakimov A, Kulikova V, Svetlova M, Solovjeva L, Khodorkovskiy M, Gambaryan S, Cunningham R, Migaud ME, et al. NAD Metabolome Analysis in Human Cells Using 1H NMR Spectroscopy. International Journal of Molecular Sciences. 2018; 19(12):3906. https://doi.org/10.3390/ijms19123906
Chicago/Turabian StyleShabalin, Konstantin, Kirill Nerinovski, Alexander Yakimov, Veronika Kulikova, Maria Svetlova, Ljudmila Solovjeva, Mikhail Khodorkovskiy, Stepan Gambaryan, Richard Cunningham, Marie E. Migaud, and et al. 2018. "NAD Metabolome Analysis in Human Cells Using 1H NMR Spectroscopy" International Journal of Molecular Sciences 19, no. 12: 3906. https://doi.org/10.3390/ijms19123906
APA StyleShabalin, K., Nerinovski, K., Yakimov, A., Kulikova, V., Svetlova, M., Solovjeva, L., Khodorkovskiy, M., Gambaryan, S., Cunningham, R., Migaud, M. E., Ziegler, M., & Nikiforov, A. (2018). NAD Metabolome Analysis in Human Cells Using 1H NMR Spectroscopy. International Journal of Molecular Sciences, 19(12), 3906. https://doi.org/10.3390/ijms19123906







