Potential Causes of Titanium Particle and Ion Release in Implant Dentistry: A Systematic Review
Abstract
1. Introduction
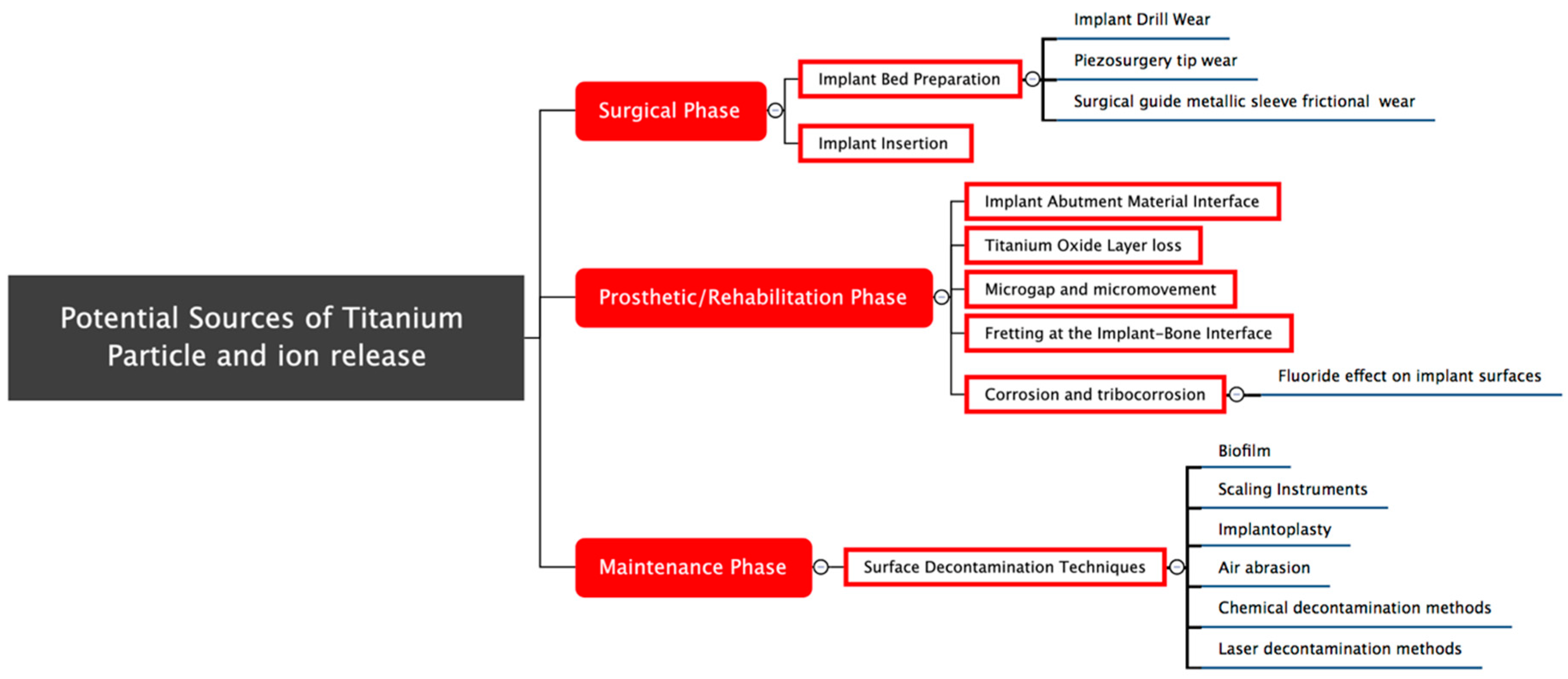
2. Results
2.1. Potential Causes of Titanium Particle Release during the Surgical Phase
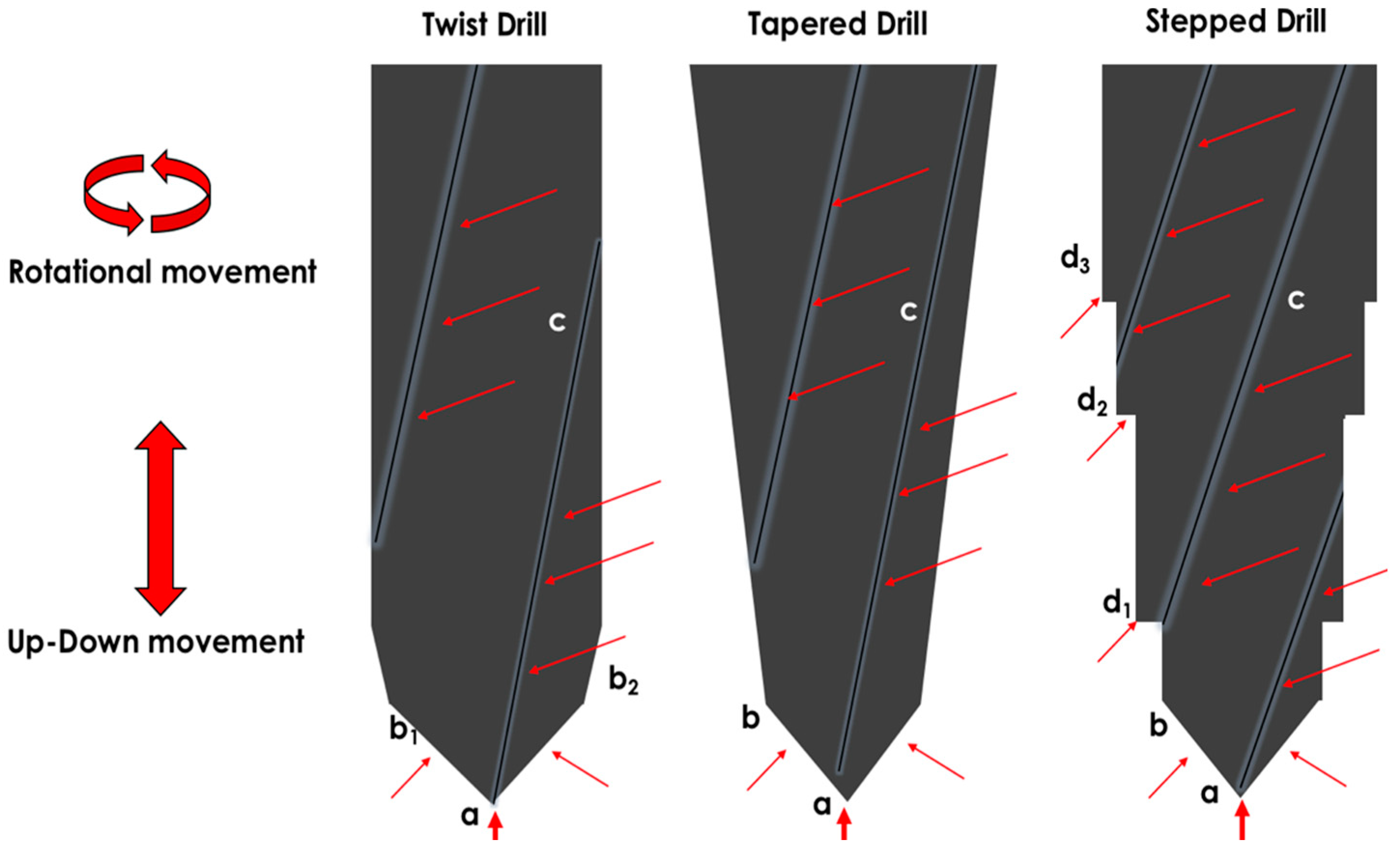
2.1.1. Implant Bed Preparation
Remarks
2.1.2. Implant Insertion
Remarks
2.2. Potential Causes of Titanium Particle and Ion Release in the Prosthetic Phase
2.2.1. Implant-Abutment Material Interface
Remarks
2.2.2. Microgap and Micromovement
Remarks
2.2.3. Titanium Oxide Layer Loss
Remarks
2.2.4. Corrosion
2.2.5. Tribocorrosion
2.2.6. Fluoride and Titanium Corrosion
Remarks
2.3. Potential Causes of Titanium Particle and Ion Release during the Maintenance Phase
2.3.1. Biofilms
2.3.2. Scaling Instruments
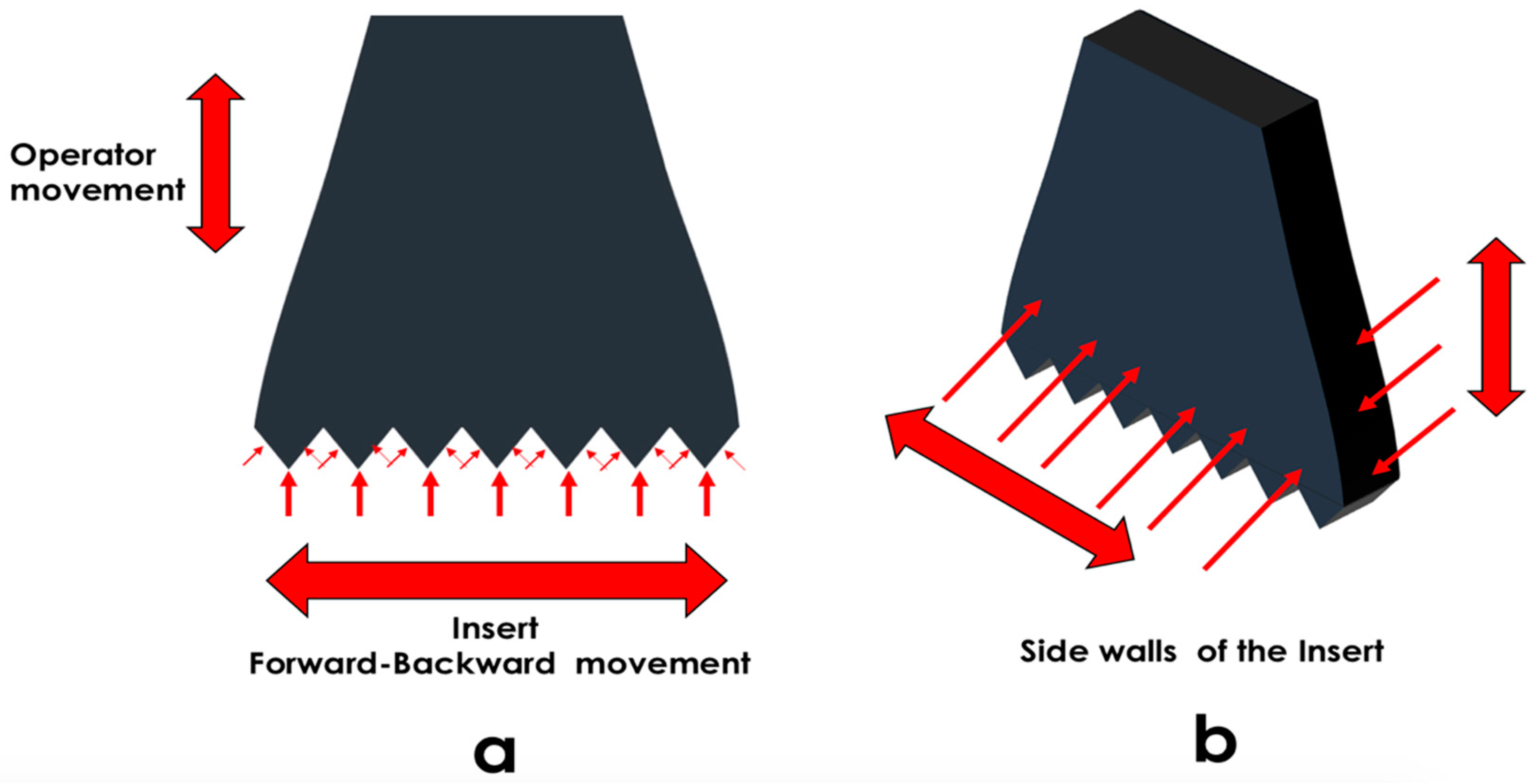
2.3.3. Implantoplasty
Remarks
2.3.4. Air-Abrasion
Remarks
2.3.5. Chemical Decontamination Methods
Remarks
2.3.6. Laser Decontamination Methods
Remarks on Laser-Assisted Decontamination Methods
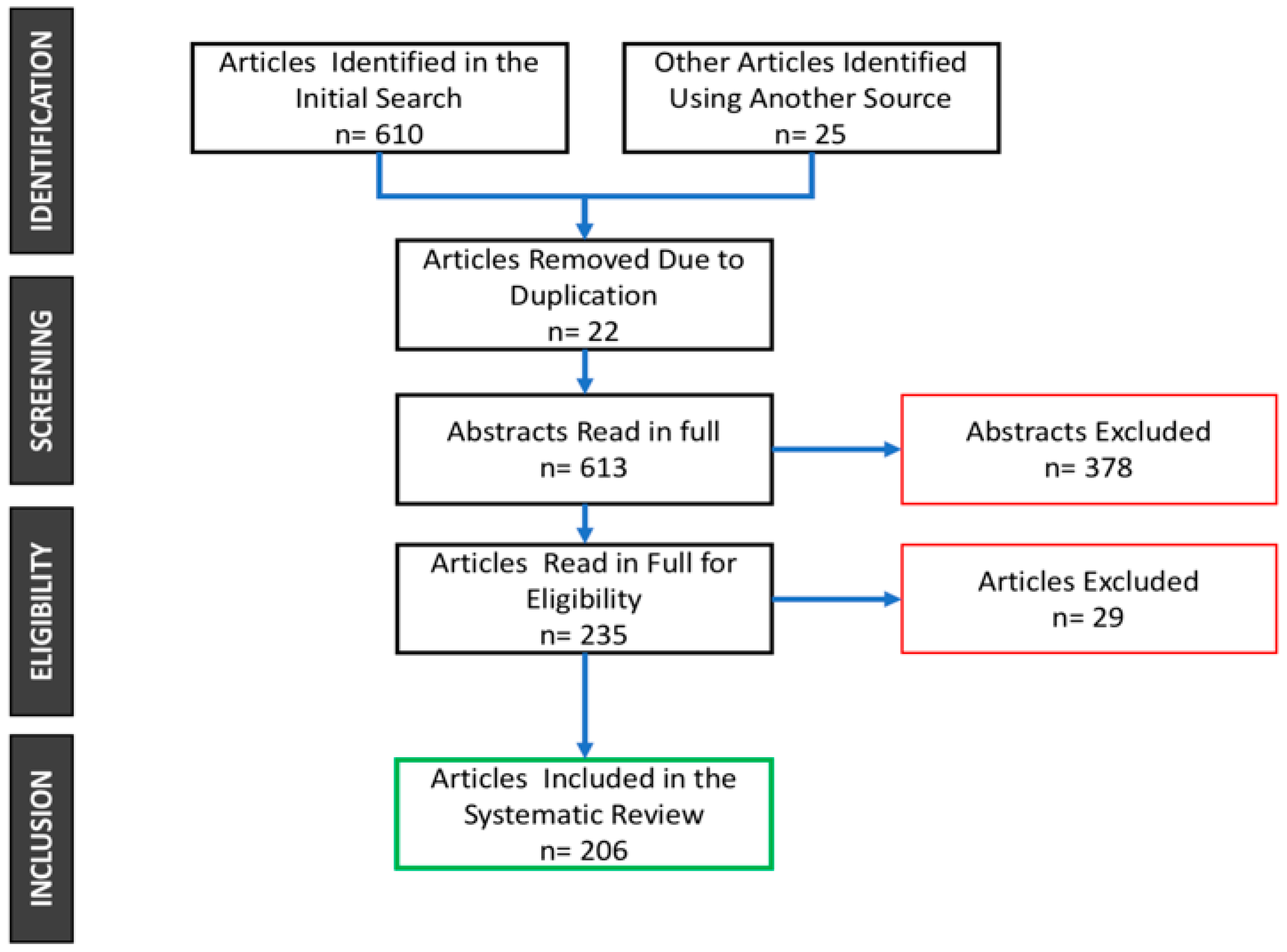
3. Materials and Methods
3.1. For the Surgical Phase
3.2. For the Prosthetic Phase
3.3. For the Maintenance Phase
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Staroveski, T.; Brezak, D.; Udiljak, T. Drill wear monitoring in cortical bone drilling. Med. Eng. Phys. 2015, 37, 560–566. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, A.B.; Laing, P.G.; Hodge, E.S. The ionization of metal implants in living tissues. J. Bone Jt. Surg. 1960, 42, 77–90. [Google Scholar] [CrossRef]
- Ferguson, A.B.; Akahoshi, Y.; Laing, P.G.; Hodge, E.S. Characteristics of trace ion release from embedded metal implants in the rabbit. J. Bone Jt. Surg. 1962, 44, 317–336. [Google Scholar] [CrossRef]
- Meachim, G.; Williams, D.F. Changes in nonosseous tissue adjacent to titanium implants. J. Biomed. Mater. Res. 1973, 7, 555–572. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, N.; Ichinose, S.; Kimijima, Y.; Mimura, M. Investigation of titanium leak to bone tissue surrounding dental titanium implant: Electron microscopic findings and analysis by electron diffraction. Med. Electron Microsc. 2000, 33, 96–101. [Google Scholar] [CrossRef] [PubMed]
- Senna, P.; Cury, A.A.D.; Kates, S.; Meirelles, L. Surface damage on dental implants with release of loose particles after insertion into bone. Clin. Implant Dent. Relat. Res. 2015, 17, 681–692. [Google Scholar] [CrossRef] [PubMed]
- Bianco, P.D.; Ducheyne, P.; Cuckler, J.M. Titanium serum and urine levels in rabbits with a titanium implant in the absence of wear. Biomaterials 1996, 17, 1937–1942. [Google Scholar] [CrossRef]
- Browne, M.; Gregson, P.J. Effect of mechanical surface pretreatment on metal ion release. Biomaterials 2000, 21, 385–392. [Google Scholar] [CrossRef]
- Ferguson, A.B. Metals in living tissues. Surg. Clin. N. Am. 1960, 40, 521–529. [Google Scholar] [CrossRef]
- Frateur, I.; Cattarin, S.; Musiani, M.; Tribollet, B. Electrodissolution of Ti and p-Si in acidic fluoride media: Formation ratio of oxide layers from electrochemical impedance spectroscopy. J. Electroanal. Chem. 2000, 482, 202–210. [Google Scholar] [CrossRef]
- Mabilleau, G.; Bourdon, S.; Joly-Guillou, M.L.; Filmon, R.; Baslé, M.F.; Chappard, D. Influence of fluoride, hydrogen peroxide and lactic acid on the corrosion resistance of commercially pure titanium. Acta Biomater. 2006, 2, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Siirila, H.S.; Kononen, M. The effect of oral topical fluorides on the surface of commercially pure titanium. Int. J. Oral Maxillofac. Implant. 1991, 6, 50–54. [Google Scholar]
- Toumelin-Chemla, F.; Rouelle, F.; Burdairon, G. Corrosive properties of fluoride-containing odontologic gels against titanium. J. Dent. 1996, 24, 109–115. [Google Scholar] [CrossRef]
- Barbieri, M.; Mencio, F.; Papi, P.; Rosella, D.; di Carlo, S.; Valente, T.; Pompa, G. Corrosion behavior of dental implants immersed into human saliva: Preliminary results of an in vitro study. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 3543–3548. [Google Scholar] [PubMed]
- Woodman, J.L.; Jacobs, J.J.; Galante, J.O.; Urban, R.M. Metal ion release from titanium-based prosthetic segmental replacements of long bones in baboons: A long-term study. J. Orthop. Res. 1984, 1, 421–430. [Google Scholar] [CrossRef] [PubMed]
- Louropoulou, A.; Slot, D.E.; van der Weijden, F.A. Titanium surface alterations following the use of different mechanical instruments: A systematic review. Clin. Oral Implants Res. 2012, 23, 643–658. [Google Scholar] [CrossRef] [PubMed]
- Ruhling, A.; Kocher, T.; Kreusch, J.; Plagmann, H.C. Treatment of subgingival implant surfaces with TeflonR-coated sonic and ultrasonic scaler tips and various implant curettes. An in vitro study. Clin. Oral Implant. Res. 1994, 5, 19–29. [Google Scholar] [CrossRef]
- Hallmon, W.W.; Waldrop, T.C.; Meffert, R.M.; Wade, B.W. A comparative study of the effects of metallic, nonmetallic, and sonic instrumentation on titanium abutment surfaces. Int. J. Oral Maxillofac. Implant. 1996, 11, 96–100. [Google Scholar] [CrossRef]
- Homiak, A.W.; Cook, P.A.; DeBoer, J. Effect of hygiene instrumentation on titanium abutments: A scanning electron microscopy study. J. Prosthet. Dent. 1992, 67, 364–369. [Google Scholar] [CrossRef]
- Cross-Poline, G.N.; Shaklee, R.L.; Stach, D.J. Effect of implant curettes on titanium implant surfaces. Am. J. Dent. 1997, 10, 41–45. [Google Scholar] [PubMed]
- Bertoldi, C.; Pradelli, J.M.; Consolo, U.; Zaffe, D. Release of elements from retrieved maxillofacial plates and screws. J. Mater. Sci. Mater. Med. 2005, 16, 857–861. [Google Scholar] [CrossRef] [PubMed]
- Thomas, P.; Bandl, W.-D.; Maier, S.; Summer, B.; Przybilla, B. Hypersensitivity to titanium osteosynthesis with impaired fracture healing, eczema, and T-cell hyperresponsiveness in vitro: Case report and review of the literature. Contact Dermat. 2006, 55, 199–202. [Google Scholar] [CrossRef] [PubMed]
- Rashad, A.; Sadr-Eshkevari, P.; Weuster, M.; Schmitz, I.; Prochnow, N.; Maurer, P. Material attrition and bone micromorphology after conventional and ultrasonic implant site preparation. Clin. Oral Implant. Res. 2013, 24, 110–114. [Google Scholar] [CrossRef] [PubMed]
- Ercoli, C.; Funkenbusch, P.D.; Lee, H.J.; Moss, M.E.; Graser, G.N. The influence of drill wear on cutting efficiency and heat production during osteotomy preparation for dental implants: A study of drill durability. Int. J. Oral Maxillofac. Implant. 2004, 19, 335–349. [Google Scholar]
- Carvalho, A.C.; Queiroz, T.P.; Okamoto, R.; Margonar, R.L.; Garcia, I.R., Jr.; Filho, O.M. Evaluation of bone heating, immediate bone cell viability, and wear of high-resistance drills after the creation of implant osteotomies in rabbit tibias. Int. J. Oral Maxillofac. Implant. 2011, 26, 1193–1201. [Google Scholar]
- De-Melo, J.F.; Gjerdet, N.R.; Erichsen, E.S. Metal release from cobalt-chromium partial dentures in the mouth. Acta Odontol. Scand. 1983, 41, 71–74. [Google Scholar] [CrossRef] [PubMed]
- Queiroz, T.P.; Souza, F.Á.; Okamoto, R.; Margonar, R.; Pereira-Filho, V.A.; Garcia, I.R.; Vieira, E.H. Evaluation of immediate bone-cell viability and of drill wear after implant osteotomies: Immunohistochemistry and scanning electron microscopy analysis. J. Oral Maxillofac. Surg. 2008, 66, 1233–1240. [Google Scholar] [CrossRef] [PubMed]
- Hochscheidt, C.J.; Shimizu, R.H.; Andrighetto, A.R.; Pierezan, R.; Thomé, G.; Salatti, R. Comparative analysis of cutting efficiency and surface maintenance between different types of implant drills. Implant Dent. 2017, 26, 723–729. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V.; Pandey, P.M. In-situ tool wear monitoring and its effects on the performance of porcine cortical bone drilling: A comparative in-vitro investigation. Mech. Adv. Mater. Mod. Process. 2017, 3, 1–7. [Google Scholar] [CrossRef]
- Dos Santos, P.L.; Queiroz, T.P.; Margonar, R.; de Souza Carvalho, A.C.; Betoni, W., Jr.; Rezende, R.R.; dos Santos, P.H.; Garcia, I.R., Jr. Evaluation of bone heating, drill deformation, and drill roughness after implant osteotomy: Guided surgery and classic drilling procedure. Int. J. Oral Maxillofac. Implant. 2014, 29, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Allsobrook, O.F.L.; Leichter, J.; Holborow, D.; Swain, M. Descriptive study of the longevity of dental implant surgery drills. Clin. Implant Dent. Relat. Res. 2011, 13, 244–254. [Google Scholar] [CrossRef] [PubMed]
- Chacon, G.E.; Bower, D.L.; Larsen, P.E.; McGlumphy, E.A.; Beck, F.M. Heat production by 3 implant drill systems after repeated drilling and sterilization. J. Oral Maxillofac. Surg. 2006, 64, 265–269. [Google Scholar] [CrossRef] [PubMed]
- Cooley, R.L.; Marshall, T.D.; Young, J.M.; Huddleston, A.M. Effect of sterilization on the strength and cutting efficiency of twist drills. Quintessence Int. 1990, 21, 919–923. [Google Scholar] [PubMed]
- Harris, B.H.; Kohles, S.S. Effects of mechanical and thermal fatigue on dental drill performance. Int. J. Oral Maxillofac. Implant. 2001, 16, 819–826. [Google Scholar]
- Mendes, G.C.B.; Padovan, L.E.M.; Ribeiro-Júnior, P.D.; Sartori, E.M.; Valgas, L.; Claudino, M. Influence of implant drill materials on wear, deformation, and roughness after repeated drilling and sterilization. Implant Dent. 2014, 23, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Sartori, E.M.; Shinohara, É.H.; Ponzoni, D.; Padovan, L.E.M.; Valgas, L.; Golin, A.L. Evaluation of deformation, mass loss, and roughness of different metal burs after osteotomy for osseointegrated implants. J. Oral Maxillofac. Surg. 2012, 70, e608–e621. [Google Scholar] [CrossRef] [PubMed]
- Guan, H.; van Staden, R.C.; Johnson, N.W.; Loo, Y.-C. Dynamic modelling and simulation of dental implant insertion process—A finite element study. Finite Elem. Anal. Des. 2011, 47, 886–897. [Google Scholar] [CrossRef]
- Wawrzinek, C.; Sommer, T.; Fischer-Brandies, H. Microdamage in cortical bone due to the overtightening of orthodontic microscrews. J. Orofac. Orthop. 2008, 69, 121–134. [Google Scholar] [CrossRef] [PubMed]
- Cvijović-Alagić, I.; Cvijović, Z.; Mitrović, S.; Panić, V.; Rakin, M. Wear and corrosion behaviour of Ti–13Nb–13Zr and Ti–6Al–4V alloys in simulated physiological solution. Corros. Sci. 2011, 53, 796–808. [Google Scholar] [CrossRef]
- Hokkirigawa, K.; Kato, K. An experimental and theoretical investigation of ploughing, cutting and wedge formation during abrasive wear. Tribol. Int. 1988, 21, 51–57. [Google Scholar] [CrossRef]
- Ni, W.; Cheng, Y.-T.; Lukitsch, M.J.; Weiner, A.M.; Lev, L.C.; Grummon, D.S. Effects of the ratio of hardness to Young’s modulus on the friction and wear behavior of bilayer coatings. Appl. Phys. Lett. 2004, 85, 4028–4030. [Google Scholar] [CrossRef]
- Rigney, D.A. Some thoughts on sliding wear. Wear 1992, 152, 187–192. [Google Scholar] [CrossRef]
- Schliephake, H.; Reiss, G.; Urban, R.; Neukam, F.W.; Guckel, S. Metal release from titanium fixtures during placement in the mandible: An experimental study. Int. J. Oral Maxillofac. Implant. 1993, 8, 502–511. [Google Scholar]
- Seki, Y.; Bessho, K.; Sugatani, T.; Kageyama, T.; Inui, M.; Tagawa, T. Clinicopathological study on titanium miniplates. J. Oral Maxillofac. Surg. 1994, 40, 892–896. [Google Scholar] [CrossRef]
- Kim, Y.-K.; Yeo, H.-H.; Lim, S.-C. Tissue response to titanium plates: A transmitted electron microscopic study. J. Oral Maxillofac. Surg. 1997, 55, 322–326. [Google Scholar] [CrossRef]
- Martini, D.; Fini, M.; Franchi, M.; Pasquale, V.D.; Bacchelli, B.; Gamberini, M.; Tinti, A.; Taddei, P.; Giavaresi, G.; Ottani, V.; et al. Detachment of titanium and fluorohydroxyapatite particles in unloaded endosseous implants. Biomaterials 2003, 24, 1309–1316. [Google Scholar] [CrossRef]
- Franchi, M.; Bacchelli, B.; Martini, D.; Pasquale, V.D.; Orsini, E.; Ottani, V.; Fini, M.; Giavaresi, G.; Giardino, R.; Ruggeri, A. Early detachment of titanium particles from various different surfaces of endosseous dental implants. Biomaterials 2004, 25, 2239–2246. [Google Scholar] [CrossRef] [PubMed]
- Geis-Gerstorfer, J.; Sauer, K.H.; Passler, K. Ion release from Ni-Cr-Mo and Co-Cr-Mo casting alloys. Int. J. Prosthodont. 1991, 4, 152–158. [Google Scholar] [PubMed]
- Pettersson, M.; Pettersson, J.; Thorén, M.M.; Johansson, A. Release of titanium after insertion of dental implants with different surface characteristics—An ex vivo animal study. Acta Biomater. Odontol. Scand. 2017, 3, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Wennerberg, A.; Ide-Ektessabi, A.; Hatkamata, S.; Sawase, T.; Johansson, C.; Albrektsson, T.; Martinelli, A.; Sodervall, U.; Odelius, H. Titanium release from implants prepared with different surface roughness. An in vitro and in vivo study. Clin. Oral Implant. Res. 2004, 15, 505–512. [Google Scholar] [CrossRef] [PubMed]
- Deppe, H.; Wolff, C.; Bauer, F.; Ruthenberg, R.; Sculean, A.; Mücke, T. Dental implant surfaces after insertion in bone: An in vitro study in four commercial implant systems. Clin. Oral Implant. Res. 2018, 22, 1593–1600. [Google Scholar] [CrossRef] [PubMed]
- Mints, D.; Elias, C.; Funkenbusch, P.; Meirelles, L. Integrity of implant surface modifications after insertion. Int. J. Oral Maxillofac. Implant. 2014, 29, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Salerno, M.; Itri, A.; Frezzato, M.; Rebaudi, A. Surface microstructure of dental implants before and after insertion: An in vitro study by means of scanning probe microscopy. Implant Dent. 2015, 24, 248–255. [Google Scholar] [CrossRef] [PubMed]
- Deppe, H.; Grünberg, C.; Thomas, M.; Sculean, A.; Benner, K.-U.; Bauer, F.J.M. Surface morphology analysis of dental implants following insertion into bone using scanning electron microscopy: A pilot study. Clin. Oral Implant. Res. 2015, 26, 1261–1266. [Google Scholar] [CrossRef] [PubMed]
- Flatebø, R.S.; Høl, P.J.; Leknes, K.N.; Kosler, J.; Lie, S.A.; Gjerdet, N.R. Mapping of titanium particles in peri-implant oral mucosa by laser ablation inductively coupled plasma mass spectrometry and high-resolution optical darkfield microscopy. J. Oral Pathol. Med. 2011, 40, 412–420. [Google Scholar] [CrossRef] [PubMed]
- Sridhar, S.; Wilson, T.G.; Valderrama, P.; Watkins-Curry, P.; Chan, J.Y.; Rodrigues, D.C. In vitro evaluation of titanium exfoliation during simulated surgical insertion of dental implants. J. Oral Implantol. 2016, 42, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Cundy, W.J.; Mascarenhas, A.R.; Antoniou, G.; Freeman, B.J.C.; Cundy, P.J. Local and systemic metal ion release occurs intraoperatively during correction and instrumented spinal fusion for scoliosis. J. Child. Orthop. 2015, 9, 39–43. [Google Scholar] [CrossRef] [PubMed]
- Sampson, B.; Hart, A. Clinical usefulness of blood metal measurements to assess the failure of metal-on-metal hip implants. Ann. Clin. Biochem. 2012, 49, 118–131. [Google Scholar] [CrossRef] [PubMed]
- USFDA. FDA Executive Summary Memorandum. Metal-On-Metal Hip Implant Systems; USFDA: Washington, DC, USA, 2012.
- Meyer, U.; Bühner, M.; Büchter, A.; Kruse-Lösler, B.; Stamm, T.; Wiesmann, H.P. Fast element mapping of titanium wear around implants of different surface structures. Clin. Oral Implant. Res. 2006, 17, 206–211. [Google Scholar] [CrossRef] [PubMed]
- Klotz, M.W.; Taylor, T.D.; Goldberg, A.J. Wear at the titanium-zirconia implant-abutment interface: A pilot study. Int. J. Oral Maxillofac. Implant. 2011, 26, 970–975. [Google Scholar]
- Stimmelmayr, M.; Edelhoff, D.; Güth, J.-F.; Erdelt, K.; Happe, A.; Beuer, F. Wear at the titanium–titanium and the titanium–zirconia implant–abutment interface: A comparative in vitro study. Dent. Mater. 2012, 28, 1215–1220. [Google Scholar] [CrossRef] [PubMed]
- Aboushelib, M.N.; Kleverlaan, C.J.; Feilzer, A.J. Evaluation of a high fracture toughness composite ceramic for dental applications. J. Prosthodont. 2008, 17, 538–544. [Google Scholar] [CrossRef] [PubMed]
- Elias, C.N.; Fernandes, D.J.; Resende, C.R.S.; Roestel, J. Mechanical properties, surface morphology and stability of a modified commercially pure high strength titanium alloy for dental implants. Dent. Mater. 2015, 31, e1–e13. [Google Scholar] [CrossRef] [PubMed]
- Cavusoglu, Y.; Akça, K.; Gürbüz, R.; Cehreli, M.C. A pilot study of joint stability at the zirconium or titanium abutment/titanium implant interface. Int. J. Oral Maxillofac. Implant. 2014, 29, 338–343. [Google Scholar] [CrossRef] [PubMed]
- Yüzügüllü, B.; Avci, M. The implant-abutment interface of alumina and zirconia abutments. Clin. Implant Dent. Relat. Res. 2008, 10, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Beuer, F.; Korczynski, N.; Rezac, A.; Naumann, M.; Gernet, W.; Sorensen, J.A. Marginal and internal fit of zirconia based fixed dental prostheses fabricated with different concepts. Clin. Cosmet. Investig. Dent. 2010, 2, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Gratton, D.G.; Aquilino, S.A.; Stanford, C.M. Micromotion and dynamic fatigue properties of the dental implant–abutment interface. J. Prosthet. Dent. 2001, 85, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Quek, H.C.; Tan, K.B.; Nicholls, J.I. Load fatigue performance of four implant-abutment interface designs: Effect of torque level and implant system. Int. J. Oral Maxillofac. Implant. 2008, 23, 253–262. [Google Scholar]
- Seetoh, Y.L.; Tan, K.B.; Chua, E.K.; Quek, H.C.; Nicholls, J.I. Load fatigue performance of conical implant-abutment connections. Int. J. Oral Maxillofac. Implant. 2011, 26, 797–806. [Google Scholar]
- Braian, M.; Bruyn, H.; Fransson, H.; Christersson, C.; Wennerberg, A. Tolerance measurements on internal- and external-hexagon implants. Int. J. Oral Maxillofac. Implant. 2014, 29, 846–852. [Google Scholar] [CrossRef] [PubMed]
- Rack, T.; Zabler, S.; Rack, A.; Riesemeier, H.; Nelson, K. An in vitro pilot study of abutment stability during loading in new and fatigue-loaded conical dental implants using synchrotron-based radiography. Int. J. Oral Maxillofac. Implant. 2013, 28, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Karl, M.; Taylor, T. Parameters determining micromotion at the implant-abutment interface. Int. J. Oral Maxillofac. Implant. 2014, 29, 1338–1347. [Google Scholar] [CrossRef] [PubMed]
- Alqutaibi, A.Y.; Aboalrejal, A.N. Microgap and micromotion at the implant abutment interface cause marginal bone loss around dental implant but more evidence is needed. J. Evid. Based Dent. Pract. 2018, 18, 171–172. [Google Scholar] [CrossRef] [PubMed]
- Blum, K.; Wiest, W.; Fella, C.; Balles, A.; Dittmann, J.; Rack, A.; Maier, D.; Thomann, R.; Spies, B.C.; Kohal, R.J.; et al. Fatigue induced changes in conical implant–abutment connections. Dent. Mater. 2015, 31, 1415–1426. [Google Scholar] [CrossRef] [PubMed]
- Pereira, J.; Morsch, C.; Henriques, B.; Nascimento, R.; Benfatti, C.; Silva, F.; López-López, J.; Souza, J. Removal torque and biofilm accumulation at two dental implant–abutment joints after fatigue. Int. J. Oral Maxillofac. Implant. 2016, 31, 813–819. [Google Scholar] [CrossRef] [PubMed]
- Prado, A.; Pereira, J.; Henriques, B.; Benfatti, C.; Magini, R.; López-López, J.; Souza, J. Biofilm affecting the mechanical integrity of implant-abutment joints. Int. J. Prosthodont. 2016, 29, 381–383. [Google Scholar] [CrossRef] [PubMed]
- Lopes, P.A.; Carreiro, A.F.P.; Nascimento, R.M.; Vahey, B.R.; Henriques, B.; Souza, J.C.M. Physicochemical and microscopic characterization of implant-abutment joints. Eur. J. Dent. 2018, 12, 100–104. [Google Scholar] [CrossRef] [PubMed]
- Tsuge, T.; Hagiwara, Y.; Matsumura, H. Marginal fit and microgaps of implant-abutment interface with internal anti-rotation configuration. Dent. Mater. J. 2008, 27, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Binon, P.P. The effect of implant/abutment hexagonal misfit on screw joint stability. Int. J. Prosthodont. 1996, 9, 149–160. [Google Scholar] [PubMed]
- Kuromoto, N.K.; Simão, R.A.; Soares, G.A. Titanium oxide films produced on commercially pure titanium by anodic oxidation with different voltages. Mater. Charact. 2007, 58, 114–121. [Google Scholar] [CrossRef]
- Rodriguez, L.L.; Sundaram, P.A.; Rosim-Fachini, E.; Padovani, A.M.; Diffoot-Carlo, N. Plasma electrolytic oxidation coatings on γTiAl alloy for potential biomedical applications. J. Biomed. Mater. Res. Part B Appl. Biomater. 2014, 102, 988–1001. [Google Scholar] [CrossRef] [PubMed]
- Gittens, R.A.; Scheideler, L.; Rupp, F.; Hyzy, S.L.; Geis-Gerstorfer, J.; Schwartz, Z.; Boyan, B.D. A review on the wettability of dental implant surfaces II: Biological and clinical aspects. Acta Biomater. 2014, 10, 2907–2918. [Google Scholar] [CrossRef] [PubMed]
- Khanlou, H.M.; Ang, B.C.; Barzani, M.M.; Silakhori, M.; Talebian, S. Prediction and characterization of surface roughness using sandblasting and acid etching process on new non-toxic titanium biomaterial: Adaptive-network-based fuzzy inference System. Neural Comput. Appl. 2015, 26, 1751–1761. [Google Scholar] [CrossRef]
- Long, M.; Rack, H.J. Titanium alloys in total joint replacement—A materials science perspective. Biomaterials 1998, 19, 1621–1639. [Google Scholar] [CrossRef]
- Addison, O.; Davenport, A.J.; Newport, R.J.; Kalra, S.; Monir, M.; Mosselmans, J.F.W.; Proops, D.; Martin, R.A. Do ‘passive’ medical titanium surfaces deteriorate in service in the absence of wear? J. R. Soc. Interface 2012, 9, 3161–3164. [Google Scholar] [CrossRef] [PubMed]
- Abey, S.; Mathew, M.T.; Lee, D.J.; Knoernschild, K.L.; Wimmer, M.A.; Sukotjo, C. Electrochemical behavior of titanium in artificial saliva: Influence of pH. J. Oral Implantol. 2014, 40, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Mathew, M.T.; Abbey, S.; Hallab, N.J.; Hall, D.J.; Sukotjo, C.; Wimmer, M.A. Influence of pH on the tribocorrosion behavior of CpTi in the oral environment: Synergistic interactions of wear and corrosion. J. Biomed. Mater. Res. Part B Appl. Biomater. 2012, 100B, 1662–1671. [Google Scholar] [CrossRef] [PubMed]
- Souza, J.C.M.; Henriques, M.; Oliveira, R.; Teughels, W.; Celis, J.P.; Rocha, L.A. Do oral biofilms influence the wear and corrosion behavior of titanium? Biofouling 2010, 26, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, T.P. An overview of the corrosion aspect of dental implants (titanium and its alloys). Indian J. Dent. Res. 2009, 20, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Mouhyi, J.; Ehrenfest, D.M.D.; Albrektsson, T. The peri-implantitis: Implant surfaces, microstructure, and physicochemical aspects. Clin. Implant Dent. Relat. Res. 2012, 14, 170–183. [Google Scholar] [CrossRef] [PubMed]
- Bhola, R.; Bhola, S.M.; Mishra, B.; Olson, D.L. Corrosion in titanium dental implants/ prostheses—A review. Trends Biomater. Artif. Organs 2011, 25, 34–46. [Google Scholar]
- Reclaru, L.; Meyer, J.M. Study of corrosion between a titanium implant and dental alloys. J. Dent. 1994, 22, 159–168. [Google Scholar] [CrossRef]
- Souza, J.C.M.; Ponthiaux, P.; Henriques, M.; Oliveira, R.; Teughels, W.; Celis, J.-P.; Rocha, L.A. Corrosion behaviour of titanium in the presence of Streptococcus mutans. J. Dent. 2013, 41, 528–534. [Google Scholar] [CrossRef] [PubMed]
- Souza, J.C.M.; Barbosa, S.L.; Ariza, E.A.; Henriques, M.; Teughels, W.; Ponthiaux, P.; Celis, J.-P.; Rocha, L.A. How do titanium and Ti6Al4V corrode in fluoridated medium as found in the oral cavity? An in vitro study. Mater. Sci. Eng. C 2015, 47, 384–393. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, D.; Valderrama, P.; Wilson, T.; Palmer, K.; Thomas, A.; Sridhar, S.; Adapalli, A.; Burbano, M.; Wadhwani, C. Titanium corrosion mechanisms in the oral environment: A retrieval study. Materials 2013, 6, 5258–5274. [Google Scholar] [CrossRef] [PubMed]
- Olmedo, D.G.; Tasat, D.R.; Duffo, G.; Guglielmotti, M.B.; Cabrini, R.L. The issue of corrosion in dental implants: A review. Acta Odontol. Latinoam. 2009, 22, 3–9. [Google Scholar] [PubMed]
- Nikolopoulou, F. Saliva and dental implants. Implant Dent. 2006, 15, 372–376. [Google Scholar] [CrossRef] [PubMed]
- Henry, P.J. Clinical experiences with dental implants. Adv. Dent. Res. 1999, 13, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Gittens, R.A.; Olivares-Navarrete, R.; Tannenbaum, R.; Boyan, B.D.; Schwartz, Z. Electrical implications of corrosion for osseointegration of titanium implants. J. Dent. Res. 2011, 90, 1389–1397. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, J.J.; Gilbert, J.L.; Urban, R.M. Corrosion of metal orthopaedic implants. J. Bone Jt. Surg. 1998, 80, 268–282. [Google Scholar] [CrossRef]
- Geis-Gerstorfer, J. In vitro corrosion measurements of dental alloys. J. Dent. 1994, 22, 247–251. [Google Scholar] [CrossRef]
- Branemark, P.I.; Hansson, B.O.; Adell, R.; Breine, U.; Lindstrom, J.; Hallen, O.; Ohman, A. Osseointegrated implants in the treatment of the edentulous jaw. Experience from a 10-year period. Scand. J. Plast. Reconstr. Surg. Suppl. 1997, 16, 1–132. [Google Scholar]
- Adell, R.; Lekholm, U.; Rockler, B.; Brånemark, P.I. A 15-year study of osseointegrated implants in the treatment of the edentulous jaw. Int. J. Oral Surg. 1981, 10, 387–416. [Google Scholar] [CrossRef]
- Galante, J.O.; Lemons, J.; Spector, M.; Wilson, P.D.; Wright, T.M. The biologic effects of implant materials. J. Orthop. Res. 1991, 9, 760–775. [Google Scholar] [CrossRef] [PubMed]
- Hallab, N.J.; Mikecz, K.; Vermes, C.; Skipor, A.; Jacobs, J.J. Orthopaedic implant related metal toxicity in terms of human lymphocyte reactivity to metal-protein complexes produced from cobalt-base and titanium-base implant alloy degradation. Mol. Cell. Biochem. 2001, 222, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Hallab, N.J.; Jacobs, J.J.; Skipor, A.; Black, J.; Mikecz, K.; Galante, J.O. Systemic metal-protein binding associated with total joint replacement arthroplasty. J. Biomed. Mater. Res. 2000, 49, 353–361. [Google Scholar] [CrossRef]
- Correa, C.B.; Pires, J.R.; Fernandes-Filho, R.B.; Sartori, R.; Vaz, L.G. Fatigue and fluoride corrosion on Streptococcus mutansadherence to titanium-based implant/component surfaces. J. Prosthodont. 2009, 18, 382–387. [Google Scholar] [CrossRef] [PubMed]
- Fathi, M.H.; Salehi, M.; Saatchi, A.; Mortazavi, V.; Moosavi, S.B. In vitro corrosion behavior of bioceramic, metallic, and bioceramic–metallic coated stainless steel dental implants. Dent. Mater. 2003, 19, 188–198. [Google Scholar] [CrossRef]
- Yu, F.; Addison, O.; Baker, S.J.; Davenport, A.J. Lipopolysaccharide inhibits or accelerates biomedical titanium corrosion depending on environmental acidity. Int. J. Oral Sci. 2015, 7, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Simon, B.I.; Goldman, H.M.; Ruben, M.P.; Baker, E. The role of endotoxin in periodontal disease. I. A reproducible, quantitative method for determining the amount of endotoxin in human gingival exudate. J. Periodontol. 1969, 40, 695–701. [Google Scholar] [CrossRef] [PubMed]
- Casarin, R.C.V.; Ribeiro, É.P.; Mariano, F.S.; Nociti, F.H., Jr.; Casati, M.Z.; Gonçalves, R.B. Levels of aggregatibacter actinomycetemcomitans, Porphyromonas gingivalis, inflammatory cytokines and species-specific immunoglobulin G in generalized aggressive and chronic periodontitis. J. Periodontal. Res. 2010, 45, 635–642. [Google Scholar] [CrossRef] [PubMed]
- Mathew, M.T.; Barão, V.A.; Yuan, J.C.-C.; Assunção, W.G.; Sukotjo, C.; Wimmer, M.A. What is the role of lipopolysaccharide on the tribocorrosive behavior of titanium? J. Mech. Behav. Biomed. Mater. 2012, 8, 71–85. [Google Scholar] [CrossRef] [PubMed]
- Gil, F.J.; Canedo, R.; Padros, A.; Baneres, M.V.; Arano, J.M. Fretting corrosion behaviour of ball-and-socket joint on dental implants with different prosthodontic alloys. Biomed. Mater. Eng. 2003, 13, 27–34. [Google Scholar] [PubMed]
- Hjalmarsson, L.; Smedberg, J.I.; Wennerberg, A. Material degradation in implant-retained cobalt-chrome and titanium frameworks. J. Oral Rehabil. 2010, 38, 61–71. [Google Scholar] [CrossRef] [PubMed]
- Wataha, J.C. Biocompatibility of dental casting alloys: A review. J. Prosthet. Dent. 2000, 83, 223–234. [Google Scholar] [CrossRef]
- Brune, D. Metal release from dental biomaterials. Biomaterials 1986, 7, 163–175. [Google Scholar] [CrossRef]
- Wataha, J.C.; Malcolm, C.T.; Hanks, C.T. Correlation between cytotoxicity and the elements released by dental casting alloys. Int. J. Prosthodont. 1995, 8, 9–14. [Google Scholar] [PubMed]
- Sarkar, N.; Fuys, R.; Stanford, J.W. Applications of electrochemical techniques to characterize the corrosion of dental alloys. In Corrosion and Degradation of Implant Materials; Syrett, B.C., Acharya, A., Eds.; ASTM: West Conshohocken, PA, USA, 1979; pp. 277–294. [Google Scholar]
- Revathi, A.; Borrás, A.D.; Muñoz, A.I.; Richard, C.; Manivasagam, G. Degradation mechanisms and future challenges of titanium and its alloys for dental implant applications in oral environment. Mater. Sci. Eng. C 2017, 76, 1354–1368. [Google Scholar] [CrossRef] [PubMed]
- Mathew, M.T.; Pai, P.S.; Pourzal, R.; Fischer, A.; Wimmer, M.A. Significance of tribocorrosion in biomedical applications: Overview and current status. Adv. Tribol. 2009, 2009, 1–12. [Google Scholar] [CrossRef]
- Landolt, D.; Mischler, S.; Stemp, M. Electrochemical methods in tribocorrosion: A critical appraisal. Electrochim. Acta 2001, 46, 3913–3929. [Google Scholar] [CrossRef]
- Licausi, M.P.; Muñoz, A.I.; Borrás, V.A. Influence of the fabrication process and fluoride content on the tribocorrosion behaviour of Ti6Al4V biomedical alloy in artificial saliva. J. Mech. Behav. Biomed. Mater. 2013, 20, 137–148. [Google Scholar] [CrossRef] [PubMed]
- Dodds, M.W.J.; Johnson, D.A.; Yeh, C.-K. Health benefits of saliva: A review. J. Dent. 2005, 33, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Cruz, H.V.; Souza, J.C.M.; Henriques, M.; Rocha, L.A. Tribocorrosion and bio-tribocorrosion in the oral environment: The case of dental implants. In Biomedical Tribology; Davim, J.P., Ed.; Nova Science Publishers: New York, NY, USA, 2011; pp. 1–33. [Google Scholar]
- Souza, J.C.M.; Henriques, M.; Teughels, W.; Ponthiaux, P.; Celis, J.-P.; Rocha, L.A. Wear and corrosion interactions on titanium in oral environment: Literature review. J. Bio-Tribo-Corros. 2015, 1, 13. [Google Scholar] [CrossRef]
- Mischler, S. Triboelectrochemical techniques and interpretation methods in tribocorrosion: A comparative evaluation. Tribol. Int. 2008, 41, 573–583. [Google Scholar] [CrossRef]
- Schiff, N.; Grosgogeat, B.; Lissac, M.; Dalard, F. Influence of fluoride content and pH on the corrosion resistance of titanium and its alloys. Biomaterials 2002, 23, 1995–2002. [Google Scholar] [CrossRef]
- Huang, H.-H. Effects of fluoride concentration and elastic tensile strain on the corrosion resistance of commercially pure titanium. Biomaterials 2002, 23, 59–63. [Google Scholar] [CrossRef]
- Kaneko, K.; Yokoyama, K.I.; Moriyama, K.; Asaoka, K.; Sakai, J.I.; Nagumo, M. Delayed fracture of beta titanium orthodontic wire in fluoride aqueous solutions. Biomaterials 2003, 24, 2113–2120. [Google Scholar] [CrossRef]
- Ratner, B.D.; Johnston, A.B.; Lenk, T.J. Biomaterial surfaces. J. Biomed. Mater. Res. 1987, 21, 59–89. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.C.; Pilliar, R.M.; Chernecky, R. Dental implant materials. I. Some effects of preparative procedures on surface topography. J. Biomed. Mater. Res. 1991, 25, 1045–1068. [Google Scholar] [CrossRef] [PubMed]
- Christersson, C.E.; Dunford, R.G.; Glantz, P.O.; Baier, R.E. Effect of critical surface tension on retention of oral microorganisms. Scand. J. Dent. Res. 1989, 97, 247–256. [Google Scholar] [CrossRef] [PubMed]
- Teughels, W.; van Assche, N.; Sliepen, I.; Quirynen, M. Effect of material characteristics and/or surface topography on biofilm development. Clin. Oral Implant. Res. 2006, 17, 68–81. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Lassiter, M.O.; Banas, J.A.; Galperín, M.Y.; Taylor, K.G.; Doyle, R.J. Multiple glucan-binding proteins of Streptococcus sobrinus. J. Bacteriol. 1996, 178, 1572–1577. [Google Scholar] [CrossRef] [PubMed]
- Steinberg, D.; Eyal, S. Early formation of Streptococcus sobrinus biofilm on various dental restorative materials. J. Dent. 2001, 30, 47–51. [Google Scholar] [CrossRef]
- Zitzmann, N.U.; Abrahamsson, I.; Berglundh, T.; Lindhe, J. Soft tissue reactions to plaque formation at implant abutments with different surface topography. An experimental study in dogs. J. Clin. Periodontol. 2002, 29, 456–461. [Google Scholar] [CrossRef] [PubMed]
- Dmytiyk, J.J.; Fox, S.C.; Moriarty, J.D. The effects of scaling titanium implant surfaces with metal and plastic instruments on cell attachment. J. Periodontol. 1990, 61, 491–496. [Google Scholar] [CrossRef] [PubMed]
- Fox, S.C.; Moriarty, J.D.; Kusy, R.P. The effects of scaling a titanium implant surface with metal and plastic instruments: An in vitro study. J. Periodontol. 1990, 61, 485–490. [Google Scholar] [CrossRef] [PubMed]
- Schou, S.; Berglundh, T.; Lang, N.P. Surgical treatment of peri-implantitis. Int. J. Oral Maxillofac. Implant. 2004, 19, 140–149. [Google Scholar]
- Claffey, N.; Clarke, E.; Polyzois, I.; Renvert, S. Surgical treatment of peri-implantitis. J. Clin. Periodontol. 2008, 35, 316–332. [Google Scholar] [CrossRef] [PubMed]
- Augthun, M.; Tinschert, J.; Huber, A. In vitro studies on the effect of cleaning methods on different implant surfaces. J. Periodontol. 1998, 69, 857–864. [Google Scholar] [CrossRef] [PubMed]
- Brookshire, F.V.G.; Nagy, W.W.; Dhuru, V.B.; Ziebert, G.J.; Chada, S. The qualitative effects of various types of hygiene instrumentation on commercially pure titanium and titanium alloy implant abutments: An in vitro and scanning electron microscope study. J. Prosthet. Dent. 1997, 78, 286–294. [Google Scholar] [CrossRef]
- Meier, R.M.; Pfammatter, C.; Zitzmann, N.U.; Filippi, A.; Kuhl, S. Surface quality after implantoplasty. Schweiz. Monatsschr. Zahnmed. 2012, 122, 714–724. [Google Scholar] [PubMed]
- Ramaglia, L.; di Lauro, A.E.; Morgese, F.; Squillace, A. Profilometric and standard error of the mean analysis of rough implant surfaces treated with different instrumentations. Implant Dent. 2006, 15, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Barbour, M.E.; O’Sullivan, D.J.; Jenkinson, H.F.; Jagger, D.C. The effects of polishing methods on surface morphology, roughness and bacterial colonisation of titanium abutments. J. Mater. Sci. Mater. Med. 2007, 18, 1439–1447. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, F.; Sahm, N.; Iglhaut, G.; Becker, J. Impact of the method of surface debridement and decontamination on the clinical outcome following combined surgical therapy of peri-implantitis: A randomized controlled clinical study. J. Clin. Periodontol. 2011, 38, 276–284. [Google Scholar] [CrossRef] [PubMed]
- Rimondini, L.; Simoncini, F.C.; Carrassi, A. Micro-morphometric assessment of titanium plasma-sprayed coating removal using burs for the treatment of peri-implant disease. Clin. Oral Implant. Res. 2000, 11, 129–138. [Google Scholar] [CrossRef]
- Valderrama, P.; Wilson, T.G., Jr. Detoxification of implant surfaces affected by peri-implant disease: An overview of surgical methods. Int. J. Dent. 2013, 2013, 740680. [Google Scholar] [CrossRef] [PubMed]
- Ramel, C.F.; Lüssi, A.; Özcan, M.; Jung, R.E.; Hämmerle, C.H.F.; Thoma, D.S. Surface roughness of dental implants and treatment time using six different implantoplasty procedures. Clin. Oral Implant. Res. 2016, 27, 776–781. [Google Scholar] [CrossRef] [PubMed]
- Rosen, P.; Qari, M.; Froum, S.; Dibart, S.; Chou, L. A pilot study on the efficacy of a treatment algorithm to detoxify dental implant surfaces affected by peri-implantitis. Int. J. Periodontics Restor. Dent. 2018, 38, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Moëne, R.; Décaillet, F.; Andersen, E.; Mombelli, A. Subgingival plaque removal using a new air-polishing device. J. Periodontol. 2010, 81, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Kozlovsky, A.; Soldinger, M.; Sperling, I. The effectiveness of the air-powder abrasive device on the tooth and periodontium: An overview. Clin. Prev. Dent. 1989, 11, 7–11. [Google Scholar] [PubMed]
- Atkinson, D.R.; Cobb, C.M.; Killoy, W.J. The effect of an air-powder abrasive system on in vitro root surfaces. J. Periodontol. 1984, 55, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Petersilka, G.J.; Bell, M.; Haberlein, I.; Mehl, A.; Hickel, R.; Flemmig, T.F. In vitro evaluation of novel low abrasive air polishing powders. J. Clin. Periodontol. 2003, 30, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Petersilka, G.J.; Tunkel, J.; Barakos, K.; Heinecke, A.; Häberlein, I.; Flemmig, T.F. Subgingival plaque removal at interdental sites using a low-abrasive air polishing powder. J. Periodontol. 2003, 74, 307–311. [Google Scholar] [CrossRef] [PubMed]
- Petersilka, G.J.; Steinmann, D.; Haberlein, I.; Heinecke, A.; Flemmig, T.F. Subgingival plaque removal in buccal and lingual sites using a novel low abrasive air-polishing powder. J. Clin. Periodontol. 2003, 30, 328–333. [Google Scholar] [CrossRef] [PubMed]
- Tastepe, C.S.; Lin, X.; Donnet, M.; Wismeijer, D.; Liu, Y. Parameters that improve cleaning efficiency of subgingival air polishing on titanium implant surfaces: An in vitro study. J. Periodontol. 2017, 88, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Ronay, V.; Merlini, A.; Attin, T.; Schmidlin, P.R.; Sahrmann, P. In vitro cleaning potential of three implant debridement methods. Simulation of the non-surgical approach. Clin. Oral Implant. Res. 2017, 28, 151–155. [Google Scholar] [CrossRef] [PubMed]
- Duarte, P.M.; Reis, A.F.; de Freitas, P.M.; Ota-Tsuzuki, C. Bacterial adhesion on smooth and rough titanium surfaces after treatment with different instruments. J. Periodontol. 2009, 80, 1824–1832. [Google Scholar] [CrossRef] [PubMed]
- Kreisler, M.; Al Haj, H.; d’Hoedt, B. Clinical efficacy of semiconductor laser application as an adjunct to conventional scaling and root planing. Lasers Surg. Med. 2005, 37, 350–355. [Google Scholar] [CrossRef] [PubMed]
- Kreisler, M.; Kohnen, W.; Christoffers, A.-B.; Götz, H.; Jansen, B.; Duschner, H.; D’Hoedt, B. In vitro evaluation of the biocompatibility of contaminated implant surfaces treated with an Er:YAG laser and an air powder system. Clin. Oral Implant. Res. 2005, 16, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Razzoog, M.E.; Koka, S. In vitro analysis of the effects of two air-abrasive prophylaxis systems and inlet air pressure on the surface of titanium abutment cylinders. J. Prosthodont. 1994, 3, 103–107. [Google Scholar] [CrossRef] [PubMed]
- Finnegan, M.; Linley, E.; Denyer, S.P.; McDonnell, G.; Simons, C.; Maillard, J.Y. Mode of action of hydrogen peroxide and other oxidizing agents: Differences between liquid and gas forms. J. Antimicrob. Chemother. 2010, 65, 2108–2115. [Google Scholar] [CrossRef] [PubMed]
- Gosau, M.; Hahnel, S.; Schwarz, F.; Gerlach, T.; Reichert, T.E.; Burgers, R. Effect of six different peri-implantitis disinfection methods on in vivo human oral biofilm. Clin. Oral Implant. Res. 2010, 21, 866–872. [Google Scholar] [CrossRef]
- Waal, Y.C.M.; Raghoebar, G.M.; Meijer, H.J.A.; Winkel, E.G.; van Winkelhoff, A.J. Implant decontamination with 2% chlorhexidine during surgical peri-implantitis treatment: A randomized, double-blind, controlled trial. Clin. Oral Implant. Res. 2014, 26, 1015–1023. [Google Scholar] [CrossRef] [PubMed]
- Valderrama, P.; Gonzalez, M.G.; Cantu, M.G.; Wilson, T.G. Detoxification of implant surfaces affected by peri-implant disease: An overview of non-surgical methods. Open Dent. J. 2014, 8, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Wheelis, S.E.; Gindri, I.M.; Valderrama, P.; Wilson, T.G.; Huang, J.; Rodrigues, D.C. Effects of decontamination solutions on the surface of titanium: Investigation of surface morphology, composition, and roughness. Clin. Oral Implant. Res. 2016, 27, 329–340. [Google Scholar] [CrossRef] [PubMed]
- Könönen, M.H.O.; Lavonius, E.T.; Kivilahti, J.K. SEM observations on stress corrosion cracking of commercially pure titanium in a topical fluoride solution. Dent. Mater. 1995, 11, 269–272. [Google Scholar] [CrossRef]
- Rodrigues, D.C.; Urban, R.M.; Jacobs, J.J.; Gilbert, J.L. In vivo severe corrosion and hydrogen embrittlement of retrieved modular body titanium alloy hip-implants. J. Biomed. Mater. Res. Part B Appl. Biomater. 2009, 88, 206–219. [Google Scholar] [CrossRef] [PubMed]
- Sartori, R.; Correa, C.B.; Marcantonio, E., Jr.; Vaz, L.G. Influence of a fluoridated medium with different pHs on commercially pure titanium-based implants. J. Prosthodont. 2009, 18, 130–134. [Google Scholar] [CrossRef] [PubMed]
- Muguruma, T.; Iijima, M.; Brantley, W.A.; Yuasa, T.; Kyung, H.-M.; Mizoguchi, I. Effects of sodium fluoride mouth rinses on the torsional properties of miniscrew implants. Am. J. Orthod. Dentofac. Orthop. 2011, 139, 588–593. [Google Scholar] [CrossRef] [PubMed]
- Toniollo, M.B.; Galo, R.; Macedo, A.P.; Rodrigues, R.C.S.; Ribeiro, R.F.; Mattos, M.D.G. Effect of fluoride sodium mouthwash solutions on cpTI: Evaluation of physicochemical properties. Braz. Dent. J. 2012, 23, 496–501. [Google Scholar] [CrossRef] [PubMed]
- Wiedmer, D.; Petersen, F.C.; Lönn-Stensrud, J.; Tiainen, H. Antibacterial effect of hydrogen peroxide-titanium dioxide suspensions in the decontamination of rough titanium surfaces. Biofouling 2017, 33, 451–459. [Google Scholar] [CrossRef] [PubMed]
- Ntrouka, V.I.; Slot, D.E.; Louropoulou, A.; van der Weijden, F. The effect of chemotherapeutic agents on contaminated titanium surfaces: A systematic review. Clin. Oral Implant. Res. 2011, 22, 681–690. [Google Scholar] [CrossRef] [PubMed]
- Ntrouka, V.; Hoogenkamp, M.; Zaura, E.; van der Weijden, F. The effect of chemotherapeutic agents on titanium-adherent biofilms. Clin. Oral Implants Res. 2011, 22, 1227–1234. [Google Scholar] [CrossRef] [PubMed]
- Ramesh, D.; Sridhar, S.; Siddiqui, D.A.; Valderrama, P.; Rodrigues, D.C. Detoxification of titanium implant surfaces: Evaluation of surface morphology and bone-forming cell compatibility. J. Bio-Tribo-Corros. 2017, 3, 1–13. [Google Scholar] [CrossRef]
- Oliveira, M.N.; Schunemann, W.V.H.; Mathew, M.T.; Henriques, B.; Magini, R.S.; Teughels, W.; Souza, J.C.M. Can degradation products released from dental implants affect peri-implant tissues? J. Periodontal Res. 2018, 53, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Souza, J.G.S.; Cordeiro, J.M.; Lima, C.V.; Barão, V.A.R. Citric acid reduces oral biofilm and influences the electrochemical behavior of titanium: An in situ and in vitro study. J. Periodontol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Ungvari, K.; Pelsoczi, I.K.; Kormos, B.; Oszko, A.; Rakonczay, Z.; Kemeny, L.; Radnai, M.; Nagy, K.; Fazekas, A.; Turzo, K. Effects on titanium implant surfaces of chemical agents used for the treatment of peri-implantitis. J. Biomed. Mater. Res. B Appl. Biomater. 2010, 94, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Soukos, N.S.; Mulholland, S.E.; Socransky, S.S.; Doukas, A.G. Photodestruction of human dental plaque bacteria: Enhancement of the photodynamic effect by photomechanical waves in an oral biofilm model. Lasers Surg. Med. 2003, 33, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Kreisler, M.; Kohnen, W.; Marinello, C.; Schoof, J.; Langnau, E.; Jansen, B.; d’Hoedt, B. Antimicrobial efficacy of semiconductor laser irradiation on implant surfaces. Int. J. Oral Maxillofac. Implant. 2003, 18, 706–711. [Google Scholar]
- Cohen, M.I.; Epperson, J.P. Application of lasers to microelectronic fabrication. In Electron Beam and Laser Beam Technology; Marton, L., El-Kareh, A.B., Eds.; Academic Press: New York, NY, USA, 1968; pp. 139–186. [Google Scholar]
- Wilson, M. Photolysis of oral bacteria and its potential use in the treatment of caries and periodontal disease. J. Appl. Bacteriol. 1993, 75, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Hall, R.R. The healing of tissues incised by a carbon-dioxide laser. Br. J. Surg. 1971, 58, 222–225. [Google Scholar] [CrossRef] [PubMed]
- Kreisler, M.; Gotz, H.; Duschner, H. Effect of Nd:YAG, Ho:YAG, Er: YAG, CO2, and GaAIAs laser irradiation on surface properties of endosseous dental implants. Int. J. Oral Maxillofac. Implant. 2002, 17, 202–211. [Google Scholar]
- Shibli, J.A.; Theodoro, L.H.; Haypek, P.; Garcia, V.G.; Marcantonio, E. The effect of CO2 laser irradiation on failed implant surfaces. Implant. Dent. 2004, 13, 342–351. [Google Scholar] [CrossRef] [PubMed]
- Park, C.-Y.; Kim, S.-G.; Kim, M.-D.; Eom, T.-G.; Yoon, J.-H.; Ahn, S.-G. Surface properties of endosseous dental implants after NdYAG and CO2 laser treatment at various energies. J. Oral Maxillofac. Surg. 2005, 63, 1522–1527. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Heo, S.J.; Koak, J.Y.; Kim, S.K.; Han, C.H.; Lee, J.H. Effects of laser irradiation on machined and anodized titanium disks. Int. J. Oral Maxillofac. Implant. 2012, 27, 265–272. [Google Scholar]
- Romanos, G.; Ko, H.-H.; Froum, S.; Tarnow, D. The use of CO2 laser in the treatment of peri-implantitis. Photomed. Laser Surg. 2009, 27, 381–386. [Google Scholar] [CrossRef] [PubMed]
- Stuebinger, S.; Etter, C.; Miskiewicz, M.; Homann, F.; Saldamli, B.; Wieland, M.; Sader, R. Surface alterations of polished and sandblasted and acid-etched titanium implants after Er:YAG, carbon dioxide, and diode laser irradiation. Int. J. Oral Maxillofac. Implant. 2010, 25, 104–111. [Google Scholar]
- Aoki, A.; Ando, Y.; Watanabe, H.; Ishikawa, I. In vitro studies on laser scaling of subgingival calculus with an erbium:YAG laser. J. Periodontol. 1994, 65, 1097–1106. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, I.; Aoki, A.; Takasaki, A.A. Clinical application of erbium: YAG laser in periodontology. J. Int. Acad. Periodontol. 2008, 10, 22–30. [Google Scholar] [PubMed]
- Schmage, P.; Thielemann, J.; Nergiz, I.; Scorziello, T.M.; Pfeiffer, P. Effects of 10 cleaning instruments on four different implant surfaces. Int. J. Oral Maxillofac. Implant. 2012, 27, 308–317. [Google Scholar]
- Shin, S.-I.; Min, H.-K.; Park, B.-H.; Kwon, Y.-H.; Park, J.-B.; Herr, Y.; Heo, S.-J.; Chung, J.-H. The effect of Er:YAG laser irradiation on the scanning electron microscopic structure and surface roughness of various implant surfaces: An in vitro study. Lasers Med. Sci. 2011, 26, 767–776. [Google Scholar] [CrossRef] [PubMed]
- Galli, C.; Macaluso, G.M.; Elezi, E.; Ravanetti, F.; Cacchioli, A.; Gualini, G.; Passeri, G. The effects of Er:YAG laser treatment on titanium surface profile and osteoblastic cell activity: An in vitro study. J. Periodontol. 2011, 82, 1169–1177. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, Y.; Aoki, A.; Mizutani, K.; Takeuchi, Y.; Ichinose, S.; Takasaki, A.A.; Schwarz, F.; Izumi, Y. Optimal Er:YAG laser irradiation parameters for debridement of microstructured fixture surfaces of titanium dental implants. Lasers Med. Sci. 2013, 28, 1057–1068. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.-I.; Lee, E.-K.; Kim, J.-H.; Lee, J.-H.; Kim, S.-H.; Kwon, Y.-H.; Herr, Y.; Chung, J.-H. The effect of Er:YAG laser irradiation on hydroxyapatite-coated implants and fluoride-modified TiO2-blasted implant surfaces: A microstructural analysis. Lasers Med. Sci. 2013, 28, 823–831. [Google Scholar] [CrossRef] [PubMed]
- Ayobian-Markazi, N.; Karimi, M.; Safar-Hajhosseini, A. Effects of Er:YAG laser irradiation on wettability, surface roughness, and biocompatibility of SLA titanium surfaces: An in vitro study. Lasers Med. Sci. 2015, 30, 561–566. [Google Scholar] [CrossRef] [PubMed]
- Takagi, T.; Aoki, A.; Ichinose, S.; Taniguchi, Y.; Tachikawa, N.; Shinoki, T.; Meinzer, W.; Sculean, A.; Izumi, Y. Effective removal of calcified deposits on microstructured titanium fixture surfaces of dental implants with erbium lasers. J. Periodontol. 2018, 89, 680–690. [Google Scholar] [CrossRef] [PubMed]
- Romanos, G.E.; Everts, H.; Nentwig, G.H. Effects of diode and Nd:YAG laser irradiation on titanium discs: A scanning electron microscope examination. J. Periodontol. 2000, 71, 810–815. [Google Scholar] [CrossRef] [PubMed]
- Castro, G.L.; Gallas, M.; Núñez, I.R.; Borrajo, J.L.L.; Álvarez, J.C.; Varela, L.G. Scanning electron microscopic analysis of diode laser-treated titanium implant surfaces. Photomed. Laser Surg. 2007, 25, 124–128. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, F.; Zanetti, A.L.; Zanetti, R.V.; Martelli, F.S.; Avila-Campos, M.J.; Tomazinho, L.F.; Granjeiro, J.M. Effectiveness of 980-mm diode and 1064-nm extra-long-pulse neodymium-doped yttrium aluminum garnet lasers in implant disinfection. Photomed. Laser Surg. 2010, 28, 273–280. [Google Scholar] [CrossRef] [PubMed]
- Leja, C.; Geminiani, A.; Caton, J.; Romanos, G.E. Thermodynamic effects of laser irradiation of implants placed in bone: An in vitro study. Lasers Med. Sci. 2013, 28, 1435–1440. [Google Scholar] [CrossRef] [PubMed]
- Geminiani, A.; Caton, J.G.; Romanos, G.E. Temperature change during non-contact diode laser irradiation of implant surfaces. Lasers Med. Sci. 2012, 27, 339–342. [Google Scholar] [CrossRef] [PubMed]
- Giannelli, M.; Lasagni, M.; Bani, D. Thermal effects of λ = 808 nm GaAlAs diode laser irradiation on different titanium surfaces. Lasers Med. Sci. 2015, 30, 2341–2352. [Google Scholar] [CrossRef] [PubMed]

| Author and Year of Publication | Original Implant Surface | Animal Model and Area of Implant Insertion | Localization of the Metal Particles | Method of Detection | Metal Detected | Particle Size/Recovered Particle Weight | Particle Geometry |
|---|---|---|---|---|---|---|---|
| Schliephake et al. [43] | Titanium, machined | Minipig mandible | Peri-implant bone and implant surface |
| Titanium particles | 5–30 µm | Solid and leaf-like particles |
| Lungs, liver and kidneys |
| Titanium concentration as ng/mg dry weight of the organ | Kidneys: 2.92 ± 0.69 ng/mg Liver: 11.5 ± 1.35 ng/mg Lungs: 135.7 ± 12.42 ng/mg | - | |||
| Tanaka et al. [5] | TPS | Dog mandible | Implant-bone interface and surrounding bone tissue |
| Titanium particles | 1.8–3.2 µm | - |
| Martini et al. [46] |
| Mongrel sheep femoral and tibial diaphysis |
| (EDS) | Titanium particles | - | - |
| Franchi et al. [47] |
| Sheep femur and tibia |
| SEM | Titanium granules | 3–60 µm | - |
| Wennerberg et al. [50] |
| New Zealand rabbit tibia |
|
| Titanium concentration as ng/mg dry weight of implant | Turned: 206.7 ± 25.2 ng/mg Sandblasted: 210 ± 35.2 ng/mg | - |
| Meyer et al. [60] |
| Minipig mandible |
|
|
| 20 nm to a few microns |
|
| Flatebo et al. [55] |
|
|
|
|
| 140–2300 nm | - |
| Senna et al. [6] |
|
|
|
|
| 10 nm to 20 µm | - |
| Deppe et al. [54] |
|
|
|
|
| - | - |
| Sridhar et al. [56] |
|
|
|
| - | - | - |
| Deppe et al. [51] |
|
|
|
|
| - | - |
| Pettersson et al. [49] |
|
|
|
| Titanium particles were detected |
| - |
| P (Probelem) | Subgroup | P (Population) | I (Interventions) | C (Comparisons) | O (Outcomes) | |
|---|---|---|---|---|---|---|
| The potential sources of titanium particle and ion release are not known or compiled in the literature | Surgical phase | Implant bed preparation | Experimental, animal and human studies | Bone drilling | Implant drills and other implant bed preparation methods |
|
| Implant placement | Experimental, animal and human studies | Implant Insertion | Implant after Insertion |
| ||
| Implant removal | Experimental, animal and human studies | Bone drilling | Other methods for implant removal |
| ||
| Prosthetic phase | Implant abutment connection | Experimental, animal and human studies | Functional load at the implant abutment connection | Type of connection, misfit gap material |
| |
| Maintenance phase | Implant cleaning and decontamination techniques | Experimental, animal and human studies | Implant cleaning, disinfection and polishing | Scaling |
| |
| Ultrasonication | ||||||
| Rubber cups and brushes | ||||||
| Air-polishing | ||||||
| Lasers | ||||||
| Cleaning and antibacterial substances | ||||||
| Author and Year of Publication | Original Implant Surface | Animal Model and Area of Implant Insertion | Localization of the Metal Particles | Method of Detection | Metal Detected | Particle Size/Recovered Particle Weight | Particle Geometry |
|---|---|---|---|---|---|---|---|
| Schliephake et al. [43] | Titanium, machined | Minipig mandible | Peri-implant bone and implant surface |
| Titanium particles | 5–30 µm | Solid and leaf-like particles |
| Lungs, liver and kidneys |
| Titanium concentration as ng/mg dry weight of the organ | Kidneys: 2.92 ± 0.69 ng/mg Liver: 11.5 ± 1.35 ng/mg Lungs: 135.7 ± 12.42 ng/mg | - | |||
| Tanaka et al. [5] | TPS | Dog mandible | Implant-bone interface and surrounding bone tissue |
| Titanium particles | 1.8–3.2 µm | - |
| Martini et al. [46] |
| Mongrel sheep femoral and tibial diaphysis |
| (EDS) | Titanium particles | - | - |
| Franchi et al. [47] |
| Sheep femur and tibia |
| SEM | Titanium granules | 3–60 µm | - |
| Wennerberg et al. [50] |
| New Zealand rabbit tibia |
|
|
| Turned: 206.7 ± 25.2 ng/mg Sandblasted: 210 ± 35.2 ng/mg | - |
| Meyer et al. [60] |
| Minipig mandible |
|
|
| 20 nm to a few microns |
|
| Flatebo et al. [55] |
|
|
|
|
| 140–2300 nm | - |
| Senna et al. [6] |
|
|
|
|
| 10 nm to 20 µm | - |
| Deppe et al. [54] |
|
|
|
|
| - | - |
| Sridhar et al. [56] |
|
|
|
| - | - | - |
| Deppe et al. [51] |
|
|
|
|
| - | - |
| Pettersson et al. [49] |
|
|
|
| Titanium particles were detected |
| - |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Delgado-Ruiz, R.; Romanos, G. Potential Causes of Titanium Particle and Ion Release in Implant Dentistry: A Systematic Review. Int. J. Mol. Sci. 2018, 19, 3585. https://doi.org/10.3390/ijms19113585
Delgado-Ruiz R, Romanos G. Potential Causes of Titanium Particle and Ion Release in Implant Dentistry: A Systematic Review. International Journal of Molecular Sciences. 2018; 19(11):3585. https://doi.org/10.3390/ijms19113585
Chicago/Turabian StyleDelgado-Ruiz, Rafael, and Georgios Romanos. 2018. "Potential Causes of Titanium Particle and Ion Release in Implant Dentistry: A Systematic Review" International Journal of Molecular Sciences 19, no. 11: 3585. https://doi.org/10.3390/ijms19113585
APA StyleDelgado-Ruiz, R., & Romanos, G. (2018). Potential Causes of Titanium Particle and Ion Release in Implant Dentistry: A Systematic Review. International Journal of Molecular Sciences, 19(11), 3585. https://doi.org/10.3390/ijms19113585






