Endometrial Intracrinology: Oestrogens, Androgens and Endometrial Disorders
Abstract
1. What Do We Mean by ‘Intracrinology’?
2. Endometrium—A Sex Hormone-Dependent Multicellular Tissue
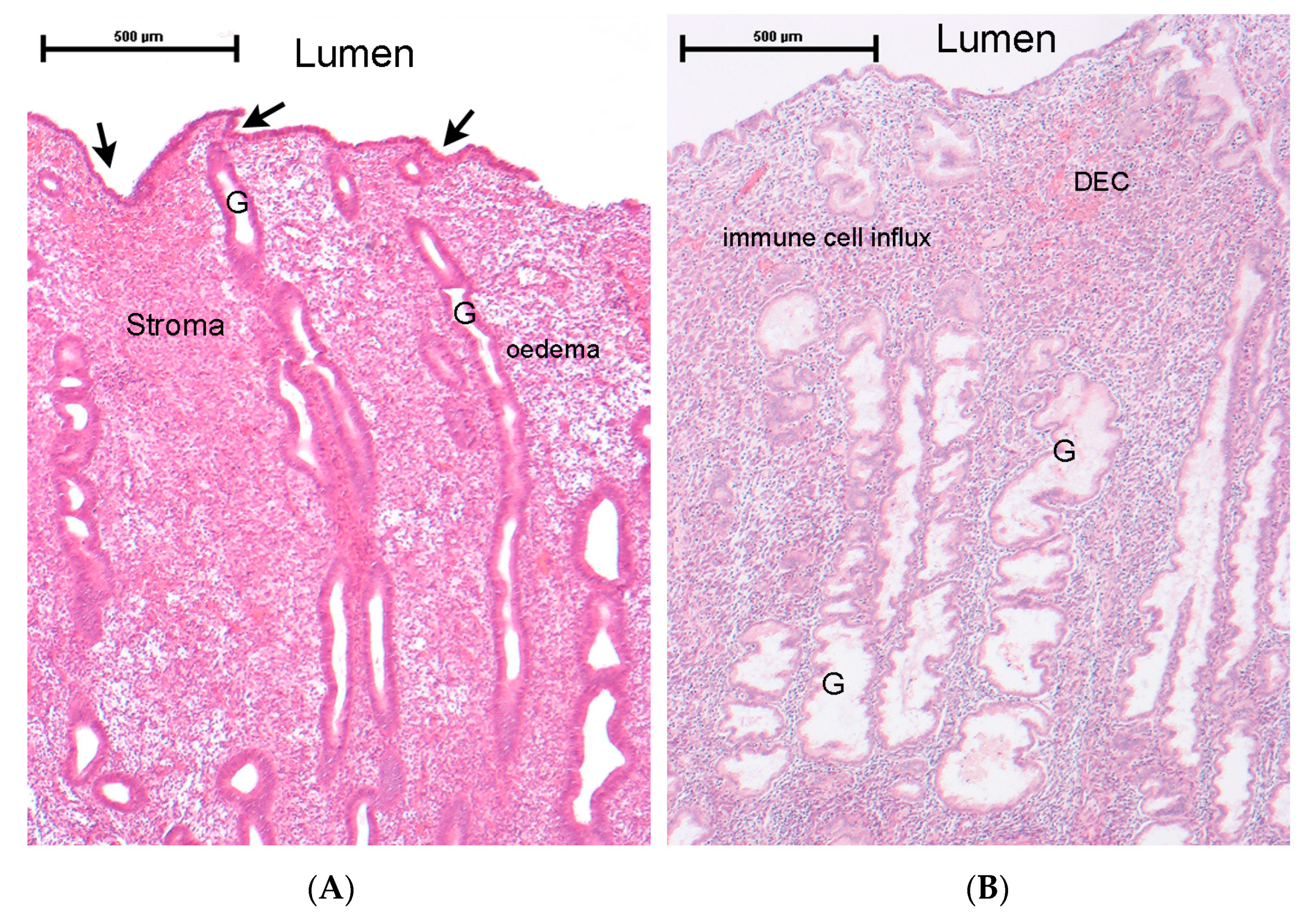
2.1. Endometrial Tissue Structure and Response to Ovarian-Derived Hormones
2.2. Expression of Androgen and Oestrogen Receptors in the Endometrium, Endometriosis and Endometrial Cancer
3. Methodology
4. Intracrine Steroid Biosynthesis in the Normal Endometrium
4.1. Insights Gained from Measurement of Steroid Precursors and Metabolites in Endometrial Tissue
4.2. Androgen Activation and Metabolism
4.3. Oestrogen Biosynthesis and Metabolism
4.4. Role of Sulphated Steroids as a Source of Endometrial Androgens and Oestrogens
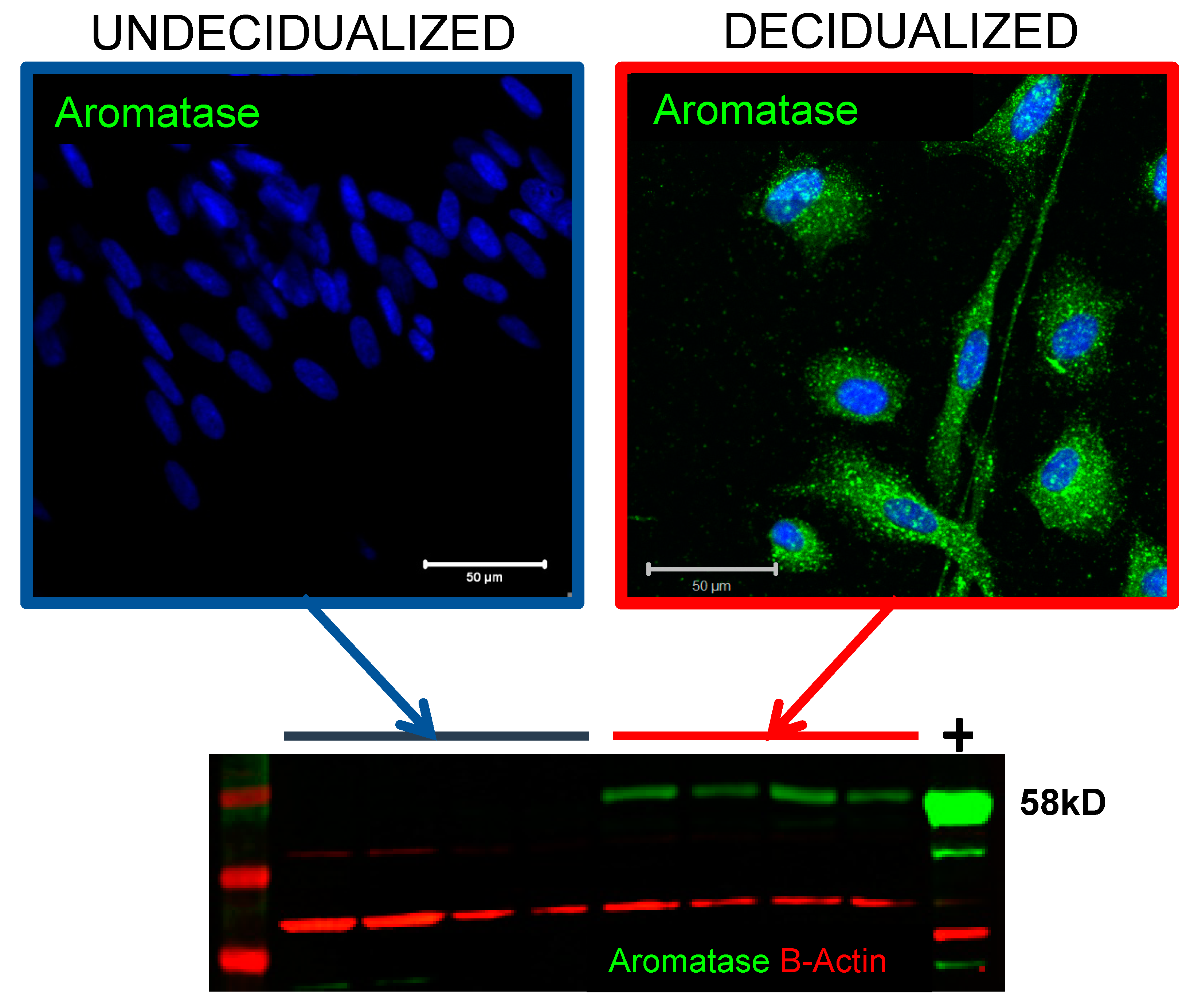
4.5. Intracrine and Paracrine Impact of Tissue Biosynthesis of Androgens and Oestrogens in Endometrium
5. Evidence for the Importance of Intracrinology in Endometrial Disorders
5.1. Endometriosis
5.2. Endometrial Cancer
6. Intracrinology and Metabolism
7. New Therapeutic Approaches for Treatment of Endometrial Disorders Based on Intracrine Targets
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Labrie, F. Intracrinology. Mol. Cell. Endocrinol. 1991, 78, C113–C118. [Google Scholar] [CrossRef]
- Re, R.N. The cellular biology of angiotensin: Paracrine, autocrine and intracrine actions in cardiovascular tissues. J. Mol. Cell. Cardiol. 1989, 21 (Suppl. 5), 63–69. [Google Scholar] [CrossRef]
- Huhtinen, K.; Saloniemi-Heinonen, T.; Keski-Rahkonen, P.; Desai, R.; Laajala, D.; Stahle, M.; Hakkinen, M.R.; Awosanya, M.; Suvitie, P.; Kujari, H.; et al. Intra-tissue steroid profiling indicates differential progesterone and testosterone metabolism in the endometrium and endometriosis lesions. J. Clin. Endocrinol. Metab. 2014, 99, E2188–E2197. [Google Scholar] [CrossRef] [PubMed]
- Huhtinen, K.; Stahle, M.; Perheentupa, A.; Poutanen, M. Estrogen biosynthesis and signaling in endometriosis. Mol. Cell. Endocrinol. 2012, 358, 146–154. [Google Scholar] [CrossRef] [PubMed]
- Labrie, F.; Luu-The, V.; Labrie, C.; Simard, J. DHEA and its transformation into androgens and estrogens in peripheral target tissues: Intracrinology. Front. Neuroendocrinol. 2001, 22, 185–212. [Google Scholar] [CrossRef] [PubMed]
- Labrie, F.; Luu-The, V.; Lin, S.X.; Simard, J.; Labrie, C.; El-Alfy, M.; Pelletier, G.; Belanger, A. Intracrinology: Role of the family of 17 beta-hydroxysteroid dehydrogenases in human physiology and disease. J. Mol. Endocrinol. 2000, 25, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Labrie, F.; Martel, C.; Belanger, A.; Pelletier, G. Androgens in women are essentially made from DHEA in each peripheral tissue according to intracrinology. J. Steroid Biochem. Mol. Biol. 2017, 168, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Moriya, T.; Ishida, T.; Ohuchi, N.; Sasano, H. Intracrine mechanism of estrogen synthesis in breast cancer. Biomed. Pharmacother. 2003, 57, 460–462. [Google Scholar] [CrossRef] [PubMed]
- McNamara, K.M.; Sasano, H. The intracrinology of breast cancer. J. Steroid Biochem. Mol. Biol. 2015, 145, 172–178. [Google Scholar] [CrossRef] [PubMed]
- Smuc, T.; Rupreht, R.; Sinkovec, J.; Adamski, J.; Rizner, T.L. Expression analysis of estrogen-metabolizing enzymes in human endometrial cancer. Mol. Cell. Endocrinol. 2006, 248, 114–117. [Google Scholar] [CrossRef] [PubMed]
- Ito, K.; Miki, Y.; Suzuki, T.; McNamara, K.M.; Sasano, H. In situ androgen and estrogen biosynthesis in endometrial cancer: Focus on androgen actions and intratumoral production. Endocr.-Relat. Cancer 2016, 23, R323–R335. [Google Scholar] [CrossRef] [PubMed]
- Rizner, T.L. Estrogen biosynthesis, phase I and phase II metabolism, and action in endometrial cancer. Mol. Cell. Endocrinol. 2013, 381, 124–139. [Google Scholar] [CrossRef] [PubMed]
- Gibson, D.A.; McInnes, K.J.; Critchley, H.O.; Saunders, P.T. Endometrial Intracrinology—Generation of an estrogen-dominated microenvironment in the secretory phase of women. J. Clin. Endocrinol. Metab. 2013, 98, E1802–E1806. [Google Scholar] [CrossRef] [PubMed]
- Rizner, T.L. Estrogen metabolism and action in endometriosis. Mol. Cell. Endocrinol. 2009, 307, 8–18. [Google Scholar] [CrossRef] [PubMed]
- DallenbachHellweg, G.; Schmidt, D.; Dallenbach, F. Atlas of Endometrial Histopathology, 3rd ed.; Springer: Berlin/Heidelberg, Germany, 2010; 245p. [Google Scholar]
- Critchley, H.O.; Saunders, P.T. Hormone receptor dynamics in a receptive human endometrium. Reprod. Sci. 2009, 16, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Thiruchelvam, U.; Dransfield, I.; Saunders, P.T.; Critchley, H.O. The importance of the macrophage within the human endometrium. J. Leukocyte Biol. 2013, 93, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Henderson, T.A.; Saunders, P.T.; Moffett-King, A.; Groome, N.P.; Critchley, H.O. Steroid receptor expression in uterine natural killer cells. J. Clin. Endocrinol. Metab. 2003, 88, 440–449. [Google Scholar] [CrossRef] [PubMed]
- Wilkens, J.; Male, V.; Ghazal, P.; Forster, T.; Gibson, D.A.; Williams, A.R.; Brito-Mutunayagam, S.L.; Craigon, M.; Lourenco, P.; Cameron, I.T.; et al. Uterine NK cells regulate endometrial bleeding in women and are suppressed by the progesterone receptor modulator asoprisnil. J. Immunol. 2013, 191, 2226–2235. [Google Scholar] [CrossRef] [PubMed]
- Moffett-King, A. Natural killer cells and pregnancy. Nat. Rev. Immunol. 2002, 2, 656–663. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.; Salamonsen, L.A. Inflammation, leukocytes and menstruation. Rev. Endocr. Metab. Disord. 2012, 13, 277–288. [Google Scholar] [CrossRef] [PubMed]
- Maybin, J.A.; Critchley, H.O. Menstrual physiology: Implications for endometrial pathology and beyond. Hum. Reprod. Update 2015, 21, 748–761. [Google Scholar] [CrossRef] [PubMed]
- Noyes, R.W.; Hertig, A.T.; Rock, J. Dating the endometrial biopsy. Fertil. Steril. 1950, 1, 3–25. [Google Scholar] [CrossRef]
- Garry, R.; Hart, R.; Karthigasu, K.A.; Burke, C. Structural changes in endometrial basal glands during menstruation. BJOG 2011, 117, 1175–1185. [Google Scholar] [CrossRef] [PubMed]
- Gibson, D.A.; Saunders, P.T. Estrogen dependent signaling in reproductive tissues—A role for estrogen receptors and estrogen related receptors. Mol. Cell. Endocrinol. 2012, 348, 361–372. [Google Scholar] [CrossRef] [PubMed]
- Gibson, D.A.; Simitsidellis, I.; Collins, F.; Saunders, P.T. Evidence of androgen action in endometrial and ovarian cancers. Endocr. Relat. Cancer 2014, 21, T203–T218. [Google Scholar] [CrossRef] [PubMed]
- Saunders, P.T.K.; Millar, M.R.; Williams, K.; Macpherson, S.; Harkiss, D.; Anderson, R.A.; Orr, B.; Groome, N.P.; Scobie, G.; Fraser, H.M. Differential expression of estrogen receptor-α and -β and androgen receptor in the ovaries of marmosets and humans. Biol. Reprod. 2000, 63, 1098–1105. [Google Scholar] [CrossRef] [PubMed]
- Marshall, E.; Lowrey, J.; Macpherson, S.; Maybin, J.A.; Collins, F.; Critchley, H.O.; Saunders, P.T. In silico analysis identifies a novel role for androgens in the regulation of human endometrial apoptosis. J. Clin. Endocrinol. Metab. 2011, 96, E1746–E1755. [Google Scholar] [CrossRef] [PubMed]
- Critchley, H.O.; Brenner, R.M.; Henderson, T.A.; Williams, K.; Nayak, N.R.; Slayden, O.D.; Millar, M.R.; Saunders, P.T. Estrogen receptor β, but not estrogen receptor α, is present in the vascular endothelium of the human and nonhuman primate endometrium. J. Clin. Endocrinol. Metab. 2001, 86, 1370–1378. [Google Scholar] [PubMed]
- Whitaker, L.H.; Murray, A.A.; Matthews, R.; Shaw, G.; Williams, A.R.; Saunders, P.T.; Critchley, H.O. Selective progesterone receptor modulator (SPRM) ulipristal acetate (UPA) and its effects on the human endometrium. Hum. Reprod. 2017, 32, 531–543. [Google Scholar] [CrossRef] [PubMed]
- Turner, K.J.; Macpherson, S.; Millar, M.R.; McNeilly, A.S.; Williams, K.; Cranfield, M.; Groome, N.P.; Sharpe, R.M.; Fraser, H.M.; Saunders, P.T. Development and validation of a new monoclonal antibody to mammalian aromatase. J. Endocrinol. 2002, 172, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Winuthayanon, W.; Lierz, S.L.; Delarosa, K.C.; Sampels, S.R.; Donoghue, L.J.; Hewitt, S.C.; Korach, K.S. Juxtacrine Activity of Estrogen Receptor alpha in Uterine Stromal Cells is Necessary for Estrogen-Induced Epithelial Cell Proliferation. Sci. Rep. 2017, 7, 8377. [Google Scholar] [CrossRef] [PubMed]
- Dupont, S.; Krust, A.; Gansmuller, A.; Dierich, A.; Chambon, P.; Mark, M. Effect of single and compound knockouts of estrogen receptors alpha (ERalpha) and beta (ERbeta) on mouse reproductive phenotypes. Development 2000, 127, 4277–4291. [Google Scholar] [PubMed]
- Critchley, H.O.; Henderson, T.A.; Kelly, R.W.; Scobie, G.S.; Evans, L.R.; Groome, N.P.; Saunders, P.T. Wild-type estrogen receptor (ERbeta1) and the splice variant (ERbetacx/beta2) are both expressed within the human endometrium throughout the normal menstrual cycle. J. Clin. Endocrinol. Metab. 2002, 87, 5265–5273. [Google Scholar] [CrossRef] [PubMed]
- Bulun, S.E.; Monsavais, D.; Pavone, M.E.; Dyson, M.; Xue, Q.; Attar, E.; Tokunaga, H.; Su, E.J. Role of estrogen receptor-beta in endometriosis. Semin. Reprod. Med. 2012, 30, 39–45. [Google Scholar] [PubMed]
- Hausknecht, V.; de la Osa, E.L.; Gurpide, E. In vitro metabolism of C19 steroids in human endometrium. J. Steroid Biochem. 1982, 17, 621–629. [Google Scholar] [CrossRef]
- Keski-Rahkonen, P.; Huhtinen, K.; Desai, R.; Harwood, D.T.; Handelsman, D.J.; Poutanen, M.; Auriola, S. LC-MS analysis of estradiol in human serum and endometrial tissue: Comparison of electrospray ionization, atmospheric pressure chemical ionization and atmospheric pressure photoionization. J. Mass Spectrom. 2013, 48, 1050–1058. [Google Scholar] [CrossRef] [PubMed]
- Burger, H.G. Androgen production in women. Fertil. Steril. 2002, 77 (Suppl. 4), S3–S5. [Google Scholar] [CrossRef]
- Simitsidellis, I.; Saunders, P.T.K.; Gibson, D.A. Androgens and endometrium: New insights and new targets. Mol. Cell. Endocrinol. 2018, 465, 48–60. [Google Scholar] [CrossRef] [PubMed]
- Gibson, D.A.; Simitsidellis, I.; Kelepouri, O.; Critchley, H.O.D.; Saunders, P.T.K. Dehydroepiandrosterone enhances decidualization in women of advanced reproductive age. Fertil. Steril. 2018, 109, 728–734. [Google Scholar] [CrossRef] [PubMed]
- Rhee, H.S.; Oh, S.H.; Ko, B.J.; Han, D.M.; Jeon, B.H.; Park, H.; Moon, H.B.; Kim, W.S. Expression of 3β-hydroxysteroid dehydrogenase and P450 side chain cleavage enzyme in the human uterine endometrium. Exp. Mol. Med. 2003, 35, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Catalano, R.D.; Wilson, M.R.; Boddy, S.C.; Jabbour, H.N. Comprehensive expression analysis of prostanoid enzymes and receptors in the human endometrium across the menstrual cycle. Mol. Hum. Reprod. 2011, 17, 182–192. [Google Scholar] [CrossRef] [PubMed]
- Gibson, D.A.; Simitsidellis, I.; Cousins, F.L.; Critchley, H.O.; Saunders, P.T. Intracrine Androgens Enhance Decidualization and Modulate Expression of Human Endometrial Receptivity Genes. Sci. Rep. 2016, 6, 19970. [Google Scholar] [CrossRef] [PubMed]
- Ito, K.; Utsunomiya, H.; Suzuki, T.; Saitou, S.; Akahira, J.; Okamura, K.; Yaegashi, N.; Sasano, H. 17β-hydroxysteroid dehydrogenases in human endometrium and its disorders. Mol. Cell. Endocrinol. 2006, 248, 136–140. [Google Scholar] [CrossRef] [PubMed]
- Mustonen, M.V.; Isomaa, V.V.; Vaskivuo, T.; Tapanainen, J.; Poutanen, M.H.; Stenback, F.; Vihko, R.K.; Vihko, P.T. Human 17β-hydroxysteroid dehydrogenase type 2 messenger ribonucleic acid expression and localization in term placenta and in endometrium during the menstrual cycle. J. Clin. Endocrinol. Metab. 1998, 83, 1319–1324. [Google Scholar] [CrossRef] [PubMed]
- Sivik, T.; Gunnarsson, C.; Fornander, T.; Nordenskjold, B.; Skoog, L.; Stal, O.; Jansson, A. 17β-Hydroxysteroid dehydrogenase type 14 is a predictive marker for tamoxifen response in oestrogen receptor positive breast cancer. PLoS ONE 2012, 7, e40568. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.L.; Huang, Y.W.; Lin, S.X. Purification, reconstitution, and steady-state kinetics of the trans-membrane 17β-hydroxysteroid dehydrogenase 2. J. Biol. Chem. 2002, 277, 22123–22130. [Google Scholar] [CrossRef] [PubMed]
- Gibson, D.A.; Foster, P.A.; Simitsidellis, I.; Critchley, H.O.D.; Kelepouri, O.; Collins, F.; Saunders, P.T.K. Sulfation Pathways: A role for steroid sulphatase in intracrine regulation of endometrial decidualisation. J. Mol. Endocrinol. 2018, 61, M57–M65. [Google Scholar] [CrossRef] [PubMed]
- Das, A.; Mantena, S.R.; Kannan, A.; Evans, D.B.; Bagchi, M.K.; Bagchi, I.C. De novo synthesis of estrogen in pregnant uterus is critical for stromal decidualization and angiogenesis. Proc. Natl. Acad. Sci. USA 2009, 106, 12542–12547. [Google Scholar] [CrossRef] [PubMed]
- Das, A.; Li, Q.; Laws, M.J.; Kaya, H.; Bagchi, M.K.; Bagchi, I.C. Estrogen-induced expression of Fos-related antigen 1 (FRA-1) regulates uterine stromal differentiation and remodeling. J. Biol. Chem. 2012, 287, 19622–19630. [Google Scholar] [CrossRef] [PubMed]
- Dassen, H.; Punyadeera, C.; Kamps, R.; Delvoux, B.; Van Langendonckt, A.; Donnez, J.; Husen, B.; Thole, H.; Dunselman, G.; Groothuis, P. Estrogen metabolizing enzymes in endometrium and endometriosis. Hum. Reprod. 2007, 22, 3148–3158. [Google Scholar] [CrossRef] [PubMed]
- Sinreih, M.; Knific, T.; Anko, M.; Hevir, N.; Vouk, K.; Jerin, A.; Frkovic Grazio, S.; Rizner, T.L. The Significance of the Sulfatase Pathway for Local Estrogen Formation in Endometrial Cancer. Front. Pharmacol. 2017, 8, 368. [Google Scholar] [CrossRef] [PubMed]
- Mueller, J.W.; Gilligan, L.C.; Idkowiak, J.; Arlt, W.; Foster, P.A. The Regulation of Steroid Action by Sulfation and Desulfation. Endocr. Rev. 2015, 36, 526–563. [Google Scholar] [CrossRef] [PubMed]
- Rubin, G.L.; Harrold, A.J.; Mills, J.A.; Falany, C.N.; Coughtrie, M.W. Regulation of sulphotransferase expression in the endometrium during the menstrual cycle, by oral contraceptives and during early pregnancy. Mol. Hum. Reprod. 1999, 5, 995–1002. [Google Scholar] [CrossRef] [PubMed]
- Greaves, E.; Temp, J.; Esnal-Zufiurre, A.; Mechsner, S.; Horne, A.W.; Saunders, P.T. Estradiol Is a Critical Mediator of Macrophage-Nerve Cross Talk in Peritoneal Endometriosis. Am. J. Pathol. 2015, 185, 2286–2297. [Google Scholar] [CrossRef] [PubMed]
- De Leo, B.; Esnal-Zufiaurre, A.; Collins, F.; Critchley, H.O.D.; Saunders, P.T.K. Immunoprofiling of human uterine mast cells identifies three phenotypes and expression of ERβ and glucocorticoid receptor. F1000Research 2017, 6, 667. [Google Scholar] [CrossRef] [PubMed]
- Gibson, D.A.; Greaves, E.; Critchley, H.O.; Saunders, P.T. Estrogen-dependent regulation of human uterine natural killer cells promotes vascular remodelling via secretion of CCL2. Hum. Reprod. 2015, 30, 1290–1301. [Google Scholar] [CrossRef] [PubMed]
- Greaves, E.; Collins, F.; Critchley, H.O.; Saunders, P.T. ERbeta-dependent effects on uterine endothelial cells are cell specific and mediated via Sp1. Hum. Reprod. 2013, 28, 2490–2501. [Google Scholar] [CrossRef] [PubMed]
- Gibson, D.A.; Simitsidellis, I.; Saunders, P.T. Regulation of androgen action during establishment of pregnancy. J. Mol. Endocrinol. 2016, 57, R35–R47. [Google Scholar] [CrossRef] [PubMed]
- Cloke, B.; Christian, M. The role of androgens and the androgen receptor in cycling endometrium. Mol. Cell. Endocrinol. 2012, 358, 166–175. [Google Scholar] [CrossRef] [PubMed]
- Cloke, B.; Huhtinen, K.; Fusi, L.; Kajihara, T.; Yliheikkila, M.; Ho, K.K.; Teklenburg, G.; Lavery, S.; Jones, M.C.; Trew, G.; et al. The androgen and progesterone receptors regulate distinct gene networks and cellular functions in decidualizing endometrium. Endocrinology 2008, 149, 4462–4474. [Google Scholar] [CrossRef] [PubMed]
- Cousins, F.L.; Kirkwood, P.M.; Murray, A.A.; Collins, F.; Gibson, D.A.; Saunders, P.T. Androgens regulate scarless repair of the endometrial “wound” in a mouse model of menstruation. FASEB J. 2016, 30, 2802–2811. [Google Scholar] [CrossRef] [PubMed]
- Rothman, M.S.; Carlson, N.E.; Xu, M.; Wang, C.; Swerdloff, R.; Lee, P.; Goh, V.H.; Ridgway, E.C.; Wierman, M.E. Reexamination of testosterone, dihydrotestosterone, estradiol and estrone levels across the menstrual cycle and in postmenopausal women measured by liquid chromatography-tandem mass spectrometry. Steroids 2011, 76, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Vercellini, P.; Vigano, P.; Somigliana, E.; Fedele, L. Endometriosis: Pathogenesis and treatment. Nat. Rev. Endocrinol. 2014, 10, 261–275. [Google Scholar] [CrossRef] [PubMed]
- Foti, P.V.; Farina, R.; Palmucci, S.; Vizzini, I.A.A.; Libertini, N.; Coronella, M.; Spadola, S.; Caltabiano, R.; Iraci, M.; Basile, A.; et al. Endometriosis: Clinical features, MR imaging findings and pathologic correlation. Insights Imaging 2018, 9, 149–172. [Google Scholar] [CrossRef] [PubMed]
- Bulun, S.E.; Lin, Z.; Zhao, H.; Lu, M.; Amin, S.; Reierstad, S.; Chen, D. Regulation of aromatase expression in breast cancer tissue. Ann. N. Y. Acad. Sci. 2009, 1155, 121–131. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.; Farquhar, C. Endometriosis: An overview of Cochrane Reviews. Cochrane Database Syst. Rev. 2014, 3, CD009590. [Google Scholar] [CrossRef] [PubMed]
- Bulun, S.E.; Monsivais, D.; Kakinuma, T.; Furukawa, Y.; Bernardi, L.; Pavone, M.E.; Dyson, M. Molecular biology of endometriosis: From aromatase to genomic abnormalities. Semin. Reprod. Med. 2015, 33, 220–224. [Google Scholar] [PubMed]
- Attar, E.; Bulun, S.E. Aromatase and other steroidogenic genes in endometriosis: Translational aspects. Hum. Reprod. Update 2006, 12, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Cheong, Y.C.; Shelton, J.B.; Laird, S.M.; Richmond, M.; Kudesia, G.; Li, T.C.; Ledger, W.L. IL-1, IL-6 and TNF-α concentrations in the peritoneal fluid of women with pelvic adhesions. Hum. Reprod. 2002, 17, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Skrzypczak, J.; Jedrzejczak, P.; Kasprzak, M.; Puk, E.; Kurpisz, M. Inflammatory cytokines in peritoneal fluid of women with endometriosis. Ann. N. Y. Acad. Sci. 1995, 762, 496–498. [Google Scholar] [CrossRef] [PubMed]
- Noble, L.S.; Takayama, K.; Zeitoun, K.M.; Putman, J.M.; Johns, D.A.; Hinshelwood, M.M.; Agarwal, V.R.; Zhao, Y.; Carr, B.R.; Bulun, S.E. Prostaglandin E2 stimulates aromatase expression in endometriosis-derived stromal cells. J. Clin. Endocrinol. Metab. 1997, 82, 600–666. [Google Scholar] [CrossRef] [PubMed]
- Karck, U.; Reister, F.; Schafer, W.; Zahradnik, H.P.; Breckwoldt, M. PGE2 and PGF2α release by human peritoneal macrophages in endometriosis. Prostaglandins 1996, 51, 49–60. [Google Scholar] [CrossRef]
- Attar, E.; Tokunaga, H.; Imir, G.; Yilmaz, M.B.; Redwine, D.; Putman, M.; Gurates, B.; Attar, R.; Yaegashi, N.; Hales, D.B.; et al. Prostaglandin E2 via steroidogenic factor-1 coordinately regulates transcription of steroidogenic genes necessary for estrogen synthesis in endometriosis. J. Clin. Endocrinol. Metab. 2009, 94, 623–631. [Google Scholar] [CrossRef] [PubMed]
- Crosbie, E.J.; Zwahlen, M.; Kitchener, H.C.; Egger, M.; Renehan, A.G. Body mass index, hormone replacement therapy, and endometrial cancer risk: A meta-analysis. Cancer Epidemiol. Biomarkers Prev. 2010, 19, 3119–3130. [Google Scholar] [CrossRef] [PubMed]
- Sanderson, P.A.; Critchley, H.O.; Williams, A.R.; Arends, M.J.; Saunders, P.T. New concepts for an old problem: The diagnosis of endometrial hyperplasia. Hum. Reprod. Update 2017, 23, 232–254. [Google Scholar] [CrossRef] [PubMed]
- Rizner, T.L.; Thalhammer, T.; Ozvegy-Laczka, C. The Importance of Steroid Uptake and Intracrine Action in Endometrial and Ovarian Cancers. Front. Pharmacol. 2017, 8, 346. [Google Scholar] [CrossRef] [PubMed]
- Simpson, E.R. Aromatase: Biologic relevance of tissue-specific expression. Semin. Reprod. Med. 2004, 22, 11–23. [Google Scholar] [PubMed]
- Sales, K.J.; Jabbour, H.N. Cyclooxygenase enzymes and prostaglandins in pathology of the endometrium. Reproduction 2003, 126, 559–567. [Google Scholar] [CrossRef] [PubMed]
- Bulun, S.E.; Chen, D.; Lu, M.; Zhao, H.; Cheng, Y.; Demura, M.; Yilmaz, B.; Martin, R.; Utsunomiya, H.; Thung, S.; et al. Aromatase excess in cancers of breast, endometrium and ovary. J. Steroid Biochem. Mol. Biol. 2007, 106, 81–96. [Google Scholar] [CrossRef] [PubMed]
- Sasano, H.; Kaga, K.; Sato, S.; Yajima, A.; Nagura, H.; Harada, N. Aromatase cytochrome P450 gene expression in endometrial carcinoma. Br. J. Cancer 1996, 74, 1541–1544. [Google Scholar] [CrossRef] [PubMed]
- Utsunomiya, H.; Ito, K.; Suzuki, T.; Kitamura, T.; Kaneko, C.; Nakata, T.; Niikura, H.; Okamura, K.; Yaegashi, N.; Sasano, H. Steroid sulfatase and estrogen sulfotransferase in human endometrial carcinoma. Clin. Cancer Res. 2004, 10, 5850–5856. [Google Scholar] [CrossRef] [PubMed]
- Utsunomiya, H.; Suzuki, T.; Kaneko, C.; Takeyama, J.; Nakamura, J.; Kimura, K.; Yoshihama, M.; Harada, N.; Ito, K.; Konno, R.; et al. The analyses of 17β-hydroxysteroid dehydrogenase isozymes in human endometrial hyperplasia and carcinoma. J. Clin. Endocrinol. Metab. 2001, 86, 3436–3443. [Google Scholar] [PubMed]
- Cornel, K.M.; Krakstad, C.; Delvoux, B.; Xanthoulea, S.; Jori, B.; Bongers, M.Y.; Konings, G.F.; Kooreman, L.F.; Kruitwagen, R.F.; Salvesen, H.B.; et al. High mRNA levels of 17β-hydroxysteroid dehydrogenase type 1 correlate with poor prognosis in endometrial cancer. Mol. Cell. Endocrinol. 2017, 442, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Segawa, T.; Shozu, M.; Murakami, K.; Kasai, T.; Shinohara, K.; Nomura, K.; Ohno, S.; Inoue, M. Aromatase expression in stromal cells of endometrioid endometrial cancer correlates with poor survival. Clin. Cancer Res. 2005, 11, 2188–2194. [Google Scholar] [CrossRef] [PubMed]
- Kamal, A.M.; Bulmer, J.N.; DeCruze, S.B.; Stringfellow, H.F.; Martin-Hirsch, P.; Hapangama, D.K. Androgen receptors are acquired by healthy postmenopausal endometrial epithelium and their subsequent loss in endometrial cancer is associated with poor survival. Br. J. Cancer 2016, 114, 688–696. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, S.; Miki, Y.; Hashimoto, C.; Takagi, K.; Doe, Z.; Li, B.; Yaegashi, N.; Suzuki, T.; Ito, K. The role of 5α-reductase type 1 associated with intratumoral dihydrotestosterone concentrations in human endometrial carcinoma. Mol. Cell. Endocrinol. 2015, 401, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Schiffer, L.; Arlt, W.; Storbeck, K.H. Intracrine androgen biosynthesis, metabolism and action revisited. Mol. Cell. Endocrinol. 2018, 465, 4–26. [Google Scholar] [CrossRef] [PubMed]
- Dubois, V.; Laurent, M.; Boonen, S.; Vanderschueren, D.; Claessens, F. Androgens and skeletal muscle: Cellular and molecular action mechanisms underlying the anabolic actions. Cell. Mol. Life Sci. 2012, 69, 1651–1667. [Google Scholar] [CrossRef] [PubMed]
- Dubois, V.; Laurent, M.R.; Sinnesael, M.; Cielen, N.; Helsen, C.; Clinckemalie, L.; Spans, L.; Gayan-Ramirez, G.; Deldicque, L.; Hespel, P.; et al. A satellite cell-specific knockout of the androgen receptor reveals myostatin as a direct androgen target in skeletal muscle. FASEB J. 2014, 28, 2979–2994. [Google Scholar] [CrossRef] [PubMed]
- Dalton, J.T.; Barnette, K.G.; Bohl, C.E.; Hancock, M.L.; Rodriguez, D.; Dodson, S.T.; Morton, R.A.; Steiner, M.S. The selective androgen receptor modulator GTx-024 (enobosarm) improves lean body mass and physical function in healthy elderly men and postmenopausal women: Results of a double-blind, placebo-controlled phase II trial. J. Cachexia Sarcopenia Muscle 2011, 2, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Dalton, J.T.; Taylor, R.P.; Mohler, M.L.; Steiner, M.S. Selective androgen receptor modulators for the prevention and treatment of muscle wasting associated with cancer. Curr. Opin. Support. Palliat. Care 2013, 7, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Labrie, F.; Archer, D.F.; Bouchard, C.; Girard, G.; Ayotte, N.; Gallagher, J.C.; Cusan, L.; Baron, M.; Blouin, F.; Waldbaum, A.S.; et al. Prasterone has parallel beneficial effects on the main symptoms of vulvovaginal atrophy: 52-week open-label study. Maturitas 2015, 81, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Martel, C.; Labrie, F.; Archer, D.F.; Ke, Y.; Gonthier, R.; Simard, J.N.; Lavoie, L.; Vaillancourt, M.; Montesino, M.; Balser, J.; et al. Serum steroid concentrations remain within normal postmenopausal values in women receiving daily 6.5 mg intravaginal prasterone for 12 weeks. J. Steroid Biochem. Mol. Biol. 2016, 159, 142–153. [Google Scholar] [CrossRef] [PubMed]
- Labrie, F.; Martel, C. A low dose (6.5 mg) of intravaginal DHEA permits a strictly local action while maintaining all serum estrogens or androgens as well as their metabolites within normal values. Horm. Mol. Biol. Clin. Investig. 2017, 29, 39–60. [Google Scholar] [CrossRef] [PubMed]
- Caruso, S.; Cianci, S.; Vitale, S.G.; Matarazzo, M.G.; Amore, F.F.; Cianci, A. Effects of ultralow topical estriol dose on vaginal health and quality of life in postmenopausal women who underwent surgical treatment for pelvic organ prolapse. Menopause 2017, 24, 900–907. [Google Scholar] [CrossRef] [PubMed]
- Labrie, F. Intracrinology and menopause: The science describing the cell-specific intracellular formation of estrogens and androgens from DHEA and their strictly local action and inactivation in peripheral tissues. Menopause 2018. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Fang, Z.; Suzuki, T.; Sasano, H.; Zhou, J.; Gurates, B.; Tamura, M.; Ferrer, K.; Bulun, S. Regulation of aromatase P450 expression in endometriotic and endometrial stromal cells by CCAAT/enhancer binding proteins (C/EBPs): Decreased C/EBPbeta in endometriosis is associated with overexpression of aromatase. J. Clin. Endocrinol. Metab. 2002, 87, 2336–2345. [Google Scholar] [CrossRef] [PubMed]
- Pavone, M.E.; Bulun, S.E. Aromatase inhibitors for the treatment of endometriosis. Fertil. Steril. 2012, 98, 1370–1379. [Google Scholar] [CrossRef] [PubMed]
- Colette, S.; Donnez, J. Are aromatase inhibitors effective in endometriosis treatment? Expert Opin. Investig. Drugs 2011, 20, 917–931. [Google Scholar] [CrossRef] [PubMed]
- O’Reilly, M.W.; Kempegowda, P.; Walsh, M.; Taylor, A.E.; Manolopoulos, K.N.; Allwood, J.W.; Semple, R.K.; Hebenstreit, D.; Dunn, W.B.; Tomlinson, J.W.; et al. AKR1C3-mediated adipose androgen generation drives lipotoxicity in women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 2017, 102, 3327–3339. [Google Scholar] [CrossRef] [PubMed]
- Day, J.M.; Foster, P.A.; Tutill, H.J.; Parsons, M.F.; Newman, S.P.; Chander, S.K.; Allan, G.M.; Lawrence, H.R.; Vicker, N.; Potter, B.V.; et al. 17β-hydroxysteroid dehydrogenase Type 1, and not Type 12, is a target for endocrine therapy of hormone-dependent breast cancer. Int. J. Cancer 2008, 122, 1931–1940. [Google Scholar] [CrossRef] [PubMed]
- Delvoux, B.; D’Hooghe, T.; Kyama, C.; Koskimies, P.; Hermans, R.J.; Dunselman, G.A.; Romano, A. Inhibition of type 1 17β-hydroxysteroid dehydrogenase impairs the synthesis of 17β-estradiol in endometriosis lesions. J. Clin. Endocrinol. Metab. 2014, 99, 276–284. [Google Scholar] [CrossRef] [PubMed]
- Konings, G.F.; Cornel, K.M.; Xanthoulea, S.; Delvoux, B.; Skowron, M.A.; Kooreman, L.; Koskimies, P.; Krakstad, C.; Salvesen, H.B.; van Kuijk, K.; et al. Blocking 17β-hydroxysteroid dehydrogenase type 1 in endometrial cancer: A potential novel endocrine therapeutic approach. J. Pathol. 2018, 244, 203–214. [Google Scholar] [CrossRef] [PubMed]
- Rizner, T.L. The Important Roles of Steroid Sulfatase and Sulfotransferases in Gynecological Diseases. Front. Pharmacol. 2016, 7, 30. [Google Scholar] [CrossRef] [PubMed]
- Piccinato, C.A.; Neme, R.M.; Torres, N.; Sanches, L.R.; Derogis, P.; Brudniewski, H.F.; Rosa, E.S.J.C.; Ferriani, R.A. Effects of steroid hormone on estrogen sulfotransferase and on steroid sulfatase expression in endometriosis tissue and stromal cells. J. Steroid Biochem. Mol. Biol. 2016, 158, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Purohit, A.; Foster, P.A. Steroid sulfatase inhibitors for estrogen- and androgen-dependent cancers. J. Endocrinol. 2012, 212, 99–110. [Google Scholar] [CrossRef] [PubMed]
- Colette, S.; Defrere, S.; Lousse, J.C.; Van Langendonckt, A.; Gotteland, J.P.; Loumaye, E.; Donnez, J. Inhibition of steroid sulfatase decreases endometriosis in an in vivo murine model. Hum. Reprod. 2011, 26, 1362–1370. [Google Scholar] [CrossRef] [PubMed]
- Salah, M.; Abdelsamie, A.S.; Frotscher, M. First Dual Inhibitors of Steroid Sulfatase (STS) and 17β-Hydroxysteroid Dehydrogenase Type 1 (17beta-HSD1): Designed Multiple Ligands as Novel Potential Therapeutics for Estrogen-Dependent Diseases. J. Med. Chem. 2017, 60, 4086–4092. [Google Scholar] [CrossRef] [PubMed]
- Potter, B.V.L. Sulfation Pathways: Steroid sulphatase inhibition via aryl sulphamates: Clinical progress, mechanism and future prospects. J. Mol. Endocrinol. 2018, 61, T233–T252. [Google Scholar] [CrossRef] [PubMed]
- Ferlita, A.; Battaglia, R.; Andronico, F.; Caruso, S.; Cianci, A.; Purrello, M.; Pietro, C.D. Non-Coding RNAs in Endometrial Physiopathology. Int. J. Mol. Sci. 2018, 19, 2120. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gibson, D.A.; Simitsidellis, I.; Collins, F.; Saunders, P.T.K. Endometrial Intracrinology: Oestrogens, Androgens and Endometrial Disorders. Int. J. Mol. Sci. 2018, 19, 3276. https://doi.org/10.3390/ijms19103276
Gibson DA, Simitsidellis I, Collins F, Saunders PTK. Endometrial Intracrinology: Oestrogens, Androgens and Endometrial Disorders. International Journal of Molecular Sciences. 2018; 19(10):3276. https://doi.org/10.3390/ijms19103276
Chicago/Turabian StyleGibson, Douglas A., Ioannis Simitsidellis, Frances Collins, and Philippa T.K. Saunders. 2018. "Endometrial Intracrinology: Oestrogens, Androgens and Endometrial Disorders" International Journal of Molecular Sciences 19, no. 10: 3276. https://doi.org/10.3390/ijms19103276
APA StyleGibson, D. A., Simitsidellis, I., Collins, F., & Saunders, P. T. K. (2018). Endometrial Intracrinology: Oestrogens, Androgens and Endometrial Disorders. International Journal of Molecular Sciences, 19(10), 3276. https://doi.org/10.3390/ijms19103276





