Enhancing Skin Health: By Oral Administration of Natural Compounds and Minerals with Implications to the Dermal Microbiome
Abstract
1. Introduction
Topical Application of Cosmetics, Cosmeceuticals, and Oral Supplementation via Nutritionals
2. Collagen
2.1. Structure of Collagen
2.2. Sources of Collagen
2.3. Biological/Biomedical Properties of Collagen
2.4. In Vitro Studies: Collagen Enhances Fibroblasts and Extracellular Matrix Proteins and Decreases Metalloproteinases (MMPs)
2.5. Oral Administration and Bioavailability of Collagen: GI Absorption, Distribution into the Bloodstream, and Deposition into the Skin
2.6. Oral Administration of Collagen Improves Skin (and Nail) Parameters in Animal Studies
2.7. Oral Administration of Collagen Improves Skin (and Nail) Parameters in Human Studies
3. Ceramides
3.1. Ceramides: Structure and Sources
3.2. Biological and Biomedical Properties of Ceramides in Skin
3.3. Ceramides—In Vitro and Animal Studies
3.4. Ceramides in Human Skin Research
4. Carotenoids (β-Carotene)
4.1. β-Carotene: Structure and Sources
4.2. Biological and Biomedical Properties of β-Carotene in Skin
4.3. β-Carotene Skin Research
4.4. In Vitro Studies: β-Carotene in Human Skin
4.5. In Vivo Studies: β-Carotene in Human Skin
5. Astaxanthin
5.1. Astaxanthin: Structure and Sources
5.2. Biological and Biomedical Properties of Astaxanthin
5.3. Astaxanthin—Skin Research
5.4. Astaxanthin—Oral Clinical Studies
6. Coenzyme Q10
6.1. Coenzyme Q10 Structure and Sources
6.2. Biological and Biomedical Properties of Coenzyme Q10 in Skin
6.3. In Vivo and In Vivo (Clinical) Studies: Coenzyme Q10 in Human Skin
7. Colostrum—Source and Composition
Colostrum and Human Skin
8. Zinc-Essential Mineral Element: Biological, Biomedical Properties and Sources
Zinc and Human Skin
9. Selenium—Essential Mineral Element: Biological and Biomedical Properties and Sources
Selenium and Human Skin
10. Other Selected Natural Compounds and Vitamin D: Effects on Skin Biology
11. Human Microbiome—History and Background
11.1. The Microbiome, Skin, and Dermal Disorders
11.2. The Microbiome, Inflammation, Chemoprotection, and the Future
12. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| Da | Dalton |
| kDa | kilo Dalton |
| g | gram |
| kg | kilogram |
| L | Liter |
| mol | mole |
| mg | milligram |
| ppm | parts per million |
| μg | microgram |
| μmol | micro mole |
| μM | micro molar |
| 3H | radioactive hydrogen (tritium) |
| 14C | radioactive carbon 14 |
| 5α-DHT | 5α-dihydrotestosterone |
| 5α-Reductase | Steroid enzyme that converts testosterone into 5α-DHT |
| AD | Atopic dermatitis |
| Akt | Protein kinase B (a serine/threonine-specific protein kinase) |
| ALA | Alanine |
| α-MSH | α-melanocyte stimulating hormone |
| AMP | Antimicrobial peptides |
| AP-1 | Activator protein 1 |
| ATP | adenosine triphosphate |
| BPH | Benign prostatic hyperplasia |
| COX | Cyclo-oxygenase |
| DNA | Deoxyribonucleic acid |
| EGF | Epidermal growth factor |
| ERβ | Estrogen receptor β |
| FGF | Fibroblast growth factor |
| GI | Gastrointestinal tract |
| G2 | Gap 2 phase (cell cycle) |
| Glc-Cer | Glucosylceramide |
| Gly | Glycine |
| GPX | Glutathione peroxidase |
| HaCaT | Human keratinoctyes |
| Hyp | Hydroxy proline |
| HMP | Human microbiome project |
| HDL | High-density lipoprotein |
| HO-1 | Heme oxygenase 1 |
| IL | interleukins |
| IgA, IgG, IgM | Immunoglobulin A, G, or M |
| IGF | Insulin-like growth factor |
| KOD | 36. amino acid synthetic collagen. The sequence of the peptide is (Pro-Lys-Gly) (Pro-Hyp-Gly) (Asp-Hyp-Gly), and in single-letter amino acid abbreviation is (P-K-G); (P-O-G) (D-O-G), giving it the name KOD |
| LC | Langerhans cells |
| LDL | Low-density lipoproteins |
| MCH | Major histocompatibility complex |
| MDA | Malondialdehyde |
| MMP | Matrix metalloproteinases |
| NF-κB | Nuclear factor κ-light-chain-enhancer of activated B cells |
| Na2SeO3 | Sodium selenite |
| Nrf2 | Nuclear factor (erythroid-derived 2)-like 2 (basic leucine zipper) |
| PDGF | Platelet derived-growth factor |
| Pro | Proline |
| PRR | Pattern recognition receptor |
| RDA | Recommended dietary allowance |
| SEPW1 | Seleno-protein 1 |
| RNA | Ribonucleic acid |
| ROS | Reactive oxygen species (oxidative stress) |
| RSSC | Residual skin surface components |
| SeMet | Seleno-L-methionine |
| Ser | Serine |
| SIRT | NAD-dependent deacetylase sirtuin-1 |
| TEWL | Trans-epidermal water loss |
| TGF | Transforming growth factor |
| Th17 | Helper T cell 17 |
| TMP | Tissue inhibitor of matrix metalloproteinases |
| TLR | Toll-like receptors |
| TNF-α | Tumor necrosis factor α |
| T-reg | Regulator T cells |
| US | United States |
| USA | United States of America |
| USFDA | United States Federal Drug Administration |
| UV | Ultraviolet |
| ZIP2 | Zinc transporter-2 |
References
- Farage, M.A.; Miller, K.W.; Elsner, P.; Maibach, H.I. Degenerative Changes in Aging Skin. In Textbook of Skin Aging; Farage, M.A., Miller, K.W., Maibach, H.I., Eds.; Springer: Berlin/Heidelberg, Germany, 2017; pp. 15–30. [Google Scholar]
- Kohl, E.; Steinbauer, J.; Landthaler, M.; Szeimies, R.M. Skin aging. J. Eur. Acad. Dermatol. Venereol. 2011, 25, 873–884. [Google Scholar] [CrossRef] [PubMed]
- Lephart, E.D. Equol’s anti-aging effects protect against environmental assaults by increasing skin antioxidant defense and ECM proteins while decreasing oxidative stress and Inflammation. Cosmetics 2018, 5, 16. [Google Scholar] [CrossRef]
- Slominski, A.T.; Zmijewski, M.A.; Plonka, P.M.; Szaflarski, J.P.; Paus, R. How UV light touches the brain and endocrine system through skin, and why. Endocrinol. 2018, 5, 1992–2007. [Google Scholar] [CrossRef] [PubMed]
- Feingold, R.; Denda, M. Regulation of permeability barrier homeostasis. Clin. Dermatol. 2009, 27, 248–251. [Google Scholar]
- Slominski, A.T.; Zmijewski, M.A.; Skobowiat, C.; Zbytek, B.; Slominski, R.M.; Steketee, J.D. Sensing the environment: regulation of local and global homeostasis by the skin’s neuroendocrine system. Adv. Anat. Embryol. Cell Biol. 2012, 212, 1–115. [Google Scholar]
- Krishnamurthy, P.; Wadhwani, A. Antioxidant Enzymes and Human Health, Chapter 1; El-Missiry, M.A., Ed.; Antioxidant Enzyme-InTech Science, Technology & Medicine: Vienna, Austria, 2012; pp. 4–18. [Google Scholar]
- Shindo, Y.; Witt, E.; Han, D.; Epstein, W.; Packer, L. Enzymic and non-enzymic antioxidants in epidermis and dermis in human skin. J. Invest. Dermatol. 1994, 102, 122–124. [Google Scholar] [CrossRef] [PubMed]
- Lephart, E.D. Skin aging and oxidative stress: Equol’s anti-aging effects via biochemical and molecular mechanisms. Ageing Res. Rev. 2016, 31, 36–54. [Google Scholar] [CrossRef] [PubMed]
- Rinnerthaler, M.; Bischof, J.; Streubi, M.K.; Trost, A.; Richter, K. Oxidative stress in aging human skin. Biomolecules 2015, 5, 545–589. [Google Scholar] [CrossRef] [PubMed]
- Chaudhri, S.K.; Jain, N.K. History of cosmetics. Asian J. Pharmaceutics 2009, 3, 164–167. [Google Scholar]
- Kligman, A. The future of cosmeceuticals: An interview with Albert Kligman, MD, Ph.D. Interviewed by Dr. Zoe Diana Draelos. Dermatol. Surg. 2005, 31, 890–981. [Google Scholar] [PubMed]
- Schagen, S.K.; Zampeli, V.A.; Makrantonaki, E.; Zouboulis, C.C. Discovering the link between nutrition and skin aging. Dermato-Endocrinol. 2012, 4, 298–307. [Google Scholar] [CrossRef] [PubMed]
- Moyad, M.; Lee, J. The Supplement Handbook: A Trusted Expert’s Guide to What Works & What’s Worthless for More than 100 Conditions; Rodale Books Press: New York, NY, USA, 2014. [Google Scholar]
- Di Cerbo, A.; Laurino, C.; Palmieri, B.; Iannitti, T.A. Dietary supplement improves facial photoaging and skin sebum, hydration and tonicity modulating serum fibronectin, neutrophil elastase 2, hyaluronic acid and carbonylated proteins. J. Photochem. Photobiol. B-Biology 2015, 144, 94–103. [Google Scholar] [CrossRef] [PubMed]
- Costa, A.; Pereira, E.S.P.; Assumpcao, E.C.; dos Santos, F.B.C.; Ota, F.S.; Pereira, M.D.; Favaro, F.; Langen, S.S.B.; de Arruda, L.H.F. Assessment of clinical effects and safety of an oral supplement based on marine protein, vitamin C, grape seed extract, zinc, and tomatoextract in the improvement of visible signs of skin aging in men. Clin. Cosmetic Investig. Derm. 2015, 8, 319–328. [Google Scholar] [CrossRef] [PubMed]
- Meinke, M.C.; Nowbary, C.K.; Schanzer, S.; Vollert, H.; Lademan, J.; Darvin, M.E. Influences of orally taken carotenoid-rich curly kale on collagen I/elastin index of the skin. Nutrients 2017, 9, 775. [Google Scholar] [CrossRef] [PubMed]
- Ricard-Blum, S. The collagen family. Cold Springs Har. Perspect. Biol. 2011, 3, a004978. [Google Scholar] [CrossRef]
- Wyckoff, R.; Corey, R.; Biscoe, J. X-ray reflections of long spacing from tendon. Science 1935, 82, 175. [Google Scholar] [CrossRef] [PubMed]
- Clark, G.; Parker, E.; Schaad, J.; Warren, W.J. New measurements of previously unknown large interplanar spacings in natural materials. J. Am. Chem. Soc. 1935, 57, 1509. [Google Scholar] [CrossRef]
- Rodriguez, M.I.A.; Barroso, L.G.R.; Sanchez, M.L. Collagen: A review on it sources and potential cosmetic applications. J. Cosmet. Dermatol. 2017, 17, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Nikoo, M.; Boran, G.; Zhou, P. Regenstein JM. Collagen and Gelatin. Annu. Rev. Food Sci. Tech. 2015, 6, 527–557. [Google Scholar] [CrossRef] [PubMed]
- Zague, V.; Bussodor do Amaral, J.; Rezende-Texeria, P.; de Oliveira Niero, E.; Lauand, C.; Machado-Santelli, G.M. Collagen peptides modulate the metabolism of extracellular matrix by human dermal fibroblasts derived from sun-protected and sun-exposed body sites. Cell Biol. Int. 2018, 42, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Oesser, S.; Adam, M.; Babel, W.; Seifert, J. Oral administration of 14 C labeled gelatin hydrolysate leads to an accumulation of radioactivity in cartilage in mice (C57/BL). J. Nutr. 1999, 129, 1891–1895. [Google Scholar] [CrossRef] [PubMed]
- Ohara, H.; Matsumoto, H.; Ito, K.; Iwai, K.; Sato, K. Comparisons of quantity and structures of hydroxyproline containing peptides in human blood after oral ingestion of gelatin hydrolysates from different sources. J. Agr. Food Chem. 2007, 55, 1532–1535. [Google Scholar] [CrossRef] [PubMed]
- Borumand, M.; Sibilla, S. Daily consumption of the collagen supplement Pure Gold Collagen(R), reduces visible signs of aging. Clin. Interv. Aging 2014, 9, 1747–1758. [Google Scholar] [PubMed]
- Yazaki, M.; Ito, Y.; Yamada, M.; Goulas, S.; Teramoto, S.; Nakaya, M.A.; Ohno, S.; Yamajuchi, K. Oral ingestion of collagen hydrolysate leads to the transportation of highly concentrated Gly-Pro-Hyp and its hydrolyzed form of Pro-Hyp into the bloodstream and skin. J. Agr. Food Chem. 2017, 65, 2315–2322. [Google Scholar] [CrossRef] [PubMed]
- Hakuta, A.; Yamaguchi, Y.; Okawa, T.; Yamamoto, S.; Sakai, Y.; Aihara, M. Anti-inflammatory effect of collagen tripeptide in atopic dermatitis. J. Dermatol. Sci. 2017, 88, 357–364. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, N.; Koyama, Y.; Hosaka, Y.; Udea, H.; Watanabe, T. Effects of ingestion of collagen peptide on collagen fibrils and gylcoaminoglycans in the dermis. J. Nutr. Sci. Vita. 2006, 52, 211–215. [Google Scholar] [CrossRef]
- Tanka, M.; Koyama, Y.; Nomura, Y. Effects of collagen peptide ingestion on UV-B-induced skin damage. Biosci. Biotechnol. Biochem. 2009, 73, 930–932. [Google Scholar] [CrossRef] [PubMed]
- Watanabe-Kamiyama, M.; Shimizu, M.; Kamiyama, S.; Taguchi, Y.; Sone, H. Absorption and effectiveness of orally administered low molecular weight collagen hydrolysate in rats. J. Agr. Food Chem. 2010, 58, 835–841. [Google Scholar] [CrossRef] [PubMed]
- Zague, V.; de Freitas, V.; Rosa, M.D.; de Castro, G.A.; Jaeger, R.G.; Machado-Santelli, G.M. Collagen hydrolysate intake increased skin collagen expression and suppresses matrix metalloproteinase 2 activity. J. Medicinal Food 2011, 14, 618–624. [Google Scholar] [CrossRef] [PubMed]
- Okawa, T.; Yamaguchi, Y.; Takada, S.; Sakai, Y.; Numata, N.; Nakamura, F.; Nagashima, Y.; Ikezawa, Z.; Aihara, M. Oral administration of collagen tripeptide improves dryness and pruritus in the acetoneinduced dry skin model. J. Dermatol. Sci. 2012, 66, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Song, H.D.; Meng, M.F.; Cheng, X.F.; Wang, C.T. The effect of collagen hydrolysates from silver carp (Hypophthalmichthys molitrix) skin on UV-induced photoaging in mice; molecular weight affects skin repair. Food Func. 2017, 8, 1538–1546. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.R.; Hou, H.; Wang, S.K.; Zhao, X.; Li, B.F. Effects of early enteral nutrition supplement with collagen peptides on post-burn inflammatory responses in a mouse model. Food Func. 2017, 8, 1933–1941. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Zhang, L.; Luo, Y.; Zhang, S.; Li, B. Effects of collagen peptides intake on skin ageing and platelet release in chronologically aged mice revealed by cytokine array analysis. J. Cell Mole. Med. 2018, 22, 277–288. [Google Scholar] [CrossRef] [PubMed]
- Iwai, K.; Hasegawa, T.; Taguchi, Y.; Morimastu, F.; Sato, K. Identification of food-derived collagen peptides in human blood after oral ingestion of gelatin hydroylsates. J. Agr. Food Chem. 2005, 53, 6531–6536. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, H.; Ohara, H.; Itoh, K.; Nakamura, Y.; Takahashi, S. Clinical effect of fish type I collagen hydrolysate on skin properties. ITE Lett. 2006, 7, 386–390. [Google Scholar]
- Schwartz, S.R.; Park, J. Ingestions of BioCell Collagen (R), a novel hydrolyzed chicken sternal cartilage extract; enhanced blood microcirculation and reduced facial aging signs. Clin. Intervent. Aging 2012, 7, 267–273. [Google Scholar]
- Proksch, E.; Segger, D.; Degwert, J.; Schunch, M.; Zague, V.; Oesser, S. Oral supplementation of specific collagen peptides has beneficial effects on human skin physiology: A double-blind, placebo-controlled study. Skin Pharmacol. Physiol. 2014, 27, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Proksch, E.; Schunck, M.; Zague, V.; Segger, D.; Degwart, J.; Oesser, S. Oral intake of specific bioactive collagen peptides reduces skin wrinkles and increases dermal matrix synthesis. Skin Pharmacol. Physiol. 2014, 27, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Lipp, T. Aging beautifully proven-skin health benefits of collagen peptides preclinical and clinical studies substantiate the beneficial effects of orally administered collagen peptides. Agro. Food Ind. Hi-Tech. 2016, 6, 11–14. [Google Scholar]
- Asserin, J.; Lati, E.; Shioya, T.; Prawitt, J. The effects of oral collagen peptide supplementation on skin moisture and the dermal collagen network: evidence from an ex vivo model and randomized, placebo-controlled clinical trials. J. Cosmet. Dermatol. 2015, 14, 291–301. [Google Scholar] [CrossRef] [PubMed]
- Hexsel, D.; Zague, V.; Schunch, M.; Siega, C.; Camozzato, F.O.; Oesser, S. Oral supplementation with specific bioactive collagen peptides improves nail growth and reduces symptoms of brittle nails. J. Cosmet. Dermatol. 2017, 16, 520–526. [Google Scholar] [CrossRef]
- Harding, C.R.; Moore, D.J.; Rawlings, A.V. Ceramides and the skin. In Textbook of Cosmetic Dermatology, 3rd ed.; Baran, R., Maibach, H.I., Eds.; Taylor & Francis: London, UK, 2005; pp. 171–186. [Google Scholar]
- Cha, H.-J.; He, C.; Zhao, H.; Dong, Y.; An, I.-S.; An, S. Intercellular and intracellular functions of ceramides and their metabolites in skin (Review). Int. J. Mole. Med. 2016, 38, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Meckfessel, M.H.; Brandt, S. The structure, function, and importance of ceramides in skin and their use as therapeutic agents in skin-care products. J. Am. Acad. Derm. 2014, 71, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Alonso, A.; Goni, F.M. The physical properties of ceramides in membranes. Annu. Rev. Biophysics 2018, 47, 633–654. [Google Scholar] [CrossRef] [PubMed]
- Castro, B.M.; Prieto, M.; Silva, L.C. Ceramide: A simple sphingolipid with unique biophysical properties. Prog. Lipid Res. 2014, 54, 53–67. [Google Scholar] [CrossRef] [PubMed]
- Uchida, Y.; Kim, Y.-I.; Park, K. Signaling roles of ceramides and its metabolites in cutaneous antimicrobial defense. Dermatol. Sin. 2015, 33, 78–83. [Google Scholar] [CrossRef]
- Ueda, O.; Hasegawa, M.; Kitamura, S. Distribution in skin of ceramide after oral administration to rats. Drug Metab. Pharmacokinet. 2009, 24, 180–184. [Google Scholar] [CrossRef] [PubMed]
- Ueda, O.; Uchiyama, T.; Nakashima, M. Distribution and metabolism of sphingosine in skin after oral administration to mice. Drug Metab. Pharmacokinet. 2010, 25, 456–465. [Google Scholar] [CrossRef] [PubMed]
- Shimoda, H.; Terazawa, S.; Hitoe, S.; Tanaka, J.; Nakumura, S.; Matsuda, H.; Yoshikawa, M. Changes in ceramides and glucosylceramides in mouse skin and human epidermal equivalents by rice-derived glucosylceramide. J. Med. Food 2012, 15, 1064–1072. [Google Scholar] [CrossRef] [PubMed]
- Jennemann, R.; Rabionet, M.; Gorgas, K.; Epstein, S.; Dalpke, A.; Rothermel, U.; Bayerle, A.; van der Hoeven, F.; Imgrund, S.; Kirsch, J.; et al. Loss of ceramide synthease 3 causes lethal skin barrier disruption. Hum. Mole. Genet. 2012, 21, 586–608. [Google Scholar] [CrossRef] [PubMed]
- Kawamura, J.; Kotoura, S.; Okuyama, T.; Furumoto, M.; Fuchuu, H.; Miake, K.; Sugiyama, M.; Ohnishi, M. Effect of oral administration of defatted chicken skin powder on day skin in humans. J. Japanese Soc. Food Sci. Tech. Nippon Shokuhin Kagaku Kogaku Kaishi 2013, 5, 218–224. [Google Scholar] [CrossRef]
- Imokawa, G.; Abe, A.; Jin, K.; Higaki, Y.; Kawashima, M.; Hidano, A. Decreased level of ceramide in stratum corneum of atopic dermatitis: an etiologic factor in atopic dry skin? J. Invest. Dermatol. 1991, 96, 523–536. [Google Scholar] [CrossRef] [PubMed]
- Darlenski, R.; Fluhr, J.W. Influence of skin type, race, sex, and anatomic location on epidermal barrier function. Clin. Dermatol. 2012, 30, 269–273. [Google Scholar] [CrossRef] [PubMed]
- Tessema, E.N.; Gebre-Mariam, T.; Neubert, R.H.H.; Wohlrab, J. Potential applications of phyto-derived ceramides in improving epidermal barrier function. Skin Pharmacol. Physiol. 2017, 30, 115–138. [Google Scholar] [CrossRef] [PubMed]
- Park, J.W.; Park, W.J.; Futerman, A.H. Ceramide synthases as potential targets for therapeutic intervention in human disease. Biochim. Biophys. Acta 2014, 1841, 671–681. [Google Scholar] [CrossRef] [PubMed]
- Levy, M.; Futerman, A.H. Mammalian ceramide synthases. IUBMB Life 2010, 62, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Sourkes, T.L. The discovery and early history of carotene. Bull. Hist. Chem. 2009, 34, 32–38. [Google Scholar]
- Cicero, A.F.G.; Colletti, A. Effects of carotenoids on health: are all the same? Results from clinical trials. Curr. Pharm. Des. 2017, 23, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Garcia, E. Skin protection against UV light by dietary antioxidants. Food Func. 2014, 5, 1994–2003. [Google Scholar] [CrossRef] [PubMed]
- Fiedor, J.; Burda, K. Potential role of carotenoids as antioxidants in human health and disease. Nutrients 2014, 6, 466–488. [Google Scholar] [CrossRef] [PubMed]
- US Department of Agriculture, Agricultural Research Service. USDA Nutrient Database for Standard Reference. Release 28. 2015. [Google Scholar]
- Hendler, S.S.; Rorvik, D.M. PDR for Nutritional Supplements, 2nd ed.; Thomson Reuters Press: Toronto, ON, Canada, 2008. [Google Scholar]
- Davinelli, S.; Nielsen, M.E.; Scapagnini, G. Astaxanthin in skin health, repair, and disease: A comprehensive review. Nutrients 2018, 10, 522. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.-P.; Peng, J.; Yin, K.; Wang, J.-H. Potential health-promoting effects of astaxanthin: A high-value carotenoid mostly from microalgae. Mol. Nutr. Food Res. 2011, 55, 150–165. [Google Scholar] [CrossRef] [PubMed]
- Kidd, P. Astaxanthin, cell membrane nutrient with diverse clinical benefits and anti-aging potential. Altern. Med. Rev. 2011, 16, 355–364. [Google Scholar] [PubMed]
- Crane, F.L.; Hatefi, Y.; Lester, R.; Widmer, C. Isolation of a quinone from beef heart mitochondria. Biochim. Biophys. Acta 1957, 25, 220–221. [Google Scholar] [CrossRef]
- Hargreaves, I.P. Coenzyme Q10 as a therapy for mitochondrial disease. Int. J. Biochem. Cell Biol. 2014, 49, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Guo, X.; Wang, W.; Medeiros, D.; Clarke, S.L.; Lucas, E.A.; Smith, B.J.; Lin, D. Molecular aspects of β, β-carotene-9’, 10’-oxygenase 2 in carotenoid metabolism and disease. Exp. Biol. Med. 2016, 241, 1879–1887. [Google Scholar] [CrossRef] [PubMed]
- Addor, F.A.S. Antioxidants in dermatology. An. Bras. Dermatol. 2017, 92, 356–362. [Google Scholar] [CrossRef] [PubMed]
- Mathews-Roth, M.M.; Pathak, M.A.; Fitzpatrick, T.B.; Harber, L.C.; Kass, E.H. β-Carotene as a photoprotective agent in erythropoietic protoporphyria. New Eng. J. Med. 1970, 282, 1231–1234. [Google Scholar] [CrossRef] [PubMed]
- Mathews-Roth, M.M.; Pathak, M.A.; Parrish, J.; Fitzpatrick, T.B.; Kass, E.H.; Toda, K.; Clemens, W. A clinical trial of the effects of oral β-carotene on the responses of human skin to solar radiation. J. Invest. Derm. 1972, 50, 349–353. [Google Scholar] [CrossRef]
- Mathews-Roth, M.M.; Pathak, U.A.; Fitzpatrick, T.B.; Harber, L.C.; Kass, E.H. β-carotene as an oral photoprotective agent in erythropoietic protophorphyria. JAMA 1974, 228, 1004–1008. [Google Scholar] [CrossRef] [PubMed]
- Grune, T.; Lietz, G.; Palou, A.; Ross, A.C.; Stahl, W.; Tang, G.; Thurnham, D.; Yin, S.; Biesalski, H.K. β-Carotene is an important vitamin A source for humans. J. Nutr. 2010, 140, 2268S–2285S. [Google Scholar] [CrossRef] [PubMed]
- van Het Hof, K.H.; West, C.E.; Weststrate, J.A.; Hautvast, J.G. Dietary factors that affect the bioavailability of carotenoids. J. Nutr. 2000, 130, 503–506. [Google Scholar] [CrossRef] [PubMed]
- Priyadarshani, A.M. A review on factors influencing bioaccessibility and bioefficacy of carotenoids. Crit. Rev. Food Sci. Nutr. 2017, 57, 1710–1717. [Google Scholar] [CrossRef] [PubMed]
- Tanumihardjo, S.A.; Palacios, N.; Pixley, K.V. Provitamin a carotenoid bioavailability:what really matters? Int. J. Vitam. Nutr. Res. 2010, 80, 336–350. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.; Lee, D.H.; Won, C.H.; Kim, S.M.; Lee, S.; Lee, M.L.; Chung, J.H. Differential effects of low-dose and high dose β-carotene supplementation on the signs of photoaging and type I procollagen gene expression in human skin in vivo. Dermatol. 2010, 221, 160–170. [Google Scholar] [CrossRef] [PubMed]
- Richelle, M.; Sabatier, M.; Steiling, H.; Williamson, G. Skin bioavailability of dietary vitamin E, carotenoids, polyphenols, vitamin C, zinc and selenium. Br. J. Nutr. 2006, 96, 227–238. [Google Scholar] [PubMed]
- Meinke, M.C.; Friedrich, A.; Tscherch, K.; Haag, S.F.; Darvin, M.E.; Vollert, H.; Groth, N.; Lademann, J.; Rohn, S. Influence of dietary carotenoids on radical scavenging capacity of the skin and skin lipids. Eur. J. Pharm. Biopharm. 2013, 84, 365–373. [Google Scholar] [CrossRef] [PubMed]
- Gimeno, A.; Zaragoza, R.; Vivo-Sese, I.; Vina, J.R.; Miralles, V.J. Retinol, at concentrations greater than the physiological limit, induces oxidative stress and apoptosis in human dermal fibroblasts. Exp. Derm. 2004, 13, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Lohan, S.B.; Vitt, K.; Scholz, P.; Keck, C.M.; Meinke, M.C. ROS production and glutathione response in keratinocytes after application of β-carotene and VIS/NIR irradiation. Chem. Biol. Interact. 2018, 280, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Trekli, M.C.; Riss, G.; Goralczyk, R.; Tyrrell, R.M. β-carotene suppresses UVA-induced HO1 gene expression in cultured FEK4. Free Rad. Biol. Med. 2003, 34, 456–464. [Google Scholar] [CrossRef]
- Wertz, K.; Hunziker, P.B.; Seifert, N.; Riss, G.; Neeb, M.; Steiner, G.; Hunziker, W.; Goralczyk, R. β-Carotene interferes with ultraviolet light A-induced gene expression by multiple pathways. J. Invest. Derm. 2005, 124, 428–434. [Google Scholar] [CrossRef] [PubMed]
- Wertz, K.; Seifert, N.; Hunziker, P.B.; Riss, G.; Wyss, A.; Hunziker, W.; Goralczyk, R. β-Carotene interference with UVA-induced gene expression by multiple pathways. Pure Appl. Chem. 2006, 78, 1539–1560. [Google Scholar] [CrossRef]
- Camera, E.; Mastrofrancesco, A.; Fabbi, C.; Daubrawa, F.; Picardo, M.; Sies, H.; Stahl, W. Astaxanthin, canthaxanthin and β-carotene differently affect UVA-induced oxidative damage and expression of oxidative stress-responsive enzyme. Exp. Derm. 2009, 18, 222–231. [Google Scholar] [CrossRef] [PubMed]
- Pappas, A.; Liakou, A.; Zouboulis, C.C. Nutrition and skin. Rev. Endocr. Metab. Disord. 2016, 17, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Souyoul, S.A.; Saussy, K.P.; Lupo, M.P. Nutraceuticals: A review. Dermatol. Ther. 2018, 8, 5–16. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, U.; Garnter, C.; Wiebusch, M.; Eichler, O.; Sies, H.; Tronnier, H.; Stahl, W. Supplementation with β-carotene or a similar amount of mixed carotenoids protects humans from UV-induced erythema. J. Nutr. 2003, 133, 98–101. [Google Scholar] [CrossRef] [PubMed]
- Kopckel, W.; Kurtmann, J. Protection from sunburn with β-cartoene- A meta-analysis. Photochem. Photobiol. 2008, 84, 284–288. [Google Scholar] [CrossRef] [PubMed]
- Valacchi, G.; Percrelli, A.; Mencarelli, M.; Maioli, E.; Davis, P.A. β-carotene prevents ozone-induced proinflammatory markers in murine skin. Toxicol. Ind. Health 2009, 25, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Darvin, M.E.; Fluhr, J.W.; Schanzer, S.; Richter, H.; Patzelt, A.; Meinke, M.C.; Zastrow, L.; Gloz, K.; Doucet, O.; Sterry, W.; et al. Dermal carotenoid levels and kinetics after topical and systemic administration of antioxidants: Enrichment strategies in a controlled in vivo study. J. Dermatol. Sci. 2011, 64, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Lademann, J.; Schanzer, S.; Meinke, M.; Sterry, W.; Darvin, M.E. Interactions between carotenoids and free radicals in human skin. Skin Pharmacol. Physiol. 2011, 24, 238–244. [Google Scholar] [CrossRef] [PubMed]
- Kuhn, R.; Sorensen, N.A. The coloring matters of the lobster (Astacus Gammarus L.). Z. Angew. Chem. 1938, 51, 465–466. [Google Scholar] [CrossRef]
- Pashkow, F.J.; Watumull, D.G.; Campbell, C.L. Astaxanthin: A novel potential treatment for oxidative stress and inflammation in cardiovascular disease. Am. J. Cardiol. 2008, 101, 58D–68D. [Google Scholar] [CrossRef] [PubMed]
- Coral-Hinstroza, G.N.; Ytrestoyl, T.; Ruyter, B.; Bjerkeng, B. Plasma appearance of unesterified astaxanthin geometrical E/Z and optical R/S isomers in men given a single dose of a mixture of optical 3 and 3’R/S isomers of astaxanthin fatty acyl diesters. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2004, 110, 99–110. [Google Scholar] [CrossRef] [PubMed]
- Okada, Y.; Ishikura, M.; Maoka, T. Bioavailability of astaxanthin in Haemtococcus algal extract: the effects of timing of diet and smoking habits. Biosci. Biotechnol. Biochem. 2009, 73, 1928–1932. [Google Scholar] [CrossRef] [PubMed]
- Herisset, A. Antioxidant properties of carotenoids and their derivatives. Weekly Report of Academy of Sciences Meetings, Volume 223, Paris, July–December 1946, Gauthier-Villars, Imprimeur-Libraire.
- Grangaud, R. Research on Astaxanthin, a New Vitamin A Factor. Doctoral Thesis, University of Lyon, Lyon, France, 1951. [Google Scholar]
- Miki, W. Biological functions and activities an animal carotenoids. Pure Appl. Chem. 1991, 63, 141–146. [Google Scholar] [CrossRef]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Scientific opinion on the safety of astaxanthin-rich ingredients (AstraREAL A1010 and AstraREAL L10) as novel food ingredients EFSA panel on dietetic products, nutrition and allergies (NDA). EFSA J. 2014, 12, 1–35. [Google Scholar]
- EFSA Panel on Additives and Products or Substances used in Animal Feed (FEEDAP). Scientific opinion on the safety and efficacy of synthetic astaxanthin as feed additive for salmon and trout, other fish, ornamental fish, crustaceans and ornamental birds. EFSA J. 2014, 12, 3724. [Google Scholar]
- Visioli, F.; Artaria, C. Astaxanthin in cardiovascular health and disease: mechanisms of action, therapeutic merits, and knowledge gaps. Food Func. 2017, 8, 39–63. [Google Scholar] [CrossRef] [PubMed]
- Anderson, M.L. A preliminary investigation of the enzymatic inhibition of 5α-reductase and growth of prostatic carcinoma cell line LNCap-FGC by natural astaxanthin and saw palmetto lipid extract in vitro. J. Herb. Pharmcother. 2005, 5, 17–26. [Google Scholar] [CrossRef]
- Angwafor, F., III; Anderson, M.L. An open label, dose response study to determine the effect of a dietary supplement on dihydrotestosterone, testosterone and estradiol levels in healthy males. J. Int. Soc. Sports Nutr. 2008, 5, 12. [Google Scholar] [CrossRef] [PubMed]
- Anderson, M.L. Evaluation of Resetin® on serum hormone levels in sedentary males. J. Int. Soc. Sports Nutr. 2014, 11, 43. [Google Scholar] [CrossRef] [PubMed]
- Suganuma, K.; Nakajima, H.; Ohtsuki, M.; Imokawa, G. Astaxanthin attenuates the UVA-induced up-regulation of matrix-metalloproteinase-1 and skin fibroblast elastase in human dermal fibroblasts. J. Dermatol. Sci. 2010, 58, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Tominaga, K.; Hongo, N.; Karato, M.; Yamashita, E. Cosmetic benefits of astaxanthin on human subjects. Acta Biochim. Pol. 2012, 59, 43–47. [Google Scholar] [PubMed]
- Nakajima, H.; Fukazawa, K.; Wakabayashi, Y.; Wakamatsu, K.; Senda, K.; Imokawa, G. Abrogating effect of a xanthophyll carotenoid astaxanthin on the stem cell factor-induced stimulation of human epidermal pigmentation. Arch. Derm. Res. 2012, 304, 803–816. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, H.; Terazawa, S.; Niwano, T.; Yamamoto, Y.; Imokawa, G. The inhibitory effects of antioxidants on ultraviolet-induced up-regulation of the wrinkling-inducing enzyme neutral endopeptidase in human fibroblasts. PLoS ONE 2016, 20, 11. [Google Scholar]
- Imokawa, G.; Ishida, K. Inhibitors of intracellular signaling pathways that lead to stimulated epidermal pigmentation: perspective of anti-pigmenting agents. Int. J. Mole. Sci. 2014, 15, 8293–8315. [Google Scholar] [CrossRef] [PubMed]
- Chou, G.; Lee, C.; Pan, J.; Wen, Z.; Huang, S.; Lan, C.; Liu, W.; Hour, T.; Hseu, Y.; Hwang, B.; et al. Enriched astaxanthin extract from Haematococcus pluvialis augments growth factor secretions to increase cell proliferation and induces MMP1 degradation to enhance collagen production in human dermal fibroblasts. Int. J. Mole. Sci. 2016, 17, 955. [Google Scholar] [CrossRef] [PubMed]
- Yoshihisa, Y.; Andoh, T.; Matsunaga, K.; Rehman, M.; Shimizu, T. Efficacy of astaxanthin for the treatment of atopic dermatitis in a murine model. PLoS ONE 2016, 29, 11. [Google Scholar] [CrossRef] [PubMed]
- Yoon, H.; Cho, H.; Cho, S.; Lee, S.; Shin, M.; Chung, J. Supplementing with dietary astaxanthin combined with collagen hydrolysate improves facial elasticity and decreases matrix metallproteinas-1 and -12 expression: A comparative study with placebo. J. Med. Food 2014, 17, 810–816. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, D.; Jena, G. Astaxanthin intervention ameliorates cyclophosphamide-induced oxidative stress, DNA damage and early hepato-carcinogenesis in rat: role of Nrf2, p53, p38 and phase-II enzymes. Mutat. Res. 2010, 696, 69–80. [Google Scholar] [CrossRef] [PubMed]
- Mizutani, Y.; Sakata, O.; Hoshino, T.; Honda, Y.; Yamashita, M.; Arakane, K.; Suzuki, T. Preventive effects of carotenoids on photoaging and its application for cosmetics. Jap. J. Cos. Chem. 2005, 29, 9–19. [Google Scholar]
- Kavitha, K.; Thiyagarajan, P.; Rathna Nandhini, J.; Mishra, R.; Nagini, S. Chemopreventive effects of diverse dietary phytochemicals against DMBA-induced hamster buccal pouch carcinogenesis via the induction of Nrf2-mediated cytoprotective antioxidant, detoxification, and DNA repair enzymes. Biochimie 2013, 95, 1629–1639. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.; Kim, C.; Lee, Y. Astaxanthin protects against MPTP/MPP+-induced mitochondrial dysfunction and ROS production in vivo and in vitro. Food Chem. Tox. 2011, 49, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Naidoo, K.; Hanna, R.; Birch-Machin, M.A. What is the role of mitochondrial dysfunction in skin photoaging? Exp. Derm. 2018, 27, 124–128. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, E. The effects of a dietary supplement containing astaxanthin on skin condition. Carotenoid Sci. 2006, 10, 91–95. [Google Scholar]
- Park, J.S.; Chyun, J.H.; Kim, Y.K.; Line, L.L.; Chew, B.P. Astaxanthin decreased oxidative stress and inflammation and enhanced immune response in humans. Nutr. Metab. 2010, 7, 18. [Google Scholar] [CrossRef] [PubMed]
- Chalyk, N.E.; Klochkov, V.A.; Bandaletova, T.Y.; Kyle, N.H.; Petyaev, I.M. Continuous astaxanthin intake reduces oxidative stress and reverses age-related morphological changes of residual skin surface components in middle-age volunteers. Nutr. Res. 2017, 48, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Tominaga, K.; Hongo, N.; Fujishita, M.; Takahashi, Y.; Adachi, Y. Protective effects of astaxanthin on skin deterioration. J. Clin. Biochem. Nutr. 2017, 61, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Ziosi, M.; Di Meo, I.; Kleiner, G.; Gao, X.-H.; Barca, E.; Sanchez-Qintero, M.J.; Tadesse, S.; Jiang, H.; Qiao, C.; Rodenburg, R.J.; et al. Coenzyme Q10 deficiency causes impairment of the sulfide oxidation pathway. EMBO Mole. Med. 2017, 9, 96–111. [Google Scholar] [CrossRef] [PubMed]
- Ramasarma, T. Natural occurrence and distribution of coenzyme Q10. In Coenzyme Q. Biochemistry; Biogenetics and Clinical Applications of Ubiquinone; Lenaz, G., Ed.; John Wiley and Sons: New York, NY, USA, 1985; pp. 67–81. [Google Scholar]
- Acosta, M.J.; Vazquez Fonseca, L.; Desbats, M.A.; Cerqua, C.; Zordan, R.; Trevisson, E.; Salviati, L. Coenzyme Q biosynthesis in health and disease. Biochim. Biophys. Acta 2016, 1857, 1079–1085. [Google Scholar] [CrossRef] [PubMed]
- Kaikkonen, J.; Tuomainen, E.P.; Nyyssonen, K.; Salonen, J.T. Coenzyme Q10: Absorption, antioxidative properties, determinants and plasma levels. Free Rad. Res. 2002, 36, 389–397. [Google Scholar] [CrossRef]
- Mattila, P.; Kumpulainen, J. Coenzyme Q9 and Q10: contents in foods and dietary intake. J. Food Comp. Anal. 2001, 14, 409–417. [Google Scholar] [CrossRef]
- Overvard, K.; Diamant, B.; Holm, L.; Holmer, G.; Mortensen, S.A.; Stenders, S. Coenzyme Q10 in health and disease. Euro. J. Clin. Nutr. 1999, 53, 764–770. [Google Scholar] [CrossRef]
- Beyer, R.E. The participation of coenzyme Q10 in free radical production and antioxidation. Free Rad Biol. Med. 1990, 8, 545–565. [Google Scholar] [CrossRef]
- Inui, M.; Boe, M.; Kujii, K.; Mataunaka, H.; Yoshida, M.; Ichihashi, M. Mechanisms of inhibitory effects of CoQ10 on UVB-induced wrinkle formation in vitro and in vivo. Biofactors 2008, 32, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Dang, L.; Guo, F.; Wang, X.; Zhao, W.; Zhao, R. Coenzyme Q10 enhances dermal elastin expression, inhibits IL-1α production and melanin synthesis in vitro. Int. J. Cos. Sci. 2012, 34, 273–279. [Google Scholar] [CrossRef] [PubMed]
- Muta-Takada, K.; Terada, T.; Yamanishi, H. Coenzyme Q10 protects against oxidative stress induced cell death and enhances the synthesis of basement membrane components in dermal and epidermal cells. Biofactors 2009, 35, 435–441. [Google Scholar] [CrossRef] [PubMed]
- Fuller, B.; Smith, D.; Howerton, A. Anti-inflammatory effects of CoQ10 and colorless carotenoids. J. Cosmet. Dermatol. 2006, 5, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Hoppe, W.; Bergemann, J.; Diembeck, W.; Ennen, J.; Gohla, S.; Jacob, J.; Kielholz, J.; Mei, W.; Poliet, D. Coenzyme Q10, a cutaneous antioxidant and energier. Biofactors 1999, 9, 371–378. [Google Scholar] [CrossRef] [PubMed]
- Blatt, T.; Lenz, H.; Koop, U.; Jaspers, S.; Weber, T.; Mummert, C.; Wittern, K.-P.; Stab, F.; Wenck, H. Stimulation of skin’s energy metabolism provides multiple benefits for mature human skin. Biofactors 2005, 25, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Ashida, Y.; Yamaishi, H.; Terada, T.; Oota, N.; Sekine, S.; Watabe, K. CoQ10 supplementation elevates the epidermal CoQ10 levels in adult hairless mice. Biofactors 2005, 25, 175–178. [Google Scholar] [CrossRef] [PubMed]
- Prahl, S.; Kueper, T.; Biernoth, T.; Wohrmann, Y.; Munster, A.; Furstenau, M.; Schmidt, M.; Schulze, C.; Wittern, K.P.; Wenck, H. Aging skin is functionally anaerobic: importance of coenzyme Q10 for antiaging skin care. Biofactors 2008, 32, 245–255. [Google Scholar] [CrossRef] [PubMed]
- Kharaeva, Z.; Gostova, E.; De Luca, C.; Raskovic, D.; Korkina, L. Clinical and biochemical effects of coenzyme Q10, vitamin E., and selenium supplementation to psoriasis patients. Nutrition 2009, 25, 295–302. [Google Scholar] [CrossRef] [PubMed]
- Knott, A.; Achterberg, V.; Mielke, H.; Sperling, G.; Dunckelman, K.; Vogelsang, A.; Kruger, A.; Schwengler, H.; Behtash, M.; Kristof, S.; et al. Topical treatment with coenzyme Q10-containing formulas improves skin’s Q10 levels and provides antioxidative effects. Biofactors 2015, 41, 383–390. [Google Scholar] [CrossRef] [PubMed]
- Schniertshauer, D.; Muller, S.; Mayr, T.; Sonntag, T.; Gebhard, D.; Bergemann, J. Accelerated regeneration of ATP levels after irradiation in human skin fibroblasts by coenzyme Q10. Photochem. Photobiol. 2016, 92, 488–494. [Google Scholar] [CrossRef] [PubMed]
- Kaci, M.; Belfaffef, A.; Meziane, S.; Dostert, G.; Menu, P.; Velot, E.; Desobry, S.; Arab-Tehrany, E. Nanoemulsions and topical creams for the safe and effective delivery of lipophilic antioxidant coenzyme Q10. Colloids Surfaces B: Biointerfaces 2018, 167, 165–175. [Google Scholar] [CrossRef] [PubMed]
- Zmitke, K.; Pogacnik, T.; Mervic, L.; Zmitek, J.; Pravst, I. The effect of dietary intake of coenzyme Q10 of skin parameters and condition: results of a randomized, placebo-controlled, double-blind study. Biofactors 2017, 43, 132–140. [Google Scholar] [CrossRef] [PubMed]
- McGrath, B.A.; Fox, P.F.; McSweeny, P.L.H.; Kelly, A.L. Composition and properties of bovine colostrum: A review. Diary Sci. Technol. 2016, 96, 133–158. [Google Scholar] [CrossRef]
- Ballard, O.; Morrow, A.L. Human milk composition: nutrients and bioactive factors. Pediatric Clin. North. Am. 2013, 60, 49–74. [Google Scholar] [CrossRef] [PubMed]
- Saint, L.; Smith, M.; Hartmann, P.E. The yield and nutrient content of colostrum and milk of women from giving birth to 1-month post-partum. Br. J. Nutr. 1984, 52, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Xu, R.J. Development of the newborn GI tract and its relation to colostrum/milk intake: A review. Reprod. Fert. Dev. 1996, 8, 35–48. [Google Scholar] [CrossRef]
- Quigley, J.D.; Drewry, J.J. Nutrient and immunity transfer from cow to calf pre- and postcalving. J. Dairy Sci. 1998, 81, 2779–2790. [Google Scholar] [CrossRef]
- Balfour, W.E.; Comline, R.S. Acceleration of the absorption of unchanged globulin in the new-born calf by factors in colostrum. J. Physiol. 1962, 160, 234–257. [Google Scholar] [CrossRef] [PubMed]
- Playford, R.J.; Macdonald, C.E.; Johnson, W.S. Colostrum and milk-derived peptide growth factors for the treatment of gastrointestinal disorders. Am. J. Clin. Nutr. 2000, 72, 5–14. [Google Scholar] [CrossRef] [PubMed]
- Tokuyama, Y.; Tokuyama, H. Purification and identification of TGF-β2-related growth factor from bovine colostrum. J. Dairy Res. 1993, 60, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Buescher, E.S.; McIlheran, S.M. Antioxidant properties of human colostrum. Pediatrics Res. 1988, 24, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Yoon, K.; Ha, S.; Kim, N.; Kim, K. The effects of bovine colostrum on human skin. J. Invest. Derm. 2002, 119, 311. [Google Scholar]
- Amiot, J.; Germain, L.; Turgeon, S.; Lemay, M.; Ory-Salam, C.; Auger, F.A. Peptides from milk protein hydrolysates to improve the growth of human keratinocytes in culture. Int. Dairy J. 2004, 14, 619–626. [Google Scholar] [CrossRef]
- Zava, S.; Barello, C.; Pessione, A.; Perono Garoffo, L.; Fatton, P.; Montorfano, G.; Conti, A.; Giunta, C.; Pessione, E.; Berra, B.; et al. Mare’s colostrum globules stimulate fibroblast growth in vitro: A biochemical study. J. Medicinal Food 2009, 12, 836–845. [Google Scholar] [CrossRef] [PubMed]
- National Institutes of Health, Zinc-Health Professional Fact Sheet. Available online: https://pds.od.nih.gov/factsheets/Zinc-HealthProfessional/ (accessed on 13 August 2018).
- Prasad, A.S. Zinc: An overview. Nutrition 1995, 11, 93–99. [Google Scholar] [PubMed]
- Heyneman, C.A. Zinc deficiency and taste disorders. Annu. Pharmacother. 1996, 30, 186–187. [Google Scholar] [CrossRef] [PubMed]
- Prasad, A.S. Chapter 20-Discovery of Zinc for Human Health and Biomarkers of Zinc Deficiency. In Molecular, Genetic, and Nutritional Aspects of Major and Minor Trace Minerals; Collins, J.F., Ed.; Academic Press (Elsevier): London, UK, 2017; pp. 241–260. [Google Scholar]
- Michaelsson, G.; Ljunghall, K.; Danielson, B.G. Zinc in epidermis and dermis in healthy subjects. Acta Derm. Venereol. 1980, 60, 295–299. [Google Scholar] [PubMed]
- Fabris, N.; Mocchegiani, E. Zinc, human diseases and aging. Aging Clin. Exp. Res. 1995, 7, 77–93. [Google Scholar] [CrossRef]
- Rink, L.; Gabriel, P. Zinc and the immune system. Proc. Nutr. Soc. 2000, 59, 541–552. [Google Scholar] [CrossRef] [PubMed]
- Wintergest, E.S.; Maggini, S.; Hornig, D.H. Contribution of selected vitamins and trace elements to immune function. Annu. Nutr. Metab. 2007, 51, 301–323. [Google Scholar] [CrossRef] [PubMed]
- Lansdown, A.B.; Mirastschijski, U.; Stubbs, N.; Scanlon, E.; Agren, M.S. Zinc in wound healing: theroretical, experimental, and clinical aspects. Wound Repair Regen. 2007, 15, 2–16. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, J.R.; Marsh, R.G.; Draelos, Z.D. Zinc and skin health: overview of physiology and pharmacology. Dermatol. Surg. 2005, 31, 837–847. [Google Scholar] [CrossRef] [PubMed]
- Newman, M.D.; Stotland, M.; Ellis, J.I. The safety of nanosized particles in titanium dioxide- and zinc dioxide-based sunscreens. J. Am. Acad. Derm. 2009, 61, 685–692. [Google Scholar] [CrossRef] [PubMed]
- Derry, J.E.; McLean, W.M.; Freeman, J.B. A study of the percutaneous absorption from topically applied zinc ointment. JPEN J. Parenter. Enteral Nutr. 1983, 7, 131–135. [Google Scholar] [CrossRef] [PubMed]
- Halmans, G. Healing of experimentally induced burn wounds. A comparative study of the healing of exposed burn wounds and burn wounds covered with adhesive zinc-tape and a scanning electron microscopic study of the microvasculature of wound healing in the rabbit ear. Scand. J. Plast. Reconstr. Surg. 1978, 12, 105–109. [Google Scholar] [CrossRef]
- Kim, K.B.; Kim, Y.W.; Lim, S.K.; Roh, T.H.; Bang, D.Y.; Choi, S.M.; Lim, D.S.; Kim, Y.J.; Baek, S.H.; Kim, M.K. Risk assessment of zinc oxide, a cosmetic ingredient used as a UV filter of sunscreens. J. Tox. Environ. Health-Part B-Critical Rev. 2017, 20, 155–182. [Google Scholar] [CrossRef] [PubMed]
- Landsdown, A.B. Metallothioneins: potential therapeutic aids for wound healing in the skin. Wound Repair Regen. 2002, 10, 130–132. [Google Scholar] [CrossRef]
- Steiling, H.; Munz, B.; Werner, S.; Brauchle, M. Different types of ROS-scavenging enzymes are expressed during cutaneous wound repair. Exp. Cell Res. 1999, 247, 484–494. [Google Scholar] [CrossRef] [PubMed]
- Lin, P.H.; Sermersheim, M.; Li, H.C.; Lee, P.H.U.; Steinberg, S.M.; Ma, J.J. Zinc in wound healing modulation. Nutrients 2018, 10, 16. [Google Scholar] [CrossRef] [PubMed]
- Mulhern, S.A.; Stroube, W.B., Jr.; Jacobs, R.M. Alopecia induced in young mice by exposure to excess dietary zinc. Experientia 1986, 42, 551–553. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, Y.; Kawamura, T.; Shimada, S. Zinc and skin biology. Arch. Biochem. Biophys. 2016, 611, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Kogan, S.; Sood, A.; Granick, M.S. Zinc and wound healing: A review of zinc physiology and clinical applications. Wounds-A Compendium Clin. Res. Pract. 2017, 29, 102–106. [Google Scholar]
- Ogawa, Y.; Kinoshita, M.; Shimada, S.; Kawamura, T. Zinc and skin disorders. Nutrients 2018, 10, 199. [Google Scholar] [CrossRef] [PubMed]
- Inoue, Y.; Hasegawa, S.; Ban, S.; Yamada, T.; Date, Y.; Mizutani, H.; Nakata, S.; Tanaka, M.; Hirashima, N. ZIP2 protein, a zinc transporter, is associated with keratinocyte differentiation. J. Biol. Chem. 2014, 289, 21451–21462. [Google Scholar] [CrossRef] [PubMed]
- National Institutes of Health, Selenium-Health Professional Fact Sheet. Available online: https://pds.od.nih.gov/factsheets/Selenium-HealthProfessional/ (accessed on 13 August 2018).
- Berzelius, J.J. Additional Observations on Lithion and Selenium. Annu. Philosophy 1818, 11, 373. [Google Scholar]
- Sunde, R.A. Selenium. In Present Knowledge in Nutrition, 9th ed.; Erdman, J.W., Macdonald, I.A., Zeisel, S.H., Eds.; Wiley-Blackwell: Washington, DC, USA, 2006; pp. 480–497. [Google Scholar]
- Terry, E.N.; Diamond, A.M. Selenium. In Present Knowledge in Nutrition, 10th ed.; Erdman, J.W., Macdonald, I.A., Zeisel, S.H., Eds.; Wiley-Blackwell: Washington, DC, USA, 2012; pp. 568–587. [Google Scholar]
- Sunde, R.A. Selenium. In Modern Nutrition in Health and Disease, 11th ed.; Ross, A.C., Caballero, B., Cousins, R.J., Tucker, K.L., Ziegler, T.R., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2012; pp. 225–237. [Google Scholar]
- Institute of Medicine, Food and Nutrition Board. Dietary Reference Intakes: Vitamin C, Vitamin E, Selenium, and Carotenoids; National Academy Press: Washington, DC, USA, 2000. [Google Scholar]
- McKenzie, R.C. Selenium, ultraviolet radiation and the skin. Clin. Exp. Dermatol. 2000, 25, 631–636. [Google Scholar] [CrossRef] [PubMed]
- Rafferty, T.S.; McKenzie, R.C.; Hunter, J.A. Differential expression of selenoproteins by human skin cells and protection by selenium from UVB-radiation-induced cell death. Biochem. J. 1998, 332, 231–236. [Google Scholar] [CrossRef] [PubMed]
- Schallreuter, K.U.; Pittelkow, M.R.; Wood, J.M. Free radical reduction by thioredoxin reductase at the surface of normal and vitiliginous human keratinocytes. J. Invest. Derm. 1986, 87, 728–732. [Google Scholar] [CrossRef] [PubMed]
- Burke, K.E.; Combs, G.F., Jr.; Gross, E.G.; Bhuyan, K.C.; Abu-Libdeh, H. The effects of oral L-selenomethionine on pigmentation and skin cancer induced by ultraviolet irradiation. Nutr. Cancer 1992, 17, 123–137. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, P.; Sharma, S.; Agarwal, U.S. Selenium toxicity: A rare diagnosis. Indian J. Dermatol. Venereol. Leprol. 2016, 82, 690–693. [Google Scholar] [CrossRef] [PubMed]
- Davis, T.Z.; Hall, J.O. Chapter 36—Selenium. In Reproductive Developmental Toxicology; Elsevier: New York, NY, USA, 2011; pp. 461–468. [Google Scholar]
- Hwang, S.W.; Lee, H.J.; Suh, K.S.; Kim, S.T.; Park, S.W.; Hur, D.Y.; Lee, D.; Seo, J.K.; Sung, H.S. Changes in murine hair with dietary selenium excess or deficiency. Exp. Derm. 2011, 20, 367–369. [Google Scholar] [CrossRef] [PubMed]
- Polelka, T.G.; Bianchini, R.J.; Shapiro, S. Interaction of mineral salts with the skin: A literature survey. Int. J. Cosmet. Dermatol. 2012, 34, 416–423. [Google Scholar] [CrossRef] [PubMed]
- Sengupta, A.; Lichti, U.F.; Carlson, B.A. Selenoproteins are essential for proper keratinocyte function and development. PLos One 2010, 5, e12249. [Google Scholar] [CrossRef] [PubMed]
- Mirastschijski, U.; Martin, A.; Jorgensen, L.N.; Sampson, B.; Agren, M.S. Zinc, copper, and selenium tissue levels and their relation to subcutaneous abscess, minor surgery, and wound healing in humans. Biol. Trace Elem. Res. 2013, 153, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Naziroglu, M.; Yildiz, K.; Tamturk, B.; Erturan, I.; Flores-Arce, M. Selenium and psoriasis. Biol. Trace Elem. Res. 2012, 150, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Michaelson, G.; Berne, B.; Carlmark, B.; Strand, A. Selenium in whole blood and plasma is decreased in patients with moderate and severe psoriasis. Acta Derm. Venereol. 1989, 69, 29–34. [Google Scholar]
- Cassidy, P.B.; Fain, H.D.; Cassidy, J.P., Jr.; Tran, S.M.; Moos, P.J.; Boucher, K.M.; Gerads, R.; Florell, S.R.; Grossman, D.; Leachman, S.A. Selenium for the prevention of cutaneous melanoma. Nutrients 2013, 5, 725–749. [Google Scholar] [CrossRef] [PubMed]
- Hazane-Puch, F.; Champelovier, P.; Arnaud, J.; Garrel, C.; Ballester, B.; Faure, P.; Laporte, F. Long-term selenium supplementation in HaCaT cells: importance of chemical form for antagonist (protective versus toxic) activities. Bio. Trace Elem. Res. 2013, 154, 288–298. [Google Scholar] [CrossRef] [PubMed]
- Hazane-Puch, F.; Champelovier, P.; Arnaud, J.; Trocme, C.; Garrel, C.; Faure, P.; Laporte, F. Six-day selenium supplementation led to either UVA-photoprotection or toxic effects in human fibroblasts depending on the chemical form and dose of Se. Metallomics 2014, 6, 1683–1692. [Google Scholar] [CrossRef] [PubMed]
- Landsdown, A.B.C. Physiological and toxicological changes in the skin resulting from the action and interaction of metal ions. CRC Crit. Rev. Toxicol. 1995, 25, 397–462. [Google Scholar] [CrossRef] [PubMed]
- Anunciato, T.P.; da Rocha Filho, P.A. Carotenoids and polyphenols in nutricosmetics, nutraceuticals, and cosmeceuticals. J. Cosmet. Dermatol. 2012, 11, 51–54. [Google Scholar] [CrossRef] [PubMed]
- Davinelli, S.; Bertoglio, J.C.; Polimeni, A.; Scapagnini, G. Cytoprotective polyphenols against chronological skin aging and cutaneous photodamage. Curr. Pharm. Des. 2018, 24, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Scapagnini, G.; Davinelli, S.; Di Renzo, L.; De Lorenzo, A.; Olarte, H.H.; Micali, G.; Cicero, A.F.; Gonzalez, S. Cocoa bioactive compounds: significance and potential for maintenance of skin health. Nutrients 2014, 6, 3202–3213. [Google Scholar] [CrossRef] [PubMed]
- Lephart, E.D.; Sommerfeldt, J.M.; Andrus, M.B. Resveratrol: influences on gene expression in human skin. J. Funct. Foods 2014, 10, 377–384. [Google Scholar] [CrossRef]
- Irrera, N.; Pizzino, G.; D’Anna, R.; Vaccaro, M.; Arcoraci, V.; Squadrito, F.; Altavilla, D.; Bitto, A. Dietary management of skin health: the role of genistein. Nutrients 2017, 9, 622. [Google Scholar] [CrossRef] [PubMed]
- Lephart, E.D. Resveratrol, 4’ acetoxy resveratrol, R-equol, racemic equol or S-equol as cosmeceuticals to improve dermal health. Int. J. Mole. Sci. 2017, 18, 1193. [Google Scholar] [CrossRef] [PubMed]
- Tsiaras, W.G.; Weinstock, M.A. Factors influencing vitamin D status. Acta Derm. Venereol. 2011, 91, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Bikle, D.D. Vitamin D metabolism and function in the skin. Mol. Cell. Endocrinol. 2011, 347, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.; Wortsman, J. Neuroendocrinology of the skin. Endocr. Rev. 2000, 21, 457–487. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.; Zbytek, B.; Nikolakis, G.; Manna, P.R.; Skobowiat, C.; Zmijewski, M.; Li, W.; Janjetovic, Z.; Postelthwaite, A.; Zoubloulis, C.C.; et al. Steroidogenesis in the skin: implications for local immune functions. J. Steroid Biochem. Mole. Biol. 2013, 137, 107–123. [Google Scholar]
- Slominski, A. Neuroendocrine system of the skin. Dermatology 2005, 211, 199–208. [Google Scholar] [CrossRef] [PubMed]
- Skobowiat, C.; Dowdy, J.C.; Sayre, R.M.; Tuckey, R.C.; Slominski, A. Cutaneous hypothalamic-pituitary-adrenal axis homolog: regulation by ultraviolet radiation. Am. J. Physiol. Endocrinol. Metab. 2011, 301, E484–E493. [Google Scholar] [CrossRef] [PubMed]
- Umar, M.; Sastry, K.; Al Ali, F.; Al-Khulaifi, A.; Wang, E.; Chouchane, A.I. Vitamin D and the pathophysiology of inflammatory skin diseases. Skin Pharmacol. Physiol. 2018, 31, 74–86. [Google Scholar] [CrossRef] [PubMed]
- Kong, H.H. Skin microbiome: genomics-based insights into the diversity and role of skin microbes. Trends Mol. Med. 2011, 17, 320–328. [Google Scholar] [CrossRef] [PubMed]
- Relman, D.A. New technologies, human-microbe interactions, and the search for previously unrecognized pathogens. J. Infect. Dis. 2002, 186, S254–S258. [Google Scholar] [CrossRef] [PubMed]
- Prescott, S.L. History of medicine: Origin of the term microbiome and why it matters. Hum. Microbiome J. 2017, 4, 24–25. [Google Scholar] [CrossRef]
- Human Microbiome Project. 23 April 2018. Available online: https://commonfund.nih.gov/hmp (accessed on 13 August 2018).
- Kong, H.H.; Segre, J.A. Skin microbiome: looking back to move forward. J. Invest. Dermatol. 2012, 132, 933–939. [Google Scholar] [PubMed]
- Byrd, A.L.; Belkaid, Y.; Segre, J.A. The human skin microbiome. Nat. Rev. Microbiol. 2018, 16, 143–155. [Google Scholar] [CrossRef] [PubMed]
- Vaughn, A.; Notay, M.; Clark, A.K.; Sivamani, R.K. Skin-gut axis: The relationship between intestinal bacteria and skin health. World J. Dermatol. 2017, 6, 52–58. [Google Scholar] [CrossRef]
- Scharschmidt, T.C.; Fischbach, M.A. What lives on our skin: Ecology, genomics and therapeutic opportunities of the skin microbiome. Drug Discov. Today Dis. Mech. 2013, 10, pii e83–pii e89. [Google Scholar] [CrossRef] [PubMed]
- Belkaid, Y.; Segre, J.A. Dialogue between skin microbiota and immunity. Science 2014, 346, 954–959. [Google Scholar] [CrossRef] [PubMed]
- Grice, E.A. The intersection of microbiome and host at the skin interface: genomic- and metagenomic based insights. Genome Res. 2015, 25, 1514–1520. [Google Scholar] [CrossRef] [PubMed]
- Mueller, N.T.; Bakacs, E.; Combellick, J.; Grigoryan, Z.; Dominguez-Bello, M.G. The infant microbiome development: mom matters. Trends Mol. Med. 2015, 21, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Dominguez-Bello, M.G.; Costello, E.K.; Contreras, M.; Magris, M.; Hidalgo, G.; Fierer, N.; Knight, R. Delivery mode shapes the acquisition and structure of the initial microbiota across multiple body habitats in newborns. Proc. Natl. Acad. Sci. USA 2010, 107, 11971–11975. [Google Scholar] [CrossRef] [PubMed]
- Cundell, A.M. Microbial Ecology of the Human Skin. Microb. Ecol. 2018, 76, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Iebba, V.; Totino, V.; Gagliardi, A.; Santangelo, F.; Cacciotti, F.; Trancassini, M.; Mancini, C.; Cicerone, C.; Corazziari, E.; Pantanella, F.; et al. Eubiosis and dysbiosis: the two sides of the microbiota. New Microbiol. 2016, 39, 1–12. [Google Scholar] [PubMed]
- Christensen, G.J.; Bruggemann, H. Bacterial skin commensals and their role as host guardians. Benef. Microbes. 2014, 5, 201–215. [Google Scholar] [CrossRef] [PubMed]
- Gallo, R.L.; Nakatsuji, T. Microbial symbiosis with the innate immune defense system of the skin. J. Invest. Dermatol. 2011, 131, 1974–1980. [Google Scholar] [CrossRef] [PubMed]
- Bastos, M.C.; Ceotto, H.; Coelho, M.L.; Nascimento, J.S. Staphylococcal antimicrobial peptides: Relevant properties and potential biotechnological applications. Curr. Pharm. Biotechnol. 2009, 10, 38–61. [Google Scholar] [CrossRef] [PubMed]
- Iwase, T.; Uehara, Y.; Shinji, H.; Tajima, A.; Seo, H.; Takada, K.; Agata, T.; Mizunoe, Y. Staphylococcus epidermidis Esp inhibits Staphylococcus aureus biofilm formation and nasal colonization. Nature 2010, 465, 346–349. [Google Scholar] [CrossRef] [PubMed]
- Lai, Y.; Di Nardo, A.; Nakatsuji, T.; Leichtle, A.; Yang, Y.; Cogen, A.L.; Wu, Z.R.; Hooper, L.V.; Schmidt, R.R.; von Aulock, S.; et al. Commensal bacteria regulate Toll-like receptor 3-dependent inflammation after skin injury. Nat. Med. 2009, 15, 1377–1382. [Google Scholar] [CrossRef] [PubMed]
- Yuki, T.; Yoshida, H.; Akazawa, Y.; Komiya, A.; Sugiyama, Y.; Inoue, S. Activation of TLR2 enhances tight junction barrier in epidermal keratinocytes. J. Immunol. 2011, 187, 3230–3237. [Google Scholar] [CrossRef] [PubMed]
- Costello, E.K.; Lauber, C.L.; Hamady, M.; Fierer, N.; Gordon, J.I.; Knight, R. Bacterial community variation in human body habitats across space and time. Science 2009, 36, 1694–1697. [Google Scholar] [CrossRef] [PubMed]
- Zlotogorski, A. Distribution of skin surface pH on the forehead and cheek of adults. Arch. Derm. Res. 1987, 279, 398–401. [Google Scholar]
- Rieg, S.; Seeber, S.; Steffen, H.; Hameny, A.; Kalbacher, H.; Stevanovic, S.; Kimura, A.; Garbe, C.; Schittek, B. Generation of multiple stable dermcidin-derived antimicrobial peptides in sweat of different body sites. J. Invest. Dermatol. 2006, 126, 354–365. [Google Scholar] [CrossRef] [PubMed]
- Rieg, S.; Garbe, C.; Sauer, B.; Kalbacher, H.; Schittek, B. Dermcidin is constitutively produced by eccrine sweat glands and is not induced in epidermal cells under inflammatory skin conditions. Br. J. Dermatol. 2004, 151, 534–539. [Google Scholar] [CrossRef]
- Murakami, M.; Ohtake, T.; Dorschner, R.A.; Chittek, B.; Gallo, R.L. Cathelicidin anti-microbial peptide expression in sweat, an innate defense system for the skin. J. Invest. Dermatol. 2002, 119, 1090–1095. [Google Scholar] [CrossRef] [PubMed]
- Gallo, R.L.; Nizet, V. Innate barriers against infection and associated disorders. Drug Discov. Today Dis. Mech. 2008, 5, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Deckers, J.; Hammad, H.; Hoste, E. Langerhans Cells: Sensing the Environment in Health and Disease. Front. Immunol. 2018, 9, 93. [Google Scholar] [CrossRef] [PubMed]
- Lynn, D.D.; Umari, T.; Dunnick, C.A.; Dellavalle, R.P. The epidemiology of acne vulgaris in late adolescence. Adolesc. Health Med. Ther. 2016, 7, 13–25. [Google Scholar] [CrossRef] [PubMed]
- Williams, H.C.; Dellavalle, R.P.; Garner, S. Acne vulgaris. Lancet 2012, 379, 361–372. [Google Scholar] [CrossRef]
- Fitz-Gibbon, S.; Tomida, S.; Chiun, B.H.; Nguyen, L.; Du, C.; Liu, M.; Elashoff, D.; Erfe, M.C.; Loncaric, A.; Kim, J.; et al. Propionibacterium acnes strain populations in the human skin microbiome associated with acne. J. Invest. Dermatol. 2013, 139, 2152–2160. [Google Scholar] [CrossRef] [PubMed]
- Benhadou, F.; Mintoff, D.; Schnebert, B.; Thio, H.B. Psoriasis and Microbiota: A Systematic Review. Disease 2018, 6, 2. [Google Scholar] [CrossRef] [PubMed]
- Kong, H.H.; Oh, J.; Deming, C.; Conlan, S.; Grice, E.A.; Beaton, M.A.; Nomicos, E.; Polley, E.C.; Komarrow, H.D.; NISC Sequence Program; et al. Temporal shifts in the skin microbiome associated with disease flares and treatment in children with atopic dermatitis. Genome Res. 2012, 22, 850–859. [Google Scholar] [CrossRef] [PubMed]
- Ellebrecht, C.T.; Srinivas, G.; Bieber, K.; Banczyk, D.; Kalies, K.; Kunzel, S.; Hammers, C.M.; Baines, J.F.; Zillikeno, D.; Ludwig, R.J.; et al. Skin microbiota-associated inflammation precedes autoantibody induced tissue damage in experimental epidermolysis bullosa acquisita. J. Autoimmun. 2016, 68, 14–22. [Google Scholar] [CrossRef] [PubMed]
- Jarmuda, S.; O’Reily, N.; Zaba, R.; Jakubowicz, O.; Szkaradkiewicz, A.; Kavanagh, K. Potential role of Demodex mites and bacteria in the induction of rosacea. J. Med. Microbiol. 2012, 61, 1504–1510. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Sheha, H.; Tseng, S.C. Pathogenic role of Demodex mites in blepharitis. Curr. Opin. Allergy Clin. Immunol. 2010, 10, 505–510. [Google Scholar] [CrossRef] [PubMed]
- Christoph, T.; Muller-Rover, S.; Audring, H.; Tobin, D.J.; Hermes, B.; Cotsarelis, G.; Ruckert, R.; Paus, R. The human hair follicle immune system: cellular composition and immune privilege. Br. J. Dermatol. 2000, 142, 862–873. [Google Scholar] [CrossRef] [PubMed]
- Ito, T.; Ito, N.; Betterman, A.; Tokura, Y.; Takigawa, M.; Paus, R. Collapse and restoration of MHC class-I-dependent immune privilege: exploiting the human hair follicle as a model. Am. J. Pathol. 2004, 164, 623–634. [Google Scholar] [CrossRef]
- Paus, R.; Eichmuller, S.; Hofmann, U.; Czarnetzki, B.M.; Robinson, P. Expression of classical and non-classical MHC class I antigens in murine hair follicles. Br. J. Dermatol. 1994, 131, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Sherwani, M.A.; Tufail, S.; Muzaffar, A.F.; Yusuf, N. The skin microbiome and immune system: Potential target for chemoprevention? Photoderm. Photoimmunol. Photomed. 2018, 34, 25–34. [Google Scholar] [CrossRef] [PubMed]
- von Hertzen, L.C.; Joensuu, H.; Haahtela, T. Microbial deprivation, inflammation and cancer. Cancer Metastasis Rev. 2011, 30, 211–223. [Google Scholar] [CrossRef] [PubMed]
- Burns, E.M.; Yusuf, N. Toll-like receptors and skin cancer. Front. Immunol. 2014, 5, 135. [Google Scholar] [CrossRef] [PubMed]
- Akaza, H.; Akaza, H.; Iwasaki, A.; Ohtani, M.; Ikeda, N.; Nijima, K.; Toida, I.; Koiso, K. Expression of antitumor response. Role of attachment and viability of bacillus Calmette-Guerin to bladder cancer cells. Cancer 1993, 72, 558–563. [Google Scholar] [CrossRef]
- Juckett, D.A.; Aylsworth, C.F.; Quensen, J.M. Intestinal protozoa are hypothesized to stimulate immunosurveillance against colon cancer. Med. Hypotheses 2008, 71, 104–110. [Google Scholar] [CrossRef] [PubMed]
- Enterline, P.E.; Sykora, J.L.; Keleti, G.; Lange, J.H. Endotoxins, cotton dust, and cancer. Lancet 1985, 2, 934–935. [Google Scholar] [CrossRef]
- Probiotics: In Depth. 31 July 2018. Available online: https://nccih.nih.gov/health/probiotics/introduction.htm (accessed on 13 August 2018).
- Bowe, W.; Patel, N.B.; Logan, A.C. Acne vulgaris, probiotics and the gut-brain-skin axis: from anecdote to translational medicine. Benef. Microbes 2014, 5, 185–199. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Kuo, S.; Shu, M.; Yu, J.; Huang, S.; Dai, A.; Two, A.; Gallo, R.L.; Huang, C.M. Staphylococcus epidermidis in the human skin microbiome mediates fermentation to inhibit the growth of Propionibacterium acnes: implications of probiotics in acne vulgaris. Appl. Microbiol. Biotechnol. 2014, 98, 411–424. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Kao, M.S.; Yu, J.; Huang, S.; Marito, S.; Gallo, R.L.; Huang, C.M. A Precision microbiome approach using sucrose for selective augmentation of Staphylococcus epidermidis fermentation against Propionibacterium acnes. Int. J. Mol. Sci. 2016, 17, 11. [Google Scholar] [CrossRef] [PubMed]
- Myles, I.A.; Williams, K.W.; Reckhow, J.D.; Jammeh, M.L.; Pincus, N.B.; Sastalla, I.; Saleem, D.; Stone, K.D.; Datta, S.K. Transplantation of human skin microbiota in models of atopic dermatitis. JCI Insight 2016, 1, 10. [Google Scholar] [CrossRef] [PubMed]
- Juge, R.; Rouaud-Tinguely, P.; Breugnot, J.; Servaes, K.; Grimaldi, C.; Roth, M.-P.; Coppin, H.; Closs, B. Shift in skin microbiota of Western European women across aging. J. Appl. Microbiol. 2018, 125, 907–916. [Google Scholar] [CrossRef] [PubMed]
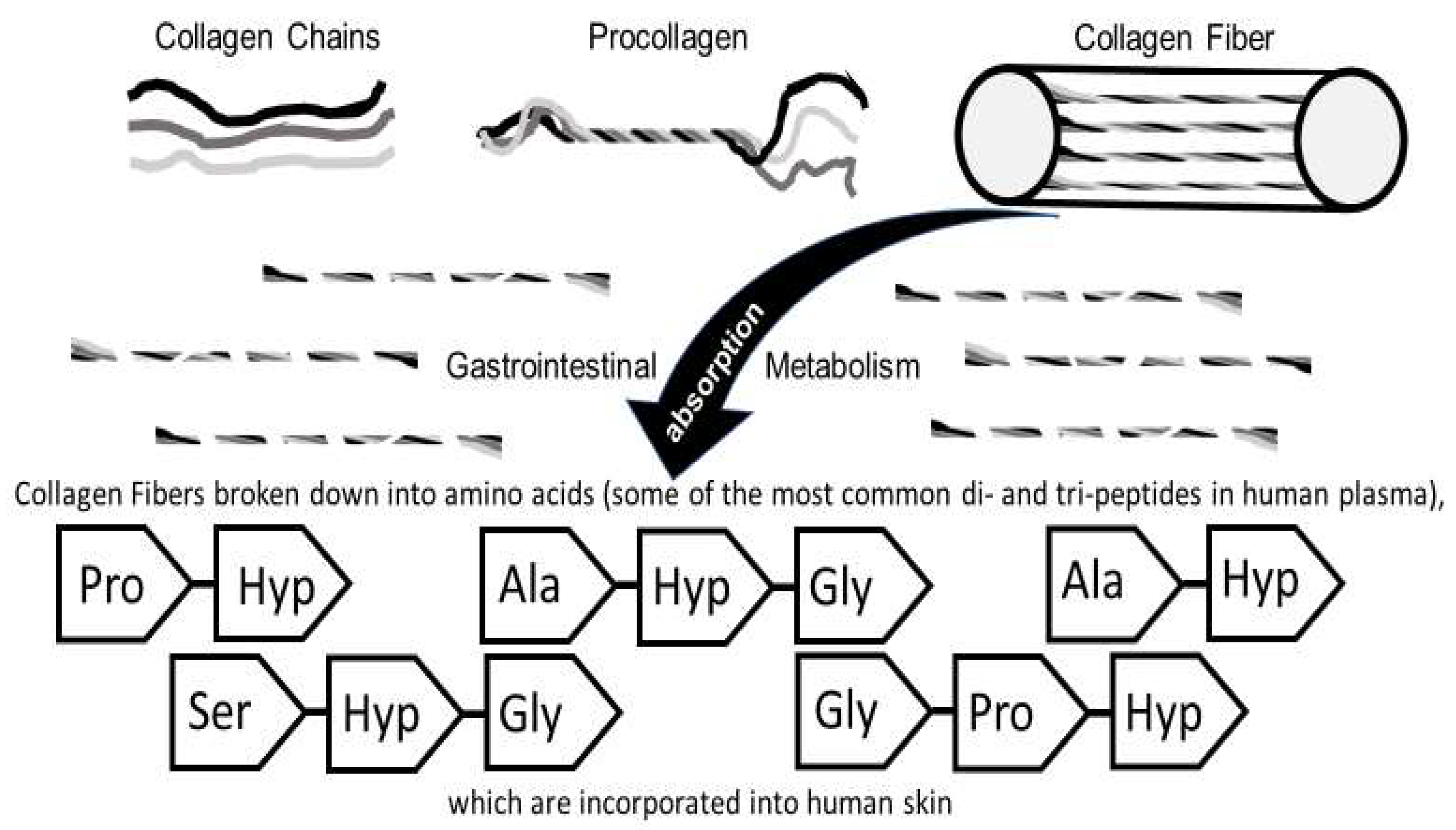
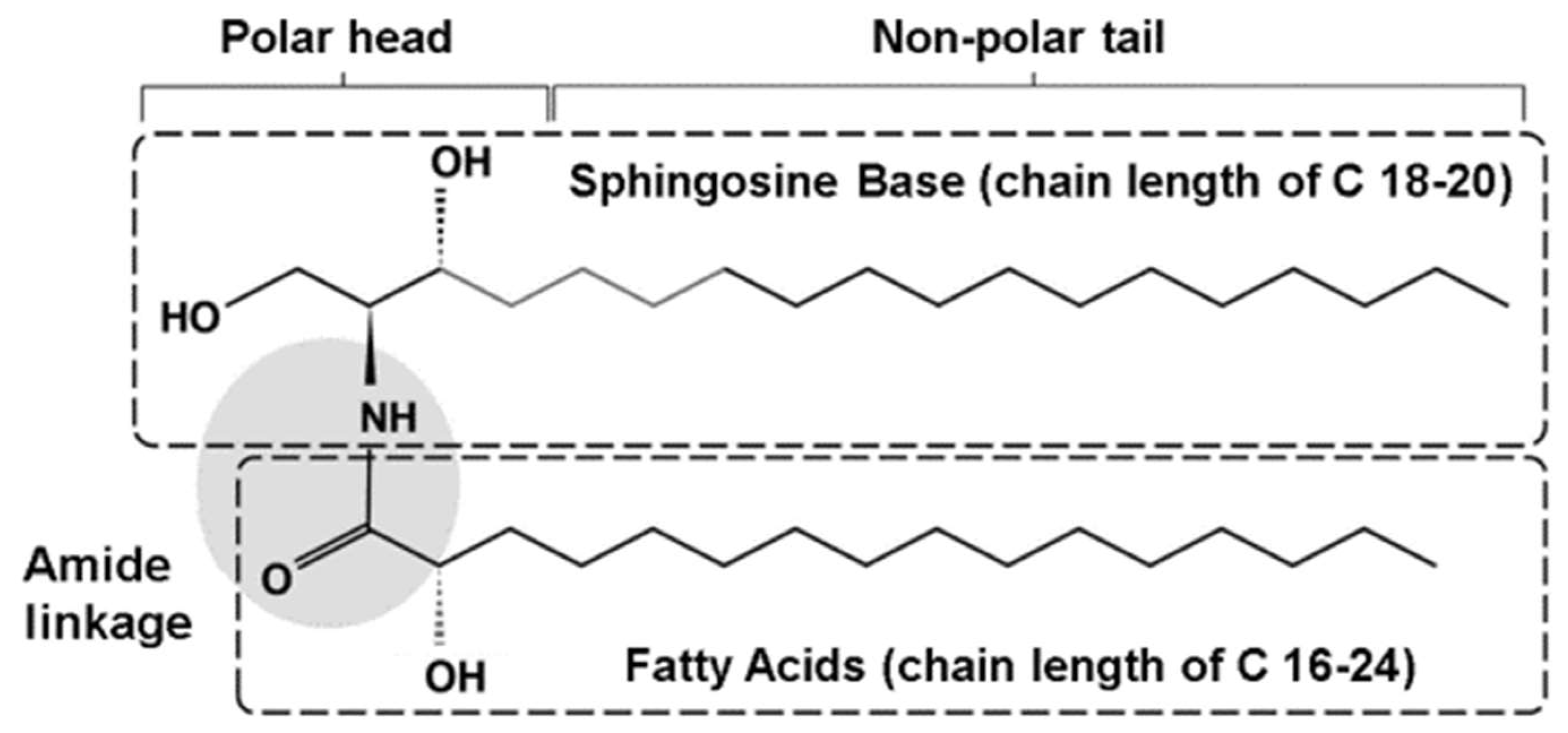
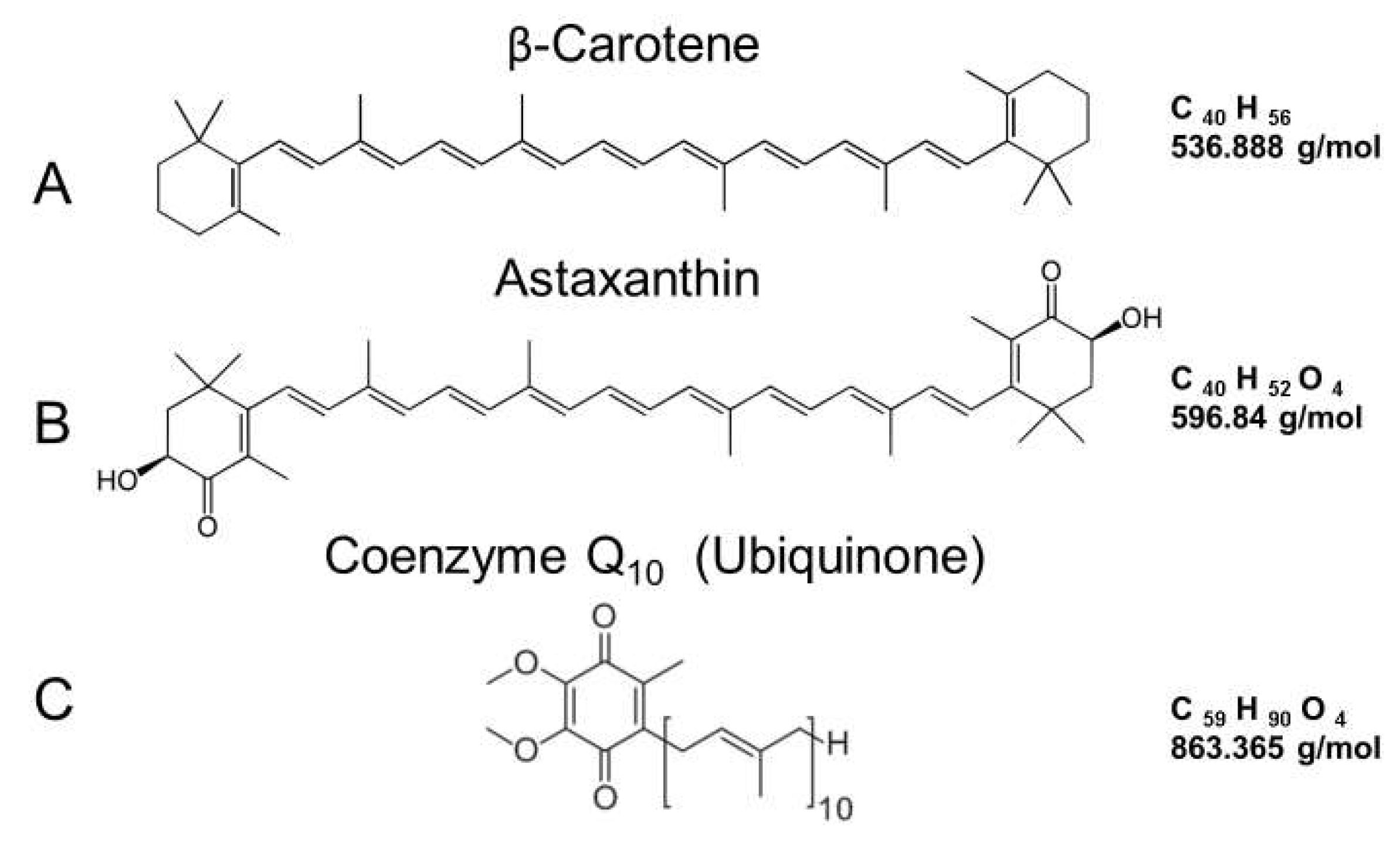
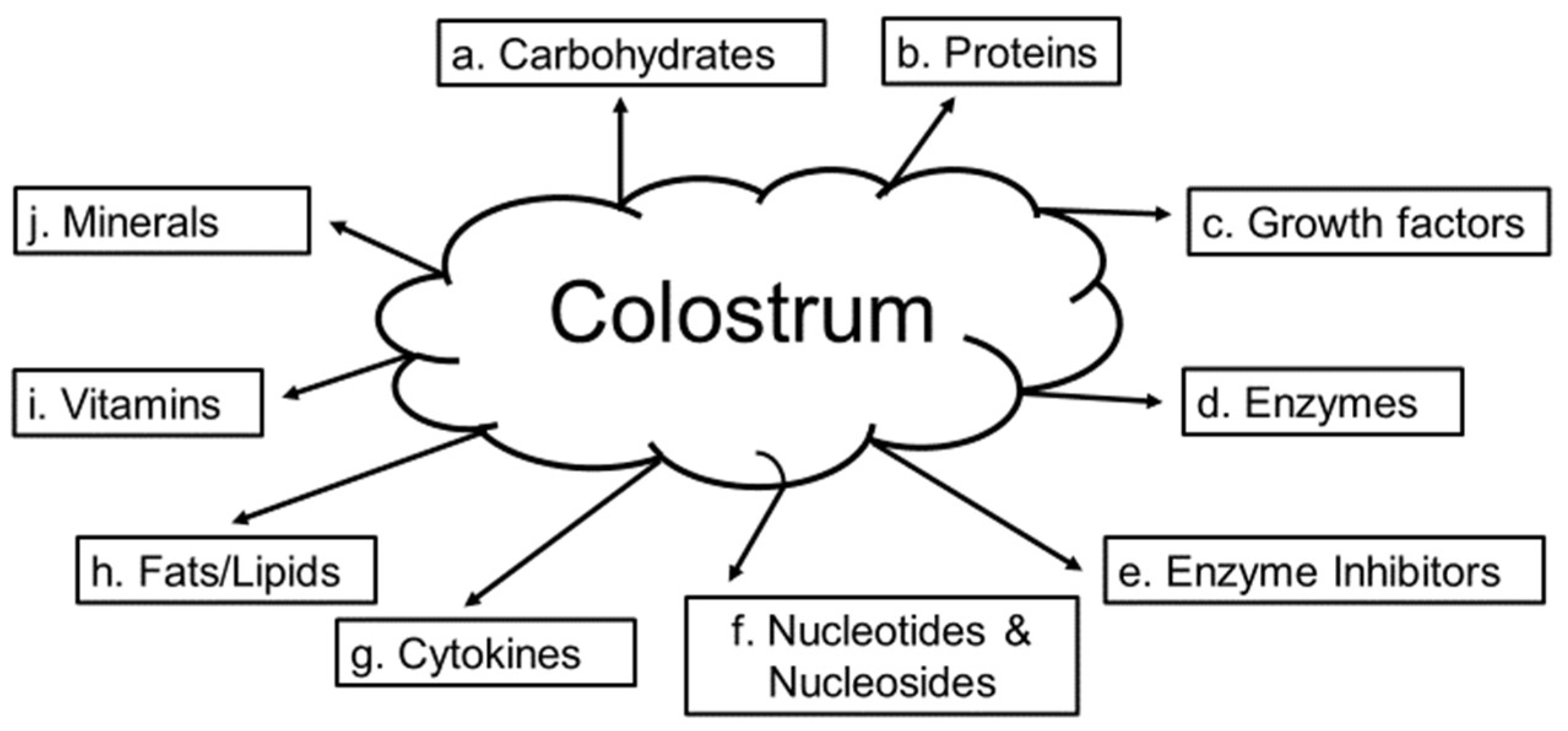
| Natural Compound or Mineral | Mechanism of Action(s) Involved in Maintaining Skin Health |
|---|---|
| 1. Collagen (Section 2) | building block of collagen and elastin fibers-improves skin and nail health; inhibits matrix metalloproteinases (MMPs); stimulates fibroblast function |
| 2. Ceramides (Section 3) | provides the major component of the lipid “mortar” of the stratum corneum essential in the structure and maintenance of skin barrierintegrity; also involved in cell proliferation, differentiation and apoptosis |
| 3. Beta Carotene (Section 4) | provitamin A molecule, acts as an antioxidant, anti-inflammatory agent and blocks ROS formation and/or ability to quench free radicals; prevents cellular damage, premature skin aging and skin cancer |
| 4. Astaxanthin (Section 5) | potent antioxidant; anti-inflammatory agent; prevents DNA damage & enhancemitochondrial function, provides UV protection; activates the Nrf2 pathway toto stimulate production of other antioxidants; inhibits MMPs; stimulates collagenproduction and wound healing |
| 5. Coenzyme Q10 (Section 6) | antioxidant; anti-aging properties-enhances collagen; potential treatment for psoriasis; accelerates generation of ATP levels after irradiation of fibroblasts |
| 6. Colostrum (Section 7) | contains, growth factors and other immune regulatory factors that promote growth of keratinocytes and wound healing |
| 7. Zinc (Section 8) | importance for skin morphogenesis, repair and maintenance such as wound healing |
| 8. Selenium (Section 9) | acts as a cofactor for glutathione peroxidase (GPX) removing harmful peroxides; involved in DNA synthesis and repair; prevents oxidative stress and UVB-radiation; also acts as an antioxidant |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vollmer, D.L.; West, V.A.; Lephart, E.D. Enhancing Skin Health: By Oral Administration of Natural Compounds and Minerals with Implications to the Dermal Microbiome. Int. J. Mol. Sci. 2018, 19, 3059. https://doi.org/10.3390/ijms19103059
Vollmer DL, West VA, Lephart ED. Enhancing Skin Health: By Oral Administration of Natural Compounds and Minerals with Implications to the Dermal Microbiome. International Journal of Molecular Sciences. 2018; 19(10):3059. https://doi.org/10.3390/ijms19103059
Chicago/Turabian StyleVollmer, David L., Virginia A. West, and Edwin D. Lephart. 2018. "Enhancing Skin Health: By Oral Administration of Natural Compounds and Minerals with Implications to the Dermal Microbiome" International Journal of Molecular Sciences 19, no. 10: 3059. https://doi.org/10.3390/ijms19103059
APA StyleVollmer, D. L., West, V. A., & Lephart, E. D. (2018). Enhancing Skin Health: By Oral Administration of Natural Compounds and Minerals with Implications to the Dermal Microbiome. International Journal of Molecular Sciences, 19(10), 3059. https://doi.org/10.3390/ijms19103059





