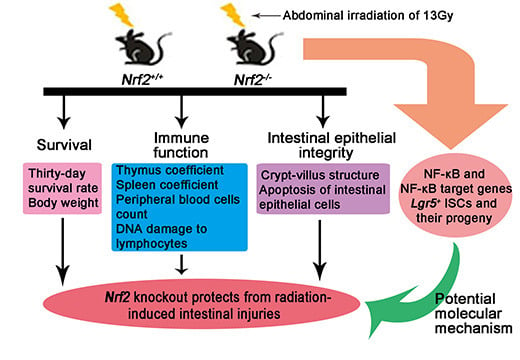
Nrf2-Knockout Protects from Intestinal Injuries in C57BL/6J Mice Following Abdominal Irradiation with γ Rays
Abstract
:1. Introduction
2. Results
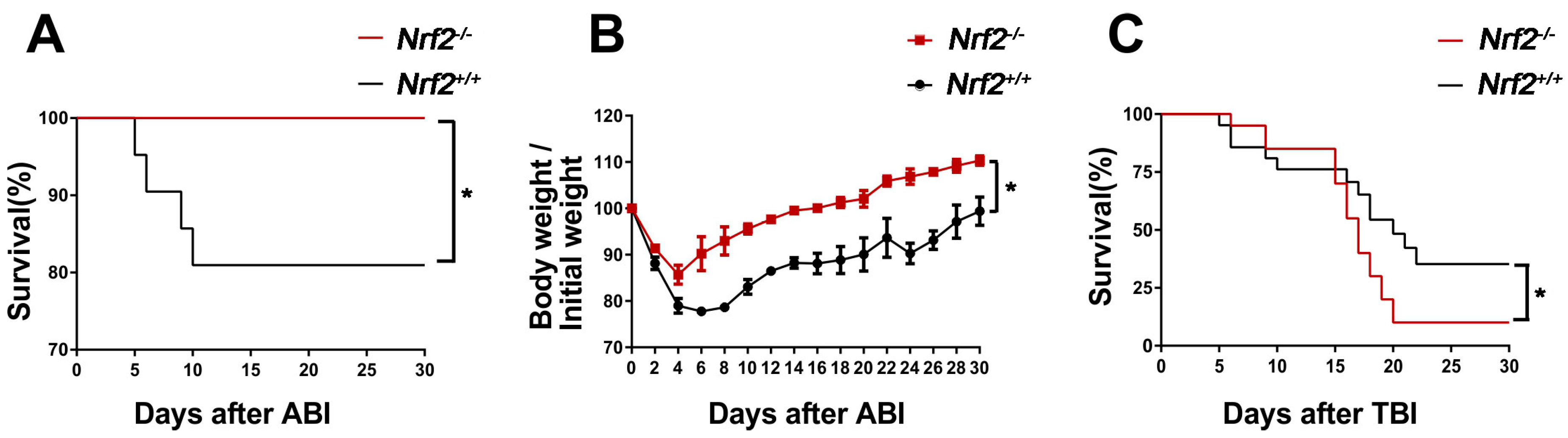
2.1. Nrf2 Knockout Improved the Survival of Mice after Abdominal Irradiation
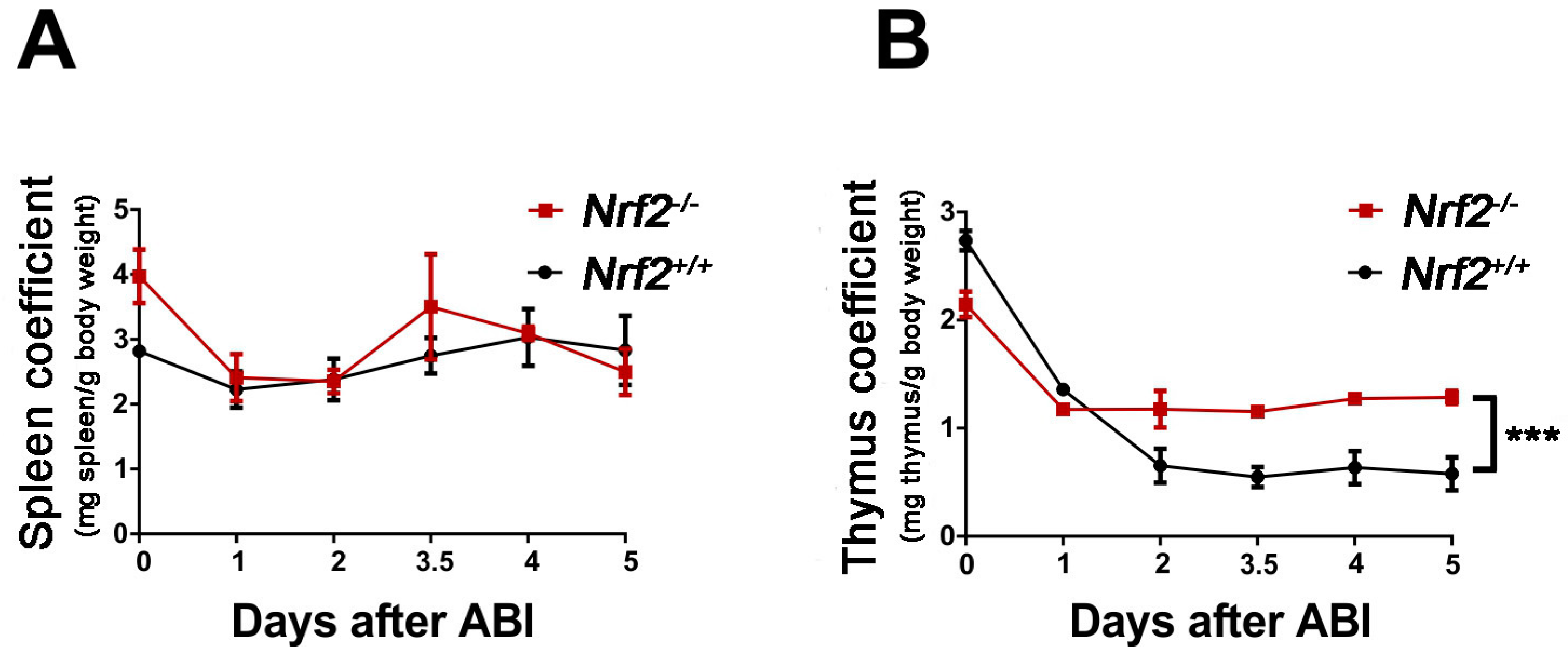
2.2. Nrf2 Knockout Increased the Immune Organ Coefficient of Mice after Abdominal Irradiation
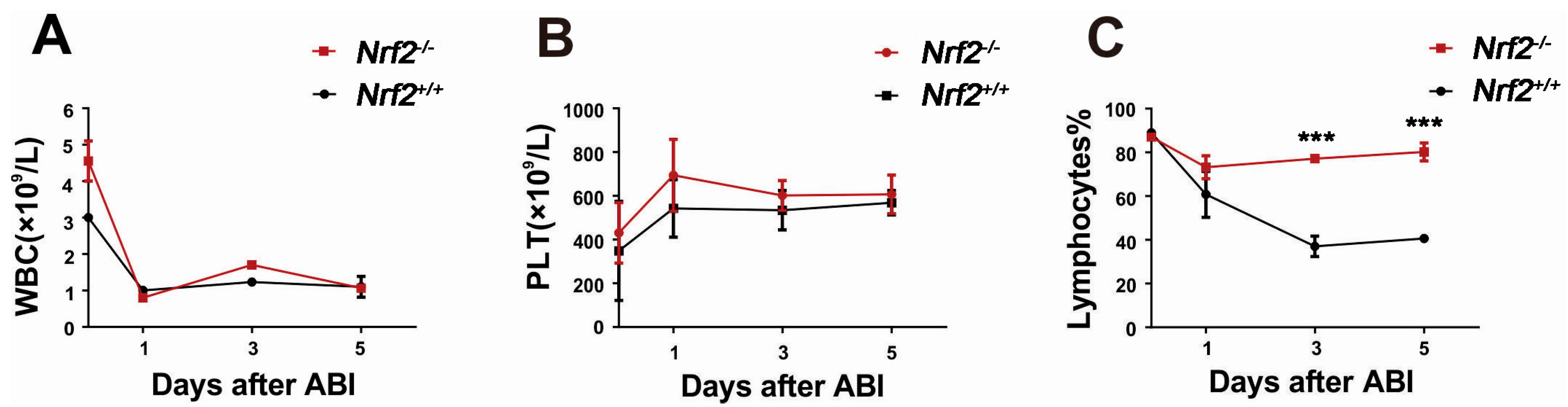
2.3. Nrf2 Knockout Mitigated Peripheral Blood Lymphocyte Count Decreases of Mice after Abdominal Irradiation
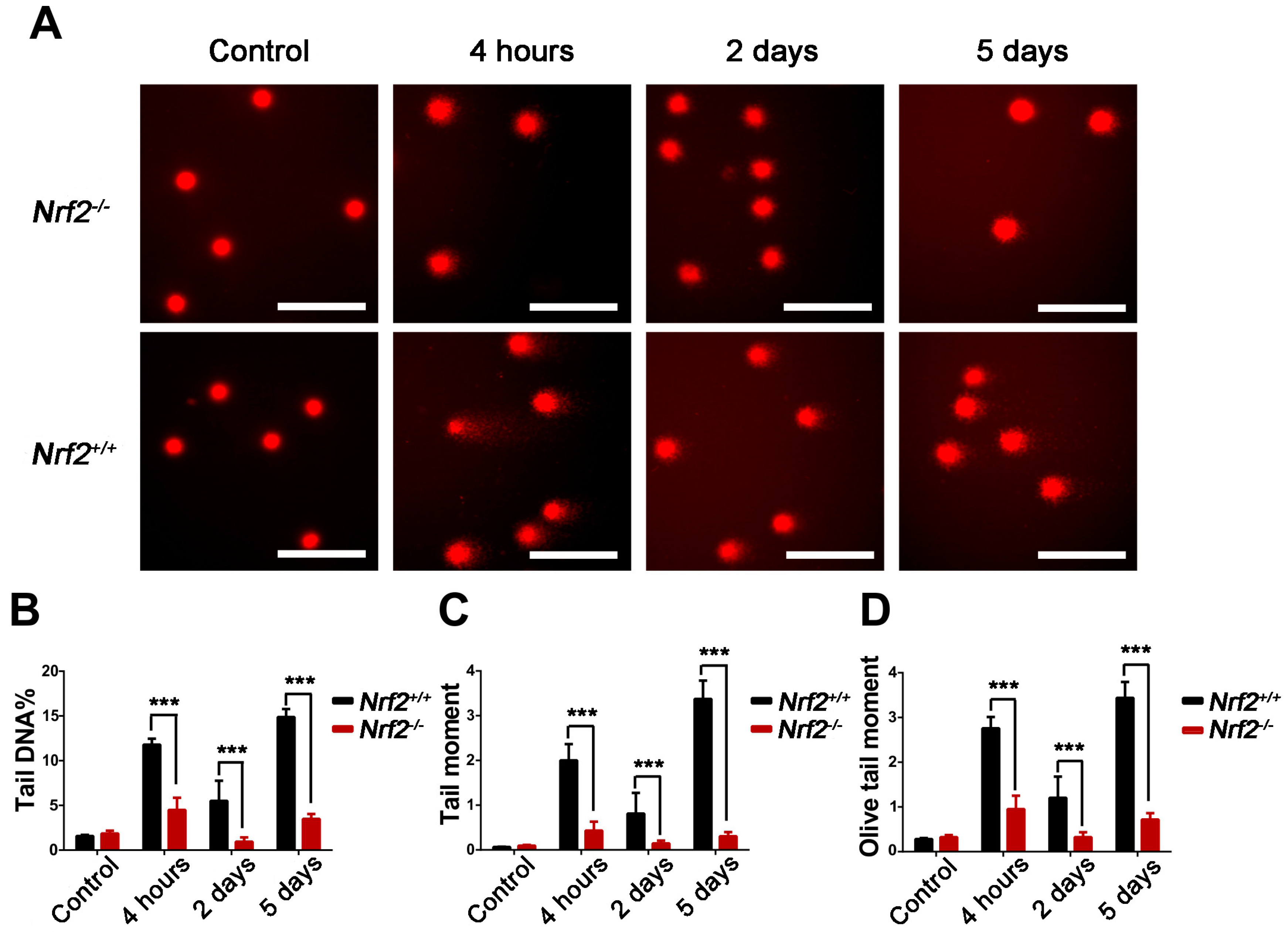
2.4. Nrf2 Knockout Attenuated DNA Damage to Peripheral Blood Lymphocytes of Mice After Abdominal Irradiation
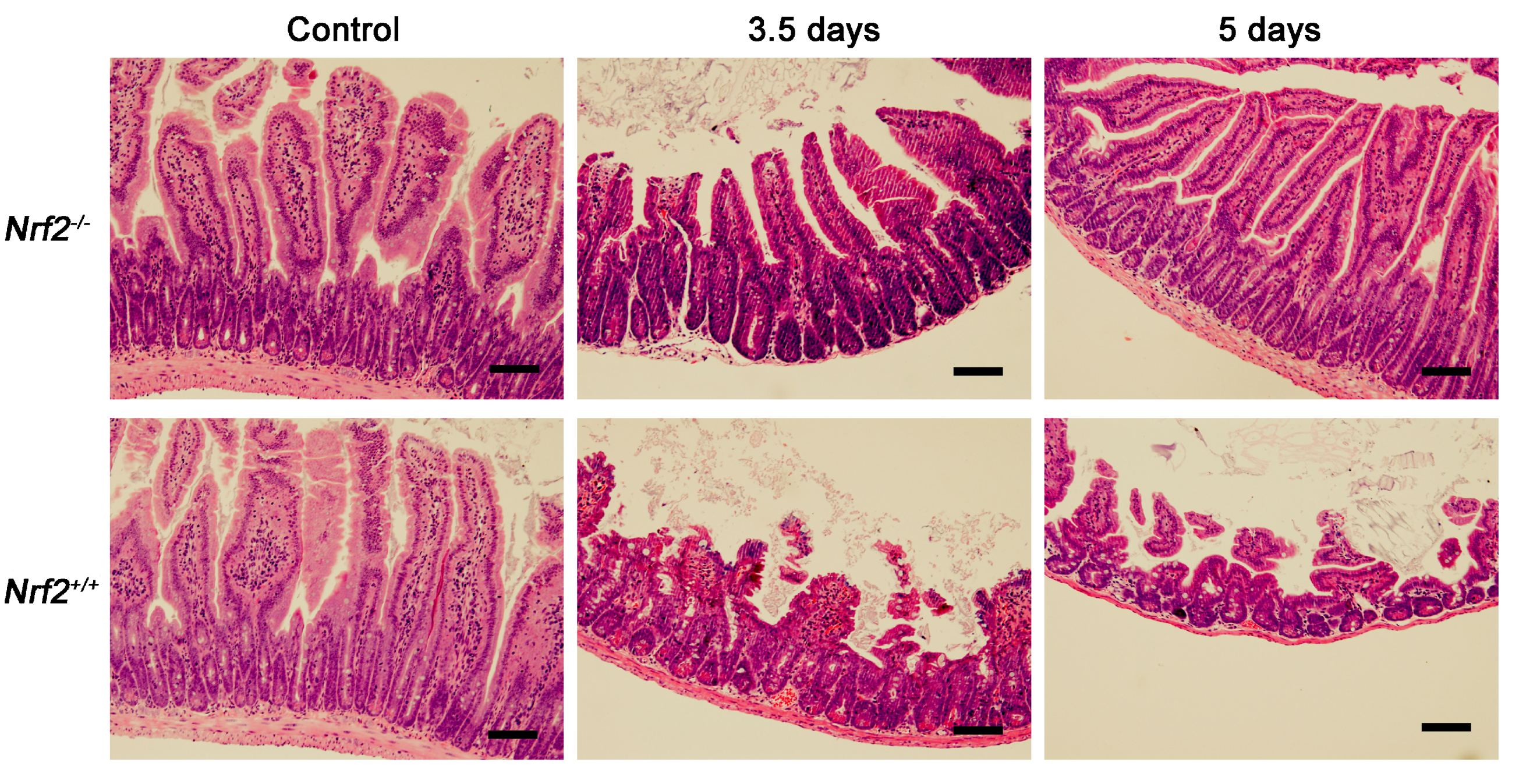
2.5. Nrf2 Knockout Alleviated the Damage to Crypt-Villus of the Small Intestine from Mice after Abdominal Irradiation
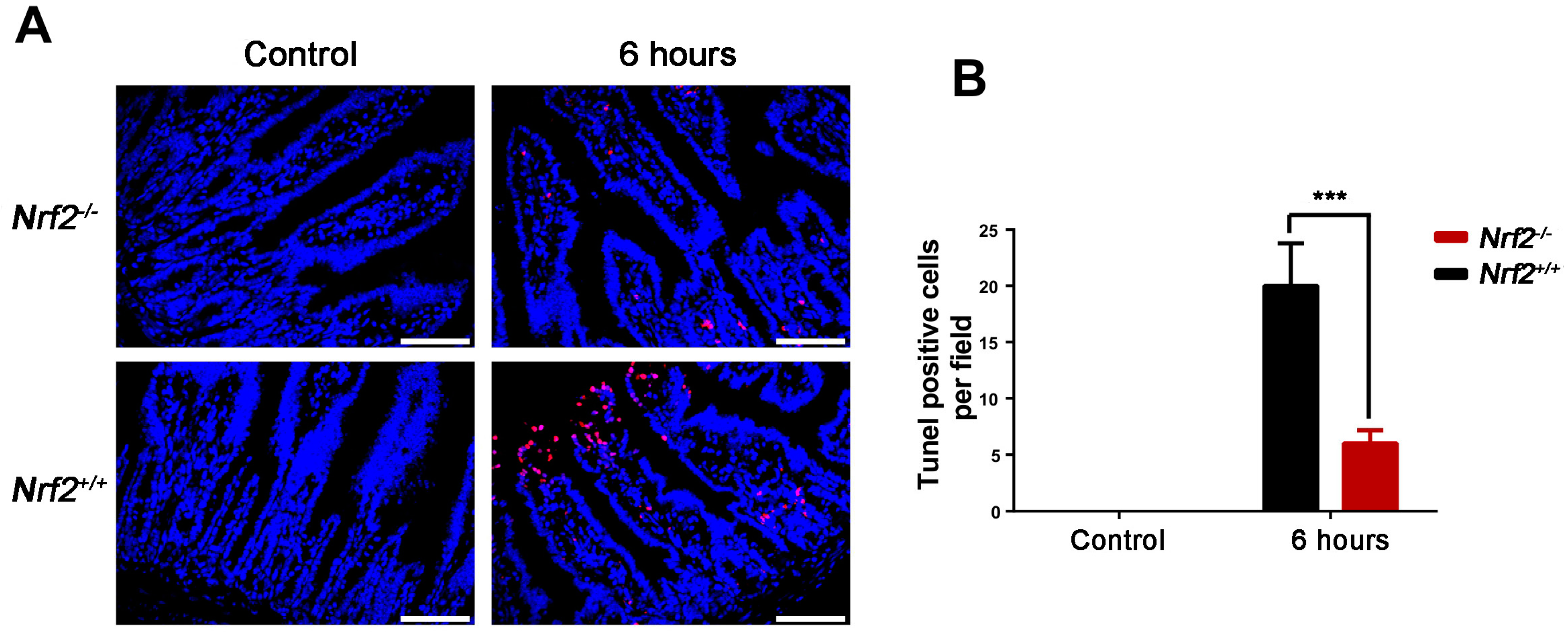
2.6. Nrf2 Knockout Decreased the Rate of Apoptosis of the Small Intestine from Mice after Abdominal Irradiation
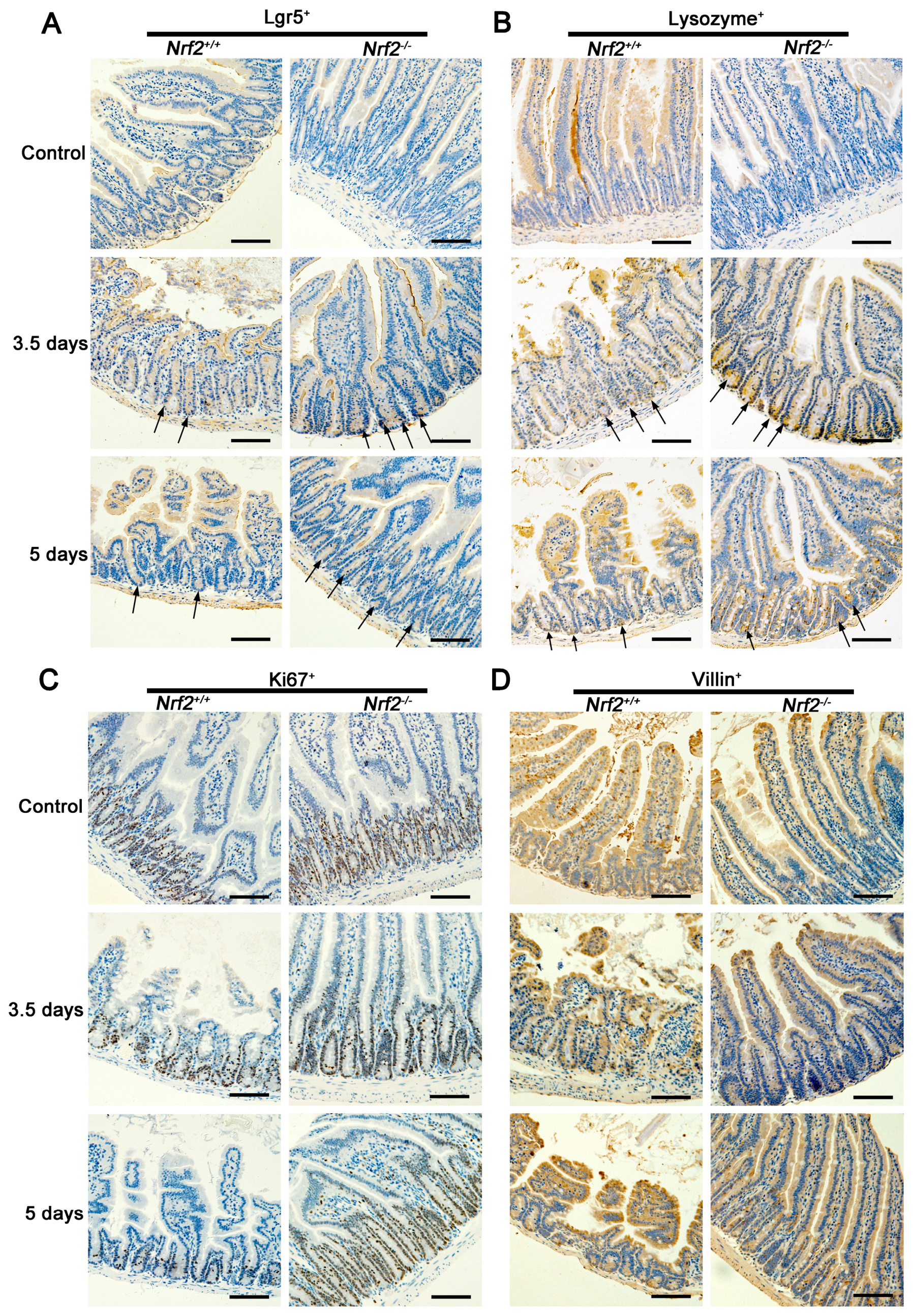
2.7. Nrf2 Knockout Resulted in an Increase in Lgr5+ Intestinal Stem Cells and Their Progeny of the Small Intestine from Mice after Abdominal Irradiation
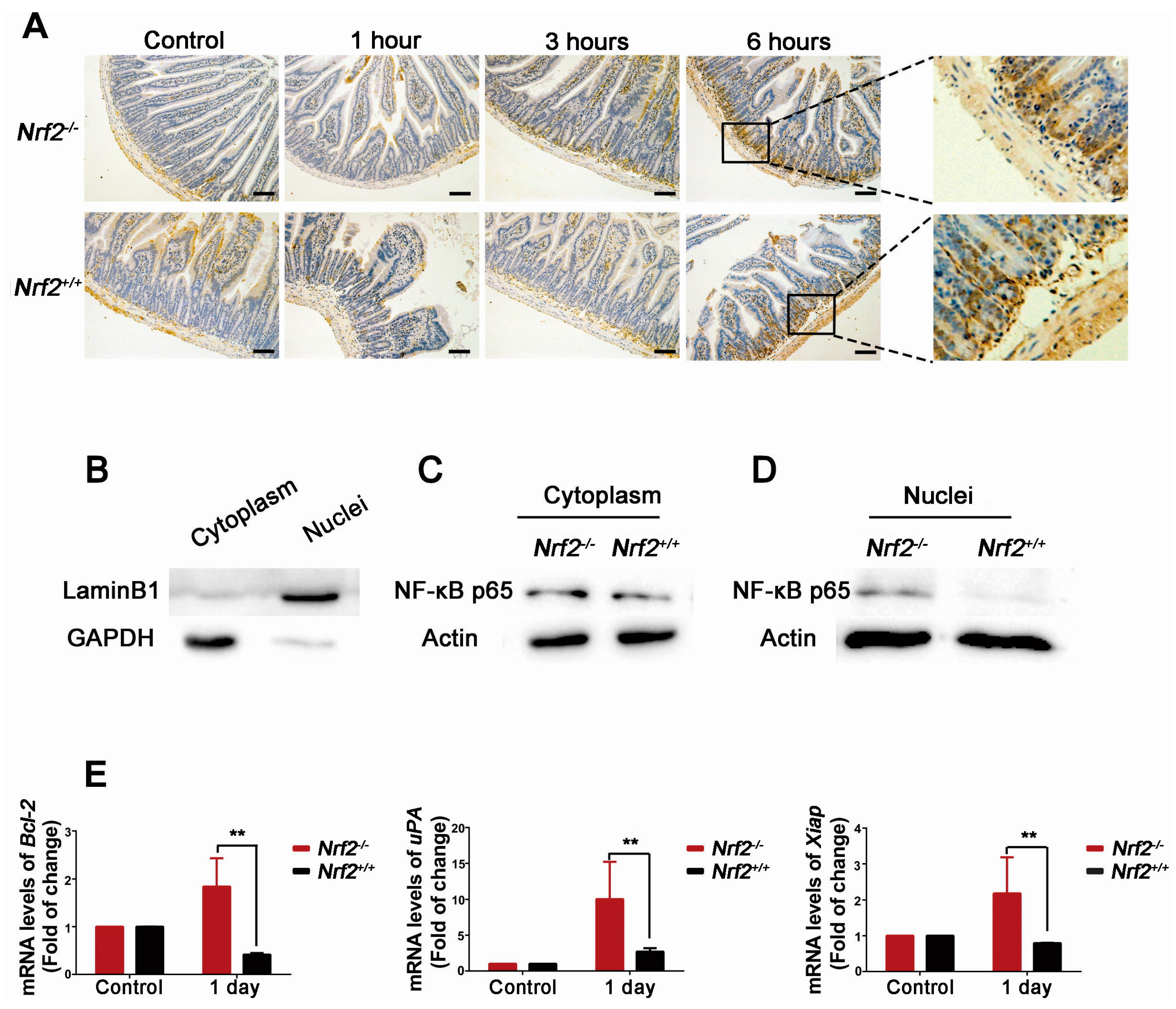
2.8. Nrf2 Knockout Enhanced the Activation of NF-κB of the Small Intestine from Mice after Abdominal Irradiation
3. Discussion
4. Materials and Methods
4.1. Mice
4.2. Irradiation of Mice
4.3. Analysis of the 30-Day Survival Rate
4.4. Organ Coefficient Calculation
4.5. Peripheral Blood Cells Counts
4.6. Analysis of DNA Damage to Peripheral Blood Lymphocytes
4.7. Pathological Analysis
4.8. Immunohistochemical Analysis
4.9. Apoptosis Assay
4.10. Isolation of Crypt Single Cells
4.11. Preparation of Cytoplasmic and Nuclear Extracts
4.12. Western Blot Analysis
4.13. RNA Isolation and Real-Time Polymerase Chain Reaction
4.14. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Potten, C.S. Radiation, the ideal cytotoxic agent for studying the cell biology of tissues such as the small intestine. Radiat. Res. 2004, 161, 123–136. [Google Scholar] [CrossRef] [PubMed]
- Moi, P.; Chan, K.; Asunis, I.; Cao, A.; Kan, Y.W. Isolation of NF-E2-related factor 2 (Nrf2), a NF-E2-like basic leucine zipper transcriptional activator that binds to the tandem NF-E2/AP1 repeat of the β-globin locus control region. Proc. Natl. Acad. Sci. USA. 1994, 91, 9926–9930. [Google Scholar] [CrossRef] [PubMed]
- Hirotsu, Y.; Katsuoka, F.; Funayama, R.; Nagashima, T.; Nishida, Y.; Nakayama, K.; Engel, J.D.; Yamamoto, M. Nrf2-MafG heterodimers contribute globally to antioxidant and metabolic networks. Nucleic Acids Res. 2012, 40, 10228–10239. [Google Scholar] [CrossRef] [PubMed]
- Hayes, J.D.; Dinkova-Kostova, A.T. The Nrf2 regulatory network provides an interface between redox and intermediary metabolism. Trends Biochem. Sci. 2014, 39, 199–218. [Google Scholar] [CrossRef] [PubMed]
- Rwigema, J.C.; Beck, B.; Wang, W.; Doemling, A.; Epperly, M.W.; Shields, D.; Goff, J.P.; Franicola, D.; Dixon, T.; Frantz, M.C.; et al. Two strategies for the development of mitochondrion-targeted small molecule radiation damage mitigators. Int. J. Radiat. Oncol. Biol. Phys. 2011, 80, 860–868. [Google Scholar] [CrossRef] [PubMed]
- Merchant, A.A.; Singh, A.; Matsui, W.; Biswal, S. The redox-sensitive transcription factor Nrf2 regulates murine hematopoietic stem cell survival independently of ROS levels. Blood 2011, 118, 6572–6579. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Mao, A.; Guo, R.; Zhang, L.; Yan, J.; Sun, C.; Tang, J.; Ye, Y.; Zhang, Y.; Zhang, H. Suppression of radiation-induced migration of non-small cell lung cancer through inhibition of Nrf2-Notch axis. Oncotarget 2017, 8, 36603–36613. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Mao, A.; Yan, J.; Sun, C.; Di, C.; Zhou, X.; Li, H.; Guo, R.; Zhang, H. Downregulation of Nrf2 promotes radiation-induced apoptosis through Nrf2 mediated Notch signaling in non-small cell lung cancer cells. Int. J. Oncol. 2016, 48, 765–773. [Google Scholar] [CrossRef] [PubMed]
- Jeong, Y.; Hoang, N.T.; Lovejoy, A.; Stehr, H.; Newman, A.M.; Gentles, A.J.; Kong, W.; Truong, D.; Martin, S.; Chaudhuri, A.; et al. Role of KEAP1/NRF2 and TP53 mutations in lung squamous cell carcinoma development and radiation resistance. Cancer Discov. 2017, 7, 86–101. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Thimmulappa, R.K.; Kumar, V.; Cui, W.; Kumar, S.; Kombairaju, P.; Zhang, H.; Margolick, J.; Matsui, W.; Macvittie, T.; et al. NRF2-mediated Notch pathway activation enhances hematopoietic reconstitution following myelosuppressive radiation. J. Clin. Investig. 2014, 124, 730–741. [Google Scholar] [CrossRef] [PubMed]
- Chute, J.P. NRF2 mitigates radiation-induced hematopoietic death. J. Clin. Investig. 2014, 124, 960–961. [Google Scholar] [CrossRef] [PubMed]
- Rana, T.; Schultz, M.A.; Freeman, M.L.; Biswas, S. Loss of Nrf2 accelerates ionizing radiation-induced bone loss by upregulating RANKL. Free Radic. Biol. Med. 2012, 53, 2298–2307. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.B.; Pandita, R.K.; Eskiocak, U.; Ly, P.; Kaisani, A.; Kumar, R.; Cornelius, C.; Wright, W.E.; Pandita, T.K.; Shay, J.W. Targeting of Nrf2 induces DNA damage signaling and protects colonic epithelial cells from ionizing radiation. Proc. Natl. Acad. Sci. USA. 2012, 109, E2949–E2955. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.B.; Ly, P.; Kaisani, A.; Zhang, L.; Wright, W.E.; Shay, J.W. Mitigation of radiation-induced damage by targeting EGFR in noncancerous human epithelial cells. Radiat. Res. 2013, 180, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Dutta, A.; Gupta, M.L.; Kalita, B. The combination of the active principles of Podophyllum hexandrum supports early recovery of the gastrointestinal system via activation of Nrf2–HO-1 signaling and the hematopoietic system, leading to effective whole-body survival in lethally irradiated mice. Free Radical Res. 2015, 49, 317–330. [Google Scholar]
- Wang, K.P.; Zhang, C.; Zhang, S.G.; Liu, E.D.; Dong, L.; Kong, X.Z.; Cao, P.; Hu, C.P.; Zhao, K.; Zhan, Y.Q.; et al. 3-(3-pyridylmethylidene)-2-indolinone reduces the severity of colonic injury in a murine model of experimental colitis. Oxid. Med. Cell Longev. 2015, 2015, 959253. [Google Scholar] [CrossRef] [PubMed]
- Berhane, H.; Epperly, M.W.; Cao, S.; Goff, J.P.; Franicola, D.; Wang, H.; Greenberger, J.S. Radioresistance of bone marrow stromal and hematopoietic progenitor cell lines derived from Nrf2−/− homozygous deletion recombinant-negative mice. In Vivo 2013, 27, 571–582. [Google Scholar] [PubMed]
- Kirsch, D.G.; Santiago, P.M.; di Tomaso, E.; Sullivan, J.M.; Hou, W.S.; Dayton, T.; Jeffords, L.B.; Sodha, P.; Mercer, K.L.; Cohen, R.; et al. p53 Controls radiation-induced gastrointestinal syndrome in mice independent of apoptosis. Science 2010, 327, 593–596. [Google Scholar] [CrossRef] [PubMed]
- Metcalfe, C.; Kljavin, N.M.; Ybarra, R.; de Sauvage, F.J. Lgr5+ stem cells are indispensable for radiation-induced intestinal regeneration. Cell Stem Cell 2014, 14, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Bhanja, P.; Saha, S.; Kabarriti, R.; Liu, L.; Roy-Chowdhury, N.; Roy-Chowdhury, J.; Sellers, R.S.; Alfieri, A.A.; Guha, C. Protective role of R-spondin1, an intestinal stem cell growth factor, against radiation-induced gastrointestinal syndrome in mice. Plos ONE 2009, 4, e8014. [Google Scholar] [CrossRef] [PubMed]
- Hua, G.; Thin, T.H.; Feldman, R.; Haimovitz-Friedman, A.; Clevers, H.; Fuks, Z.; Kolesnick, R. Crypt base columnar stem cells in small intestines of mice are radioresistant. Gastroenterology 2012, 143, 1266–1276. [Google Scholar] [CrossRef] [PubMed]
- Gong, W.; Guo, M.; Han, Z.; Wang, Y.; Yang, P.; Xu, C.; Wang, Q.; Du, L.; Li, Q.; Zhao, H.; et al. Mesenchymal stem cells stimulate intestinal stem cells to repair radiation-induced intestinal injury. Cell Death Dis. 2016, 7, e2387. [Google Scholar] [CrossRef] [PubMed]
- Umar, S. Intestinal stem cells. Curr. Gastroenterol. Rep. 2010, 12, 340–348. [Google Scholar] [CrossRef] [PubMed]
- Potten, C.S.; Merritt, A.; Hickman, J.; Hall, P.; Faranda, A. Characterization of radiation-induced apoptosis in the small intestine and its biological implications. Int. J. Radiat. Biol. 1994, 65, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Merritt, A.J.; Potten, C.S.; Kemp, C.J.; Hickman, J.A.; Balmain, A.; Lane, D.P.; Hall, P.A. The role of p53 in spontaneous and radiation-induced apoptosis in the gastrointestinal tract of normal and p53-deficient mice. Cancer Res. 1994, 54, 614–617. [Google Scholar] [PubMed]
- Ripp, T.; Ryabov, V.; Karpov, R. Significance changes endothelial dysfunction after enhanced external conterpulsation treatment in hypertensive patients with stable angina. J. Hypertens. 2007, 25, S59. [Google Scholar]
- Sato, T.; Clevers, H. Growing self-organizing mini-guts from a single intestinal stem cell: Mechanism and applications. Science 2013, 340, 1190–1194. [Google Scholar] [CrossRef] [PubMed]
- Sato, T.; Vries, R.G.; Snippert, H.J.; van de Wetering, M.; Barker, N.; Stange, D.E.; van Es, J.H.; Abo, A.; Kujala, P.; Peters, P.J.; et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 2009, 459, 262–265. [Google Scholar] [CrossRef] [PubMed]
- Jin, W.; Wang, H.; Yan, W.; Xu, L.; Wang, X.; Zhao, X.; Yang, X.; Chen, G.; Ji, Y. Disruption of Nrf2 enhances upregulation of nuclear factor-κB activity, proinflammatory cytokines, and intercellular adhesion molecule-1 in the brain after traumatic brain injury. Mediat. Inflamm. 2008, 2008, 725174. [Google Scholar] [CrossRef] [PubMed]
- Mao, L.; Wang, H.; Qiao, L.; Wang, X. Disruption of Nrf2 enhances the upregulation of nuclear factor-κB activity, tumor necrosis factor-α, and matrix metalloproteinase-9 after spinal cord injury in mice. Mediat. Inflamm. 2010, 2010, 238321. [Google Scholar] [CrossRef] [PubMed]
- Pan, H.; Wang, H.; Wang, X.; Zhu, L.; Mao, L. The absence of Nrf2 enhances NF-κB-dependent inflammation following scratch injury in mouse primary cultured astrocytes. Mediat. Inflamm. 2012, 2012, 217580. [Google Scholar] [CrossRef] [PubMed]
- Cuadrado, A.; Martín-Moldes, Z.; Ye, J.; Lastres-Becker, I. Transcription factors NRF2 and NF-κB are coordinated effectors of the Rho family, GTP-binding protein RAC1 during inflammation. J. Biol. Chem. 2014, 289, 15244–15258. [Google Scholar] [CrossRef] [PubMed]
- Jin, W.; Zhu, L.; Guan, Q.; Chen, G.; Wang, Q.F.; Yin, H.X.; Hang, C.H.; Shi, J.X.; Wang, H.D. Influence of Nrf2 genotype on pulmonary NF-κB activity and inflammatory response after traumatic brain injury. Ann. Clin. Lab. Sci. 2008, 38, 221–227. [Google Scholar] [PubMed]
- Wang, Y.; Meng, A.; Lang, H.; Brown, S.A.; Konopa, J.L.; Kindy, M.S.; Schmiedt, R.A.; Thompson, J.S.; Zhou, D. Activation of nuclear factor κB in vivo selectively protects the murine small intestine against ionizing radiation-induced damage. Cancer Res. 2004, 64, 6240–6246. [Google Scholar] [CrossRef] [PubMed]
- Van der Heijden, M.; Zimberlin, C.D.; Nicholson, A.M.; Colak, S.; Kemp, R.; Meijer, S.L.; Medema, J.P.; Greten, F.R.; Jansen, M.; Winton, D.J.; et al. Bcl-2 is a critical mediator of intestinal transformation. Nat. Commun. 2016, 7, 10916. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, B.B. Apoptosis and nuclear factor-κB: A tale of association and dissociation. Biochem. Pharmacol. 2000, 60, 1033–1039. [Google Scholar] [CrossRef]
- Hochmuth, C.E.; Biteau, D.; Bohmann, D.; Jasper, H. Redox regulation by Keap1 and Nrf2 controls intestinal stem cell proliferation in Drosophila. Cell Stem Cell 2011, 8, 188–199. [Google Scholar] [CrossRef] [PubMed]
- Tsai, J.J.; Dudakov, J.A.; Takahashi, K.; Shieh, J.H.; Velardi, E.; Holland, A.M.; Singer, N.V.; West, M.L.; Smith, O.M.; Young, L.F.; et al. Nrf2 regulates haematopoietic stem cell function. Nat. Cell Biol. 2013, 15, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Banath, J.P.; Fushiki, M.; Olive, P.L. Rejoining of DNA single- and double-strand breaks in human white blood cells exposed to ionizing radiation. Int. J. Radiat. Biol. 1998, 73, 649–660. [Google Scholar] [CrossRef] [PubMed]
- Konca, K.; Lankoff, A.; Banasik, A.; Lisowska, H.; Kuszewski, T.; Gozdz, S.; Koza, Z.; Wojcik, A. A cross-platform public domain PC image-analysis program for the comet assay. Mutat. Res. 2003, 534, 15–20. [Google Scholar] [CrossRef]
- Mahe, M.M.; Aihara, E.; Schumacher, M.A.; Zavros, Y.; Montrose, M.H.; Helmrath, M.A.; Sato, T.; Shroyer, N.F. Establishment of gastrointestinal epithelial organoids. Curr. Protoc. Mouse Biol. 2013, 3, 217–240. [Google Scholar] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, W.; Sun, Z.; Yang, B.; Wang, Q. Nrf2-Knockout Protects from Intestinal Injuries in C57BL/6J Mice Following Abdominal Irradiation with γ Rays. Int. J. Mol. Sci. 2017, 18, 1656. https://doi.org/10.3390/ijms18081656
Yang W, Sun Z, Yang B, Wang Q. Nrf2-Knockout Protects from Intestinal Injuries in C57BL/6J Mice Following Abdominal Irradiation with γ Rays. International Journal of Molecular Sciences. 2017; 18(8):1656. https://doi.org/10.3390/ijms18081656
Chicago/Turabian StyleYang, Wenyan, Zhijuan Sun, Bing Yang, and Qin Wang. 2017. "Nrf2-Knockout Protects from Intestinal Injuries in C57BL/6J Mice Following Abdominal Irradiation with γ Rays" International Journal of Molecular Sciences 18, no. 8: 1656. https://doi.org/10.3390/ijms18081656
APA StyleYang, W., Sun, Z., Yang, B., & Wang, Q. (2017). Nrf2-Knockout Protects from Intestinal Injuries in C57BL/6J Mice Following Abdominal Irradiation with γ Rays. International Journal of Molecular Sciences, 18(8), 1656. https://doi.org/10.3390/ijms18081656