Antibacterial and Antifungal Activities of Spices
Abstract
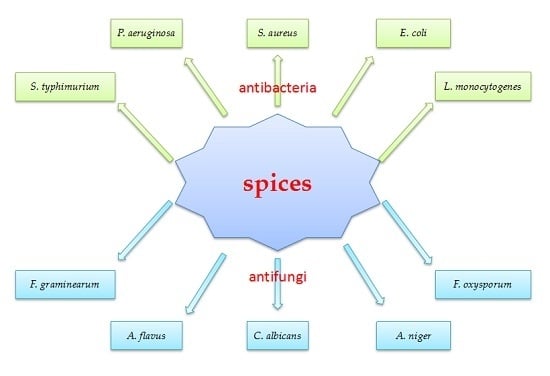
:1. Introduction
2. Clove
2.1. Antimicrobial Activities of Clove
2.2. Comparison of Antimicrobial Activities of Clove and Other Spices
2.3. The Application of Clove as Antimicrobial Agents in Food Packaging
3. Oregano
3.1. Antimicrobial Activities of Oregano
3.2. Comparison of Antimicrobial Activities of Oregano and Other Spices
3.3. The Application of Oregano as Antimicrobial Agents in Food Packaging
4. Thyme
4.1. Antimicrobial Activities of Thyme
4.2. Comparison of Antimicrobial Activities of Thyme and Other Spices
5. Cinnamon
5.1. Antimicrobial Activities of Cinnamon
5.2. Comparison of Antimicrobial Activities of Cinnamon and Other Spices
6. Cumin
6.1. Antimicrobial Activities of Cumin
6.2. Comparison of Antimicrobial Activities of Cumin and Other Spices
7. Rosemary
7.1. Antimicrobial Activities of Rosemary
7.2. Comparison of Antimicrobial Activities of Rosemary and Other Spices
8. Garlic
8.1. Antimicrobial Activities of Garlic
8.2. Comparison of Antimicrobial Activities of Garlic and Other Spices
9. Ginger
9.1. Antimicrobial Activities of Ginger
9.2. Comparison of Antimicrobial Activities of Ginger and Other Spices
10. Basil
10.1. Antimicrobial Activities of Basil
10.2. Comparison of Antimicrobial Activities of Basil and Other Spices
11. Fennel
11.1. Antimicrobial Activities of Fennel
11.2. Comparison of Antimicrobial Activities of Fennel and Other Spices
12. Coriander
12.1. Antimicrobial Activities of Coriander
12.2. Comparison of Antimicrobial Activities of Coriander and Other Spices
13. Galangal
14. Black Pepper
15. Other Spices
16. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Miladi, H.; Zmantar, T.; Chaabouni, Y.; Fedhila, K.; Bakhrouf, A.; Mandouani, K.; Chaieb, K. Antibacterial and efflux pump inhibitors of thymol and carvacrol against food-borne pathogens. Microb. Pathog. 2016, 99, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Brul, S.; Coote, P. Preservative agents in foods. Mode of action and microbial resistance mechanisms. Int. J. Food Microbiol. 1999, 50, 1–17. [Google Scholar] [CrossRef]
- De Souza, E.L.; Stamford, T.; Lima, E.D.; Trajano, V.N.; Barbosa, J.M. Antimicrobial effectiveness of spices: An approach for use in food conservation systems. Braz. Arch. Biol. Technol. 2005, 48, 549–558. [Google Scholar] [CrossRef]
- Silva, F.; Domingues, F.C. Antimicrobial activity of coriander oil and its effectiveness as food preservative. Crit. Rev. Food Sci. Nutr. 2017, 57, 35–47. [Google Scholar] [CrossRef] [PubMed]
- Tajkarimi, M.M.; Ibrahim, S.A.; Cliver, D.O. Antimicrobial herb and spice compounds in food. Food Control 2010, 21, 1199–1218. [Google Scholar] [CrossRef]
- Nabavi, S.M.; Marchese, A.; Izadi, M.; Curti, V.; Daglia, M.; Nabavi, S.F. Plants belonging to the genus Thymus as antibacterial agents: From farm to pharmacy. Food Chem. 2015, 173, 339–347. [Google Scholar] [CrossRef] [PubMed]
- Marchese, A.; Barbieri, R.; Sanches-Silva, A.; Daglia, M.; Nabavi, S.F.; Jafari, N.J.; Izadi, M.; Ajami, M.; Nabavi, S.M. Antifungal and antibacterial activities of allicin: A review. Trends Food Sci. Technol. 2016, 52, 49–56. [Google Scholar] [CrossRef]
- Paphitou, N.I. Antimicrobial resistance: Action to combat the rising microbial challenges. Int. J. Antimicrob. Agents 2013, 42, S25–S28. [Google Scholar] [CrossRef] [PubMed]
- Högberg, L.D.; Heddini, A.; Cars, O. The global need for effective antibiotics: Challenges and recent advances. Trends Pharmacol. Sci. 2010, 31, 509–515. [Google Scholar] [CrossRef] [PubMed]
- Lai, P.K.; Roy, J. Antimicrobial and chemopreventive properties of herbs and spices. Curr. Med. Chem. 2004, 11, 1451–1460. [Google Scholar] [CrossRef] [PubMed]
- Nabavi, S.F.; di Lorenzo, A.; Izadi, M.; Sobarzo-Sánchez, E.; Daglia, M.; Nabavi, S.M. Antibacterial effects of cinnamon: From farm to food, cosmetic and pharmaceutical industries. Nutrients 2015, 7, 7729–7748. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Zhou, Y.; Li, Y.; Xu, D.P.; Li, S.; Li, H.B. Spices for prevention and treatment of cancers. Nutrients 2016, 8, 495. [Google Scholar] [CrossRef] [PubMed]
- De, M.; De, A.K.; Banerjee, A.B. Antimicrobial screening of some Indian spices. Phytother. Res. 1999, 13, 616–618. [Google Scholar] [CrossRef]
- Arora, D.S.; Kaur, J. Antimicrobial activity of spices. Int. J. Antimicrob. Agents 1999, 12, 257–262. [Google Scholar] [CrossRef]
- Chaieb, K.; Hajlaoui, H.; Zmantar, T.; Kahla-Nakbi, A.B.; Rouabhia, M.; Mahdouani, K.; Bakhrouf, A. The chemical composition and biological activity of clove essential oil, Eugenia caryophyllata (Syzigium aromaticum L. Myrtaceae): A short review. Phytother. Res. 2007, 21, 501–506. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.G.; Liu, T.; Hu, Q.P.; Cao, X.M. Chemical composition, antibacterial properties and mechanism of action of essential oil from clove buds against Staphylococcus aureus. Molecules 2016, 21, 1194. [Google Scholar] [CrossRef] [PubMed]
- Lomarat, P.; Phanthong, P.; Wongsariya, K.; Chomnawang, M.T.; Bunyapraphatsara, N. Bioautography-guided isolation of antibacterial compounds of essential oils from Thai spices against histamine-producing bacteria. Pak. J. Pharm. Sci. 2013, 26, 473–477. [Google Scholar] [PubMed]
- Nassan, M.A.; Mohamed, E.H.; Abdelhafez, S.; Ismail, T.A. Effect of clove and cinnamon extracts on experimental model of acute hematogenous pyelonephritis in albino rats: Immunopathological and antimicrobial study. Int. J. Immunopathol. Pharmacol. 2015, 28, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Kuang, X.; Li, B.; Kuang, R.; Zheng, X.D.; Zhu, B.; Xu, B.L.; Ma, M.H. Granularity and antibacterial activities of ultra-fine cinnamon and clove powders. J. Food Saf. 2011, 31, 291–296. [Google Scholar] [CrossRef]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef] [PubMed]
- Badei, A.; Faheld, S.; El-Akel, A.; Mahmoud, B. Application of some spices in flavoring and preservation of cookies: 2-Antimicrobial and sensory properties of cardamom, cinnamon and clove. Dtsch. Lebensm. Rundsch. 2002, 98, 261–265. [Google Scholar]
- Schmidt, E.; Jirovetz, L.; Wlcek, K.; Buchbauer, G.; Gochev, V.; Girova, T.; Stoyanova, A.; Geissler, M. Antifungal activity of eugenol and various eugenol-containing essential oils against 38 clinical isolates of Candida albicans. J. Essent. Oil Bear. Plants 2007, 10, 421–429. [Google Scholar] [CrossRef]
- Angienda, P.O.; Onyango, D.M.; Hill, D.J. Potential application of plant essential oils at sub-lethal concentrations under extrinsic conditions that enhance their antimicrobial effectiveness against pathogenic bacteria. Afr. J. Microbiol. Res. 2010, 4, 1678–1684. [Google Scholar]
- Shan, B.; Cai, Y.Z.; Brooks, J.D.; Corke, H. Antibacterial and antioxidant effects of five spice and herb extracts as natural preservatives of raw pork. J. Sci. Food Agric. 2009, 89, 1879–1885. [Google Scholar] [CrossRef]
- Bayoub, K.; Baibai, T.; Mountassif, D.; Retmane, A.; Soukri, A. Antibacterial activities of the crude ethanol extracts of medicinal plants against Listeria monocytogenes and some other pathogenic strains. Afr. J. Biotechnol. 2010, 9, 4251–4258. [Google Scholar]
- Cui, H.Y.; Gabriel, A.A.; Nakano, H. Antimicrobial efficacies of plant extracts and sodium nitrite against Clostridium botulinum. Food Control 2010, 21, 1030–1036. [Google Scholar] [CrossRef]
- Keskin, D.; Toroglu, S. Studies on antimicrobial activities of solvent extracts of different spices. J. Environ. Biol. 2011, 32, 251–256. [Google Scholar] [PubMed]
- Liang, Z.W.; Cheng, Z.H.; Mittal, G.S. Inactivation of microorganisms in apple cider using spice powders, extracts and oils as antimicrobials with and without low-energy pulsed electric field. J. Food Agric. Environ. 2003, 1, 28–33. [Google Scholar]
- Badhe, S.R.; Fairoze, M.N. Antibacterial efficacy of clove powder of chicken legs spiked with pathogenic reference strains under refrigeration temperature (8 ± 2 °C). Indian J. Anim. Res. 2012, 46, 371–375. [Google Scholar]
- Hernández-Ochoa, L.; Gonzales-Gonzales, A.; Gutierrez-Mendez, N.; Munoz-Castellanos, L.N.; Quintero-Ramos, A. Study of the antibacterial activity of chitosan-based films prepared with different molecular weights including spices essential oils and functional extracts as antimicrobial agents. Rev. Mex. Ing. Quim. 2011, 10, 455–463. [Google Scholar]
- Hernández-Ochoa, L.; Macías-Castañeda, C.A.; Nevárez-Moorillón, G.V.; Salas-Muñoz, E.; Sandoval-Salas, F. Antimicrobial activity of chitosan-based films including spices’ essential oils and functional extracts. CyTA J. Food 2012, 10, 85–91. [Google Scholar] [CrossRef]
- Liu, G.Q.; Zhang, L.L.; Zong, K.; Wang, A.M.; Yu, X.F. Effects of spices essential oils on the spoilage-related microbiota in chilled pork stored in antimicrobial pack. Food Sci. Technol. Res. 2012, 18, 695–704. [Google Scholar] [CrossRef]
- Rodriguez-Garcia, I.; Silva-Espinoza, B.A.; Ortega-Ramirez, L.A.; Leyva, J.M.; Siddiqui, M.W.; Cruz-Valenzuela, M.R.; Gonzalez-Aguilar, G.A.; Ayala-Zavala, J.F. Oregano essential oil as an antimicrobial and antioxidant additive in food products. Crit. Rev. Food Sci. Nutr. 2016, 56, 1717–1727. [Google Scholar] [CrossRef] [PubMed]
- Babacan, O.; Cengiz, S.; Akan, M. Detection of antibacterial effect of oregano plant on various Salmonella serotypes. Ank. Univ. Vet. Fak. Derg. 2012, 59, 103–106. [Google Scholar]
- Santoyo, S.; Cavero, S.; Jaime, L.; Ibanez, E.; Senorans, F.J.; Reglero, G. Supercritical carbon dioxide extraction of compounds with antimicrobial activity from Origanum vulgare L.: Determination of optimal extraction parameters. J. Food Prot. 2006, 69, 369–375. [Google Scholar] [CrossRef] [PubMed]
- De Souza, E.L.; Stamford, T.; Lima, E.; Barbosa, J.M.; Marques, M. Interference of heating on the antimicrobial activity and chemical composition of Origanum vulgare L. (Lamiaceae) essential oil. Cienc. Tecnol. Aliment. 2008, 28, 418–422. [Google Scholar] [CrossRef]
- Lima, I.O.; Pereira, F.D.O.; de Oliveira, W.A.; Lima, E.D.O.; Menezes, E.A.; Cunha, F.A.; Formiga Melo Diniz, M.D.F. Antifungal activity and mode of action of carvacrol against Candida albicans strains. J. Essent. Oil Res. 2013, 25, 138–142. [Google Scholar] [CrossRef]
- Ultee, A.; Kets, E.; Smid, E.J. Mechanisms of action of carvacrol on the food-borne pathogen Bacillus cereus. Appl. Environ. Microb. 1999, 65, 4606–4610. [Google Scholar]
- Ozcan, M.; Boyraz, N. Antifungal properties of some herb decoctions. Eur. Food Res. Technol. 2000, 212, 86–88. [Google Scholar]
- Ai-Turki, A.I.; Ei-Ziney, M.G.; Abdel-Salam, A.M. Chemical and anti-bacterial characterization of aqueous extracts of oregano, marjoram, sage and licorice and their application in milk and labneh. J. Food Agric. Environ. 2008, 6, 39–44. [Google Scholar]
- Marques, J.D.; Volcao, L.M.; Funck, G.D.; Kroning, I.S.; da Silva, W.P.; Fiorentini, A.M.; Ribeiro, G.A. Antimicrobial activity of essential oils of Origanum vulgare L. and Origanum majorana L. against Staphylococcus aureus isolated from poultry meat. Ind. Crop. Prod. 2015, 77, 444–450. [Google Scholar] [CrossRef]
- Bozin, B.; Mimica-Dukic, N.; Simin, N.; Anackov, G. Characterization of the volatile composition of essential oils of some Lamiaceae spices and the antimicrobial and antioxidant activities of the entire oils. J. Agric. Food Chem. 2006, 54, 1822–1828. [Google Scholar] [CrossRef] [PubMed]
- Viuda-Martos, M.; Ruiz-Navajas, Y.; Fernández-López, J.; Pérez-Álvarez, J.A. Antibacterial activity of different essential oils obtained from spices widely used in Mediterranean diet. Int. J. Food Sci. Technol. 2008, 43, 526–531. [Google Scholar] [CrossRef]
- Santurio, D.F.; da Costa, M.M.; Maboni, G.; Cavalheiro, C.P.; de Sá, M.F.; Dal Pozzo, M.; Alves, S.H.; Fries, L. Antimicrobial activity of spice essential oils against Escherichia coli strains isolated from poultry and cattle. Cienc. Rural 2011, 41, 1051–1056. [Google Scholar] [CrossRef]
- Khosravi, A.R.; Shokri, H.; Kermani, S.; Dakhili, M.; Madani, M.; Parsa, S. Antifungal properties of Artemisia sieberi and Origanum vulgare essential oils against Candida glabrata isolates obtained from patients with vulvovaginal candidiasis. J. Mycol. Med. 2011, 21, 93–99. [Google Scholar] [CrossRef]
- Dal Pozzo, M.; Viégas, J.; Santurio, D.F.; Rossatto, L.; Soares, I.H.; Alves, S.H.; da Costa, M.M. Antimicrobial activities of essential oils extracted from spices against Staphylococcus spp. isolated from goat mastitis. Cienc. Rural 2011, 41, 667–672. [Google Scholar] [CrossRef]
- Santos, J.C.; Carvalho, C.D.; Barros, T.F.; Guimaraes, A.G. In vitro antimicrobial activity of essential oils from oregano, garlic, clove and lemon against pathogenic bacteria isolated from Anomalocardia brasiliana. Semin. Cienc. Agrar. 2011, 32, 1557–1564. [Google Scholar]
- Hyun, J.E.; Bae, Y.M.; Song, H.; Yoon, J.H.; Lee, S.Y. Antibacterial effect of various essential oils against pathogens and spoilage microorganisms in fresh produce. J. Food Saf. 2015, 35, 206–219. [Google Scholar] [CrossRef]
- Zivanovic, S.; Chi, S.; Draughon, A.F. Antimicrobial activity of chitosan films enriched with essential oils. J. Food Sci. 2005, 70, M45–M51. [Google Scholar] [CrossRef]
- Assiri, A.M.A.; Elbanna, K.; Abulreesh, H.H.; Ramadan, M.F. Bioactive compounds of cold-pressed thyme (Thymus vulgaris) oil with antioxidant and antimicrobial properties. J. Oleo Sci. 2016, 65, 629–640. [Google Scholar] [CrossRef] [PubMed]
- Al Maqtari, M.; Alghalibi, S.M.; Alhamzy, E.H. Chemical composition and antimicrobial activity of essential oil of Thymus vulgaris from Yemen. Turk. J. Biochem. 2011, 36, 342–349. [Google Scholar]
- Marino, M.; Bersani, C.; Comi, G. Antimicrobial activity of the essential oils of Thymus vulgaris L. measured using a bioimpedometric method. J. Food Prot. 1999, 62, 1017–1023. [Google Scholar] [CrossRef] [PubMed]
- Varga, E.; Bardocz, A.; Belak, A.; Maraz, A.; Boros, B.; Felinger, A.; Boszormenyi, A.; Horvath, G. Antimicrobial activity and chemical composition of thyme essential oils and the polyphenolic content of different thymus extracts. Farmacia 2015, 63, 357–361. [Google Scholar]
- Tiwari, B.K.; Valdramidis, V.P.; O’Donnell, C.P.; Muthukumarappan, K.; Bourke, P.; Cullen, P.J. Application of natural antimicrobials for food preservation. J. Agric. Food Chem. 2009, 57, 5987–6000. [Google Scholar] [CrossRef] [PubMed]
- Al-Turki, A.I. Antibacterial effect of thyme, peppermint, sage, black pepper and garlic hydrosols against Bacillus subtilis and Salmonella enteritidis. J. Food Agric. Environ. 2007, 5, 92–94. [Google Scholar]
- Hussien, J.; Teshale, C.; Mohammed, J. Assessment of the antimicrobial effects of some ethiopian aromatic spice and herb hydrosols. Int. J. Pharmacol. 2011, 7, 635–640. [Google Scholar] [CrossRef]
- Girova, T.; Gochev, V.; Jirovetz, L.; Buchbauer, G.; Schmidt, E.; Stoyanova, A. Antimicrobial activity of essential oils from spices against psychrotrophic food spoilage microorganisms. Biotechnol. Biotechnol. Equip. 2010, 24, 547–552. [Google Scholar] [CrossRef]
- Hajlaoui, H.; Snousi, M.; Noumi, E.; Zaneti, S.; Ksouri, R.; Bakhrouf, A. Chemical composition, antioxidant and antibacterial activities of the essential oils of five Tunisian aromatic plants. Ital. J. Food Sci. 2010, 22, 320–329. [Google Scholar]
- Viuda-Martos, M.; Mohamady, M.A.; Fernández-López, J.; Abd ElRazik, K.A.; Omer, E.A.; Perez-Alvarez, J.A.; Sendra, E. In vitro antioxidant and antibacterial activities of essentials oils obtained from Egyptian aromatic plants. Food Control 2011, 22, 1715–1722. [Google Scholar] [CrossRef]
- Aliakbarlu, J.; Shameli, F. In vitro antioxidant and antibacterial properties and total phenolic contents of essential oils from Thymus vulgaris, T. Kotschyanus, Ziziphora tenuior and Z. Clinopodioides. Turk. J. Biochem. 2013, 38, 425–431. [Google Scholar] [CrossRef]
- Bagamboula, C.F.; Uyttendaele, M.; Debevere, J. Antimicrobial effect of spices and herbs on Shigella sonnei and Shigella flexneri. J. Food Prot. 2003, 66, 668–673. [Google Scholar] [CrossRef] [PubMed]
- Martins, N.; Barros, L.; Santos-Buelga, C.; Silva, S.; Henriques, M.; Ferreira, I. Decoction, infusion and hydroalcoholic extract of cultivated thyme: Antioxidant and antibacterial activities, and phenolic characterisation. Food Chem. 2015, 167, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Ozturk, I. Antifungal activity of propolis, thyme essential oil and hydrosol on natural mycobiota of sucuk, a turkish fermented sausage: Monitoring of their effects on microbiological, color and aroma properties. J. Food Process Preserv. 2015, 39, 1148–1158. [Google Scholar] [CrossRef]
- Aman, S.; Naim, A.; Siddiqi, R.; Naz, S. Antimicrobial polyphenols from small tropical fruits, tea and spice oilseeds. Food Sci. Technol. Int. 2014, 20, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Aznar, A.; Fernandez, P.S.; Periago, P.M.; Palop, A. Antimicrobial activity of nisin, thymol, carvacrol and cymene against growth of Candida lusitaniae. Food Sci. Technol. Int. 2015, 21, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Ranasinghe, P.; Jayawardana, R.; Galappaththy, P.; Constantine, G.R.; de Vas Gunawardana, N.; Katulanda, P. Efficacy and safety of “true” cinnamon (Cinnamomum zeylanicum) as a pharmaceutical agent in diabetes: A systematic review and meta-analysis. Diabet. Med. 2012, 29, 1480–1492. [Google Scholar] [CrossRef] [PubMed]
- Khasnavis, S.; Pahan, K. Sodium benzoate, a metabolite of cinnamon and a food additive, upregulates neuroprotective Parkinson disease protein DJ-1 in astrocytes and neurons. J. Neuroimmune Pharm. 2012, 7, 424–435. [Google Scholar] [CrossRef] [PubMed]
- Brierley, S.M.; Kelber, O. Use of natural products in gastrointestinal therapies. Curr. Opin. Pharmacol. 2011, 11, 604–611. [Google Scholar] [CrossRef] [PubMed]
- Al-Jiffri, O.; El-Sayed, Z.M.F.; Al-Sharif, F.M. Urinary tract infection with Esherichia coli and antibacterial activity of some plants extracts. Int. J. Microbiol. Res. 2011, 2, 1–7. [Google Scholar]
- Gupta, C.; Garg, A.P.; Uniyal, R.C.; Kumari, A. Comparative analysis of the antimicrobial activity of cinnamon oil and cinnamon extract on some food-borne microbes. Afr. J. Microbiol. Res. 2008, 2, 247–251. [Google Scholar]
- Ceylan, E.; Fung, D.; Sabah, J.R. Antimicrobial activity and synergistic effect of cinnamon with sodium benzoate or potassium sorbate in controlling Escherichia coli O157: H7 in apple juice. J. Food Sci. 2004, 69, M102–M106. [Google Scholar] [CrossRef]
- Cui, H.Y.; Li, W.; Li, C.Z.; Vittayapadung, S.; Lin, L. Liposome containing cinnamon oil with antibacterial activity against methicillin-resistant Staphylococcus aureus biofilm. Biofouling 2016, 32, 215–225. [Google Scholar] [CrossRef] [PubMed]
- Shreaz, S.; Wani, W.A.; Behbehani, J.M.; Raja, V.; Irshad, M.; Karched, M.; Ali, I.; Siddiqi, W.A.; Hun, L.T. Cinnamaldehyde and its derivatives, a novel class of antifungal agents. Fitoterapia 2016, 112, 116–131. [Google Scholar] [CrossRef] [PubMed]
- Mvuemba, H.N.; Green, S.E.; Tsoomo, A.; Avis, T.J. Antimicrobial efficacy of cinnamon, ginger, horseradish and nutmeg extracts against spoilage pathogens. Phytoprotection 2009, 90, 65–70. [Google Scholar] [CrossRef]
- Wang, Q.; Ou, Z.B.; Lei, H.W.; Zeng, X.H.; Ying, Y.; Bai, W.D. Antimicrobial activities of a new formula of spice water extracts against foodborne bacteria. J. Food Process Preserv. 2012, 36, 374–381. [Google Scholar] [CrossRef]
- Agaoglu, S.; Dostbil, N.; Alemdar, S. Antimicrobial activity of some spices used in the meat industry. Bull. Vet. Inst. Pulawy 2007, 51, 53–57. [Google Scholar]
- Revati, S.; Bipin, C.; Chitra, P.; Minakshi, B. In vitro antibacterial activity of seven Indian spices against high level gentamicin resistant strains of enterococci. Arch. Med. Sci. 2015, 11, 863–868. [Google Scholar] [CrossRef] [PubMed]
- Nanasombat, S.; Wimuttigosol, P. Antimicrobial and antioxidant activity of spice essential oils. Food Sci. Biotechnol. 2011, 20, 45–53. [Google Scholar] [CrossRef]
- Trajano, V.N.; Lima, E.D.; de Souza, E.L.; Travassos, A. Antibacterial property of spice essential oils on food contaminating bacteria. Ciencia Tecnol. Aliment. 2009, 29, 542–545. [Google Scholar] [CrossRef]
- Reza, Z.M.; Atefeh, J.Y.; Faezeh, F. Effect of ã-irradiation on the antibacterial activities of Cuminum cyminum L. essential oils in vitro and in vivo systems. J. Essent. Oil Bear. Plants 2015, 18, 582–591. [Google Scholar] [CrossRef]
- Allahghadri, T.; Rasooli, I.; Owlia, P.; Nadooshan, M.J.; Ghazanfari, T.; Taghizadeh, M.; Astaneh, S. Antimicrobial property, antioxidant capacity, and cytotoxicity of essential oil from cumin produced in Iran. J. Food Sci. 2010, 75, H54–H61. [Google Scholar] [CrossRef] [PubMed]
- Kedia, A.; Prakash, B.; Mishra, P.K.; Dubey, N.K. Antifungal and antiaflatoxigenic properties of Cuminum cyminum (L.) seed essential oil and its efficacy as a preservative in stored commodities. Int. J. Food Microbiol. 2014, 168, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Abd El Mageed, M.A.; Mansour, A.F.; El Massry, K.F.; Ramadan, M.M.; Shaheen, M.S.; Shaaban, H. Effect of microwaves on essential oils of coriander and cumin seeds and on their antioxidant and antimicrobial activities. J. Essent. Oil Bear. Plants 2012, 15, 614–627. [Google Scholar] [CrossRef]
- Chaudhry, N.; Tariq, P. In vitro antibacterial activities of kalonji, cumin and poppy seed. Pak. J. Bot. 2008, 40, 461–467. [Google Scholar]
- Iacobellis, N.S.; Lo Cantore, P.; Capasso, F.; Senatore, F. Antibacterial activity of Cuminum cyminum L. and Carum carvi L. essential oils. J. Agric. Food Chem. 2005, 53, 57–61. [Google Scholar] [CrossRef] [PubMed]
- Ozcan, M.; Erkmen, O. Antimicrobial activity of the essential oils of Turkish plant spices. Eur. Food Res. Technol. 2001, 212, 658–660. [Google Scholar]
- Stefanini, M.B.; Figueiredo, R.O.; Ming, L.C.; Junior, A.F. Antimicrobial activity of the essential oils of some spice herbs. In Proceedings of the International Conference on Medicinal and Aromatic Plants Possibilities and Limitations of Medicinal and Aromatic Plant Production in the 21st Century, Budapest, Hungary, 8–11 July 2001; Szoke, E., Mathe, I., Blunden, G., Kery, A., Eds.; International Society for Horticultural Science: Leuven, Belgium, 2003; pp. 215–216. [Google Scholar]
- Toroglu, S. In vitro antimicrobial activity and synergistic/antagonistic effect of interactions between antibiotics and some spice essential oils. J. Environ. Biol. 2011, 32, 23–29. [Google Scholar] [PubMed]
- Bag, A.; Chattopadhyay, R.R. Evaluation of synergistic antibacterial and antioxidant efficacy of essential oils of spices and herbs in combination. PLoS ONE 2015, 10, e0131321. [Google Scholar] [CrossRef] [PubMed]
- Azizkhani, M.; Tooryan, F. Antioxidant and antimicrobial activities of rosemary extract, mint extract and a mixture of tocopherols in beef sausage during storage at 4C. J. Food Saf. 2015, 35, 128–136. [Google Scholar] [CrossRef]
- Tavassoli, S.K.; Mousavi, S.M.; Emam-Djomeh, Z.; Razavi, S.H. Chemical composition and evaluation of antimicrobial properties of Rosmarinus officinalis L. essential oil. Afr. J. Biotechnol. 2011, 10, 13895–13899. [Google Scholar]
- Bozin, B.; Mlmica-Dukic, N.; Samojlik, I.; Jovin, E. Antimicrobial and antioxidant properties of rosemary and sage (Rosmarinus officinalis L. and Salvia officinalis L., Lamiaceae) essential oils. J. Agric. Food Chem. 2007, 55, 7879–7885. [Google Scholar] [CrossRef] [PubMed]
- Weerakkody, N.S.; Caffin, N.; Turner, M.S.; Dykes, G.A. In vitro antimicrobial activity of less-utilized spice and herb extracts against selected food-borne bacteria. Food Control 2010, 21, 1408–1414. [Google Scholar] [CrossRef]
- Krajcova, E.; Greifova, M.; Schmidt, S. Study of antimicrobial activity of selected plant extracts against bacterial food contaminants. J. Food Nutr. Res. 2008, 47, 125–130. [Google Scholar]
- Zhang, H.Y.; Kong, B.H.; Xiong, Y.; Sun, X. Antimicrobial activities of spice extracts against pathogenic and spoilage bacteria in modified atmosphere packaged fresh pork and vacuum packaged ham slices stored at 4 °C. Meat Sci. 2009, 81, 686–692. [Google Scholar] [CrossRef] [PubMed]
- Kozlowska, M.; Laudy, A.E.; Przybyl, J.; Ziarno, M.; Majewska, E. Chemical composition and antibacterial activity of some medicinal plants from Lamiaceae family. ACTA Pol. Pharm. 2015, 72, 757–767. [Google Scholar] [PubMed]
- Weerakkody, N.S.; Caffin, N.; Dykes, G.A.; Turner, M.S. Effect of antimicrobial spice and herb extract combinations on Listeria monocytogenes, Staphylococcus aureus, and Spoilage Microflora growth on cooked ready-to-eat vacuum-packaged shrimp. J. Food Prot. 2011, 74, 1119–1125. [Google Scholar] [CrossRef] [PubMed]
- Nejad, A.; Shabani, S.; Bayat, M.; Hosseini, S.E. Antibacterial effect of garlic aqueous extract on Staphylococcus aureus in hamburger. Jundishapur J. Microbiol. 2014, 7, e13134. [Google Scholar]
- Rahman, M.S.; Al-Sheibani, H.I.; Al-Riziqi, M.H.; Mothershaw, A.; Guizani, N.; Bengtsson, G. Assessment of the anti-microbial activity of dried garlic powders produced by different methods of drying. Int. J. Food Prop. 2006, 9, 503–513. [Google Scholar] [CrossRef]
- Sallam, K.I.; Ishloroshi, M.; Samejima, K. Antioxidant and antimicrobial effects of garlic in chicken sausage. LWT-Food Sci. Technol. 2004, 37, 849–855. [Google Scholar] [CrossRef] [PubMed]
- Aydin, A.; Bostan, K.; Erkan, M.E.; Bingol, B. The antimicrobial effects of chopped garlic in ground beef and raw meatball (Çið Köfte). J. Med. Food 2007, 10, 203–207. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Shi, Q.; Dai, H.; Liang, Q.; Xie, X.; Huang, X.; Zhao, G.; Zhang, L. Antifungal activity, kinetics and molecular mechanism of action of garlic oil against Candida albicans. Sci. Rep. UK 2016, 6, 22805. [Google Scholar] [CrossRef] [PubMed]
- Indu, M.N.; Hatha, A.; Abirosh, C.; Harsha, U.; Vivekanandan, G. Antimicrobial activity of some of the south-Indian spices against serotypes of Escherichia coli, Salmonella, Listeria monocytogenes and Aeromonas hydrophila. Braz. J. Microbiol. 2006, 37, 153–158. [Google Scholar] [CrossRef]
- Joe, M.M.; Jayachitra, J.; Vijayapriya, M. Antimicrobial activity of some common spices against certain human pathogens. J. Med. Plants Res. 2009, 3, 1134–1136. [Google Scholar]
- Geremew, T.; Kebede, A.; Andualem, B. The role of spices and lactic acid bacteria as antimicrobial agent to extend the shelf life of metata ayib (traditional Ethiopian spiced fermented cottage cheese). J. Food Sci. Technol. Mysore 2015, 52, 5661–5670. [Google Scholar] [CrossRef] [PubMed]
- Touba, E.P.; Zakaria, M.; Tahereh, E. Anti-fungal activity of cold and hot water extracts of spices against fungal pathogens of roselle (Hibiscus sabdariffa) in vitro. Microb. Pathog. 2012, 52, 125–129. [Google Scholar] [CrossRef] [PubMed]
- Park, M.; Bae, J.; Lee, D.S. Antibacterial activity of [10]-gingerol and [12]-gingerol isolated from ginger rhizome against periodontal bacteria. Phytother. Res. 2008, 22, 1446–1449. [Google Scholar] [CrossRef] [PubMed]
- Sa-Nguanpuag, K.; Kanlayanarat, S.; Srilaong, V.; Tanprasert, K.; Techavuthiporn, C. Ginger (Zingiber officinale) oil as an antimicrobial agent for minimally processed produce: A case study in shredded green papaya. Int. J. Agric. Biol. 2011, 13, 895–901. [Google Scholar]
- Singh, G.; Maurya, S.; Catalan, C.; de Lampasona, M.P. Studies on essential oils, Part 42: Chemical, antifungal, antioxidant and sprout suppressant studies on ginger essential oil and its oleoresin. Flavour Frag. J. 2005, 20, 1–6. [Google Scholar] [CrossRef]
- Yoo, M.J.; Kim, Y.S.; Shin, D.H. Antibacterial effects of natural essential oils from ginger and mustard against Vibrio species inoculated on sliced raw flatfish. Food Sci. Biotechnol. 2006, 15, 462–465. [Google Scholar]
- Thongson, C.; Davidson, P.M.; Mahakarnchanakul, W.; Vibulsresth, P. Antimicrobial effect of Thai spices against Listeria monocytogenes and Salmonella typhimurium DT104. J. Food Prot. 2005, 68, 2054–2058. [Google Scholar] [CrossRef] [PubMed]
- Suppakul, P.; Miltz, J.; Sonneveld, K.; Bigger, S.W. Antimicrobial properties of basil and its possible application in food packaging. J. Agric. Food Chem. 2003, 51, 3197–3207. [Google Scholar] [CrossRef] [PubMed]
- Hussain, A.I.; Anwar, F.; Sherazi, S.; Przybylski, R. Chemical composition, antioxidant and antimicrobial activities of basil (Ocimum basilicum) essential oils depends on seasonal variations. Food Chem. 2008, 108, 986–995. [Google Scholar] [CrossRef] [PubMed]
- Kaya, I.; Yigit, N.; Benli, M. Antimicrobial activity of various extracts of Ocimum basilicum L. and observation of the inhibition effect on bacterial cells by use of scanning electron microscopy. Afr. J. Tradit. Complement. Altern. Med. 2008, 5, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Kocic-Tanackov, S.; Dimic, G.; Levic, J.; Tanackov, I.; Tuco, D. Antifungal activities of basil (Ocimum basilicum L.) extract on Fusarium species. Afr. J. Biotechnol. 2011, 10, 10188–10195. [Google Scholar]
- Beatovic, D.; Krstic-Milosevic, D.; Trifunovic, S.; Siljegovic, J.; Glamoclija, J.; Ristic, M.; Jelacic, S. Chemical composition, antioxidant and antimicrobial activities of the essential oils of twelve Ocimum basilicum L. cultivars grown in Serbia. Rec. Nat. Prod. 2015, 9, 62–75. [Google Scholar]
- El-Habib, R. Antifungal activity of some essential oils on Aspergillus flavus growth and aflatoxin production. J. Food Agric. Environ. 2012, 10, 274–279. [Google Scholar]
- Singh, G.; Maurya, S.; de Lampasona, M.P.; Catalan, C. Chemical constituents, antifungal and antioxidative potential of Foeniculum vulgare volatile oil and its acetone extract. Food Control 2006, 17, 745–752. [Google Scholar] [CrossRef]
- Diao, W.R.; Hu, Q.P.; Zhang, H.; Xu, J.G. Chemical composition, antibacterial activity and mechanism of action of essential oil from seeds of fennel (Foeniculum vulgare Mill.). Food Control 2014, 35, 109–116. [Google Scholar] [CrossRef]
- Park, J.S.; Baek, H.H.; Bai, D.H.; Oh, T.K.; Lee, C.H. Antibacterial activity of fennel (Foeniculum vulgare Mill.) seed essential oil against the growth of Streptococcus mutans. Food Sci. Biotechnol. 2004, 13, 581–585. [Google Scholar]
- Zellagui, A.; Gherraf, N.; Elkhateeb, A.; Hegazy, M.; Mohamed, T.A.; Touil, A.; Shahat, A.A.; Rhouati, S. Chemical constituents from Algerian Foeniculum Vulgare aerial parts and evaluation of antimicrobial activity. J. Chil. Chem. Soc. 2011, 56, 759–763. [Google Scholar] [CrossRef]
- Bisht, D.S.; Menon, K.; Singhal, M.K. Comparative antimicrobial activity of essential oils of Cuminum cyminum L. and Foeniculum vulgare Mill seeds against Salmonella typhimurium and Escherichia coli. J. Essent. Oil Bear. Plants 2014, 17, 617–622. [Google Scholar] [CrossRef]
- Nguyen, S.; Huang, H.; Foster, B.C.; Tam, T.W.; Xing, T.; Smith, M.L.; Arnason, J.T.; Akhtar, H. Antimicrobial and P450 inhibitory properties of common functional foods. J. Pharm. Pharm. Sci. 2014, 17, 254–265. [Google Scholar] [CrossRef]
- Duarte, A.; Luis, A.; Oleastro, M.; Domingues, F.C. Antioxidant properties of coriander essential oil and linalool and their potential to control Campylobacter spp. Food Control 2016, 61, 115–122. [Google Scholar] [CrossRef]
- Duarte, A.F.; Ferreira, S.; Oliveira, R.; Domingues, F.C. Effect of coriander oil (Coriandrum sativum) on planktonic and biofilm cells of Acinetobacter baumannii. Nat. Prod. Commun. 2013, 8, 673–678. [Google Scholar]
- Duarte, A.; Ferreira, S.; Silva, F.; Domingues, F.C. Synergistic activity of coriander oil and conventional antibiotics against Acinetobacter baumannii. Phytomedicine 2012, 19, 236–238. [Google Scholar] [CrossRef] [PubMed]
- Freires, I.D.; Murata, R.M.; Furletti, V.F.; Sartoratto, A.; de Alencar, S.M.; Figueira, G.M.; Rodrigues, J.; Duarte, M.; Rosalen, P.L. Coriandrum sativum L. (coriander) essential oil: Antifungal activity and mode of action on Candida spp., and molecular targets affected in human whole-genome expression. PLoS ONE 2014, 9, e99086. [Google Scholar]
- Silva, F.; Ferreira, S.; Queiroz, J.A.; Domingues, F.C. Coriander (Coriandrum sativum L.) essential oil: Its antibacterial activity and mode of action evaluated by flow cytometry. J. Med. Microbiol. 2011, 60, 1479–1486. [Google Scholar] [CrossRef] [PubMed]
- Acimovic, M.G.; Grahovac, M.S.; Stankovic, J.M.; Cvetkovic, M.T.; Masirevic, S.N. Essential oil composition of different coriander (Coriandrum sativum L.) accessions and their influence on mycelial growth of Colletotrichum spp. Acta Sci. Pol. Hortorum Cultus 2016, 15, 35–44. [Google Scholar]
- Singh, G.; Maurya, S.; de Lampasona, M.P.; Catalan, C. Studies on essential oils, Part 41. Chemical composition, antifungal, antioxidant and sprout suppressant activities of coriander (Coriandrum sativum) essential oil and its oleoresin. Flavour Frag. J. 2006, 21, 472–479. [Google Scholar] [CrossRef]
- Yakout, S.M.; Abd-Alrahman, S.H.; Mostafa, A.; Salem-Bekhit, M.M. Antimicrobial effect of seed ethanolic extract of coriander. J. Pure Appl. Microbiol. 2013, 7, 459–463. [Google Scholar]
- Dimic, G.; Kocic-Tanackov, S.; Mojovic, L.; Pejin, J. Antifungal activity of lemon essential oil, coriander and cinnamon extracts on foodborne molds in direct contact and the vapor phase. J. Food Process. Pres. 2015, 39, 1778–1787. [Google Scholar] [CrossRef]
- Rao, K.; Ch, B.; Narasu, L.M.; Giri, A. Antibacterial activity of Alpinia galanga (L.) willd crude extracts. Appl. Biochem. Biotechnol. 2010, 162, 871–884. [Google Scholar] [CrossRef] [PubMed]
- Weerakkody, N.S.; Caffin, N.; Lambert, L.K.; Turner, M.S.; Dykes, G.A. Synergistic antimicrobial activity of galangal (Alpinia galanga), rosemary (Rosmarinus officinalis) and lemon iron bark (Eucalyptus staigerana) extracts. J. Sci. Food Agric. 2011, 91, 461–468. [Google Scholar] [CrossRef] [PubMed]
- Wong, L.F.; Lim, Y.Y.; Omar, M. Antioxidant and antimicrobial activities of some Alpina species. J. Food Biochem. 2009, 33, 835–851. [Google Scholar] [CrossRef]
- Singh, G.; Marimuthu, P.; Catalan, C.; DeLampasona, M.P. Chemical, antioxidant and antifungal activities of volatile oil of black pepper and its acetone extract. J. Sci. Food Agric. 2004, 84, 1878–1884. [Google Scholar] [CrossRef]
- Singh, G.; Marimuthu, P.; Murali, H.S.; Bawa, A.S. Antioxidative and antibacterial potentials of essential oils and extracts isolated from various spice materials—Part 48. J. Food Saf. 2005, 25, 130–145. [Google Scholar] [CrossRef]
- Zarai, Z.; Boujelbene, E.; Ben Salem, N.; Gargouri, Y.; Sayari, A. Antioxidant and antimicrobial activities of various solvent extracts, piperine and piperic acid from Piper nigrum. LWT-Food Sci. Technol. 2013, 50, 634–641. [Google Scholar] [CrossRef]
- Zhang, J.; Ye, K.; Zhang, X.; Pan, D.; Sun, Y.; Cao, J. Antibacterial activity and mechanism of action of black pepper essential oil on meat-borne Escherichia coli. Front. Microbiol. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Baris, D.; Kizil, M.; Aytekin, C.; Kizil, G.; Yavuz, M.; Ceken, B.; Ertekin, A.S. In vitro antimicrobial and antioxidant activity of ethanol extract of three Hypericum and three Achillea species from Turkey. Int. J. Food Prop. 2011, 14, 339–355. [Google Scholar] [CrossRef]
- Tajik, H.; Jalali, F.; Sobhani, A.; Shahbazi, Y.; Zadeh, M.S. In vitro assessment of antimicrobial efficacy of alcoholic extract of Achillea millefolium in comparison with penicillin derivatives. J. Anim. Vet. Adv. 2008, 7, 508–511. [Google Scholar]
- Eyob, S.; Martinsen, B.K.; Tsegaye, A.; Appelgren, M.; Skrede, G. Antioxidant and antimicrobial activities of extract and essential oil of korarima (Aframomum corrorima (Braun) P.C.M. Jansen). Afr. J. Biotechnol. 2008, 7, 2585–2592. [Google Scholar]
- Ismail, S.; Jalilian, F.A.; Talebpour, A.H.; Zargar, M.; Shameli, K.; Sekawi, Z.; Jahanshiri, F. Chemical composition and antibacterial and cytotoxic activities of Allium hirtifolium Boiss. BioMed Res. Int. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Najjaa, H.; Ammar, E.; Neffati, M. Antimicrobial activities of protenic extracts of Allium roseum L., a wild edible species in North Africa. J. Food Agric. Environ. 2009, 7, 150–154. [Google Scholar]
- Mihaylova, D.S.; Lante, A.; Tinello, F.; Krastanov, A.I. Study on the antioxidant and antimicrobial activities of Allium ursinum L. pressurised-liquid extract. Nat. Prod. Res. 2014, 28, 2000–2005. [Google Scholar] [CrossRef] [PubMed]
- Diao, W.R.; Zhang, L.L.; Feng, S.S.; Xu, J.G. Chemical composition, antibacterial activity, and mechanism of action of the essential oil from Amomum kravanh. J. Food Prot. 2014, 77, 1740–1746. [Google Scholar] [CrossRef]
- Singh, G.; Maurya, S.; de Lampasona, M.P.; Catalan, C. Chemical constituents, antimicrobial investigations, and antioxidative potentials of Anethum graveolens L. essential oil and acetone extract: Part 52. J. Food Sci. 2005, 70, M208–M215. [Google Scholar] [CrossRef]
- Akkoyun, H.T.; Dostbil, N. Antibacterial activity of some species of Umbelliferae. Asian J. Chem. 2007, 19, 4862–4866. [Google Scholar]
- Sofia, P.K.; Prasad, R.; Vijay, V.K.; Srivastava, A.K. Evaluation of antibacterial activity of Indian spices against common foodborne pathogens. Int. J. Food Sci. Technol. 2007, 42, 910–915. [Google Scholar] [CrossRef]
- Sekine, T.; Sugano, M.; Majid, A.; Fujii, Y. Antifungal effects of volatile compounds from black zira (Bunium persicum) and other spices and herbs. J. Chem. Ecol. 2007, 33, 2123–2132. [Google Scholar] [CrossRef] [PubMed]
- Mishra, P.K.; Shukla, R.; Singh, P.; Prakash, B.; Dubey, N.K. Antifungal and antiaflatoxigenic efficacy of Caesulia axillaris Roxb essential oil against fungi deteriorating some herbal raw materials, and its antioxidant activity. Ind. Crop. Prod. 2012, 36, 74–80. [Google Scholar] [CrossRef]
- Wahba, N.M.; Ahmed, A.S.; Ebraheim, Z.Z. Antimicrobial effects of pepper, parsley, and dill and their roles in the microbiological quality enhancement of traditional Egyptian kareish cheese. Foodborne Pathog. Dis. 2010, 7, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Gurnani, N.; Gupta, M.; Shrivastava, R.; Mehta, D.; Mehta, B.K. Effect of extraction methods on yield, phytochemical constituents, antibacterial and antifungal activity of Capsicum frutescens L. Indian J. Nat. Prod. Resour. 2016, 7, 32–39. [Google Scholar]
- Singh, G.; Kapoor, I.; Pandey, S.K.; Singh, U.K.; Singh, R.K. Studies on essential oils: Part 10; Antibacterial activity of volatile oils of some spices. Phytother. Res. 2002, 16, 680–682. [Google Scholar] [CrossRef] [PubMed]
- Oroojalian, F.; Kasra-Kermanshahi, R.; Azizi, M.; Bassami, M.R. Phytochemical composition of the essential oils from three Apiaceae species and their antibacterial effects on food-borne pathogens. Food Chem. 2010, 120, 765–770. [Google Scholar] [CrossRef]
- Shan, B.; Cai, Y.Z.; Brooks, J.D.; Corke, H. Antibacterial properties and major bioactive components of cinnamon stick (Cinnamomum burmannii): Activity against foodborne pathogenic bacteria. J. Agric. Food Chem. 2007, 55, 5484–5490. [Google Scholar] [CrossRef] [PubMed]
- Pandey, A.K.; Mishra, A.K.; Mishra, A. Antifungal and antioxidative potential of oil and extracts derived from leaves of Indian spice plant Cinnamomum tamala. Cell. Mol. Biol. 2012, 58, 142–147. [Google Scholar] [PubMed]
- Shreya, A.; Manisha, D.; Sonali, J. Phytochemical screening and anti-microbial activity of cinnamon spice against urinary tract infection and fungal pathogens. Int. J. Life Sci. Pharma Res. 2015, 5, P30–P38. [Google Scholar]
- Naveed, R.; Hussain, I.; Mahmood, M.S.; Akhtar, M. In vitro and in vivo evaluation of antimicrobial activities of essential oils extracted from some indigenous spices. Pak. Vet. J. 2013, 33, 413–417. [Google Scholar]
- Naveed, R.; Hussain, I.; Tawab, A.; Tariq, M.; Rahman, M.; Hameed, S.; Mahmood, M.S.; Siddique, A.; Iqbal, M. Antimicrobial activity of the bioactive components of essential oils from Pakistani spices against Salmonella and other multi-drug resistant bacteria. BMC Complement. Altern. Med. 2013, 13, 265. [Google Scholar] [CrossRef] [PubMed]
- Hashemi, S.; Amininezhad, R.; Shirzadinezhad, E.; Farahani, M.; Yousefabad, S. The antimicrobial and antioxidant effects of Citrus aurantium L. flowers (Bahar narang) extract in traditional yoghurt stew during refrigerated storage. J. Food Saf. 2016, 36, 153–161. [Google Scholar] [CrossRef]
- Castilho, P.; Liu, K.; Rodrigues, A.I.; Feio, S.; Tomi, F.; Casanova, J. Composition and antimicrobial activity of the essential oil of Clinopodium ascendens (Jordan) Sampaio from Madeira. Flavour Frag. J. 2007, 22, 139–144. [Google Scholar] [CrossRef]
- Oussalah, M.; Caillet, S.; Saucier, L.; Lacroix, M. Antimicrobial effects of selected plant essential oils on the growth of a Pseudomonas putida strain isolated from meat. Meat Sci. 2005, 73, 236–244. [Google Scholar] [CrossRef] [PubMed]
- Sani, A.M.; Yaghooti, F. Antibacterial effects and chemical composition of essential oil from Cotoneaster nummularioides leaves extract on typical food-borne pathogens. J. Essent. Oil Bear. Plants 2016, 19, 290–296. [Google Scholar] [CrossRef]
- Daouda, T.; Prevost, K.; Gustave, B.; Joseph, D.A.; Nathalie, G.; Raphael, O.; Rubens, D.; Claude, C.J.; Mireille, D.; Felix, T. Terpenes, antibacterial and modulatory antibiotic activity of essential oils from Croton hirtus L’ Hér. (Euphorbiaceae) from Ivory Coast. J. Essent. Oil Bear. Plants 2014, 17, 607–616. [Google Scholar] [CrossRef]
- Ani, V.; Varadaraj, M.C.; Naidu, K.A. Antioxidant and antibacterial activities of polyphenolic compounds from bitter cumin (Cuminum nigrum L.). Eur. Food Res. Technol. 2006, 224, 109–115. [Google Scholar] [CrossRef]
- Li, X.F.; Jin, C.; He, J.; Zhou, J.; Wang, H.T.; Dai, B.; Yan, D.; Wang, J.B.; Zhao, Y.L.; Xiao, X.H. Microcalorimetric investigation of the antibacterial activity of curcumin on Staphylococcus aureus coupled with multivariate analysis. J. Therm. Anal. Calorim. 2012, 109, 395–402. [Google Scholar] [CrossRef]
- Sandri, I.G.; Zacaria, J.; Fracaro, F.; Delamare, A.; Echeverrigaray, S. Antimicrobial activity of the essential oils of Brazilian species of the genus Cunila against foodborne pathogens and spoiling bacteria. Food Chem. 2007, 103, 823–828. [Google Scholar] [CrossRef]
- Fankam, A.G.; Kuete, V.; Voukeng, I.K.; Kuiate, J.R.; Pages, J.M. Antibacterial activities of selected Cameroonian spices and their synergistic effects with antibiotics against multidrug-resistant phenotypes. BMC Complement. Altern. Med. 2011, 11, 104. [Google Scholar] [CrossRef] [PubMed]
- Tekwu, E.M.; Askun, T.; Kuete, V.; Nkengfack, A.E.; Nyasse, B.; Etoa, F.X.; Beng, V.P. Antibacterial activity of selected Cameroonian dietary spices ethno-medically used against strains of Mycobacterium tuberculosis. J. Ethnopharmacol. 2012, 142, 374–382. [Google Scholar] [CrossRef] [PubMed]
- El Malti, J.; Mountassif, D.; Amarouch, H. Antimicrobial activity of Elettaria cardamomum: Toxicity, biochemical and histological studies. Food Chem. 2007, 104, 1560–1568. [Google Scholar] [CrossRef]
- Singh, G.; Kim, S.; Marimuthu, P.; Isidorov, V.; Vinogorova, V. Antioxidant and antimicrobial activities of essential oil and various oleoresins of Elettaria cardamomum (seeds and pods). J. Sci. Food Agric. 2008, 88, 280–289. [Google Scholar] [CrossRef]
- Bey-Ould Si Said, Z.; Haddadi-Guemghar, H.; Boulekbache-Makhlouf, L.; Rigou, P.; Remini, H.; Adjaoud, A.; Khoudja, N.K.; Madani, K. Essential oils composition, antibacterial and antioxidant activities of hydrodistillated extract of Eucalyptus globulus fruits. Ind. Crop. Prod. 2016, 89, 167–175. [Google Scholar] [CrossRef]
- Nikbakht, M.R.; Rahimi-Nasrabadi, M.; Ahmadi, F.; Gandomi, H.; Abbaszadeh, S.; Batooli, H. The chemical composition and in vitro antifungal activities of essential oils of five Eucalyptus species. J. Essent. Oil Bear. Plants 2015, 18, 666–677. [Google Scholar] [CrossRef]
- Luís, Â.; Duarte, A.; Gominho, J.; Domingues, F.; Duarte, A.P. Chemical composition, antioxidant, antibacterial and anti-quorum sensing activities of Eucalyptus globulus and Eucalyptus radiata essential oils. Ind. Crop. Prod. 2016, 79, 274–282. [Google Scholar] [CrossRef]
- Puangpronpitag, D.; Niamsa, N.; Sittiwet, C. Anti-microbial properties of clove (Eugenia caryophyllum Bullock and Harrison) aqueous extract against food-borne pathogen bacteria. Int. J. Pharmacol. 2009, 5, 281–284. [Google Scholar]
- Ozcan, M.M.; Chalchat, J.C.; Arslan, D.; Ates, A.; Unver, A. Comparative essential oil composition and antifungal effect of bitter fennel (Foeniculum vulgare ssp. piperitum) fruit oils obtained during different vegetation. J. Med. Food 2006, 9, 552–561. [Google Scholar] [PubMed]
- Soureshjan, E.H.; Heidari, M. In vitro variation in antibacterial activity plant extracts on Glaucium elegans and saffron (Crocus sativus). Bangladesh J. Pharmacol. 2014, 9, 275–278. [Google Scholar]
- Khan, H.; Khan, M.A.; Mahmood, T.; Choudhary, M.I. Antimicrobial activities of Gloriosa superba Linn (Colchicaceae) extracts. J. Enzym. Inhib. Med. Chem. 2008, 23, 855–859. [Google Scholar] [CrossRef] [PubMed]
- Albayrak, S.; Aksoy, A.; Sagdic, O.; Hamzaoglu, E. Compositions, antioxidant and antimicrobial activities of Helichrysum (Asteraceae) species collected from Turkey. Food Chem. 2010, 119, 114–122. [Google Scholar] [CrossRef]
- Albayrak, S.; Aksoy, A.; Sagdic, O.; Budak, U. Phenolic compounds and antioxidant and antimicrobial properties of Helichrysum species collected from eastern Anatolia, Turkey. Turk. J. Biol. 2010, 34, 463–473. [Google Scholar]
- Czapska, A.; Balasinska, B.; Szczawinski, J. Antimicrobial and antioxidant properties of aqueous extracts from selected spice plants. Med. Weter. 2006, 62, 302–305. [Google Scholar]
- Džamiæ, A.M.; Sokoviæ, M.D.; Novakoviæ, M.; Jadranin, M.; Ristiæ, M.S.; Teševiæ, V.; Marin, P.D. Composition, antifungal and antioxidant properties of Hyssopus officinalis L. subsp pilifer (Pant.) Murb essential oil and deodorized extracts. Ind. Crop. Prod. 2013, 51, 401–407. [Google Scholar] [CrossRef]
- Parlatan, A.; Saricoban, C.; Ozcan, M.M. Chemical composition and antimicrobial activity of the extracts of Kefe cumin (Laser trilobum L.) fruits from different regions. Int. J. Food Sci. Nutr. 2009, 60, 606–617. [Google Scholar] [CrossRef] [PubMed]
- El Malti, J.; Amarouch, H. Antibacterial effect, histological impact and oxidative stress studies Fromlaurus nobilis extract. J. Food Qual. 2009, 32, 190–208. [Google Scholar] [CrossRef]
- Ramos, C.; Teixeira, B.; Batista, I.; Matos, O.; Serrano, C.; Neng, N.R.; Nogueira, J.; Nunes, M.L.; Marques, A. Antioxidant and antibacterial activity of essential oil and extracts of bay laurel Laurus nobilis Linnaeus (Lauraceae) from Portugal. Nat. Prod. Res. 2012, 26, 518–529. [Google Scholar] [CrossRef] [PubMed]
- Ceyhan, N.; Keskin, D.; Ugur, A. Antimicrobial activities of different extracts of eight plant species from four different family against some pathogenic microoorganisms. J. Food Agric. Environ. 2012, 10, 193–197. [Google Scholar]
- Marín, I.; Sayas-Barberá, E.; Viuda-Martos, M.; Navarro, C.; Sendra, E. Chemical composition, antioxidant and antimicrobial activity of essential oils from organic fennel, parsley, and lavender from Spain. Foods 2016, 5, 18. [Google Scholar] [CrossRef]
- Basile, A.; Rigano, D.; Loppi, S.; Di Santi, A.; Nebbioso, A.; Sorbo, S.; Conte, B.; Paoli, L.; de Ruberto, F.; Molinari, A.M. Antiproliferative, antibacterial and antifungal activity of the lichen Xanthoria parietina and its secondary metabolite parietin. Int. J. Mol. Sci. 2015, 16, 7861–7875. [Google Scholar] [CrossRef] [PubMed]
- Sarrazin, S.; Oliveira, R.B.; Barata, L.; Mourao, R. Chemical composition and antimicrobial activity of the essential oil of Lippia grandis Schauer (Verbenaceae) from the western Amazon. Food Chem. 2012, 134, 1474–1478. [Google Scholar] [CrossRef] [PubMed]
- Asowata-Ayodele, A.M.; Otunola, G.A.; Afolayan, A.J. Assessment of the polyphenolic content, free radical scavenging, anti-inflammatory, and antimicrobial activities of acetone and aqueous extracts of Lippia javanica (Burm.F.) Spreng. Pharmacogn. Mag. 2016, 12, S353–S362. [Google Scholar] [PubMed]
- Oliveira, D.R.; Leitao, G.G.; Bizzo, H.R.; Lopes, D.; Alviano, D.S.; Alviano, C.S.; Leitao, S.G. Chemical and antimicrobial analyses of essential oil of Lippia origanoides HBK. Food Chem. 2007, 101, 236–240. [Google Scholar] [CrossRef]
- Li, W.R.; Shi, Q.S.; Liang, Q.; Xie, X.B.; Huang, X.M.; Chen, Y.B. Antibacterial activity and kinetics of Litsea cubeba oil on Escherichia coli. PLoS ONE 2014, 9, e110983. [Google Scholar] [CrossRef] [PubMed]
- Stanojevic, D.; Comic, L.; Stefanovic, O.; Sukdolak, S.S. In vitro synergistic antibacterial activity of Melissa officinalis L. and some preservatives. Span. J. Agric. Res. 2010, 8, 109–115. [Google Scholar] [CrossRef]
- Sharma, M.; Sharma, M. Antimicrobial potential of essential oil from Mentha piperita L. against anthropophilic dermatophytes. J. Essent. Oil Bear. Plants 2012, 15, 263–269. [Google Scholar] [CrossRef]
- Arumugam, P.; Murugan, R.; Subathra, M.; Ramesh, A. Superoxide radical scavenging and antibacterial activities of different fractions of ethanol extract of Mentha spicata (L.). Med. Chem. Res. 2010, 19, 664–673. [Google Scholar] [CrossRef]
- Esekhiagbe, M.; Agatemor, M.; Agatemor, C. Phenolic content and antimicrobial potentials of Xylopia aethiopica and Myristica argentea. Maced. J. Chem. Chem. Eng. 2009, 28, 159–162. [Google Scholar]
- Shafiei, Z.; Shuhairi, N.N.; Yap, N.; Sibungkil, C.; Latip, J. Antibacterial activity of Myristica fragrans against oral pathogens. Evid. Based Complement. Altern. 2012, 2012. [Google Scholar] [CrossRef]
- Mhamdi, B.; Abbassi, F.; Marzouki, L. Antimicrobial Activities effects of the essential oil of spice food Myrtus communis leaves vr. Italica. J. Essent. Oil Bear. Plants 2014, 17, 1361–1366. [Google Scholar] [CrossRef]
- Ozcan, M.M.; Uyar, B.; Unver, A. Antibacterial effect of myrtle (Myrtus communis L.) leaves extract on microorganisms. J. Food Saf. Food Qual. 2015, 66, 18–21. [Google Scholar]
- Popovici, J.; Bertrand, C.; Bagnarol, E.; Fernandez, M.P.; Comte, G. Chemical composition of essential oil and headspace-solid microextracts from fruits of Myrica gale L. and antifungal activity. Nat. Prod. Res. 2008, 22, 1024–1032. [Google Scholar] [CrossRef] [PubMed]
- Aboee-Mehrizi, F.; Rustaiyan, A.; Zare, M. Chemical composition and antimicrobial activity of the essential oils of Nepeta alpina growing wild in Iran. J. Essent. Oil Bear. Plants 2016, 19, 236–240. [Google Scholar] [CrossRef]
- Arslan, U.; Ilhan, K.; Karabulut, O.A. Antifungal activity of aqueous extracts of spices against bean rust (Uromyces appendiculatus). Allelopath. J. 2009, 24, 207–213. [Google Scholar]
- Arici, M.; Sagdic, O.; Gecgel, U. Antibacterial effect of Turkish black cumin (Nigella sativa L.) oils. Grasas Aceites 2005, 56, 259–262. [Google Scholar] [CrossRef]
- Wouatsa, N.; Misra, L.; Kumar, R.V. Antibacterial activity of essential oils of edible spices, Ocimum canum and Xylopia aethiopica. J. Food Sci. 2014, 79, M972–M977. [Google Scholar] [CrossRef] [PubMed]
- Prakash, B.; Shukla, R.; Singh, P.; Mishra, P.K.; Dubey, N.K.; Kharwar, R.N. Efficacy of chemically characterized Ocimum gratissimum L. essential oil as an antioxidant and a safe plant based antimicrobial against fungal and aflatoxin B1 contamination of spices. Food Res. Int. 2011, 44, 385–390. [Google Scholar] [CrossRef]
- Kumar, A.; Dubey, N.K.; Srivastaya, S. Antifungal evaluation of Ocimum sanctum essential oil against fungal deterioration of raw materials of Rauvolfia serpentina during storage. Ind. Crop. Prod. 2013, 45, 30–35. [Google Scholar] [CrossRef]
- Saharkhiz, M.J.; Kamyab, A.A.; Kazerani, N.K.; Zomorodian, K.; Pakshir, K.; Rahimi, M.J. Chemical compositions and antimicrobial activities of Ocimum sanctum L. essential oils at different harvest stages. Jundishapur J. Microbiol. 2015, 8, e13720. [Google Scholar] [CrossRef] [PubMed]
- Runyoro, D.; Ngassapa, O.; Vagionas, K.; Aligiannis, N.; Graikou, K.; Chinou, I. Chemical composition and antimicrobial activity of the essential oils of four Ocimum species growing in Tanzania. Food Chem. 2010, 119, 311–316. [Google Scholar] [CrossRef]
- Ali, N.H.; Faizi, S.; Kazmi, S.U. Antibacterial activity in spices and local medicinal plants against clinical isolates of Karachi, Pakistan. Pharm. Biol. 2011, 49, 833–839. [Google Scholar] [CrossRef] [PubMed]
- Yano, Y.; Satomi, M.; Oikawa, H. Antimicrobial effect of spices and herbs on Vibrio parahaemolyticus. Int. J. Food Microbiol. 2006, 111, 6–11. [Google Scholar] [CrossRef] [PubMed]
- Seydim, A.C.; Sarikus, G. Antimicrobial activity of whey protein based edible films incorporated with oregano, rosemary and garlic essential oils. Food Res. Int. 2006, 39, 639–644. [Google Scholar] [CrossRef]
- Ho, C.H.; Noryati, I.; Sulaiman, S.F.; Rosma, A. In vitro antibacterial and antioxidant activities of Orthosiphon stamineus Benth extracts against food-borne bacteria. Food Chem. 2010, 122, 1168–1172. [Google Scholar] [CrossRef]
- Boyd, F.; Benkeblia, N. In vitro evaluation of antimicrobial activity of crude extracts of Pimenta dioica L. (Merr.). In Proceedings of the 3rd International Conference on Postharvest and Quality Management of Horticultural Products of Interest for Tropical Regions, Port of Spain, Trinid & Tobago, 1–5 July 2013; Mohammed, M., Francis, J.A., Eds.; International Society for Horticultural Science: Leuven, Belgium, 2014; pp. 199–205. [Google Scholar]
- Ozcan, M.M.; Chalchat, J.C. Chemical composition and antifungal effect of anise (Pimpinella anisum L.) fruit oil at ripening stage. Ann. Microbiol. 2006, 56, 353–358. [Google Scholar] [CrossRef]
- Woguem, V.; Maggi, F.; Fogang, H.; Tapondjou, L.A.; Womeni, H.M.; Quassinti, L.; Bramucci, M.; Vitali, L.A.; Petrelli, D.; Lupidi, G.; et al. Antioxidant, antiproliferative and antimicrobial activities of the volatile oil from the wild pepper piper capense used in Cameroon as a culinary spice. Nat. Prod. Commun. 2013, 8, 1791–1796. [Google Scholar] [PubMed]
- Eruteya, O.C.; Odunfa, S.A. Antimicrobial properties of three spices used in the preparation of suya condiment against organisms isolated from formulated samples and individual ingredients. Afr. J. Biotechnol. 2009, 8, 2316–2320. [Google Scholar]
- Toroglu, S.; Cenet, M. Comparison of antimicrobial activities of essential oil and solvent extracts of endemic Phlomis oppositiflora Boiss. & Hausskn. from Turkey. Pak. J. Zool. 2013, 45, 475–482. [Google Scholar]
- Sahin, S.; Oran, S.; Sahinturk, P.; Demir, C.; Ozturk, S. Ramalina lichens and their major metabolites as possible natural antioxidant and antimicrobial agents. J. Food Biochem. 2015, 39, 471–477. [Google Scholar] [CrossRef]
- Fazeli, M.R.; Amin, G.; Attari, M.; Ashtiani, H.; Jamalifar, H.; Samadi, N. Antimicrobial activities of Iranian sumac and avishan-e shirazi. (Zataria multiflora) against some food-borne bacteria. Food Control 2007, 18, 646–649. [Google Scholar] [CrossRef]
- Aliakbarlu, J.; Mohammadi, S.; Khalili, S. A study on antioxidant potency and antibacterial activity of water extracts of some spices widely consumed in Iranian diet. J. Food Biochem. 2014, 38, 159–166. [Google Scholar] [CrossRef]
- Mekinic, I.G.; Skroza, D.; Ljubenkov, I.; Simat, V.; Mozina, S.S.; Katalinic, V. In vitro antioxidant and antibacterial activity of Lamiaceae phenolic extracts: A correlation study. Food Technol. Biotechnol. 2014, 52, 119–127. [Google Scholar]
- Bouajaj, S.; Benyamna, A.; Bouamama, H.; Romane, A.; Falconieri, D.; Piras, A.; Marongiu, B. Antibacterial, allelopathic and antioxidant activities of essential oil of Salvia officinalis L. growing wild in the Atlas Mountains of Morocco. Nat. Prod. Res. 2013, 27, 1673–1676. [Google Scholar] [CrossRef] [PubMed]
- Mehr, H.M.; Hosseini, Z.; Khodaparast, M.; Edalatian, M.R. Study on the antimicrobial effect of Salvia leriifolia (nowroozak) leaf extract powder on the growth of Staphylococcus aureus in hamburger. J. Food Saf. 2010, 30, 941–953. [Google Scholar] [CrossRef]
- Djeddi, S.; Djebile, K.; Hadjbourega, G.; Achour, Z.; Argyropoulou, C.; Skaltsa, H. In vitro Antimicrobial properties and chemical composition of Santolina chamaecyparissus essential oil from Algeria. Nat. Prod. Commun. 2012, 7, 937–940. [Google Scholar] [PubMed]
- Oke, F.; Aslim, B.; Ozturk, S.; Altundag, S. Essential oil composition, antimicrobial and antioxidant activities of Satureja cuneifolia Ten. Food Chem. 2009, 112, 874–879. [Google Scholar] [CrossRef]
- Mihajilov-Krstev, T.; Kitic, D.; Radnovic, D.; Ristic, M.; Mihajlovic-Ukropina, M.; Zlatkovic, B. Chemical composition and antimicrobial activity of Satureja kitaibelii essential oil against pathogenic microbial strains. Nat. Prod. Commun. 2011, 6, 1167–1172. [Google Scholar] [PubMed]
- Yucel, N.; Aslim, B. Antibacterial activity of the essential oil of Satureja wiedemanniana against Bacillus species isolated from chicken meat. Foodborne Pathog. Dis. 2011, 8, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Farzaneh, M.; Kiani, H.; Sharifi, R.; Reisi, M.; Hadian, J. Chemical composition and antifungal effects of three species of Satureja (S. hortensis, S. spicigera and S. khuzistanica) essential oils on the main pathogens of strawberry fruit. Postharvest Biol. Technol. 2015, 109, 145–151. [Google Scholar]
- Toroglu, S.; Keskin, D.; Dadandi, M.Y.; Yildiz, K. Comparision of antimicrobial activity of Silene laxa Boiss. & Kotschy and Silene caramanica Boiss. & Heldr different extracts from Turkey. J. Pure Appl. Microbiol. 2013, 7, 1763–1768. [Google Scholar]
- Ozcan, M.M.; Al Juhaimi, F.Y. Antioxidant and antifungal activity of some aromatic plant extracts. J. Med. Plants Res. 2011, 5, 1361–1366. [Google Scholar]
- El Abed, N.; Kaabi, B.; Smaali, M.I.; Chabbouh, M.; Habibi, K.; Mejri, M.; Marzouki, M.N.; Ahmed, S.B. Chemical composition, antioxidant and antimicrobial activities of Thymus capitata essential oil with its preservative effect against Listeria monocytogenes inoculated in minced beef meat. Evid. Based Complement. Altern. 2014, 2014. [Google Scholar] [CrossRef]
- Ballester-Costa, C.; Sendra, E.; Fernandez-Lopez, J.; Perez-Alvarez, J.A.; Viuda-Martos, M. Chemical composition and in vitro antibacterial properties of essential oils of four Thymus species from organic growth. Ind. Crop. Prod. 2013, 50, 304–311. [Google Scholar] [CrossRef]
- Albayrak, S.; Aksoy, A. Essential oil composition and in vitro antioxidant and antimicrobial activities of Thymus cappadocicus Boiss. J. Food Process Preserv. 2013, 37, 605–614. [Google Scholar] [CrossRef]
- Toroglu, S. In vitro antimicrobial activity and antagonistic effect of essential oils from plant species. J. Environ. Biol. 2007, 28, 551–559. [Google Scholar] [PubMed]
- Ruiz-Navajas, Y.; Viuda-Martos, M.; Sendra, E.; Perez-Alvarez, J.A.; Fernández-López, J. Chemical characterization and antibacterial activity of Thymus moroderi and Thymus piperella essential oils, two Thymus endemic species from southeast of Spain. Food Control 2012, 27, 294–299. [Google Scholar] [CrossRef]
- Georgescu, C.; Mironescu, M. Obtaining, characterisation and screening of the antifungal activity of the volatile oil extracted from Thymus serpyllum. J. Environ. Prot. Ecol. 2011, 12, 2294–2302. [Google Scholar]
- Singh, G.; Maurya, S.; Catalan, C.; de Lampasona, M.P. Chemical constituents, antifungal and antioxidative effects of ajwain essential oil and its acetone extract. J. Agric. Food Chem. 2004, 52, 3292–3296. [Google Scholar] [CrossRef] [PubMed]
- Adesegun, E.A.; Adebayo, O.S.; Akintokun, A.K. Antifungal activity of spices extracts against Sclerotium rolfsii. In Proceedings of the 28th International Horticultural Congress on Science and Horticulture for People (IHC)/International Symposium on Organic Horticulture—Productivity and Sustainability, Lisbon, Portugal, 22–27 August 2010; Mourao, I., Aksoy, U., Eds.; International Society for Horticultural Science: Leuven, Belgium, 2012; pp. 415–419. [Google Scholar]
- Kusuda, M.; Inada, K.; Ogawa, T.O.; Yoshida, T.; Shiota, S.; Tsuchiya, T.; Hatano, T. Polyphenolic constituent structures of Zanthoxylum piperitum fruit and the antibacterial effects of its polymeric procyanidin on methicillin-resistant Staphylococcus aureus. Biosci. Biotechnol. Biochem. 2006, 70, 1423–1431. [Google Scholar] [CrossRef] [PubMed]
- Diao, W.R.; Hu, Q.P.; Feng, S.S.; Li, W.Q.; Xu, J.G. Chemical composition and antibacterial activity of the essential oil from green huajiao (Zanthoxylum schinifolium) against selected foodborne pathogens. J. Agric. Food Chem. 2013, 61, 6044–6049. [Google Scholar] [CrossRef] [PubMed]
| Type of Samples | Bacteria and Fungi | Main Results | Reference |
|---|---|---|---|
| Clove and cinnamon water extracts | Staphylococcus aureus and Escherichia coli | Both in vivo and in vitro results confirmed the efficacy of clove extract as natural antimicrobials. | [18] |
| Ultra-fine powders of ball-milled clove | E. coli, S. aureus, Brochothrix thermosphacta, Lactobacillus rhamnosus, Pseudomonas fluorescens | Clove powder showed a strong inhibitory effect with the minimum inhibitory concentrations (MICs) ranging from 1.0% to 2.0% w/v. | [19] |
| Cardamom, cinnamon, clove essential oils (EOs) and phenol | 13 bacterial strains, 7 molds and 2 yeasts | Clove EO possessed the highest antimicrobial activities. | [21] |
| 4 spice EOs | Candida albicans | Clove EO possessed the strongest activities against all C. albicans strains. | [22] |
| 4 spice EOs | Salmonella typhimurium, E. coli, Bacillus cereus, Listeria innocua | Clove EO showed the most effective inhibition with the MICs ranging from 1.25% to 2.50% v/v. | [23] |
| 9 spice EOs | Morganella morganii | Clove EO was the most effective with MIC of 0.13% v/v. | [17] |
| Ethanol extracts from 5 spices and herbs | Listeria monocytogenes, S. aureus, Salmonella enterica | Clove extract was the most effective against bacteria tested. | [24] |
| 13 plant ethanol extracts | L. monocytogenes | Clove extract was the most effective with the MIC of 0.24 mg/mL. | [25] |
| 90 plant extracts | Clostridium spp. | Clove water extract was the most effective among all the water extracts with the MIC ranging from 0.1% to 0.2%. | [26] |
| Ethyl acetate, acetone and methanol extracts of 12 plant species | Kluyveromyces marxianus, Rhodotorula rubra, Klebsiella pneumoniae, Bacillus megaterium, Pseudomonas aeruginosa, S. aureus, E. coli, Enterobacter cloacae, Corynebacterium xerosis, Streptococcus faecalis | Clove possessed the most effective inhibitory effects. | [27] |
| 7 spices, their extracts and EOs | Microorganisms in apple cider | Clove products had the strongest antimicrobial activities compared with other spices tested. | [28] |
| Many spice and herb powders | S. aureus, S. typhimurium, E. coli, B. cereus | 2% level of clove powder was more effective against S. aureus followed by E.coli and S. typhimurium under refrigeration. | [29] |
| EOs and functional extracts of cumin and clove. | E. coli, S. typhimurium, S. aureus, B. cereus, L. monocytogenes | The films of low molecular weight chitosan with a concentration of 2% of EO of clove and clove ethyl heptanoate extract had antimicrobial activities against most strains tested. | [30] |
| EOs and functional extracts of cumin, clove, and elecampane | E. coli, S. typhimurium, B. cereus, S. aureus, L. monocytogenes | Chitosan films added with clove EO showed the best inhibitory effects with the MICs of 500 mg/L. | [31] |
| 3 spice EOs | Enterobacteriaceae, S. aureus, Pseudomonas sp., Lactic acid bacteria, Brocithrix thermosphacta | Clove EO was the most effective against microorganisms tested. | [32] |
| Type of Study | Bacteria and Fungi | Main Results | Reference |
|---|---|---|---|
| Oregano extract | Salmonella gallinarum, Salmonella enteritidis, S. typhimurium | Oregano extract had antibacterial effects on Salmonella serotypes. | [34] |
| EO-rich fractions of oregano | S. aureus, B. subtilis, E. coli, P. aeruginosa, C. albicans, Aspergillus niger | All of the supercritical fluid extraction fractions showed antimicrobial activities against all tested microorganisms. | [35] |
| Oregano EO | C. albicans, Candida krusei, Candida tropicalis, B. cereus, E. coli, S. aureus, Yersinia enterocolitica, S. enterica, Serratia marcescens | Heating treatment showed no significant effects on the antimicrobial activities of EO. | [36] |
| 4 spice decoctions | F. oxysporum f. sp. phaseoli, Macrophomina phaseoli, Botrytis cinerea, Rhizoctonia solani, Alternaria solani, Alternaria parasiticus | The 10% level of oregano decoction was 100% inhibitive to mycelial growth in the culture medium. | [39] |
| 4 plant aqueous extracts | E. coli and B. subtilis | Oregano extract had the highest antibacterial activities against all tested bacteria. | [40] |
| Oregano and marjoram EOs | S. aureus isolated from poultry meat. | All the isolates tested were sensitive to EO of oregano. | [41] |
| 3 spice EOs | 13 bacterial strains and 6 fungi | Oregano EO showed the most effective antibacterial activities. | [42] |
| 6 spice EOs | Staphylococcus xylosus, Staphylococcus carnosus, Lactobacillus sakei, Lactobacillus curvatus, Enterobacter gergoviae, Enterobacter amnigenus | Oregano EO was the most effective. | [43] |
| 8 spice EOs | E. coli strains isolated from poultry and cattle faeces. | Oregano EO was the most effective against E. coli. | [44] |
| Oregano and A. sieberi EOs | Candida glabrata isolated from patients with vulvovaginal candidiasis. | The MICs of oregano EO ranged from 0.5 to 1100 μg/mL for all tested C. glabrata isolates. | [45] |
| 7 spice EOs and the majority constituents | 33 Staphylococcus spp. isolates | Oregano and thyme EOs possessed the equal and strongest antimicrobial activities among EOs. | [46] |
| 4 spice EOs | S. aureus and E. coli isolated from vongole and bacteria standard ATCC: E. coli, S. aureus, Salmonella choleraesuis | Oregano presented antimicrobial activities against all tested bacteria. | [47] |
| Various spice EOs | Microorganisms in fresh leaf lettuce and radish sprouts. | Oregano-2 (in the USA) oil was the most effective at maintaining the reduced levels of total mesophilic microorganisms. | [48] |
| Pure EOs of 4 spices and chitosan-EOs films | L. monocytogenes and E. coli | Both oregano EO alone and incorporated in the films possessed the best antimicrobial activities. | [49] |
| Type of Samples | Tested Bacteria and Fungi | Main Results | Reference |
|---|---|---|---|
| Thyme EO | B. subtilis, S. aureus, Staphylococcus epidermidis, P. aeruginosa, E. coli, Mycobacterium smegmatis, C. albicans, Candida vaginalis | MICs ranged from 75 to 1100 μg/mL for bacteria, and from 80 to 97 μg/mL for fungi. | [51] |
| Thyme EOs of 4 ontogenetic stages | E. coli, Proteus mirabilis, Proteus vulgaris, S. typhimurium, S. marcescens, Y. enterocolitica, P. fluorescens, Pseudomonas putida, Micrococcus spp., S. flava, S. aureus, Bacillus licheniformis, Bacillus thuringiensis, L. innocua | All the thyme EOs had significant antibacterial activities against the microorganisms tested. | [52] |
| 4 Thymus species EOs | P. aeruginosa, Cronobacter sakazakii, L. innocua, Streptococcus pyogenes, C.albicans, Saccharomyces cerevisiae | Thyme EO was the most efficient on all the tested bacteria and yeast both in original and half-diluted concentrations. | [53] |
| 5 spice hydrosols | B. subtilis and S. enteritidis | Thyme hydrosol was more effective than sage, peppermint, and black pepper. | [55] |
| 6 plant hydrosols | S. aureus, E. coli, S. typhimurium, P. aeruginosa, C. albicans | 15% hydrosol concentration of thyme completely inhibited E. coli and S. typhimurium. | [56] |
| 5 plant EOs | P. fluorescens, P. putida, Pseudomonas fragi, Brochothrix thermosphacta C. albicans, P. aeruginosa | Thyme EO showed the highest antimicrobial activities with MICs ranging from 0.05% to 0.8% w/v. | [57] |
| 5 aromatic plant EOs | 13 Vibrio alginolyticus strains | The MICs of thyme EO ranged from 0.078 to 0.31 mg/mL, and MBCs ranged from 0.31 to 1.25 mg/mL. | [58] |
| 5 spice EOs | L. innocua, S. marcescens, P. fluorescens | Only the thyme EO showed inhibition effects on all tested bacteria at all added doses. | [59] |
| 4 spice EOs | B. cereus, L. monocytogenes, S. typhimurium, E. coli | MICs of thyme EO ranged from 0.312 to 1.25 μL/mL. | [60] |
| Various EOs | 18 pathogens and 15 spoilage bacteria | Thyme EO showed the strongest antibacterial activities against spoilage bacteria. | [48] |
| 17 spices and herbs | Shigella sonnei and Shigella flexneri | MICs of thyme ranged from 0.5% to 1% (w/v) depending on the Shigella strains used. | [61] |
| Thyme infusion, decoction and hydroalcoholic extracts | S. aureus, S. epidermidis, E. coli, Klebsiella spp., P. aeruginosa, Enterobacter aerogenes, P. vulgaris, Enterobacter sakazakii | Decoction presented the most pronounced effects. | [62] |
| Thyme EO, hydrosol and propolis extracts | Natural mycobiota on the surface of sucuk | Thyme EO and propolis extract provided reductions of 2.45 and 2.05 log CFU/g in yeast-mold counts respectively. | [63] |
| Polyphenolic fractions and oil fractions from 4 spice oilseeds | 35 bacterial strains | Thyme oil fraction had the highest antibacterial activities comparing with other spices oilseeds. | [64] |
| Thymol, nisin, carvacrol, cymene | Candida lusitaniae | Thymol completely inhibited the yeast growth at concentrations over 1 mmol/L for at least 21 days at 25 °C. | [65] |
| Type of Samples | Tested Bacteria and Fungi | Mian Results | Reference |
|---|---|---|---|
| Cinnamon extract and oil | 7 Gram-positive bacteria, 3 Gram-negative bacteria, and 7 fungi | Cinnamon oil was more effective than cinnamon extract with MICs ranging from 1.25% to 5% v/v. | [70] |
| Cinnamon, sodium benzoate, potassium sorbate | E. coli | E. coli was reduced by 1.6 log CFU/mL at 8 °C and 2.0 log CFU/mL at 25 °C by 0.3% cinnamon. | [71] |
| Cinnamon oil and liposome-encapsulated cinnamon oil | Methicillin resistant Staphylococcus aureus (MRSA) | Cinnamon oil possessed effective antibacterial activity and prominent anti-biofilm activity against MRSA. | [72] |
| 4 spice aqueous extracts | A. niger, Fusarium sambucinum, Pythium sulcatum, Rhizopus stolonifer | 0.05 g/mL of cinnamon extract completely inhibited A. niger and P. sulcatum, 0.10 g/mL of cinnamon extract completely inhibited F. sambucinum. | [74] |
| 5 plant aqueous extracts | S. aureus, Lactobacillus sp., B. thermosphacta, Pseudomonas spp., E. coli | Cinnamon aqueous extract inhibited all the tested microorganisms at the concentration of 1%. | [75] |
| 6 spice hydrosols | S. aureus, E. coli, S. typhimurium, P. aeruginosa, C. albicans | The percent inhibition ranged from 10% to 33.8% at 5% hydrosol of cinnamon. | [56] |
| 6 spice diethyl ether extracts | S. aureus, K. pneumoniae, P. aeruginosa, E. coli, Enterococcus faecalis, M. smegmatis, Micrococcus luteus, C. albicans | Cinnamon possessed inhibitory activities against all the tested microorganisms. | [76] |
| Ethyl acetate, acetone, and methanol extracts from 12 plants | K. pneumonia, B. megaterium, P. aeruginosa, S. aureus, E. coli, E. cloacae, C. xerosis, S. faecalis, K. marxianus, R. rubra | The methanol extract of cinnamon showed antibacterial activities against all the microorganisms tested. | [27] |
| 7 Indian spice ethanol extracts | 215 enterococcal strains | Crude ethanol extract of cinnamon was the most effective against all the clinical isolates. | [77] |
| 8 spice EOs | B. cereus, E. coli, L. monocytogenes, S. rissen, P. fluorescens, S. aureus, Candida lipolytica, Hanseniaspora uvarum, Pichia membranaefaciens, Rhodotorula glutinis, Schizosaccharomyces pombe, Zygosaccharomyces rouxii, A. flavus, Aspergillus versicolor, A. parasiticus, Fusarium moniliforme | Cinnamon EO possessed the strongest inhibition effects with the MICs ranging from 0.015 to 2.0 mg/mL. | [78] |
| 10 spice EOs | B. cereus, B. subtilis, E. coli, K. pneumoniae, L. monocytogenes, P. aeruginosa, S. aureus, S. enterica, S. marcencens, Y. enterocolitica | Cinnamon EO was efficient in inhibiting all tested bacterial strains. | [79] |
| Type of Study | Bacteria and Fungi | Main Results | Reference |
|---|---|---|---|
| Cumin EO | E. coli, S. aureus, S. faecalis, P. aeruginosa, K. pneumoniae | E. coli, S. aureus, and S. faecalis were sensitive to various cumin EO dilutions. | [81] |
| Cumin seeds EO | 1230 fungal isolates obtained from food samples | The EO was fungicidal against most of the fungal species at MIC of 0.6 μL/mL. | [82] |
| Cumin seeds EOs | E. coli, S. aureus, P. aeruginosa, A. niger, A. parasiticus, C. albicans | Both microwave and conventionally (oven) roasted cumin oils showed higher effects than raw oils. | [83] |
| Cumin EO | E. coli, P. aeruginosa, B. cereus, S. aureus | ã-Irradiation to cumin seeds at 10 and 25 kGy had no significant effects on the antibacterial effects. | [80] |
| Aqueous infusions and aqueous decoctions from kalonji, cumin and poppy seed | 188 bacterial isolates isolated from oral cavity of apparently healthy individuals | Aqueous decoction of cumin inhibited 73% of the tested bacteria. | [84] |
| Cumin and C. carvi EOs | E. coli, the genera Pseudomonas, Clavibacter, Curtobacterium, Rhodococcus, Erwinia, Xanthomonas, Ralstonia, Agrobacterium | Cumin EO showed antibacterial activities against all tested bacteria except Pseudomonas viridiflava. | [85] |
| 9 spice EOs | S. typhimurium, B. cereus, S. aureus, E. faecalis, E. coli. Y. enterocolitica, S. cerevisiae, Candida rugosa, Rhizopus oryzae, A. niger | Cumin EO was effective against all bacterial species and two fungi (S. cerevisiae and Candida rugosa). | [86] |
| 4 spice EOs | S. aureus, Salmonella sp., E. coli, P. aeruginosa, etc. | Cumin EO was effective against E. coli, P. aeruginosa and Salmonella sp. | [87] |
| 6 spice EOs | L. curvatus, L. sakei, S. carnosus, S. xylosus, E. gergoviae, E. amnigenus | Cumin EO was the second effective among tested spices. | [43] |
| 5 spice EOs | M. luteus, B. megaterium, Brevibacillus brevis, E. faecalis, Pseudomonas pyocyaneus, M. smegmatis, E. coli, Aeromonas hydrophila, Y. enterocolitica, S. aureus, S. faecalis, S. cerevisiae, Kluvyeromyces fragilis | Cumin inhibited all tested bacteria and fungi and showed synergistic and antagonistic effect with antibiotics. | [88] |
| EOs of 9 spices in combination | B. cereus, L. monocytogenes, M. luteus, S. aureus, E. coli, S. typhimurium | Coriander/cumin seed oil combination showed synergistic interactions on antibacterial activities. | [89] |
| Type of Study | Bacteria and Fungi | Main Results | Reference |
|---|---|---|---|
| Rosemary EO | Leuconostoc mesenteroides, Lactobacillus delbruekii, S. cerevisiae, C. krusei | Rosemary EO showed higher effects against bacteria tested than yeasts. | [91] |
| Rosemary and sage EOs | C. albicans, Trichophyton mentagrophytes, Trichophyton tonsurans, Trichophyton rubrum, Epidermophyton floccosum, Microsporum canis, P. aeruginosa, E. coli, S. typhimurium, S. enteritidis, Shigella sonei, Micrococcus flavus, Sarcina lutea, S. aureus, S. epidermidis, B. subtilis | The EO of rosemary showed significant antifungal activities and antibacterial activities. | [92] |
| 7 spice and herb extracts | E. coli, S. typhimurium, L. monocytogenes, S. aureus | The hexane extract of rosemary exhibited significantly higher antibacterial activities than ethanol and water extracts. | [93] |
| 5 plant ethanol extracts | B. cereus, E. coli, P. aeruginosa, S. aureus, L. monocytogenes | Rosemary extract was the most effective against all the tested microorganisms. | [94] |
| 14 spice ethanol extracts and their mixture | L. monocytogenes, E. coli, P. fluorescens, L. sake | The mixture of rosemary and liquorice extracts was the most effective against all tested bacteria. | [95] |
| 5 spice aqueous extracts | 8 Gram-positive bacteria and 12 Gram-negative bacteria | MICs ranged from 0.125 to 0.5 mg/mL for Gram-positive bacteria and 0.25–0.5 mg/mL for Gram-negative bacteria. | [96] |
| 2 spice and herb extract combinations | L. monocytogenes, S. aureus and naturally present spoilage microflora on cooked ready-to-eat shrimp stored for 16 days at 4 or 8 °C | Both combination of galangal, rosemary, and lemon and combination of galangal and rosemary significantly reduced levels of aerobic bacteria and lactic acid bacteria. | [97] |
| Rosemary, mint and a mixture of tocopherols | Microorganisms from the sausages | The addition of rosemary resulted in significant inhibition of microbial growth and showed possible synergistic effects with mint. | [90] |
| 5 spice EOs | M. luteus, B. megaterium, B. brevis, E. faecalis, P. pyocyaneus, M. smegmatis, E. coli, A. hydrophila, Y. enterocolitica, S. aureus, S. faecalis, S. cerevisiae, K. fragilis | Rosemary EO showed synergic effects with cephalothin. | [88] |
| Type of Study | Bacteria and Fungi | Main Results | Reference |
|---|---|---|---|
| Fresh garlic, garlic powder, garlic oil | Microorganisms in raw chicken sausage | The order of antimicrobial activities were fresh garlic > garlic powder > garlic oil > butylated hydroxyanisole. | [100] |
| Garlic powder | S. aureus, E. coli, S. typhimurium, B. cereus, and a mixed lactic culture consisting of Lactobacillus delbrueckii subsp. bulgaricus and Streptococcus thermophilus | Fresh garlic produced the greatest inhibition followed by freeze-dried powder. | [99] |
| Chopped garlic | Microorganisms in ground beef and raw meatball | Chopped garlic had slowing-down effects on microbiological growth. | [101] |
| 5 spice extracts | 20 serogroups of E. coli, 8 serotypes of Salmonella, L. monocytogenes and A. hydrophila | Garlic extract exhibited significant activities against microorganisms except L. monocytogenes at all concentrations. | [103] |
| 3 ethanol extracts | K. pneumoniae, S. aureus, M. morganii, C. albicans, E. coli, P. vulgaris | Garlic extract exerted superior antibacterial activities at all concentrations | [104] |
| 6 spice crude ethanol, hexane and acetone extracts | E. coli, S. aureus, S. flexneri, Streptococcus pneumoniae | Garlic was the most effective against all the tested pathogens except S. flexneri. | [105] |
| 7 spice crude extracts | Phoma exigua, Fusarium nygamai, R. solani | Cold water extract of garlic exhibited good antifungal activities against all three tested fungi. | [106] |
| Garlic aqueous extract | S. aureus | The first and second week samples were significantly decreased by all the 1, 2, and 3-mL garlic extracts. | [98] |
| 5 spice hydrosols | B. subtilis and S. enteritidis | Garlic hydrosols demonstrated stronger antibacterial activities than other spices hydrosols. | [54] |
| Type of Sample | Bacteria and Fungi | Main Results | Reference |
|---|---|---|---|
| 3 spice extracts and EOs | 5 strains of L. monocytogenes, 4 strains of S. typhimurium DT104 | Commercial EOs of ginger inhibited all L. monocytogenes at ≤ 0.6 | [111] |
| Ginger EO and oleoresin | Aspergillus terrus, A. niger, A. flavus, Trichothecium roseum, Fusarium graminearum, Fusarium oxysporum, Fusarium monoliforme, Curvularia palliscens | EO and oleoresin of ginger were 100% antifungal against F. oxysporum and A. niger, respectively. | [109] |
| Ginger ethanol and n-hexane extracts | Porphyromonas gingivalis, Porphyromonas endodontalis, Prevotella intermedia | Only [10]-gingerol and [12]-gingerol effectively inhibited the growth of tested bacteria at a MIC range of 6–30 µg/mL. | [107] |
| Ginger oil extracted by hydrodistillation and solvent extraction method | B. subtilis, Bacillus nutto, P. aerugenosa, Rhodoturola sp., Samonella newport, S. enteritidis, Fusarium sp. | Extracts obtained by both extraction methods inhibited listed microorganisms. | [108] |
| Ginger and mustard EOs | Vibrio species | Ginger and mustard EOs inhibited the growth of Vibrio parahaemolyticus and Vibrio vulnificus. | [110] |
| 5 spice extracts | 20 serogroups of E. coli, 8 serotypes of Salmonella, L. monocytogenes and A. hydrophila. | Ginger extract possessed inhibitory effects on two serogroups of E. coli. | [102] |
| 4 spice water extracts | A. niger, F. sambucinum, P. sulcatum, R. stolonifera | Ginger extract significantly inhibited the mycelial growth of tested microorganisms. | [74] |
| 7 spice crude extracts | P. exigua, F. nygamai, R. solani | In the case of the hot water extracts, garlic and ginger showed the best antifungal activities. | [106] |
| 7 spice ethanol extracts | 215 enterococcal strains isolated from clinical samples | Ginger was found to have antibacterial activities against all the isolates. | [76] |
| Type of Samples | Bacteria and Fungi | Main Results | Reference |
|---|---|---|---|
| EO from aerial parts of basil | S. aureus, E. coli, B. subtilis, Pasteurella multocida, A. niger, Mucor mucedo, F. solani, Botryodiplodia theobromae, R. solani | All the tested microorganisms were sensitive to EOs of basil. | [113] |
| Chloroform, acetone and methanol extracts of basil | E. gallinarum, E. faecalis, B. subtilis, E. coli, Shigella sp., S. pyogenes, S. aureus, L. monocytogenes, P. aeruginosa, S. cerevisiae, C. albicans, C. crusei | The methanol extract inhibited P. aeruginosa, Shigella sp., L. monocytogenes, S. aureus, and two strains of E. coli. | [114] |
| Basil extracts | Fusarium oxysporum, Fusarium proliferatum, Fusarium subglutinans, Fusarium verticillioides | At the concentration of 1.50% v/v, basil extract completely inhibited Fusarium spp. tested. | [115] |
| EOs from 12 cumin cultivars | B. cereus, M. flavus, S. aureus, E. faecalis, E. coli, P. aeruginosa, S. typhimurium, L. monocytogenes, 7 fungi, Aspergillus fumigatus, A. niger, A. versicolor, Aspergillus ochraceus, Penicillium funiculosum, Penicillium ochrochloron, Trichoderma viride | MICs of basil EOs ranged from 0.009 to 23.48 μg/mL for bacteria and 0.08–5.00 μg/mL for fungi. | [116] |
| 7 spice EOs | A. flavus | Basil EO completely inhibited A. flavus at 150 μL/100 mL. | [117] |
| 8 spice EOs | Histamine-producing bacteria including M. morganii | Basil EO inhibited M. morganii with the MIC of 2.39 mg/mL. | [17] |
| Type of Sample | Bacteria and Fungi | Main Results | Reference |
|---|---|---|---|
| Fennel seeds EO | Streptococcus mutans | MICs: 80 ppm | [120] |
| Fennel seeds EO | Streptomyces albus, B. subtilis, S. typhimurium, P. aeruginosa, Shigella dysenteriae, E. coli | EO of fennel seeds inhibited several foodborne pathogens with lowest MIC of 0.125 mg/mL. | [119] |
| Fennel crude extract | E. coli, S. blanc, P. merabilis, P. vulgaris, S. epidemidis, Staphylococcus saprophyticus, A. versicolor, A. fumigates, Penicilium camemberti | Fennel crude extract had antimicrobial activities against all nine microorganisms, especially fungi. | [121] |
| Fennel EO and acetone extract | A. niger, A. flavus , Aspergillus oryzae, A. ochraceus, F. graminearum, F. moniliforme, P. ctrium, Penicillium viridicatum, Penicillium madriti, Curvularia lunata | Fennel EO showed complete zone inhibition against several strains at 6 μL dose. | [118] |
| Cumin and fennel EOs | S. typhimurium and E. coli | The MICs of fennel EO was 0.031% v/v against S. typhimurium and 0.062% v/v E. coli. | [122] |
| 8 spice methanol and ethanol extracts | B. subtilis, E. faecalis, L. innocua, E. coli, P. putida, Providencia stuartii, Acetobacter calcoaceticus | Fennel seeds extracts showed the largest zones of inhibitions in six out of the seven bacteria. | [123] |
| Type of Sample | Bacteria and Fungi | Main Results | Reference |
|---|---|---|---|
| Coriander EO and linalool | Campylobactor jejuni and Campylobactor coli strains | MICs ranged between 0.5 and 1 mL/mL. | [124] |
| Coriander EO | A. baumannii strains | MICs and MBCs ranged between 1 and 4 μL/mL. | [125] |
| Coriander EO and 6 antibacterial drugs | A. baumannii strains | Coriander EO showed synergistic action with chloramphenicol, ciprofloxacin, and tetracycline. | [126] |
| Coriander leaves EO | Candida spp. | MICs ranged from 15.6 to 31.2 µg/mL, and MFCs ranged from 31.2 to 62.5 µg/mL. | [127] |
| Coriander EO | 12 bacterial strians | MICs of coriander against all tsted bacteria ranged from 0.1% to 1.6%, v/v. | [128] |
| EOs of 6 coriander accessions | Colletotrichum acutatum and Colletotrichum gloeosporioides | Coriander EOs could inhibit Colletotrichum genus at higher application rates. | [129] |
| Coriander EO and oleoresin | Aspergillus terreus, A. niger, F. graminearum, F. oxysporum | Both EO and oleoresin of coriander were effective against tested fungi. | [130] |
| Coriander ethanol and aqueous-ethanol extracts | B. subtilis, S. aureus, P. vulgaris, E. coil, P. aeruginosa, K. peunomonia, L. monocytogenes, C. albicans | The ethanol extract showed clear difference and more potent against tested microorganisms in comparison with the aqueous-ethanol extract. | [131] |
| Coriander EO | - | Microwave and conventionally roasted oils exhibit similar antimicrobial effects but were higher effect than raw oils. | [83] |
| 4 spice EOs | Microorganisms isolated from clinical specimens of patients | Coriander oil was active only against Salmonella sp. | [87] |
| Lemon EO, coriander extract and cinnamon extract | A. parasiticus, Cladosporium cladosporioides, Eurotium herbariorum, Penicillium chrysogenum and Aspergillus carbonarius | Coriander extract had the best antifungal activities in the vapor phase | [132] |
| Type of Sample | Bacteria and Fungi | Main Results | Reference |
|---|---|---|---|
| 7 spice and herb extracts | E. coli, S. typhimurium, L. monocytogenes, S. aureus | Galangal hexane and ethanol extracts had strong antimicrobial activities against S. aureus and L. monocytogenes. | [93] |
| Combination of extracts from galangal, rosemary and lemon iron bark | S. aureus, L. monocytogenes, E. coli, S. typhimurium, Clostridium perfringens | Galangal showed synergistic activities against tested microorganisms with rosemary and lemon iron bark. | [134] |
| Galangal methanol, acetone and diethyl ether extracts | B. subtilis, E. aerogenes, E. cloacae, E. faecalis, E. coli, K. pneumoniae, P. aeruginosa, S. typhimurium, S. aureus, S. epidermis | The activities of methanol extract at pH 5.5 were excellent with MICs ranging from 0.04 to 1.28 mg/mL. | [133] |
| 4 Alpinia strains methanol extracts | 6 strains of bacteria and 4 strains of fungi | Galangal flower possessed the highest activity against M. luteus and only the extract from galangal rhizome showed antifungal activity toward A. niger. | [135] |
| Type of Samples | Bacteria and Fungi | Main Results | Reference |
|---|---|---|---|
| Black pepper EO and acetone extract | A. flavus, A. ochraceus, A.oryzae, A. niger, F. moniliforme, F. graminearum, Penicillium citrinum, Penicillium viridcatum, P. madriti, Curvularia lunata | The EO was effective against F. graminearum, while the acetone extract was effective against P. viridcatum and A. ochraceus. | [136] |
| 4 spice EOs and acetone extracts | S. aureus, B. cereus, B. subtilis, E. coli, S. typhimurium, P. aeruginosa | Black pepper extracts showed complete reduction of colonies against tested bacterial strains at 5 and 10 μL levels. | [137] |
| Various solvent extracts, piperine and piperic acid from pepper | E. coli, K. pneumonia, S. enterica, S. aureus, S. epidermidis, E. faecalis, B. subtilis | The ethanol extract was the most effective with the MICs ranging from 156.25 to 1250 μg/mL. | [138] |
| Bacteria and Fungi | Spices | Type of Samples | Reference |
|---|---|---|---|
| E. coli | Clove | Aqueous extract | [18] |
| Acetone extract | [27] | ||
| EO | [23,30,31] | ||
| Ethyl acetate extract | [27] | ||
| Ethyl heptanoate extract | [30] | ||
| Methanol extract | [27] | ||
| Powder | [19,29] | ||
| Oregano | Aqueous extract | [40] | |
| EO | [36,44,47,49] | ||
| EO-rich fractions | [35] | ||
| Thyme | Decoction | [62] | |
| EO | [51,52,60] | ||
| Hydroalcoholic extract | [62] | ||
| Hydrosol | [56] | ||
| Infusion | [62] | ||
| Cinnamon | Acetone extract | [27] | |
| Aqueous extract | [75] | ||
| Diethyl ether extract | [76] | ||
| EO | [78,79] | ||
| Ethyl acetate extract | [27] | ||
| Hydrosol | [56] | ||
| Methanol extract | [27] | ||
| Powder | [71] | ||
| Cumin | EO | [80,81,83,85,86,87,88,89] | |
| Rosemary | Aqueous extract | [93] | |
| EO | [88,92] | ||
| Ethanol extract | [93,94,95] | ||
| Hexane extract | [93] | ||
| Garlic | Acetone extract | [105] | |
| Aqueous extract | [103] | ||
| Ethanol extract | [104,105] | ||
| Hexane extract | [105] | ||
| Powder | [99] | ||
| Ginger | Aqueous extract | [103] | |
| Basil | Acetone extract | [114] | |
| Chloroform extract | [114] | ||
| EO | [113,116] | ||
| Methanol extract | [114] | ||
| Fennel | Crude extract | [121] | |
| EO | [121,122] | ||
| Ethanol extract | [123] | ||
| Methanol extract | [123] | ||
| Coriander | Aqueous-ethanol extract | [131] | |
| Ethanol extract | [131] | ||
| Galangal | Acetone extract | [133] | |
| Diethyl ether extract | [133] | ||
| Hexane extract | [134] | ||
| Methanol extract | [133] | ||
| Black pepper | Acetone extract | [137] | |
| EO | [137] | ||
| Ethanol extract | [138] | ||
| S. aureus | Clove | Acetone extract | [27] |
| Aqueous extract | [18] | ||
| EO | [30,31,32] | ||
| Ethanol extract | [24] | ||
| Ethyl heptanoate extract | [30,31] | ||
| Methanol extract | [27] | ||
| Powder | [19,29] | ||
| Oregano | EO | [36,41,47] | |
| EO-rich fractions | [35] | ||
| Thyme | Decoction | [62] | |
| EO | [51,52] | ||
| Hydroalcoholic extract | [62] | ||
| Infusion | [62] | ||
| Cinnamon | Acetone extract | [27] | |
| Aqueous extract | [75] | ||
| Diethyl ether extract | [76] | ||
| EO | [72,78,79] | ||
| Ethyl acetate extract | [27] | ||
| Hydrosol | [56] | ||
| Methanol extract | [27] | ||
| Cumin | EO | [80,81,83,86,88,89] | |
| Rosemary | EO | [88,92] | |
| Ethanol extract | [94,97] | ||
| Hexane extract | [93] | ||
| Garlic | Aqueous extract | [98] | |
| Acetone extract | [105] | ||
| Ethanol extract | [104,105] | ||
| Hexane extract | [105] | ||
| Powder | [99] | ||
| Basil | Acetone extract | [114] | |
| Chloroform extract | [114] | ||
| EO | [113,116] | ||
| Methanol extract | [114] | ||
| Coriander | Aqueous-ethanol extract | [131] | |
| Ethanol extract | [131] | ||
| Galangal | Acetone extract | [133] | |
| Diethyl ether extract | [133] | ||
| Ethanol extract | [93] | ||
| Hexane extract | [93,134] | ||
| Methanol | [133] | ||
| Black pepper | Acetone extract | [137] | |
| EO | [137] | ||
| Ethanol extract | [138] | ||
| L. monocytogenes | Clove | Ethanol extract | [24,25] |
| Ethyl heptanoate extract | [30,31] | ||
| EO | [30,31] | ||
| Oregano | EO | [49] | |
| Thyme | EO | [60] | |
| Cinnamon | EO | [78,79] | |
| Cumin | EO | [89] | |
| Rosemary | Aqueous extract | [93] | |
| Ethanol extract | [93,94,95,97] | ||
| Hexane extract | [93] | ||
| Garlic | Aqueous extract | [103] | |
| Ginger | EO | [111] | |
| Basil | Acetone extract | [114] | |
| Chloroform | [114] | ||
| EO | [116] | ||
| Methanol extract | [114] | ||
| Coriander | Aqueous-ethanol extract | [131] | |
| Ethanol extract | [131] | ||
| Galangal | Ethanol extract | [93] | |
| Hexane extract | [93,134] | ||
| S. typhimurium | Clove | EO | [23,30,31] |
| Ethyl heptanoate extract | [30] | ||
| Powder | [29] | ||
| Oregano | Extract | [34] | |
| Thyme | EO | [60,52] | |
| Hydrosol | [56] | ||
| Cumin | EO | [86,89] | |
| Rosemary | Aqueous extract | [93] | |
| EO | [92] | ||
| Ethanol extract | [93] | ||
| Hexane extract | [93] | ||
| Garlic | Powder | [99] | |
| Basil | EO | [116] | |
| Fennel | EO | [121,122] | |
| Galangal | Acetone extract | [133] | |
| Diethyl ether extract | [133] | ||
| Hexane extract | [134] | ||
| Methanol extract | [133] | ||
| Black pepper | Acetone extract | [137] | |
| EO | [137] | ||
| P. aeruginosa | Clove | Acetone extract | [27] |
| EO | [32] | ||
| Ethyl acetate extract | [27] | ||
| Methanol extract | [27] | ||
| Oregano | EO-rich fractions | [35] | |
| Thyme | Decoction | [62] | |
| EO | [51,53,57] | ||
| Hydroalcoholic extract | [62] | ||
| Infusion | [62] | ||
| Cinnamon | Diethyl ether extract | [76] | |
| EO | [79] | ||
| Hydrosol | [56] | ||
| Methanol extract | [27] | ||
| Cumin | EO | [83,87] | |
| Rosemary | EO | [92] | |
| Ethanol extract | [94] | ||
| Ginger | EO | [108] | |
| Basil | Acetone extract | [114] | |
| Chloroform | [114] | ||
| EO | [116] | ||
| Methanol extract | [114] | ||
| Fennel | EO | [121] | |
| Coriander | Aqueous-ethanol extract | [131] | |
| Ethanol extract | [131] | ||
| Galangal | Acetone extract | [133] | |
| Diethyl ether extract | [133] | ||
| Methanol extract | [133] | ||
| Black pepper | Acetone extract | [137] | |
| EO | [137] | ||
| B. subtilis | Oregano | Aqueous extract | [40] |
| EO-rich fractions | [35] | ||
| Thyme | EO | [51] | |
| Hydrosol | [55] | ||
| Cinnamon | EO | [79] | |
| Rosemary | EO | [92] | |
| Garlic | Hydrosol | [55] | |
| Ginger | EO | [108] | |
| Basil | EO | [113] | |
| Fennel | EO | [121] | |
| Ethanol extract | [123] | ||
| Methanol extract | [123] | ||
| Coriander | Aqueous-ethanol extract | [131] | |
| Ethanol extract | [131] | ||
| Galangal | Acetone extract | [133] | |
| Diethyl ether extract | [133] | ||
| Methanol extract | [133] | ||
| Black pepper | Acetone extract | [137] | |
| EO | [137] | ||
| Ethanol extract | [138] | ||
| B. cereus | Clove | EO | [23,30,31] |
| Ethyl heptanoate extract | [30] | ||
| Oregano | EO | [36] | |
| Thyme | EO | [60] | |
| Cinnamon | EO | [78,79] | |
| Cumin | EO | [80,86,89] | |
| Rosemary | Ethanol extract | [94] | |
| Garlic | Powder | [99] | |
| Basil | EO | [116] | |
| Black pepper | Acetone extract | [137] | |
| EO | [137] | ||
| E. faecalis | Cinnamon | Diethyl ether extract | [76] |
| Cumin | EO | [86,88] | |
| Rosemary | EO | [88] | |
| Basil | EO | [116] | |
| Fennel | Ethanol extract | [123] | |
| Methanol extract | [123] | ||
| Galangal | Acetone extract | [133] | |
| Diethyl ether extract | [133] | ||
| Methanol extract | [133] | ||
| Black pepper | Ethanol extract | [138] | |
| E. faecalis | Cinnamon | Diethyl ether extract | [76] |
| Cumin | EO | [86,88] | |
| Basil | EO | [116] | |
| Fennel | Ethanol extract | [123] | |
| Methanol extract | [123] | ||
| Galangal | Acetone extract | [133] | |
| Diethyl ether extract | [133] | ||
| Methanol extract | [133] | ||
| Black pepper | Ethanol extract | [138] | |
| K. pneumoniae | Clove | Acetone extract | [27] |
| Ethyl acetate | [27] | ||
| Methanol extract | [27] | ||
| Cinnamon | Acetone extract | [27] | |
| Diethyl ether extract | [76] | ||
| EO | [79] | ||
| Ethyl acetate | [27] | ||
| Methanol extract | [27] | ||
| Garlic | Ethanol extract | [104] | |
| Galangal | Acetone extract | [133] | |
| Diethyl ether extract | [133] | ||
| Methanol extract | [133] | ||
| Black pepper | Ethanol extract | [138] | |
| P. vulgaris | Thyme | Decoction | [62] |
| EO | [52] | ||
| Hydroalcoholic extract | [62] | ||
| Infusion | [62] | ||
| Garlic | Ethanol extract | [104] | |
| Fennel | Crude extract | [121] | |
| Coriander | Aqueous-ethanol extract | [131] | |
| Ethanol extract | [131] | ||
| P. fluorescens | Clove | Powder | [19] |
| Thyme | EO | [57,59,52] | |
| Cinnamon | EO | [78] | |
| Rosemary | Ethanol extract | [95] | |
| L. innocua | Clove | EO | [23] |
| Thyme | EO | [52,59,52] | |
| Fennel | Ethanol extract | [123] | |
| Methanol extract | [123] | ||
| S. faecalis | Clove | Acetone extract | [27] |
| Ethyl acetate | [27] | ||
| Methanol extract | [27] | ||
| Cinnamon | Acetone extract | [27] | |
| Ethyl acetate | [27] | ||
| Methanol extract | [27] | ||
| Cumin | Decoctions | [81] | |
| EO | [88] | ||
| Infusions | [81] | ||
| S. enteritidis | Thyme | Hydrosol | [55] |
| Garlic | Hydrosol | [55] | |
| Ginger | EO | [108] | |
| M. luteus | Cinnamon | Diethyl ether extract | [76] |
| Cumin | EO | [88] | |
| Galangal | Methanol extract | [135] | |
| B. megaterium | Clove | Acetone extract | [27] |
| Ethyl acetate | [27] | ||
| Methanol extract | [27] | ||
| Cinnamon | Acetone extract | [27] | |
| Ethyl acetate | [27] | ||
| Methanol extract | [27] | ||
| Cumin | EO | [88] | |
| A. hydrophila | Cumin | EO | [88] |
| Garlic | Aqueous extract | [103] | |
| S. epidermidis | Thyme | Decoction | [62] |
| EO | [51] | ||
| Hydroalcoholic extract | [62] | ||
| Infusion | [62] | ||
| Black pepper | Ethanol extract | [138] | |
| C. albicans | Clove | EO | [22] |
| Oregano | EO | [35,36] | |
| Thyme | EO | [51,57] | |
| Cinnamon | Hydrosol | [56,76] | |
| Diethyl ether extract | [76] | ||
| Cumin | EO | [83] | |
| Rosemary | EO | [92] | |
| Garlic | Ethanol extract | [104] | |
| Coriander | Aqueous-ethanol extract | [131] | |
| EO | [127] | ||
| Ethanol extract | [131] | ||
| A. niger | Oregano | EO-rich fraction | [35] |
| Cinnamon | Aqueous extract | [74] | |
| Cumin | EO | [83] | |
| Ginger | Aqueous extract | [74,109] | |
| EO | [109] | ||
| Oleoresin | [109] | ||
| Basil | EO | [113,116] | |
| Fennel | Acetone extract | [118] | |
| EO | [118] | ||
| Coriander | EO | [130] | |
| Galangal | Methanol extract | [135] | |
| A. flavus | Basil | EO | [117] |
| Fennel | Acetone extract | [118] | |
| EO | [118] | ||
| F. oxysporum | oregano | Decoction | [39] |
| ginger | EO | [109] | |
| basil | Extract | [115] | |
| coriander | EO | [130] | |
| Oleoresin | [130] | ||
| F. graminearum | Fennel | Acetone extract | [118] |
| EO | [118] | ||
| Coriander | EO | [130] | |
| Oleoresin | [130] | ||
| Black pepper | Acetone extract | [136] | |
| EO | [136] | ||
| S. cerevisiae | Thyme | EO | [52] |
| Cumin | EO | [86] |
| Spices | Type of Samples | Bacteria and Fungi | Main Results | Reference |
|---|---|---|---|---|
| Achillea species | Ethanol extract | K. pneumoniae, E. cloacae, S. typhimurium, S. epidermis, E. coli, E. aerogenes, S. aureus, Klebsiella oxytoca, S. pyogenes, P. aeruginosa, C. albicans | Achillea species showed a broad spectrum of strong antibacterial activities against all tested microorganisms. | [140] |
| Achillea millefolium | Ethanol extract | S. aureus, S. enteritidis, E. coli, S. pneumoniae, K. pneumoniae, P. aeruginosa, E. aerogenes, P. mirabilis, A. niger, C. albicans | The antibacterial activities of A. millefolium were greater or similar to other penicillin derivatives but lesser than Ampicillin. | [141] |
| Aframomum corrorima | Seeds, pods, leaves and rhizomes extract | A. flavus and Penicillum expansum | A. corrorima crude seed extract was the most active against A. flavus and P. expansum at concentration of 0.4 mg/mL. | [142] |
| Allium hirtifolium Boiss. | Hydromethanol extract | MRSA, S. epidermidis, S. pneumoniae, E. coli, S. typhimurium, P. mirabilis, K. pneumoniae | A. hirtifolium extract was effective against 10 species of pathogenic bacteria with MICs ranging from 1.88 to 7.50 mg/mL. | [143] |
| Allium roseum L. | Extracts of bulbs, leaves, flowers and seeds by 3 extraction methods | S. aureu, S. epidermidis, M. luteus, B. cereus, B. subtilis, E. faecalis, S. typhimurium, E. coli, P. aeruginosa, C. albicans | A. roseum extract showed very significant antimicrobial activities to strains such as C. albicans (MICs: 1.00–3.44 μg/μL) and E. coli (MICs: 2.00–3.44 μg/μL). | [144] |
| Allium ursinum L. | Pressurized-liquid extract | S. aureus and A. niger | A. ursinum extract showed antimicrobial activities against S. aureus with DIZs of 12 and 10 mm (two parallel determinations) and A. niger of 6 mm. | [145] |
| Amomum kravanh | EO | Different foodborne pathogens | A. kravanh EO exhibited the best antibacterial activities against B. subtilis and E. coli. | [146] |
| Anethum graveolens L. | EO and acetone extract | P. citrinum, A. niger, S. aureus, B. cereus, P. aeruginosa | EO and extract showed different but both effective activities against tested microorganisms. | [147] |
| Anethum graveolens L. | diethyl-ether extract | P. aeruginosa, E. coli, K. pneumoniae, M. luteus, E. faecalis, B. megaterium, S. aureus | A. graveolens extract affected all of the bacteria tested. | [148] |
| Anethum graveolens L. | EO | A. flavus | A. graveolens EO is the most effective against aflatoxin production. | [117] |
| Brassica jancea | EO | Vibrio parahaemolyticus and Vibrio vulnificus | B. jancea EO could inhibit V. parahaemolyticus and Vibrio vulnificus inoculated sliced raw flatfish at 5 °C of storage. | [110] |
| Brassica jancea | Water extract | E. coli, S. aureus, B. cereus | B. jancea extract showed good inhibitory action at 1% concentration. | [149] |
| Bunium persicum | Volatile compounds | F. oxysporum | B. persicum showed the strongest effect compared with other 51 spices and herbs. | [150] |
| Caesulia axillaris Roxb. | EO | A. flavus | C. axillaris EO showed complete inhibition against A. flavus at 1.0 μg/mL. | [151] |
| Capsicum froutescens | Ethanol extract | S. aureus | C. froutescens extract showed the highest activity. | [152] |
| Capsicum frutescens L. | n-hexane, chloroform, ethyl acetate, acetone, and methanol extracts of dried seeds | B. cereus, S. aureus, MRSA, E. coli, S. typhimurium, P. aeruginosa, K. pneumoniae, P. vulgaris, C. albicans, C. krusei | Microwave assisted solvent extracts showed significant activities and n-hexane extract was effective against P. aeruginosa and C. albicans, while ethyl acetate extract was effective against C. krusei. | [153] |
| Carum capticum | EO | Corynebacterium diphtheriae, S. aureus, Staphylococcus haemolyticus, B. subtilis, P. aeruginosa, E. coli, Klebsiella species, P. vulgaris | C. capticum was very effective against all tested bacteria. | [154] |
| Carum copticum | EO | S. aureus, B. cereus, E. coli, S. enteritidis, L. monocytogenes | C. copticum EO was the most effective against tested bacteria with MICs of 0.03–0.5 mg/mL compared with two other spices. | [155] |
| Cinnamomum burmannii | Methanol crude extract | B. cereus, L. monocytogenes, S. aureus, E. coli, Salmonella anatum | MIC and MBC for B. cereus were 625 and 2500 μg/mL respectively, for four other bacteria were more than 2500 μg/mL. | [156] |
| Cinnamomum cassia | Ultra-fine powder | E. coli, S. aureus, P. fluorescens, L. rhamnosus, B. thermosphacta | C. cassia powder significantly reduced the microorganisms tested at the concentration ≤2.5% w/v and the inhibitory effects were positive correlated with concentrations. | [19] |
| Cinnamomum tamala | Leaves EO | C. albicans, A. niger, A. fumigatus, R. stolonifer, Penicillium spp. | The MFCs of EO against all the tested fungi were 230 μg/mL. | [157] |
| Cinnamomum verum | Bark and leaf extracts and EO | Bacteria isolated from urine samples, and A. niger | C. verum oil possessed stronger antimicrobial activities than extracts. A. niger showed no growth in the presence of oil. | [158] |
| Cinnamomum verum | EO | E. coli, S. typhimurium, S. aureus, B. subtilis, A. flavus, C. albicans | C. verum EO treated group showed significant decrease in viable bacterial counts. | [159] |
| Cinnamomum verum | EO | S. typhimurium, S. paratyphi, E. coli, S. aureus, P. fluorescens, B. licheniformis | C. verum bark EO showed the best antibacterial activities with mean MICs ranging from 2.9 to 4.8 mg/mL. | [160] |
| Citrus aurantium L. | Ethanol extract | E. coli, P. aeruginosa, S. aureus, B. cereus | C. aurantium showed strong antimicrobial activities against tested bacteria. | [161] |
| Clinopodium ascendens | EO | S. aureus, S. faecium, S. mutans, Agrobacterium tumefasciens, E. coli, B. cinerea, C. albicans | C. ascendens exhibited remarkable activity against E. coli and was active against A. tumefasciens, S. aureus, and B. cinerea. | [162] |
| Corydothymus capitatus | EO | P. putida | C. capitatus EO was the most active with a MIC of 0.025% w/v and a MTC of 0.006% w/v. | [163] |
| Cotoneaster nummularioides | Leaves EO | B. cereus, S. aureus, Salmonella entrica, E. coli | The extract of C. nummularioides showed strong effects on two Gram-positive microorganisms tested with higher sensitivity for B. cereus (MIC: 3.125 mg/mL). | [164] |
| Croton hirtus | EO | E. coli, S. aureus | C. hirtus EO was effective against S. aureus with MIC of 512 μg/mL. | [165] |
| Cuminum nigrum L. | Polyphenolic compounds | B. subtilis, B. cereus, Enterobacter spp., E. coli, L. monocytogenes, S. aureus, Y. enterocolitica | C. nigrum extract possessed significantly inhibitory effects on B. subtilis, B. cereus, and S. aureus. | [166] |
| Curcuma longa | Curcumin | S. aureus | Antibacterial activity of curcumin against S. aureus was enhanced with the increase of the concentration. | [167] |
| Cunila galioides | EO from aerial parts | 15 bacterial species including Bacillus sp., L. monocytogenes, S. aureus, A. hydrophila, E. faecalis etc. | The oil of C. galioides citral efficiently controlled some microorganisms, showing both contact and gaseous activity. | [168] |
| Dichrostachys glomerata | Methanol extract | Providencia stuartii, P. aeruginosa, K.pneumoniae, E. coli, E. aerogenes, E. cloacae | D. glomerata extract inhibited the growth of all the 29 tested bacteria with MICs ≤ 1024 μg/mL. | [169] |
| Echinops giganteus | Methanol extract | Mycobacterium tuberculosis H(37)Rv, Mycobacterium tuberculosis H37Ra | The extract of E. giganteus was the most effective with MICs of 32 μg/mL and 16 μg/mL, respectively against H37Ra and H(37)Rv, compared with other 19 spices. | [170] |
| Elettaria cardamomum | Ethanol extract | 4 strains of Gram-positive bacteria and 12 strains of Gram-negative bacteria | E. cardamomum extract was effective against a majority of the pathogens, MICs ranged from 9.4 to 18.75 mg/mL except E. coli, B. cereus, and E. cloacae which had a great sensitivity to the spice extract (MICs < 2.34 mg/mL). | [171] |
| Elettaria cardamomum | EO and various oleoresins | S. aureus, B. cereus, E. coli, S. typhimurium, A. terreus, Penicillium purpurogenum, F. graminearum, Penicillium madriti | The EO showed strong effects against bacteria tested at 3000 ppm, and the methanol and ethanol oleoresins gave the best results against A. terreus at 3000 ppm. | [172] |
| Eucalyptus globulus | Hydrodistillated extract | S. aureus, B. subtilis, L. innocua, E. coli, P. aeruginosa | E. globulus extract showed an inhibition effects against all the tested bacteria with MIC of 3 and 4 mg/mL. | [173] |
| Eucalyptus largiflorens | EO | A. flavus, A. parasiticus, A. niger, Penicillium chryzogenum, P. citrinum | The leaf oil of E. largiflorens showed higher antifungal activities than four other Eucalyptus spices. | [174] |
| Eucalyptus radiata | EO | P. aeruginosa, E. coli , K. pneumoniae, S. typhimurium, Acinetobacter baumannii, P. aeruginosa, K. pneumoniae | E. radiate showed better antibacterial activities with MICs ranging from 8 to 32μL/mL. | [175] |
| Eugenia caryophyllum Bullock and Harrison | Aqueous extract | S. aureus, S. typhimurium, E. coli, S. epidermidis, L. plantarum, P. vulgaris | The MICs and MBCs against all tested bacteria ranged from 1 to 4 g/L and 2 to 8 g/L, respectively. | [176] |
| Foeniculum vulgare ssp. piperitum | EO | A. alternate, F. oxysporum, R. solani | 100% fungistatic effects were observed with 40 ppm doses of F. vulgare oils. | [177] |
| Glaucium elegans | Methanol extract | E. coli, S. aureus, S. enteritidis, Bacillus anthracis, Proteus | G. elegans methanol extract had significant antibacterial effects. | [178] |
| Gloriosa superba Linn | Methanol extract and fractions in different solvent systems | C. albicans, Candida glaberata, Trichophyton longifusus, M. canis, S. aureus, E. coli, B. subtilis, K. pneumonae, S. flexneri, S. typhimurium | The n-butanol fraction of G. superba showed excellent antifungal activities and chloroform fraction showed the highest antibacterial activity against S. aureus. | [179] |
| Helichrysum species | Methanol extracts | 13 bacteria and 2 yeasts | All the extracts showed significant antimicrobial activities against all tested microorganisms. | [180] |
| 4 Helichrysum Mill. plants | Methanol extracts | A. hydrophila, Bacillus brevis, B. cereus, K. pneumoniae, P. aeruginosa, S. aureus, E. coli, M. morganii, M. smegmatis, P. mirabilis, Y. enterocolitica, S. cerevisiae | The methanol extracts had antibacterial activities against the first six microorganisms listed. | [181] |
| horseradish | Aqueous extract | S. aureus | Horseradish water extract showed a higher biological activity. | [182] |
| Hyssopus officinalis L. | EO | A. niger, A. ochraceus, A. versicolor, A. fumigatus, Cladosporium cladosporioides, Cladosporium fulvum, Penicillium funiculosum, Penicillium ochrochloron, Trichoderma viride, C. albicans | All tested EO and deodorized extracts showed activities with the MICs ranging from 4 to 16 mg/mL. | [183] |
| Laser trilobum L. | Methanol extract | S. aureus, P. vulgaris, P. mirabilis, B. cereus, A. hydrophila, E. faecalis, K. pneumoniae, S. typhimurium, E. aerogenes, E. coli | The fruit extract had significant antimicrobial effects on pathogen bacteria. | [184] |
| Laurus nobilis | Ethanol extract | 4 Gram-positive bacteria and 12 Gram-negative bacteria | L. nobilis extract was effective in inhibiting a majority of the pathogens, MICs ranged from 4.7 to 9.4 mg/mL. | [185] |
| Laurus nobilis L. | EO and leaves ethanol, water and hot water extract | B. thermosphacta, E. coli, L. innocua, L. monocytogenes, P. putida, S. typhimurium, Shewanella putrefaciens | L. nobilis EO exhibited strong antibacterial activities against all tested bacteria. | [186] |
| Laurus nobilis L. | Aqueous, ethanol, ethyl acetate and hexane extracts | B. cereus, S. aureus, E. coli, K. pneumoniae, C. albicans | Only aqueous extract of L. nobilis showed anticandidal activities among the tested 8 plants. | [187] |
| Lavandula officinalis | EO | L. innocua and P. fluorescens | L. officinalis EO showed the highest activity against L. innocua. | [188] |
| Lichen Xanthoria parietina | Acetone extract | S. aureus, E. faecalis, P. vulgaris, P. mirabilis, S. typhimurium, E. cloacae, E. aerogenes, P. aeruginosa, K. pneumoniae, R. solani, Botridis cinerea, C. albicans | X. parietina acetone extract and parietin showed similar activities on the nine bacteria tested, but less active than parietin on the three fungi tested. | [189] |
| Lippia grandis Schauer. | EO | E. coli, P. aeruginosa, K. pneumoniae, S. aureus, E. faecalis | The EO was effective against 75% of the microorganisms analyzed especially S. aureus, E. faecalis, and E. coli. | [190] |
| Lippia javanica | Acetone and aqueous extracts | S. aureus, L. monocytogenes, S. typhimurium, E. coli, A. fumigatus, A. niger, M. canis, Microsporum gypseum, T. tonsurans, T. rubrum, T. mucoides, Penicillium aurantiogriseum, Penicillium chrysogenum | The aqueous and acetone extracts were active against the bacterial strains, and the acetone extract exhibited the antifungal activities higher than even the reference drugs. | [191] |
| Lippia origanoides H.B.K. | EO | C. albicans, Candida parapsilosis, Candida guilliermondii, Cryptococcus neoformans, Trichophyton rubrum, Fonsecaea pedrosoi, S. aureus, Lactobacillus casei, S. mutans | L. origanoides EO showed highly significant inhibition zones for all microorganisms tested. | [192] |
| Litsea cubeba | EO | E. coli | The MIC and MBC of L. cubeba against E. coli were both 0.125% v/v. | [193] |
| Melissa officinalis L. | Ethanol, ethyl acetate and aqueous extracts | Agrobacterium tumefaciens, Bacillus mycoides, B. subtilis, E. cloaceae, Erwinia carotovora, E. coli, Proteus sp., P. fluorescens, S. aureus | M. officinalis ethanol, ethyl acetate, and aqueous extracts significantly enhanced the effectiveness of tested preservatives (sodium benzoate, sodium nitrite, and potassium sorbate). | [194] |
| Mentha piperita L. | EO | T. rubrum, T. tonsurans, T. schoenleinii, T. mentagrophytes, M. canis, M. fulvum | For effective concentration of M. piperita oil against tested antropophilic dermatophytes, and MICs ranged from 0.1 to 1.5 μL/mL. | [195] |
| Mentha spicata L. | hexane, chloroform, ethyl acetate, and aqueous fractions of ethanol extract | Salmonella paratyphi, Shigella boydii, S. aureus, E. coli, Vibrio cholera, P. aeruginosa, E. faecalis, S. typhimurium, P. vulgaris, K. pneumoniae | M. spicata ethanol extract and its solvent fractions effectively inhibited half of the microorganism growth. | [196] |
| Myristica argentea | Water extract | E. coli and S. aureus | M. argentea were more effective against E. coli (MIC of 9.80 mg/mL) and S. aureus (MIC of 6.20 mg/mL). | [197] |
| Myristica fragrans | - | 20 different serogroups of E. coli, 8 serotypes of Salmonella, L. monocytogenes, A. hydrophila | M. fragrans showed good anti-listerial activity, although activities against E. coli and Salmonella were serotype dependent. | [103] |
| Myristica fragrans | Ethyl acetate and ethanol extracts of flesh, mace and seed | S. mutans, Streptococcus mitis, Streptococcus salivarius, Aggregatibacter actinomycetemcomitans, P. gingivalis, Fusobacterium nucleatum | Flesh ethyl acetate extract had the highest effects against tested bacteria with mean MICs ranging from 0.625 to 1.25 mg/mL among all tested extracts. | [198] |
| Myrtus communis | EO | P. aeruginosa, S. typhimurium, E. coli, A. hydrophila, L. monocytogenes, C. albicans | M. communis EO exhibited antimicrobial activities against all tested microorganisms, especially Gram-negative bacteria. | [199] |
| Myrtus communis L. | Methanol, ethyl acetate, acetone extracts | S. aureus, P. vulgaris, P. mirabilis | The most effective extract was the methanol extract from M. communis leaves against S. aureus. | [200] |
| Myrica gale L. | EO | A. flavus, Cladosporium cladosporioides, Penicillium expansum | A complete antifungal activity was observed at 1000 ppm of M. gale EO against Cladosporium cladosporioides. | [201] |
| Nepeta alpina | EO | Bacillus pumilus, E. coli, Kocuria varians, L. monocytogenes, P. aeruginosa, S. typhimurium, A. niger, A. flavus, C. glabrata | The EO was active against L. monocytogenes with MIC of 32 μg/mL. | [202] |
| Nigella saliva L. | Aqueous extracts | Uromyces appendiculatus | N. saliva extract was effective against U. appendiculatus and controlled rust similar to mancozeb fungicide at 2 and 3% concentrations. | [203] |
| Nigella sativa L. | n-hexan extract | 24 pathogenic, spoilage and lactic acid bacteria | N. sativa oil showed antibacterial activities against all the bacteria at all concentrations (0.5%, 1.0% and 2.0%) tested. | [204] |
| Ocimum canum | EO | B. subtilis, E. coli, K. pneumoniae, M. luteus, P. aeruginosa, Raoultella planticola, S. typhimurium, S. mutans | MICs of O. canum ranged from 0.43 to 2.08 μL/mL against 7 out of 10 bacteria tested. | [205] |
| Ocimum gratissimum L. | EO | A. flavus, A. niger, Aspergillus fumigatus, Aspergillus terreus, Aspergillus sydowi, Aspergillus alternate, Penicillium italicum, Fusarium nivale, C. lunata, Cladosporium spp. | The EO exhibited antifungal activities against fungal isolates from some spices and showed better efficacy as fungi toxicant than prevalent fungicide Wettasul-80. | [206] |
| Ocimum sanctum | EO | A. flavus | MIC: 0.3 μL/mL. | [207] |
| Ocimum sanctum L. | EO | A. flavus, Aspergillus fumigatus, Aspergillus clavatus, Aspergillus orizae S. aureus, E. faecalis, E. coli, enterohemorrhagic E. coli, P. aeruginosa, S. flexneri | O. sanctum EO exhibited antimicrobial activities against all tested pathogens at concentrations of 0.125–32 μL/mL except P. aeruginosa. | [208] |
| Ocimum suave | EO | S. aureus, S. epidermidis, S. mutans, S. viridans, E. coli, E. cloacae, K. pneumoniae, P. aeruginosa, C. albicans, C. tropicalis, C. glabrata | O. suave EO showed the strongest antibacterial activities with MICs ranging from 0.05 to 1.37 mg/mL. | [209] |
| Olea europaea L. | Methanol extract | S. aureus, S. epidermidis, S. pyogenes, Streptococcus agalactiae, S. enterica serovar Typhi, P. aeruginosa, Acetobacter calcoaceticus, C. albicans, P. vulgaris, S. faecalis, S. dysenteriae, K. pneumoniae, E. coli, V. cholera, C. xerosis | O. europaea methanol extract showed strong antibacterial activities against S. aureus, S. epidermidis, and S. pyogenes at MICs range of 31.25–62.5 μg/mL. | [210] |
| Origanum marjorana | Water extract | Vibrio parahaemolyticus | O. marjorana showed the lowest MICs against V. parahaemolyticus both in a nutrient rich and poor medium. | [211] |
| Origanum minutiflorum | EO | E. coli, S. aureus, S. enteritidis, L. monocytogenes, L. plantarum | Whey protein based edible films incorporated with O. minutiflorum EO was the most effective at 2% level. | [212] |
| Orthosiphon stamineus Benth. | Methanol and aqueous extracts | V. parahaemolyticus | V. parahaemolyticus was more susceptible to 50–100% methanol extracts of O. stamineus. | [213] |
| Peganum harmala L. | Methanol extract | S. aureus, S. epidermidis, S. pyogenes, S. agalactiae, S. enterica serovar Typhi, P. aeruginosa, Acetobacter calcoaceticus, C. albicans, P. vulgaris, S. faecalis, S. dysenteriae, K. pneumoniae, E. coli, V. cholera, C. xerosis | P. harmala seed showed MICs of 31.25–62.5, 250, 125–250, and 31.25–250 μg/mL, respectively for S. aureus, S. enterica serovar Typhi, Acetobacter calcoaceticus, and C. albicans. | [210] |
| Pimenta dioica L. | Alcoholic and hexane extracts | P. fluorescens, B. megaterium, A. niger, Penicillium sp. | Alcoholic and hexane extracts of P. dioica exerted significant inhibitory effects on both the bacteria and fungi. | [214] |
| Pimpinella anisum L. | EO of fruit | A. alternate, A. niger, A. parasiticus | The most sensitive fungus for P. anisum oil was A. parasiticus. | [215] |
| Pimpinella anisum L. | EO | 16 microorganisms | P. anisum EO exhibited strong antifungal activities against R. glutinis, A. ochraceus, and F. moniliforme. | [78] |
| Pimpinella anisum L. | EO | C. lipolytica, H. uvarum, Pichia membranaefaciens, R. glutinis, S. pombe, Z. rouxii, A. flavus, A. ochraceus, A. parasiticus, F. moniliforme | P. anisum EO completely inhibited the growth of tested fungi. | [78] |
| Piper capense | EO | S. aureus, E. faecalis, C. albicans | P. capense showed moderate activities against tested microorganisms. | [216] |
| Piper guineense | powder | B. cereus, Bacillus coagulans, B. enterobacter sp., A. niger, R. stolonifer | P. guineense inhibited R. stolonifer at concentrations above 0.5%. | [217] |
| Phlomis oppositiflora | Methanol, ethanol, ethyl acetate extracts and EO | E. coli, S. aureus, K. pneumonia, M. smegmatis, P. aeruginosa, E. cloacae, B. megaterium, M. luteus, R. rubra, C. albicans, K. marxianus | P. oppositiflora contains antimicrobial components against various microorganisms. | [218] |
| Ramalina species | Acetone, methanol and ethanol extracts | E. coli and S. aureus | The MICs of all extracts ranged from 64 to 512 g/mL for all bacterial strains tested. | [219] |
| Rhus coriaria L. | 80% (v/v) aqueous alcohol extract | S. aureus, B. cereus, E. coli, S. typhimurium, P. vulgaris, S. flexneri | The MICs of R. coriaria extract against the tested bacteria ranged from 0.04% to 0.2%. | [220] |
| Rhus coriaria | Water extract | B. cereus, L. monocytogenes, E. coli, S. typhimurium | R. coriaria extract was the most effective against the four bacteria tested. | [221] |
| Salvia officinalis L. | EO | 13 bacterial strains and 6 fungi | Sage EO was more effective against E. coli, S. typhimurium, S. enteritidis, and S. sonei. | [92] |
| Salvia officinalis L. (sage) | 80% ethanol extract | Campylobacter coli, E. coli, Streptococcus infantis, B. cereus, L. monocytogenes, S. aureus | Sage extract showed the best antibacterial activities compared with four other plants, especially against Gram-positive bacteria and C. coli. | [222] |
| Salvia officinalis L. | EO | E. coli, P. aeruginosa, Enterobacter sp., S. aureus | Microwave-EO of S. officinalis possessed good antibacterial activities than the hydrodistilled oil. | [223] |
| Salvia leriifolia | Methanol extract | S. aureus | S. leriifolia extract exhibited antimicrobial activity against S. aureus. | [224] |
| Santolina chamaecyparissus L. | EO | K. pneumonia and C. albicans | S. chamaecyparissus EO was very active against the two microorganisms listed. | [225] |
| Satureja cuneifolia Ten. | EO | E. coli, Campylobacter jejuni, S. sonnei, S. aureus, L. monocytogenes, B. cereus, P. aeruginosa, S. enteritidis | MICs of S. cuneifolia EO for tested bacteria were in the range of 600–1400 μg/mL. | [226] |
| Satureja kitaibelii | EO | 30 pathogenic microorganisms | S. kitaibelii EO showed significant activities against foodborne microbes (MIC: 0.18–25.5 μg/mL), multiresistant bacterial isolates (MIC: 6.25–50.0 μg/mL), and dermatophyte strains (MIC: 12.5–50.0 μg/mL). | [227] |
| Satureja wiedemanniana | EO | 37 Bacillus strains | Both S. wiedemanniana EO and its main component p-cymene exhibited strong antimicrobial activities against some Bacillus strains. | [228] |
| Satureja species | EOs | A. niger, Penicillium digitatum, B. cinerea, R. stolonifer | The EOs exhibited fungicidal activities against P. digitatum, B. cinereal, and R. stolonifer. | [229] |
| Silene laxa | Ethyl acetate, chloroform, methanol, ethanol and acetone extract | P. aeruginosa, E. cloacae, B. megaterium, E. cloacae, S. aureus | S. laxa leaves ethanol extract showed the best activities against P. aeruginosa, E. cloacae, B. megaterium, while the methanol extracts of S. laxa fruits showed the best antibacterial activity against B.megaterium. | [230] |
| Summer savory | - | A. niger, A. alternate, A. parasiticus | 0.5% summer savory extract showed 100% inhibition till the seventh day of incubation. | [231] |
| Syzygium aromaticum L. | Water extract | S. aureus, S. epidermidis, S. pyogenes, S. agalactiae, S. enterica serovar Typhi, P. aeruginosa, Acetobacter calcoaceticus, C. albicans, P. vulgaris, S. faecalis, S. dysenteriae, K. pneumoniae, E. coli, V. cholera, C. xerosis | S. aromaticum water extract showed antibacterial activities with MICs in the range of 31.25–250 μg/mL for S. aureus, S. epidermidis, S. pyogenes, S. enterica serovar Typhi, Acetobacter calcoaceticus, and P. aeruginosa. | [210] |
| Thymbra spicata L. | Decoction | F. oxysporum f. sp. phaseoli, M. phaseoli, B. cinerea, R. solani, A. solani, A. parasiticus | T. spicata completely inhibited the mycelial growth of fungi and showed a complete fungicidal effect on molds. | [39] |
| Thymus capitata | EO | L. monocytogenes | MICs ranged from 0.32 to 20 mg/mL. | [232] |
| Thymus capitatus | EO | L. innocua, S. marcescens, P. fragi, P. fluorescens, A. hydrophila, Shewanella putrefaciens, Achromobacter denitrificans, E. amnigenus, E. gergoviae, Alcaligenes faecalis, Leuconostoc carnosum | T. capitatus EOs showed inhibitory effects on the 10 tested bacteria with MICs ranging from 1.87 to 7.5 μL/mL. | [233] |
| Thymus cappadocicus Boiss. | EO | 13 bacteria and 2 yeasts | T. cappadocicus EO showed great antimicrobial activities against microorganisms tested. | [234] |
| Thymus eigii | EO | M. luteus, B. megaterium, B. brevis, E. faecalis, P. pyocyaneus, M. smegmatis, E. coli, A. hydrophila, Y. enterocolitica, S. aureus, S. faecalis, S. cerevisiae, K. fragilis | T. eigii EO showed the highest antimicrobial activities compared with two other plants. | [235] |
| Thymus piperella | EO | L. innocua, S. marcescens , P. fragi, P. fluorescens, A. hydrophila, S. putrefaciens, A. denitrificans, E. amnigenus, E. gergoviae, A. faecalis, L. carnosum | T. piperella EO had inhibitory effects on 5 of the 11 bacteria tested. | [236] |
| Thymus serpyllum | EO | Penicillium sp., Alternaria sp., Aureobasidium sp. | 8 mg/disc EO of T. serpyllum has a good efficiency by inhibiting the germination of spores from 80% to 100%. | [237] |
| Trachyspermum ammi L. | EO | A. niger, A. flavus, A. oryzae, A. ochraceus, F. monoliforme, F. graminearum, Pencillium citrium, P. viridicatum, P. madriti, C. lunata | T. ammi EO exhibited a broad spectrum of fungi toxic behavior against all tested fungi. | [238] |
| Xylopia aethiopica | - | Sclerotium rolfsii | X. aethiopica extract was the most effective against S. rolfsii compared with four other spices. | [239] |
| Zanthoxylum piperitum | Polymeric procyanidin | S. aureus | A polymeric proanthocyanidin purified from the fruit of Z. piperitum, noticeably decreased the MICs of β-lactam antibiotics for MRSA. | [240] |
| Zanthoxylum schinifolium | EO | S. aureus, S. epidermidis, B. subtilis, S. typhimurium, P. aeruginosa, S. dysenteriae, E. coli | Z. schinifolium EO was particularly strong against S. epidermidis, with MIC 2.5 mg/mL. | [241] |
| Zataria multiflora Boiss. | 80% (v/v) aqueous alcohol extract | S. aureus, B. cereus, E. coli, S. typhimurium, P. vulgaris, S. flexneri | The MICs of Z. multiflora against the tested bacteria ranged from 0.4% to 0.8%. | [220] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Q.; Meng, X.; Li, Y.; Zhao, C.-N.; Tang, G.-Y.; Li, H.-B. Antibacterial and Antifungal Activities of Spices. Int. J. Mol. Sci. 2017, 18, 1283. https://doi.org/10.3390/ijms18061283
Liu Q, Meng X, Li Y, Zhao C-N, Tang G-Y, Li H-B. Antibacterial and Antifungal Activities of Spices. International Journal of Molecular Sciences. 2017; 18(6):1283. https://doi.org/10.3390/ijms18061283
Chicago/Turabian StyleLiu, Qing, Xiao Meng, Ya Li, Cai-Ning Zhao, Guo-Yi Tang, and Hua-Bin Li. 2017. "Antibacterial and Antifungal Activities of Spices" International Journal of Molecular Sciences 18, no. 6: 1283. https://doi.org/10.3390/ijms18061283
APA StyleLiu, Q., Meng, X., Li, Y., Zhao, C.-N., Tang, G.-Y., & Li, H.-B. (2017). Antibacterial and Antifungal Activities of Spices. International Journal of Molecular Sciences, 18(6), 1283. https://doi.org/10.3390/ijms18061283