Resilience and Vulnerability to Pain and Inflammation in the Hippocampus
Abstract
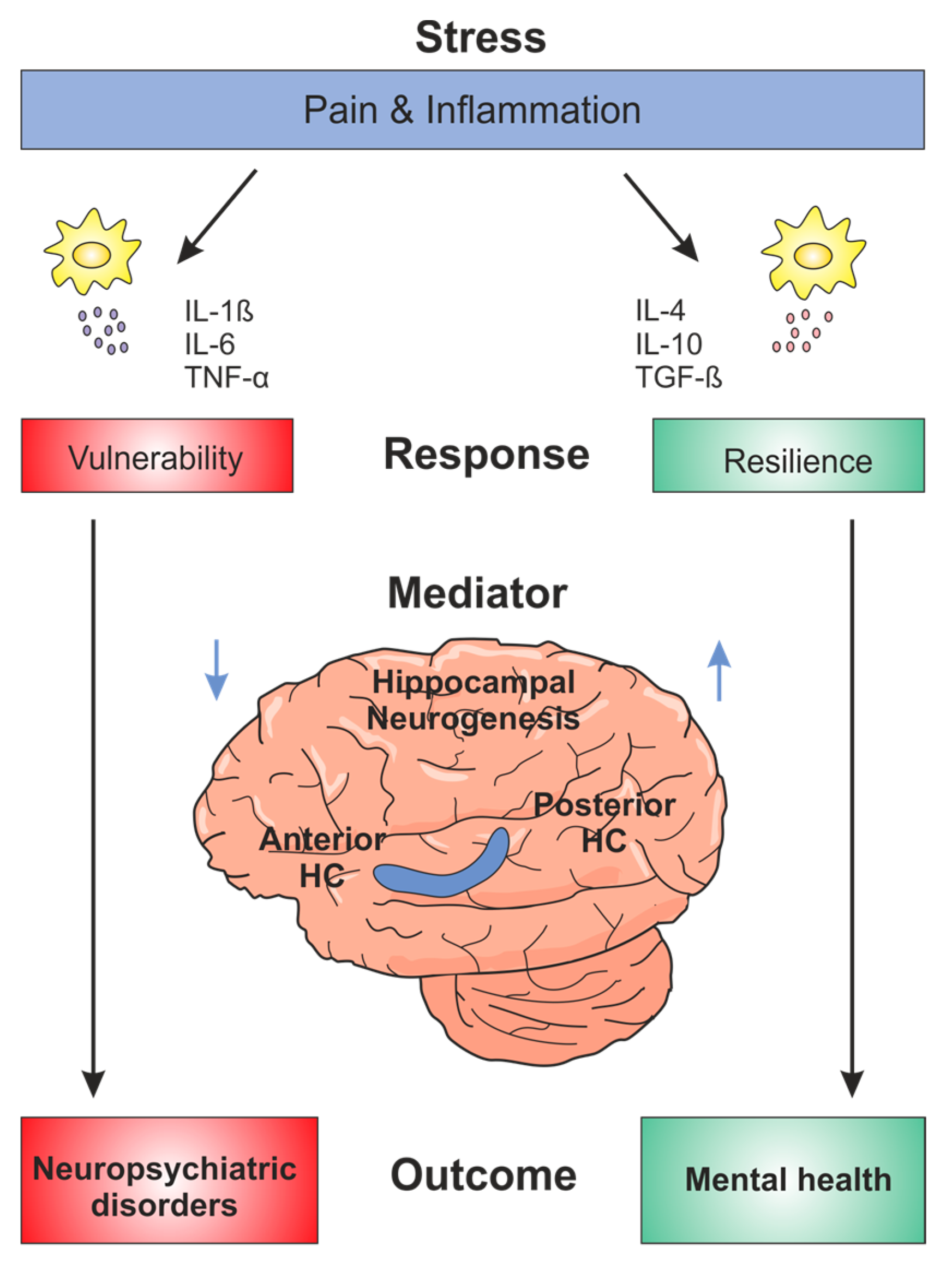
:1. Introduction
2. Hippocampus
2.1. Structure and Function
2.2. Hippocampal Neurogenesis
3. Inflammation
3.1. Inflammation and Neurogenesis
3.2. Inflammation and Neurological Disorders
4. Pain
4.1. Pain and Neurogenesis
4.2. Pain and Neurological Disorders
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| CA1-3 | Cornu Ammonis |
| DG | Dentate gyrus |
| SVZ | Subventricular zone |
| SGZ | Subgranular zone |
| NSCs | Neural stem cells |
| NPCs | Neural progenitor cells |
| CNS | Central nervous system |
| TNF | Tumor necrosis factor |
| IL | Interleukin |
| TGF-β | Transforming growth factor-β |
| IGF-1 | Insulin-like growth factor |
| NGF-1 | Nerve growth factor |
| BDNF | Brain-derived neurotrophic factor |
| LTP | Long-term potentiation |
| LPS | Lipopolysaccharide |
| SE | Status epilepticus |
References
- Kempermann, G.; Kuhn, H.G.; Gage, F.H. More hippocampal neurons in adult mice living in an enriched environment. Nature 1997, 386, 493. [Google Scholar] [CrossRef] [PubMed]
- Shimazu, K.; Zhao, M.; Sakata, K.; Akbarian, S.; Bates, B.; Jaenisch, R.; Lu, B. Nt-3 facilitates hippocampal plasticity and learning and memory by regulating neurogenesis. Learn. Mem. 2006, 13, 307–315. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Li, D.; Shimazu, K.; Zhou, Y.-X.; Lu, B.; Deng, C.-X. Fibroblast growth factor receptor-1 is required for long-term potentiation, memory consolidation, and neurogenesis. Biol. Psychiatry 2007, 62, 381–390. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, M.L.; Brachman, R.A.; Martinowich, K.; Schloesser, R.J.; Herkenham, M. Glucocorticoids orchestrate divergent effects on mood through adult neurogenesis. J. Neurosci. 2013, 33, 2961–2972. [Google Scholar] [CrossRef] [PubMed]
- Parihar, V.K.; Hattiangady, B.; Kuruba, R.; Shuai, B.; Shetty, A.K. Predictable chronic mild stress improves mood, hippocampal neurogenesis and memory. Mol. Psychiatry 2011, 16, 171–183. [Google Scholar] [CrossRef] [PubMed]
- Driscoll, I.; Howard, S.; Stone, J.; Monfils, M.; Tomanek, B.; Brooks, W.; Sutherland, R. The aging hippocampus: A multi-level analysis in the rat. Neuroscience 2006, 139, 1173–1185. [Google Scholar] [CrossRef] [PubMed]
- Kempermann, G. Regulation of adult hippocampal neurogenesis–implications for novel theories of major depression. Bipolar Disord. 2002, 4, 17–33. [Google Scholar] [CrossRef] [PubMed]
- Ferri, A.L.; Cavallaro, M.; Braida, D.; di Cristofano, A.; Canta, A.; Vezzani, A.; Ottolenghi, S.; Pandolfi, P.P.; Sala, M.; de Biasi, S. Sox2 deficiency causes neurodegeneration and impaired neurogenesis in the adult mouse brain. Development 2004, 131, 3805–3819. [Google Scholar] [CrossRef] [PubMed]
- Mathers, C.D.; Loncar, D. Projections of global mortality and burden of disease from 2002 to 2030. PLoS Med. 2006, 3, e442. [Google Scholar] [CrossRef] [PubMed]
- Murray, C.J.; Vos, T.; Lozano, R.; Naghavi, M.; Flaxman, A.D.; Michaud, C.; Ezzati, M.; Shibuya, K.; Salomon, J.A.; Abdalla, S. Disability-adjusted life years (dalys) for 291 diseases and injuries in 21 regions, 1990–2010: A systematic analysis for the global burden of disease study 2010. Lancet 2013, 380, 2197–2223. [Google Scholar] [CrossRef]
- Slavich, G.M.; Irwin, M.R. From stress to inflammation and major depressive disorder: A social signal transduction theory of depression. Psychol. Bull. 2014, 140, 774. [Google Scholar] [CrossRef] [PubMed]
- Kolber, B.J.; Wieczorek, L.; Muglia, L.J. Hypothalamic–pituitary–adrenal axis dysregulation and behavioral analysis of mouse mutants with altered glucocorticoid or mineralocorticoid receptor function: Review. Stress 2008, 11, 321–338. [Google Scholar] [CrossRef] [PubMed]
- Herman, J.P.; Cullinan, W.E. Neurocircuitry of stress: Central control of the hypothalamo–pituitary–adrenocortical axis. Trends Neurosci. 1997, 20, 78–84. [Google Scholar] [CrossRef]
- Morimoto, M.; Morita, N.; Ozawa, H.; Yokoyama, K.; Kawata, M. Distribution of glucocorticoid receptor immunoreactivity and mRNA in the rat brain: An immunohistochemical and in situ hybridization study. Neurosci. Res. 1996, 26, 235–269. [Google Scholar] [CrossRef]
- Bomholt, S.F.; Harbuz, M.S.; Blackburn-Munro, G.; Blackburn-Munro, R.E. Involvement and role of the hypothalamo-pituitary-adrenal (HPA) stress axis in animal models of chronic pain and inflammation. Stress 2004, 7, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Strange, B.A.; Witter, M.P.; Lein, E.S.; Moser, E.I. Functional organization of the hippocampal longitudinal axis. Nat. Rev. Neurosci. 2014, 15, 655–669. [Google Scholar] [CrossRef] [PubMed]
- Moser, M.-B.; Moser, E.I.; Forrest, E.; Andersen, P.; Morris, R. Spatial learning with a minislab in the dorsal hippocampus. Proc. Natl. Acad. Sci. USA 1995, 92, 9697–9701. [Google Scholar] [CrossRef] [PubMed]
- Bannerman, D.; Deacon, R.; Offen, S.; Friswell, J.; Grubb, M.; Rawlins, J. Double dissociation of function within the hippocampus: Spatial memory and hyponeophagia. Behav. Neurosci. 2002, 116, 884–900. [Google Scholar] [CrossRef] [PubMed]
- Pothuizen, H.H.; Zhang, W.N.; Jongen-Rêlo, A.L.; Feldon, J.; Yee, B.K. Dissociation of function between the dorsal and the ventral hippocampus in spatial learning abilities of the rat: A within-subject, within-task comparison of reference and working spatial memory. Eur. J. Neurosci. 2004, 19, 705–712. [Google Scholar] [CrossRef] [PubMed]
- Fanselow, M.S.; Dong, H.-W. Are the dorsal and ventral hippocampus functionally distinct structures? Neuron 2010, 65, 7–19. [Google Scholar] [CrossRef] [PubMed]
- Bannerman, D.; Grubb, M.; Deacon, R.; Yee, B.; Feldon, J.; Rawlins, J. Ventral hippocampal lesions affect anxiety but not spatial learning. Behav. Brain Res. 2003, 139, 197–213. [Google Scholar] [CrossRef]
- Kjelstrup, K.G.; Tuvnes, F.A.; Steffenach, H.-A.; Murison, R.; Moser, E.I.; Moser, M.-B. Reduced fear expression after lesions of the ventral hippocampus. Proc. Natl. Acad. Sci. USA 2002, 99, 10825–10830. [Google Scholar] [CrossRef] [PubMed]
- Green, H.F.; Nolan, Y.M. Inflammation and the developing brain: Consequences for hippocampal neurogenesis and behavior. Neurosci. Biobehav. Rev. 2014, 40, 20–34. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, P.S.; Perfilieva, E.; Björk-Eriksson, T.; Alborn, A.-M.; Nordborg, C.; Peterson, D.A.; Gage, F.H. Neurogenesis in the adult human hippocampus. Nat. Med. 1998, 4, 1313–1317. [Google Scholar] [CrossRef] [PubMed]
- Gage, F.H. Mammalian neural stem cells. Science 2000, 287, 1433–1438. [Google Scholar] [CrossRef] [PubMed]
- Kempermann, G.; Jessberger, S.; Steiner, B.; Kronenberg, G. Milestones of neuronal development in the adult hippocampus. Trends Neurosci. 2004, 27, 447–452. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Teng, E.M.; Summers, R.G.; Ming, G.-l.; Gage, F.H. Distinct morphological stages of dentate granule neuron maturation in the adult mouse hippocampus. J. Neurosci. 2006, 26, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Santarelli, L.; Saxe, M.; Gross, C.; Surget, A.; Battaglia, F.; Dulawa, S.; Weisstaub, N.; Lee, J.; Duman, R.; Arancio, O. Requirement of hippocampal neurogenesis for the behavioral effects of antidepressants. Science 2003, 301, 805–809. [Google Scholar] [CrossRef] [PubMed]
- Danielson, N.B.; Kaifosh, P.; Zaremba, J.D.; Lovett-Barron, M.; Tsai, J.; Denny, C.A.; Balough, E.M.; Goldberg, A.R.; Drew, L.J.; Hen, R. Distinct contribution of adult-born hippocampal granule cells to context encoding. Neuron 2016, 90, 101–112. [Google Scholar] [CrossRef] [PubMed]
- Kheirbek, M.A.; Drew, L.J.; Burghardt, N.S.; Costantini, D.O.; Tannenholz, L.; Ahmari, S.E.; Zeng, H.; Fenton, A.A.; Hen, R. Differential control of learning and anxiety along the dorsoventral axis of the dentate gyrus. Neuron 2013, 77, 955–968. [Google Scholar] [CrossRef] [PubMed]
- Breunig, J.J.; Silbereis, J.; Vaccarino, F.M.; Šestan, N.; Rakic, P. Notch regulates cell fate and dendrite morphology of newborn neurons in the postnatal dentate gyrus. Proc. Natl. Acad. Sci. USA 2007, 104, 20558–20563. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.H.; Bicker, F.; Nikolic, I.; Meister, J.; Babuke, T.; Picuric, S.; Müller-Esterl, W.; Plate, K.H.; Dikic, I. Epidermal growth factor-like domain 7 (EGFL7) modulates Notch signalling and affects neural stem cell renewal. Nat. Cell Biol. 2009, 11, 873–880. [Google Scholar] [CrossRef] [PubMed]
- Lie, D.-C.; Colamarino, S.A.; Song, H.-J.; Désiré, L.; Mira, H.; Consiglio, A.; Lein, E.S.; Jessberger, S.; Lansford, H.; Dearie, A.R. Wnt signalling regulates adult hippocampal neurogenesis. Nature 2005, 437, 1370–1375. [Google Scholar] [CrossRef] [PubMed]
- Lai, K.; Kaspar, B.K.; Gage, F.H.; Schaffer, D.V. Sonic hedgehog regulates adult neural progenitor proliferation in vitro and in vivo. Nat. Neurosci. 2003, 6, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Apkarian, A.V.; Mutso, A.A.; Centeno, M.V.; Kan, L.; Wu, M.; Levinstein, M.; Banisadr, G.; Gobeske, K.T.; Miller, R.J.; Radulovic, J. Role of adult hippocampal neurogenesis in persistent pain. Pain 2016, 157, 418. [Google Scholar] [CrossRef] [PubMed]
- Stankovic, N.D.; Teodorczyk, M.; Ploen, R.; Zipp, F.; Schmidt, M.H. Microglia–blood vessel interactions: A double-edged sword in brain pathologies. Acta Neuropathol. 2016, 131, 347–363. [Google Scholar] [CrossRef] [PubMed]
- Sierra, A.; Encinas, J.M.; Deudero, J.J.; Chancey, J.H.; Enikolopov, G.; Overstreet-Wadiche, L.S.; Tsirka, S.E.; Maletic-Savatic, M. Microglia shape adult hippocampal neurogenesis through apoptosis-coupled phagocytosis. Cell Stem Cell 2010, 7, 483–495. [Google Scholar] [CrossRef] [PubMed]
- Ryan, S.M.; Nolan, Y.M. Neuroinflammation negatively affects adult hippocampal neurogenesis and cognition: Can exercise compensate? Neurosci. Biobehav. Rev. 2016, 61, 121–131. [Google Scholar] [CrossRef] [PubMed]
- Kostianovsky, A.M.; Maier, L.M.; Anderson, R.C.; Bruce, J.N.; Anderson, D.E. Astrocytic regulation of human monocytic/microglial activation. J. Immunol. 2008, 181, 5425–5432. [Google Scholar] [CrossRef] [PubMed]
- Van Rossum, D.; Hanisch, U.-K. Microglia. Metab. Brain Dis. 2004, 19, 393–411. [Google Scholar] [CrossRef] [PubMed]
- Belarbi, K.; Rosi, S. Modulation of adult-born neurons in the inflamed hippocampus. Front. Cell. Neurosci. 2013, 7, 145. [Google Scholar] [CrossRef] [PubMed]
- Sierra, A.; Beccari, S.; Diaz-Aparicio, I.; Encinas, J.M.; Comeau, S.; Tremblay, M.-È. Surveillance, phagocytosis, and inflammation: How never-resting microglia influence adult hippocampal neurogenesis. Neural Plast. 2014, 2014, 610343. [Google Scholar] [CrossRef] [PubMed]
- Jolivel, V.; Bicker, F.; Binamé, F.; Ploen, R.; Keller, S.; Gollan, R.; Jurek, B.; Birkenstock, J.; Poisa-Beiro, L.; Bruttger, J. Perivascular microglia promote blood vessel disintegration in the ischemic penumbra. Acta Neuropathol. 2015, 129, 279–295. [Google Scholar] [CrossRef] [PubMed]
- Vallières, L.; Campbell, I.L.; Gage, F.H.; Sawchenko, P.E. Reduced hippocampal neurogenesis in adult transgenic mice with chronic astrocytic production of interleukin-6. J. Neurosci. 2002, 22, 486–492. [Google Scholar] [PubMed]
- Monje, M.L.; Toda, H.; Palmer, T.D. Inflammatory blockade restores adult hippocampal neurogenesis. Science 2003, 302, 1760–1765. [Google Scholar] [CrossRef] [PubMed]
- Monje, F.J.; Cabatic, M.; Divisch, I.; Kim, E.-J.; Herkner, K.R.; Binder, B.R.; Pollak, D.D. Constant darkness induces IL-6-dependent depression-like behavior through the NF-κB signaling pathway. J. Neurosci. 2011, 31, 9075–9083. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.; McCloskey, M.A.; Blong, C.C.; Bendickson, L.; Nilsen-Hamilton, M.; Sakaguchi, D.S. Astrocyte-derived interleukin-6 promotes specific neuronal differentiation of neural progenitor cells from adult hippocampus. J. Neurosci. Res. 2010, 88, 2798–2809. [Google Scholar] [CrossRef] [PubMed]
- Goshen, I.; Kreisel, T.; Ben-Menachem-Zidon, O.; Licht, T.; Weidenfeld, J.; Ben-Hur, T.; Yirmiya, R. Brain interleukin-1 mediates chronic stress-induced depression in mice via adrenocortical activation and hippocampal neurogenesis suppression. Mol. Psychiatry 2008, 13, 717–728. [Google Scholar] [CrossRef] [PubMed]
- Ryan, S.M.; O’Keeffe, G.W.; O’Connor, C.; Keeshan, K.; Nolan, Y.M. Negative regulation of TLX by IL-1β correlates with an inhibition of adult hippocampal neural precursor cell proliferation. Brain Behav. Immun. 2013, 33, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, N.; Kudo, K.; Mabuchi, T.; Takemoto, K.; Fujimaki, K.; Wati, H.; Iguchi, H.; Tezuka, H.; Kanba, S. Suppression of cell proliferation by interferon-alpha through interleukin-1 production in adult rat dentate gyrus. Neuropsychopharmacology 2006, 31, 2619. [Google Scholar] [CrossRef] [PubMed]
- Zunszain, P.A.; Anacker, C.; Cattaneo, A.; Choudhury, S.; Musaelyan, K.; Myint, A.M.; Thuret, S.; Price, J.; Pariante, C.M. Interleukin-1β: A new regulator of the kynurenine pathway affecting human hippocampal neurogenesis. Neuropsychopharmacology 2012, 37, 939–949. [Google Scholar] [CrossRef] [PubMed]
- Johansson, S.; Price, J.; Modo, M. Effect of inflammatory cytokines on major histocompatibility complex expression and differentiation of human neural stem/progenitor cells. Stem Cells 2008, 26, 2444–2454. [Google Scholar] [CrossRef] [PubMed]
- Brambilla, R.; Ashbaugh, J.J.; Magliozzi, R.; Dellarole, A.; Karmally, S.; Szymkowski, D.E.; Bethea, J.R. Inhibition of soluble tumour necrosis factor is therapeutic in experimental autoimmune encephalomyelitis and promotes axon preservation and remyelination. Brain 2011, 134, 2736–2754. [Google Scholar] [CrossRef] [PubMed]
- Martuscello, R.T.; Spengler, R.N.; Bonoiu, A.C.; Davidson, B.A.; Helinski, J.; Ding, H.; Mahajan, S.; Kumar, R.; Bergey, E.J.; Knight, P.R. Increasing TNF levels solely in the rat hippocampus produces persistent pain-like symptoms. Pain 2012, 153, 1871–1882. [Google Scholar] [CrossRef] [PubMed]
- Iosif, R.E.; Ekdahl, C.T.; Ahlenius, H.; Pronk, C.J.; Bonde, S.; Kokaia, Z.; Jacobsen, S.-E.W.; Lindvall, O. Tumor necrosis factor receptor 1 is a negative regulator of progenitor proliferation in adult hippocampal neurogenesis. J. Neurosci. 2006, 26, 9703–9712. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Palmer, T.D. Differential roles of tnfr1 and TNFR2 signaling in adult hippocampal neurogenesis. Brain Behav. Immun. 2013, 30, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Battista, D.; Ferrari, C.C.; Gage, F.H.; Pitossi, F.J. Neurogenic niche modulation by activated microglia: Transforming growth factor β increases neurogenesis in the adult dentate gyrus. Eur. J. Neurosci. 2006, 23, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Butovsky, O.; Ziv, Y.; Schwartz, A.; Landa, G.; Talpalar, A.E.; Pluchino, S.; Martino, G.; Schwartz, M. Microglia activated by IL-4 or IFN-γ differentially induce neurogenesis and oligodendrogenesis from adult stem/progenitor cells. Mol. Cell. Neurosci. 2006, 31, 149–160. [Google Scholar] [CrossRef] [PubMed]
- Annenkov, A. The insulin-like growth factor (IGF) receptor type 1 (IGF1R) as an essential component of the signalling network regulating neurogenesis. Mol. Neurobiol. 2009, 40, 195–215. [Google Scholar] [CrossRef] [PubMed]
- Cacci, E.; Ajmone-Cat, M.A.; Anelli, T.; Biagioni, S.; Minghetti, L. In vitro neuronal and glial differentiation from embryonic or adult neural precursor cells are differently affected by chronic or acute activation of microglia. Glia 2008, 56, 412–425. [Google Scholar] [CrossRef] [PubMed]
- Bachstetter, A.D.; Morganti, J.M.; Jernberg, J.; Schlunk, A.; Mitchell, S.H.; Brewster, K.W.; Hudson, C.E.; Cole, M.J.; Harrison, J.K.; Bickford, P.C. Fractalkine and CX3CR1 regulate hippocampal neurogenesis in adult and aged rats. Neurobiol. Aging 2011, 32, 2030–2044. [Google Scholar] [CrossRef] [PubMed]
- Rimmerman, N.; Schottlender, N.; Reshef, R.; Dan-Goor, N.; Yirmiya, R. The hippocampal transcriptomic signature of stress resilience in mice with microglial fractalkine receptor (CX3CR1) deficiency. Brain Behav. Immun. 2016. [Google Scholar] [CrossRef] [PubMed]
- Smith, R. The macrophage theory of depression. Med. Hypotheses 1991, 35, 298–306. [Google Scholar] [CrossRef]
- Kim, Y.K.; Maes, M. The role of the cytokine network in psychological stress. Acta Neuropsychiatr. 2003, 15, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Rao, J.S.; Harry, G.J.; Rapoport, S.I.; Kim, H.-W. Increased excitotoxicity and neuroinflammatory markers in postmortem frontal cortex from bipolar disorder patients. Mol. Psychiatry 2010, 15, 384–392. [Google Scholar] [CrossRef] [PubMed]
- Donzis, E.J.; Tronson, N.C. Modulation of learning and memory by cytokines: Signaling mechanisms and long term consequences. Neurobiol. Learn. Mem. 2014, 115, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Tannenholz, L.; Jimenez, J.C.; Kheirbek, M.A. Local and regional heterogeneity underlying hippocampal modulation of cognition and mood. Front. Behav. Neurosci. 2014, 8, 147. [Google Scholar] [CrossRef] [PubMed]
- Kang, E.; Wen, Z.; Song, H.; Christian, K.M.; Ming, G.-L. Adult neurogenesis and psychiatric disorders. Cold Spring Harbor Perspect. Biol. 2016, 8, a019026. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Arruda-Carvalho, M.; Wang, J.; Janoschka, S.R.; Josselyn, S.A.; Frankland, P.W.; Ge, S. Optical controlling reveals time-dependent roles for adult-born dentate granule cells. Nat. Neurosci. 2012, 15, 1700–1706. [Google Scholar] [CrossRef] [PubMed]
- Denny, C.A.; Kheirbek, M.A.; Alba, E.L.; Tanaka, K.F.; Brachman, R.A.; Laughman, K.B.; Tomm, N.K.; Turi, G.F.; Losonczy, A.; Hen, R. Hippocampal memory traces are differentially modulated by experience, time, and adult neurogenesis. Neuron 2014, 83, 189–201. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, C.; Sanderson, D.J. Malaise in the water maze: Untangling the effects of LPS and IL-1β on learning and memory. Brain Behav. Immun. 2008, 22, 1117–1127. [Google Scholar] [CrossRef] [PubMed]
- Hein, A.M.; Stasko, M.R.; Matousek, S.B.; Scott-McKean, J.J.; Maier, S.F.; Olschowka, J.A.; Costa, A.C.; O’Banion, M.K. Sustained hippocampal IL-1β overexpression impairs contextual and spatial memory in transgenic mice. Brain Behav. Immun. 2010, 24, 243–253. [Google Scholar] [CrossRef] [PubMed]
- Butler, M.; O’connor, J.; Moynagh, P. Dissection of tumor-necrosis factor-α inhibition of long-term potentiation (LTP) reveals a p38 mitogen-activated protein kinase-dependent mechanism which maps to early—But not late—Phase ltp. Neuroscience 2004, 124, 319–326. [Google Scholar] [CrossRef] [PubMed]
- Tancredi, V.; D’Antuono, M.; Cafè, C.; Giovedì, S.; Buè, M.C.; D’Arcangelo, G.; Onofri, F.; Benfenati, F. The inhibitory effects of interleukin-6 on synaptic plasticity in the rat hippocampus are associated with an inhibition of mitogen-activated protein kinase ERK. J. Neurochem. 2000, 75, 634–643. [Google Scholar] [CrossRef] [PubMed]
- Stojanovic, A.; Martorell, L.; Montalvo, I.; Ortega, L.; Monseny, R.; Vilella, E.; Labad, J. Increased serum interleukin-6 levels in early stages of psychosis: Associations with at-risk mental states and the severity of psychotic symptoms. Psychoneuroendocrinology 2014, 41, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Raison, C.L.; Rutherford, R.E.; Woolwine, B.J.; Shuo, C.; Schettler, P.; Drake, D.F.; Haroon, E.; Miller, A.H. A randomized controlled trial of the tumor necrosis factor antagonist infliximab for treatment-resistant depression: The role of baseline inflammatory biomarkers. JAMA Psychiatry 2013, 70, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Schoenfeld, T.J.; Cameron, H.A. Adult neurogenesis and mental illness. Neuropsychopharmacology 2015, 40, 113–128. [Google Scholar] [CrossRef] [PubMed]
- Yun, S.; Reynolds, R.P.; Masiulis, I.; Eisch, A.J. Re-evaluating the link between neuropsychiatric disorders and dysregulated adult neurogenesis. Nat. Med. 2016, 22, 1239–1247. [Google Scholar] [CrossRef] [PubMed]
- Jensen, T.S.; Baron, R.; Haanpää, M.; Kalso, E.; Loeser, J.D.; Rice, A.S.; Treede, R.-D. A new definition of neuropathic pain. Pain 2011, 152, 2204–2205. [Google Scholar] [CrossRef] [PubMed]
- Mutso, A.A.; Radzicki, D.; Baliki, M.N.; Huang, L.; Banisadr, G.; Centeno, M.V.; Radulovic, J.; Martina, M.; Miller, R.J.; Apkarian, A.V. Abnormalities in hippocampal functioning with persistent pain. J. Neurosci. 2012, 32, 5747–5756. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, M.E.; Pan, J.W.; Hetherington, H.P.; Lipton, M.L.; Baigi, K.; Lipton, R.B. Hippocampal correlates of pain in healthy elderly adults a pilot study. Neurology 2009, 73, 1567–1570. [Google Scholar] [CrossRef] [PubMed]
- Tse, Y.C.; Montoya, I.; Wong, A.S.; Mathieu, A.; Lissemore, J.; Lagace, D.C.; Wong, T.P. A longitudinal study of stress-induced hippocampal volume changes in mice that are susceptible or resilient to chronic social defeat. Hippocampus 2014, 24, 1120–1128. [Google Scholar] [CrossRef] [PubMed]
- Duric, V.; McCarson, K.E. Persistent pain produces stress-like alterations in hippocampal neurogenesis and gene expression. J. Pain 2006, 7, 544–555. [Google Scholar] [CrossRef] [PubMed]
- Lempel, A.A.; Coll, L.; Schinder, A.F.; Uchitel, O.D.; Piriz, J. Chronic pregabalin treatment decreases excitability of dentate gyrus and accelerates maturation of adult-born granule cells. J. Neurochem. 2016, 140, 257–267. [Google Scholar] [CrossRef] [PubMed]
- Altmann, C.; Vasic, V.; Hardt, S.; Heidler, J.; Häussler, A.; Wittig, I.; Schmidt, M.H.; Tegeder, I. Progranulin promotes peripheral nerve regeneration and reinnervation: Role of Notch signaling. Mol. Neurodegener. 2016, 11, 69. [Google Scholar] [CrossRef] [PubMed]
- Leggio, M.G.; Mandolesi, L.; Federico, F.; Spirito, F.; Ricci, B.; Gelfo, F.; Petrosini, L. Environmental enrichment promotes improved spatial abilities and enhanced dendritic growth in the rat. Behav. Brain Res. 2005, 163, 78–90. [Google Scholar] [CrossRef] [PubMed]
- Terada, M.; Kuzumaki, N.; Hareyama, N.; Imai, S.; Niikura, K.; Narita, M.; Yamazaki, M.; Suzuki, T.; Narita, M. Suppression of enriched environment-induced neurogenesis in a rodent model of neuropathic pain. Neurosci. Lett. 2008, 440, 314–318. [Google Scholar] [CrossRef] [PubMed]
- Dellarole, A.; Morton, P.; Brambilla, R.; Walters, W.; Summers, S.; Bernardes, D.; Grilli, M.; Bethea, J.R. Neuropathic pain-induced depressive-like behavior and hippocampal neurogenesis and plasticity are dependent on TNFR1 signaling. Brain Behav. Immun. 2014, 41, 65–81. [Google Scholar] [CrossRef] [PubMed]
- Spalding, K.L.; Bergmann, O.; Alkass, K.; Bernard, S.; Salehpour, M.; Huttner, H.B.; Boström, E.; Westerlund, I.; Vial, C.; Buchholz, B.A. Dynamics of hippocampal neurogenesis in adult humans. Cell 2013, 153, 1219–1227. [Google Scholar] [CrossRef] [PubMed]
- Vachon-Presseau, E.; Roy, M.; Martel, M.-O.; Caron, E.; Marin, M.-F.; Chen, J.; Albouy, G.; Plante, I.; Sullivan, M.J.; Lupien, S.J. The stress model of chronic pain: Evidence from basal cortisol and hippocampal structure and function in humans. Brain 2013, 136, 815–827. [Google Scholar] [CrossRef] [PubMed]
- Dimitrov, E.L.; Tsuda, M.C.; Cameron, H.A.; Usdin, T.B. Anxiety-and depression-like behavior and impaired neurogenesis evoked by peripheral neuropathy persist following resolution of prolonged tactile hypersensitivity. J. Neurosci. 2014, 34, 12304–12312. [Google Scholar] [CrossRef] [PubMed]
- Zautra, A.J.; Johnson, L.M.; Davis, M.C. Positive affect as a source of resilience for women in chronic pain. J. Consult. Clin. Psychol. 2005, 73, 212. [Google Scholar] [CrossRef] [PubMed]
- Stone, A.A.; Neale, J.M.; Cox, D.S.; Napoli, A.; Valdimarsdottir, H.; Kennedy-Moore, E. Daily events are associated with a secretory immune response to an oral antigen in men. Health Psychol. 1994, 13, 440. [Google Scholar] [CrossRef] [PubMed]
- Snow-Turek, A.L.; Norris, M.P.; Tan, G. Active and passive coping strategies in chronic pain patients. Pain 1996, 64, 455–462. [Google Scholar] [CrossRef]
- Strahl, C.; Kleinknecht, R.A.; Dinnel, D.L. The role of pain anxiety, coping, and pain self-efficacy in rheumatoid arthritis patient functioning. Behav. Res. Ther. 2000, 38, 863–873. [Google Scholar] [CrossRef]
- Esteve, R.; Ramírez-Maestre, C.; López-Martínez, A.E. Adjustment to chronic pain: The role of pain acceptance, coping strategies, and pain-related cognitions. Ann. Behav. Med. 2007, 33, 179–188. [Google Scholar] [CrossRef] [PubMed]
- Lawlor, P.G. The panorama of opioid-related cognitive dysfunction in patients with cancer. Cancer 2002, 94, 1836–1853. [Google Scholar] [CrossRef] [PubMed]
- Kodama, D.; Ono, H.; Tanabe, M. Altered hippocampal long-term potentiation after peripheral nerve injury in mice. Eur. J. Pharmacol. 2007, 574, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Ren, W.-J.; Liu, Y.; Zhou, L.-J.; Li, W.; Zhong, Y.; Pang, R.-P.; Xin, W.-J.; Wei, X.-H.; Wang, J.; Zhu, H.-Q. Peripheral nerve injury leads to working memory deficits and dysfunction of the hippocampus by upregulation of TNF-α in rodents. Neuropsychopharmacology 2011, 36, 979–992. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.-D.; Barde, S.; Yang, T.; Lei, B.; Eriksson, L.I.; Mathew, J.P.; Andreska, T.; Akassoglou, K.; Harkany, T.; Hökfelt, T.G. Orthopedic surgery modulates neuropeptides and bdnf expression at the spinal and hippocampal levels. Proc. Natl. Acad. Sci. USA 2016, 113, E6686–E6695. [Google Scholar] [CrossRef] [PubMed]
- Duman, R.S.; Monteggia, L.M. A neurotrophic model for stress-related mood disorders. Biol. Psychiatry 2006, 59, 1116–1127. [Google Scholar] [CrossRef] [PubMed]
- Jensen, M.P.; Chodroff, M.J.; Dworkin, R.H. The impact of neuropathic pain on health-related quality of life review and implications. Neurology 2007, 68, 1178–1182. [Google Scholar] [CrossRef] [PubMed]
- Bair, M.J.; Robinson, R.L.; Katon, W.; Kroenke, K. Depression and pain comorbidity: A literature review. Arch. Intern. Med. 2003, 163, 2433–2445. [Google Scholar] [CrossRef] [PubMed]
- Van Praag, H.; Kempermann, G.; Gage, F.H. Running increases cell proliferation and neurogenesis in the adult mouse dentate gyrus. Nat. Neurosci. 1999, 2, 266–270. [Google Scholar] [PubMed]
- Voss, M.W.; Vivar, C.; Kramer, A.F.; van Praag, H. Bridging animal and human models of exercise-induced brain plasticity. Trends Cogn. Sci. 2013, 17, 525–544. [Google Scholar] [CrossRef] [PubMed]
- Clark, P.; Brzezinska, W.; Thomas, M.; Ryzhenko, N.; Toshkov, S.; Rhodes, J. Intact neurogenesis is required for benefits of exercise on spatial memory but not motor performance or contextual fear conditioning in c57bl/6j mice. Neuroscience 2008, 155, 1048–1058. [Google Scholar] [CrossRef] [PubMed]
- Fishbain, D. Evidence-based data on pain relief with antidepressants. Ann. Med. 2000, 32, 305–316. [Google Scholar] [CrossRef] [PubMed]
- Micó, J.A.; Ardid, D.; Berrocoso, E.; Eschalier, A. Antidepressants and pain. Trends Pharmacol. Sci. 2006, 27, 348–354. [Google Scholar] [CrossRef] [PubMed]
- Russo, S.J.; Murrough, J.W.; Han, M.-H.; Charney, D.S.; Nestler, E.J. Neurobiology of resilience. Nat. Neurosci. 2012, 15, 1475–1484. [Google Scholar] [CrossRef] [PubMed]
| Pain | Neurogenesis | Effect on Neurogenesis | Publication |
|---|---|---|---|
| Neuropathic pain | ↓ | Suppressed neurogenesis | [91] |
| Persistent pain; spared nerve injury | ↓ | Reduced BrdU/DCX cells | [80] |
| Persistent pain; spared nerve injury | ↑ | Neurogenesis mouse models | [35] |
| Persistent pain | ↓ | Reduced BrdU cells | [83] |
| Neuropathic pain | ↓ | Reduced NeuroD cells | [87] |
| Neuropathic pain | ↓ | Reduced BrdU/NeuN cells | [88] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vasic, V.; Schmidt, M.H.H. Resilience and Vulnerability to Pain and Inflammation in the Hippocampus. Int. J. Mol. Sci. 2017, 18, 739. https://doi.org/10.3390/ijms18040739
Vasic V, Schmidt MHH. Resilience and Vulnerability to Pain and Inflammation in the Hippocampus. International Journal of Molecular Sciences. 2017; 18(4):739. https://doi.org/10.3390/ijms18040739
Chicago/Turabian StyleVasic, Verica, and Mirko H. H. Schmidt. 2017. "Resilience and Vulnerability to Pain and Inflammation in the Hippocampus" International Journal of Molecular Sciences 18, no. 4: 739. https://doi.org/10.3390/ijms18040739
APA StyleVasic, V., & Schmidt, M. H. H. (2017). Resilience and Vulnerability to Pain and Inflammation in the Hippocampus. International Journal of Molecular Sciences, 18(4), 739. https://doi.org/10.3390/ijms18040739







