Effects of Melatonin on Liver Injuries and Diseases
Abstract
:1. Introduction
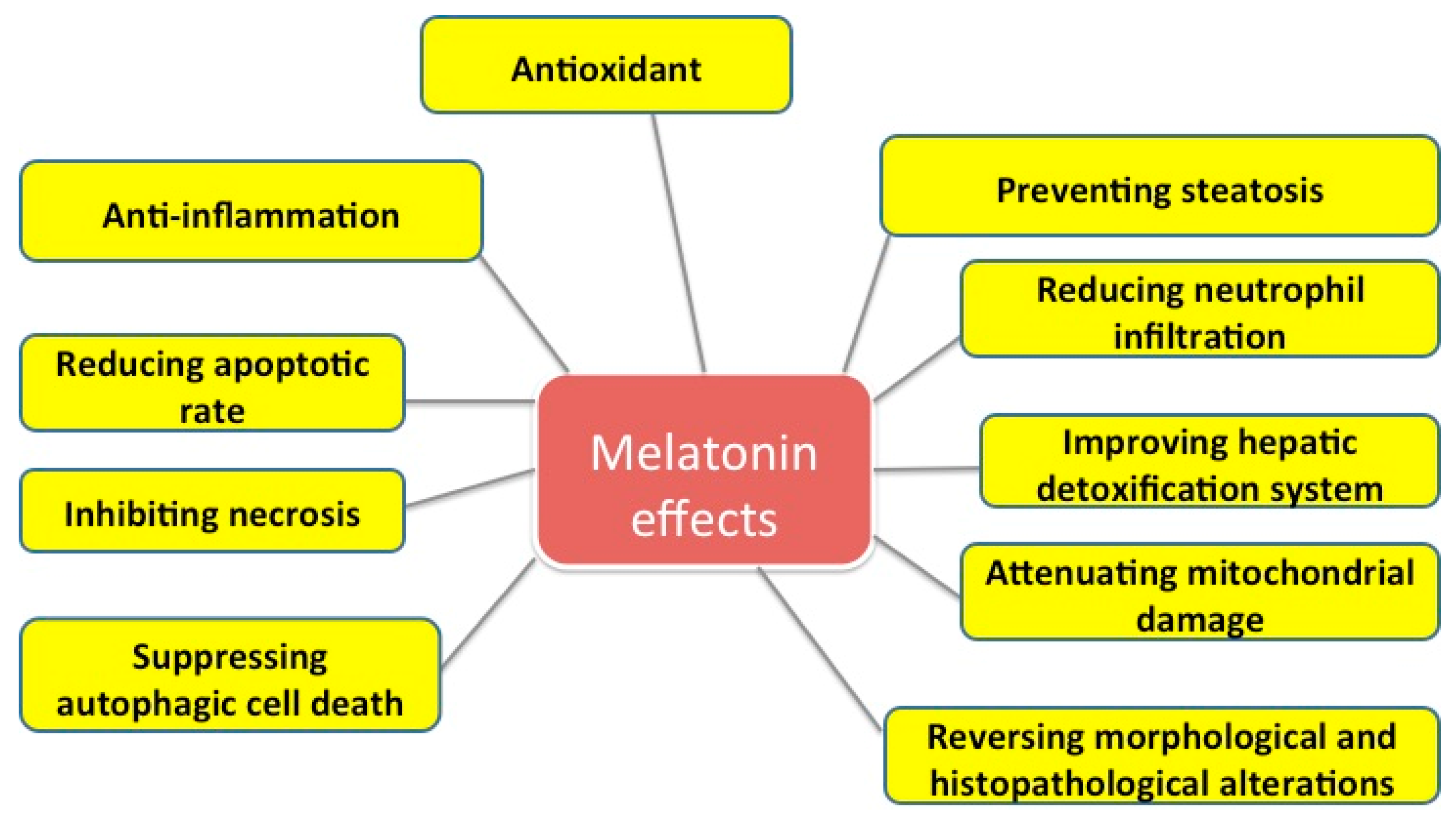
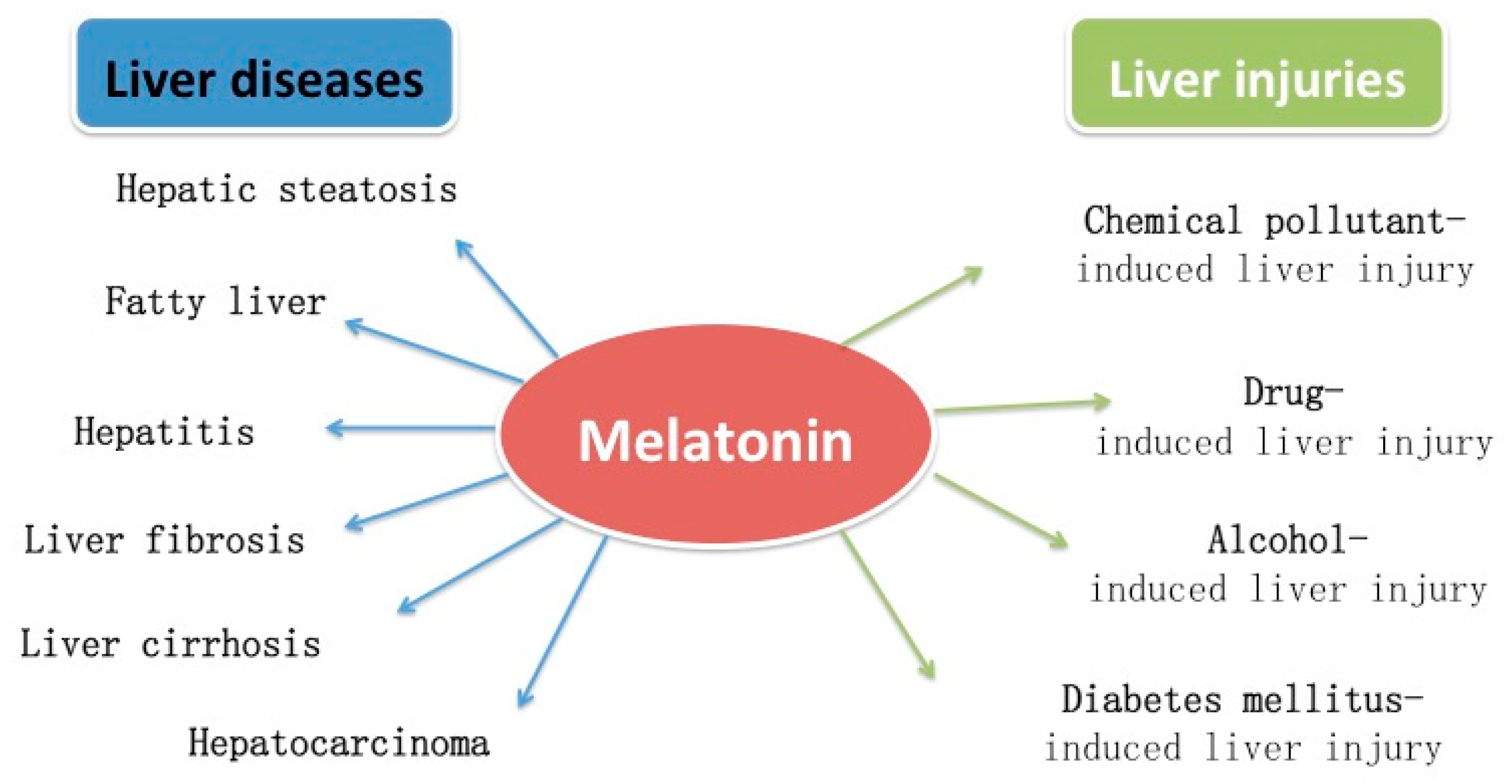
2. Protective Effects of Melatonin on Liver Injuries
2.1. Protective Effects of Melatonin on Chemical Pollutant-Induced Liver Injuries
2.2. Protective Effects of Melatonin on Drug-Induced Liver Injuries
2.3. Protective Effects of Melatonin on Alcohol-Induced Liver Injury
2.4. Protective Effects of Melatonin on Other Factor-Induced Liver Injuries
3. Protective Effects of Melatonin on Hepatic Steatosis
4. Protective Effects of Melatonin on Non-Alcoholic Fatty Liver
5. Protective Effects of Melatonin on Hepatitis
6. Protective Effects of Melatonin on Liver Fibrosis
7. Protective Effects of Melatonin on Liver Cirrhosis
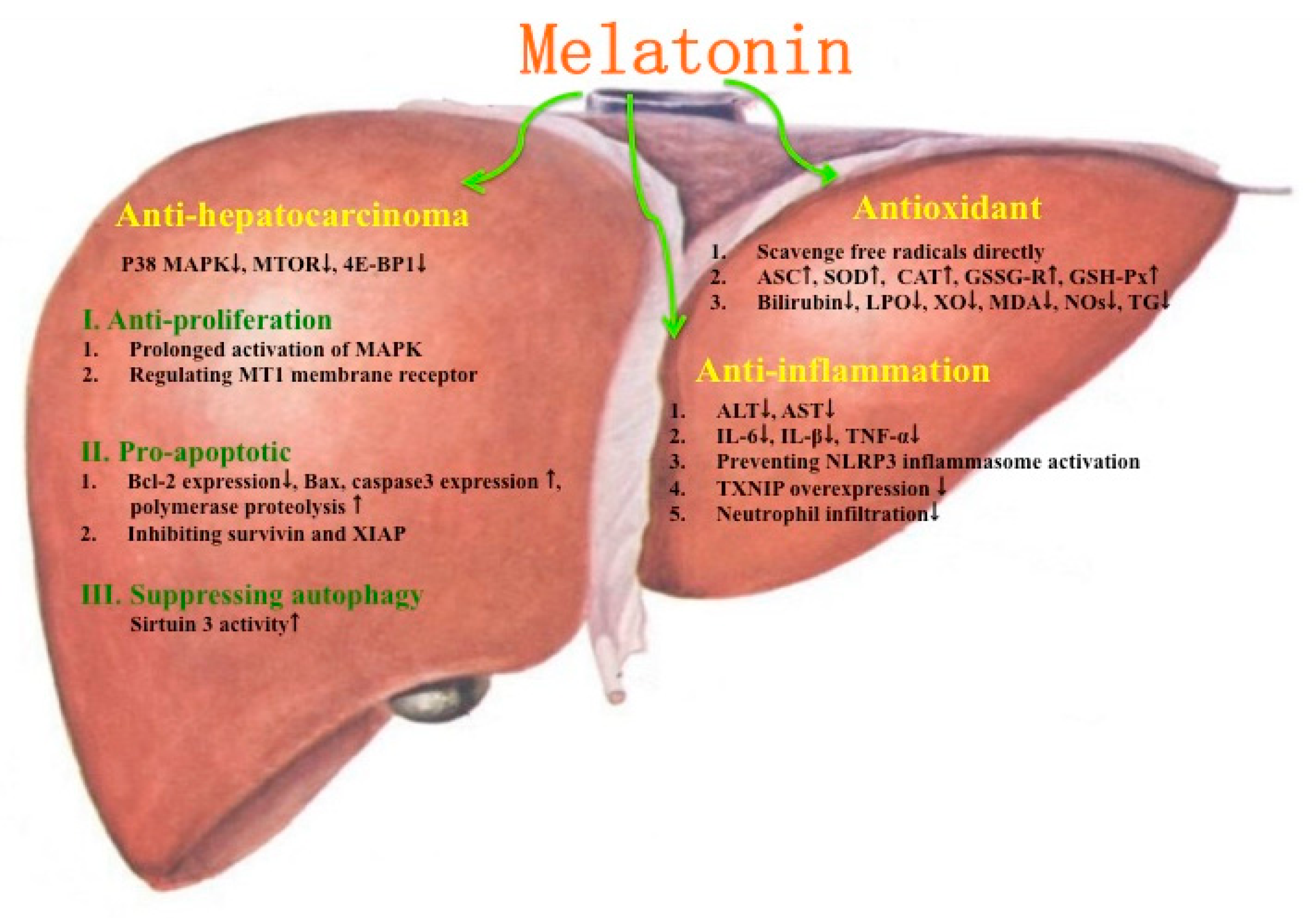
8. Protective Effects of Melatonin on Hepatocarcinoma
9. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Guerra, S.; Mamede, A.C.; Carvalho, M.J.; Laranjo, M.; Tralhao, J.G.; Abrantes, A.M.; Maia, C.J.; Botelho, M.F. Liver diseases: What is known so far about the therapy with human amniotic membrane? Cell Tissue Bank. 2016, 17, 653–663. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Manchester, L.C.; Esteban-Zubero, E.; Zhou, Z.; Reiter, R.J. Melatonin as a potent and inducible endogenous antioxidant: Synthesis and metabolism. Molecules 2015, 20, 18886–18906. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, P.; Mirunalini, S.; Dakshayani, K.B.; Pandi-Perumal, S.R.; Trakht, I.; Cardinali, D.P. Prevention by melatonin of hepatocarcinogenesis in rats injected with N-nitrosodiethylamine. J. Pineal Res. 2007, 43, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Ren, W.K.; Liu, G.; Chen, S.; Yin, J.; Wang, J.; Tan, B.; Wu, G.Y.; Bazer, F.W.; Peng, Y.Y.; Li, T.; et al. Melatonin signaling in T cells: Functions and applications. J. Pineal Res. 2017. [Google Scholar] [CrossRef] [PubMed]
- Kennaway, D.J. Are the proposed benefits of melatonin-rich foods too hard to swallow? Crit. Rev. Food Sci. Nutr. 2017, 57, 958–962. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.R.; Lee, C.; Crowley, S.J.; Fogg, L.F.; Eastman, C.I. Morning melatonin has limited benefit as a soporific for daytime sleep after night work. Chronobiol. Int. 2005, 22, 873–888. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Huang, F.F.; Qu, S. Melatonin: A potential intervention for hepatic steatosis. Lipids Health Dis. 2015, 14, 75. [Google Scholar] [CrossRef] [PubMed]
- Manchester, L.C.; Coto-Montes, A.; Boga, J.A.; Andersen, L.P.H.; Zhou, Z.; Galano, A.; Vriend, J.; Tan, D.X.; Reiter, R.J. Melatonin: An ancient molecule that makes oxygen metabolically tolerable. J. Pineal Res. 2015, 59, 403–419. [Google Scholar] [CrossRef] [PubMed]
- Bali, I.; Bilir, B.; Emir, S.; Turan, F.; Yilmaz, A.; Gokkus, T.; Aydin, M. The effects of melatonin on liver functions in arsenic-induced liver damage. Turk. J. Surg. 2016, 32, 233–237. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Mayo, J.C.; Tan, D.X.; Sainz, R.M.; Alatorre-Jimenez, M.; Qin, L.L. Melatonin as an antioxidant: Under promises but over delivers. J. Pineal Res. 2016, 61, 253–278. [Google Scholar] [CrossRef] [PubMed]
- Ohta, Y.; Kongo, M.; Sasaki, E.; Nishida, K.; Ishiguro, I. Therapeutic effect of melatonin on carbon tetrachloride-induced acute liver injury in rats. J. Pineal Res. 2000, 28, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Noyan, T.; Komuroglu, U.; Bayram, I.; Sekeroglu, M.R. Comparison of the effects of melatonin and pentoxifylline on carbon tetrachloride-induced liver toxicity in mice. Cell Biol. Toxicol. 2006, 22, 381–391. [Google Scholar] [CrossRef] [PubMed]
- Ohta, Y.; Kongo-Nishimura, M.; Matsura, T.; Yamada, K.; Kitagawa, A.; Kishikawa, T. Melatonin prevents disruption of hepatic reactive oxygen species metabolism in rats treated with carbon tetrachloride. J. Pineal Res. 2004, 36, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Kus, I.; Ogeturk, M.; Oner, H.; Sahin, S.; Yekeler, H.; Sarsilmaz, M. Protective effects of melatonin against carbon tetrachloride-induced hepatotoxicity in rats: A light microscopic and biochemical study. Cell Biochem. Funct. 2005, 23, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Zavodnik, L.B.; Zavodnik, I.B.; Lapshina, E.A.; Belonovskaya, E.B.; Martinchik, D.I.; Kravchuk, R.I.; Bryszewska, M.; Reiter, R.J. Protective effects of melatonin against carbon tetrachloride hepatotoxicity in rats. Cell Biochem. Funct. 2005, 23, 353–359. [Google Scholar] [CrossRef] [PubMed]
- Oner, J.; Kus, I.; Oner, H. Melatonin increases the expression of insulin-like growth factor I in rats with carbon tetrachlorid-induced hepatic damage. J. Anim. Vet. Adv. 2009, 8, 2256–2261. [Google Scholar]
- Cheshchevik, V.T.; Lapshina, E.A.; Dremza, I.K.; Zabrodskaya, S.V.; Reiter, R.J.; Prokopchik, N.I.; Zavodnik, I.B. Rat liver mitochondrial damage under acute or chronic carbon tetrachloride-induced intoxication: Protection by melatonin and cranberry flavonoids. Toxicol. Appl. Pharmacol. 2012, 261, 271–279. [Google Scholar] [CrossRef] [PubMed]
- Maksimchik, Y.Z.; Dremza, I.K.; Lapshina, E.A.; Cheshchevik, V.T.; Sudnikovich, E.Y.; Zabrodskaya, S.V.; Zavodnik, I.B. Rat liver mitochondria impairment under acute carbon tetrachloride-induced intoxication. Effects of melatonin. Biol. Membr. 2010, 27, 262–271. [Google Scholar] [CrossRef]
- Ebaid, H.; Bashandy, S.A.E.; Alhazza, I.M.; Rady, A.; El-Shehry, S. Folic acid and melatonin ameliorate carbon tetrachloride-induced hepatic injury, oxidative stress and inflammation in rats. Nutr. Metab. 2013, 10, 20. [Google Scholar] [CrossRef] [PubMed]
- Aranda, M.; Albendea, C.D.; Lostale, F.; Lopez-Pingarron, L.; Fuentes-Broto, L.; Martinez-Ballarin, E.; Reiter, R.J.; Perez-Castejon, M.C.; Garcia, J.J. In vivo hepatic oxidative stress because of carbon tetrachloride toxicity: Protection by melatonin and pinoline. J. Pineal Res. 2010, 49, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Ogeturk, M.; Kus, I.; Pekmez, H.; Yekeler, H.; Sahin, S.; Sarsilmaz, M. Inhibition of carbon tetrachloride-mediated apoptosis and oxidative stress by melatonin in experimental liver fibrosis. Toxicol. Ind. Health 2008, 24, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Rana, S.V.S. Melatonin improves liver function in benzene-treated rats. Arh. Hig. Rada Toksikol. 2013, 64, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Rana, S.V.S. Melatonin inhibits benzene-induced lipid peroxidation in rat liver. Arh. Hig. Rada Toksikol. 2010, 61, 11–18. [Google Scholar] [CrossRef]
- Tas, U.; Ogeturk, M.; Meydan, S.; Kus, I.; Kuloglu, T.; Ilhan, N.; Kose, E.; Sarsilmaz, M. Hepatotoxic activity of toluene inhalation and protective role of melatonin. Toxicol. Ind. Health 2011, 27, 465–473. [Google Scholar] [CrossRef] [PubMed]
- Pi, H.F.; Xu, S.C.; Reiter, R.J.; Guo, P.; Zhang, L.; Li, Y.M.; Li, M.; Cao, Z.W.; Tian, L.; Xie, J.; et al. SIRT3-SOD2-mROS-dependent autophagy in cadmium-induced hepatotoxicity and salvage by melatonin. Autophagy 2015, 11, 1037–1051. [Google Scholar] [CrossRef] [PubMed]
- Kara, H.; Cevik, A.; Konar, V.; Dayangac, A.; Servi, K. Effects of selenium with vitamin E and melatonin on cadmium-induced oxidative damage in rat liver and kidneys. Biol. Trace Elem. Res. 2008, 125, 236–244. [Google Scholar] [CrossRef] [PubMed]
- El-Sokkary, G.H.; Nafady, A.A.; Shabash, E.H. Melatonin administration ameliorates cadmium-induced oxidative stress and morphological changes in the liver of rat. Ecotox. Environ. Saf. 2010, 73, 456–463. [Google Scholar] [CrossRef] [PubMed]
- Guo, P.; Pi, H.F.; Xu, S.C.; Zhang, L.; Li, Y.M.; Li, M.; Cao, Z.W.; Tian, L.; Xie, J.; Li, R.Y.; et al. Melatonin improves mitochondrial function by promoting MT1/SIRT1/PGC-1 alpha-dependent mitochondrial biogenesis in cadmium-induced hepatotoxicity in vitro. Toxicol. Sci. 2014, 142, 182–195. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.W.; Fang, Y.L.; Lu, Y.H.; Tan, D.X.; Du, C.H.; Li, Y.M.; Ma, Q.L.; Yu, J.M.; Chen, M.Y.; Zhou, C.; et al. Melatonin alleviates cadmium-induced liver injury by inhibiting the TXNIP-NLRP3 inflammasome. J. Pineal Res. 2017. [Google Scholar] [CrossRef] [PubMed]
- Romero, A.; Ramos, E.; de Los Rios, C.; Egea, J.; del Pino, J.; Reiter, R.J. A review of metal-catalyzed molecular damage: Protection by melatonin. J. Pineal Res. 2014, 56, 343–370. [Google Scholar] [CrossRef] [PubMed]
- El-Sokkary, G.H.; bdel-Rahman, G.H.; Kamel, E.S. Melatonin protects against lead-induced hepatic and renal toxicity in male rats. Toxicology 2005, 213, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Hedayati, M.T.; Omran, S.M.; Soleymani, A.; Taghizadeh, M. Aflatoxins in food products in Iran: A review of the literature. Jundishapur J. Microbiol. 2016, 9, e33235. [Google Scholar] [CrossRef]
- Akinrinmade, F.J.; Akinrinde, A.S.; Amid, A. Changes in serum cytokine levels, hepatic and intestinal morphology in aflatoxin B1-induced injury: Modulatory roles of melatonin and flavonoid-rich fractions from Chromolena odorata. Mycotoxin Res. 2016, 32, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Meki, A.R.M.A.; Abdel-Ghaffar, S.K.; El-Gibaly, I. Aflatoxin B1 induces apoptosis in rat liver: Protective effect of melatonin. Neuroendocrinol. Lett. 2001, 22, 417–426. [Google Scholar] [PubMed]
- Meki, A.R.M.A.; Esmail, E.E.D.F.; Hussein, A.A.; Hassanein, H.M. Caspase-3 and heat shock protein-70 in rat liver treated with aflatoxin B1: Effect of melatonin. Toxicon 2004, 43, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Malir, F.; Ostry, V.; Pfohl-Leszkowicz, A.; Malir, J.; Toman, J. Ochratoxin A: 50 years of research. Toxins 2016, 8, 191. [Google Scholar] [CrossRef] [PubMed]
- Meki, A.R.M.A.; Hussein, A.A.A. Melatonin reduces oxidative stress induced by ochratoxin A in rat liver and kidney. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2001, 130, 305–313. [Google Scholar] [CrossRef]
- Sutken, E.; Aral, E.; Ozdemir, F.; Uslu, S.; Alatas, O.; Colak, O. Protective role of melatonin and coenzyme Q(10) in ochratoxin a toxicity in rat liver and kidney. Int. J. Toxicol. 2007, 26, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Aydin, G.; Ozcelik, N.; Cicek, E.; Soyoz, M. Histopathologic changes in liver and renal tissues induced by ochratoxin A and melatonin in rats. Hum. Exp. Toxicol. 2003, 22, 383–391. [Google Scholar] [PubMed]
- Cullen, J.M.; Faiola, B.; Melich, D.H.; Peterson, R.A.; Jordan, H.L.; Kimbrough, C.L.; Miller, R.T. Acute α-naphthylisothiocyanate-induced liver toxicity in germfree and conventional male rats. Toxicol. Pathol. 2016, 44, 987–997. [Google Scholar] [CrossRef] [PubMed]
- Ohta, Y.; Kongo, M.; Sasaki, E.; Ishiguro, I.; Harada, N. Protective effect of melatonin against α-naphthylisothiocyanate-induced liver injury in rats. J. Pineal Res. 2000, 29, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Ohta, Y.; Kongo, M.; Kishikawa, T. Preventive effect of melatonin on the progression of α-naphthylisothiocyanate-induced acute liver injury in rats. J. Pineal Res. 2003, 34, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Ohta, Y.; Kongo, M.; Kishikawa, T. Effect of melatonin on changes in hepatic antioxidant enzyme activities in rats treated with α-naphthylisothiocyanate. J. Pineal Res. 2001, 31, 370–377. [Google Scholar] [CrossRef] [PubMed]
- Calvo, J.R.; Reiter, R.J.; Garcia, J.J.; Ortiz, G.G.; Tan, D.X.; Karbownik, M. Characterization of the protective effects of melatonin and related indoles against α-naphthylisothiocyanate-induced liver injury in rats. J. Cell Biochem. 2001, 80, 461–470. [Google Scholar] [CrossRef]
- Ohta, Y.; Kongo-Nishimura, M.; Imai, Y.; Matsura, T.; Kitagawa, A.; Yamada, K. Alpha-tocopherol protects against α-naphthylisothiocyanate-induced hepatotoxicity in rats less effectively than melatonin. Chem. Biol. Interact. 2006, 161, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Kurcer, Z.; Oguz, E.; Iraz, M.; Fadillioglu, E.; Baba, F.; Koksal, M.; Olmez, E. Melatonin improves methanol intoxication-induced oxidative liver injury in rats. J. Pineal Res. 2007, 43, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Chawla, S.L.; Yadav, R.; Shah, D.; Rao, M.V. Protective action of melatonin against fluoride-induced hepatotoxicity in adult female mice. Fluoride 2008, 41, 44–51. [Google Scholar]
- Abdel-Wahab, W.M. AlCl3-induced toxicity and oxidative stress in liver of male rats: Protection by melatonin. Life Sci. J. 2012, 9, 1173–1182. [Google Scholar]
- Jung, K.H.; Hong, S.W.; Zheng, H.M.; Lee, D.H.; Hong, S.S. Melatonin downregulates nuclear erythroid 2-related factor 2 and nuclear factor-κB during prevention of oxidative liver injury in a dimethylnitrosamine model. J. Pineal Res. 2009, 47, 173–183. [Google Scholar] [CrossRef] [PubMed]
- Bruck, R.; Aeed, H.; Avni, Y.; Shirin, H.; Matas, Z.; Shahmurov, M.; Avinoach, I.; Zozulya, G.; Weizman, N.; Hochman, A. Melatonin inhibits nuclear factor kappa B activation and oxidative stress and protects against thioacetamide induced liver damage in rats. J. Hepatol. 2004, 40, 86–93. [Google Scholar] [CrossRef]
- El-Sokkary, G.H.; Cuzzocrea, S.; Reiter, R.J. Effect of chronic nicotine administration on the rat lung and liver: Beneficial role of melatonin. Toxicology 2007, 239, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Rubio, L.; Matas, P.; Miguez, M.P. Protective effect of melatonin on paraquat-induced cytotoxicity in isolated rat hepatocytes. Hum. Exp. Toxicol. 2005, 24, 475–480. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.; Lee, Y.J.; Park, K.K. The pathogenesis of drug-induced liver injury. Expert Rev. Gastroenterol. Hepatol. 2016, 10, 1175–1185. [Google Scholar] [CrossRef] [PubMed]
- Taskin, E.; Guven, C.; Sahin, L.; Dursun, N. The cooperative effect of local angiotensin-II in liver with adriamycin hepatotoxicity on mitochondria. Med. Sci. Monit. 2016, 22, 1013–1021. [Google Scholar] [CrossRef] [PubMed]
- Rapozzi, V.; Comelli, M.; Mavelli, I.; Sentjurc, M.; Schara, M.; Perissin, L.; Giraldi, T. Melatonin and oxidative damage in mice liver induced by the prooxidant antitumor drug, adriamycin. In Vivo 1999, 13, 45–50. [Google Scholar] [PubMed]
- Karakilcik, A.Z.; Bitiren, M.; Zerin, M.; Celik, H.; Aksoy, N. Melatonin increased vitamin C and antioxidant enzyme values in the plasma, heart, liver, and kidney of adriamycin-treated rats. Turk. J. Biol. 2015, 39, 925–931. [Google Scholar] [CrossRef]
- Lee, I.C.; Kim, S.H.; Baek, H.S.; Moon, C.; Bae, C.S.; Kim, S.H.; Yun, W.K.; Nam, K.H.; Kim, H.C.; Kim, J.C. Melatonin improves adriamycin-induced hepatic oxidative damage in rats. Mol. Cell. Toxicol. 2013, 9, 257–265. [Google Scholar] [CrossRef]
- Jahovic, N.; Cevik, H.; Sehirli, A.O.; Yegen, B.C.; Sener, G. Melatonin prevents methotrexate-induced hepatorenal oxidative injury in rats. J. Pineal Res. 2003, 34, 282–287. [Google Scholar] [CrossRef] [PubMed]
- Shokrzadeh, M.; Ahmadi, A.; Naghshvar, F.; Chabra, A.; Jafarinejhad, A. Prophylactic efficacy of melatonin on cyclophosphamide-induced liver toxicity in mice. Biomed Res. Int. 2014, 2014, 470425. [Google Scholar] [CrossRef] [PubMed]
- Aydin, M.; Oktar, S.; Ozkan, O.V.; Alcin, E.; Ozturk, O.H.; Nacar, A. Letrozole induces hepatotoxicity without causing oxidative stress: The protective effect of melatonin. Gynecol. Endocrinol. 2011, 27, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Rezzani, R.; Buffoli, B.; Rodella, L.; Stacchiotti, A.; Bianchi, R. Protective role of melatonin in cyclosporine A-induced oxidative stress in rat liver. Int. Immunopharmacol. 2005, 5, 1397–1405. [Google Scholar] [CrossRef] [PubMed]
- Kurus, M.; Esrefoglu, M.; Sogutlu, G.; Atasever, A. Melatonin prevents cyclosporine-induced hepatotoxicity in rats. Med. Princ. Pract. 2009, 18, 407–410. [Google Scholar] [CrossRef] [PubMed]
- Karabulut, A.B.; Ara, C. Melatonin ameliorates tacrolimus (FK-506)’s induced immunosupressive effect in rat liver. Transplant. Proc. 2009, 41, 1875–1877. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.H.; Chi, N.F.; Kuan, Y.C.; Chan, L.; Hu, C.J.; Chiou, H.Y.; Chien, L.N. Efficacy of phenytoin, valproic acid, carbamazepine and new antiepileptic drugs on control of late-onset post-stroke epilepsy in Taiwan. Eur. J. Neurol. 2015, 22, 1459–1468. [Google Scholar] [CrossRef] [PubMed]
- Eghbal, M.A.; Taziki, S.; Sattari, M.R. Protective role of melatonin and taurine against carbamazepine-induced toxicity in freshly isolated rat hepatocytes. Int. J. Morphol. 2013, 31, 1081–1089. [Google Scholar] [CrossRef]
- Ramantani, G. Of phenobarbital for neonatal seizures: Response rate and refractoriness predictors. Neuropediatrics 2016, 47, 281–282. [Google Scholar] [CrossRef] [PubMed]
- Eghbal, M.A.; Taziki, S.; Sattari, M.R. Mechanisms of phenytoin-induced toxicity in freshly isolated rat hepatocytes and the protective effects of taurine and/or melatonin. J. Biochem. Mol. Toxicol. 2014, 28, 111–118. [Google Scholar] [CrossRef]
- El-Sokkary, G.H. An autoradiographic study of cellular proliferaton, DNA synthesis and cell cycle variability in the rat liver caused by phenobarbital-induced oxidative stress: The protective role of melatonin. Cell Mol. Biol. Lett. 2007, 12, 317–330. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.F.; Sousa, D.S.; Medeiros, A.M.; Macedo, P.T.; Leao, A.H.; Ribeiro, A.M.; Izidio, G.S.; Silva, R.H. Sex and estrous cycle influence diazepam effects on anxiety and memory: Possible role of progesterone. Prog. Neuro Psychopharmacol. Biol. Psychiatry 2016, 70, 68–76. [Google Scholar] [CrossRef] [PubMed]
- El-Sokkary, G.H. Melatonin and vitamin C administration ameliorate diazepam-induced oxidative stress and cell proliferation in the liver of rats. Cell Prolif. 2008, 41, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Khouzam, H.R. A review of trazodone use in psychiatric and medical conditions. Postgrad. Med. 2016, 2016, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Taziki, S.; Sattari, M.R.; Eghbal, M.A. Mechanisms of trazodone-induced cytotoxicity and the protective effects of melatonin and/or taurine toward freshly isolated rat hepatocytes. J. Biochem. Mol. Toxicol. 2013, 27, 457–462. [Google Scholar] [CrossRef] [PubMed]
- Saha, K.B.; Bo, L.; Zhao, S.; Xia, J.; Sampson, S.; Zaman, R.U. Chlorpromazine versus atypical antipsychotic drugs for schizophrenia. Cochrane Database Syst Rev. 2016, 4, CD010631. [Google Scholar] [PubMed]
- Sulaiman, A.A.; Al-Shawi, N.N.; Jwaied, A.H.; Mahmood, D.M.; Hussain, S.A. Protective effect of melatonin against chlorpromazine-induced liver disease in rats. Saudi Med. J. 2006, 27, 1477–1482. [Google Scholar] [PubMed]
- Ghanem, C.I.; Perez, M.J.; Manautou, J.E.; Mottino, A.D. Acetaminophen from liver to brain: New insights into drug pharmacological action and toxicity. Pharmacol. Res. 2016, 109, 119–131. [Google Scholar] [CrossRef] [PubMed]
- Du, K.; Ramachandran, A.; Jaeschke, H. Oxidative stress during acetaminophen hepatotoxicity: Sources, pathophysiological role and therapeutic potential. Redox Biol. 2016, 10, 148–156. [Google Scholar] [CrossRef] [PubMed]
- Matsura, T.; Nishida, T.; Togawa, A.; Horie, S.; Kusumoto, C.; Ohata, S.; Nakada, J.; Ishibe, Y.; Yamada, K.; Ohta, Y. Mechanisms of protection by melatonin against acetaminophen-induced liver injury in mice. J. Pineal Res. 2006, 41, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.L.; Zhang, Z.H.; Liu, X.J.; Liu, X.Q.; Tao, L.; Zhang, Y.F.; Wang, H.; Zhang, C.; Chen, X.; Xu, D.X. Melatonin protects against apoptosis-inducing factor (AIF)-dependent cell death during acetaminophen-induced acute liver failure. PLoS ONE 2012, 7, e51911. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Zheng, J.; Li, S.; Zhou, T.; Zhang, P.; Li, H.B. Alcoholic beverage consumption and chronic diseases. Int. J. Environ. Res. Public Health 2016, 13, 522. [Google Scholar] [CrossRef]
- Wang, F.; Li, Y.; Zhang, Y.J.; Zhou, Y.; Li, S.; Li, H.B. Natural products for the prevention and treatment of hangover and alcohol use disorder. Molecules 2016, 21, 64. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Zhang, Y.J.; Zhou, Y.; Li, Y.; Zhou, T.; Zheng, J.; Zhang, J.J.; Li, S.; Xu, D.P.; Li, H.B. Effects of beverages on alcohol metabolism: Potential health benefits and harmful impacts. Int. J. Mol. Sci. 2016, 17, 354. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.J.; Wang, F.; Zhou, Y.; Li, Y.; Zhou, T.; Zheng, J.; Zhang, J.J.; Li, S.; Xu, D.P.; Li, H.B. Effects of 20 selected fruits on ethanol metabolism: Potential health benefits and harmful impacts. Int. J. Environ. Res. Public Health 2016, 13, 399. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.J.; Zhou, T.; Wang, F.; Zhou, Y.; Li, Y.; Zhang, J.J.; Zheng, J.; Xu, D.P.; Li, H.B. The effects of Syzygium samarangense, Passiflora edulis and Solanum muricatum on alcohol-induced liver injury. Int. J. Mol. Sci. 2016, 17, 1616. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.L.; Yin, S.; Jiang, X.D.; Huang, D.B.; Shen, G. Melatonin protects against alcoholic liver injury by attenuating oxidative stress, inflammatory response, and apoptosis. Eur. J. Pharmacol. 2009, 616, 287–292. [Google Scholar] [CrossRef] [PubMed]
- Mishra, A.; Paul, S.; Swarnakar, S. Downregulation of matrix metalloproteinase-9 by melatonin during prevention of alcohol-induced liver injury in mice. Biochimie 2011, 93, 854–866. [Google Scholar] [CrossRef] [PubMed]
- Taysi, S.; Koc, M.; Buyukokuroglu, M.E.; Altinkaynak, K.; Sahin, Y.N. Melatonin reduces lipid peroxidation and nitric oxide during irradiation-induced oxidative injury in the rat liver. J. Pineal Res. 2003, 34, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Shirazi, A.; Mihandoost, E.; Ghobadi, G.; Mohseni, M.; Ghazi-Khansari, M. Evaluation of radio-protective effect of melatonin on whole body irradiation induced liver tissue damage. Cell J. 2013, 14, 292–297. [Google Scholar]
- Karbownik, M.; Reiter, R.J.; Qi, W.B.; Garcia, J.J.; Tan, D.X.; Manchester, L.C.; Vijayalaxmi. Protective effects of melatonin against oxidation of guanine bases in DNA and decreased microsomal membrane fluidity in rat liver induced by whole body ionizing radiation. Mol. Cell. Biochem. 2000, 211, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Koc, M.; Taysi, S.; Buyukokuroglu, M.E.; Bakan, N. Melatonin protects rat liver against irradiation-induced oxidative injury. J. Radiat. Res. 2003, 44, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Djordjevic, B.; Sokolovic, D.; Kocic, G.; Veljkovic, A.; Despotovic, M.; Basic, J.; Jevtovic-Stoimenov, T.; Sokolovic, D.M. The effect of melatonin on the liver of rats exposed to microwave radiation. Bratisl. Med. J. 2015, 116, 96–100. [Google Scholar] [CrossRef]
- Taghizadieh, M.; Hajipour, B.; Asl, N.A.; Khodadadi, A.; Somi, M.H.; Banei, M. Combination effect of melatonin and dexamethasone on liver ischemia/reperfusion injury. Bratisl. Med. J. 2016, 117, 47–53. [Google Scholar] [CrossRef]
- Kang, J.W.; Cho, H.I.; Lee, S.M. Melatonin inhibits mTOR-dependent autophagy during liver ischemia/reperfusion. Cell. Physiol. Biochem. 2014, 33, 23–36. [Google Scholar] [CrossRef] [PubMed]
- Deng, W.S.; Xu, Q.; Liu, Y.; Jiang, C.H.; Zhou, H. Effects of melatonin on liver function and lipid peroxidation in a rat model of hepatic ischemia/reperfusion injury. Exp. Ther. Med. 2016, 11, 1955–1960. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.W.; Lee, S.M. Melatonin inhibits type 1 interferon signaling of toll-like receptor 4 via heme oxygenase-1 induction in hepatic ischemia/reperfusion. J. Pineal Res. 2012, 53, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Bekyarova, G.; Tzaneva, M.; Hristova, M.; Hristov, K. Melatonin protection against burn-induced liver injury. A review. Cent. Eur. J. Med. 2014, 9, 148–158. [Google Scholar] [CrossRef]
- Bekyarova, G.; Apostolova, M.; Kotzev, I. Melatonin protection against burn-induced hepatic injury by down-regulation of nuclear factor κB activation. Int. J. Immunopathol. Pharmacol. 2012, 25, 591–596. [Google Scholar] [CrossRef] [PubMed]
- Bekyarova, G.; Bratchkova, Y.; Tancheva, S.; Hristova, M. Effective melatonin protection of burn-induced hepatic disorders in rats. Cent. Eur. J. Med. 2012, 7, 533–538. [Google Scholar] [CrossRef]
- Bekyarova, G.; Tzaneva, M.; Hristova, M. Melatonin protects against burn-induced hepatic oxidative injury by inducing HO-1 via the Nrf2 pathway. Vet. Med. 2015, 60, 621–628. [Google Scholar] [CrossRef]
- Nickkholgh, A.; Schneider, H.; Sobirey, M.; Venetz, W.P.; Hinz, U.; Pelzl, L.H.; Gotthardt, D.N.; Cekauskas, A.; Manikas, M.; Mikalauskas, S.; et al. The use of high-dose melatonin in liver resection is safe: First clinical experience. J. Pineal Res. 2011, 50, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Montilla, P.; Cruz, A.; Padillo, F.J.; Tunez, I.; Gascon, F.; Munoz, M.C.; Gomez, M.; Pera, C. Melatonin versus vitamin E as protective treatment against oxidative stress after extra-hepatic bile duct ligation in rats. J. Pineal Res. 2001, 31, 138–144. [Google Scholar] [CrossRef]
- Ohta, Y.; Kongo, M.; Kishikawa, T. Melatonin exerts a therapeutic effect on cholestatic liver injury in rats with bile duct ligation. J. Pineal Res. 2003, 34, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Hsu, J.T.; Kuo, C.J.; Chen, T.H.; Wang, F.; Lin, C.J.; Yeh, T.S.; Hwang, T.L.; Jan, Y.Y. Melatonin prevents hemorrhagic shock-induced liver injury in rats through an Akt-dependent HO-1 pathway. J. Pineal Res. 2012, 53, 410–416. [Google Scholar] [CrossRef] [PubMed]
- Oner, J.; Ozan, E. Effects of melatonin on liver of rats with experimental hyperthyroid. Acta Vet. Beogr. 2005, 55, 31–39. [Google Scholar]
- Martinez-Cruz, F.; Osuna, C.; Guerrero, J.M. Mitochondrial damage induced by fetal hyperphenylalaninemia in the rat brain and liver: Its prevention by melatonin, vitamin E, and vitamin C. Neurosci. Lett. 2006, 392, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Sener, G.; Balkan, J.; Cevikbas, U.; Keyer-Uysal, M.; Uysal, M. Melatonin reduces cholesterol accumulation and prooxidant state induced by high cholesterol diet in the plasma, the liver and probably in the aorta of C57BL/6J mice. J. Pineal Res. 2004, 36, 212–216. [Google Scholar] [CrossRef] [PubMed]
- Balkan, J.; Sener, G.; Cevikbas, U.; Keyer-Uysall, M.; Uysall, M. Melatonin improved the disturbances in hepatic prooxidant and antioxidant balance and hepatotoxicity induced by a high cholesterol diet in C57BL/6J mice. Int. J. Vitam. Nutr. Res. 2004, 74, 349–354. [Google Scholar] [CrossRef] [PubMed]
- Baydas, G.; Ercel, E.; Canatan, H.; Donder, E.; Akyol, A. Effect of melatonin on oxidative status of rat brain, liver and kidney tissues under constant light exposure. Cell Biochem. Funct. 2001, 19, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Gedikli, S.; Gelen, V.; Sengul, E.; Ozkanlar, S.; Gur, C.; Agirbas, O.; Cakmak, F.; Kara, A. Therapeutic effects of melatonin on liver and kidney damages in intensive exercise model of rats. Endocr. Metab. Immune Disord. Drug Targets 2015, 15, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Wei, W.; Shen, Y.X.; Dong, C.; Zhang, L.L.; Wang, N.P.; Yue, L.; Xu, S.Y. Protective effect of melatonin against liver injury in mice induced by Bacillus Calmette-Guerin plus lipopolysaccharide. World J. Gastroenterol. 2004, 10, 2690–2696. [Google Scholar] [CrossRef] [PubMed]
- Laothong, U.; Pinlaor, P.; Hiraku, Y.; Boonsiri, P.; Prakobwong, S.; Khoontawad, J.; Pinlaor, S. Protective effect of melatonin against Opisthorchis viverrini-induced oxidative and nitrosative DNA damage and liver injury in hamsters. J. Pineal Res. 2010, 49, 271–282. [Google Scholar] [CrossRef] [PubMed]
- Tunon, M.J.; Miguel, B.S.; Crespo, I.; Jorquera, F.; Santamaria, E.; Alvarez, M.; Prieto, J.; Gonzalez-Gallego, J. Melatonin attenuates apoptotic liver damage in fulminant hepatic failure induced by the rabbit hemorrhagic disease virus. J. Pineal Res. 2011, 50, 38–45. [Google Scholar] [CrossRef] [PubMed]
- San-Miguel, B.; Crespo, I.; Vallejo, D.; Alvarez, M.; Prieto, J.; Gonzalez-Gallego, J.; Tunon, M.J. Melatonin modulates the autophagic response in acute liver failure induced by the rabbit hemorrhagic disease virus. J. Pineal Res. 2014, 56, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Norris, G.H.; Porter, C.M.; Jiang, C.; Millar, C.L.; Blesso, C.N. Dietary sphingomyelin attenuates hepatic steatosis and adipose tissue inflammation in high-fat-diet-induced obese mice. J. Nutr. Biochem. 2017, 40, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Tiao, M.M.; Huang, L.T.; Chen, C.J.; Sheen, J.M.; Tain, Y.L.; Chen, C.C.; Kuo, H.C.; Huang, Y.H.; Tang, K.S.; Chu, E.W.; Yu, H.R. Melatonin in the regulation of liver steatosis following prenatal glucocorticoid exposure. BioMed Res. Int. 2014, 2014, 942172. [Google Scholar] [CrossRef] [PubMed]
- Hermoso, D.A.M.; Shimada, L.B.C.; Gilglioni, E.H.; Constantin, J.; Mito, M.S.; Hermoso, A.P.M.; Salgueiro-Pagadigorria, C.L.; Iwamoto, E.L.I. Melatonin protects female rats against steatosis and liver oxidative stress induced by oestrogen deficiency. Life Sci. 2016, 157, 178–186. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; Kang, H.S.; Lee, J.H.; Park, J.H.; Jung, C.H.; Bae, J.H.; Oh, B.C.; Song, D.K.; Baek, W.K.; Im, S.S. Melatonin ameliorates ER stress-mediated hepatic steatosis through miR-23a in the liver. Biochem. Biophys. Res. Commun. 2015, 458, 462–469. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.; Song, Y.L.; Xu, J.M.; Gan, H.Z. Melatonin ameliorates nonalcoholic fatty liver induced by high-fat diet in rats. J. Pineal Res. 2006, 41, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Stacchiotti, A.; Favero, G.; Lavazza, A.; Golic, I.; Aleksic, M.; Korac, A.; Rodella, L.F.; Rezzani, R. Hepatic macrosteatosis is partially converted to microsteatosis by melatonin supplementation in ob/ob mice non-alcoholic fatty liver disease. PLoS ONE 2016, 11, e0148115. [Google Scholar] [CrossRef]
- Ferraro, S.M.; Lopez-Ortega, A. Antioxidant activity of melatonin on fatty liver induced by ethionine in mice. Arch. Med. Vet. 2008, 40, 51–57. [Google Scholar]
- Celinski, K.; Konurek, P.C.; Slomka, M.; Cichoz-Lach, H.; Brzozowski, T.; Konturek, S.J.; Korolczuk, A. Effects of treatment with melatonin and tryptophan on liver enzymes, parametrs of fat metabolism and plasma levels of cytokines in patients with non-alcoholic fatty liver disease-14 months follow up. J. Physiol. Pharmacol. 2014, 65, 75–82. [Google Scholar] [PubMed]
- Shima, T.; Uto, H.; Ueki, K.; Takamura, T.; Kohgo, Y.; Kawata, S.; Yasui, K.; Park, H.; Nakamura, N.; Nakatou, T.; et al. Clinicopathological features of liver injury in patients with type 2 diabetes mellitus and comparative study of histologically proven nonalcoholic fatty liver diseases with or without type 2 diabetes mellitus. J. Gastroenterol. 2013, 48, 515–525. [Google Scholar] [CrossRef] [PubMed]
- Agil, A.; Elmahallawy, E.K.; Rodriguez-Ferrer, J.M.; Adem, A.; Bastaki, S.M.; Al-abbadi, I.; Fino Solano, Y.A.; Navarro-Alarcon, M. Melatonin increases intracellular calcium in the liver, muscle, white adipose tissues and pancreas of diabetic obese rats. Food Funct. 2015, 6, 2671–2678. [Google Scholar] [CrossRef] [PubMed]
- Shieh, J.M.; Wu, H.T.; Cheng, K.C.; Cheng, J.T. Melatonin ameliorates high fat diet-induced diabetes and stimulates glycogen synthesis via a PKC zeta-Akt-GSK3 beta pathway in hepatic cells. J. Pineal. Res. 2009, 47, 339–344. [Google Scholar] [CrossRef] [PubMed]
- Zavodnik, I.B.; Lapshina, E.A.; Cheshchevik, V.T.; Dremza, I.K.; Kujawa, J.; Zabrodskaya, S.V.; Reiter, R.J. Melatonin and succinate reduce rat liver mitochondrial dysfunction in diabetes. J. Physiol. Pharmacol. 2011, 62, 421–427. [Google Scholar] [PubMed]
- Vardi, N.; Iraz, M.; Ozturk, F.; Gul, M.; Ucar, M.; Cetin, A.; Nalcaci, N.; Otlu, A. Improving effects of melatonin against on the histologic alterations of rat liver in diabetes. Turk. Klin. Tip Bilim. Derg. 2007, 27, 641–648. [Google Scholar]
- Elbe, H.; Esrefoglu, M.; Vardi, N.; Taslidere, E.; Ozerol, E.; Tanbek, K. Melatonin, quercetin and resveratrol attenuates oxidative hepatocellular injury in streptozotocin-induced diabetic rats. Hum. Exp. Toxicol. 2015, 34, 859–868. [Google Scholar] [CrossRef] [PubMed]
- Guven, A.; Yavuz, O.; Cam, M.; Ercan, F.; Bukan, N.; Comunoglu, C.; Gokce, F. Effects of melatonin on streptozotocin-induced diabetic liver injury in rats. Acta Histochem. 2006, 108, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Korkmaz, G.G.; Uzun, H.; Cakatay, U.; Aydin, S. Melatonin ameliorates oxidative damage in hyperglycemia-induced liver injury. Clin. Investig. Med. 2012, 35, 370–377. [Google Scholar]
- Agarkov, A.A.; Popova, T.N.; Matasova, L.V. Effects of melatonin on the activity of the glutathione antioxidant system and various NADPH-generating enzymes in the liver and blood of rats with type 2 diabetes mellitus. Pharm. Chem. J. 2011, 45, 385–388. [Google Scholar] [CrossRef]
- Allagui, M.S.; Feriani, A.; Bouoni, Z.; Alimi, H.; Murat, J.C.; El Feki, A. Protective effects of vitamins (C and E) and melatonin co-administration on hematological and hepatic functions and oxidative stress in alloxan-induced diabetic rats. J. Physiol. Biochem. 2014, 70, 713–723. [Google Scholar] [CrossRef] [PubMed]
- Cheshchevik, V.T.; Dremza, I.K.; Lapshina, E.A.; Zabrodskaya, S.V.; Kujawa, J.; Zavodnik, I.B. Corrections by melatonin of liver mitochondrial disorders under diabetes and acute intoxication in rats. Cell Biochem. Funct. 2011, 29, 481–488. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.Q.; Dai, J.; Hu, K.; Cai, L.; Lin, L.; Ge, P.; Zhang, L. Metformin alleviates acetaminophen-induced toxic hepatitis. Genom. Appl. Biol. 2016, 35, 1633–1637. [Google Scholar]
- Kurtgoz, P.O.; Tunay, H.; Kurban, F.; Kurtgoz, S.; Demir, K.; Acarturk, G. Toxic hepatitis associated with amoxicillin-clavulanic acid in a chronic viral hepatitis B patient: A case report. Med. J. Bakirkoy 2016, 12, 92–95. [Google Scholar] [CrossRef]
- Thomas, F.V.; Mingo, A.T.; Mantes, D.R.; Uribe, S.O. Albendazole-induced toxic hepatitis. Gastroenterol. Hepatol. 2015, 38, 436–438. [Google Scholar]
- Castiella, A.; Iglesias, U.; Zapata, E.; Zubiaurre, L.; Iribarren, A. Toxic hepatocellular hepatitis due to labetalol. Gastroenterol. Hepatol. 2015, 38, 326–327. [Google Scholar] [CrossRef] [PubMed]
- Couturier, F.J.; Colemont, L.J.; Fierens, H.; Verhoeven, V.M. Toxic hepatitis due to a food supplement: “Natural” is no synonym for “harmless”. Clin. Res. Hepatol. Gastroenterol. 2016, 40, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Pashkov, A.N.; Popov, S.S.; Semenikhina, A.V.; Rakhmanova, T.I. Glutathione system and activity of NADPH-generating enzymes in the liver of intact rats and animals with toxic hepatitis receiving melatonin. Bull. Exp. Biol. Med. 2005, 139, 565–568. [Google Scholar] [CrossRef] [PubMed]
- Laliena, A.; San Miguel, B.; Crespo, I.; Alvarez, M.; Gonzalez-Gallego, J.; Tunon, M.J. Melatonin attenuates inflammation and promotes regeneration in rabbits with fulminant hepatitis of viral origin. J. Pineal Res. 2012, 53, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Tunon, M.J.; San-Miguel, B.; Crespo, I.; Laliena, A.; Vallejo, D.; Alvarez, M.; Prieto, J.; Gonzalez-Gallego, J. Melatonin treatment reduces endoplasmic reticulum stress and modulates the unfolded protein response in rabbits with lethal fulminant hepatitis of viral origin. J. Pineal Res. 2013, 55, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Gonciarz, M.; Gonciarz, Z.; Bielanski, W.; Mularczyk, A.; Konturek, P.C.; Brzozowski, T.; Konturek, S.J. The pilot study of 3-month course of melatonin treatment of patients with nonalcoholic steatohepatitis: Effect on plasma levels of liver enzymes, lipids, and melatonin. J. Physiol. Pharmacol. 2010, 61, 705–710. [Google Scholar] [PubMed]
- Gonciarz, M.; Gonciarz, Z.; Bielanski, W.; Mularczyk, A.; Konturek, P.C.; Brzozowski, T.; Konturek, S.J. The effects of long-term melatonin treatment on plasma liver enzymes levels and plasma concentrations of lipids and melatonin in patients with nonalcoholic steatohepatitis: A pilot study. J. Physiol. Pharmacol. 2012, 63, 35–40. [Google Scholar] [PubMed]
- Wang, P.; Koyama, Y.; Liu, X.; Xu, J.; Ma, H.Y.; Liang, S.; Kim, I.H.; Brenner, D.A.; Kisseleva, T. Promising therapy candidates for liver fibrosis. Front. Physiol. 2016, 7, 47. [Google Scholar] [CrossRef] [PubMed]
- Crespo, I.; San-Miguel, B.; Fernandez, A.; De Urbina, J.O.; Gonzalez-Gallego, J.; Tunon, M.J. Melatonin limits the expression of profibrogenic genes and ameliorates the progression of hepatic fibrosis in mice. Transl. Res. 2015, 165, 346–357. [Google Scholar] [CrossRef] [PubMed]
- Shajari, S.; Laliena, A.; Heegsma, J.; Tunon, M.J.; Moshage, H.; Faber, K.N. Melatonin suppresses activation of hepatic stellate cells through ROR α-mediated inhibition of 5-lipoxygenase. J. Pineal Res. 2015, 59, 391–401. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.S.; Kang, J.W.; Lee, S.M. Melatonin attenuates carbon tetrachloride-induced liver fibrosis via inhibition of necroptosis. Transl. Res. 2015, 166, 292–303. [Google Scholar] [CrossRef] [PubMed]
- Mortezaee, K.; Sabbaghziarani, F.; Omidi, A.; Dehpour, A.R.; Omidi, N.; Ghasemi, S.; Pasbakhsh, P.; Kashani, I.R. Therapeutic value of melatonin post-treatment on CCl4-induced fibrotic rat liver. Can. J. Physiol. Pharmacol. 2016, 94, 119–130. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Wei, W.; Wang, N.P.; Gui, S.Y.; Wu, L.; Sun, W.Y. Melatonin ameliorates carbon tetrachloride-induced hepatic fibrogenesis in rats via inhibition of oxidative stress. Life Sci. 2005, 77, 1902–1915. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.W.; Hong, J.M.; Lee, S.M. Melatonin enhances mitophagy and mitochondrial biogenesis in rats with carbon tetrachloride-induced liver fibrosis. J. Pineal Res. 2016, 60, 383–393. [Google Scholar] [CrossRef] [PubMed]
- Hong, R.T.; Xu, J.M.; Mei, Q. Melatonin ameliorates experimental hepatic fibrosis induced by carbon tetrachloride in rats. World J. Gastroentero. 2009, 15, 1452–1458. [Google Scholar] [CrossRef]
- Cho, Y.A.; Noh, K.; Jue, S.S.; Lee, S.Y.; Kim, E.C. Melatonin promotes hepatic differentiation of human dental pulp stem cells: Clinical implications for the prevention of liver fibrosis. J. Pineal Res. 2015, 58, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Tahan, G.; Akin, H.; Aydogan, F.; Ramadan, S.S.; Yapicier, O.; Tarcin, O.; Uzun, H.; Tahan, V.; Zengin, K. Melatonin ameliorates liver fibrosis induced by bile-duct ligation in rats. Can. J. Surg. 2010, 53, 313–318. [Google Scholar] [PubMed]
- Czechowska, G.; Celinski, K.; Korolczuk, A.; Wojcicka, G.; Dudka, J.; Bojarska, A.; Reiter, R.J. Protective effects of melatonin against thioacetamide-induced liver fibrosis in rats. J Physiol. Pharmacol. 2015, 66, 567–579. [Google Scholar] [PubMed]
- Tahan, V.; Ozaras, R.; Canbakan, B.; Uzun, H.; Aydin, S.; Yildirim, B.; Aytekin, H.; Ozbay, G.; Mert, A.; Senturk, H. Melatonin reduces dimethylnitrosamine-induced liver fibrosis in rats. J. Pineal Res. 2004, 37, 78–84. [Google Scholar] [CrossRef]
- Cruz, A.; Padillo, F.J.; Torres, E.; Navarrete, C.M.; Munoz-Castaneda, J.R.; Caballero, F.J.; Briceno, J.; Marchal, T.; Tunez, I.; Montilla, P.; et al. Melatonin prevents experimental liver cirrhosis induced by thioacetamide in rats. J. Pineal Res. 2005, 39, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Chojnacki, C.; Walecka-Kapica, E.; Klupinska, G.; Wachowska-Kelly, P.; Zylinska, K.; Winczyk, K.; Chojnacki, J. Serotonin and melatonin secretion and metabolism in patients with liver cirrhosis. Pol. Arch. Med. Wewn. 2012, 122, 392–397. [Google Scholar] [PubMed]
- Cunnane, S.C.; Manku, M.S.; Horrobin, D.F. The pineal and regulation of fibrosis: Pinealectomy as a model of primary biliary cirrhosis: Roles of melatonin and prostaglandins in fibrosis and regulation of T lymphocytes. Med. Hypotheses 1979, 5, 403–414. [Google Scholar] [CrossRef]
- Colares, J.R.; Schemitt, E.G.; Hartmann, R.M.; Licks, F.; Soares, M.D.; Dal Bosco, A.; Marroni, N.P. Antioxidant and anti-inflammatory action of melatonin in an experimental model of secondary biliary cirrhosis induced by bile duct ligation. World J. Gastroenterol. 2016, 22, 8918–8928. [Google Scholar] [CrossRef] [PubMed]
- Celinski, K.; Konturek, P.C.; Slomka, M.; Cichoz-Lach, H.; Gonciarz, M.; Bielanski, W.; Reiter, R.J.; Konturek, S.J. Altered basal and postprandial plasma melatonin, gastrin, ghrelin, leptin and insulin in patients with liver cirrhosis and portal hypertension without and with oral administration of melatonin or tryptophan. J. Pineal Res. 2009, 46, 408–414. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Zheng, J.; Li, Y.; Xu, D.P.; Li, S.; Chen, Y.M.; Li, H.B. Natural polyphenols for prevention and treatment of cancer. Nutrients 2016, 8, 515. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Li, S.; Li, H.B.; Deng, G.F.; Ling, W.H.; Xu, X.R. Antiproliferative activities of tea and herbal infusions. Food Funct. 2013, 4, 530–538. [Google Scholar] [CrossRef] [PubMed]
- Carbajo-Pescador, S.; Martin-Renedo, J.; Garcia-Palomo, A.; Tunon, M.J.; Mauriz, J.L.; Gonzalez-Gallego, J. Changes in the expression of melatonin receptors induced by melatonin treatment in hepatocarcinoma HepG2 cells. J. Pineal Res. 2009, 47, 330–338. [Google Scholar] [CrossRef]
- Zhou, Y.; Li, Y.; Zhou, T.; Zheng, J.; Li, S.; Li, H.B. Dietary natural products for prevention and treatment of liver cancer. Nutrients 2016, 8, 156. [Google Scholar] [CrossRef] [PubMed]
- Moreira, A.J.; Ordonez, R.; Cerski, C.T.; Picada, J.N.; Garcia-Palomo, A.; Marroni, N.P.; Mauriz, J.L.; Gonzalez-Gallego, J. Melatonin activates endoplasmic reticulum stress and apoptosis in rats with diethylnitrosamine-induced hepatocarcinogenesis. PLoS ONE 2015, 10, e0144517. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Zhou, Y.; Li, Y.; Xu, D.P.; Li, S.; Li, H.B. Spices for prevention and treatment of cancers. Nutrients 2016, 8, 495. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Li, S.; Li, H.B.; Deng, G.F.; Ling, W.H.; Wu, S.; Xu, X.R.; Chen, F. Antiproliferative activity of peels, pulps and seeds of 61 fruits. J. Funct. Foods 2013, 5, 1298–1309. [Google Scholar] [CrossRef]
- Kimball, S.R.; Abbas, A.; Jefferson, L.S. Melatonin represses oxidative stress-induced activation of the MAP kinase and mTOR signaling pathways in H4IIE hepatoma cells through inhibition of Ras. J. Pineal Res. 2008, 44, 379–386. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, J.; Saegusa, Y.; Dewa, Y.; Jin, M.; Kawai, M.; Kemmochi, S.; Harada, T.; Hayashi, S.; Shibutani, M.; Mitsumori, K. Antioxidant enzymatically modified isoquercitrin or melatonin supplementation reduces oxidative stress-mediated hepatocellular tumor promotion of oxfendazole in rats. Arch. Toxicol. 2010, 84, 143–153. [Google Scholar] [CrossRef] [PubMed]
- Verma, D.; Hashim, O.H.; Jayapalan, J.J.; Subramanian, P. Effect of melatonin on antioxidant status and circadian activity rhythm during hepatocarcinogenesis in mice. J. Canc. Res. Ther. 2014, 10, 1040–1044. [Google Scholar]
- Dakshayani, K.B.; Subramanian, R.; Essa, M.M. Effect of melatonin on N-nitrosodiethylamine-induced hepatocarcinogenesis in rats with reference to biochemical circadian rhythms. Toxicol. Mech. Methods 2007, 17, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, P.; Dakshayani, K.B.; Pandi-Perumal, S.R.; Trakht, I.; Cardinali, D.P. 24-hour rhythms in oxidative stress during hepatocarcinogenesis in rats: Effect of melatonin or α-ketoglutarate. Redox Rep. 2008, 13, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Carbajo-Pescador, S.; Garcia-Palomo, A.; Martin-Renedo, J.; Piva, M.; Gonzalez-Gallego, J.; Mauriz, J.L. Melatonin modulation of intracellular signaling pathways in hepatocarcinoma HepG2 cell line: Role of the MT1 receptor. J. Pineal Res. 2011, 51, 463–471. [Google Scholar] [CrossRef] [PubMed]
- Hermann, R.; Podhajsky, S.; Jungnickel, S.; Lerchl, A. Potentiation of antiproliferative effects of tamoxifen and ethanol on mouse hepatoma cells by melatonin: Possible involvement of mitogen-activated protein kinase and induction of apoptosis. J. Pineal Res. 2002, 33, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Cid, M.A.; Ubeda, A.; Hernandez-Bule, M.L.; Martinez, M.A.; Trillo, M.A. Antagonistic effects of a 50 Hz magnetic field and melatonin in the proliferation and differentiation of hepatocarcinoma cells. Cell. Physiol. Biochem. 2012, 30, 1502–1516. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.L.; Sun, G.P.; Ma, T.; Zhong, F.; Wei, W. Melatonin overcomes apoptosis resistance in human hepatocellular carcinoma by targeting Survivin and XIAP. J. Pineal Res. 2013, 55, 174–183. [Google Scholar] [CrossRef] [PubMed]
- Martin-Renedo, J.; Mauriz, J.L.; Jorquera, F.; Ruiz-Andres, O.; Gonzalez, P.; Gonzalez-Gallego, J. Melatonin induces cell cycle arrest and apoptosis in hepatocarcinoma HepG2 cell line. J. Pineal Res. 2008, 45, 532–540. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.L.; Sun, G.P.; Wei, W.; Wang, Z.G.; Ge, L.; Fu, W.Z.; Wang, H. Melatonin and doxorubicin synergistically induce cell apoptosis in human hepatoma cell lines. World J. Gastroenterol. 2010, 16, 1473–1481. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.L.; Sun, G.P.; Ma, T.; Zhong, F.; Lei, Y.; Li, X.Q.; Wei, W. Melatonin reverses tunicamycin-induced endoplasmic reticulum stress in human hepatocellular carcinoma cells and improves cytotoxic response to doxorubicin by increasing CHOP and decreasing Survivin. J. Pineal Res. 2013, 55, 184–194. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Jia, Z.L.; Zhang, X.; Hou, J.C.; Wang, L.; Hao, S.L.; Ruan, X.J.; Yu, Z.H.; Zheng, Y.Q. Involvement of melatonin in autophagy-mediated mouse hepatoma H22 cell survival. Int. Immunopharmacol. 2012, 12, 394–401. [Google Scholar] [CrossRef] [PubMed]
- Ordonez, R.; Fernandez, A.; Prieto-Dominguez, N.; Martinez, L.; Garcia-Ruiz, C.; Fernandez-Checa, J.C.; Mauriz, J.L.; Gonzalez-Gallego, J. Ceramide metabolism regulates autophagy and apoptotic cell death induced by melatonin in liver cancer cells. J. Pineal Res. 2015, 59, 178–189. [Google Scholar] [CrossRef] [PubMed]
- Ordonez, R.; Carbajo-Pescador, S.; Prieto-Dominguez, N.; Garcia-Palomo, A.; Gonzalez-Gallego, J.; Mauriz, J.L. Inhibition of matrix metalloproteinase-9 and nuclear factor kappa B contribute to melatonin prevention of motility and invasiveness in HepG2 liver cancer cells. J. Pineal Res. 2014, 56, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Lin, Z.Y.; Chuang, W.L. Pharmacologic concentrations of melatonin have diverse influence on differential expressions of angiogenic chemokine genes in different hepatocellular carcinoma cell lines. Biomed. Pharmacother. 2010, 64, 659–662. [Google Scholar] [CrossRef] [PubMed]
- Carbajo-Pescador, S.; Ordonez, R.; Benet, M.; Jover, R.; Garcia-Palomo, A.; Mauriz, J.L.; Gonzalez-Gallego, J. Inhibition of VEGF expression through blockade of Hif1α and STAT3 signalling mediates the anti-angiogenic effect of melatonin in HepG2 liver cancer cells. Br. J. Cancer 2013, 109, 83–91. [Google Scholar] [CrossRef] [PubMed]
| Toxins | Subjects | Methods of Melatonin Administration | Duration of Melatonin Treatment | Melatonin Doses | Melatonin Effects | Ref. |
|---|---|---|---|---|---|---|
| Methanol | Rats | Intragastric gavage | 6 or 24 h | 10 mg/kg BW or 3 g/kg BW | Reducing the MDA level significantly, restoring the protein carbonylation level, preventing the increase in nitrite level and MPO activity and the reduction in the antioxidant enzyme activities, and returning piecemeal necrosis, lobular lytic necrosis and portal inflammation to normal histologic appearances at a dose of 10 mg/kg | [46] |
| Fluoride | Mice | Peritoneal injection | 30 days | 10 mg/kg BW/daily | Preventing the decrease in body and liver weight as well as the decrease in liver enzyme activity of succinate dehydrogenase (SDH), acid phosphatase (ACP), alkaline phosphatase (ALP), and total liver protein level and diminishing the increase in serum glutamate oxaloacetate transaminase (SGOT) and serum glutamate pyruvate transaminase (SGPT) activities in the liver | [47] |
| Aluminum chloride | Rats | Oral administration | 30 days | 5 mg/kg BW/daily | Alleviating the increases in the plasma of the ALT, AST, ALP, total bilirubin, total lipids, total cholesterol, TG and glucose levels, and attenuating the decrease in total proteins, reducing oxidative stress, and improving histological changes | [48] |
| Dimethyl-nitrosamine | Rats | Intraperitoneal injection | 14 days | 50 mg/kg BW/daily | Improving serum and antioxidant enzyme activities, reducing the infiltration of inflammatory cells and necrosis in the liver, and increasing the expression of nicotinamide adenine dinucleotide phosphate (NADPH): quinone oxidoreductase-1, HO-1, and SOD2, and increasing novel transcription factor expression, nuclear erythroid 2-related factor 2(Nrf2) and decreasing inflammatory mediators expression | [49] |
| Thio-acetamide | Rats | Intraperitoneal injection | 24 h | 3 mg/kg BW | Decreasing serum liver enzymes and blood ammonia levels, improving liver histological changes, decreasing mortality of rats, inhibiting the increase in nuclear binding of nuclear factor kappa B (NFκB), and decreasing the hepatic level of thiobarbituric acid reactive substances, protein carbonyls and inducible NO synthase, improving survival and reducing liver damage and oxidative stress | [50] |
| Nicotine | Rats | Subcutaneous injection | 30 days | 10 mg/kg BW/daily | Attenuating the increase in LPO products and restoring the SOD activity and GSH level, and reducing both nitrotyrosine reactivity and tissue damage | [51] |
| Paraquat | Rats and hepatocytes | Preincubation with melatonin in vitro | 30 min | 0.5, 1 or 2 mM | Preventing in a dose- and time- dependent manner the loss of viability, the leakage of lactate dehydrogenase, depletion of intracellular glutathione and MDA accumulation, and inhibiting cell damage completely at 2 mM dose | [52] |
| Factors | Subjects | Methods of Melatonin Administration | Duration of Melatonin Treatment | Melatonin Doses | Melatonin Effects | Ref. |
|---|---|---|---|---|---|---|
| Liver resection | Patients | Through a nasogastric tube | A single dose | 50 mg/kg BW | Resulting in lower postoperative transaminases, and inducing a trend toward shorter ICU stay and total hospital stay | [99] |
| Bile duct ligation | Rats | Injection or oral administration | 8 days | 500 μg/kg BW/daily, and 10, 100 mg/kg BW daily | Resulting a significant recovery of antioxidant enzymes and a reduction in the negative parameters of cholestasis at the concentration of 500 mg/kg, and attenuating cholestatic liver injury and reducing the increases in serum and hepatic TBARS concentrations and hepatic MPO activity at the concentration of 10 and 100 mg/kg | [100,101] |
| Hemorrhagic shock | Rats | Intravenous injection | A single dose | 2 mg/kg BW | Normalizing liver Akt phosphorylation, increasing mTOR activation and HO-1 expression, and reducing cleaved caspase-3 level | [102] |
| Experimental hyperthyroid | Rats | Intraperitoneal injection | 20 days | 6 mg/kg BW/daily | Increasing the number of Kupffer cells, lipid vacuoles of Ito cells and microvilli of hepatocytes, and enlarging the spaces of disse | [103] |
| Hyperphenylalaninemia | Rats | Subcutaneous injection | From mating day until delivery | 20 mg/kg BW/daily | Preventing the accumulation of LPO products | [104] |
| High cholesterol diet | Mice | Oral administration | 4 months | 10 mg/L in drinking water | Reducing plasma, liver cholesterol, hepatic MDA, diene conjugate (DC) and liver TG levels, increasing hepatic α-tocopherol and ascorbic acid levels and liver GSH-Px and GST activities, and attenuating the histopathological lesions | [105,106] |
| Constant light exposure | Rats | Subcutaneous injection | 14 days | 1 mg/kg BW/daily | Decreasing lipid peroxidation, and increasing GSH-Px activity | [107] |
| Intensive exercise | Rats | Intra-peritoneal injection | 10 days | 10 mg/kg BW/daily | Increasing the parameters of enzymes in serum, liver and kidney, and decreasing cellular degenerations | [108] |
| Bacillus Calmette Guerin and lipopolysaccharide | Mice, kupffer cells and hepatocytes | Using feeding needle in vivo or culture in vitro | 10 days in vivo or 48 h in vitro | 0.25, 1.0, 4.0 mg/kg BW/daily in vivo, 10−9, 10−8, 10−7, 10−6, 10−5 M in vitro | Decreasing serum ALT, AST activities at the concentration of 0.25, 1.0, 4.0 mg/kg, reducing MDA content, pro-inflammatory mediators (TNF-α, IL-1, NO) and immigration of inflammatory cells, upregulating SOD, attenuating the area and extent of necrosis and inhibiting TNF-α at the concentrations of 10−8–10−6 M, and decreasing IL-1 production of kupffer cells at the concentration of 10−6 M | [109] |
| Opisthorchis viverrini | Hamsters | Oral administration | 30 days | 5, 10, and 20 mg/kg BW/daily | Decreasing the formation of oxidative and nitrosative DNA lesions, 8-oxo-7, 8-dihydro-2’-deoxyguanosine, 3-nitrotyrosine and 8-nitroguanine in the nucleus of bile duct epithelium and inflammatory cells, reducing the HO-1 expression, mRNA expression of oxidant-generating genes (inducible NO synthase, NFκB, and cyclooxygenase-2) and proinflammatory cytokines (TNF-α and IL-1β), cytokeratin 19, nitrate/nitrite, 8-isoprostane and vitamin E levels, ALT activity and bile duct proliferation in the liver and increasing antioxidant genes (Nrf2 and Mn-SOD) expression | [110] |
| Rabbit hemorrhagic disease virus | Rabbits | Dissolved into dilutions | 24 h | 10 or 20 mg/kg BW | Inhibiting autophagic response significantly, and attenuating apoptosis | [111,112] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, J.-J.; Meng, X.; Li, Y.; Zhou, Y.; Xu, D.-P.; Li, S.; Li, H.-B. Effects of Melatonin on Liver Injuries and Diseases. Int. J. Mol. Sci. 2017, 18, 673. https://doi.org/10.3390/ijms18040673
Zhang J-J, Meng X, Li Y, Zhou Y, Xu D-P, Li S, Li H-B. Effects of Melatonin on Liver Injuries and Diseases. International Journal of Molecular Sciences. 2017; 18(4):673. https://doi.org/10.3390/ijms18040673
Chicago/Turabian StyleZhang, Jiao-Jiao, Xiao Meng, Ya Li, Yue Zhou, Dong-Ping Xu, Sha Li, and Hua-Bin Li. 2017. "Effects of Melatonin on Liver Injuries and Diseases" International Journal of Molecular Sciences 18, no. 4: 673. https://doi.org/10.3390/ijms18040673
APA StyleZhang, J.-J., Meng, X., Li, Y., Zhou, Y., Xu, D.-P., Li, S., & Li, H.-B. (2017). Effects of Melatonin on Liver Injuries and Diseases. International Journal of Molecular Sciences, 18(4), 673. https://doi.org/10.3390/ijms18040673







