Thyroid Disrupting Chemicals
Abstract
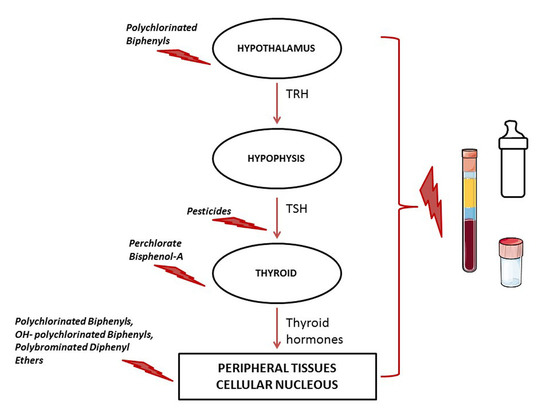
1. Introduction
2. Industrial Chemicals
2.1. Polychlorinated Biphenyls (PCBs)
2.2. Polybrominated Diphenyl Ethers (PBDEs)
2.3. Perchlorate
2.4. Bisphenol-A and Phthalates
3. Pesticides
4. Perfluoroalkyl
5. Potential New Models to Evaluate EDCs
6. Conclusions
Author Contributions
Conflicts of Interest
References
- Diamanti-Kandarakis, E.; Bourguignon, J.P.; Giudice, L.C.; Hauser, R.; Prins, G.S.; Soto, A.M.; Zoeller, R.T.; Gore, A.C. Endocrine-disrupting chemicals: An Endocrine Society scientific statement. Endocr. Rev. 2009, 30, 293–342. [Google Scholar] [CrossRef] [PubMed]
- Zoeller, R.T.; Brown, T.R.; Doan, L.L.; Gore, A.C.; Skakkebaek, N.E.; Soto, A.M.; Woodruff, T.J.; Vom Saal, F.S. Endocrine-disrupting chemicals and public health protection: A statement of principles from The Endocrine Society. Endocrinology 2012, 153, 4097–4110. [Google Scholar] [CrossRef] [PubMed]
- Bunay, J.; Larriba, E.; Patino-Garcia, D.; Cruz-Fernandes, L.; Castaneda-Zegarra, S.; Rodriguez-Fernandez, M.; Del Mazo, J.; Moreno, R.D. Differential Effects of Exposure to Single versus a Mixture of Endocrine-Disrupting Chemicals on Steroidogenesis Pathway in Mouse Testes. Toxicol. Sci. 2017. [Google Scholar] [CrossRef]
- Vandenberg, L.N.; Colborn, T.; Hayes, T.B.; Heindel, J.J.; Jacobs, D.R.; Lee, D.H.; Shioda, T.; Soto, A.M.; vom Saal, F.S.; Welshons, W.V.; et al. Hormones and Endocrine-Disrupting Chemicals: Low-Dose Effects and Nonmonotonic Dose Responses. Endocr. Rev. 2012, 33, 378–455. [Google Scholar] [CrossRef] [PubMed]
- Lagarde, F.; Beausoleil, C.; Belcher, S.M.; Belzunces, L.P.; Emond, C.; Guerbet, M.; Rousselle, C. Non-monotonic dose-response relationships and endocrine disruptors: A qualitative method of assessment. Environ. Health 2015, 14, 13. [Google Scholar] [CrossRef] [PubMed]
- Brouwer, A.; Morse, D.C.; Lans, M.C.; Schuur, A.G.; Murk, A.J.; Klasson-Wehler, E.; Bergman, A.; Visser, T.J. Interactions of persistent environmental organohalogens with the thyroid hormone system: Mechanisms and possible consequences for animal and human health. Toxicol. Ind. Health 1998, 14, 59–84. [Google Scholar] [CrossRef] [PubMed]
- Duntas, L.H. Chemical contamination and the thyroid. Endocrine 2015, 48, 53–64. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Endocrine Disruptors: Mejor Step Towards Protecting Citizens and Environment; European Commission: Brussels, Belgium, 2017. [Google Scholar]
- Boas, M.; Feldt-Rasmussen, U.; Main, K.M. Thyroid effects of endocrine disrupting chemicals. Mol. Cell. Endocrinol. 2012, 355, 240–248. [Google Scholar] [CrossRef] [PubMed]
- Gore, A.C.; Chappell, V.A.; Fenton, S.E.; Flaws, J.A.; Nadal, A.; Prins, G.S.; Toppari, J.; Zoeller, R.T. EDC-2, The Endocrine Society’s Second Scientific Statement on Endocrine-Disrupting Chemicals. Endocr. Rev. 2015, 36, E1–E150. [Google Scholar] [CrossRef] [PubMed]
- Koopmanesseboom, C.; Morse, D.C.; Weisglaskuperus, N.; Brouwer, A.; Sauer, P.J. Effects of Dioxins and Polychlorinated-Biphenyls on Thyroid-Hormone Status of Pregnant-Women and Their Infants. Pediatr. Res. 1994, 36, 468–473. [Google Scholar] [CrossRef] [PubMed]
- Longnecker, M.P.; Gladen, B.C.; Patterson, D.G.; Rogan, W.J. Polychlorinated biphenyl (PCB) exposure in relation to thyroid hormone levels in neonates. Epidemiology 2000, 11, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Soechitram, S.D.; Berghuis, S.A.; Visser, T.J.; Sauer, P.J. Polychlorinated biphenyl exposure and deiodinase activity in young infants. Sci. Total Environ. 2017, 574, 1117–1124. [Google Scholar] [CrossRef] [PubMed]
- Abdelouahab, N.; Langlois, M.F.; Lavoie, L.; Corbin, F.; Pasquier, J.C.; Takser, L. Maternal and cord-blood thyroid hormone levels and exposure to polybrominated diphenyl ethers and polychlorinated biphenyls during early pregnancy. Am. J. Epidemiol. 2013, 178, 701–713. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; He, C.T.; Chen, S.J.; Yan, X.; Guo, M.N.; Wang, M.H.; Yu, Y.J.; Yang, Z.Y.; Mai, B.X. Disruption of thyroid hormone (TH) levels and TH-regulated gene expression by polybrominated diphenyl ethers (PBDEs), polychlorinated biphenyls (PCBs), and hydroxylated PCBs in e-waste recycling workers. Environ. Int. 2017, 102, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Fowles, J.R.; Fairbrother, A.; Baecher-Steppan, L.; Kerkvliet, N.I. Immunologic and endocrine effects of the flame-retardant pentabromodiphenyl ether (DE-71) in C57BL/6J mice. Toxicology 1994, 86, 49–61. [Google Scholar] [CrossRef]
- Lee, E.; Kim, T.H.; Choi, J.S.; Nabanata, P.; Kim, N.Y.; Ahn, M.Y.; Jung, K.K.; Kang, I.H.; Kim, T.S.; Kwack, S.J.; et al. Evaluation of liver and thyroid toxicity in Sprague-Dawley rats after exposure to polybrominated diphenyl ether BDE-209. J. Toxicol. Sci. 2010, 35, 535–545. [Google Scholar] [CrossRef] [PubMed]
- Mazdai, A.; Dodder, N.G.; Abernathy, M.P.; Hites, R.A.; Bigsby, R.M. Polybrominated diphenyl ethers in maternal and fetal blood samples. Environ. Health Perspect. 2003, 111, 1249–1252. [Google Scholar] [CrossRef] [PubMed]
- Julander, A.; Karlsson, M.; Hagstrom, K.; Ohlson, C.G.; Engwall, M.; Bryngelsson, I.L.; Westberg, H.; van Bavel, B. Polybrominated diphenyl ethers—Plasma levels and thyroid status of workers at an electronic recycling facility. Int. Archiv. Occup. Environ. Health 2005, 78, 584–592. [Google Scholar] [CrossRef] [PubMed]
- Vuong, A.M.; Webster, G.M.; Romano, M.E.; Braun, J.M.; Zoeller, R.T.; Hoofnagle, A.N.; Sjodin, A.; Yolton, K.; Lanphear, B.P.; Chen, A. Maternal Polybrominated Diphenyl Ether (PBDE) Exposure and Thyroid Hormones in Maternal and Cord Sera: The HOME Study, Cincinnati, USA. Environ. Health Perspect. 2015, 123, 1079–1085. [Google Scholar] [CrossRef] [PubMed]
- Stapleton, H.M.; Eagle, S.; Anthopolos, R.; Wolkin, A.; Miranda, M.L. Associations between Polybrominated Diphenyl Ether (PBDE) Flame Retardants, Phenolic Metabolites, and Thyroid Hormones during Pregnancy. Environ. Health Perspect. 2011, 119, 1454–1459. [Google Scholar] [CrossRef] [PubMed]
- Zheng, M.Y.; Li, X.H.; Zhang, Y.; Yang, Y.L.; Wang, W.Y.; Tian, Y. Partitioning of polybrominated biphenyl ethers from mother to fetus and potential health-related implications. Chemosphere 2017, 170, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Kronborg, T.M.; Hansen, J.F.; Rasmussen, A.K.; Vorkamp, K.; Nielsen, C.H.; Frederiksen, M.; Hofman-Bang, J.; Hahn, C.H.; Ramhoj, L.; Feldt-Rasmussen, U. The flame retardant DE-71 (a mixture of polybrominated diphenyl ethers) inhibits human differentiated thyroid cell function in vitro. PLoS ONE 2017, 12, e0179858. [Google Scholar] [CrossRef] [PubMed]
- Leung, A.M.; Pearce, E.N.; Braverman, L.E. Environmental perchlorate exposure: Potential adverse thyroid effects. Curr. Opin. Endocrinol. Diabetes Obes. 2014, 21, 372–376. [Google Scholar] [CrossRef] [PubMed]
- McMullen, J.; Ghassabian, A.; Kohn, B.; Trasande, L. Identifying Subpopulations Vulnerable to the Thyroid-Blocking Effects of Perchlorate and Thiocyanate. J. Clin. Endocrinol. Metab. 2017, 102, 2637–2645. [Google Scholar] [CrossRef] [PubMed]
- Blount, B.C.; Pirkle, J.L.; Osterloh, J.D.; Valentin-Blasini, L.; Caldwell, K.L. Urinary perchlorate and thyroid hormone levels in adolescent and adult men and women living in the United States. Environ. Health Perspect. 2006, 114, 1865–1871. [Google Scholar] [CrossRef] [PubMed]
- Suh, M.; Abraham, L.; Hixon, J.G.; Proctor, D.M. The effects of perchlorate, nitrate, and thiocyanate on free thyroxine for potentially sensitive subpopulations of the 2001–2002 and 2007–2008 National Health and Nutrition Examination Surveys. J. Expo. Sci. Environ. Epidemiol. 2014, 24, 579–587. [Google Scholar] [CrossRef] [PubMed]
- Taylor, P.N.; Okosieme, O.E.; Murphy, R.; Hales, C.; Chiusano, E.; Maina, A.; Joomun, M.; Bestwick, J.P.; Smyth, P.; Paradice, R.; et al. Maternal Perchlorate Levels in Women With Borderline Thyroid Function During Pregnancy and the Cognitive Development of Their Offspring: Data From the Controlled Antenatal Thyroid Study. J. Clin. Endocrinol. Metab. 2014, 99, 4291–4298. [Google Scholar] [CrossRef] [PubMed]
- Park, M.; Moon, W.J. Structural MR Imaging in the Diagnosis of Alzheimer’s Disease and Other Neurodegenerative Dementia: Current Imaging Approach and Future Perspectives. Korean J. Radiol. 2016, 17, 827–845. [Google Scholar] [CrossRef] [PubMed]
- Meeker, J.D.; Calafat, A.M.; Hauser, R. Di(2-ethylhexyl) phthalate metabolites may alter thyroid hormone levels in men. Environ. Health Perspect. 2007, 115, 1029–1034. [Google Scholar] [CrossRef] [PubMed]
- Boas, M.; Frederiksen, H.; Feldt-Rasmussen, U.; Skakkebaek, N.E.; Hegedus, L.; Hilsted, L.; Juul, A.; Main, K.M. Childhood exposure to phthalates: Associations with thyroid function, insulin-like growth factor I, and growth. Environ. Health Perspect. 2010, 118, 1458–1464. [Google Scholar] [CrossRef] [PubMed]
- Meeker, J.D.; Ferguson, K.K. Relationship between Urinary Phthalate and Bisphenol A Concentrations and Serum Thyroid Measures in US Adults and Adolescents from the National Health and Nutrition Examination Survey (NHANES) 2007–2008. Environ. Health Perspect. 2011, 119, 1396–1402. [Google Scholar] [CrossRef] [PubMed]
- Aung, M.T.; Johns, L.E.; Ferguson, K.K.; Mukherjee, B.; McElrath, T.F.; Meeker, J.D. Thyroid hormone parameters during pregnancy in relation to urinary bisphenol A concentrations: A repeated measures study. Environ. Int. 2017, 104, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Andrianou, X.D.; Gangler, S.; Piciu, A.; Charisiadis, P.; Zira, C.; Aristidou, K.; Piciu, D.; Hauser, R.; Makris, K.C. Human Exposures to Bisphenol A, Bisphenol F and Chlorinated Bisphenol A Derivatives and Thyroid Function. PLoS ONE 2016, 11, e0155237. [Google Scholar] [CrossRef] [PubMed]
- Minatoya, M.; Sasaki, S.; Araki, A.; Miyashita, C.; Itoh, S.; Yamamoto, J.; Matsumura, T.; Mitsui, T.; Moriya, K.; Cho, K.; et al. Cord Blood Bisphenol A Levels and Reproductive and Thyroid Hormone Levels of Neonates: The Hokkaido Study on Environment and Children’s Health. Epidemiology 2017, 28 (Suppl. S1), S3–S9. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Zhang, J.; Jiang, F.; Xie, Y.; Zhang, X.; Jiang, L. Higher urinary bisphenol A concentration and excessive iodine intake are associated with nodular goiter and papillary thyroid carcinoma. Biosci. Rep. 2017, 37. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Zhou, Y.; Fu, C.; Wang, H.; Huang, P.; Wang, B.; Su, M.; Jiang, F.; Fang, H.; Zhao, Q.; et al. Influence of Bisphenol A on Thyroid Volume and Structure Independent of Iodine in School Children. PLoS ONE 2015, 10, e0141248. [Google Scholar] [CrossRef] [PubMed]
- Romano, M.E.; Webster, G.M.; Vuong, A.M.; Zoeller, R.T.; Chen, A.M.; Hoofnagle, A.N.; Calafat, A.M.; Karagas, M.R.; Yolton, K.; Lanphear, B.P.; Braun, J.M. Gestational urinary bisphenol A and maternal and newborn thyroid hormone concentrations: The HOME Study. Environ. Res. 2015, 138, 453–460. [Google Scholar] [CrossRef] [PubMed]
- Freire, C.; Koifman, R.J.; Sarcinelli, P.N.; Simoes Rosa, A.C.; Clapauch, R.; Koifman, S. Long-term exposure to organochlorine pesticides and thyroid status in adults in a heavily contaminated area in Brazil. Environ. Res. 2013, 127, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Agency for Toxic Substances and Disease Registry. Toxicological Profile for DDT, DDE and DDD; Agency for Toxic Substances and Disease Registry: Atlanta, GA, USA, 2002. [Google Scholar]
- Yaglova, N.V.; Yaglov, V.V. Cytophysiological Changes in the Follicular Epithelium of the Thyroid Gland after Long-Term Exposure to Low Doses of Dichlorodiphenyltrichloroethane (DDT). Bull. Exp. Biol. Med. 2017, 162, 699–702. [Google Scholar] [CrossRef] [PubMed]
- Freire, C.; Lopez-Espinosa, M.J.; Fernandez, M.; Molina-Molina, J.M.; Prada, R.; Olea, N. Prenatal exposure to organochlorine pesticides and TSH status in newborns from Southern Spain. Sci. Total Environ. 2011, 409, 3281–3287. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Espinosa, M.J.; Vizcaino, E.; Murcia, M.; Llop, S.; Espada, M.; Seco, V.; Marco, A.; Rebagliato, M.; Grimalt, J.O.; Ballester, F. Association between thyroid hormone levels and 4,4′-DDE concentrations in pregnant women (Valencia, Spain). Environ. Res. 2009, 109, 479–485. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Mariano, J.A.; Torres-Sanchez, L.; Bassol-Mayagoitia, S.; Escamilla-Nunez, M.C.; Cebrian, M.E.; Villeda-Gutierrez, E.A.; Lopez-Rodriguez, G.; Felix-Arellano, E.E.; Blanco-Munoz, J. Effect of exposure to p,p-DDE during the first half of pregnancy in the maternal thyroid profile of female residents in a Mexican floriculture area. Environ. Res. 2017, 156, 597–604. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.E.; Choi, K. Perfluoroalkyl substances exposure and thyroid hormones in humans: Epidemiological observations and implications. Ann. Pediatr. Endocrinol. Metab. 2017, 22, 6–14. [Google Scholar] [CrossRef] [PubMed]
- Webster, G.M.; Venners, S.A.; Mattman, A.; Martin, J.W. Associations between Perfluoroalkyl acids (PFASs) and maternal thyroid hormones in early pregnancy: A population-based cohort study. Environ. Res. 2014, 133, 338–347. [Google Scholar] [CrossRef] [PubMed]
- Berg, V.; Nost, T.H.; Hansen, S.; Elverland, A.; Veyhe, A.S.; Jorde, R.; Odland, J.O.; Sandanger, T.M. Assessing the relationship between perfluoroalkyl substances, thyroid hormones and binding proteins in pregnant women; a longitudinal mixed effects approach. Environ. Int. 2015, 77, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Ballesteros, V.; Costa, O.; Iniguez, C.; Fletcher, T.; Ballester, F.; Lopez-Espinosa, M.J. Exposure to perfluoroalkyl substances and thyroid function in pregnant women and children: A systematic review of epidemiologic studies. Environ. Int. 2017, 99, 15–28. [Google Scholar] [CrossRef] [PubMed]
- Ji, C.; Jin, X.; He, J.Y.; Yin, Z. Use of TSH beta: EGFP transgenic zebrafish as a rapid in vivo model for assessing thyroid-disrupting chemicals. Toxicol. Appl. Pharmacol. 2012, 262, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Terrien, X.; Fini, J.B.; Demeneix, B.A.; Schramm, K.W.; Prunet, P. Generation of fluorescent zebrafish to study endocrine disruption and potential crosstalk between thyroid hormone and corticosteroids. Aquat. Toxicol. 2011, 105, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Mengeling, B.J.; Wei, Y.; Dobrawa, L.N.; Streekstra, M.; Louisse, J.; Singh, V.; Singh, L.; Lein, P.J.; Wulff, H.; Murk, A.J.; Furlow, J.D. A multi-tiered, in vivo, quantitative assay suite for environmental disruptors of thyroid hormone signaling. Aquat. Toxicol. 2017, 190, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Leusch, F.D.L.; Aneck-Hahn, N.H.; Cavanagh, J.E.; Du Pasquier, D.; Hamers, T.; Hebert, A.; Neale, P.A.; Scheurer, M.; Simmons, S.O.; Schriks, M. Comparison of in vitro and in vivo bioassays to measure thyroid hormone disrupting activity in water extracts. Chemosphere 2017, 191, 868–875. [Google Scholar] [CrossRef] [PubMed]
| Compound | Aim of the Study | Result | References |
|---|---|---|---|
| Polychlorinated Biphenyls | Evaluated the maternal exposure to 26 PCBs (and dioxin) in maternal plasma and umbilical cord plasma during the last month of pregnancy, in umbilical cord plasma and in human milk, and the relationship with thyroid hormones. | ↑PCB levels in human milk correlated significantly with ↓plasma levels of maternalTT3 and TT4. ↑plasma levels of TSH in the babies in the 2nd week and 3rd month. Infants exposed to ↑toxic doses had ↓plasma FT4 and TT4 in the 2nd week after birth. | [11] |
| Evaluated the correlation between several PCBs in maternal blood during pregnancy and T4, T4S, T3, rT3, TSH and TBG levels in cord blood/serum at three- and 18-month-old babies. | Positive correlation between 3 PCBs and T3 (cord serum). Negative correlation between 4 PCBs and rT3 (cord serum). After correction, 2 PCBs and the sum of the 10 PCBs showed positive correlation with the cord serum T3/rT3 ratio. No correlations between PCBs and T4, TSH and TBG in cord blood. Positive correlation between 4-OH-PCB-107 and T4 at 3 months and T4, T4S and T3 at 18 months. | [13] | |
| Evaluated the associations between maternal blood levels of 3 PCBs congeners and thyroid hormones in maternal and umbilical-cord blood in pregnant women in the first trimester of pregnancy. Thyroid hormone levels also assessed at delivery and in cord blood in 260 subjects. | At delivery, negative associations between maternal FT3 and PCBs. | [14] | |
| Analysed the relationship between serum concentrations of PCBs, levels of thyroid hormones and the mRNA levels of seven TH-regulated genes in peripheral blood leukocytes of e-waste recycling workers. | No associations of TH and PCBs. TH-regulated gene expression was associated with some PCBs and hydroxylated PCB congeners. | [15] | |
| Polybrominated Diphenyl Ethers | Evaluated the associations between levels in maternal blood of PBDEs and levels of thyroid hormones in maternal and umbilical-cord blood in a 380 pregnant women in the 1st trimester of pregnancy. Thyroid hormone levels also assessed at delivery and in cord blood in 260 subjects. | Before 20 weeks of pregnancy, inverse association between maternal PBDEs and total T3 and total T4 and a direct association with free T3 and free T4 were observed. At delivery, negative associations between maternal T T4, FT3, cord-blood FT4, and PBDEs. | [14] |
| Analyzed the relationship between serum concentrations of PBDEs, thyroid hormones TH and mRNA levels of seven TH-regulated genes in peripheral blood leukocytes of e-waste recycling workers. | ↑T4 and T3 levels associated with some lower-brominated BDEs. Negative association between highly brominated PBDE and TSH levels. The expression of most target genes was suppressed by PBDEs (mostly highly brominated congeners). | [15] | |
| Correlation between levels of PBDEs in maternal and foetal serum with thyroid hormones in an Indiana population. | No correlation between the PBDEs and thyroid hormone levels. | [18] | |
| Relationship between PBDEs congeners exposure and thyroid hormones in a population electronic recycling facility workers. | No relationship between exposure to PBDEs and thyroid hormones (small cohort). | [19] | |
| Relationship between maternal PBDE levels and thyroid hormone levels in maternal and cord sera. | ↑T3 and T4 associated with levels of PBE-28 -47 in 2nd trimester of pregnancy. Significant trend between maternal levels of TT4 and PBE7 in the 3rd trimester. No association between maternal PBDE levels and thyroid hormones levels in cord serum. | [20] | |
| Measured PBDEs and metabolites women in late pregnancy phases. Further objective was the potential association between PBDEs and maternal thyroid hormones. | Positive association between BDEs-47, -99, and -10 and increased levels of FT4 and TT3. | [21] | |
| Quantified the partitioning of selected PBDEs from mother to foetus and evaluate the effect of PBDE exposure on maternal THs levels. | Significant difference between mother’s serum levels of low brominated PBDEs and the cord blood. Significant correlation between one PBDE and maternal serum T4. | [22] | |
| Perchlorate | Evaluated the effect of NIS inhibitors on the thyroid function, and identify the sub-population at higher risk for thyroid disruption; 3151 subjects, 12–80 years. | Adolescents are the most sensitive to the action of NIS inhibitors. | [25] |
| Evaluated the relationship between urinary levels of perchlorate and serum levels of TSH and T4 in a population from the National Health and Nutrition Examination Survey (2001–2002). | Negative association between perchlorate in the urine samples and TT4, and a positive association with TSH, only in women, especially in women with low urinary iodine concentration. | [26] | |
| Evaluated the association between urinary perchlorate and serum FT4 in individuals with ↓urinary iodine levels and pregnant women | Urinary perchlorate is predictor of FT4 level only in non-pregnant women. | [27] | |
| Evaluated the impact of maternal perchlorate in the first trimester of pregnancy, in hypothyroid/hypothyroxinemic pregnant women. | Significant association with reduced Intelligence Quotient in the offsprings, in the lower 10th percentile in the offspring of mothers with the highest perchlorate levels. | [28] | |
| Bisphenol-A phthalates | Evaluated relationship between urinary concentration of phthalates metabolites and FT3, FT4 and TSH. | Inverse association between the urinary mono(2-ethylhexyl) phthalate levels and serum T3 levels. | [30] |
| Evaluated the relationship between urine concentration of different phthalates and thyroid hormones in children. | In boys, no association was found. In girls, significant negative association between T3 and phthalate metabolites. All cohort: significant negative association between phthalate metabolites and TT3 and FT3. | [31] | |
| Evaluated the association between phthalate and BP-A exposure and thyroid hormone levels in the serum. | Association between phthalate metabolites concentration in the urine samples with ↓T4 or T3 or ↑TSH. Negative association between urinary bisphenol-A and TSH. | [32] | |
| Evaluated the association between urinary BPA concentrations and plasma thyroid hormone during pregnancy. | Inverse association between urinary concentration of BPA and TSH in pregnant women. | [33] | |
| Evaluated the association between thyroid nodular disease and BP-A and -F. | In the cohort of adult women evaluated, urinary concentration of BPA and serum TSH value were significantly positively associated, but no association was found with the nodular disease. | [34] | |
| Evaluated the association between BPA in cord blood and TSH and FT4 at 3 and 7 days of age. | No association between BPA levels and TSH and FT4 was found in this cohort. | [35] | |
| Evaluation the relationship between BPA concentration in urine and serum and urinary iodine levels in subject with NG and PTC, and HC. | UBC were higher in the NG and PTC groups compared with HC. UIC were higher in the NG and PTC groups compared with HC. Significant correlation between UBC and UIC in the groups. | [36] | |
| Evaluated the association between urinary BPA thyroid volumes and thyroid nodules in a population of 718 Chinese children (aged 9–11 years). | 99.9% of urinary samples showed presence of BPA. 14% of children had thyroid nodules. Inverse association was found between urinary BPA concentration and both the thyroid volume and the risk of Thyroid nodules. | [37] | |
| Evaluate whether BPA concentration in urine samples of women at 16 and 26 weeks of pregnancy were associated with THs in maternal or cord serum. Eventual differences between boys and girls were evaluated. | No association between maternal urinary BPA concentrations and THs in cord serum; ↓cord TSH in girls was associated with maternal urine levels increased 10 folds. No association was found between maternal and cord serum BPA at 16 weeks and THs. Association was found between BPA maternal serum concentration at 26 weeks and ↓TSH in girls. | [38] |
| Compound | Aim of the Study | Results | Ref. |
|---|---|---|---|
| Pesticides | Evaluated the relationship between 19 different OC pesticides and thyroid hormones, TSH, TPOAb and thyroglobulin (cohort of 303 men and 305 women). | In men, correlation between endosulphan 2 and TT3, inverse correlation between T4 and beta-hexachlorocyclohexane and p,p′-DDT. In women, association of T3 levels and ↑ alpha-chlordane, DDT, endosulphan 2 and methoxychlor; T4 levels positively associated with HCB, heptachlor, DDT | [39] |
| Evaluated relationship between OCPs levels in placenta and TSH in umbilical cord blood. | Endrin was associated with ↑TSH in the cord blood. | [42] | |
| Evaluated the association between thyroid hormone levels and 4,4′-DDE concentrations in pregnant women | Association found between maternal serum concentration of 4.40-DDE and ↑TSH and ↓FT4 | [43] | |
| Evaluate the effect of exposure to p,p′-DDE during the first half of pregnancy in thyroid profile | Significant positive association between p,p′-DDE and T3 levels. | [44] |
| Compound | Aim of the Study | Results | Ref. |
|---|---|---|---|
| Perfluoroalkyl | Evaluate the levels of different PFAS, TSH and TPOAb in the maternal serum of euthyroid pregnant women. | Positive association between TSH and PFASs in TPOAb positive women, plus weak association with ↓FT4. | [46] |
| Evaluate the potential association between thyroid hormones, thyroid binding proteins, TPOAb and different PFASs in three samples of maternal blood in the second trimester of pregnancy and 3 days and 6 weeks after delivery. | Positive association between the compound and TSH levels in women in the highest quartile of PFOS. Women in the highest quartile of PFDA and PFUnDA had reduced ↓ of TT3 and FT3. | [47] | |
| Evaluate the epidemiological studies focussing on the relationship between TSH, T3, T4 and different PFAS in pregnant women or young children. | Positive association between PFHxS and PFOS and TSH levels in maternal blood, Positive association between PFNA and TSH in boys older than 11 year of age. | [48] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Calsolaro, V.; Pasqualetti, G.; Niccolai, F.; Caraccio, N.; Monzani, F. Thyroid Disrupting Chemicals. Int. J. Mol. Sci. 2017, 18, 2583. https://doi.org/10.3390/ijms18122583
Calsolaro V, Pasqualetti G, Niccolai F, Caraccio N, Monzani F. Thyroid Disrupting Chemicals. International Journal of Molecular Sciences. 2017; 18(12):2583. https://doi.org/10.3390/ijms18122583
Chicago/Turabian StyleCalsolaro, Valeria, Giuseppe Pasqualetti, Filippo Niccolai, Nadia Caraccio, and Fabio Monzani. 2017. "Thyroid Disrupting Chemicals" International Journal of Molecular Sciences 18, no. 12: 2583. https://doi.org/10.3390/ijms18122583
APA StyleCalsolaro, V., Pasqualetti, G., Niccolai, F., Caraccio, N., & Monzani, F. (2017). Thyroid Disrupting Chemicals. International Journal of Molecular Sciences, 18(12), 2583. https://doi.org/10.3390/ijms18122583