Gut Fermentation of Dietary Fibres: Physico-Chemistry of Plant Cell Walls and Implications for Health
Abstract
1. Introduction
2. Plant Cell Walls and Their Components-Definition and Physico-Chemical Properties
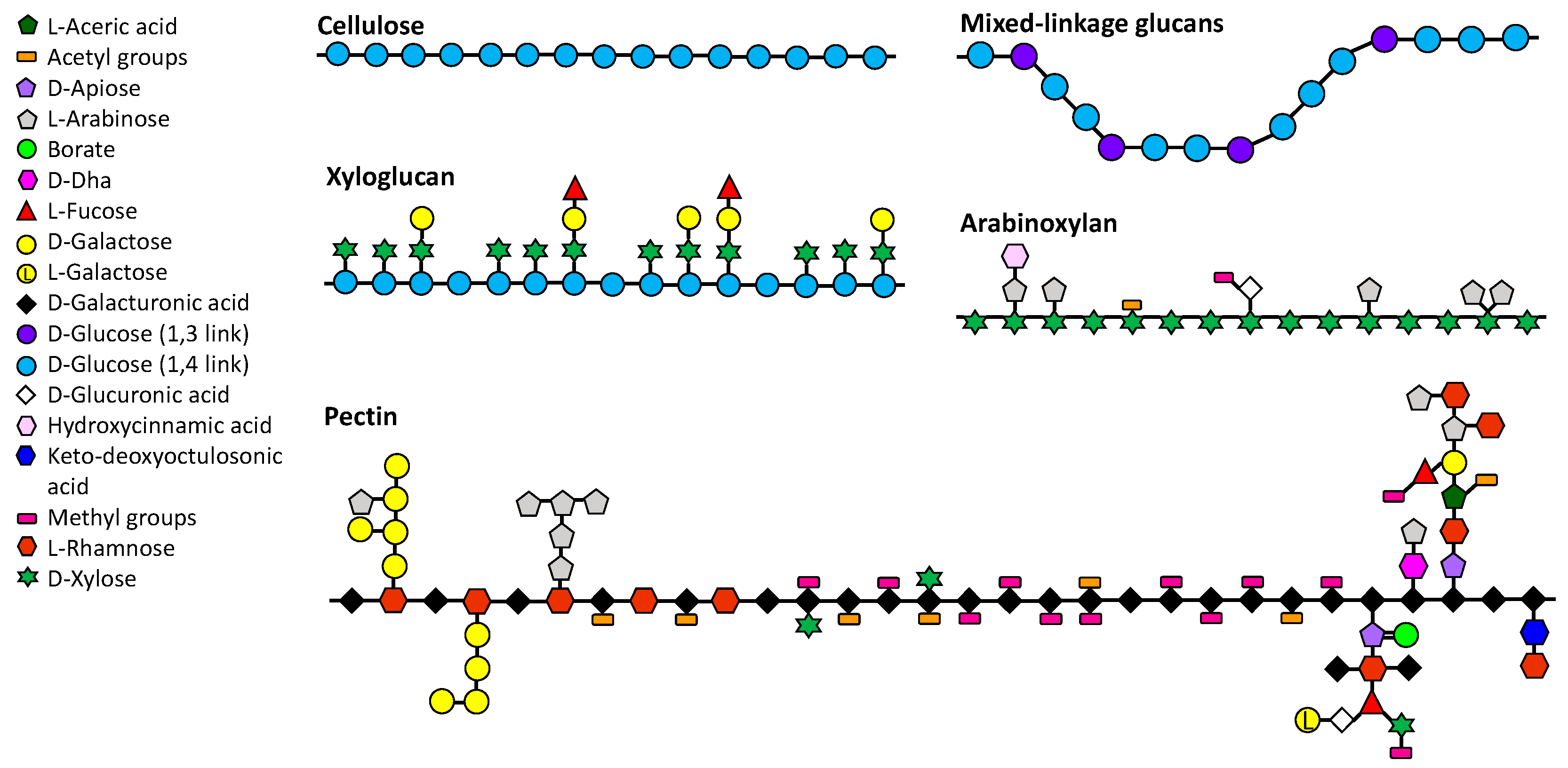
2.1. “Soluble” Dietary Fibres
2.1.1. Pectin
2.1.2. Arabinoxylan
2.1.3. Mixed-Linkage Glucans
2.1.4. Xyloglucans
2.1.5. Others
2.2. “Insoluble” Dietary Fibres
2.2.1. Cellulose
2.2.2. Lignin
2.3. Limitations of Classification by Polymer Type
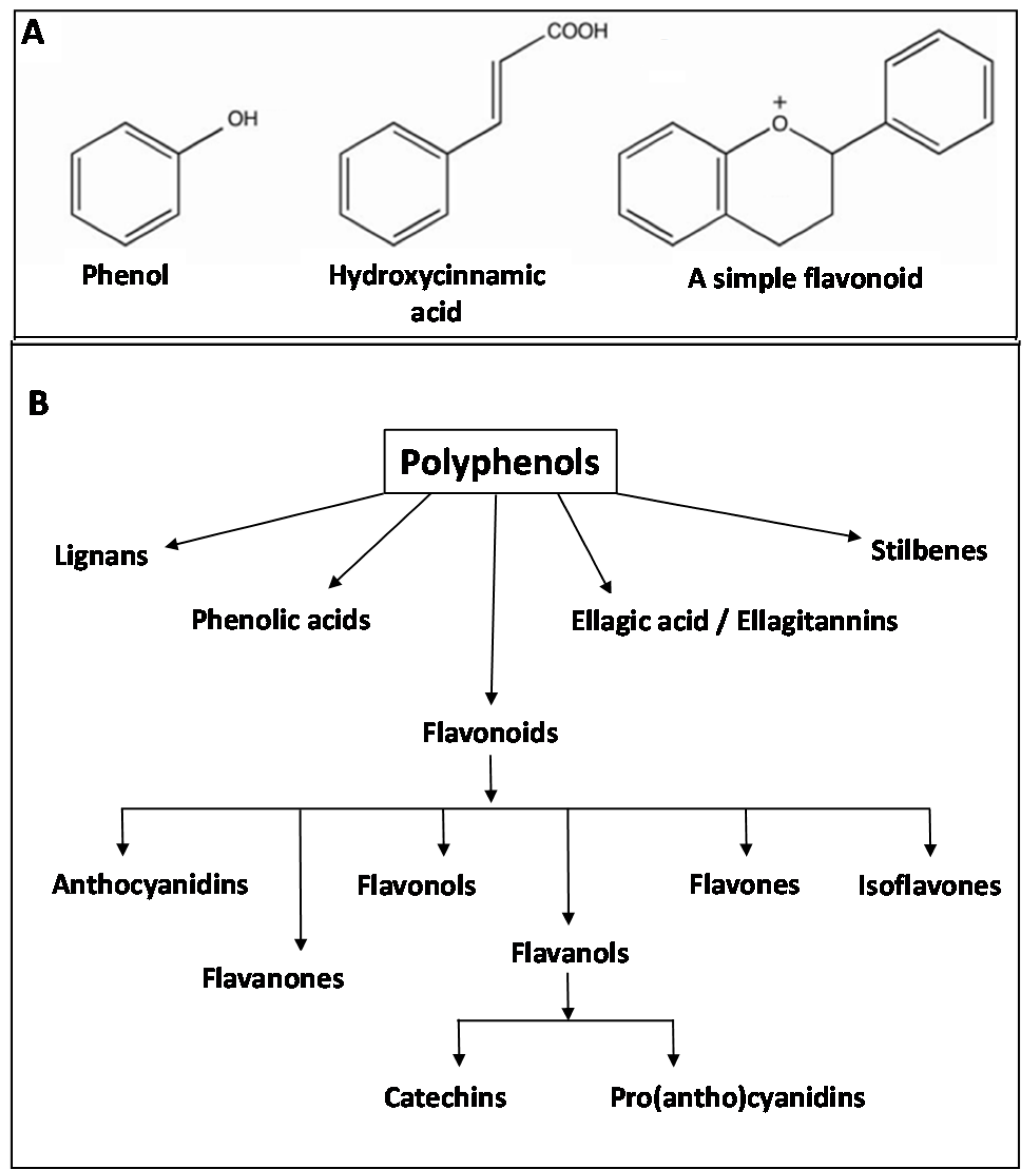
2.4. Phytonutrients from Plants
2.5. Effects of Food Processing on Plant Cell Walls
3. Gastro-Intestinal Tract Fermentation and Effects on Health
3.1. Gastro-Intestinal Tract Microbiota
3.2. Microbial Function
3.2.1. Low Dietary Fibre Diets and Pathogenesis Associated with Microbiota
3.2.2. Diet and microbiota Stability
3.2.3. Diet and Microbial Diversity
3.3. Fermentation End-Products Including Short-Chain Fatty Acids, Ammonia and Others
3.3.1. Carbohydrate Fermentation
3.3.2. Protein Fermentation
3.3.3. Fermentation of Polyphenolic Compounds
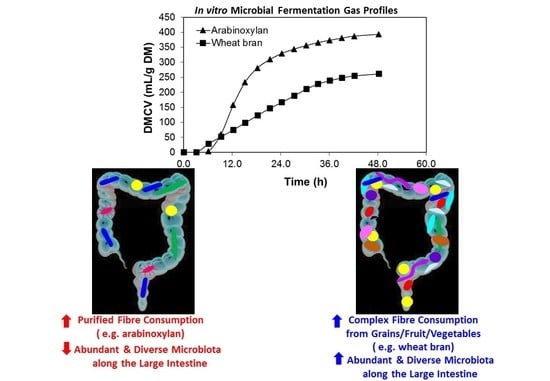
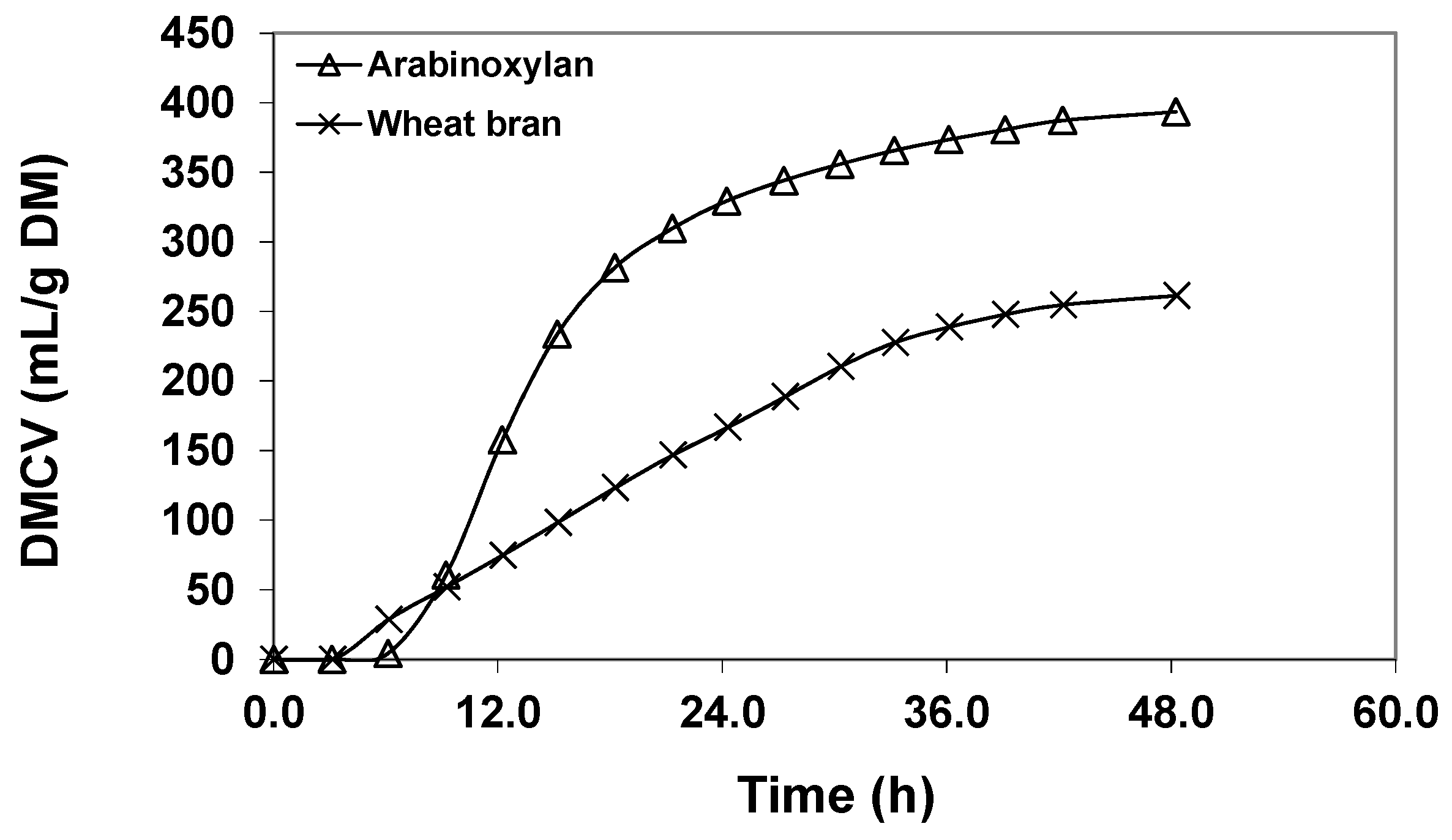
4. Comparing Fermentation of Purified Dietary Fibre (DF) and Whole Plant Foods
4.1. Purified Compounds Affecting GIT Microbiota
4.2. “Purified” Dietary Fibre
4.3. Whole Plant-Based Food Dietary Fibre and Microbiota
4.4. Whole Grains
4.5. Fruits and Vegetables
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- World Health Organisation. Diet, Nutrition and the Prevention of Chronic Diseases. Report of a Joint WHO FAO Expert Consultation; World Health Organisation Technical Report Series; World Health Organisation: Geneva, Switzerland, 2003; Volume 916, pp. 1–149. [Google Scholar]
- World Cancer Research Fund and American Institute for Cancer Research. Policy and Action for Cancer Prevention. In Food, Nutrition, and Physical Activity: A Global Perspective; Research, A.I.C.R: Washington, DC, USA, 2009. [Google Scholar]
- Astorg, P.; Boutron-Ruoult, M.C.; Andrieux, C.; Blachier, F.; Blottiere, H.; Bonithon-Kopp, C.; Boutron-Ruault, M.C.; Cassand, P.; Chaumontet, C.; Cherbut, C.; et al. Dietary fibers and colorectal cancer. Experimental studies, epidemiology, mechanisms. Gastroenterol. Clin. Biol. 2002, 26, 893–912. [Google Scholar] [PubMed]
- Bazzano, L.A.; He, J.; Ogden, L.G.; Loria, C.M.; Vupputuri, S.; Myers, L.; Whelton, P.K. Fruit and vegetable intake and risk of cardiovascular disease in US adults: The first National Health and Nutrition Examination Survey Epidemiologic Follow-up Study. Am. J. Clin. Nutr. 2002, 76, 93–99. [Google Scholar] [PubMed]
- Chuang, S.-C.; Norat, T.; Murphy, N.; Olsen, A.; Tjønneland, A.; Overvad, K.; Boutron-Ruault, M.C.; Perquier, F.; Dartois, L.; Kaaks, R.; et al. Fiber intake and total and cause-specific mortality in the European Prospective Investigation into Cancer and Nutrition cohort. Am. J. Clin. Nutr. 2012, 96, 164–174. [Google Scholar] [CrossRef] [PubMed]
- De Leeuw, J.A.; Jongbloed, A.W.; Verstegen, M.W.A. Dietary fiber stabilizes blood glucose and insulin levels and reduces physical activity in sows (Sus scrofa). J. Nutr. 2004, 134, 1481–1486. [Google Scholar] [PubMed]
- Jenkins, D.J.A.; Kendall, C.W.C.; Vuksan, V.; Augustin, L.S.A.; Li, Y.M.; Lee, B.; Mehling, C.C.; Parker, T.; Faulkner, D.; Seyler, H.; et al. The effect of wheat bran particle size on laxation and colonic fermentation. J. Am. Coll. Nutr. 1999, 18, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Deschrijver, R.; Fremaut, D.; Verheyen, A. Cholesterol-lowering effects and utilization of protein, lipid, fiber and energy in rats fed unprocessed and baked oat bran. J. Nutr. 1992, 122, 1318–1324. [Google Scholar]
- Xu, C.; Lv, J.; Lo, Y.M.; Cui, S.W.; Hu, X.; Fan, M. Effects of oat beta-glucan on endurance exercise and its anti-fatigue properties in trained rats. Carbohydr. Polym. 2013, 92, 1159–1165. [Google Scholar] [CrossRef] [PubMed]
- DeVries, J.W. Validating official methodology commensurate with dietary fibre research and definitions. In Dietary Fibre New Frontiers for Food and Health; Van der Kamp, J.W., Jones, J., McCleary, B., Topping, D., Eds.; Wageningen Academic Publishers: Wageningen, The Netherlands, 2009; pp. 29–48. [Google Scholar]
- Eckburg, P.B.; Bik, E.M.; Bernstein, C.N.; Purdom, E.; Dethlefsen, L.; Sargent, M.; Gill, S.R.; Nelson, K.E.; Relman, D.A. Diversity of the human intestinal microbial flora. Science 2005, 308, 1635–1638. [Google Scholar] [CrossRef] [PubMed]
- Lupton, J.R. Codex definition of dietary fibre and issues requiring resolution. In Dietary Fibre New Frontiers for Food and Health; Van der Kamp, J.W., Jones, J., McCleary, B., Topping, D., Eds.; Wageningen Academic Publishers: Wageningen, The Netherlands, 2009; pp. 15–24. [Google Scholar]
- Sims, I.M.; Monro, J.A. Fiber: Composition, structures, and functional properties. Adv. Food Nutr. Res. 2013, 68, 81–99. [Google Scholar] [CrossRef] [PubMed]
- Brownlee, I.A. The physiological roles of dietary fibre. Food Hydrocolloids 2011, 25, 238–250. [Google Scholar] [CrossRef]
- Leclercq, S.; Matamoros, S.; Cani, P.D.; Francois, J.; Starkal, P.; Windey, K.; Tremaroli, V.; Backhed, F.; Verbeke, K.; de Timary, P.; et al. Increased intestinal permeability is related to bacterial dysbiosis and behavioral markers of addiction severity in alcohol-dependent subjects. Alcohol Alcohol. 2014, 49, i33. [Google Scholar] [CrossRef][Green Version]
- Wu, H.; Tremaroli, V.; Backhed, F. Linking Microbiota to Human Diseases: A Systems Biology Perspective. Trends Endocrinol. Metab. 2015, 26, 758–770. [Google Scholar] [CrossRef] [PubMed]
- Leclercq, S.; Delzenne, N.; Cani, P.; Matamoros, S.; Starkel, P.; Backhed, F.; Tremaroli, V.; Verbeke, K.; de Timary, P. A possible role of gut microbiota in the behavioral control of alcohol-dependent subjects. Alcohol. Clin. Exp. Res. 2014, 38, 176A. [Google Scholar]
- Larsson, E.; Tremaroli, V.; Lee, Y.S.; Koren, O.; Nookaew, I.; Fricker, A.; Nielsen, J.; Ley, R.E.; Backhed, F. Analysis of gut microbial regulation of host gene expression along the length of the gut and regulation of gut microbial ecology through MyD88. Gut 2012, 61, 1124–1131. [Google Scholar] [CrossRef] [PubMed]
- Mudgil, D.; Barak, S. Composition, properties and health benefits of indigestible carbohydrate polymers as dietary fiber: A review. Int. J. Biol. Macromol. 2013, 61, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Chawla, R.; Patil, G.R. Soluble Dietary Fiber. Compr. Rev. Food Sci. Food Saf. 2010, 9, 178–196. [Google Scholar] [CrossRef]
- Maathuis, A.; Hoffman, A.; Evans, A.; Sanders, L.; Venema, K. The Effect of the Undigested Fraction of Maize Products on the Activity and Composition of the Microbiota Determined in a Dynamicin VitroModel of the Human Proximal Large Intestine. J. Am. Coll. Nutr. 2009, 28, 657–666. [Google Scholar] [CrossRef] [PubMed]
- Phillips, G.O. Dietary fibre: A chemical category or a health ingredient? Bioact. Carbohydr. Diet. Fibre 2013, 1, 3–9. [Google Scholar] [CrossRef]
- Williams, B.A.; Oostdam, A.J.; Groot, J.C.J.; Boer, H.; Tamminga, S. Effects of ageing on the in vitro fermentation of cell walls and cell contents of entire, fractionated and composite leaves of Italian ryegrass. J. Sci. Food Agric. 2000, 80, 484–490. [Google Scholar] [CrossRef]
- Dhital, S.; Dolan, G.; Stokes, J.R.; Gidley, M.J. Enzymatic hydrolysis of starch in the presence of cereal soluble fibre polysaccharides. Food Funct. 2014, 5, 579–586. [Google Scholar] [CrossRef] [PubMed]
- Lattimer, J.M.; Haub, M.D. Effects of Dietary Fiber and Its Components on Metabolic Health. Nutrients 2010, 2, 1266–1289. [Google Scholar] [CrossRef] [PubMed]
- Abuajah, C.I.; Ogbonna, A.C.; Osuji, C.M. Functional components and medicinal properties of food: A review. J. Food Sci. Technol. Mysore 2015, 52, 2522–2529. [Google Scholar] [CrossRef] [PubMed]
- Mohnen, D. Pectin structure and biosynthesis. Curr. Opin. Plant Biol. 2008, 11, 266–277. [Google Scholar] [CrossRef] [PubMed]
- Yapo, B.M.; Lerouge, P.; Thibault, J.-F.; Ralet, M.-C. Pectins from citrus peel cell walls contain homogalacturonans homogenous with respect to molar mass, rhamnogalacturonan I and rhamnogalacturonan II. Carbohydr. Polym. 2007, 69, 426–435. [Google Scholar] [CrossRef]
- Morris, G.A.; Foster, T.J.; Harding, S.E. The effect of the degree of esterification on the hydrodynamic properties of citrus pectin. Food Hydrocoll. 2000, 14, 227–235. [Google Scholar] [CrossRef]
- Ridley, B.L.; O’Neill, M.A.; Mohnen, D. Pectins: Structure, biosynthesis, and oligogalacturonide-related signaling. Phytochemistry 2001, 57, 929–967. [Google Scholar] [CrossRef]
- Ndeh, D.; Rogowski, A.; Cartmell, A.; Luis, A.S.; Baslé, A.; Gray, J.; Venditto, I.; Briggs, J.; Zhang, X.; Labourel, A.; et al. Complex pectin metabolism by gut bacteria reveals novel catalytic functions. Nature 2017, 544, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Shewry, P.R.; Saulnier, L.; Guillon, F.; Gebruers, K.; Courtin, C.; Delcour, J.; Toole, G.; Boros, D.; Salse, J.; Ravel, C.; et al. Improving the benefits of wheat as a source of dietary fibre. In Dietary Fibre New Frontiers for Food and Health; Van der Kamp, J.W., Jones, J., McCleary, B., Topping, D., Eds.; Wageningen Academic Publishers: Wageningen, The Netherlands, 2009; pp. 65–78. [Google Scholar]
- Williams, B.A.; Mikkelsen, D.; le Paih, L.; Gidley, M.J. In vitro fermentation kinetics and end-products of cereal arabinoxylans and (1,3;1,4)-β-glucans by porcine faeces. J. Cereal Sci. 2011, 53, 53–58. [Google Scholar] [CrossRef]
- Williams, B.A.; Zhang, D.; Lisle, A.; Mikkelsen, D.; McSweeney, C.; Kang, S.; Bryden, W.L.; Gidely, M.J. Soluble arabinoxylan enhances large intestinal microbial health biomarkers in pigs fed a red meat-containing diet. Nutrition 2016, 32, 491–497. [Google Scholar] [CrossRef] [PubMed]
- Glitso, L.V.; Jensen, B.B.; Knudsen, K.E.B. In vitro fermentation of rye carbohydrates including arabinoxylans of different structure. J. Sci. Food Agric. 2000, 80, 1211–1218. [Google Scholar] [CrossRef]
- Rogowski, A.; Briggs, J.A.; Mortimer, J.C.; Tryfona, T.; Terrapon, N.; Lowe, E.C.; Baslé, A.; Morland, C.; Day, A.M.; Zheng, H.; et al. Glycan complexity dictates microbial resource allocation in the large intestine. Nat. Commun. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Lazaridou, A.; Biliaderis, C.G. Molecular aspects of cereal β-glucan functionality: Physical properties, technological applications and physiological effects. J. Cereal Sci. 2007, 46, 101–118. [Google Scholar] [CrossRef]
- Tosh, S.M.; Brummer, Y.; Wolever, T.M.S.; Wood, P.J. Glycemic response to oat bran muffins treated to vary molecular weight of beta-glucan. Cereal Chem. 2008, 85, 211–217. [Google Scholar] [CrossRef]
- Wood, P.J. Oat and rye beta-glucan: Properties and function. Cereal Chem. 2010, 87, 315–330. [Google Scholar] [CrossRef]
- Gunness, P.; Michiels, J.; Vanhaecke, L.; De Smet, S.; Kravchuk, O.; Van de Meene, A.; Gidley, M.J. Reduction in circulating bile acid and restricted diffusion across the intestinal epithelium are associated with a decrease in blood cholesterol in the presence of oat beta-glucan. FASEB J. 2016, 30, 4227–4238. [Google Scholar] [CrossRef] [PubMed]
- Hughes, S.A.; Shewry, P.R.; Gibson, G.R.; McCleary, B.V.; Rastall, R.A. In vitro fermentation of oat and barley derived β-glucans by human faecal microbiota. FEMS Microbiol. Ecol. 2008, 64, 482–493. [Google Scholar] [CrossRef] [PubMed]
- Bauer, E.; Williams, B.A.; Voigt, C.; Mosenthin, R.; Verstegen, M.W.A. Microbial activities of faeces from unweaned and adult pigs, in relation to selected fermentable carbohydrates. Anim. Sci. 2001, 73, 313–322. [Google Scholar] [CrossRef]
- Drzikova, B.; Dongowski, G.; Gebhardt, E. Dietary fibre-rich oat-based products affect serum lipids, microbiota, formation of short-chain fatty acids and steroids in rats. Br. J. Nutr. 2005, 94, 1012–1025. [Google Scholar] [CrossRef] [PubMed]
- Johansen, H.N.; Knudsen, K.E.B.; Wood, P.J.; Fulcher, R.G. Physico-chemical properties and the degradation of oat bran polysaccharides in the gut of pigs. J. Sci. Food Agric. 1997, 73, 81–92. [Google Scholar] [CrossRef]
- Knudsen, K.E.B.; Canibe, N. Breakdown of plant carbohydrates in the digestive tract of pigs fed on wheat- or oat-based rolls. J. Sci. Food Agric. 2000, 80, 1253–1261. [Google Scholar] [CrossRef]
- Jha, R.; Rossnagel, B.; Pieper, R.; Van Kessel, A.; Leterme, P. Barley and oat cultivars with diverse carbohydrate composition alter ileal and total tract nutrient digestibility and fermentation metabolites in weaned piglets. Animal 2010, 4, 724–731. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, U.; Johansson, M.; Nilsson, A.; Bjoerck, I.; Nyman, M. Dietary supplementation with beta-glucan enriched oat bran increases faecal concentration of carboxylic acids in healthy subjects. Eur. J. Clin. Nutr. 2008, 62, 978–984. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, M.; Jia, Z.; Pena, M.J.; Cash, M.; Harper, A.; Blackburn, A.R., 2nd; Darvill, A.; York, W.S. Structural analysis of xyloglucans in the primary cell walls of plants in the subclass Asteridae. Carbohydr. Res. 2005, 340, 1826–1840. [Google Scholar] [CrossRef] [PubMed]
- Larsbrink, J.; Rogers, T.E.; Hemsworth, G.R.; McKee, L.S.; Tauzin, A.S.; Spadiut, O.; Klinter, S.; Pudlo, N.A.; Urs, K.; Koropatkin, N.M.; et al. A discrete genetic locus confers xyloglucan metabolism in select human gut Bacteroidetes. Nature 2014, 506, 498–502. [Google Scholar] [CrossRef] [PubMed]
- Burton, R.A.; Gidley, M.J.; Fincher, G.B. Heterogeneity in the chemistry, structure and function of plant cell walls. Nat. Chem. Biol. 2010, 6, 724–732. [Google Scholar] [CrossRef] [PubMed]
- Cockburn, D.W.; Koropatkin, N.M. Polysaccharide degradation by the intestinal microbiota and its influence on human health and disease. J. Mol. Biol. 2014, 428, 3230–3252. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Sungryul, Y. Discovery of cellulose as a smart material. Macromolecules 2006, 39, 4202–4206. [Google Scholar] [CrossRef]
- McCann, M.C.; Wells, B.; Roberts, K. Direct visualization of cross-links in the primary plant cell wall. J. Cereal Sci. 1990, 96, 323–334. [Google Scholar]
- Sjogren, K.; Engdahl, C.; Henning, P.; Lerner, U.H.; Tremaroli, V.; Lagerquist, M.K.; Backhed, F.; Ohlsson, C. The gut microbiota regulates bone mass in mice. J. Bone Min. Res. 2012, 27, 1357–1367. [Google Scholar] [CrossRef] [PubMed]
- Grundy, M.M.; Edwards, C.H.; Mackie, A.R.; Gidley, M.J.; Butterworth, P.J.; Ellis, P.R. Re-evaluation of the mechanisms of dietary fibre and implications for macronutrient bioaccessibility, digestion and postprandial metabolism. Br. J. Nutr. 2016, 116, 816–833. [Google Scholar] [CrossRef] [PubMed]
- Dhingra, D.; Michael, M.; Rajput, H.; Patil, R.T. Dietary fibre in foods: A review. J. Food Sci. Technol. 2012, 49, 255–266. [Google Scholar] [CrossRef] [PubMed]
- Dehority, B.A. Rumen Microbiology; Nottingham University Press: Nottingham, UK, 2003. [Google Scholar]
- Castillo-González, A.R.; Burrola-Barraza, M.E.; Domínguez-Viveros, J.; Chávez-Martínez, A. Rumen microorganisms and fermentation. Arch. Med. Vet. 2014, 46, 349–361. [Google Scholar] [CrossRef]
- Yan, H.; Potu, R.; Lu, H.; de Almeida, V.V.; Stewart, T.; Ragland, D.; Armstrong, A.; Adeola, O.; Nakatsu, C.H.; Ajuwon, K.M.; et al. Dietary Fat Content and Fiber Type Modulate Hind Gut Microbial Community and Metabolic Markers in the Pig. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Alberts, B.; Johnson, A.; Lewis, J.; Roberts, K.; Raff, M.; Walter, P. Cell junctions, cell adhesion and the extra-cellular matrix. In Molecular Biology of the Cell, 4th ed.; Garland Science: New York, NY, USA, 2002. [Google Scholar]
- Neutelings, G. Lignin variability in plant cell walls: Contribution of new models. Plant Sci. 2011, 181, 379–386. [Google Scholar] [CrossRef] [PubMed]
- Marlett, J.A.; Cheung, T.F. Database and quick methods of assessing typical dietary fiber intakes using data for 228 commonly consumed foods. J. Am. Diet. Assoc. 1997, 97, 1139–1148. [Google Scholar] [CrossRef]
- Acosta-Estrada, B.A.; Gutierrez-Uribe, J.A.; Serna-Saldivar, S.O. Bound phenolics in foods, a review. Food Chem. 2014, 152, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Antolovich, M.; Prenzler, P.; Robards, K.; Ryan, D. Sample preparation in the determination of phenolic compounds in fruits. Analyst 2000, 125, 989–1009. [Google Scholar] [CrossRef]
- Neveu, V.; Perez-Jimenez, J.; Vos, F.; Crespy, V.; du Chaffaut, L.; Mennen, L.; Knox, C.; Eisner, R.; Cruz, J.; Wishart, D.; et al. Phenol-Explorer: An online comprehensive database on polyphenol contents in foods. Database J. Biol. Databases Curation 2010, 2010. [Google Scholar] [CrossRef] [PubMed]
- Selma, M.V.; Espin, J.C.; Tomas-Barberan, F.A. Interaction between phenolics and gut microbiota: Role in human health. J. Agric. Food Chem. 2009, 57, 6485–6501. [Google Scholar] [CrossRef] [PubMed]
- Cardona, F.; Andres-Lacueva, C.; Tulipani, S.; Tinahones, F.J.; Queipo-Ortuno, M.I. Benefits of polyphenols on gut microbiota and implications in human health. J. Nutr. Biochem. 2013, 24, 1415–1422. [Google Scholar] [CrossRef] [PubMed]
- Khoddami, A.; Wilkes, M.A.; Roberts, T.H. Techniques for analysis of plant phenolic compounds. Molecules 2013, 18, 2328–2375. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Holvoet, S.; Mercenier, A. Dietary polyphenols in the prevention and treatment of allergic diseases. Clin. Exp. Allergy 2011, 41, 1346–1359. [Google Scholar] [CrossRef] [PubMed]
- Bingham, M. The metabolism of dietary polyphenols by the human gut microbiota. In Gastrointestinal Microbiology; Ouwehand, A.C., Vaughan, E.E., Eds.; Taylor & Francis Group: New York, NY, USA, 2006; pp. 155–168. [Google Scholar]
- Padayachee, A.; Day, L.; Howell, K.; Gidley, M.J. Complexity and Health Functionality of Plant Cell Wall Fibres from Fruits and Vegetables. Crit. Rev. Food Sci. Nutr. 2015, 57, 59–81. [Google Scholar] [CrossRef] [PubMed]
- Phan, A.D.T.; Netzel, G.; Wang, D.; Flanagan, B.M.; D’Arcy, B.R.; Gidley, M.J. Binding of dietary polyphenols to cellulose: Structural and nutritional aspects. Food Chem. 2015, 171, 388–396. [Google Scholar] [CrossRef] [PubMed]
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jiménez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [PubMed]
- Wang, D.; Williams, B.A.; Ferruzzi, M.G.; D’Arcy, B.R. Microbial metabolites, but not other phenolics derived from grape seed phenolic extract, are transported through differentiated Caco-2 cell monolayers. Food Chem. 2013, 138, 1564–1573. [Google Scholar] [CrossRef] [PubMed]
- Tuohy, K.M.; Conterno, L.; Gasperotti, M.; Viola, R. Up-regulating the human intestinal microbiome using whole plant foods, polyphenols, and/or fiber. J. Agric. Food Chem. 2012, 60, 8776–8782. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, C.A. Nutrition and health. The issue is not food, nor nutrients, so much as processing. Public Health Nutr. 2009, 12, 729–731. [Google Scholar] [CrossRef] [PubMed]
- Slavin, J.L.; Martini, M.C.; Jacobs, D.R., Jr.; Marquart, L. Plausible mechanisms for the protectiveness of whole grains. Am. J. Clin. Nutr. 1999, 70, 459s–463s. [Google Scholar] [PubMed]
- Liu, R.H. Health benefits of fruit and vegetables are from additive and synergistic combinations of phytochemicals. Am. J. Clin. Nutr. 2003, 78, 517s–520s. [Google Scholar] [PubMed]
- Kiely, E.M.; Ajayi, N.A.; Wheeler, R.A.; Malone, M. Diversion procto-colitis: Response to treatment with short-chain fatty acids. J. Pediatr. Surg. 2001, 36, 1514–1517. [Google Scholar] [CrossRef] [PubMed]
- Hemsworth, G.R.; Thompson, A.J.; Stepper, J.; Sobala, L.F.; Coyle, T.; Larsbrink, J.; Spadiut, O.; Goddard-Borger, E.D.; Stubbs, K.A.; Brumer, H.; et al. Structural dissection of a complex Bacteroides ovatus gene locus conferring xyloglucan metabolism in the human gut. Open Biol. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Martens, E.C.; Kelly, A.G.; Tauzin, A.S.; Brumer, H. The devil lies in the details: How variations in polysaccharide fine-structure impact the physiology and evolution of gut microbes. J. Mol. Biol. 2014, 426, 3851–3865. [Google Scholar] [CrossRef] [PubMed]
- Sommer, F.; Backhed, F. The gut microbiota-masters of host development and physiology. Nat. Rev. Microbiol. 2013, 11, 227–238. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.; McKenzie, C.; Potamitis, M.; Thorburn, A.N.; Mackay, C.R.; Macia, L. The role of short-chain Fatty acids in health and disease. Adv. Immunol. 2014, 121, 91–119. [Google Scholar] [CrossRef] [PubMed]
- Zoetendal, E.G.; Raes, J.; Van Den Bogert, B.; Arumugam, M.; Booijink, C.C.G.M.; Troost, F.J.; Bork, P.; Wels, M.; Vos, W.M.D.; Kleerebezem, M. The human small intestinal microbiota is driven by rapid uptake and conversion of simple carbohydrates. ISME J. 2012, 6, 1415–1426. [Google Scholar] [CrossRef] [PubMed]
- Diaz Heijtz, R.; Wang, S.; Anuar, F.; Qian, Y.; Bjorkholm, B.; Samuelsson, A.; Hibberd, M.L.; Forssberg, H.; Pettersson, S. Normal gut microbiota modulates brain development and behavior. Proc. Natl. Acad. Sci. USA 2011, 108, 3047–3052. [Google Scholar] [CrossRef] [PubMed]
- Maslowski, K.M.; Mackay, C.R. Diet, gut microbiota and immune responses. Nat. Immunol. 2011, 12, 5–9. [Google Scholar] [CrossRef] [PubMed]
- Gong, J.; Yang, C. Advances in the methods for studying gut microbiota and their relevance to the research of dietary fiber functions. Food Res. Int. 2012, 48, 916–929. [Google Scholar] [CrossRef]
- Rajilic-Stojanovic, M. Function of the microbiota. Best Pract. Res. Clin. Gastroenterol. 2013, 27, 5–16. [Google Scholar] [CrossRef] [PubMed]
- Egert, M.; de Graaf, A.A.; Smidt, H.; de Vos, W.M.; Venema, K. Beyond diversity: Functional microbiomics of the human colon. Trends Microbiol. 2006, 14, 86–91. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.D.; Bushmanc, F.D.; Lewis, J.D. Diet, the human gut microbiota, and IBD. Anaerobe 2013, 24, 117–120. [Google Scholar] [CrossRef] [PubMed]
- Peterson, D.A.; Frank, D.N.; Pace, N.R.; Gordon, J.I. Metagenomic approaches for defining the pathogenesis of inflammatory bowel diseases. Cell Host Microbe 2008, 3, 417–427. [Google Scholar] [CrossRef] [PubMed]
- Donaldson, G.P.; Lee, S.M.; Mazmanian, S.K. Gut biogeography of the bacterial microbiota. Nat. Rev. Microbiol. 2016, 14, 20–32. [Google Scholar] [CrossRef] [PubMed]
- Qin, J.; Li, R.; Raes, J.; Arumugam, M.; Burgdorf, K.S.; Manichanh, C.; Nielsen, T.; Pons, N.; Levenez, F.; Yamada, T.; et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 2010, 464, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Sunagawa, S.; Mende, D.R.; Zeller, G.; Izquierdo-Carrasco, F.; Berger, S.A.; Kultima, J.R.; Coelho, L.P.; Arumugam, M.; Tap, J.; Nielsen, H.B.; et al. Metagenomic species profiling using universal phylogenetic marker genes. Nat. Methods 2013, 10, 1196–1199. [Google Scholar] [CrossRef] [PubMed]
- Gevers, D.; Cohan, F.M.; Lawrence, J.G.; Spratt, B.G.; Coenye, T.; Feil, E.J.; Stackebrandt, E.; Van de Peer, Y.; Vandamme, P.; Thompson, F.L.; et al. Opinion: Re-evaluating prokaryotic species. Nat. Rev. Microbiol. 2005, 3, 733–739. [Google Scholar] [CrossRef] [PubMed]
- Sunil, T.; Izard, J.; Walsh, E.; Batich, K.; Chongsathidkiet, P.; Clarke, G.; Sela, D.A.; Muller, A.J.; Mullin, J.M.; Albert, K.; et al. The host microbiome regulates and maintains human health: A primer and perspective for non-microbiologists. Cancer Res. 2017, 77, 1783–1812. [Google Scholar] [CrossRef]
- Marshall, B.J. Helicobacter pylori The Etiologic Agent for Peptic Ulcer. J. Am. Med. Assoc. 1995, 274, 1064–1066. [Google Scholar] [CrossRef]
- Tauzin, A.S.; Laville, E.; Xiao, Y.; Nouaille, S.; Le Bourgeois, P.; Heux, S.; Portais, J.C.; Monsan, P.; Martens, E.C.; Potocki-Veronese, G.; et al. Functional characterization of a gene locus from an uncultured gut Bacteroides conferring xylo-oligosaccharides utilization to Escherichia coli. Mol. Microbiol. 2016, 102, 579–592. [Google Scholar] [CrossRef] [PubMed]
- Croxen, M.A.; Law, R.J.; Scholz, R.; Keeney, K.M.; Wlodarska, M.; Finlay, B.B. Recent Advances in Understanding Enteric Pathogenic Escherichia coli. Clin. Microbiol. Rev. 2013, 26, 822–880. [Google Scholar] [CrossRef] [PubMed]
- Crabtree, J.E.; Taylor, J.D.; Wyatt, J.I.; Heatley, R.V.; Shallcross, T.M.; Tompkins, D.S.; Rathbone, B.J. Mucosal IgA recognition of Helico pylori 120 kDa protein, peptic ulceration, and gastric pathology. Lancet 1991, 338, 332–335. [Google Scholar] [CrossRef]
- Brace, C.; Gloor, G.B.; Ropeleski, M.; Allen-Vercoe, E.; Petrof, E.O. Microbial composition analysis of Clostridium difficile infections in an ulcerative colitis patient treated with multiple fecal microbiota transplantations. J. Crohn’s Colitis 2014, 8, 1133–1137. [Google Scholar] [CrossRef] [PubMed]
- Fujimoto, T.; Imaeda, H.; Takahashi, K.; Kasumi, E.; Bamba, S.; Fujiyama, Y.; Andoh, A. Decreased abundance of Faecalibacterium prausnitzii in the gut microbiota of Crohn’s disease. J. Gastroenterol. Hepatol. 2013, 28, 613–619. [Google Scholar] [CrossRef] [PubMed]
- Backhed, F.; Ley, R.E.; Sonnenburg, J.L.; Peterson, D.A.; Gordon, J.I. Host-bacterial mutualism in the human intestine. Science 2005, 307, 1915–1920. [Google Scholar] [CrossRef] [PubMed]
- Bauer, E.; Williams, B.A.; Verstegen, M.W.A.; Mosenthin, R. Fermentable carbohydrates: Potential dietary modulators of intestinal physiology, microbiology and immunity in pigs. In Biology of Nutrition in Growing Animals; Mosenthin, R.Z.J., Zebrowska, T., Eds.; Elsevier: Edinburgh, UK, 2006; pp. 33–63. [Google Scholar]
- Candela, M.; Maccaferri, S.; Turroni, S.; Carnevali, P.; Brigidi, P. Functional intestinal microbiome, new frontiers in prebiotic design. Int. J. Food Microbiol. 2010, 140, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Butel, M.J. Probiotics, gut microbiota and health. Med. Mal. Infect. 2014, 44, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Jeffery, I.; O’Toole, P. Diet-Microbiota interactions and their implications for healthy living. Nutrients 2013, 5, 234–252. [Google Scholar] [CrossRef] [PubMed]
- Flint, H.J.; Scott, K.P.; Duncan, S.H.; Louis, P.; Forano, E. Microbial degradation of complex carbohydrates in the gut. Gut Microbes 2012, 3, 289–306. [Google Scholar] [CrossRef] [PubMed]
- Hugenholtz, F.; Mullaney, J.A.; Kleerebezem, M.; Smidt, H.; Rosendale, D.I. Modulation of the microbial fermentation in the gut by fermentable carbohydrates. Bioact. Carbohydr. Diet. Fibre 2013, 2, 133–142. [Google Scholar] [CrossRef]
- Salyers, A.A.; Reeves, A.; D’Elia, J. Solving the problem of how to eat something as big as yourself: Diverse bacterial strategies for degrading polysaccharides. J. Ind. Microbiol. 1996, 17, 470–476. [Google Scholar] [CrossRef]
- Macfarlane, G.T.; Gibson, G.R. Carbohydrate fermentation, energy transduction and gas metabolism in the human large intestine. In Gastrointestinal Microbiology; Mackie, R.I., White, B.A., Eds.; Springer: New York, NY, USA, 1997; pp. 269–318. [Google Scholar]
- Williams, B.A.; Verstegen, M.W.; Tamminga, S. Fermentation in the large intestine of single-stomached animals and its relationship to animal health. Nutr. Res. Rev. 2001, 14, 207–228. [Google Scholar] [CrossRef] [PubMed]
- Koecher, K.J.; Thomas, W.; Slavin, J.L. Healthy subjects experience bowel changes on enteral diets: Addition of a fiber blend attenuates stool weight and gut bacteria decreases without changes in gas. J. Parenter. Enter. Nutr. 2015, 39, 337–343. [Google Scholar] [CrossRef] [PubMed]
- Lewis, S.J.; Heaton, K.W. Stool form scale as a useful guide to intestinal transit time. Scand. J. Gastroenterol. 1997, 32, 920–924. [Google Scholar] [CrossRef] [PubMed]
- Slavin, J. Fiber and prebiotics: Mechanisms and health benefits. Nutrients 2013, 5, 1417–1435. [Google Scholar] [CrossRef] [PubMed]
- Brown, K.; DeCoffe, D.; Molcan, E.; Gibson, D.L. Diet-induced dysbiosis of the intestinal microbiota and the effects on immunity and disease. Nutrients 2012, 4, 1095–1119. [Google Scholar] [CrossRef] [PubMed]
- Desai, M.S.; Seekatz, A.M.; Koropatkin, N.M.; Kamada, N.; Hickey, C.A.; Wolter, M.; Pudlo, N.A.; Kitamoto, S.; Terrapon, N.; Muller, A.; et al. A Dietary Fiber-Deprived Gut Microbiota Degrades the Colonic Mucus Barrier and Enhances Pathogen Susceptibility. Cell 2016, 167, 1339–1353. [Google Scholar] [CrossRef] [PubMed]
- Davis, C.D.; Milner, J.A. Gastrointestinal microflora, food components and colon cancer prevention. J. Nutr. Biochem. 2009, 20, 743–752. [Google Scholar] [CrossRef] [PubMed]
- Gorham, J.B. Changes to Gut Bacteria Composition and Diversity by the Addition Of Soluble Dietary Fibres to Porcine Diets: Human Health Implications. Ph.D. Thesis, The University of Queensland, Brisbane, Austria, 2017. [Google Scholar]
- Castillo, M.; Martín-Orúe, S.M.; Anguita, M.; Pérez, J.F.; Gasa, J. Adaptation of gut microbiota to corn physical structure and different types of dietary fibre. Livest. Sci. 2007, 109, 149–152. [Google Scholar] [CrossRef]
- Shanahan, F. The colonic microbiota in health and disease. Curr. Opin. Gastroenterol. 2013, 29, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Lozupone, C.A.; Stombaugh, J.I.; Gordon, J.I.; Jansson, J.K.; Knight, R. Diversity, stability and resilience of the human gut microbiota. Nature 2012, 489, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Salonen, A.; De Vos, W.M. Impact of diet on human intestinal microbiota and health. Annu. Rev. Food Sci. Technol. 2014, 5, 239–262. [Google Scholar] [CrossRef] [PubMed]
- Yatsunenko, T.; Rey, F.E.; Manary, M.J.; Trehan, I.; Dominguez-Bello, M.G.; Contreras, M.; Magris, M.; Hidalgo, G.; Baldassano, R.N.; Anokhin, A.P.; et al. Human gut microbiome viewed across age and geography. Nature 2012, 486, 222–227. [Google Scholar] [CrossRef] [PubMed]
- De Filippo, C.; Cavalieri, D.; Di Paola, M.; Ramazzotti, M.; Poullet, J.B.; Massart, S.; Collini, S.; Pieraccini, G.; Lionetti, P. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc. Natl. Acad. Sci. USA 2010, 107, 14691–14696. [Google Scholar] [CrossRef] [PubMed]
- Ou, J.; Carbonero, F.; Zoetendal, E.G.; DeLany, J.P.; Wang, M.; Newton, K.; Gaskins, H.R.; O’Keefe, S.J. Diet, microbiota, and microbial metabolites in colon cancer risk in rural Africans and African Americans. Am. J. Clin. Nutr. 2013, 98, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Abdallah Ismail, N.; Ragab, S.H.; Abd Elbaky, A.; Shoeib, A.R.; Alhosary, Y.; Fekry, D. Frequency of Firmicutes and Bacteroidetes in gut microbiota in obese and normal weight Egyptian children and adults. Arch. Med. Sci. 2011, 7, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Sokol, H.; Lay, C.; Seksik, P.; Tannock, G.W. Analysis of bacterial bowel communities of IBD patients: What has it revealed? Inflamm. Bowel Dis. 2008, 14, 858–867. [Google Scholar] [CrossRef] [PubMed]
- Andoh, A.; Tsujikawa, T.; Fujiyama, Y. Role of dietary fiber and short-chain fatty acids in the colon. Curr. Pharm. Des. 2003, 9, 347–358. [Google Scholar] [CrossRef] [PubMed]
- Tremaroli, V.; Backhed, F. Functional interactions between the gut microbiota and host metabolism. Nature 2012, 489, 242–249. [Google Scholar] [CrossRef] [PubMed]
- Macfarlane, G.T.; Macfarlane, S. Bacteria, colonic fermentation, and gastrointestinal health. J. AOAC Int. 2012, 95, 50–60. [Google Scholar] [CrossRef] [PubMed]
- Flickinger, E.A.; Van Loo, J.; Fahey, G.C., Jr. Nutritional responses to the presence of inulin and oligofructose in the diets of domesticated animals: A review. Crit. Rev. Food Sci. Nutr. 2003, 43, 19–60. [Google Scholar] [CrossRef] [PubMed]
- Sokol, H.; Pigneur, B.; Watterlot, L.; Lakhdari, O.; Bermudez-Humaran, L.G.; Gratadoux, J.J.; Blugeon, S.; Bridonneau, C.; Furet, J.P.; Corthier, G.; et al. Faecalibacterium prausnitzii is an anti-inflammatory commensal bacterium identified by gut microbiota analysis of Crohn disease patients. Proc. Natl. Acad. Sci. USA 2008, 105, 16731–16736. [Google Scholar] [CrossRef] [PubMed]
- Scheppach, W.; Sommer, H.; Kirchner, T.; Paganelli, G.-M.; Bartram, P.; Christl, S.U.; Richter, F.; DUSEL, G.; Kasper, H. Effect of butyrate enemas on the colonic mucosa in distal ulcerative colitis. Gastroenterology 1992, 103, 51–56. [Google Scholar] [CrossRef]
- Robles Alonso, V.; Guarner, F. Linking the gut microbiota to human health. Br. J. Nutr. 2013, 109, 21s–26s. [Google Scholar] [CrossRef] [PubMed]
- Hijova, E.; Chmelarova, A. Short chain fatty acids and colonic health. Bratisl. Lekarsk. List. 2007, 108, 354–358. [Google Scholar]
- Fava, F.; Lovegrove, J.A.; Tuohy, K.M.; Gibson, G.R. The potential role of the intestinal gut microbiota in obesity and the metabolic syndrome. In Food Science and Technology Bulletin: Functional Foods; IFIS Publishing: Reading, UK, 2009; Volume 5, pp. 71–92. [Google Scholar]
- Wong, J.M.W.; De Souza, R.; Kendall, C.W.C.; Emam, A.; Jenkins, A.J.A. Colonic Health: Fermentation and Short Chain Fatty Acids. J. Clin. Gastroenterol. 2006, 40, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Round, J.L.; Mazmanian, S.K. The gut microbiota shapes intestinal immune responses during health and disease. Nat. Rev. Immunol. 2009, 9, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Louis, P.; Flint, H.J. Formation of propionate and butyrate by the human colonic microbiota. Environ. Microbiol. 2017, 19, 29–41. [Google Scholar] [CrossRef] [PubMed]
- Topping, D.L.; Clifton, P.M. Short-chain fatty acids and human colonic function: Roles of resistant starch and nnstarch polysaccharides. Physiol. Rev. 2001, 81, 1031–1064. [Google Scholar] [PubMed]
- Flint, H.J.; Duncan, S.H.; Scott, K.P.; Louis, P. Interactions and competition within the microbial community of the human colon: Links between diet and health. Environ. Microbiol. 2007, 9, 1101–1111. [Google Scholar] [CrossRef] [PubMed]
- Flint, H.J.; Duncan, S.H.; Scott, K.P.; Louis, P. Links between diet, gut microbiota composition and gut metabolism. Proc. Nutr. Soc. 2015, 74, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Hague, A.; Elder, D.J.; Hicks, D.J.; Paraskeva, C. Apoptosis in colorectal tumour cells: Induction by the short chain fatty acids butyrate, propionate and acetate and by the bile salt deoxycholate. Int. J. Cancer 1995, 60, 400–406. [Google Scholar] [CrossRef] [PubMed]
- Abrahamse, S.L.; Pool-Zobel, B.L.; Rechkemmer, G. Potential of short chain fatty acids to modulate the induction of DNA damage and changes in the intracellular calcium concentration by oxidative stress in isolated rat distal colon cells. Carcinogenesis 1999, 20, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Macfarlane, S.; Macfarlane, G.T. Composition and metabolic activities of bacterial biofilms colonizing food residues in the human gut. Appl. Environ. Microbiol. 2006, 72, 6204–6211. [Google Scholar] [CrossRef] [PubMed]
- Al-Lahham, S.H.; Peppelenbosch, M.P.; Roelofsen, H.; Vonk, R.J.; Venema, K. Biological effects of propionic acid in humans; metabolism, potential applications and underlying mechanisms. Biochim. Biophys. Acta 2010, 1801, 1175–1183. [Google Scholar] [CrossRef] [PubMed]
- Andriamihaja, M.; Chaumontet, C.; Tome, D.; Blachier, F. Butyrate Metabolism in Human Colon Carcinoma Cells: Implications Concerning Its Growth-Inhibitory Effect. J. Cell. Phys. 2009, 218, 58–65. [Google Scholar] [CrossRef] [PubMed]
- Burkitt, D.P. Epidemiology of cancer of the colon and rectum. Nutrition 1988, 4, 201–212. [Google Scholar]
- Castillo Martinez, F.A.; Balciunas, E.M.; Salgado, J.M.; Domínguez González, J.M.; Converti, A.; Oliveira, R.P.d.S. Lactic acid properties, applications and production: A review. Trends Food Sci. Technol. 2013, 30, 70–83. [Google Scholar] [CrossRef]
- Pedersen, M.B.; Gaudu, P.; Lechardeur, D.; Petit, M.A.; Gruss, A. Aerobic respiration metabolism in lactic acid bacteria and uses in biotechnology. Annu. Rev. Food Sci. Technol. 2012, 3, 37–58. [Google Scholar] [CrossRef] [PubMed]
- Hogberg, A.; Lindberg, J.E. The effect of level and type of cereal non-starch polysaccharides on the performance, nutrient utilization and gut environment of pigs around weaning. Anim. Feed Sci. Technol. 2006, 127, 200–219. [Google Scholar] [CrossRef]
- Hou, N.; Huo, D.; Dignam, J.J. Prevention of colorectal cancer and dietary management. Chin. Clin. Oncol. 2013, 2, 13–33. [Google Scholar] [CrossRef] [PubMed]
- Romero-Gomez, M.; Jover, M.; Galan, J.J.; Ruiz, A. Gut ammonia production and its modulation. Metab. Brain Dis. 2009, 24, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Liu, M.; Yang, F.G.; Cui, L.H.; Lu, X.Y.; Chen, C. Dietary fibre and the risk of colorectal cancer: A case-control study. Asian Pac. J. Cancer Prev. 2015, 16, 3747–3752. [Google Scholar] [CrossRef] [PubMed]
- Eid, N.; Walton, G.; Costabile, A.; Kuhnle, G.; Spencer, J. Polyphenols, glucosinolades, dietary fibre and colon cancer: Understanding the potential of specific types of fruit and vegetables to reduce bowel cancer progression. Nutr. Aging 2014, 45–67. [Google Scholar] [CrossRef]
- Crozier, A.; Del Rio, D.; Clifford, M.N. Bioavailability of dietary flavonoids and phenolic compounds. Mol. Asp. Med. 2010, 31, 446–467. [Google Scholar] [CrossRef] [PubMed]
- Aura, A.M.; Martin-Lopez, P.; O’Leary, K.A.; Williamson, G.; Oksman-Caldentey, K.M.; Poutanen, K.; Santos-Buelga, C. In vitro metabolism of anthocyanins by human gut microflora. Eur. J. Nutr. 2005, 44, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Saura-Calixto, F.; Serrano, J.; Goñi, I. Intake and bioaccessibility of total polyphenols in a whole diet. Food Chem. 2007, 101, 492–501. [Google Scholar] [CrossRef]
- Padayachee, A.; Netzel, G.; Netzel, M.; Day, L.; Mikkelsen, D.; Gidley, M.J. Lack of release of bound anthocyanins and phenolic acids from carrot plant cell walls and model composites during simulated gastric and small intestinal digestion. Food Funct. 2013, 4, 906–916. [Google Scholar] [CrossRef] [PubMed]
- Laparra, J.M.; Sanz, Y. Interactions of gut microbiota with functional food components and nutraceuticals. Pharmacol. Res. 2010, 61, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Peterson, J.; Dwyer, J.; Adlercreutz, H.; Scalbert, A.; Jacques, P.; McCullough, M.L. Dietary lignans: Physiology and potential for cardiovascular disease risk reduction. Nutr. Rev. 2010, 68, 571–603. [Google Scholar] [CrossRef] [PubMed]
- Van’t Slot, G.; Humpf, H.U. Degradation and metabolism of catechin, epigallocatechin-3-gallate (EGCG), and related compounds by the intestinal microbiota in the pig cecum model. J. Agric. Food Chem. 2009, 57, 8041–8048. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Patan, F.; Barroso, E.; van de Wiele, T.; Jimenez-Giron, A.; Martin-Alvarez, P.J.; Moreno-Arribas, M.V.; Martinez-Cuesta, M.C.; Pelaez, C.; Requena, T.; Bartolome, B. Comparative in vitro fermentations of cranberry and grape seed polyphenols with colonic microbiota. Food Chem. 2015, 183, 273–282. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Lin, X.; Xue, B.; Luo, J.; Gao, L.; Wang, Y.; Ou, S.; Peng, X. Impact of polyphenols combined with high-fat diet on rats’ gut microbiota. J. Funct. Foods 2016, 26, 763–771. [Google Scholar] [CrossRef]
- Klinder, A.; Shen, Q.; Heppel, S.; Lovegrove, J.A.; Rowland, I.; Tuohy, K.M. Impact of increasing fruit and vegetables and flavonoid intake on the human gut microbiota. Food Funct. 2016, 7, 1788–1796. [Google Scholar] [CrossRef] [PubMed]
- Tagliabue, A.; Elli, M. The role of gut microbiota in human obesity: Recent findings and future perspectives. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Heinritz, S.N.; Weiss, E.; Eklund, M.; Aumiller, T.; Louis, S.; Rings, A.; Messner, S.; Camarinha-Silva, A.; Seifert, J.; Bischoff, S.C.; et al. Intestinal Microbiota and Microbial Metabolites Are Changed in a Pig Model Fed a High-Fat/Low-Fiber or a Low-Fat/High-Fiber Diet. PLoS ONE 2016, 11, e0154329. [Google Scholar] [CrossRef] [PubMed]
- Muegge, B.D.; Kuczynski, J.; Knights, D.; Clemente, J.C.; Gonzalez, A.; Fontana, L.; Henrissat, B.; Knight, R.; Gordon, J.I. Diet drives convergence in gut microbiome functions across mammalian phylogeny and within humans. Science 2011, 332, 970–974. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Huang, Q.; Fu, X.; Liu, R.H. In vitro fermentation of mulberry fruit polysaccharides by human fecal inocula and impact on microbiota. Food Funct. 2016, 7, 4637–4643. [Google Scholar] [CrossRef] [PubMed]
- Mikkelsen, D.; Gidley, M.J.; Williams, B.A. In vitro fermentation of bacterial cellulose composites as model dietary fibers. J. Agric. Food Chem. 2011, 59, 4025–4032. [Google Scholar] [CrossRef] [PubMed]
- Mikkelsen, D.; Gidley, M.J. Formation of cellulose-based composites with hemicelluloses and pectins using Gluconacetobacter fermentation. In Plant Cell Wall: Methods and Protocols; Popper, Z.A., Ed.; Humana Press: Totowa, NJ, USA, 2011; Volume 715, pp. 197–208. [Google Scholar]
- Gibson, G.R.; Roberfroid, M.B. Dietary modulation of the human colonic microbiota-introducing the concept of prebiotics. J. Nutr. 1995, 125, 1401–1412. [Google Scholar] [PubMed]
- Duncan, S.H.; Flint, H.J. Probiotics and prebiotics and health in ageing populations. Maturitas 2013, 75, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Roberfroid, M. Prebiotics: The concept revisited. J. Nutr. 2007, 137, 830s–837s. [Google Scholar] [PubMed]
- Bindels, L.B.; Delzenne, N.; Cani, P.; Walter, J. Towards a more comprehensive concept for prebiotics. Nat. Rev. Gastroenterol. Hepatol. 2015, 12, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Gorham, J.B.; Williams, B.A.; Gidley, M.J.; Mikkelsen, D. Visualization of microbe-dietary remnant interactions in digesta from pigs, by fluorescence in situ hybridization and staining methods; effects of a dietary arabinoxylan-rich wheat fraction. Food Hydrocoll. 2016, 52, 952–962. [Google Scholar] [CrossRef]
- De Vries, J.; Birkett, A.; Hulshof, T.; Verbeke, K.; Gibes, K. Effects of Cereal, Fruit and Vegetable Fibers on Human Fecal Weight and Transit Time: A Comprehensive Review of Intervention Trials. Nutrients 2016, 8, 130. [Google Scholar] [CrossRef] [PubMed]
- Marzorati, M.; Verhelst, A.; Luta, G.; Sinnott, R.; Verstraete, W.; Van de Wiele, T.; Possemiers, S. In vitro modulation of the human gastrointestinal microbial community by plant-derived polysaccharide-rich dietary supplements. Int. J. Food Microbiol. 2010, 139, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Dongowski, G.; Lorenz, A.; Proll, J. The Degree of Methylation Influences the Degradation of Pectin in the Intestinal Tract of Rats and In Vitro. Nutr. Metab. 2002, 132, 1935–1944. [Google Scholar]
- Konstantinov, S.R.; Favier, C.F.; Zhu, W.Y.; Williams, B.A.; Klüß, J.; Souffrant, W.B.; De Vos, W.M.; Akkermans, A.D.L.; Smidt, H. Microbial diversity studies of the porcine gastrointestinal ecosystem during weaning transition. Anim. Res. 2004, 53, 317–324. [Google Scholar] [CrossRef]
- Grootaert, C.; Van den Abbeele, P.; Marzorati, M.; Broekaert, W.F.; Courtin, C.M.; Delcour, J.A.; Verstraete, W.; Van de Wiele, T. Comparison of prebiotic effects of arabinoxylan oligosaccharides and inulin in a simulator of the human intestinal microbial ecosystem. FEMS Microbiol. Ecol. 2009, 69, 231–242. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.M.; Yu, Y.N.; Wang, J.L.; Lin, Y.W.; Kong, X.; Yang, C.Q.; Yang, L.; Liu, Z.J.; Yuan, Y.Z.; Liu, F.; et al. Decreased dietary fiber intake and structural alteration of gut microbiota in patients with advanced colorectal adenoma. Am. J. Clin. Nutr. 2013, 97, 1044–1052. [Google Scholar] [CrossRef] [PubMed]
- Hooda, S.; Boler, B.M.; Serao, M.C.; Brulc, J.M.; Staeger, M.A.; Boileau, T.W.; Dowd, S.E.; Fahey, G.C., Jr.; Swanson, K.S. 454 pyrosequencing reveals a shift in fecal microbiota of healthy adult men consuming polydextrose or soluble corn fiber. J. Nutr. 2012, 142, 1259–1265. [Google Scholar] [CrossRef] [PubMed]
- Salyers, A.A.; Vercellotti, J.R.; West, S.E.; Wilkins, T.D. Fermentation of Mucin and Plant Polysaccharides by Strains of Bacteroides from the Human Colon. Appl. Environ. Microbiol. 1977, 33, 319–322. [Google Scholar] [PubMed]
- Shinohara, K.; Ohashi, Y.; Kawasumi, K.; Terada, A.; Fujisawa, T. Effect of apple intake on fecal microbiota and metabolites in humans. Anaerobe 2010, 16, 510–515. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Zhang, Y.; Zheng, P.; Chen, X.; Yang, Y. Starch structure modulates metabolic activity and gut microbiota profile. Anaerobe 2013, 24, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Slavin, J. Why whole grains are protective: Biological mechanisms. Proc. Nutr. Soc. 2003, 62, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Lappi, J.; Salojarvi, J.; Kolehmainen, M.; Mykkanen, H.; Poutanen, K.; de Vos, W.M.; Salonen, A. Intake of whole-grain and fiber-rich rye bread versus refined wheat bread does not differentiate intestinal microbiota composition in Finnish adults with metabolic syndrome. J. Nutr. 2013, 143, 648–655. [Google Scholar] [CrossRef] [PubMed]
- Costabile, A.; Klinder, A.; Fava, F.; Napolitano, A.; Fogliano, V.; Leonard, C.; Gibson, G.R.; Tuohy, K.M. Whole-grain wheat breakfast cereal has a prebiotic effect on the human gut microbiota: A double-blind, placebo-controlled, crossover study. Br. J. Nutr. 2008, 99, 110–120. [Google Scholar] [CrossRef] [PubMed]
- Brambillasca, S.; Zunino, P.; Cajarville, C. Addition of inulin, alfalfa and citrus pulp in diets for piglets: Influence on nutritional and faecal parameters, intestinal organs, and colonic fermentation and bacterial populations. Livest. Sci. 2015, 178, 243–250. [Google Scholar] [CrossRef]
- Blatchford, P.; Stoklosinski, H.; Walton, G.; Swann, J.; Gibson, G.; Gearry, R.; Ansell, J. Kiwifruit fermentation drives positive gut microbial and metabolic changes irrespective of initial microbiota composition. Bioact. Carbohydr. Diet. Fibre 2015, 6, 37–45. [Google Scholar] [CrossRef]
- Eid, N.; Osmanova, H.; Natchez, C.; Walton, G.; Costabile, A.; Gibson, G.; Rowland, I.; Spencer, J.P. Impact of palm date consumption on microbiota growth and large intestinal health: A randomised, controlled, cross-over, human intervention study. Br. J. Nutr. 2015, 114, 1226–1236. [Google Scholar] [CrossRef] [PubMed]
- Montagne, L.; Arturo-Schaan, M.; Le Floc’h, N.; Guerra, L.; Le Gall, M. Effect of sanitary conditions and dietary fibre on the adaptation of gut microbiota after weaning. Livest. Sci. 2010, 133, 113–116. [Google Scholar] [CrossRef]
| Lactic | Succinic | Acetic | Propionic | Butyric | Mode of Action |
|---|---|---|---|---|---|
| √ | √ | Source of energy (brain, heart, muscle) | |||
| √ | Energy for colonocytes | ||||
| √ | √ | √ | √ | Reduce GIT pH | |
| √ | √ | √ | √ | Decreases NH3 absorption across epithelium | |
| √ | √ | √ | √ | Decreases growth of potential pathogens | |
| √ | Inhibits proliferation and induces apoptosis of cancerous cells | ||||
| √ | √ | Lipid Metabolism | |||
| √ | √ | √ | Increased leptin production (increased satiety) | ||
| Involvement in bacterial cross-feeding |
| Purified Ingredient | Methodology Used | Findings | Reference |
|---|---|---|---|
| Polydextrose Soluble maize fibre | 454 pyrosequencing of bacterial 16S rRNA genes (V4–V6 region) | Consumption of these fibres led to an increased abundance of faecal Clostridiaceae, Veillonellaceae, Faecalibacterium, Phascolarctobacterium, Dialister and lower Eubacteriaceae | [184] |
| Maize, Dextrin, Pullulan, Resistant starch (RS) | Micro-array analysis | All tested substrates except RS reduced species of the Bacteroides group, and increased Bifidobacteria | [21] |
| Aloe vera gel (extract and powder), Larch, U. pinnatifida fucoidans, Tragacanth gum, Ghatti gum | Real-time PCR analysis of species of interest | Increased Bifidobacteria spp. and the bacteroides-prevotella group | [179] |
| Amylose, amylopectin, dextran, xylan, polygalacturonate, pectin | Culture analysis of 10 Bacteroides spp. | Identified the polysaccharide preference (of the tested substrates) for fermentation by specific bacterial species. Most capable of plant polysaccharide fermentation | [185] |
| Apple pectin | Faeces were collected for culturing analysis of bacterial populations | Significant increase in Bifidobacteria with a decrease in species from the Clostridia class | [186] |
| High amylase maize starch | Fluorescence in situ hybridisation | Faecalibacterium prausnitzii and Eubacterium hallii were significantly increased in the cultures | [187] |
| Source of Bacteria | Ingredients | Methodology Used | Findings | Reference |
|---|---|---|---|---|
| Human faeces | Kiwifruit | In vitro batch culture fermentation, 454 pyrosequencing (V2–V3 region) | Bacteroides and Bifidobacterium species were more abundant in bacterial communities fermenting kiwifruit | [192] |
| Human faeces | High flavonoid whole-foods | Total bacterial counts by fluorescence in situ hybridisation | Flavonoid content of whole-foods led to a decreased abundance of potentially pathogenic bacteria, as per relationship to cardiovascular disorders | [166] |
| Human faeces | Dates | Bacteria enumeration via fluorescent in situ hybridisation | No significant differences of microbiota between diets reported | [193] |
| Swine in vivo digesta | Alfalfa & citrus pulp | Bacterial culturing for counts | No difference in bacterial counts reported between diets | [191] |
| Swine faeces | Wheat, wheat bran | Faecal microbiota analysis using qPCR, DNA fingerprinting, metaproteomics | Lactobacilli, bifidobacteria and Faecalibacterium prausnitzii was significantly higher (p < 0.05) in the high fibre animals. Enterobacteriaceae was more abundant in low-fibre-fed animals | [168] |
| Ground maize, Wheat bran | Real-time PCR to analyse populations of Lactobacilli and Enterobacteria | No long-term differences for Lactobacilli and Enterobacteria between the two diets containing maize and wheat bran. The feeding period of 7 to 42 days showed the enzymatic potential to degrade complex fibres adapted over time. Enzyme activity was detected for xylanase after 7 d and cellulose 42 d | [120] | |
| Swine faeces | Sugar beet pulp, Soybean hulls | Faeces collected for culturing and bacterial counts (log10 CFU/g) | Reported that DF did not affect the composition of the bacterial population cultured from faeces | [194] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Williams, B.A.; Grant, L.J.; Gidley, M.J.; Mikkelsen, D. Gut Fermentation of Dietary Fibres: Physico-Chemistry of Plant Cell Walls and Implications for Health. Int. J. Mol. Sci. 2017, 18, 2203. https://doi.org/10.3390/ijms18102203
Williams BA, Grant LJ, Gidley MJ, Mikkelsen D. Gut Fermentation of Dietary Fibres: Physico-Chemistry of Plant Cell Walls and Implications for Health. International Journal of Molecular Sciences. 2017; 18(10):2203. https://doi.org/10.3390/ijms18102203
Chicago/Turabian StyleWilliams, Barbara A., Lucas J. Grant, Michael J. Gidley, and Deirdre Mikkelsen. 2017. "Gut Fermentation of Dietary Fibres: Physico-Chemistry of Plant Cell Walls and Implications for Health" International Journal of Molecular Sciences 18, no. 10: 2203. https://doi.org/10.3390/ijms18102203
APA StyleWilliams, B. A., Grant, L. J., Gidley, M. J., & Mikkelsen, D. (2017). Gut Fermentation of Dietary Fibres: Physico-Chemistry of Plant Cell Walls and Implications for Health. International Journal of Molecular Sciences, 18(10), 2203. https://doi.org/10.3390/ijms18102203