Effects of Short-Term Probiotic Ingestion on Immune Profiles and Microbial Translocation among HIV-1-Infected Vietnamese Children
Abstract
:1. Introduction
2. Results
2.1. LcS Ingestion Did Not Induce Significant Clinical Events
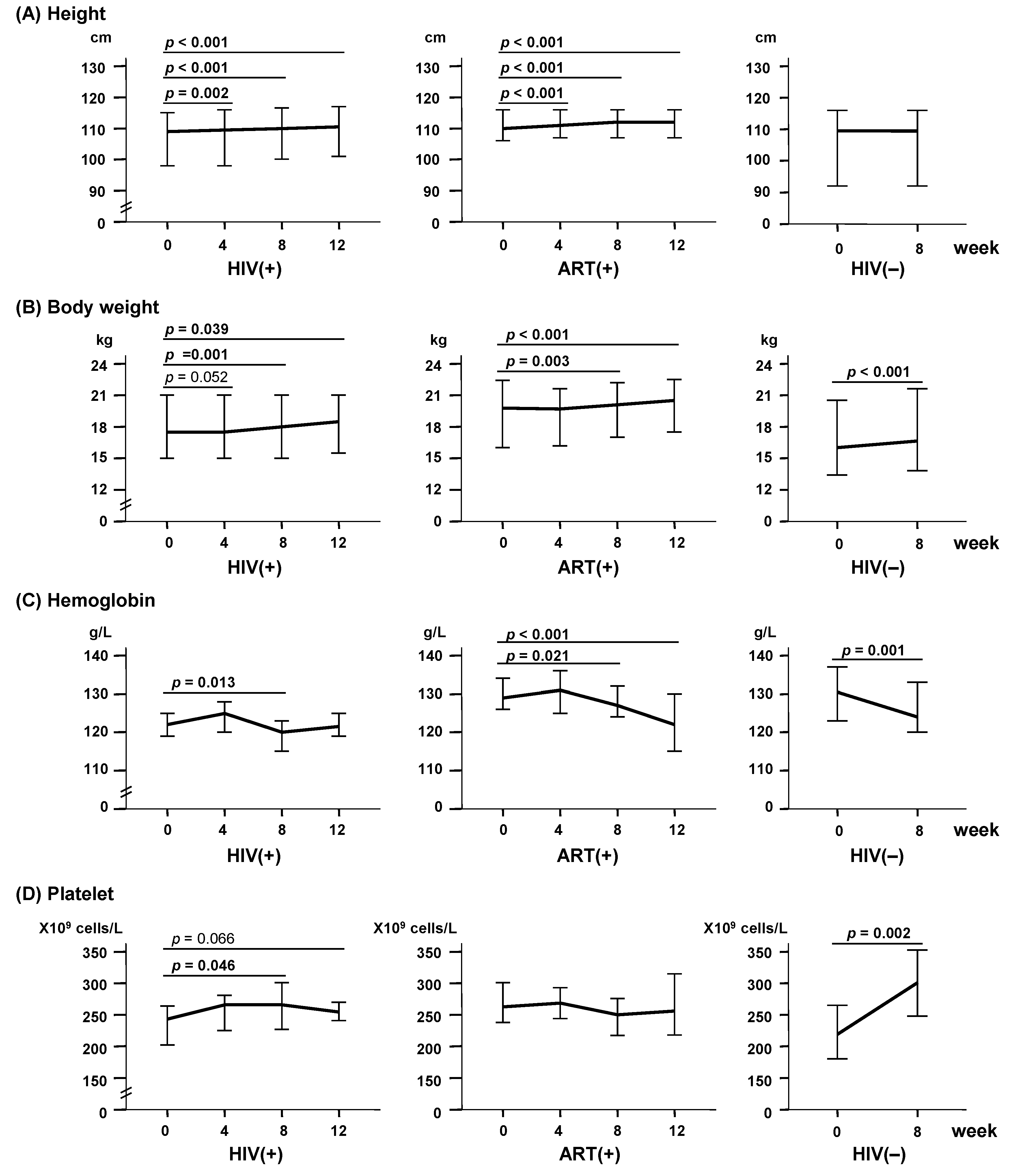
2.2. LcS Ingestion Improved Physical and Clinical Parameters
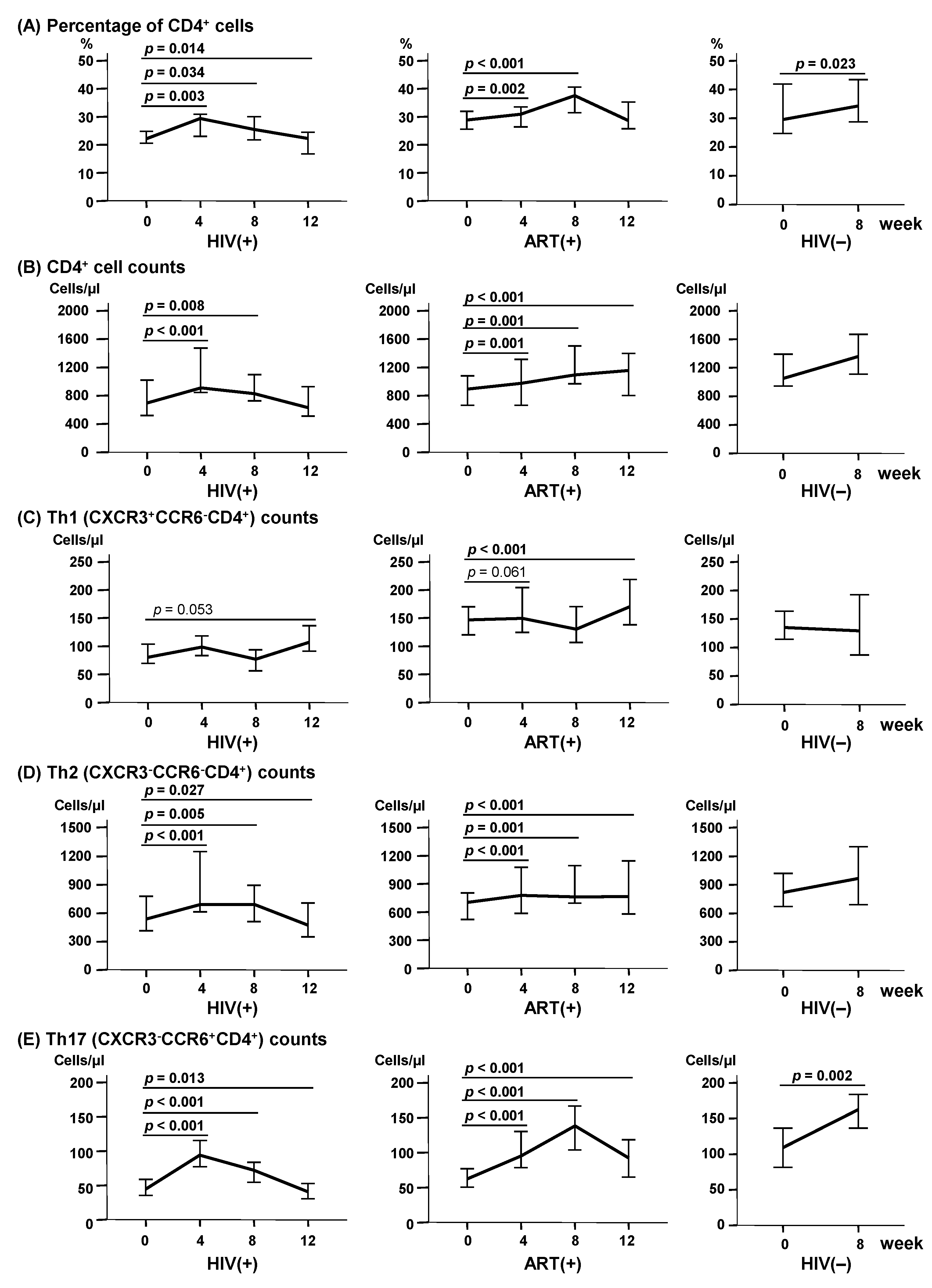
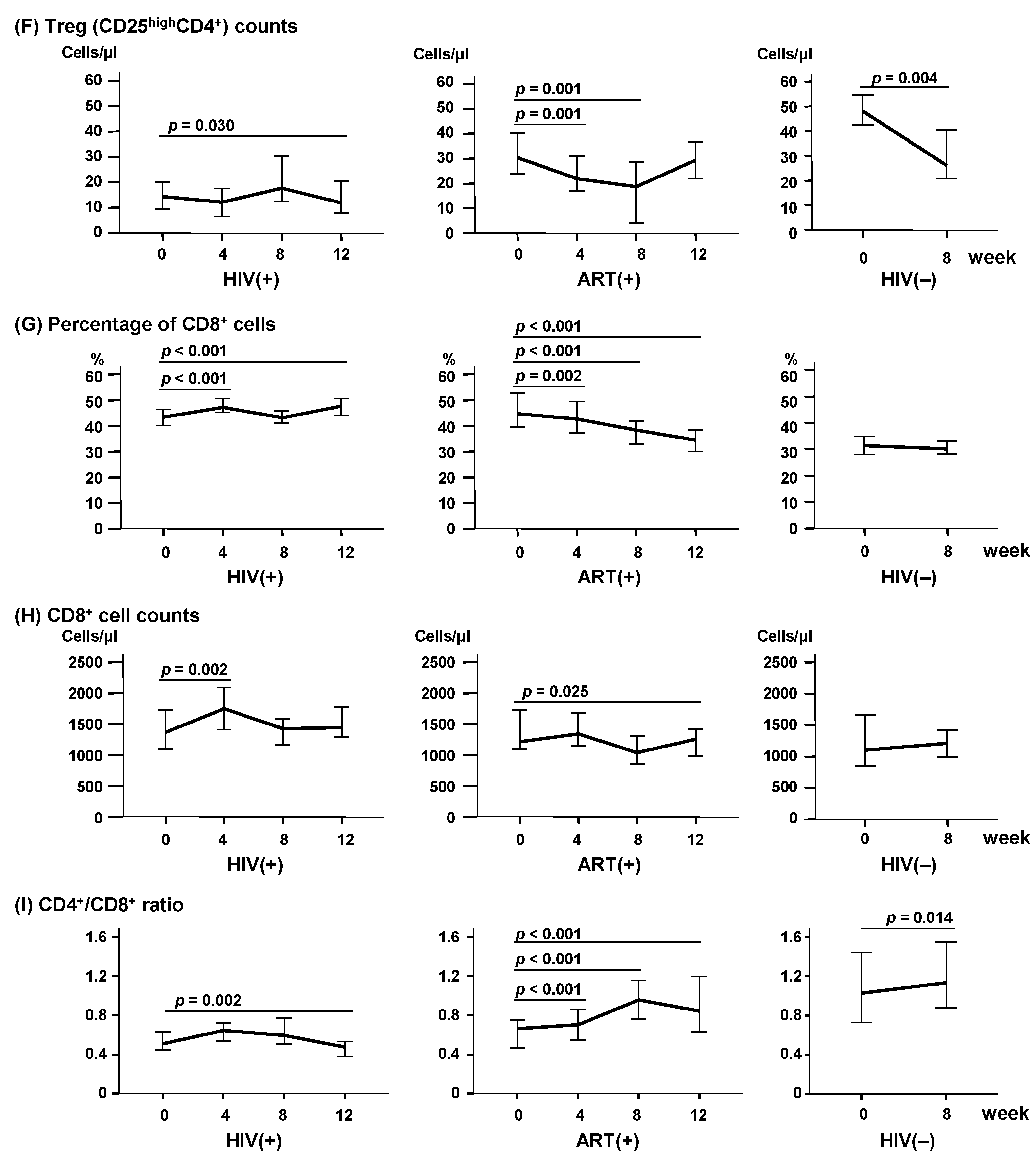
2.3. LcS Ingestion Increased CD4+ Cells, Especially Th2 and Th17 Subsets
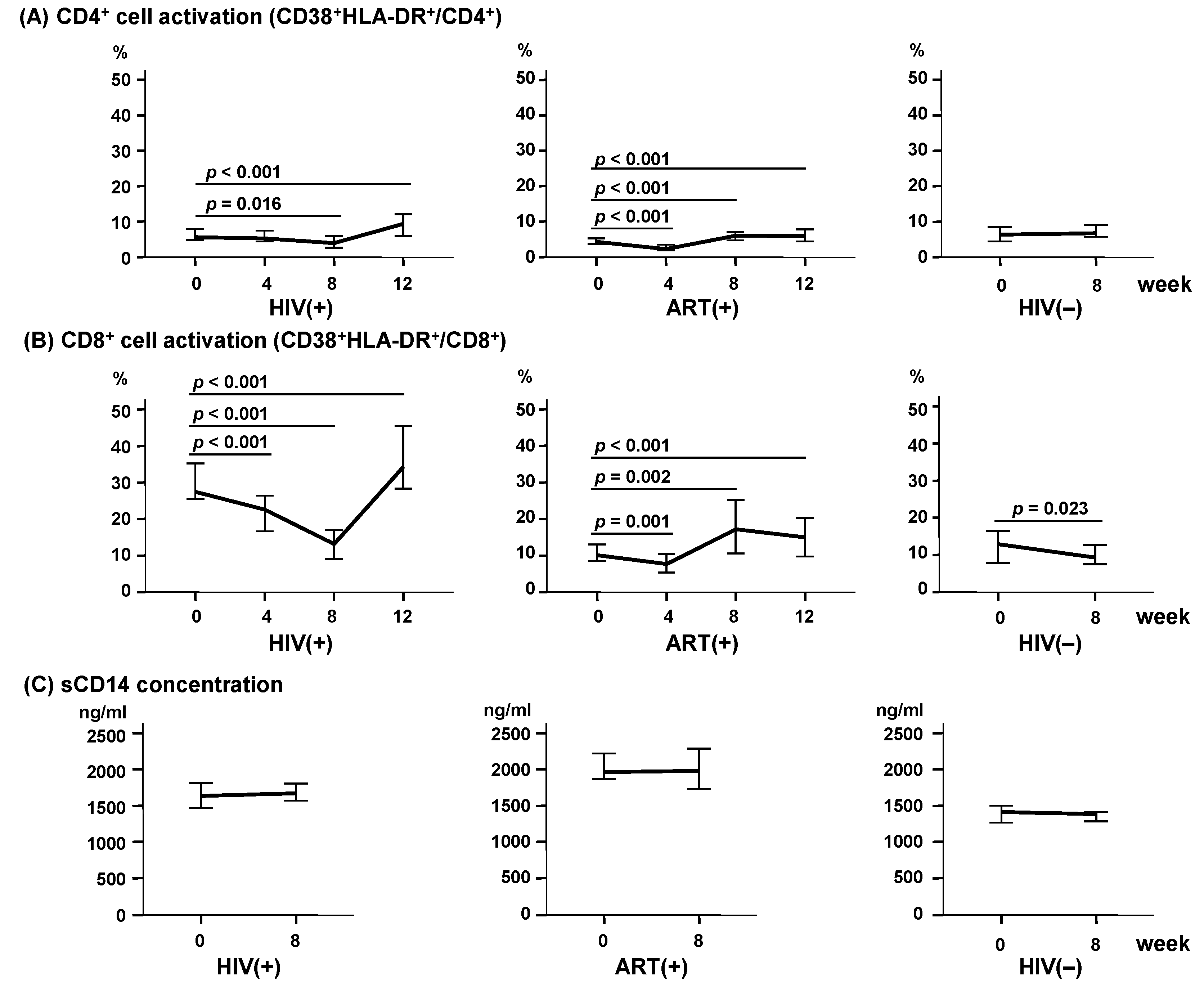
2.4. LcS Ingestion Induced a Dramatic Decline in CD8+ Cell Activation But Not in Monocyte Activation
2.5. LcS Ingestion Induced a Decrease in Plasma Viral Load (VL) in HIV(+) Children
2.6. LcS Ingestion Did Not Significantly Change Microbial Translocation
3. Discussion
4. Materials and Methods
4.1. Study Design and Subjects
4.2. Study Schedule
4.3. Clinical Laboratory Measurement
4.4. Plasma HIV Viral Load
4.5. Immunological Analysis
4.6. Detection of Bacterial Ribosomal RNA Genes (rDNA) in Plasma
4.7. Detection of Bacterial Ribosomal RNA Molecules (rRNA) in Whole Blood
4.8. Statistical Analysis
4.9. Study Approval
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mehandru, S.; Poles, M.A.; Tenner-Racz, K.; Horowitz, A.; Hurley, A.; Hogan, C.; Boden, D.; Racz, P.; Markowitz, M. Primary HIV-1 infection is associated with preferential depletion of CD4+ T lymphocytes from effector sites in the gastrointestinal tract. J. Exp. Med. 2004, 200, 761–770. [Google Scholar] [CrossRef] [PubMed]
- Hunt, P.W. Th17, gut, and HIV: Therapeutic implications. Curr. Opin. HIV AIDS 2010, 5, 189–193. [Google Scholar] [CrossRef] [PubMed]
- Brenchley, J.M.; Paiardini, M.; Knox, K.S.; Asher, A.I.; Cervasi, B.; Asher, T.E.; Scheinberg, P.; Price, D.A.; Hage, C.A.; Kholi, L.M.; et al. Differential Th17 CD4 T-cell depletion in pathogenic and nonpathogenic lentiviral infections. Blood 2008, 112, 2826–2835. [Google Scholar] [CrossRef] [PubMed]
- Gordon, S.N.; Cervasi, B.; Odorizzi, P.; Silverman, R.; Aberra, F.; Ginsberg, G.; Estes, J.D.; Paiardini, M.; Frank, I.; Silvestri, G. Disruption of intestinal CD4+ T cell homeostasis is a key marker of systemic CD4+ T cell activation in HIV-infected individuals. J. Immunol. 2010, 185, 5169–5179. [Google Scholar] [CrossRef] [PubMed]
- Estes, J.D.; Harris, L.D.; Klatt, N.R.; Tabb, B.; Pittaluga, S.; Paiardini, M.; Barclay, G.R.; Smedley, J.; Pung, R.; Oliveira, K.M.; et al. Damaged intestinal epithelial integrity linked to microbial translocation in pathogenic simian immunodeficiency virus infections. PLoS Pathog. 2010, 6, e1001052. [Google Scholar] [CrossRef] [PubMed]
- Brenchley, J.M.; Price, D.A.; Schacker, T.W.; Asher, T.E.; Silvestri, G.; Rao, S.; Kazzaz, Z.; Bornstein, E.; Lambotte, O.; Altmann, D.; et al. Microbial translocation is a cause of systemic immune activation in chronic HIV infection. Nat. Med. 2006, 12, 1365–1371. [Google Scholar] [CrossRef] [PubMed]
- Mehandru, S.; Poles, M.A.; Tenner-Racz, K.; Jean-Pierre, P.; Manuelli, V.; Lopez, P.; Shet, A.; Low, A.; Mohri, H.; Boden, D.; et al. Lack of mucosal immune reconstitution during prolonged treatment of acute and early HIV-1 infection. PLoS Med. 2006, 3, e484. [Google Scholar]
- Bi, X.; Suzuki, Y.; Gatanaga, H.; Oka, S. High frequency and proliferation of CD4+ FOXP3+ Treg in HIV-1-infected patients with low CD4 counts. Eur. J. Immunol. 2009, 39, 301–309. [Google Scholar] [CrossRef] [PubMed]
- Hardy, H.; Harris, J.; Lyon, E.; Beal, J.; Foey, A.D. Probiotics, prebiotics and immunomodulation of gut mucosal defences: Homeostasis and immunopathology. Nutrients 2013, 5, 1869–1912. [Google Scholar] [CrossRef] [PubMed]
- Hemsworth, J.C.; Hekmat, S.; Reid, G. Micronutrient supplemented probiotic yogurt for HIV-infected adults taking HAART in London, Canada. Gut Microbes 2012, 3, 414–419. [Google Scholar] [CrossRef] [PubMed]
- Trois, L.; Cardoso, E.M.; Miura, E. Use of probiotics in HIV-infected children: A randomized double-blind controlled study. J. Trop. Pediatr. 2008, 54, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Hummelen, R.; Changalucha, J.; Butamanya, N.L.; Koyama, T.E.; Cook, A.; Habbema, J.D.; Reid, G. Effect of 25 weeks probiotic supplementation on immune function of HIV patients. Gut Microbes 2011, 2, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Gori, A.; Rizzardini, G.; Van’t Land, B.; Amor, K.B.; van Schaik, J.; Torti, C.; Quirino, T.; Tincati, C.; Bandera, A.; Knol, J.; et al. Specific prebiotics modulate gut microbiota and immune activation in HAART-naive HIV-infected adults: Results of the “COPA” pilot randomized trial. Mucosal Immunol. 2011, 4, 554–563. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Hernandez, L.A.; Jave-Suarez, L.F.; Fafutis-Morris, M.; Montes-Salcedo, K.E.; Valle-Gutierrez, L.G.; Campos-Loza, A.E.; Enciso-Gomez, L.F.; Andrade-Villanueva, J.F. Synbiotic therapy decreases microbial translocation and inflammation and improves immunological status in HIV-infected patients: A double-blind randomized controlled pilot trial. Nutr. J. 2012, 11, 90. [Google Scholar] [CrossRef] [PubMed]
- Shida, K.; Nanno, M.; Nagata, S. Flexible cytokine production by macrophages and T cells in response to probiotic bacteria: A possible mechanism by which probiotics exert multifunctional immune regulatory activities. Gut Microbes 2011, 2, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Falasca, K.; Vecchiet, J.; Ucciferri, C.; Di Nicola, M.; D’Angelo, C.; Reale, M. Effect of Probiotic Supplement on Cytokine Levels in HIV-Infected Individuals: A Preliminary Study. Nutrients 2015, 7, 8335–8347. [Google Scholar] [CrossRef] [PubMed]
- Bi, X.; Ishizaki, A.; Nguyen, L.V.; Matsuda, K.; Pham, H.V.; Phan, C.T.; Ogata, K.; Giang, T.T.; Phung, T.T.; Nguyen, T.T.; et al. Impact of HIV Infection and Anti-Retroviral Therapy on the Immune Profile of and Microbial Translocation in HIV-Infected Children in Vietnam. Int. J. Mol. Sci. 2016, 17. [Google Scholar] [CrossRef] [PubMed]
- Kamada, N.; Seo, S.U.; Chen, G.Y.; Nunez, G. Role of the gut microbiota in immunity and inflammatory disease. Nat. Rev. Immunol. 2013, 13, 321–335. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, T.; Suzuki, T.; Kaji, R.; Serata, M.; Nagata, T.; Ando, M.; Iizuka, R.; Tsujibe, S.; Murakami, J.; Kiyoshima-Shibata, J.; et al. Probiotic upregulation of peripheral IL-17 responses does not exacerbate neurological symptoms in experimental autoimmune encephalomyelitis mouse models. Immunopharmacol. Immunotoxicol. 2012, 34, 423–433. [Google Scholar] [CrossRef] [PubMed]
- Klatt, N.R.; Canary, L.A.; Sun, X.; Vinton, C.L.; Funderburg, N.T.; Morcock, D.R.; Quinones, M.; Deming, C.B.; Perkins, M.; Hazuda, D.J.; et al. Probiotic/prebiotic supplementation of antiretrovirals improves gastrointestinal immunity in SIV-infected macaques. J. Clin. Investig. 2013, 123, 903–907. [Google Scholar] [CrossRef] [PubMed]
- D’Ettorre, G.; Ceccarelli, G.; Giustini, N.; Serafino, S.; Calantone, N.; De Girolamo, G.; Bianchi, L.; Bellelli, V.; Ascoli-Bartoli, T.; Marcellini, S.; et al. Probiotics Reduce Inflammation in Antiretroviral Treated, HIV-Infected Individuals: Results of the “Probio-HIV” Clinical Trial. PLoS ONE 2015, 10, e0137200. [Google Scholar] [CrossRef] [PubMed]
- D’Ettorre, G.; Rossi, G.; Scagnolari, C.; Andreotti, M.; Giustini, N.; Serafino, S.; Schietroma, I.; Scheri, G.C.; Fard, S.N.; Trinchieri, V.; et al. Probiotic supplementation promotes a reduction in T-cell activation, an increase in Th17 frequencies, and a recovery of intestinal epithelium integrity and mitochondrial morphology in ART-treated HIV-1-positive patients. Immun. Inflamm. Dis. 2017, 5, 244–260. [Google Scholar] [CrossRef] [PubMed]
- Agustina, R.; Bovee-Oudenhoven, I.M.J.; Lukito, W.; Fahmida, U.; van de Rest, O.; Zimmermann, M.B.; Firmansyah, A.; Wulanti, R.; Albers, R.; van den Heuvel, E.G.H.M.; et al. Probiotics Lactobacillus reuteri DSM 17938 and Lactobacillus casei CRL 431 Modestly Increase Growth, but Not Iron and Zinc Status, among Indonesian Children Aged 1–6 Years. J. Nutr. 2013, 143, 1184–1193. [Google Scholar] [CrossRef] [PubMed]
- Minemura, M.; Shimizu, Y. Gut microbiota and liver diseases. World J. Gastroenterol. 2015, 21, 1691–1702. [Google Scholar] [CrossRef] [PubMed]
- Lata, J.; Jurankova, J.; Kopacova, M.; Vitek, P. Probiotics in hepatology. World J. Gastroenterol. 2011, 17, 2890–2896. [Google Scholar] [CrossRef] [PubMed]
- Trifari, S.; Kaplan, C.D.; Tran, E.H.; Crellin, N.K.; Spits, H. Identification of a human helper T cell population that has abundant production of interleukin 22 and is distinct from T(H)-17, T(H)1 and T(H)2 cells. Nat. Immunol. 2009, 10, 864–871. [Google Scholar] [CrossRef] [PubMed]
- Hunt, P.W.; Martin, J.N.; Sinclair, E.; Bredt, B.; Hagos, E.; Lampiris, H.; Deeks, S.G. T cell activation is associated with lower CD4+ T cell gains in human immunodeficiency virus-infected patients with sustained viral suppression during antiretroviral therapy. J. Infect. Dis. 2003, 187, 1534–1543. [Google Scholar] [CrossRef] [PubMed]
- Maecker, H.T.; McCoy, J.P.; Nussenblatt, R. Standardizing immunophenotyping for the Human Immunology Project. Nat. Rev. Immunol. 2012, 12, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Kinter, A.; McNally, J.; Riggin, L.; Jackson, R.; Roby, G.; Fauci, A.S. Suppression of HIV-specific T cell activity by lymph node CD25+ regulatory T cells from HIV-infected individuals. Proc. Natl. Acad. Sci. USA 2007, 104, 3390–3395. [Google Scholar] [CrossRef] [PubMed]

| Target Bacteria | HIV(+) (n = 31) | ART(+) (n = 29) | HIV(−) (n = 20) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Week 0 | Week 4 | Week 8 | Week 12 | Week 0 | Week 4 | Week 8 | Week 12 | Week 0 | Week 8 | |
| Clostridium coccoides group | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND |
| Clostridium leptum subgroup | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND |
| Bacteroides fragilis group | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND |
| Bifidobacterium | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND |
| Atopobium cluster | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND |
| Prevotella | ND | ND | ND | ND | ND | 37.1 [1/29] | ND | ND | ND | ND |
| Enterobacteriaceae | ND | ND | 2.0 [1/31] | ND | ND | ND | ND | ND | ND | ND |
| Streptococcus | 17.8 [1/31] | 13.8 [2/31] | 1.8 ± 0.5 [3/31] | ND | ND | 23.7 [2/29] | 20.7 [1/29] | 3.0 [2/29] | ND | 3.5 [2/20] |
| Enterococcus | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND |
| Staphylococcus | * 35.5 ± 15.0 # [7/31] | 38.7 [2/31] | 25.6 ± 6.5 [4/31] | 19.8 ± 11.9 [9/30] | ND | 26.4 [1/29] | 49.1 [2/29] | 16.2 ± 7.8 [5/29] | 23.2 [1/20] | 35.1 ± 43.8 [4/20] |
| Pseudomonas | 2.9 [1/31] | ND | ND | ND | ND | 1.9 [2/29] | 1.5 [1/29] | ND | 3.3 [2/20] | ND |
| Lactobacillus casei subgroup | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ishizaki, A.; Bi, X.; Nguyen, L.V.; Matsuda, K.; Pham, H.V.; Phan, C.T.T.; Khu, D.T.K.; Ichimura, H. Effects of Short-Term Probiotic Ingestion on Immune Profiles and Microbial Translocation among HIV-1-Infected Vietnamese Children. Int. J. Mol. Sci. 2017, 18, 2185. https://doi.org/10.3390/ijms18102185
Ishizaki A, Bi X, Nguyen LV, Matsuda K, Pham HV, Phan CTT, Khu DTK, Ichimura H. Effects of Short-Term Probiotic Ingestion on Immune Profiles and Microbial Translocation among HIV-1-Infected Vietnamese Children. International Journal of Molecular Sciences. 2017; 18(10):2185. https://doi.org/10.3390/ijms18102185
Chicago/Turabian StyleIshizaki, Azumi, Xiuqiong Bi, Lam Van Nguyen, Kazunori Matsuda, Hung Viet Pham, Chung Thi Thu Phan, Dung Thi Khanh Khu, and Hiroshi Ichimura. 2017. "Effects of Short-Term Probiotic Ingestion on Immune Profiles and Microbial Translocation among HIV-1-Infected Vietnamese Children" International Journal of Molecular Sciences 18, no. 10: 2185. https://doi.org/10.3390/ijms18102185
APA StyleIshizaki, A., Bi, X., Nguyen, L. V., Matsuda, K., Pham, H. V., Phan, C. T. T., Khu, D. T. K., & Ichimura, H. (2017). Effects of Short-Term Probiotic Ingestion on Immune Profiles and Microbial Translocation among HIV-1-Infected Vietnamese Children. International Journal of Molecular Sciences, 18(10), 2185. https://doi.org/10.3390/ijms18102185





